Abstract
Hydra vulgaris, long known for its remarkable regenerative capabilities, is also a long-standing source of inspiration for models of spontaneous patterning. Recently it became clear that early patterning during Hydra regeneration is an integrated mechanochemical process whereby morphogen dynamics is influenced by tissue mechanics. One roadblock to understanding Hydra self-organization is our lack of knowledge about the mechanical properties of these organisms. In this study, we combined microfluidic developments to perform parallelized microaspiration rheological experiments and numerical simulations to characterize these mechanical properties. We found three different behaviors depending on the applied stresses: an elastic response, a viscoelastic response, and tissue rupture. Using models of deformable shells, we quantify their Young’s modulus, shear viscosity, and the critical stresses required to switch between behaviors. Based on these experimental results, we propose a description of the tissue mechanics during normal regeneration. Our results provide a first step toward the development of original mechanochemical models of patterning grounded in quantitative experimental data.
Significance
Hydra vulgaris is a remarkable organism owing to its regenerative abilities. One can cut this animal into several pieces which will reform into a full Hydra in a few days. In this process, the pieces have to define a new organizing axis. Recently, researchers have shown that this axis definition is under mechanical control. One roadblock to understanding the relationship between tissue mechanics and Hydra biology is our lack of knowledge about the mechanical state of this organism. Here, we perform a mechanical characterization using a combination of microaspiration setups and numerical simulations. We finally propose a description of what happens at the mechanical level during Hydra regeneration, allowing quantitative approaches to question the role of mechanical cues in axis definition.
Introduction
Hydra vulgaris has long been a model of choice in developmental biology because of its remarkable regenerative capabilities (1,2). Almost any excised tissue piece, as well as cellular re-aggregates, is capable of reforming a fully viable adult in just a few days. In the former example, an excised tissue piece folds back into a closed spherical shape with both of its epithelial monolayers, endoderm and ectoderm, engulfing a water-filled lumen. In the latter, the same tissues start by spontaneously sorting into their relative positions (3,4) before expelling excess cells to reform a hollow sphere. At this point, both regenerative trajectories converge and these tissue spheres start undergoing osmotically driven oscillations during which they swell because of water flowing from the environment to the lumen up to the point where the tension building up within the tissues becomes too large and leads to a local rupture (5,6). After rupture the samples deflate, close the resulting wound, and start another swelling-rupture cycle. These osmotic oscillations induce deformations and, therefore, stresses within the tissues with relative changes in the radii of the spheres of up to 30%. These have been referred to as phase I oscillations and are characterized by a high amplitude and a low frequency (6). Indeed, after a few of these cycles, the oscillations clearly change and enter phase II where they have lower amplitude and higher frequency (6).
In parallel with these mechanical oscillations, the samples establish a chemical pattern involving some characterized morphogens to define the oral-aboral axis of the organism. Most notably, a local expression of HyWnt3 has been shown to be an early signal of axial patterning, with this activation defining the future position of the head organizer (7). This local activation is then followed by the establishment of various chemical gradients within the spheres effectively patterning the whole axis (8,9). At that point, the originally symmetrical samples start elongating in an oblong shape at whose ends the head and foot will be regenerated, and the patterning can be considered complete. The switch between phase I and phase II oscillations was thought to be a signature of the establishment of axial patterning (6,10). Since small excised tissue pieces showed both phases, it was thought that they underwent spontaneous symmetry breaking and retained no memory of axial patterning, just as for cellular re-aggregates (5,6).
The question was then to determine how the spherical symmetry was broken during phase I oscillations and how the local head organizer was defined. For a long time, the main hypothesis was that of a purely biochemical spontaneous symmetry breaking in the form of Turing instabilities of an unknown reaction-diffusion system. In his seminal work, partially inspired by Hydra regeneration, Turing showed how a system of two interacting and diffusing chemical species, which he named morphogens, could become unstable in their homogeneous state and spontaneously start forming structures such as dots or stripes (11). These ideas were critical in developing the field of pattern formation and have been adapted for a wide variety of systems and organisms (see, e.g., (12,13) for recent reviews in the context of developmental biology). In this historical development, Hydra has retained its place as a model organism. Notably, Gierer and Meinhardt have expanded on the seminal ideas of Turing and have developed a modified version of his reaction-diffusion system specifically designed to explain regeneration and grafting experiments in Hydra (14). As a result, the so-called Gierer-Meinhardt model has long been the gold standard in the field. At its inception, it was purely speculative but, since then, some proteins have been shown to possess many of the characteristics required by the Gierer-Meinhardt model. Most notably, the protein HyWnt3, involved in the canonical Wnt pathway, is now generally considered to be the activator represented in the Gierer-Meinhardt model (15), since its expression is restricted to the head organizer, it has self-activating capacities and is the first temporal signature of symmetry breaking during regeneration. However, the necessary long-range inhibitor is notoriously missing. Some promising candidates were put forward, such as Dickkopf (Dkk) (16), Sp5 (17), and Hydra astacin-7 (18), but none of these could reproduce the predictions of these models, mostly because of differences in expression patterns.
These models also ignore the mechanical aspects of the process, most notably the osmotic oscillations, although it is now well established that they are necessary for proper regeneration (19,20). This observation suggested a possible coupling between the mechanical state of the tissues and their biological response, as has been observed in a variety of contexts and organisms with impacts on cell division or gene expression, including in the canonical Wnt pathway (21,22,23,24). Recent results have demonstrated such a coupling in Hydra by which the expression of HyWnt3 is reduced when osmotic oscillations are blocked (20), providing a potential direct coupling between tissue mechanics and chemical patterning.
Recent modeling efforts have thus been made in order to incorporate mechanochemical couplings (19,25,26), for instance by making the diffusion constant of the morphogens within the tissues dependent on tissue stretch (19). In most cases, however, these couplings were not grounded in experimental evidence. In addition, recent experimental results have started to question the assumptions described so far and underlying these models. First, it was shown that the shift from phase I to phase II oscillations was not a direct signature of axial patterning being established. Instead, the onset of phase II is due to the early apparition of the Hydra mouth (27), allowing it to regulate its osmotic imbalance by mouth opening rather than by tissue rupture. One consequence was that axial patterning had to be anterior to the shift between oscillation phases. It then became unclear whether small excised tissue pieces really went through de novo patterning or if they could inherit this patterning from their original host organism. It now appears that they do retain axial patterning, although the exact mechanism by which they do so remains unclear, whether by the organization of their ectodermal actin structures called myonemes (28) or by pre-existing biochemical gradients (29).
Although small excised tissue pieces retain axial patterning, it still remains that 1) cellular re-aggregates that cannot conserve either supracellular actin structures or chemical gradients have to show de novo axial patterning during osmotic oscillations, 2) there is evidence of a direct coupling between tissue deformations and HyWnt3 expression (20), one of the most important morphogens involved in Hydra patterning, as previously predicted from theoretical analyses of Hydra aggregates (25), and 3) osmotic oscillations are required for the proper elongation, morphogenesis, and regeneration of both excised tissue pieces and cellular re-aggregates. For all these reasons, the focus of the field is currently shifting to an integrated view of Hydra regeneration as a mechanobiological process (26,30). One clear roadblock to the development of these ideas is our lack of understanding of the rheological properties and mechanical state of regenerating Hydra tissue spheres. This often leads to assumptions as to the rheology of these samples and rough estimates of their key mechanical parameters. One exception is the development by Veschgini et al. of a two-fingered robotic hand allowing them to apply known, constant deformations on the tissue spheres and measure the resulting forces (31) giving the first local, quantitative measurements of mechanical features in Hydra. These measurements were not, however, used to deepen our understanding of the spontaneous osmotic oscillations.
