Abstract
Mice which coexpress human papillomavirus type 16 E7 and HLA A2.1 in peripheral squamous epithelium and thymic cortical epithelium are tolerant at the cytotoxic T-lymphocyte (CTL) level to E7 epitopes restricted through HLA A*0201 and H-2b (T. Doan, M. Chambers, M. Street, G. J. Fernando, K. Herd, P. Lambert, and R. Tindle, Virology 244:352–364, 1998). Here we used bone marrow-reconstituted radiation chimeras to distinguish whether E7-directed CTL tolerance was mediated peripherally by E7 expression in skin or centrally by E7 expression in thymus. In chimeric mice expressing E7 in skin and reconstituted with E7-naïve bone marrow and E7-naïve thymus, CTL responses to vaccine-administered E7 epitopes were not restored, i.e., the mice remained tolerant. In contrast, chimeric mice not expressing E7 in skin and reconstituted with E7-naïve bone marrow and E7-expressing thymus had full E7-directed CTL responses. These results demonstrate that E7 protein expression in peripheral squamous epithelium is sufficient to tolerize the E7-directed CTL precursor repertoire. The data have implications for E7-mediated tumorigenesis and for the development of E7-based immunotherapeutic strategies, since peripheral immunological tolerance of tumor-associated antigens may create a barrier to effective immunotherapy.
The E7 oncoprotein of human papillomavirus type 16 (HPV16) is a tumor-specific antigen when expressed in HPV16-associated cervical epithelial tumors, to which immunomanipulative strategies are being directed, both experimentally (see, for example, references 7, 9, and 29) and in E7-based therapeutic vaccine clinical trials (5). We recently reported studies with mice expressing HPV16 E7 protein, driven from the keratin 14 (K14) promoter, in basal epithelium of skin and in the thymic cortex (8). We showed that immunization-induced cytotoxic T-lymphocyte (CTL) responses to each of three CTL epitopes in the E7 protein restricted through two major histocompatibility complex (MHC) class 1 haplotypes were down-regulated in these E7-transgenic mice compared with non-E7 syngeneic control mice. However, in these studies we did not determine whether the down-regulation (i.e., tolerance) was induced centrally by E7 expressed in the thymus or peripherally by E7 expressed in epithelium. In the present study, we distinguish between these two possibilities by specific immunization of bone marrow-reconstituted thymus-transplanted chimeric E7 transgenic mice. We report that chimeric mice expressing the E7 transgene in peripheral epithelium but not in the thymus showed E7-specific down-regulated CTL responses to each of two E7 CTL epitopes restricted through a human and a mouse MHC class I allele, respectively, when compared with sham chimeric but non-E7 control mice. In contrast, chimeric mice expressing the E7 transgene in thymus, but not peripheral epithelium, showed E7-directed CTL responses indistinguishable from those of non-E7 control mice. Thus, we show that the expression of E7 in peripheral squamous epithelium is sufficient to induce and maintain a state of tolerance against E7.
E7-directed bone marrow-derived precursor CTLs (pCTLs) are not tolerized in mice expressing E7 in thymus but not in skin.
