Abstract
Iron is indispensable for the viablility of nearly all living organisms, and it is imperative for cells, tissues, and organisms to acquire this essential metal sufficiently and maintain its metabolic stability for survival. Disruption of iron homeostasis can lead to the development of various diseases. There is a robust connection between iron metabolism and infection, immunity, inflammation, and aging, suggesting that disorders in iron metabolism may contribute to the pathogenesis of arthritis. Numerous studies have focused on the significant role of iron metabolism in the development of arthritis and its potential for targeted drug therapy. Targeting iron metabolism offers a promising approach for individualized treatment of arthritis. Therefore, this review aimed to investigate the mechanisms by which the body maintains iron metabolism and the impacts of iron and iron metabolism disorders on arthritis. Furthermore, this review aimed to identify potential therapeutic targets and active substances related to iron metabolism, which could provide promising research directions in this field.
Keywords: Iron metabolism, Arthritis, Inflammation, Immunity, Rheumatoid arthritis, Psoriatic arthritis, Gouty arthritis
Introduction
Arthritis is a broad term that encompasses most of the pathologies that cause joint symptoms or dysfunction. Joints can be damaged due to degenerative and inflammatory changes or the deposition of physiologically harmful substances. More than 100 different types of arthritis have been identified, with osteoarthritis (OA)—categorized as a non-inflammatory type—being the most common. Other forms, including rheumatoid arthritis (RA), psoriatic arthritis (PsA), ankylosing spondylitis (AS), gouty arthritis (GA), and arthritis associated with inflammatory bowel disease (IBD), are categorized as inflammation-related arthritis. Common clinical features across all types of arthritis include monocyte infiltration, inflammation, synovial edema, proliferation of synovial tissue, joint stiffness, and cartilage degeneration. The traditional classification of primary inflammatory arthritis differs between adults and children. In adults, most cases of inflammatory arthritis fall under RA, which is further subdivided into seropositive and seronegative forms. Other forms of inflammatory arthritis in adults are generally considered seronegative. In contrast, primary inflammatory arthritis in adolescents is typically classified as juvenile idiopathic arthritis (JIA), which is further broken down into several subcategories. To bridge the age gap in the genetic classification of arthritis and to foster collaboration between adult and pediatric rheumatologists, Nigrovic et al[1] have proposed a new framework. This framework classifies inflammatory arthritis into four genetic-based categories: seropositive arthritis, seronegative arthritis, spondyloarthritis (SpA), and systemic arthritis.[1] Seropositive arthritis, often associated with RA, is characterized by the presence of rheumatoid factor and anti-citrullinated peptide antibodies in the patient’s blood. Seronegative arthritis includes RA and JIA cases that lack these antibodies. SpA typically affects the sacroiliac joints and spine, including conditions like AS, PsA, arthritis related to IBD, reactive arthritis (ReA), and JIA with spinal involvement. Systemic arthritis mainly includes systemic JIA and adult-onset Still’s disease.[2]
Iron plays a crucial role in various physiological processes in living organisms. It is involved in systemic and cellular oxygen transport, nucleic acid synthesis, electron transport, and enzyme catalysis. Iron serves as a key cofactor in many functional proteins and enzymes that participate in electron transfer and oxygen metabolism and is typically in the form of heme or iron-sulfur clusters. Additionally, iron is the central cation in the prosthetic moieties of nucleic acid synthesis. These essential functions are evolutionarily conserved from unicellular organisms to human beings.[3]
Iron and iron-related proteins are equally important for individuals with arthritis. A characteristic symptom in these patients is systemic inflammation, which affects iron homeostasis. The disruption of iron homeostasis involves the regulation of hepcidin synthesis, iron transporters, and changes in iron distribution and storage that reduce the availability of iron in the body during inflammatory conditions, subsequently leading to disruptions in iron metabolism. Iron is essential for the development, function, and survival of immune cells such as T cells, B cells, and macrophages. The immune system, which is regulated by iron homeostasis, plays a crucial role in arthritis. Disrupted iron homeostasis also contributes to common complications associated with arthritis. A typical example is inflammation-induced anemia, which is also known as anemia of chronic disease. In addition, hyperferritinemia, a condition characterized by abnormally high levels of ferritin, can occur due to various factors, including inflammation, infection, or malignancy. Arthritis has been identified as the primary cause of hyperferritinemia, followed by infections and iron overload.[4] Consequently, targeted therapies focusing on iron metabolism are expected to play a crucial role in the treatment of arthritis in the future. In this review, we examine the relationship between various types of arthritis, based on a common classification system, and iron metabolism to offer a new perspective.
In brief, the pathophysiologic processes related to iron include various conditions, such as anemia, inflammation, infection, iron overload, and ferroptosis, all of which are also commonly observed in arthritis. These processes are interconnected and mutually influential. For instance, infection can trigger inflammatory reactions that affect the regulation of iron ions. Prolonged inflammation can result in iron deficiency, impacting the production of erythrocytes and leading to anemia. Iron imbalances can also have detrimental effects on the immune system.
Systemic Iron Metabolism
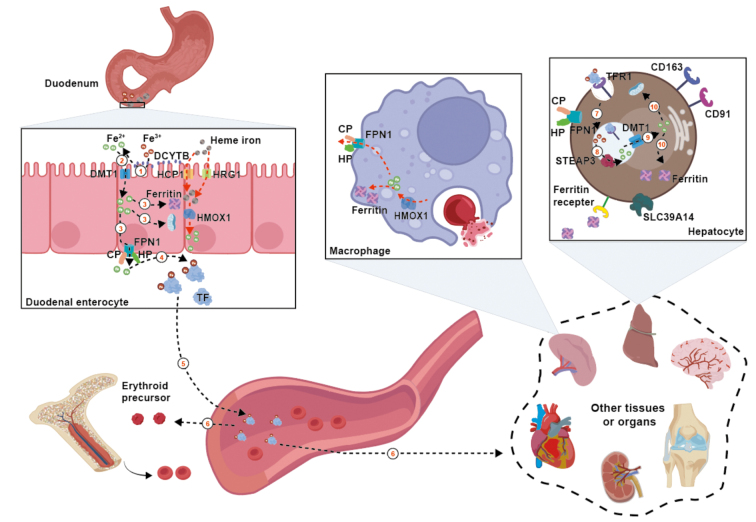
Understanding the metabolic process of iron in organisms is crucial for understanding iron metabolism-related diseases. The human body relies on dietary intake to obtain iron. Iron is absorbed exclusively in the small intestine. There are two forms of dietary iron: mostly trivalent iron and a small amount of heme iron.[5]
Non-heme-bound iron exists as trivalent iron, which remains stable in the body and can not be directly absorbed. In the intestinal lumen, trivalent iron is converted to divalent iron by duodenal cytochrome b (Dcytb). Dcytb is expressed on the apical membrane of enterocytes. Divalent iron is then transported into enterocytes by divalent metal transporter 1 (DMT1). Once absorbed by enterocytes, divalent iron can be used for various cellular metabolic processes, such as entering mitochondria, being stored in ferritin, or being released into the systemic circulation through the basolateral membrane. Divalent iron that is released from the basolateral membrane is transported to the circulation by ferroportin (FPN1), which is the only iron efflux protein. Iron is then oxidized to trivalent iron by hephaestin (HP) or ceruloplasmin (CP). Hepcidin is a peptide hormone that is mainly synthesized by hepatocytes and is the only known ligand of FPN. It binds to and triggers the internalization, ubiquitination, and degradation of FPN, thereby reducing the efflux of iron ions.[6]