In this work, we try to overcome these limitations and to offer a quantitative characterization of the mechanics of Hydra tissue spheres. To do so we used the well-established microaspiration technique (32,33,34,35), which we adapted to increase its throughput through the use of original microfluidic constructs following (36). We found three different mechanical behaviors as the aspiration pressure was increased: first an elastic response, then a viscoelastic response, and finally tissue rupture, as observed in phase I oscillations. Combining our experimental observations and measurements with the development of a rheological model of elastic shells and numerical simulations, we obtained quantitative measurements of both the main rheological parameters of Hydra tissue spheres and the critical pressures required to switch between the three regimes. Thanks to these results, we provide a description of internal tissue mechanics, strains, and stresses during osmotic oscillations and reveal that the tissue spheres behave largely as hyperelastic spherical shells during these oscillations. Hopefully, this mechanical characterization will serve as a stepping stone for the study of mechanobiochemical couplings in a quantitative manner.
Materials and methods
Hydra maintenance and lines used
Three Hydra vulgaris lines were maintained and used for experiments: a watermelon (WM) line (ectoderm GFP/endoderm DsRed2), a reverse watermelon (RWM) line (ectoderm DsRed2/endoderm GFP), and the AEP line from which embryos were obtained for making transgenic animals. Hydras were kept in Hydra medium (HM), which consists of 1 mM CaCl2, 0.1 mM MgCl2, 0.03 mM KNO3, 0.5 mM NaHCO3, and 0.08 mM MgSO4 at a pH between 7 and 7.3. Cultures were stored at 18°C in darkness in an incubator (Pol-Ekko-Aparatura, Wodzisław Śląski, Poland). Animals were fed 2–3 times per week with newly hatched Artemia (Hobby; Dohse Aquaristik, Grafschaft, Germany) and cleaned every 2 days by changing the medium in which they sat. Animals were starved for at least 24 h before the start of any experiment.
Preparation of tissue spheres
Tissue spheres were made by cutting a whole animal using a sterile scalpel (Bistouri UU no. 10; Holtex, Aix-en -Provence, France), as follows. The head and tail were removed by two transverse cuts and the remaining body column was first sliced into three or four pieces. These pieces were cut further to obtain 6–8 samples per adult animal. These samples were left to fold into tissue spheres for 3–4 h in HM.
Design and fabrication of the removable inserts
Sliding elements containing the cylindrical pipettes were fabricated in dry film according to the protocol detailed in (36). In brief, photolithography was realized on a 500-μm-thick photosensitive dry film (DF series; Nagase, Düsseldorf, Germany), laminated on a silicon wafer. Lateral dimension of the photolithography mask defined the length of the insert (20 mm) and its height (1 mm), as well as pipette diameter (50 and 100 μm were used). Square openings 100 μm wide placed at the bottom of the insert were also used for imaging purposes in Video S3 and Fig. S4. After post-exposure bake on a hotplate, the inserts were released from the wafer during overnight development in an acetone bath that both revealed the patterns and unstuck the dry films from the wafer. A single fabrication run allowed fabrication of 100 such sliding elements.
Mounting and preparing the microfluidic device
We used a microfluidic tool that allows several microaspirations to be performed in parallel. This system is composed of a chamber crossed by a removable insert bearing multiple tunnels allowing suction to occur. Both the chambers and inserts were designed using the CAD software Inventor 2020 (Autodesk, San Francisco, CA). The chamber is composed of a main channel which is 20.5 mm long, 4 mm wide, and 0.5 mm high (see Fig. S1 for the schematics). Halfway through its length, it is barred by another channel which will host the removable insert. The perpendicular channel is 13 mm long, 0.5 mm wide, and 1 mm high (Fig. S1). Details on the preparation of the device, microaspiration protocols, and threshold experiments can be found in supporting methods.
Osmotic oscillation measurements
To measure osmotic oscillations shown in Fig. S8, Hydra tissue spheres were prepared as described above. In parallel, a 1% agarose gel was prepared at the bottom of a Petri dish, left to solidify, and manually punched to create 1-mm-wide wells which then hosted the samples. These wells were used to limit the samples’ movement during osmotic oscillations. They were then imaged for 24 h at a 15min interval. The resulting images were thresholded and analyzed in ImageJ to extract the projected area as a function of time . Finally, the samples’ radii were computed as . The equilibrium radius of a given sample was defined as the minimum of over the whole recording, allowing us to estimate the strains on the inflating sphere.
Finite-element simulations
Numerical simulations were performed with the finite-element solver Comsol Multiphysics (COMSOL, Burlington, MA). The micropipette was represented as a rigid, motionless cylinder of inner radius = 50 μm. The reference state of the Hydra sphere was a shell of outer radius and inner radius , whose center was located on the cylinder’s longitudinal symmetry axis . We used contact boundary conditions ensuring that the (deformed) Hydra sphere and the micropipette never overlapped. Hydra tissue was modeled as a hyperelastic Saint-Venant Kirchhoff material. We denote by and the Young’s modulus and Poisson’s ratio of the tissue and considered nearly incompressible conditions (). The external pressure was applied on the spherical section contained within the micropipette. Simulations were performed in two dimensions, enforcing the rotational invariance of the system (Hydra + micropipette) about . The equations of elasticity were treated within a pressure formulation, adapted to approximately incompressible elastic materials. For each value of the applied pressure, we recorded the maximal deformation observed on the symmetry axis denoted , and computed as a spatial average along the shell’s thickness. We checked that our results were robust to smoothing the sharp corner of the micropipette by a fillet over a scale of the order of 5 μm. They were also insensitive to decreasing the typical mesh size.
Extraction of rheological parameters
To measure Young’s moduli in the elastic phase, each sample was treated separately. Based on the acquired images, we measured their projected area and turned them into rest radii . We next used our measurements of as a function of (Eq. 7) and fitted them by a straight line in Python. The slope of this straight line was kept and multiplied, according to Eq. 7, by to extract the Young’s modulus of that specific sample. We then repeated the procedure on many samples and report the mean and standard deviation of this distribution in the text.
In the viscoelastic phase, we measured the dynamics of the aspirated length as a function of time and fitted them in Python according to Eq. 8. To extract Young’s moduli, we used the same formula as above except that we replaced the slope of a linear fit of several points by the ratio of over and stemming from the fit of the full dynamics.
To estimate the order of magnitude of the effective viscosities in the same experiments, we used the same fit according to Eq. 8 to extract the speed of tissue flow and estimated the viscosity as with .
EDTA/blebbistatin experiments
Rheological measurements on Hydra whose cell adhesion had been inhibited were carried out by immersing the tissue spheres in solutions of either 2 mM EDTA (Sigma-Aldrich, St. Louis, MO) or 5 μm blebbistatin (Tocris Bioscience, Bristol, UK) in HM. The entire microfluidic system was immersed in the solution, and the Hydra pieces were left to bathe in it for around 10 min before aspiration to give the drug time to take effect before measuring. The rest of the protocol was identical to that of the rheological measurements explained above.