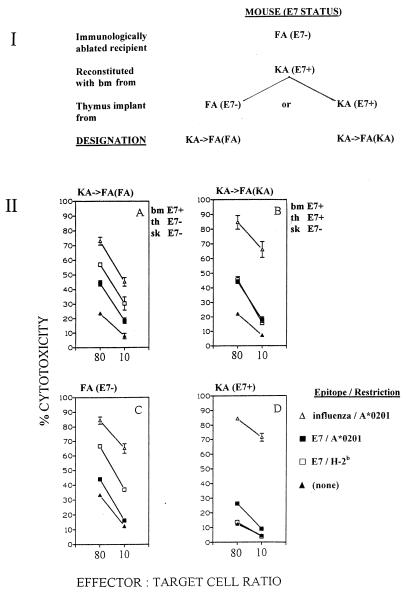
(K14E7 × A2.1Kb)F1 mice (designated KA mice) were derived by crossing male K14.HPV16E7(+/+) mice (16), which express an HPV16 E7 transgene perinatally and throughout life in skin and thymic cortical epithelium, with female HLA A2.1Kb(+/+) mice (30). (FVB × A2.1Kb)F1 mice (designated FA) are syngeneic but do not possess the E7 transgene. KA (E7+) and FA (E7−) mice are on an H-2b background. To inquire whether pCTLs from E7-transgenic mice were tolerized on E7-expressing thymus, we constructed thymus-transplanted radiation chimeras as described elsewhere (8) from immunologically depleted FA (E7−) mice reconstituted with KA (E7+) bone marrow cells. In half of the mice, [designated KA→FA(FA) mice], the bone marrow-derived T-cell precursors were made to mature through a thymus implant from an FA (E7−) donor mouse; in the other half of the mice [designated KA→FA(KA) mice], the bone marrow cells were made to mature through an E7-expressing KA thymus implant (Fig. 1, panel I). KA→FA(FA) mice, KA→FA(KA) mice, and control FA (E7−) and KA (E7+) mice were immunized for CTL response induction with a mix of peptides containing 82LLMGTLGIV90 (an HLA A*0201-restricted E7 CTL epitope [24]), 49RAHYNIVTF57 (an H-2Db-restricted E7 CTL epitope [9]), and 58GILGFVFTL66 (an HLA A*0201-restricted influenza virus matrix CTL epitope [13]). Control mice underwent surgical procedures but without receiving cell and/or organ transplants (sham). KA (E7+) mice showed the previously documented (8) down-regulated CTL response to the E7 epitopes (but not to the irrelevant influenza virus matrix epitope) compared to FA (E7−) mice (Fig. 1, panels IIC and IID). In contrast, KA→FA(KA) mice exhibited E7 (and influenza virus matrix)-directed CTL responses of the same magnitude as those of KA→FA(FA) mice and FA (E7−) mice (Fig. 1, panels IIA to IIC). These data indicate that E7-directed pCTLs from E7 transgenic mice which mature through an E7-expressing thymus, and emerge into a non-E7-expressing peripheral epithelial environment, are not tolerized.
FIG. 1.
(I) Derivation of KA→FA(FA) and KA→FA(KA) chimeric mice from immunologically ablated FA (E7−) mice. (II) CTL responses of splenocytes from chimeric mice and sham control FA (E7−) and KA (E7+) mice (three per group) immunized with a mix of peptides containing E7 CTL epitopes LLMGTLGIV and RAHYNIVTF and influenza virus matrix CTL epitope GILGFVFTL. Immunizations were given in Quil A adjuvant and tetanus toxoid as described elsewhere (8). Spleen cells were restimulated with individual peptides in vitro. Targets were EL4.A2 cells (8) pulsed with individual peptides as indicated. EL4.A2 cells are susceptible to specific CTL lysis through both HLA A*0201 and H-2b restriction elements. CTL assays were conducted as described elsewhere (8). bm, bone marrow; th, thymus; sk, skin.
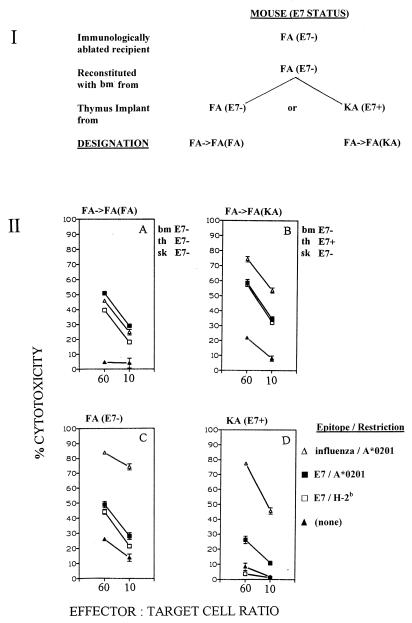
To eliminate the possibility that bone marrow-derived precursors from KA (E7+) mice had somehow previously encountered E7 protein before transfer to recipient mice, thereby influencing their immunological status in the above-described experiment, we asked whether bone marrow-derived pCTLs from FA (E7−) mice would be tolerized during maturation in an E7-expressing thymus. We constructed chimeras from immunologically ablated FA (E7−) mice by reconstitution with FA (E7−) bone marrow cells. In half the mice [designated FA→FA(KA) mice], the bone marrow cells were made to mature through an E7-expressing KA thymus implant. In the other half of the mice [designated FA→FA(FA) mice], the bone marrow cells were made to mature through a non-E7-expressing FA thymus implant (Fig. 2, panel I). FA→FA(KA) mice, FA→FA(FA) mice, and control FA (E7−) and KA (E7+) mice were immunized for CTL induction with a mix of peptides containing the HLA A*0201-restricted and H-2b-restricted E7 CTL epitopes and influenza virus matrix CTL epitope. FA→FA(KA) mice exhibited E7-directed CTL responses of the same magnitude as those of FA→FA(FA) mice and FA (E7−) mice, while KA (E7+) mice exhibited the expected down-regulated E7-directed (but not down-regulated influenza virus matrix-directed) CTL responses (Fig. 2, panel II). These data indicated that E7-naïve bone marrow-derived pCTLs which mature through an E7-expressing thymus and emerge into a non-E7 peripheral epithelial environment are not tolerized to E7.