Trivalent iron binds to transferrin (TF), the main plasma iron carrier, for further use. TF is a glycoprotein that serves as the primary transport protein for ferric iron, maintaining it in an inert state. The majority of circulating diferric TF is taken up by erythroid precursors in the bone marrow for erythropoiesis. Under normal physiological conditions, cellular iron uptake occurs through the TF cycle. In this cycle, diferric TF binds to transferrin receptor 1 (TFR1) and is internalized into the cell via endocytosis. However, under certain pathological conditions, when the iron binding capacity of TF is surpassed, non-transferrin bound iron (NTBI) is generated. One component of NTBI is labile plasma iron, which is redox-active and unavailable for erythropoiesis. Labile plasma iron can be taken up by non-hematopoietic cells, leading to parenchymal iron deposition and free radical damage. Iron and peroxide generate hydroxyl radicals and destroy polyunsaturated fatty acids through the Fenton reaction. During this destructive process, lipid peroxides are produced, and excessive accumulation of lipid peroxides can cause membrane damage, ultimately leading to ferroptosis. Glutathione peroxidase 4 (GPX4) uses reduced glutathione (GSH) to convert lipid hydroperoxides (LPO) into lipid alcohols, thereby alleviating lipid peroxidation and inhibiting ferroptosis.[7]
Heme (iron-protoporphyrin IX) is an iron-containing tetrapyrrole that is a subunit of hemoglobin and plays a crucial role in oxygen transport. Although the mechanism of non-heme iron uptake has been extensively studied, the mechanisms underlying heme iron absorption from the intestine are still not well understood. Animal studies suggest that enterocytes can take up heme iron from food through heme carrier protein 1 (HCP1) and/or heme responsive gene-1 (HRG1). Once inside the cytoplasm, heme iron can be catabolized by heme oxygenase 1 (HMOX1) to prevent the toxic effects of free heme on cells. Then, it may undergo the same transport as non-heme iron.[8]
The liver plays a crucial role in iron metabolism, and hepatocytes are responsible for the absorption, storage, and transport of iron. Hepatocytes can synthesize a large amount of ferritin, which is the main storage site for iron. After binding to specific receptors, ferritin is absorbed by cells through endocytosis. An acidic environment promotes the release of trivalent iron from TF and its reduction to divalent iron through six-transmembrane epithelial antigen of the prostate 3 (STEAP3). Iron is then transported out of the endosome via DMT1, and TF is exocytosed for reuse. In addition, solute carrier family 39 member 14 (SLC39A14) and DMT1 play roles in the transport of NTBI. To prevent the toxic effects of free heme outside the cell, extracellular free hemes form complexes by binding to hemopexin, which are then taken up by cells through the specific receptor cluster of differentiation (CD)91. Heme in hemoglobin forms a complex with haptoglobin, which binds to the cell surface receptor CD163 and is subsequently internalized via endocytosis. The complex is targeted to lysosomes for degradation or is catabolized by HMOX1, which releases iron into the intracellular iron pool.[8]
Iron recycling is crucial for meeting the daily iron requirement for erythropoiesis and other physiological needs, as the circulating iron pool is relatively small compared to the demand. The spleen plays a major role in the fragmentation and recovery of erythrocytes. Macrophages are responsible for recycling most of the iron from aged or damaged erythrocytes to efficiently reuse the iron present in hemoglobin.
In contrast, iron excretion is attributed to passive pathways that are not regulated by iron levels or other parameters related to iron homeostasis. These pathways include the sloughing of intestinal epithelial cells, the exfoliation of skin cells, and physiological blood loss that occurs during menstruation or minor trauma to epithelial linings.[9] The systemic metabolism of iron is shown in Figure 1.
Figure 1.
The systemic iron metabolism and non-heme dietary iron circulation. (1) Fe3+ is reduced to Fe2+ by DCYTB on the apical membrane of enterocytes. (2) Fe2+ enters the enterocytes via DMT1. (3) Fe2+ absorbed by enterocytes can enter mitochondria, be stored in ferritin, or be released into the systemic circulation via FPN1 on the membrane. (4, 5) The released Fe2+ is first oxidized to Fe3+ by CP or HP, and then combined with TF to enter the bloodstream for circulation. (6) Most of the circulating TF is absorbed by erythroid precursors in the bone marrow for erythrocyte production, and the rest is distributed to other tissues or organs that require iron. (7) Diferric TF binds to TFR1 and is internalized into the cell (using hepatocytes as an example) via endocytosis. (8) Within the endosome, the acidic environment causes TF to release Fe3+, which is then reduced to Fe2+ by STEAP3. (9) Fe2+ is released into the cytoplasm through DMT1 from the endosome. (10) Fe2+ iron in the cytoplasm can enter mitochondria for heme synthesis or be stored in ferritin. Enterocytes can uptake heme iron via HCP1 or HRG1. The heme iron entering the cytoplasm can be catabolized by HMOX1 to prevent toxicity. Then it may undergo the same transportation as Fe3+. The spleen is the main site for the recycling and reuse of iron in erythrocytes. Macrophages are responsible for recovering most of the iron from aged or damaged erythrocytes, effectively reusing the iron present in hemoglobin. Fe2+ catabolized by HMOX1 can be stored in ferritin or transported out of cells by FPN1 for further use. CD: Cluster of differentiation; CP: Ceruloplasmin; DCYTB: Divalent iron by duodenal cytochrome b; DMT1: Divalent metal transporter 1; FPN1: Ferroportin; HCP1: Heme carrier protein 1; HMOX1: Heme oxygenase 1; HP: Hephaestin; HRG1: Heme responsive gene-1; SLC39A14: Solute carrier family 39 member 14; STEAP3: Six-transmembrane epithelial antigen of the prostate 3; TF: Transferrin; TFR1: Transferrin receptor 1.
Iron Metabolism and Arthritis
Iron metabolism and RA
RA is a chronic inflammatory disease that affects the synovial tissue in joints. It is characterized by synovial hyperplasia, pannus formation, and destruction of cartilage and bone, leading to symptoms such as joint pain, stiffness, and swelling. The exact cause of RA is unknown, but it is accompanied by various clinical complications that affect the bone, vascular system, metabolism, and cognition. The pathogenesis of RA is complex and involves many cell types, including T cells, B cells, macrophages, chondrocytes, and fibroblasts. Synovial fibroblast-like synoviocytes (FLSs) and macrophages in synovial tissue play important roles in maintaining joint health and causing inflammation.[10] Current treatment strategies for RA focus on regulating immune responses and inhibiting the abnormal proliferation of FLSs. However, due to the increasing incidence of RA and the limitations of existing therapies, there is growing interest in exploring new treatment approaches. One emerging area is targeted iron metabolism therapy, which is a potential approach to alleviate RA symptoms.
Iron availability is low in patients with chronic inflammatory diseases such as RA. This is evidenced by the low level of TF saturation.[11] A meta-analysis was conducted to further support this finding and showed that circulating blood iron levels were decreased and negatively correlated with the Disease Activity Score in 28 Joints (DAS28)-C-reactive protein (CRP) and inflammatory markers in RA patients.[12] Low iron use may indicate abnormalities in iron metabolism-related proteins in RA. Several proteins associated with iron metabolism, including FPN, hepcidin, CD163, and HCP-1, have been identified in the synovium of RA patients.[11] Additionally, consistent findings for iron biomarkers have shown that genetically high iron status is positively associated with gout and inversely associated with RA.[13] These findings suggest that factors involved in iron metabolism could be targets for treating RA. For instance, transferrin receptor 22 (TFR2) in macrophages has been shown to alleviate the progression of arthritis by inhibiting M1-like polarization.[14] Although these studies suggest a link between iron metabolism and RA, there is a lack of in-depth research on its pathogenesis.