Mechanical description of Hydra tissue spheres
We consider a thin spherical shell of undeformed inner and outer radii and . Normal components of the Cauchy stress tensor are denoted . In brief, we re-express the strain energy function, Eq. 4, of a Saint-Venant Kirchhoff material as a function of the stretch ratios : . Denoting the shell pressure field, stress components are given by . By the assumption of plane stress, , we obtain the expression of and deduce the circumferential, or hoop stress . For an incompressible material, , where and denote the deformed radius and thickness, respectively. We denote the circumferential stretch ratio, and deduce the normal stretch ratio . The calculation follows (37,38) and yields the expression of the hoop stress of a Saint-Venant Kirchhoff spherical shell, as given by Eq. 11.
Results
Multiplexed microaspiration setup
To define and measure the rheological properties of Hydra tissue spheres, we settled on the use of microaspiration experiments. These experiments are now well established and robust and measure rheological properties at length scales and frequencies relevant for the regenerative process. In contrast to atomic force microscopy (AFM), previously used to locally probe the stiffness of adult Hydras (39), our experiments were particularly designed to study the mechanics of tissue spheres during the osmotic oscillations, which is why we employed elongations rather than indentations and probed the large deformation regime. Their main drawback is their intrinsically low throughput. In their original form, microaspiration experiments can only probe one sample at a time with a single experiment running for around 1 h (4,33). We thus started by developing and adapting a microfluidics device making use of removable inserts (36) to parallelize the experiment, in the same spirit as the technique developed in (40) but with circular pipettes, which prevent singularities and leakage. This device was composed of two different objects. First, a microfluidic channel was designed to host the samples, 3 cm in length and 500 μm in height (Figs. 1 A and S1). Halfway through this channel and perpendicular to it sat another channel meant to host the removable insert. This channel was 500 μm wide and 1 mm high. This whole construct was then manufactured in polydimethylsiloxane (PDMS). Most of the experiments presented here were carried out with removable inserts, microfabricated in dry film, bearing either 6 or 10 circular openings, 500 μm or 300 μm away from one another, 300 μm in length and 100 μm in diameter. Of note, these holes sat, vertically, at the midpoint of the samples’ channel (250 μm, Fig. 1 B). Tissue spheres aspirated in these holes therefore did not touch either the bottom or the top of the PDMS channel, which had no influence on the aspiration.
Figure 1.
Parallelized microaspiration experiments. (A) Schematic of the mold for the PDMS channel (details on dimensions can be found in Fig. S1). (B) Representation of the principle of the experiment. The insert, in orange, effectively creates an array of cylindrical tunnels, equivalent to model micropipettes. The tissue spheres, in yellow, flow toward these openings and, once sealed, are aspirated within. (C) Snapshot of a mounted channel with an insert containing ten tunnels in parallel. (D) Fluorescent imaging of the ectoderm of six samples aspirated within the tunnels. To see this figure in color, go online.
Using hydrostatic pressure, we applied a difference in pressure between the inlet and outlet of the main chamber (with , see Fig. S2), which initially created a flow of water from the Petri dish to the outlet of the chamber. Previously prepared Hydra tissue spheres were then manually pipetted into the entrance of the main channel. Thanks to the flow of water, the samples naturally aligned with the openings in the insert. Once all openings were blocked by a sample, the flow of water stopped and aspiration of the tissue spheres was observed (Fig. 1 D and Video S1). Of note, in most experiments, the pressure difference was set to 0 as soon as the samples aligned with the holes so that a controlled step in pressure could be applied thereafter.
Rheological behavior depends on applied stress
Using this multiplexed setup, we started by studying the response of newly formed Hydra tissue spheres to microaspirations in the kPa range using circular openings with a radius = 50 μm. We observed two different rheological responses depending on the applied pressure. Up to around 2.5 kPa, tissue spheres exhibited an elastic response in the sense that for a given applied pressure difference, a tongue of tissue was aspirated within the holes, the length of which increased in the first tens of seconds but then became saturated and remained constant over much longer timescales. When the pressure was released, we also observed that the samples retrieved their original shape and that aspiration and release dynamics were very similar (Fig. S3), consistent with the reversible behavior of an elastic material. To further ensure that we were not missing viscous behavior at longer timescales, we managed to trap tissue spheres for tens of hours in this elastic phase without any indication of tissue flow (Video S2).
Endoderm is shown in magenta and ectoderm in green.
However, when we increased the applied pressure, we started observing samples flowing inside the holes, a signature of viscous behavior. This demonstrates a real change in mechanical behavior of the tissue spheres akin to the behavior of yield stress fluids and requiring two different experimental and theoretical approaches.
Finally, at even higher applied stresses, we started observing rupture of the tissue and cell detachments, reminiscent of the rupture observed under normal phase I osmotic oscillations (Video S3). Since all three behaviors seemed relevant to understanding the mechanical behavior of normally regenerating tissue spheres, we decided to characterize them all, as well as the pressure thresholds between these different phases.
Endoderm is shown in magenta and ectoderm in green.
Elastic behavior at low applied stresses
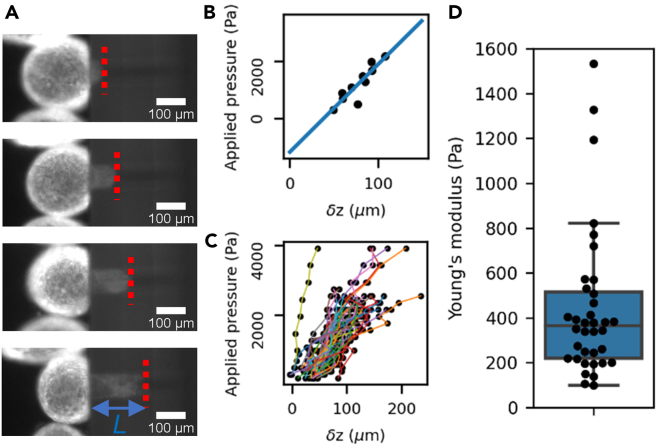
To characterize the elastic behavior of the tissue spheres, we applied steps of pressure on the same samples and recorded, for each step and for each sample, the length of the aspirated tongue (Fig. 2 A). We monitored the samples closely to ensure they did not start flowing in the holes, in which case their analysis was stopped. As expected, we found that the higher the pressure, the longer the aspirated tongue (Fig. 2 A). To estimate a Young’s modulus from these measurements, we needed a rheological model of an elastic spherical shell of mean radius and thickness .
Figure 2.
Elastic behavior quantification. (A) Snapshots of one tissue sphere aspirated in a hole at increasing pressure steps (top to bottom). The dashed red line shows the position of the aspirated tongue at each step, and L is the aspirated length. (B) Linear relationship between the applied pressure and for a single tissue sphere and 10 different pressure steps showing both the linear behavior (fitted as a blue line) and negative intercept. (C) Same measurement as in (B), repeated on n = 36 tissue spheres, each represented by a differently colored line. (D) distribution of Young’s moduli obtained by this method. Black dots are individual measurements; the box plot shows the median and quartiles of the distribution. We found E = (4.4 ± 3.3) × 102 Pa (mean ± standard deviation, n = 36). To see this figure in color, go online.