FIG. 2.
(I) Derivation of FA→FA(FA) and FA→FA(KA) chimeric mice from immunologically ablated FA (E7−) mice. (II) CTL responses of splenocytes from chimeric mice and sham control mice (three per group) immunized with a mix of peptides containing E7 CTL epitopes LLMGTLGIV and RAHYNIVTF and influenza virus matrix CTL epitope GILGFVFTL. Spleen cells were restimulated with individual peptides in vitro. Targets were EL4.A2 cells pulsed with individual peptides as indicated. CTL assays were conducted as described elsewhere (8). bm, bone marrow; th, thymus; sk, skin.
Bone marrow-derived pCTLs are specifically tolerized in mice expressing E7 in skin but not in thymus.
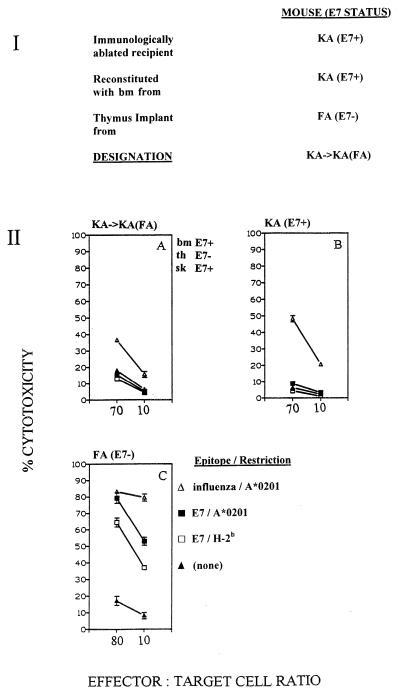
To inquire whether E7-directed pCTLs were tolerized in mice expressing E7 in skin but not in thymus, we constructed chimeric mice in which bone marrow-derived precursors were made to mature through a non-E7-expressing thymus and to emerge into an E7-expressing peripheral epithelial environment. In a first experiment, immunologically ablated KA (E7+) mice were reconstituted with KA (E7+) bone marrow cells which were made to mature through a thymus implanted from an FA (E7−) mouse. The recipient mice, designated KA→KA(FA) (Fig. 3, panel I), and control FA (E7−) and KA (E7+) mice were immunized for CTL response induction with a mix of peptides containing the HLA A*0201-restricted and H-2b-restricted E7 CTL epitopes and the influenza virus matrix CTL epitope. In KA→KA(FA) mice, E7-directed CTL responses to both E7 epitopes were down-regulated to the level seen in control KA (E7+) mice (Fig. 3, panel IIB), while control FA (E7−) mice showed the expected high responses to both E7 CTL epitopes.
FIG. 3.
(I) Derivation of KA→KA(FA) chimeric mice from immunologically ablated KA (E7+) mice. (II) CTL responses of splenocytes from chimeric mice and sham control mice (three per group) immunized with a mix of peptides containing E7 CTL epitopes LLMGTLGIV and RAHYNIVTF and influenza virus matrix CTL epitope GILGFVFTL. Spleen cells were restimulated with individual peptides in vitro. Targets were EL4.A2 cells pulsed with individual peptides as indicated. CTL assays were conducted as described elsewhere (8). bm, bone marrow; th, thymus; sk, skin.