Abnormal iron metabolism in RA is associated with inflammation. And inflammation and iron metabolism disorders impact the severity of RA. There is growing interest in regulating iron metabolism to reduce inflammation. One promising approach is the use of nanoparticles, which exhibit zinc and possess iron-chelating properties. In an inflammation model induced by Freund’s adjuvant, these nanoparticles could reduce paw edema, thermal hyperalgesia, serum interleukin-1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α) levels, and liver hepcidin expression.[15]
People with arthritis often suffer from anemia, which is more common in individuals with RA. There are two main types of anemia associated with RA: anemia of chronic diseases (ACD) and iron deficiency anemia (IDA). ACD, which is also known as inflammatory anemia, can be distinguished from IDA in RA through bone marrow iron staining and measuring hepcidin or prohepcidin levels in serum. It is often caused by iron deficiency resulting from gastrointestinal bleeding associated with the use of non-steroidal anti-inflammatory drugs (NSAIDs). The exact mechanism underlying anemia in RA is not fully understood, but it is believed to be influenced by inflammatory cytokines, impaired erythropoiesis, and iron deprivation in erythroblasts. In addition, the level of ferritin is closely associated with hemoglobin levels in patients with RA. Inflammation has the potential to raise ferritin levels and stimulate hepcidin production, subsequently limiting the availability of iron necessary for the production of red blood cells, ultimately resulting in anemia. A study revealed a correlation between the levels of ferritin and other inflammation-related indicators, such as CRP and the erythrocyte sedimentation rate (ESR).[16] The complex mechanism of anemia in RA also involves the key factors hepcidin and interleukin-6 (IL-6). Hepcidin production is suppressed by factors associated with increased erythropoiesis and iron deficiency, and IL-6 plays a role in rheumatoid anemia.[17] Therefore, regulating iron metabolism and inflammation is a promising approach for treating anemia in RA patients. Treatment with tocilizumab, which reduces systemic inflammation, has been shown to improve inflammatory anemia in RA patients. This improvement is associated with decreases in hepcidin and haptoglobin levels, as well as an increase in iron-binding capacity.[18] And, this treatment is believed to protect against ACD in various diseases, including RA.[19]
Ferroptosis is a newly discovered form of programmed cell death characterized by uncontrolled iron-dependent lipid peroxidation.[7] Based on the systemic metabolic pathway of iron, divalent iron is stored in cells as the labile iron pool (LIP), where it is bound to low molecular weight compounds such as GSH. Cystine/glutamate exchangers such as solute carrier family 3 member 2 (SLC3A2) and solute carrier family 7 member 11 (SLC7A11) mediate the transport of cystine to cells for the exchange of glutamate. GSH depletion not only inactivates GPX4 but also releases divalent iron for Fenton chemistry, leading to the propagation of lipid peroxides and ultimately triggering ferroptosis. Iron overload and ferroptosis have also been widely observed in RA.
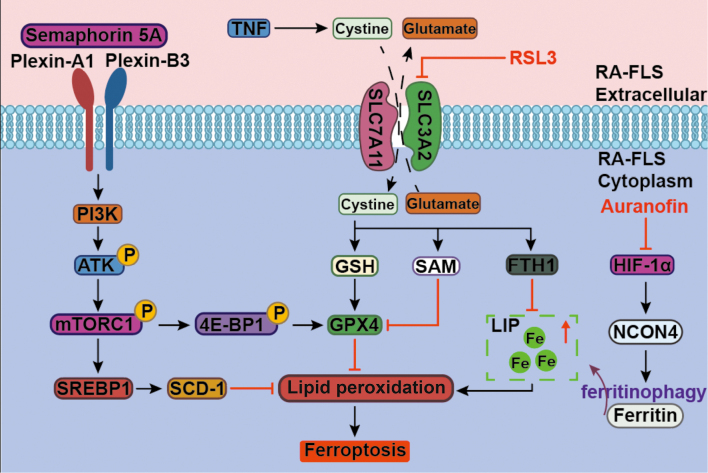
The pathogenesis of RA has been shown to be associated with the hub genes PTGS2, ENO1, and GRN, which are strongly associated with ferroptosis.[20] The abnormal proliferation of synovial fibroblasts in RA may be attributed to reduced ferroptosis sensitivity in FLSs. One mechanism involves S-adenosylmethionine (SAM), which decreases the expression of GPX4 and ferritin heavy polypeptide 1 (FTH1) in RA-FLSs by promoting and mediating GPX4 promoter methylation and reducing FTH1 expression.[21] Another mechanism involves the secretion of semaphorin 5A by synovial macrophages, which binds to Plexin-A1 and Plexin-B3 on synovial fibroblasts, activating the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT)/mammalian target of rapamycin (mTOR) signaling pathway. The activation of this process not only increases the phosphorylation of eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1), which leads to increased protein synthesis of GPX4, but also activates downstream sterol-regulatory element binding protein 1 (SREBP1)/stearoyl-coenzyme A desaturase-1 (SCD-1) signaling to suppresses lipid peroxidation, reactive oxygen species (ROS) production, and ferroptosis.[22] It is important to consider the role of inflammation, and TNF signaling can protect fibroblasts from ferroptosis by promoting cysteine uptake and GSH biosynthesis. Combining a low-dose ferroptosis inducer with a TNF antagonist can induce fibroblast ferroptosis and attenuate arthritis progression.[23] Inhibiting inflammation via the Janus kinase/signal transduction and transcription activation (JAK/STAT) signaling pathway and inducing ferroptosis in MH7A cells by inhibiting GPX4 expression can also alleviate arthritis. Additionally, wasp venom, which is a secretion produced by insect, has been shown to alleviate RA by inhibiting inflammation and inducing fibroblast ferroptosis.[24] Low-dose auranofin can promote ferroptosis in RA-FLSs by regulating the HIF-1α/ nuclear receptor coactivator 4 (NCOA4) pathway, thereby inhibiting cell proliferation and alleviating RA.[25] The mechanism by which the abnormal proliferation of fibroblasts exacerbates RA progression by decreasing the sensitivity of RA-FLSs to ferroptosis is described in Figure 2. Changes in ferroptosis in RA-FLSs can not only lead to the abnormal proliferation of synovial fibroblasts but also exacerbate the progression of arthritis by enhancing synovial angiogenesis. Ferroptosis can enhance the expression of the endosomal sorting complex required for transport in RA-FLSs, promoting the release of small extracellular vesicles from affected fibroblasts. These extracellular vesicles can then promote synovial angiogenesis in human dermal microvascular endothelial cells by upregulating Forkhead Box O1(FOXO1) and downstream vascular endothelial growth factor (VEGF), thus exacerbating arthritis progression.[26]
Figure 2.
The molecular mechanism of reduced sensitivity to ferroptosis in RA-FLS. Ferroptosis is mainly caused by iron-dependent lipid peroxidation. GPX4 is a crucial factor that inhibits ferroptosis. Decreasing the accumulation of LIP, activating of GPX4 or related signaling pathways can suppress lipid peroxidation production and prevent ferroptosis. RSL3 is a known ferroptosis inducer. The data cut-off date for the analysis was February 1, 2024. 4E-BP1: 4E binding protein 1; AKT: Protein kinase B; FLS: Fibroblast-like synoviocyte; FTH1: Ferritin heavy polypeptide 1; GPX4: Glutathione peroxidase 4; GSH: Glutathione; HIF-1α: Hypoxia-inducible factor-1 alpha; LIP: Labile iron pool; mTOR: Mammalian target of rapamycin; NCOA4: Nuclear receptor coactivator 4; PI3K: Phosphatidylinositol 3 kinase; RA: Rheumatoid arthritis; SAM: S-adenosylmethionine; SCD-1: Stearoyl-CoA desaturase 1; SLC3A2: Solute carrier family 3 member 2; SLC7A11: Solute carrier family 7 member 11; SREBP1: Sterol-regulatory element binding protein 1; TNF: Tumor necrosis factor.