As is the case for cell aggregates, we expect that intercellular adhesion and cellular cortical tension will contribute to an effective surface tension of the Hydra tissue spheres, which we denote by .
A pressure difference through the micropipette of radius generates an aspiration force . For an aspirated length , the free energy of the aspirated shell reads
| (Equation 1) |
where denotes the surface area of the sample and the factor 2 takes into account the presence of two interfaces. Assuming incompressibility, the volume of the sample is constant. For small enough deformations during aspiration, we expected that the thickness of the sample would remain constant. To validate this hypothesis, we performed spinning-disk confocal microscopy on normal tissue spheres and aspirated them to reconstruct their 3D structure and estimate their thicknesses. We found in both cases and did not observe any clear change due to microaspiration (Fig. S4). Since , the surface area of the sample also remains constant . In other words, in the case of the small deformations of an incompressible shell of approximately constant thickness, tissue surface tension does not contribute to the total force exerted on the sample:
| (Equation 2) |
Indeed, our experiments did not exhibit a clear half-sphere of radius , typical of the early entry of a full cell aggregate into a micropipette (41). In addition, Guevorkian et al. elegantly demonstrate that surface tension manifests itself as a critical pressure below which no aspiration is observed and which needs to correct all subsequent applied pressures (33). The fact that we observed aspirations with applied pressures as low as 200 Pa also indicates that surface tension can largely be ignored in our experiments.
The rest state of the system is simply that of the initial spherical shell of radius , and due to that initial curvature, the displacement of the tip of the tissue tongue inside the micropipette reads
| (Equation 3) |
As expected for an elastic material, we found a linear relationship between this displacement and the applied pressure (Fig. 2 B). We also noticed that for many samples, our linear fits had negative intercepts at the origin. This could be the signature of a stress-stiffening effect by which the effective Young’s modulus would increase with the applied pressure, i.e., the existence of nonlinear elastic behavior, as observed in suspended cell monolayers (42).
To take into account this observation, we turned to a nonlinear, hyperelastic description of the Hydra tissue, using, as previously advocated by (43), the Saint-Venant Kirchhoff model, defined by the strain energy function
| (Equation 4) |
where and denote the Lamé coefficients and is the Green-Lagrange strain tensor (37). This equation reduces to its linear elastic counterpart for small deformations, when with the infinitesimal strain tensor. Of note, the Saint-Venant Kirchhoff model is defined by the same parameters as would be the case for a linear elastic material, with the following classical relationships between Lamé coefficients and other elastic moduli: and , respectively, the Young’s modulus, and the Poisson’s ratio of the sample.
| (Equation 5) |
| (Equation 6) |
In analogy with the behavior of linearly elastic thin spherical shells (see, e.g., (44)), and taking into account the non-zero intercept of the linear fits of our data (Fig. 2 C, ), we expect that the displacement and the applied pressure should be related according to
| (Equation 7) |
where is a dimensionless, geometry-dependent pre-factor. Of note, in this description, we ignored the complex composite structure of the shell, which includes two different epithelial layers as well as the extracellular matrix.
To estimate , we performed numerical simulations of microaspiration of hyperelastic spherical shells by a single cylindrical tunnel (Fig. 3 A). In our experimental data, we had (mean ± standard deviation, n = 36) and . As we had no measurement of the Poisson’s ratio of our samples, we made an assumption of near incompressibility and set . Based on these values, we submitted spherical shells to applied pressure and measured the resulting . As shown in Fig. 3 B, these simulations confirmed the linear relationship between and over a similar range of applied pressures and displacements and allowed to estimate thanks to knowledge of the value of and . Of note, other element-based numerical simulations were performed with parameters varying in the following ranges: , , and with a fillet size varying between 2 and 10 μm. Overall, our estimate of the coefficient reads , in the vicinity of 23, suggesting that the order of magnitude of our results was unaffected by uncertainties on these different parameters. Further, our estimate of the intercept in these simulations is , consistent with the experimental values.
Figure 3.
Numerical simulations. (A) Graphical representation of the deformed shell. The color code represents the z-component of the displacement field. Black lines delineate the reference state of the system. Parameter values for these simulations are . (B) applied pressure versus the displacement for the same parameter values. The slope of the fitted line, in orange, corresponds to a value . To see this figure in color, go online.
Thanks to our multiplexed setup, we were able to measure up to six samples in parallel in one sitting. By then repeating these experiments eight different times, we achieved a total of 36 tissue spheres (Fig. 2 C) characterized in that phase. For each of them, we extracted the slope of the linear relationship between and and its initial radius , and finally obtained a quantitative measurement of its Young’s modulus using Eq. 7 (Fig. 2 D). From the above estimate of the coefficient , we found the Young’s modulus of Hydra tissue spheres to be (4.4 ± 3.3) × 102 Pa (mean ± standard deviation, n = 36).
We also observed a large variability in our measurements of individual Young’s moduli (Fig. 2 D), which could hint at subtle effects missed by our averaging approach. The different tissue spheres could differ in initial size or degree of inflation, since we did not precisely control the time they need to fold back into tissue spheres after being cut. To better understand this variability, we looked at the correlation between the samples’ radii and their extracted elastic moduli. We indeed found a positive correlation, albeit weak, between these measurements (Fig. S5). We then asked whether this correlation could be due to differences in inflation state. To do so, we ran new experiments measuring the elastic moduli of tissue spheres whose oscillations were blocked by the addition of 70 mM sucrose in the outside medium. Doing so, we found 1) that the obtained elastic moduli distributions in both cases were not statistically different (Fig. S6) and 2) that addition of sucrose abolished neither the weak positive correlation between size and elasticity (Fig. S5) nor the existence of stiffer samples. We conclude that some, but not all, of the observed variability comes from differences in initial size rather than inflation rate between different samples. A better understanding of this dependency would be a natural continuation of this work.
In addition, we validated the relevance and precision of our experimental procedure by repeating elastic measurements under two chemical modulations, EDTA and blebbistatin. EDTA is a chemical well known for weakening cell-cell adhesions in many epithelial tissues (45), whereas blebbistatin inhibits actomyosin contractility (46). We expected that both treatments should lower the samples’ elasticity either by allowing larger tissue-scale deformations in the case of EDTA or cell-scale deformations in the case of blebbistatin by making the actin cortex of each cell softer. We indeed found a clear and statistically significant decrease in Young’s modulus with both treatments (Fig. S6; EDTA: E = (3.0 ± 2.0) × 102 Pa, blebbistatin: E = (1.9 ± 0.7) × 102 Pa), confirming that the observed elastic behavior of the tissue spheres stems from a complex interplay of single-cell mechanics and tissue structure.
Overall, we characterized in a quantitative way the response of Hydra tissue spheres to applied stresses and found them to behave hyperelastically, i.e., as soft () elastic materials at small deformations, in line with AFM measurements on adult Hydra (39), while they become increasingly stiffer as the applied pressure increases.