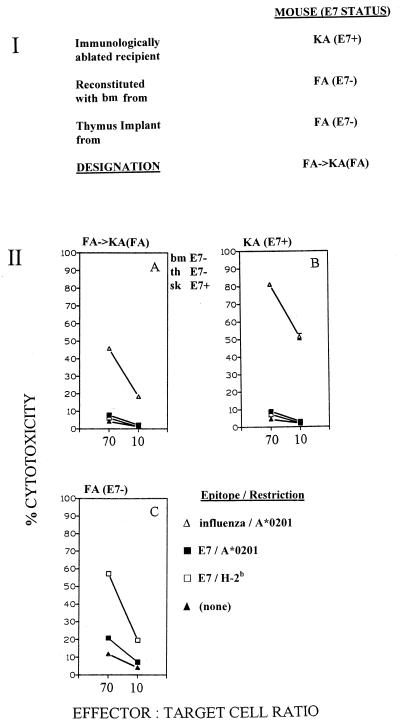
In a second experiment, immunologically ablated KA (E7+) mice were reconstituted with bone marrow from FA (E7−) mice, which was made to mature through a non-E7-expressing FA thymus. These mice, designated FA→KA(FA) mice (Fig. 4, panel I), were immunized for CTL response induction with a mix of peptides containing the HLA A*0201-restricted and the H-2b-restricted E7 CTL epitopes and the influenza virus matrix CTL epitope. As with KA→KA(FA) mice in the previous experiment, E7-directed CTL responses to both E7 epitopes were down-regulated as in KA (E7+) controls and in contrast to FA (E7−) controls, while the influenza virus matrix response confirmed adequate reconstitution.
FIG. 4.
(I) Derivation of FA→KA(FA) chimeric mice from immunologically ablated KA (E7+) mice. (II) CTL responses of splenocytes from chimeric mice and sham control mice (three per group) immunized once with a mix of peptides containing E7 CTL epitopes LLMGTLGIV and RAHYNIVTF and influenza virus matrix CTL epitope GILGFVFTL. Spleen cells were restimulated with individual peptides in vitro. Targets were EL4.A2 cells pulsed with individual peptides as indicated. CTL assays were conducted as described elsewhere (8). bm, bone marrow; th, thymus; sk, skin.
The results from these two experiments indicate that bone marrow-derived E7-directed pCTLs which mature through a non-E7-expressing thymus and emerge into an E7-expressing epithelial environment are specifically tolerized to E7.
We have previously reported pCTL tolerance to epitopes of the HPV16 E7 oncoprotein in KA mice expressing a K14 promoter-driven E7 transgene perinatally and throughout life in the thymus and in basal and/or suprabasal cells of peripheral epithelium (8). In the present experiments, we demonstrate that the E7-directed pCTL repertoire is tolerized in mice expressing E7 in peripheral epithelium in the absence of thymic expression. Conversely, the repertoire is not tolerized in mice expressing E7 in the thymus, in the absence of E7 expression in peripheral epithelium. These data indicate that expression of E7 in peripheral epithelium, and not the thymus, is sufficient to induce and maintain a state of pCTL tolerance to E7. In the thymus, the K14 promoter directs transgene expression to the cortical epithelial compartment (19), which, in other mouse models, has been shown to contribute to the shaping of the T-cell repertoire by positive rather than negative selection (for example, see reference 20). Melero et al. (21) observed no functional down-regulation of the CTL responses induced by immunization with E7 peptide epitope RAHYNIVTF in H-2b mice expressing HPV16 E7 from a K14 promoter and concluded that the mice remain immunologically ignorant of this epitope. This result contrasts with ours. Together, they provide further examples of T-cell tolerance to peripheral antigens in some systems (see, for example, references 1, 2, and 23) and T-cell ignorance in others (see, for example, references 14, 17, and 25). While determinants of immunological outcome of peripheral antigen expression are clearly complex (22), the level of expression (as well as timing and site of expression) can determine whether an antigen induces tolerance or is ignored by naïve T cells. This consideration may explain the difference between the results of Melero et al. and ours. The effect of the level of E7 expression on peripheral tolerance induction is under investigation in our laboratory.