On the other hand, an increase in chondrocyte ferroptosis accelerates the progression of RA. According to a previous study, the activation of transient receptor potential melastatin-subfamily member 7 (TRPM7) calcium ion channels via the protein kinase C α (PKCα)–NOX4 axis leads to the accumulation of intracellular calcium ions and ROS. This triggers oxidative stress, ultimately resulting in ferroptosis in chondrocytes, which contributes to the development of RA.[27] The antioxidant spermidine can inhibit ferroptosis in chondrocytes and alleviate RA.[28] Macrophage ferroptosis induced by iron overload exacerbates the development of RA, and M1 macrophages are sensitive to ferroptosis. M1 macrophages have higher iron levels and lower levels of GSH than other cells, which increases their susceptibility to ferroptosis. Therefore, modulating macrophage sensitivity to ferroptosis may be helpful in treating RA.[29] Additionally, T cells and plasma cells may also regulate RA synovitis through ferroptosis-related genes. A separate study demonstrated the presence of two ferroptosis clusters in the RA synovium that exhibit distinct immune profiles and ferroptosis sensitivity.[30] The development of appropriate clinical treatment plans for different groups of ferroptotic cells is beneficial for treating RA.
Currently, the main drugs used for the clinical treatment of RA are NSAIDs, glucocorticoids, and disease-modifying antirheumatic drugs (DMARDs).[31] Several DMARDs such as tocilizumab, methotrexate, leflunomide, infliximab, etanercept, tofacitinib, ruxolitinib, and sulfasalazine are commonly used in clinical practice for treating rheumatoid diseases. The mechanisms of action of these drugs are associated with iron metabolism.[14,23,32,33,34,35] Additionally, auranofin, an organic gold compound approved by the U.S. Food and Drug Administration (FDA) for RA treatment, has also been reported to affect iron metabolism.[25] More details can be found in Supplementary Table 1, http://links.lww.com/CM9/C37.
Iron metabolism and SpA
SpA is a group of related but phenotypically distinct disorders that can affect the sacroiliac joints, spine (axial SpA), and/or peripheral joints (peripheral SpA). PsA, arthritis related to IBD, ReA, JIA, and AS are the most common clinical subtypes.[2] Compared to axial SpA, which has a clear radiological basis, peripheral SpA is more ambiguous and often used interchangeably with some subsets, such as PsA, ReA, and undifferentiated SpA. Currently, medical history (e.g., family history, back pain characteristics, response to NSAIDs, history of enthesitis/arthritis/dactylitis or concomitant extra-musculoskeletal manifestations [e.g., acute anterior uveitis, psoriasis, or IBD]), physical examinations (e.g., joint swelling, tenderness, restricted mobility), laboratory tests (e.g., CRP, ESR, and human leukocyte antigen [HLA]-B27), and imaging examinations (e.g., magnetic resonance imaging [MRI] and X-ray) are used to comprehensively determine the presence of SpA and classify SpA.[2,36] Inflammation and immune dysfunction are common in different subtypes of spinal arthritis. Considering the strong correlation among inflammation, immunity, and iron metabolism, iron metabolism may play an important role in SpA.
Although no causal genetic relationship has been established between AS and iron homeostasis, it is important to acknowledge that there may be a correlation between these factors at non-genetic levels.[37] The genes encoding several enzymes related to microbial iron chelation are enriched in AS patients.[38] Changes in the inflammatory response in the immune microenvironment, which are mediated by DNA damage inducible transcript 3 (DDIT3) and heat shock protein family B (small) member 1 (HSPB1), may also induce ferroptosis in AS cells.[39] Understanding the role of ferroptosis in AS is crucial because it can help categorize AS into subtypes based on ferroptosis-related gene expression. Tailoring treatment strategies according to the ferroptosis status of AS patients holds promise as a direction for future research.[40]
In addition to abnormal iron metabolism in AS, abnormal iron metabolism is also observed in patients with other types of peripheral arthritis. Histone-induced inflammation is one of the causes of JIA. Free iron activates neutrophils to generate neutrophil extracellular traps and release histones. Histones may further promote the release of neutral extracellular traps, thereby exacerbating arthritis progression.[41] C-type lectin domain family 2 member B (CLEC2B) may participate in the progression of PsA by regulating ferroptosis and the killing function of natural killer cells.[42] Directly or indirectly regulating the iron metabolism in SpA will be beneficial for disease treatment. The presence of acrylamide in food has been associated with an increased risk of AS because it inhibits cell proliferation, induces autophagy-dependent ferroptosis, and activates AMP-activated protein kinase (AMPK)- UNC51-like kinase-1 (ULK1)-mTOR signaling in human chondrocytes.[43] Children with JIA who take dietary iron supplements have more stable ferritin levels, which is beneficial for reducing inflammation.[44]
Currently, in addition to physical therapy, NSAIDs are the preferred drugs for the clinical treatment of SpA. As a second-line treatment, TNF inhibitors and IL-17 inhibitors are available. TNF-α and IL-23/IL-17 are the major proinflammatory cytotoxic pathways in axial SpA. Iron homeostasis is regulated to varying degrees by TNF-α. For example, TNF-α can increase iron uptake in astrocytes and microglia, but the pathways are different.[45] This suggests that TNF-α may affect iron metabolism in key cell subpopulations in this disease. The key role of TNF signaling has been described previously.[23] Treatment with anti-TNF drugs can decrease TF, iron, and ferritin levels in the serum of axial SpA patients.[46] Unfortunately, the key pathogenic cell subpopulations involved in axial SpA remain poorly understood. Currently, it is believed that abnormal activation and dysfunction of T cells, B cells, natural killer cells, and macrophages (especially M2 macrophages) may be important factors in the pathogenesis of axial SpA.[36] In addition to the immune cell population, another studies showed that targeted chondrocyte therapy for functional impairments could alleviate axial SpA-like bone disease in mice.[47] The complexity of pathogenic cell subpopulations in axial SpA and the numerous interactions among them have become key factors impeding progress of axial SpA research. The role of iron metabolism in specific cell subpopulations in axial SpA has received widespread attention in recent years and is a promising direction for exploring the pathogenesis of axial SpA.
IL-17 is a cytokine primarily produced by T helper 17 (Th17) cells stimulated by the proinflammatory cytokine IL-23. In addition, abnormal invariant natural killer T cells, γδ T cells, and mucosal-associated invariant T cells in axial SpA can also produce IL-17. IL-17 has been widely reported to participate in the pathological process of atherosclerosis by promoting the inflammatory response and inducing inflammatory cell infiltration. Strategies targeting IL-17 have become important for treating axial SpA. By inhibiting the activity of IL-17, inflammation can be alleviated, and symptoms in axial SpA patients can be improved.[36] IL-17 can also regulate bone formation through iron metabolism. A recent study revealed that IL-17 induced ferritin expression in osteoblasts, which further enhanced osteogenic differentiation through autophagy activation.[48] Although IL-17 is associated with the occurrence of axial SpA and iron metabolism homeostasis, research on whether IL-17 participates in the pathogenesis of axial SpA through iron metabolism pathways is still very limited and is an interesting research direction. Inflammatory factors such as IL-1, IL-6, IL-22, and TGF-β also affect the pathological process of axial SpA. Similarly, these inflammatory factors also regulate different diseases through iron metabolism. Linking inflammation with axial SpA through iron metabolism will be a promising research direction.