Viscoelastic behavior at intermediate applied stresses
We then turned our attention to the next observed behavior, which involved flowing of the samples within the tunnels. This viscoelastic behavior has long been observed and studied, both on single cells (32,47,48) and multicellular spheroids (4,33,41). We followed the usual approach in that situation where a single, controlled pressure step was applied in the device at t = 0 after the samples were already placed close to the entrance of the tunnels. The dynamics of the aspirated length was then recorded by fluorescent microscopy for tens of minutes (Fig. 4 A). It was important to keep recording on these longer timescales in order to be certain of the behavior adopted by each sample, as a very slow flow could easily be misinterpreted as an elastic response if the observation timescale was too short.
Figure 4.
Viscoelastic flow at large applied pressure. (A) Snapshots of a tissue sphere showing flow at a constant applied pressure. (B) four examples of typical dynamics of the flow as a function of time. Dots of different colors represent four independent samples and solid lines fits by Eq. 8. (C) Distribution of measured viscosities as black dots . The box plot shows the median and quartiles of the distribution. We found it to be (2.4 ± 1.2) × 106 Pa s (mean ± standard deviation, n = 22). To see this figure in color, go online.
Since the behavior was qualitatively similar to that observed for cellular aggregates aspirated in cylindrical micropipettes, we first applied the well-known modified Maxwell model developed in (33). In brief, a dashpot representing tissue-scale viscosity is combined in series with a standard linear solid unit incorporating cell-scale viscoelasticity as well as tissue-scale elasticity. We fitted our data with the form of predicted by this model:
| (Equation 8) |
where is linked to the short-term elastic deformation, is a constant linked to the relative contributions of the two springs in the standard linear solid unit, is the cell-scale viscoelastic relaxation time, and is the long-term viscous flow rate. We found that this functional form was also relevant to describe our data quantitatively (Fig. 4 B) despite the morphological differences between our samples (empty shells) and ball-shaped aggregates (full spheres). We could, however, employ a scaling argument similar to the one applied in this model to estimate an order of magnitude of the effective viscosity of our samples. Regarding cellular aggregates, friction between the tissue and the tunnel walls can be neglected, since the long-term aspirated length scales not as the square root of time but as a linear function of time. Assuming that dissipation is due to cell rearrangements at the entrance of the tunnels, thus acting on a length scale of order , the total dissipative force can be approximated as , with the viscosity of the samples and a dimensionless, geometrical pre-factor equal to for a ball-shaped aggregate and unknown in our geometry. This viscous force has to be balanced by the aspiration force that we have shown to be , which leads to
| (Equation 9) |
To the best of our knowledge, the computation of has never been done for spherical viscous shells and in the same geometry as the one in our experiments. We will see below that a precise measurement of this effective viscosity has little impact on the description of Hydra osmotic oscillations, and we limited ourselves to obtaining an order of magnitude estimation of the viscosity by taking . Note that assuming that tissue-scale dissipation acts across the thickness h of this multilayered tissue does not change our estimate, as and have the same order of magnitude. Again, our microaspiration setup allowed us to efficiently gather data on multiple samples and combining them, whereby we found an estimation of the viscosity of (2.4 ± 1.2) × 106 Pa s (mean ± standard deviation, n = 22, Fig. 4 C).
The full dynamics of aspiration in that phase can also yield measurements of the Young’s modulus of the samples. In line with our analysis of the purely elastic response, we expect here that
| (Equation 10) |
Similar to Eq. 7, with the immediate elastic aspirated length in the notation of this model. Applying this relationship to the results of our fitted data, we found (Fig. S7), in accordance with our results on the purely elastic behavior of Hydra tissue spheres.
These viscoelastic experiments were made difficult by the fact that in many instances we observed, in addition to the flow of the samples, cells detaching at the tip of the aspirated tongue, indicative that the tissues were already rupturing. These cases were obviously discarded from the analysis, limiting the amount of available data. Overall, it appeared that at these applied pressures the viscoelastic phase and rupture were often occurring together.
Critical pressures between different behaviors
To further study this question, we then turned our attention to the critical pressures required to transition between the three regimes: hyperelastic, viscoelastic, and rupture. We often found cases where, for a given applied pressure, a fraction of the samples loaded in the same setup would behave as elastic solids while others behaved as viscoelastic fluids (Fig. 5 A). To characterize the first critical pressure, we therefore measured, at different applied pressures, the fraction of samples showing a viscoelastic versus elastic behavior while discarding rupturing ones. We fitted this fraction as a function of the applied pressure by a sigmoid (see materials and methods; Fig. 5 B). We defined the critical pressure as the center of this sigmoid where 50% of samples had switched from one behavior to the other. Following this method, we found the critical pressure between elastic and viscoelastic behaviors to be 2.22 ± 0.14 kPa (error bars represent the 95% confidence interval of the sigmoid fit). We then employed the same strategy to characterize the critical pressure leading to tissue rupture (Fig. 5 C). We found it to be 2.37 ± 0.08 kPa, very close to the previous value, confirming our intuition that both behaviors were linked together and that tissue flows inside the holes were already a signature of their lack of integrity.
Figure 5.
Critical stresses between different mechanical behaviors. (A) Snapshot of an experiment showing two similar samples displaying different behaviors at the same applied pressure. (B) Quantification of the fraction of viscous behavior at different applied pressures. (C) Quantification of the fraction of samples showing tissue rupture as a function of applied pressure. In (B) and (C), dots are data and solid lines fit by a sigmoid function. In black are control samples and in red samples pretreated with 2 mM EDTA. Each point is derived from 18–40 samples stemming from eight different experiments in (B) and four in (C). (D) Numerical simulations. The color code represents the hoop stress within the aspirated shell when aspirated at a pressure of 1 kPa. Parameter values are the same as in Fig. 3. To see this figure in color, go online.
The most natural explanation for this lack of integrity is the weakening and rupture of cell-cell contacts, since we often observed detachment of single cells, making a possible de-adhesion of the tissues to the extracellular matrix unlikely. To verify this hypothesis, we repeated our measurement of the critical pressures required to induce either a viscous behavior or tissue rupture on tissue spheres pretreated with 2 mM EDTA, as we expected this treatment to facilitate the induction of a viscous behavior and tissue rupture. This is indeed what we found, with a shift of the critical pressure from 2.22 kPa to 1.19 ± 0.16 kPa for the viscous behavior (Fig. 5 B) and from 2.37 kPa to 1.29 ± 0.19 kPa for rupture (Fig. 5 C).
It is important to note that all these values are difference of pressure applied between the two sides of the holes and not necessarily the stress actually acting in the tissue, called the hoop stress. Said differently, the applied pressure required to rupture the tissue should not be directly taken as a measurement of the ultimate tensile stress of the whole tissue. To relate the applied pressure and internal tissue stresses, we turned again to numerical simulations. Within the range of parameters studied, the maximal values of the hoop stress we found was on the order of a 2–7 kPa (see Fig. 5 D for an example).
Since in normal conditions the threshold for viscous behavior cannot be clearly distinguished from the threshold for rupture, our measurements revealed that up to rupture, which is also observed in normal osmotic oscillations, regenerating Hydra tissue spheres behave as hyperelastic spherical shells with a stress-stiffening effect.