Specific CTL tolerance has implications for E7-mediated tumorigenesis. Nascent E7-expressing tumor cells will escape surveillance where little or no positive priming of cognate pCTLs by endogenous E7 occurs. Additionally, specific CTL tolerance which inhibits the generation of an immunization-induced CTL response will detract from effective immunotherapy (26). We have previously reported that (K14.E7 × C57)F1 mice fail to control a challenge with an E7-expressing tumor following immunization with E7 CTL epitope RAHYNIVTF, whereas in immunized non-E7-transgenic control mice the tumors did not become established (12). Failure to control the tumor was correlated with a lack of an inducible RAHYNIVTF-directed CTL response in E7-transgenic mice, in contrast to non-E7-transgenic control mice, where a powerful CTL response was observed. In further experiments, multiple immunization of KA mice with E7 CTL epitopes or whole E7 protein failed to arrest the development of E7-associated endogenous tumors (8), again being correlated with a lack of E7-directed CTL responses.
The current therapeutic vaccine strategy for HPV16-associated cervical carcinoma targets the E7 tumor-specific antigen by CTL induction (5, 28). The possibility arises that chronic expression of E7 in transformed cervical epithelial cells during the life of the tumor functionally tolerizes E7-directed pCTLs.
Ongoing experiments in our laboratory will distinguish between presentation of E7 to pCTLs directly by keratinocytes and cross presentation of exogenously acquired E7 by bone marrow-derived professional antigen-presenting cells. Presentation of antigen by either of these routes can be tolerogenic (3, 6, 15, 27). Additionally, we will determine whether loss of functional E7-directed CTLs results from pCTL deletion (4, 11) or anergy (10, 25). Peripheral tolerance of tissue-specific antigen depends, at least in some cases, on the generation of regulatory CD4+ cells (see, for example, reference 18). That E7-directed CTL tolerance in KA (E7+) mice reflects an impairment of cognate CD4+ help is unlikely in view of our finding that (K14.E7 × C57)F1 mice immunized with full-length E7 and displaying E7-specific pCTL tolerance showed concomitant enhanced E7-directed CD4+ T-helper responses (12).
The data reported in the present study demonstrate the induction of peripheral tolerance in E7-directed pCTLs by HPV16 E7 expressed in squamous epithelial cells, in the context of human (and mouse) MHC class 1 haplotypes. There are direct implications for the development and progression of cervical cancers which express E7 in transformed squamous epithelium and for the design of E7-based immunotherapeutic strategies for cervical cancer. In the broader context, there are implications for CTL response induction to any foreign or aberrant protein expressed constitutively in squamous epithelial cells as a result of infection, tumorigenesis, or appearance of autoantigen.
Acknowledgments
The work was supported by grants from the Royal Children’s Hospital Foundation, the Queensland Cancer Fund, and the Sir Albert Sakzewski Foundation.
Linda Sherman gave permission to use the HLA A2.1Kb transgenic mice. Donna West and her staff provided animal husbandry. We are indebted to members of the Centre for Immunology and Cancer Research (Brisbane, Australia) and Francis Carbone for helpful discussion.
REFERENCES
- 1.Adler A J, Marsh D W, Yochum G S, Guzzo J L, Nigam A, Nelson W G, Pardoll D M. CD4+ T cell tolerance to parenchymal self-antigens requires presentation by bone marrow-derived antigen-presenting cells. J Exp Med. 1998;187:1555–1564. doi: 10.1084/jem.187.10.1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Antoniou A, McCormick D, Scott D, Yeoman H, Chandler P, Mellor A, Dyson J. T cell tolerance and activation to a transgene-encoded tumor antigen. Eur J Immunol. 1996;26:1094–1102. doi: 10.1002/eji.1830260521. [DOI] [PubMed] [Google Scholar]