Overall, despite the new perspective of iron metabolism in the pathogenesis and treatment of SpA, research on the interaction among inflammation, immunity, and iron metabolism in SpA is still limited. Further studies are needed to elucidate their roles in the progression of SpA.
Iron metabolism and OA
Iron overload commonly occurs in patients with OA. Synovial fluid and serum iron concentrations were significantly increased in patients with OA compared to healthy subjects.[49,50] Conversely, the serum total iron binding capacity was decreased in OA patients. A positive correlation was observed between serum iron levels and the risk of OA in women.[51] Additionally, elevated serum ferritin levels were associated with the incidence and severity of symptomatic knee OA in men.[52] Direct intake of iron or iron metabolism-related proteins could partially alleviate the progression of OA. Furthermore, systemic iron levels may contribute to knee OA pathogenesis, with a short-term deficit in dietary iron potentially reducing the severity of knee cartilage lesions.[53]
Inflammation and iron overload in articular cartilage are key factors in the development of OA. Animal experiments have shown that systemic iron overload leads to the accumulation of iron in the knee joint. This accumulation is associated with an increase in the expression of local inflammatory mediators and the early onset and progression of knee joint OA.[54] Iron overload is common in osteoarthritic joints (especially articular cartilage). Currently, there are no clinically available drugs for the treatment of OA that can eliminate iron overload in osteoarthritic joints. Some of the latest research has made some progress in this area. Treatment with deferoxamine (DFO), which is a systemic iron reduction agent, has been shown to decrease the development of cartilage lesions associated with OA in the knee joints.[55] Additionally, treatment with ferric ammonium citrate has been shown to increase ferritin expression and significantly decrease the expression of hepcidin, FPN1, TFR1, and TFR2. This compromised the functional integrity of chondrocytes, leading to oxidative stress and apoptosis.[56] Articular cartilage is a connective tissue composed of cartilage cells and surrounding extracellular matrix (ECM). As the only cellular component, chondrocytes participate in the synthesis of ECM, maintain matrix circulation, and maintain the functional and structural integrity of cartilage. Therefore, the disruption of homeostasis of chondrocyte iron metabolism is crucial for the occurrence and development of OA. Inflammatory and fibrocartilage chondrocytes are important subpopulations in hand OA, and recent research suggests that ferroptosis may be a significant process involved in this condition. This finding provides new insights into the underlying mechanisms and potential therapeutic targets for hand OA.[57] Proinflammatory cytokines disrupt the iron balance in chondrocytes by increasing the expression of the iron influx transporter TFR1 and decreasing the expression of the iron efflux transporter FPN. This disruption leads to the accumulation of iron in chondrocytes, which in turn promotes the expression of the catabolic markers matrix metallopeptidase 3 (MMP3) and MMP13. Furthermore, oxidative stress and mitochondrial dysfunction play crucial roles in the degeneration of cartilage caused by iron overload. Studies have shown that reducing iron concentrations with iron chelators or antioxidant drugs can inhibit the expression of these catabolic markers and prevent mitochondrial dysfunction associated with iron overload.[58] Another study demonstrated that sterol carrier protein 2 (SCP2) facilitated the transport of cytoplasmic LPO to mitochondria, contributing to the spread of intracellular LPO and accelerating chondrocyte ferroptosis.[59]
Bioinformatics analysis and machine learning techniques identified several ferroptosis-related genes involved in OA. The genes ATF3, IL6, CDKN1A, IL1B, EGR1, CD44, AQP8, BRD7, IFNA4, and ARHGEF26-AS1 have been identified as potential target genes for reducing iron overload during OA treatment.[60,61] Additionally, transferrin receptor (TFRC) has been implicated in ferroptosis in IL-1β-stimulated chondrocytes, and ferroptosis inhibitors have been shown to inhibit ROS in these chondrocytes.[62] Furthermore, certain long non-coding RNAs have been identified as biomarkers of OA based on their association with ferroptosis-related genes.[63] These findings can serve as early diagnostic and therapeutic targets for OA.
In this review, we summarized the emerging evidence regarding the impact of ferroptosis on various pathological processes involved in the progression of OA. Some key ferroptosis-related targets, such as DMT1, transient receptor potential vanilloid 1 (TRPV1), staphylococcal nuclease domain containing 1 (SND1), Piezo1, SLC3A2, GPX4, NCOA4, excitatory amino acid transporter 1 (EAAT1), SCP2, and FOXO3, are potential new drug research targets for treating OA.[54,59,64,65,66,67,68,69,70,71] Recent research has shown that various natural medicinal factors (such as stigmasterol, D-mannose, naringenin, astaxanthin, metformin, theaflavin-3,3′-digallate, biochanin A, sarsasapogenin, kukoamine A, cardamonin, calcipotriol, brevilin A, and baicalein) have therapeutic benefits in combating the occurrence of OA by inhibiting the ferroptosis pathway and have multiple targets and pathways, providing new ideas and methods for the treatment of OA.[72,73,74,75,76,77,78,79,80,81,82,83,84,85] Some chemical reagents, such as DFO and the calcium chelator BAPTA-AM, are potential therapeutic drug components.[86,87] Similarly, microbiota and its metabolite capsiate are also potential therapeutic drug active substances.[49] With an increased understanding of the role of microRNA (miRNAs) in OA, miRNAs are expected to be potential targets for treating OA.[88,89] We have compiled research conducted within the past five years on the mechanism and treatment of iron overload and ferroptosis in OA chondrocytes, and the results are presented in Supplementary Table 2, http://links.lww.com/CM9/C37. However, most of the related research is still in the basic experimental stage, and it is not possible to clearly explain the clinical efficacy of iron chelators, ferroptosis inhibitors, antioxidants, calcium chelators, stigmasterol, D-mannose, or other drugs in patients. Further research is needed to provide more ideas and treatment strategies and develop targeted treatment plans. The clinical drugs currently used for the treatment of knee OA include NSAIDs, glucocorticoids, opioids, symptomatic drugs, chondroprotective agents, and anticytokines.[90] Some clinical drugs or preclinical agents, such as metformin, astaxanthin, and ethinyl estradiol, may also treat OA by regulating iron metabolism, as summarized in Supplementary Table 1, http://links.lww.com/CM9/C37.[60,75,76] Additionally, we described the mechanism of chondrocyte ferroptosis in OA in Figure 3. By examining the relationship between ferroptosis and OA pathology, our aim was to provide valuable insights into potential therapeutic approaches for treating OA.
Figure 3.
The molecular mechanism of ferroptosis exacerbating OA and potential therapeutic targets. Ferroptosis of articular chondrocytes is associated with the pathogenesis of OA. The pathways and networks through which ferroptosis regulates the progression of OA can be divided into four basic categories, specifically amino acid metabolism (blue background section), lipid metabolism (green background section), iron metabolism (red background section), and others (yellow background section). The red font indicates potential therapeutic drugs or reagents. The data cut-off date for the analysis was February 1, 2024. AMPK: Adenosine 5′-monophosphate-activated protein kinase; CX43: Connexin 43; DFO: Deferoxamine; DMT1: Divalent metal transporter 1; FOXO3: Forkhead box O3; FPN1: Ferroportin; GPX4: Glutathione peroxidase 4; GSH: Glutathione; HIF-1α: Hypoxia-inducible factor-1 alpha; HIF-2α: Hypoxia-inducible factor-2 alpha; HMOX1: Heme oxygenase 1; HSPA5: Heat shock protein family a member 5; JNK: C-Jun N-terminal kinase; LIP: Labile iron pool; MAPK: Mitogen-activated protein kinase; NCOA4: Nuclear receptor coactivator 4; NF-κB: Nuclear factor kappa B; NRF2: Nuclear factor erythroid 2-related factor 2; OA: Osteoarthritis; SCP2: Sterol carrier protein 2; SIRT1: Silent mating type information regulation 2 homolog-1; SLC2A1: Solute carrier family 2 member 1; SLC3A2: Solute carrier family 3 member 2; SLC7A11: Solute carrier family 7 member 11; SND1: Staphylococcal nuclease domain containing 1; SREBF2: Sterol regulatory element binding transcription factor 2; STEAP3: Six-transmembrane epithelial antigen of the prostate 3; TF: Transferrin; TF3: Theaflavin-3,3′-digallate; TFR1: Transferrin receptor 1; TGF-β1: Transforming growth factor-beta 1; TRPV1: Transient receptor potential vanilloid 1; YAP1: Yes-associated protein 1.