Mechanical description of oscillating Hydra tissue spheres during regeneration
After folding back into a spherical shape, regenerating Hydra tissue spheres experience a series of swelling-rupture cycles, which have been shown to be driven by the difference in osmolarity between the inside and outside of the spheres. These oscillations are required for the proper morphogenesis and regeneration of the samples, although their exact role remains unknown. Thanks to previous work on the topic (5,49) and our own mechanical characterization, we can provide a good understanding of the internal tissue mechanics occurring during this phase.
The key quantity is the circumferential or hoop stress, noted here , which is known to control tissue integrity and to be able to alter key biological processes. It is given for an incompressible, thin elastic spherical shell of a Saint-Venant Kirchhoff material (see (37,38) and materials and methods) by the expression
| (Equation 11) |
where is the stretch ratio and and the Lamé coefficients. This gives the nonlinear constitutive relationship between stresses and strains in the spherical shell during the oscillations, which allows to directly extract the hoop stress from a measurement of the radius of the sample as a function of time (see Fig. S8). One can check that Eq. 11 reduces to the relationship expected of a linear elastic thin shell in the limit of small deformations where :
| (Equation 12) |
At larger deformations, the tissues get stiffer and the experimentally measured values of imply that, in this regime, the differential elastic modulus is on the order of 10 kPa, in accordance with previous estimates of the sphere’s stiffness during osmotic oscillations (5).
Based on our experimental observations, we will approximate our samples as hyperelastic, symmetric, spherical shells of time-dependent thickness and radius (Fig. S8) submitted to a difference in osmolite concentrations . The fact that the spheres are constantly swelling indicates a constant influx of water into their lumen and that the system never reaches osmotic equilibrium. Furthermore, Kücken et al. (5) showed that the increase of can be described by a Darcy-type law of the form
| (Equation 13) |
where is the volume of the sphere, is the water permeability coefficient, and is Boltzmann’s constant. The dynamics of swelling is thus entirely controlled by the influx of water, which imposes a constantly increasing deformation on the tissue sphere with a growth rate of the radius on the order of . This influx of water effectively inflates the sphere and creates a restoring elastic pressure , which our mechanical description can also predict. Following the mechanics of spherical thin shells, this pressure can be written as
| (Equation 14) |
Using the experimental measurement of we previously obtained and the same value of the Poisson’s ratio, , we were able to approximate the values of and (). Using these values and experimental measurements of during the oscillations allowed us to estimate , which we found to be on the order of a few kPa (Fig. S8). Most notably, this approach allowed us to estimate the critical stress, within the tissue, to induce rupture, which we found to be on the order of 4–6 kPa. This is the same order of magnitude as the maximum hoop stress obtained by numerical simulations of the elastic deformation during micropipette aspiration. Overall, we demonstrate that our results allow mapping of measurements of the radius of the tissue spheres, which are experimentally easy to access, into both internal stresses within the tissue and restoring elastic pressure acting on the fluid inside of the spheres.
Discussion
The effect of stretching on the biology of epithelial tissues has emerged in recent years as an important topic in developmental biology, since developing organisms often undergo substantial deformations which have an increasingly recognized impact on their biological response. In the context of Hydra regeneration, Ferenc et al. observed that the levels of expression of Wnt ligands, especially HyWnt3, are directly linked to the cumulative amount of stretching in the tissue (20). In a similar fashion, stretching adhering monolayers of epithelial cells has been shown to induce cell division through the activation of Piezo1 mechanosensitive ion channels (22) to restore proper cell density. Anisotropic deformations also have a large impact on the tissue as uniaxial cyclic stretch leads to reorientation of the cells perpendicular to the direction of application (50,51), as was observed for the mitotic spindle of single cells (52). In many of these experiments, however, stresses within the tissues could not be recorded after stretching.
One notable exception was achieved on freely suspended epithelia axially stretched with a measurable force (42). This experimental setup allowed the authors to characterize the rheology of Madine-Darby canine kidney monolayers. Interestingly, they observed a nonlinear response with three distinct phases: a linear one at small deformations with a small equivalent elastic modulus, a second linear one for intermediate values of stresses and strains with much higher stiffness, and a third one corresponding to the rupture of the tissue. This behavior is very similar to the one we observed in this work. The authors quantified the second phase by linear fitting of the stress-strain curves with negative intercepts, as we did. Since they approximated the tissue in this phase as linearly elastic, they found a high elastic modulus on the order of 20 kPa, similar to our observations of Hydra tissue sphere differential moduli at intermediate stresses. Finally, the authors found that the monolayer was able to withstand deformations of up to 60% before rupturing, a value slightly higher than what we observed for Hydra tissue spheres. Our results are thus well in line with these observations, and both ask the question of the biological origin of the nonlinear behavior of epithelial tissues, which remains elusive for the moment.
Regarding the specific rheology of Hydra, our work is the first to focus specifically on regenerating tissue spheres, but measurements exist on full adults. Naik et al. employed a modified AFM experimental procedure to generate indentations on fixed, full Hydra with a 20-μm-wide sphere (39). By co-recording the deformation of the tissue and the force exerted, they observed a linear relationship between both, which allowed them to estimate the elastic modulus of these samples. One key result was the observation that the elasticity of the body column was inhomogeneous, the tissue being roughly three times stiffer near the head than close to the foot. Although it is a well-known fact that different experimental approaches can lead to different estimations of rheological properties (53) and potential differences between fully mature Hydras and tissue spheres, our results are in good agreement with this work. Indeed, in the softest part of the adult, which we would imagine to most resemble tissue spheres, the authors report an elastic modulus on the order of 400 Pa. Since the deformations involved are of a few micrometers, we expect these measurements to probe the small deformation regime where our own measurements also find an elastic modulus on the order of 400 Pa. In the large deformation regime, our results also confirm those of (5) which, in order to properly reproduce the osmotic oscillations based on chemical considerations, implemented an elastic modulus . Still, AFM measurements on tissue spheres, albeit technically challenging, would be a great complement to our work to obtain a comprehensive understanding of the mechanics of these samples.
Another consequence of our focus on osmotic oscillations and tissue-scale mechanics is that our mechanical description of the tissue spheres assumes a homogeneous shell, ignoring the complex three-layer (endoderm-extracellular matrix-ectoderm) structure of the samples. It is clearly possible that the deformations and stresses induced by the osmotic oscillations are differently transmitted and have different effects on these different layers, a refinement that will need to be addressed in the future. In particular, it was recently suggested that the mechanical properties of the extracellular matrix, which were not specifically measured here, could be linked to morphogenesis in Hydra (54).
Since we believe deformations and stresses within the tissues to be central in the morphogenesis of Hydra, another natural question is the existence of inhomogeneities of deformations, forces, or rheological properties within the same sample. Mechanical fluctuations, if coupled to biochemistry, could well be sufficient to destabilize a reaction-diffusion system into a patterned state. Our own experimental setup could allow us to measure fluctuations in rheological properties by repeating measurements on the same samples with small holes but at different locations, the main limitation being the lack of control of the position of the tested zone. Using the same logic, it has been proposed that the amount of HyWnt3 protein could locally affect the Young’s modulus of the tissue (25), which could couple the chemical patterning of the head organizer and morphogenesis. If the future location of the head becomes softer, the homogeneous osmotic pressure will deform this region more, leading to a deviation from a spherical shape to an oblong one. Here, too, our setup could allow measurement of the Young’s modulus of tissue spheres overexpressing HyWnt3 to directly test this hypothesis.