- 3.Bal V, McIndoe A, Denton G, Hudson D, Lombardi G, Lamb J, Lechler R. Antigen presentation by keratinocytes induces tolerance in human T cells. Eur J Immunol. 1990;20:1893–1897. doi: 10.1002/eji.1830200904. [DOI] [PubMed] [Google Scholar]
- 4.Bertolino P, Heath W R, Hardy C L, Morahan G, Miller J F. Peripheral deletion of autoreactive CD8+ T cells in transgenic mice expressing H-2Kb in the liver. Eur J Immunol. 1995;25:1932–1942. doi: 10.1002/eji.1830250721. [DOI] [PubMed] [Google Scholar]
- 5.Borysiewicz L K, Fiander A, Nimako M, Man S, Wilkinson G W, Westmoreland D, Evans A S, Adams M, Stacey S N, Boursnell M E, Rutherford E, Hickling J K, Inglis S C. A recombinant vaccinia virus encoding human papillomavirus types 16 and 18, E6 and E7 proteins as immunotherapy for cervical cancer. Lancet. 1996;347:1523–1527. doi: 10.1016/s0140-6736(96)90674-1. [DOI] [PubMed] [Google Scholar]
- 6.Carbone F, Kurts C, Bennett S, Miller J, Heath W. Cross-presentation: a general mechanism for CTL immunity and tolerance. Immunol Today. 1998;19:368–373. doi: 10.1016/s0167-5699(98)01301-2. [DOI] [PubMed] [Google Scholar]
- 7.Chen L, Thomas E K, Hu S-L, Hellstrom I, Hellstrom K E. Human papillomavirus type 16 nucleoprotein E7 is a tumour rejection antigen. Proc Natl Acad Sci USA. 1992;88:110–114. doi: 10.1073/pnas.88.1.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Doan T, Chambers M, Street M, Fernando G J, Herd K, Lambert P, Tindle R. Mice expressing the E7 oncogene of HPV16 in epithelium show central tolerance, and evidence of peripheral anergising tolerance, to E7-encoded cytotoxic T-lymphocyte epitopes. Virology. 1998;244:352–364. doi: 10.1006/viro.1998.9128. [DOI] [PubMed] [Google Scholar]
- 9.Feltkamp M C, Smits H L, Vierboom M P, Minnaar R P, de Jongh B M, Drijfhout J W, ter Schegget J, Melief C J, Kast W M. Vaccination with cytotoxic T lymphocyte epitope-containing peptide protects against a tumor induced by human papillomavirus type 16-transformed cells. Eur J Immunol. 1993;23:2242–2249. doi: 10.1002/eji.1830230929. [DOI] [PubMed] [Google Scholar]
- 10.Ferber I, Schonrich G, Schenkel J, Mellor A L, Hammerling G J, Arnold B. Levels of peripheral T cell tolerance induced by different doses of tolerogen. Science. 1994;263:674–676. doi: 10.1126/science.8303275. [DOI] [PubMed] [Google Scholar]
- 11.Fields L E, Loh D Y. Organ injury associated with extrathymic induction of immune tolerance in doubly transgenic mice. Proc Natl Acad Sci USA. 1992;89:5730–5734. doi: 10.1073/pnas.89.13.5730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Frazer I H, Fernando G J P, Fowler N, Leggatt G R, Lambert P F, Liem A, Malcolm K, Tindle R W. Split tolerance to a viral antigen expressed in thymic epithelium and keratinocytes. Eur J Immunol. 1998;28:2791–2800. doi: 10.1002/(SICI)1521-4141(199809)28:09<2791::AID-IMMU2791>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 13.Gotch F, McMichael A, Smith G, Moss B. Identification of viral molecules recognized by influenza-specific human cytotoxic T lymphocytes. J Exp Med. 1987;165:408–416. doi: 10.1084/jem.165.2.408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Heath W R, Karamalis F, Donoghue J, Miller J F. Autoimmunity caused by ignorant CD8+ T cells is transient and depends on avidity. J Immunol. 1995;155:2339–2349. [PubMed] [Google Scholar]
- 15.Heath W R, Kurts C, Miller J F, Carbone F R. Cross-tolerance: a pathway for inducing tolerance to peripheral tissue antigens. J Exp Med. 1998;187:1549–1553. doi: 10.1084/jem.187.10.1549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Herber R, Liem A, Pitot H, Lambert P F. Squamous epithelial hyperplasia and carcinoma in mice transgenic for the human papillomavirus type 16 E7 oncogene. J Virol. 1996;70:1873–1881. doi: 10.1128/jvi.70.3.1873-1881.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herd K, Fernando G J, Dunn L A, Frazer I H, Lambert P, Tindle R W. E7 oncoprotein of human papillomavirus type 16 expressed constitutively in the epidermis has no effect on E7-specific B- or Th-repertoires or on the immune response induced or sustained after immunization with E7 protein. Virology. 1997;231:155–165. doi: 10.1006/viro.1997.8491. [DOI] [PubMed] [Google Scholar]