Iron metabolism and GA
GA is a type of joint inflammation caused by the deposition of monosodium urate (MSU) crystals in joints and extra-articular structures in patients with hyperuricemia. Uric acid is a chemical produced through metabolism. When there is too much uric acid in the blood, uric acid crystals form and deposit in the joints and surrounding tissues, causing joint inflammation and pain.[91] Iron metabolism abnormalities often occur in GA. Elevated levels of ferritin are associated with gout and the frequency of attacks.[92] Patients with gout have smaller global and regional brain volumes and higher brain iron levels.[93] Elevated serum uric acid, which is involved in iron metabolism, has been increasingly recognized as a risk factor for gout.[94] Iron supplementation exacerbates joint inflammation in mice with acute GA.[95] Targeting iron metabolism to treat GA is also a promising approach. MSU crystals and other particulate matter can induce ferroptosis in various cells, but ferroptosis inhibitors cannot directly prevent MSU crystal-induced necrosis in mouse macrophages.[96] In addition, polydatin ameliorates sodium urate-induced inflammation and oxidative stress by modulating PPAR-γ and ferritin activation in GA model mice.[97]
Future Directions and Challenges
In addition to the relationship between arthritis and iron metabolism mentioned above, there are changes in the iron metabolism of the microbial community in arthritis patients. Functionally, the redox environment, transport, and metabolism of iron are altered in the microbiota of individuals with RA.[98] Arthritis induced by ferroptosis inducers is associated with an abnormal gut microbiota.[99] Although RA is associated with ferroptosis, which is driven by lipid peroxidation, the direct use of antioxidants appears to have limited therapeutic effects on RA. Hence, treating arthritis by targeting iron metabolism still requires further investigation. Conducting research on iron metabolism to develop reliable biomarkers would not only aid in patient stratification and the monitoring of treatment response but also facilitate evaluating the efficacy of targeted treatment of ferroptosis in clinical trials. But, further groundbreaking work is necessitated. Elderly patients with arthritis are at an increased risk of developing IDA and other gastrointestinal injuries when they take NSAIDs such as celecoxib, ibuprofen, and naproxen. These findings indicate that NSAIDs treatment may interfere with the iron metabolism balance in elderly patients with joint disease. However, more research is needed to determine the extent of this effect and whether regulating iron metabolism can improve this type of anemia and help treat arthritis.
The study of ferroptosis in arthritis is a dynamic and active research field with extensive research and application prospects. However, there are still several unresolved issues in targeted ferroptosis treatment for arthritis. For instance, TNF-α exacerbates RA by protecting fibroblasts from ferroptosis while simultaneously increasing inflammation. Concurrently, TNF-α has been extensively documented to promote ferroptosis by amplifying oxidative stress and bolstering iron accumulation. Currently, there is limited understanding of the specificity and effectiveness of drugs targeting ferroptosis in RA, as well as the optimal dose and timing. Further research is required to optimize the specificity and efficacy of these drugs, and there is still a lack of supporting clinical data. The current understanding of ferroptosis and arthritis remains relatively superficial, posing a challenge for achieving precision medicine based on this approach.
Conclusion and Perspectives
Maintaining iron homeostasis is crucial in the field of arthritis. However, there is a lack of thorough research on the impact of iron metabolism on arthritis, including the effects of iron chelators, antioxidants, and ferroptosis inhibitors on arthritis treatment. Additional research is needed regarding the pathogenic mechanism of iron metabolism disorders in arthritis. Therefore, focusing on balancing iron levels in the body can provide new insights into the development of arthritis and innovative therapeutic approaches. There are currently no clinical drugs that specifically target iron metabolism to treat arthritis. Targeting iron metabolism has opened up new possibilities for treating arthritis. By regulating iron metabolism, we can effectively suppress inflammatory reactions, alleviate pathological damage, and improve patient quality of life. Precision medicine targeting iron metabolism can also be personalized based on individual differences, reducing drug side effects and improving effectiveness. Iron metabolism and its regulatory networks hold potential as therapeutic targets. The study of iron metabolism in the treatment of arthritis has significant clinical applications and is a rapidly evolving research field. It is expected to witness the emergence of appropriate approaches targeting iron metabolism for arthritis therapy and prevention in the foreseeable future.
Funding
The study was supported by grants from the National Natural Science Foundation of China (Nos. 82030003 and 82001726), the CAMS Innovation Fund for Medical Sciences (No. 2019-I2M-5-066), and the Shanghai Municipal Science and Technology Major Project (No. 2023SHZDZX02).
Conflicts of interest
None.
Supplementary Material
Footnotes
Dachun Zhuo and Wenze Xiao contributed equally to this work.