Conclusions
In this work, we have employed an original parallelized microaspiration device to characterize the mechanical behavior of Hydra tissue spheres. Although we found different rheological behaviors depending on the applied stresses, the main result we obtained is that during normal osmotic oscillations, these samples behave largely as hyperelastic thin shells, displaying a nonlinear, stress-stiffening rheology. Doing so, we also obtained quantitative measurements of their relevant rheological parameters ( s, critical applied pressures for viscous behavior and tissue rupture). This allowed us to estimate the stresses acting within the tissues and, in particular, the critical stress at which they start rupturing. These results, by allowing a quantification of both stresses and deformations during Hydra regeneration, will hopefully pave the way for quantitative approaches aiming at correlating these mechanical cues with molecular ones to understand Hydra patterning as an integrated mechanobiochemical process.
Author contributions
O.C.-E. designed the research. Z.B.M., M.D., and P.J. manufactured the inserts. T.P., A.B.-B., P.M., and O.C.-E. performed the research and analyzed data. T.P., P.M., and O.C.-E. wrote the manuscript, which was reviewed by all authors.
Acknowledgments
We thank H. Delanoë-Ayari for fruitful discussions.
This work was funded by the Mission pour les Initiatives Transverses et Interdisciplinaires of CNRS (Project MeChemReg to O.C.-E. and P.M.).
Declaration of interests
The authors declare no competing interests.
Editor: Paul Janmey.
Footnotes
Supporting material can be found online at https://doi.org/10.1016/j.bpj.2024.05.022.
Supporting material
References
- 1.Galliot B., Schmid V. Cnidarians as a model system for understanding evolution and regeneration. Int. J. Dev. Biol. 2002;46:39–48. [PubMed] [Google Scholar]
- 2.Technau U., Steele R.E. Evolutionary crossroads in developmental biology: Cnidaria. Development. 2011;138:1447–1458. doi: 10.1242/dev.048959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gierer A., Berking S., et al. Trenkner E. Regeneration of hydra from reaggregated cells. Nat. New Biol. 1972;239:98–101. doi: 10.1038/newbio239098a0. [DOI] [PubMed] [Google Scholar]
- 4.Cochet-Escartin O., Locke T.T., et al. Collins E.-M.S. Physical Mechanisms Driving Cell Sorting in Hydra. Biophys. J. 2017;113:2827–2841. doi: 10.1016/j.bpj.2017.10.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kücken M., Soriano J., et al. Nicola E.M. An osmoregulatory basis for shape oscillations in regenerating hydra. Biophys. J. 2008;95:978–985. doi: 10.1529/biophysj.107.117655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fütterer C., Colombo C., et al. Ott A. Morphogenetic oscillations during symmetry breaking of regenerating Hydra vulgaris cells. Europhys. Lett. 2003;64:137–143. [Google Scholar]
- 7.Broun M., Gee L., et al. Bode H.R. Formation of the head organizer in hydra involves the canonical Wnt pathway. Development. 2005;132:2907–2916. doi: 10.1242/dev.01848. [DOI] [PubMed] [Google Scholar]
- 8.Bode H.R. Axial Patterning in Hydra. Cold Spring Harbor Perspect. Biol. 2009;1 doi: 10.1101/cshperspect.a000463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Böttger A., Hassel M. Hydra, a model system to trace the emergence of boundaries in developing eumetazoans. Int. J. Dev. Biol. 2012;56:583–591. doi: 10.1387/ijdb.113454ab. [DOI] [PubMed] [Google Scholar]
- 10.Soriano J., Colombo C., Ott A. Hydra molecular network reaches criticality at the symmetry-breaking axis-defining moment. Phys. Rev. Lett. 2006;97 doi: 10.1103/PhysRevLett.97.258102. [DOI] [PubMed] [Google Scholar]
- 11.Turing A.M. The Chemical Basis of Morphogenesis. Philos. Trans. R. Soc. B Biol. Sci. 1952;237:37–72. [Google Scholar]
- 12.Marcon L., Sharpe J. Turing patterns in development: What about the horse part? Curr. Opin. Genet. Dev. 2012;22:578–584. doi: 10.1016/j.gde.2012.11.013. [DOI] [PubMed] [Google Scholar]
- 13.Schweisguth F., Corson F. Self-Organization in Pattern Formation. Dev. Cell. 2019;49:659–677. doi: 10.1016/j.devcel.2019.05.019. [DOI] [PubMed] [Google Scholar]
- 14.Gierer A., Meinhardt H. A theory of biological pattern formation. Kybernetik. 1972;12:30–39. doi: 10.1007/BF00289234. [DOI] [PubMed] [Google Scholar]
- 15.Nakamura Y., Tsiairis C.D., et al. Holstein T.W. Autoregulatory and repressive inputs localize Hydra Wnt3 to the head organizer. Proc. Natl. Acad. Sci. USA. 2011;108:9137–9142. doi: 10.1073/pnas.1018109108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Augustin R., Franke A., et al. Bosch T.C.G. Dickkopf related genes are components of the positional value gradient in Hydra. Dev. Biol. 2006;296:62–70. doi: 10.1016/j.ydbio.2006.04.003. [DOI] [PubMed] [Google Scholar]
- 17.Vogg M.C., Beccari L., et al. Galliot B. An evolutionarily-conserved Wnt3/β-catenin/Sp5 feedback loop restricts head organizer activity in Hydra. Nat. Commun. 2019;10:312. doi: 10.1038/s41467-018-08242-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ziegler B., Yiallouros I., et al. Özbek S. The Wnt-specific astacin proteinase HAS-7 restricts head organizer formation in Hydra. BMC Biol. 2021;19:120–122. doi: 10.1186/s12915-021-01046-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Soriano J., Rüdiger S., et al. Ott A. Mechanogenetic coupling of Hydra symmetry breaking and driven Turing instability model. Biophys. J. 2009;96:1649–1660. doi: 10.1016/j.bpj.2008.09.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ferenc J., Papasaikas P., et al. Tsiairis C.D. Mechanical oscillations orchestrate axial patterning through Wnt activation in Hydra. Sci. Adv. 2021;7:6897. doi: 10.1126/sciadv.abj6897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Desprat N., Supatto W., et al. Farge E. Tissue deformation modulates twist expression to determine anterior midgut differentiation in Drosophila embryos. Dev. Cell. 2008;15:470–477. doi: 10.1016/j.devcel.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 22.Gudipaty S.A., Lindblom J., et al. Rosenblatt J. Mechanical stretch triggers rapid epithelial cell division through Piezo1. Nature. 2017;543:118–121. doi: 10.1038/nature21407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pouille P.-A., Ahmadi P., et al. Farge E. Mechanical signals trigger Myosin II redistribution and mesoderm invagination in Drosophila embryos. Sci. Signal. 2009;2 doi: 10.1126/scisignal.2000098. [DOI] [PubMed] [Google Scholar]
- 24.Shinozuka T., Takada R., et al. Takada S. Wnt produced by stretched roof-plate cells is required for the promotion of cell proliferation around the central canal of the spinal cord. Devenir. 2019;146 doi: 10.1242/dev.159343. [DOI] [PubMed] [Google Scholar]