- 18.Kurts C, Carbone F R, Barnden M, Blanas E, Allison J, Heath W R, Miller J F. CD4+ T cell help impairs CD8+ T cell deletion induced by cross-presentation of self-antigens and favors autoimmunity. J Exp Med. 1997;186:2057–2062. doi: 10.1084/jem.186.12.2057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Laufer T M, DeKoning J, Markowitz J S, Lo D, Glimcher L H. Unopposed positive selection and autoreactivity in mice expressing class II MHC only on thymic cortex. Nature. 1996;383:81–85. doi: 10.1038/383081a0. [DOI] [PubMed] [Google Scholar]
- 20.Lo D, Reilly C R, Burkly L C, DeKoning J, Laufer T M, Glimcher L H. Thymic stromal cell specialization and the T-cell receptor repertoire. Immunol Res. 1997;16:3–14. doi: 10.1007/BF02786320. [DOI] [PubMed] [Google Scholar]
- 21.Melero I, Singhal M C, McGowan P, Haugen H S, Blake J, Hellstrom K E, Yang G, Clegg C H, Chen L. Immunological ignorance of an E7-encoded cytolytic T-lymphocyte epitope in transgenic mice expressing the E7 and E6 oncogenes of human papillomavirus type 16. J Virol. 1997;71:3998–4004. doi: 10.1128/jvi.71.5.3998-4004.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Miller J F, Flavell R A. T-cell tolerance and autoimmunity in transgenic models of central and peripheral tolerance. Curr Opin Immunol. 1994;6:892–899. doi: 10.1016/0952-7915(94)90010-8. [DOI] [PubMed] [Google Scholar]
- 23.Ohashi P S, Oehen S, Buerki K, Pircher H, Ohashi C T, Odermatt B, Malissen B, Zinkernagel R M, Hengartner H. Ablation of “tolerance” and induction of diabetes by virus infection in viral antigen transgenic mice. Cell. 1991;65:305–317. doi: 10.1016/0092-8674(91)90164-t. [DOI] [PubMed] [Google Scholar]
- 24.Ressing M E, van Driel W J, Celis E, Sette A, Brandt M P, Hartman M, Anholts J D, Schreuder G M, ter Harmsel W B, Fleuren G J, Trimbos B J, Kast W M, Melief C J. Occasional memory cytotoxic T-cell responses of patients with human papillomavirus type 16-positive cervical lesions against a human leukocyte antigen-A *0201-restricted E7-encoded epitope. Cancer Res. 1996;56:582–588. [PubMed] [Google Scholar]
- 25.Schonrich G, Momburg F, Malissen M, Schmitt Verhulst A M, Malissen B, Hammerling G J, Arnold B. Distinct mechanisms of extrathymic T cell tolerance due to differential expression of self antigen. Int Immunol. 1992;4:581–590. doi: 10.1093/intimm/4.5.581. [DOI] [PubMed] [Google Scholar]
- 26.Sotomayor E M, Borrello I, Levitsky H I. Tolerance and cancer: a critical issue in tumor immunology. Crit Rev Oncog. 1996;7:433–456. doi: 10.1615/critrevoncog.v7.i5-6.30. [DOI] [PubMed] [Google Scholar]
- 27.Tan K C, Hosoi J, Grabbe S, Asahina A, Granstein R D. Epidermal cell presentation of tumor-associated antigens for induction of tolerance. J Immunol. 1994;153:760–767. [PubMed] [Google Scholar]
- 28.Tindle R W. Human papillomavirus vaccines for cervical cancer. Curr Opin Immunol. 1996;8:643–650. doi: 10.1016/s0952-7915(96)80080-x. [DOI] [PubMed] [Google Scholar]
- 29.Tindle R W, Croft S, Herd K, Malcolm K, Geczy A F, Stewart T, Fernando G J. A vaccine conjugate of “ISCAR” immunocarrier and peptide epitopes of the E7 cervical cancer-associated protein of human papillomavirus type 16 elicits specific Th1- and Th2-type responses in immunized mice in the absence of oil-based adjuvants. Clin Exp Immunol. 1995;101:265–271. doi: 10.1111/j.1365-2249.1995.tb08349.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vitiello A, Marchesini D, Furze J, Sherman L A, Chesnut R W. Analysis of the HLA-restricted influenza-specific cytotoxic T lymphocyte response in transgenic mice carrying a chimeric human-mouse class I major histocompatibility complex. J Exp Med. 1991;173:1007–1015. doi: 10.1084/jem.173.4.1007. [DOI] [PMC free article] [PubMed] [Google Scholar]