How to cite this article: Zhuo DC, Xiao WZ, Tang YL, Jiang S, Geng CC, Xie JN, Ma XB, Zhang Q, Tang KH, Yu YX, Bai L, Zou HJ, Liu J, Wang JC. Iron metabolism and arthritis: Exploring connections and therapeutic avenues. Chin Med J 2024;137:1651–1662. doi: 10.1097/CM9.0000000000003169
References
- 1.Nigrovic PA, Raychaudhuri S, Thompson SD. Review: Genetics and the Classification of Arthritis in Adults and Children. Arthritis Rheumatol 2018;70:7–17. doi: 10.1002/art.40350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dougados M, Baeten D. Spondyloarthritis. Lancet 2011;377:2127–2137. doi: 10.1016/s0140-6736(11)60071-8. [DOI] [PubMed] [Google Scholar]
- 3.Cassat JE, Skaar EP. Iron in infection and immunity. Cell Host Microbe 2013;13:509–519. doi: 10.1016/j.chom.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Üsküdar Cansu D, Üsküdar Teke H, Cansu GB, Korkmaz C. Evaluation of hyperferritinemia causes in rheumatology practice: A retrospective, single-center experience. Rheumatol Int 2021;41:1617–1624. doi: 10.1007/s00296-021-04935-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Anderson GJ, Frazer DM. Current understanding of iron homeostasis. Am J Clin Nutr 2017;106(Suppl 6):1559s–1566s. doi: 10.3945/ajcn.117.155804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yang Q, Liu W, Zhang S, Liu S. The cardinal roles of ferroportin and its partners in controlling cellular iron in and out. Life Sci 2020;258:118135. doi: 10.1016/j.lfs.2020.118135. [DOI] [PubMed] [Google Scholar]
- 7.Stockwell BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications. Cell 2022;185:2401–2421. doi: 10.1016/j.cell.2022.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dutt S, Hamza I, Bartnikas TB. Molecular mechanisms of iron and heme metabolism. Annu Rev Nutr 2022;42:311–335. doi: 10.1146/annurev-nutr-062320-112625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Koleini N, Shapiro JS, Geier J, Ardehali H. Ironing out mechanisms of iron homeostasis and disorders of iron deficiency. J Clin Invest 2021;131:e148671. doi: 10.1172/jci148671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alivernini S, Firestein GS, McInnes IB. The pathogenesis of rheumatoid arthritis. Immunity 2022;55:2255–2270. doi: 10.1016/j.immuni.2022.11.009. [DOI] [PubMed] [Google Scholar]
- 11.Nieuwenhuizen L Schutgens RE van Asbeck BS Wenting MJ van Veghel K Roosendaal G, et al. Identification and expression of iron regulators in human synovium: Evidence for upregulation in haemophilic arthropathy compared to rheumatoid arthritis, osteoarthritis, and healthy controls. Haemophilia 2013;19:e218–e227. doi: 10.1111/hae.12208. [DOI] [PubMed] [Google Scholar]
- 12.Wang H Zhang R Shen J Jin Y Chang C Hong M, et al. Circulating level of blood iron and copper associated with inflammation and disease activity of rheumatoid arthritis. Biol Trace Elem Res 2023;201:90–97. doi: 10.1007/s12011-022-03148-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yuan S, Larsson S. Causal associations of iron status with gout and rheumatoid arthritis, but not with inflammatory bowel disease. Clin Nutr 2020;39:3119–3124. doi: 10.1016/j.clnu.2020.01.019. [DOI] [PubMed] [Google Scholar]
- 14.Ledesma-Colunga MG Baschant U Weidner H Alves TC Mirtschink P Hofbauer LC, et al. Transferrin receptor 2 deficiency promotes macrophage polarization and inflammatory arthritis. Redox Biol 2023;60:102616. doi: 10.1016/j.redox.2023.102616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nazemian V Kalanaky S Manaheji H Houshmandi E Mohammadi M Zaringhalam J, et al. Anti-hyperalgesia effect of nanchelating based nano particle, RAc1, can be mediated via liver hepcidin expression modulation during persistent inflammation. Int Immunopharmacol 2019;69:337–346. doi: 10.1016/j.intimp.2019.02.003. [DOI] [PubMed] [Google Scholar]
- 16.Padjen I, Öhler L, Studenic P, Woodworth T, Smolen J, Aletaha D. Clinical meaning and implications of serum hemoglobin levels in patients with rheumatoid arthritis. Semin Arthritis Rheum 2017;47:193–198. doi: 10.1016/j.semarthrit.2017.03.001. [DOI] [PubMed] [Google Scholar]
- 17.Masson C. Rheumatoid anemia. Joint Bone Spine 2011;78:131–137. doi: 10.1016/j.jbspin.2010.05.017. [DOI] [PubMed] [Google Scholar]
- 18.Isaacs JD, Harari O, Kobold U, Lee JS, Bernasconi C. Effect of tocilizumab on haematological markers implicates interleukin-6 signalling in the anaemia of rheumatoid arthritis. Arthritis Res Ther 2013;15:R204. doi: 10.1186/ar4397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Youssef SR, Hassan EH, Morad CS, Elazab Elged AA, El-Gamal RA. Erythroferrone expression in anemic rheumatoid arthritis patients: Is it disordered iron trafficking or disease activity? J Inflamm Res 2021;14:4445–4455. doi: 10.2147/jir.S327465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.He X Zhang J Gong M Gu Y Dong B Pang X, et al. Identification of potential ferroptosis-associated biomarkers in rheumatoid arthritis. Front Immunol 2023;14:1197275. doi: 10.3389/fimmu.2023.1197275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ling H Li M Yang C Sun S Zhang W Zhao L, et al. Glycine increased ferroptosis via SAM-mediated GPX4 promoter methylation in rheumatoid arthritis. Rheumatology (Oxford) 2022;61:4521–4534. doi: 10.1093/rheumatology/keac069. [DOI] [PubMed] [Google Scholar]
- 22.Cheng Q Chen M Liu M Chen X Zhu L Xu J, et al. Semaphorin 5A suppresses ferroptosis through activation of PI3K-AKT-mTOR signaling in rheumatoid arthritis. Cell Death Dis 2022;13:608. doi: 10.1038/s41419-022-05065-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu J Feng Z Chen L Li Y Bian H Geng J, et al. TNF antagonist sensitizes synovial fibroblasts to ferroptotic cell death in collagen-induced arthritis mouse models. Nat Commun 2022;13:676. doi: 10.1038/s41467-021-27948-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ni LL Che YH Sun HM Wang B Wang MY Yang ZZ, et al. The therapeutic effect of wasp venom (Vespa magnifica, Smith) and its effective part on rheumatoid arthritis fibroblast-like synoviocytes through modulating inflammation, redox homeostasis and ferroptosis. J Ethnopharmacol 2023;317:116700. doi: 10.1016/j.jep.2023.116700. [DOI] [PubMed] [Google Scholar]
- 25.Wang Y Ding H Zheng Y Wei X Yang X Wei H, et al. Alleviated NCOA4-mediated ferritinophagy protected RA FLSs from ferroptosis in lipopolysaccharide-induced inflammation under hypoxia. Inflamm Res 2024;73:363–379. doi: 10.1007/s00011-023-01842-9. [DOI] [PubMed] [Google Scholar]
- 26.Lin Z Li W Wang Y Lang X Sun W Zhu X, et al. SMSCs-derived sEV overexpressing miR-433-3p inhibits angiogenesis induced by sEV released from synoviocytes under triggering of ferroptosis. Int Immunopharmacol 2023;116:109875. doi: 10.1016/j.intimp.2023.109875. [DOI] [PubMed] [Google Scholar]
- 27.Zhou R Chen Y Li S Wei X Hu W Tang S, et al. TRPM7 channel inhibition attenuates rheumatoid arthritis articular chondrocyte ferroptosis by suppression of the PKCα-NOX4 axis. Redox Biol 2022;55:102411. doi: 10.1016/j.redox.2022.102411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cheng Q Ni L Liu A Huang X Xiang P Zhang Q, et al. Spermidine protects cartilage from IL-1β-mediated ferroptosis. Mol Cell Biochem 2023;1–10. doi: 10.1007/s11010-023-04889-8. [DOI] [PubMed] [Google Scholar]