- 25.Mercker M., Köthe A., Marciniak-Czochra A. Mechanochemical symmetry breaking in Hydra aggregates. Biophys. J. 2015;108:2396–2407. doi: 10.1016/j.bpj.2015.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brinkmann F., Mercker M., et al. Marciniak-Czochra A. Post-Turing tissue pattern formation: Advent of mechanochemistry. PLoS Comput. Biol. 2018;14 doi: 10.1371/journal.pcbi.1006259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang R., Goel T., et al. Collins E.-M.S. Mouth Function Determines the Shape Oscillation Pattern in Regenerating Hydra Tissue Spheres. Biophys. J. 2019;117:1145–1155. doi: 10.1016/j.bpj.2019.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Livshits A., Shani-Zerbib L., et al. Keren K. Structural Inheritance of the Actin Cytoskeletal Organization Determines the Body Axis in Regenerating Hydra. Cell Rep. 2017;18:1410–1421. doi: 10.1016/j.celrep.2017.01.036. [DOI] [PubMed] [Google Scholar]
- 29.Wang R., Steele R.E., Collins E.M.S. Wnt signaling determines body axis polarity in regenerating Hydra tissue fragments. Dev. Biol. 2020;467:88–94. doi: 10.1016/j.ydbio.2020.08.012. [DOI] [PubMed] [Google Scholar]
- 30.Wang R., Bialas A.L., et al. Collins E.M.S. Mechano-Chemical Coupling in Hydra Regeneration and Patterning. Integr. Comp. Biol. 2023;63:1422–1441. doi: 10.1093/icb/icad070. [DOI] [PubMed] [Google Scholar]
- 31.Veschgini M., Gebert F., et al. Tanaka M. Tracking mechanical and morphological dynamics of regenerating Hydra tissue fragments using a two fingered micro-robotic hand. Appl. Phys. Lett. 2016;108 [Google Scholar]
- 32.Evans E.A. New Membrane Concept Applied to the Analysis of Fluid Shear- and Micropipette-Deformed Red Blood Cells. Biophys. J. 1973;13:941–954. doi: 10.1016/S0006-3495(73)86036-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Guevorkian K., Colbert M.-J., et al. Brochard-Wyart F. Aspiration of Biological Viscoelastic Drops. Phys. Rev. Lett. 2010;104:218101–218104. doi: 10.1103/PhysRevLett.104.218101. [DOI] [PubMed] [Google Scholar]
- 34.Hochmuth R.M. Micropipette aspiration of living cells. J. Biomech. 2000;33:15–22. doi: 10.1016/s0021-9290(99)00175-x. [DOI] [PubMed] [Google Scholar]
- 35.Lim C.T., Zhou E.H., Quek S.T. Mechanical models for living cells--a review. J. Biomech. 2006;39:195–216. doi: 10.1016/j.jbiomech.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 36.Landiech S., Elias M., et al. Joseph P. Parallel on-chip micropipettes enabling quantitative multiplexed characterization of vesicle mechanics and cell aggregates rheology. bioRxiv. 2023 doi: 10.1101/2023.10.19.562871. Preprint at. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Holzapfel G.A. Wiley; Hoboken: 2000. Nonlinear Solid Mechanics: A Continuum Approach for Engineering Science. [Google Scholar]
- 38.Green A.E., Zerna W. Dover Publications; Mineola: 1968. Theoretical Elasticity. [Google Scholar]
- 39.Naik S., Unni M., et al. Galande S. Differential tissue stiffness of body column facilitates locomotion of Hydra on solid substrates. J. Exp. Biol. 2020;223 doi: 10.1242/jeb.232702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Boot R.C., Roscani A., et al. Boukany P.E. High-throughput mechanophenotyping of multicellular spheroids using a microfluidic micropipette aspiration chip. Lab Chip. 2023;23:1768–1778. doi: 10.1039/d2lc01060g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guevorkian K., Brochard-Wyart F., Gonzalez-Rodriguez D. Viscoelasticity and Collective Cell Migration: An Interdisciplinary Perspective across Levels of Organization. Academic Press; 2021. Flow dynamics of 3D multicellular systems into capillaries; pp. 193–223. [Google Scholar]
- 42.Harris A.R., Peter L., et al. Charras G.T. Characterizing the mechanics of cultured cell monolayers. Proc. Natl. Acad. Sci. USA. 2012;109:16449–16454. doi: 10.1073/pnas.1213301109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Brinkmann F. Ruprecht-Karls-Universität; 2020. Mathematical Models and Numerical Simulation of Mechanochemical Pattern Formation in Biological Tissues. PhD Thesis. [Google Scholar]
- 44.Ugural A.C. third edition. CRC Press; Boca Raton: 2009. Stresses in Beams, Plates, and Shells. [Google Scholar]
- 45.Takeichi M. Functional correlation between cell adhesive properties and some cell surface proteins. J. Cell Biol. 1977;75:464–474. doi: 10.1083/jcb.75.2.464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Straight A.F., Cheung A., et al. Mitchison T.J. Dissecting temporal and spatial control of cytokinesis with a myosin II inhibitor. Science. 2003;299:1743–1747. doi: 10.1126/science.1081412. [DOI] [PubMed] [Google Scholar]
- 47.Sato M., Theret D.P., et al. Nerem R.M. Application of the Micropipette Technique to the Measurement of Cultured Porcine Aortic Endothelial Cell Viscoelastic Properties. J. Biomech. Eng. 1990;112:263–268. doi: 10.1115/1.2891183. [DOI] [PubMed] [Google Scholar]
- 48.Tsai M.A., Frank R.S., Waugh R.E. Passive mechanical behavior of human neutrophils: power-law fluid. Biophys. J. 1993;65:2078–2088. doi: 10.1016/S0006-3495(93)81238-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ruiz-Herrero T., Alessandri K., et al. Mahadevan L. Organ size control via hydraulically gated oscillations. Development. 2017;144:4422–4427. doi: 10.1242/dev.153056. [DOI] [PubMed] [Google Scholar]
- 50.Lien J.C., Wang Y.L. Cyclic stretching-induced epithelial cell reorientation is driven by microtubule-modulated transverse extension during the relaxation phase. Sci. Rep. 2021;11:14803–14812. doi: 10.1038/s41598-021-93987-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gérémie L., Ilker E., et al. Descroix S. Evolution of a confluent gut epithelium under on-chip cyclic stretching. Phys. Rev. Res. 2022;4 [Google Scholar]
- 52.Fink J., Carpi N., et al. Piel M. External forces control mitotic spindle positioning. Nat. Cell Biol. 2011;13:771–778. doi: 10.1038/ncb2269. [DOI] [PubMed] [Google Scholar]
- 53.Wu P.H., Aroush D.R.B., et al. Wirtz D. A comparison of methods to assess cell mechanical properties. Nat. Methods. 2018;15:491–498. doi: 10.1038/s41592-018-0015-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Veschgini M., Suzuki R., et al. Tanaka M. Wnt/β-catenin signaling induces axial elasticity patterns of Hydra extracellular matrix. iScience. 2023;26 doi: 10.1016/j.isci.2023.106416. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Endoderm is shown in magenta and ectoderm in green.
Endoderm is shown in magenta and ectoderm in green.