- 29.Liu Y Luo X Chen Y Dang J Zeng D Guo X, et al. Heterogeneous ferroptosis susceptibility of macrophages caused by focal iron overload exacerbates rheumatoid arthritis. Redox Biol 2024;69:103008. doi: 10.1016/j.redox.2023.103008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Xie M, Zhu C, Ye Y. Ferroptosis-related molecular clusters and diagnostic model in rheumatoid arthritis. Int J Mol Sci 2023;24:7342. doi: 10.3390/ijms24087342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ding Q Hu W Wang R Yang Q Zhu M Li M, et al. Signaling pathways in rheumatoid arthritis: Implications for targeted therapy. Signal Transduct Target Ther 2023;8:68. doi: 10.1038/s41392-023-01331-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ribeiro R, Batista F, Seguro Paula F, Delgado Alves J. Changes in iron metabolism induced by anti-interleukin-6 receptor monoclonal antibody are associated with an increased risk of infection. Pharmaceuticals (Basel) 2019;12:100. doi: 10.3390/ph12030100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Knoke K Rongisch RR Grzes KM Schwarz R Lorenz B Yogev N, et al. Tofacitinib suppresses IL-10/IL-10R signaling and modulates host defense responses in human macrophages. J Invest Dermatol 2022;142(3 Pt A):559–570.e6. doi: 10.1016/j.jid.2021.07.180. [DOI] [PubMed] [Google Scholar]
- 34.Ruan L Cai X Qian R Bei S Wu L Cao J, et al. Live macrophages loaded with Fe(3)O(4) and sulfasalazine for ferroptosis and photothermal therapy of rheumatoid arthritis. Mater Today Bio 2023;24:100925. doi: 10.1016/j.mtbio.2023.100925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen J Li S Ge Y Kang J Liao JF Du JF, et al. iTRAQ and PRM-based proteomic analysis provides new insights into mechanisms of response to triple therapy in patients with rheumatoid arthritis. J Inflamm Res 2021;14:6993–7006. doi: 10.2147/jir.S340351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Navarro-Compán V, Sepriano A, El-Zorkany B, van der Heijde D. Axial spondyloarthritis. Ann Rheum Dis 2021;80:1511–1521. doi: 10.1136/annrheumdis-2021-221035. [DOI] [PubMed] [Google Scholar]
- 37.Yang M Yu H Xu K Xie J Zheng H Feng R, et al. No evidence of a genetic causal relationship between ankylosing spondylitis and iron homeostasis: A two-sample Mendelian randomization study. Front Nutr 2023;10:1047640. doi: 10.3389/fnut.2023.1047640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thompson KN Bonham KS Ilott NE Britton GJ Colmenero P Bullers SJ, et al. Alterations in the gut microbiome implicate key taxa and metabolic pathways across inflammatory arthritis phenotypes. Sci Transl Med 2023;15:eabn4722. doi: 10.1126/scitranslmed.abn4722. [DOI] [PubMed] [Google Scholar]
- 39.Li Q Chen Z Yang C Wang L Ma J He T, et al. Role of ferroptosis-associated genes in ankylosing spondylitis and immune cell infiltration. Front Genet 2022;13:948290. doi: 10.3389/fgene.2022.948290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rong T, Jia N, Wu B, Sang D, Liu B. New insights into the regulatory role of ferroptosis in ankylosing spondylitis via consensus clustering of ferroptosis-related genes and weighted gene co-expression network analysis. Genes (Basel) 2022;13:1373. doi: 10.3390/genes13081373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hu X, Xie Q, Mo X, Jin Y. The role of extracellular histones in systemic-onset juvenile idiopathic arthritis. Ital J Pediatr 2019;45:14. doi: 10.1186/s13052-019-0605-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li X, Tao X, Ding X. An integrative analysis to reveal that CLEC2B and ferroptosis may bridge the gap between psoriatic arthritis and cancer development. Sci Rep 2022;12:14653. doi: 10.1038/s41598-022-19135-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang H, Tang Z, Liu S, Xie K, Zhang H. Acrylamide induces human chondrocyte cell death by initiating autophagy‑dependent ferroptosis. Exp Ther Med 2023;25:246. doi: 10.3892/etm.2023.11945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dilandro G De Cosmi V Corona F Torcoletti M Petaccia A Filocamo G, et al. May biscuits contribute to iron balance? An observation in children with juvenile idiopatic arthritis. Int J Food Sci Nutr 2015;66:811–814. doi: 10.3109/09637486.2015.1077791. [DOI] [PubMed] [Google Scholar]
- 45.Rathore KI, Redensek A, David S. Iron homeostasis in astrocytes and microglia is differentially regulated by TNF-α and TGF-β1. Glia 2012;60:738–750. doi: 10.1002/glia.22303. [DOI] [PubMed] [Google Scholar]
- 46.Niccoli L, Nannini C, Cassarà E, Kaloudi O, Cantini F. Frequency of anemia of inflammation in patients with ankylosing spondylitis requiring anti-TNFα drugs and therapy-induced changes. Int J Rheum Dis 2012;15:56–61. doi: 10.1111/j.1756-185X.2011.01662.x. [DOI] [PubMed] [Google Scholar]
- 47.Shao F Liu Q Zhu Y Fan Z Chen W Liu S, et al. Targeting chondrocytes for arresting bony fusion in ankylosing spondylitis. Nat Commun 2021;12:6540. doi: 10.1038/s41467-021-26750-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yuan W Yang Y Wei Y Yu X Bao J Zhong J, et al. Ferritin was involved in interleukin-17A enhanced osteogenesis through autophagy activation. Int Immunopharmacol 2023;124(Pt A):110916. doi: 10.1016/j.intimp.2023.110916. [DOI] [PubMed] [Google Scholar]
- 49.Guan Z, Jin X, Guan Z, Liu S, Tao K, Luo L. The gut microbiota metabolite capsiate regulate SLC2A1 expression by targeting HIF-1α to inhibit knee osteoarthritis-induced ferroptosis. Aging Cell 2023;22:e13807. doi: 10.1111/acel.13807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Miyauchi S Umekita K Hidaka T Umeki K Aratake Y Takahashi N, et al. Increased plasma lactoferrin levels in leucocytapheresis therapy in patients with rheumatoid arthritis. Rheumatology (Oxford) 2014;53:1966–1972. doi: 10.1093/rheumatology/keu219. [DOI] [PubMed] [Google Scholar]
- 51.Qu Z Yang F Hong J Wang W Li S Jiang G, et al. Causal relationship of serum nutritional factors with osteoarthritis: A Mendelian randomization study. Rheumatology (Oxford) 2021;60:2383–2390. doi: 10.1093/rheumatology/keaa622. [DOI] [PubMed] [Google Scholar]
- 52.Zhou J Liu C Sun Y Francis M Ryu MS Grider A, et al. Genetically predicted circulating levels of copper and zinc are associated with osteoarthritis but not with rheumatoid arthritis. Osteoarthritis Cartilage 2021;29:1029–1035. doi: 10.1016/j.joca.2021.02.564. [DOI] [PubMed] [Google Scholar]
- 53.Radakovich LB Burton LH Culver LA Afzali MF Marolf AJ Olver CS, et al. Systemic iron reduction via an iron deficient diet decreases the severity of knee cartilage lesions in the Dunkin-Hartley guinea pig model of osteoarthritis. Osteoarthritis Cartilage 2022;30:1482–1494. doi: 10.1016/j.joca.2022.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lv M Cai Y Hou W Peng K Xu K Lu C, et al. The RNA-binding protein SND1 promotes the degradation of GPX4 by destabilizing the HSPA5 mRNA and suppressing HSPA5 expression, promoting ferroptosis in osteoarthritis chondrocytes. Inflamm Res 2022;71:461–472. doi: 10.1007/s00011-022-01547-5. [DOI] [PubMed] [Google Scholar]
- 55.Burton LH, Radakovich LB, Marolf AJ, Santangelo KS. Systemic iron overload exacerbates osteoarthritis in the strain 13 guinea pig. Osteoarthritis Cartilage 2020;28:1265–1275. doi: 10.1016/j.joca.2020.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Karim A, Bajbouj K, Shafarin J, Qaisar R, Hall AC, Hamad M. Iron overload induces oxidative stress, cell cycle arrest and apoptosis in chondrocytes. Front Cell Dev Biol 2022;10:821014. doi: 10.3389/fcell.2022.821014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Li H Jiang X Xiao Y Zhang Y Zhang W Doherty M, et al. Combining single-cell RNA sequencing and population-based studies reveals hand osteoarthritis-associated chondrocyte subpopulations and pathways. Bone Res 2023;11:58. doi: 10.1038/s41413-023-00292-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jing X Du T Li T Yang X Wang G Liu X, et al. The detrimental effect of iron on OA chondrocytes: Importance of pro-inflammatory cytokines induced iron influx and oxidative stress. J Cell Mol Med 2021;25:5671–5680. doi: 10.1111/jcmm.16581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dai T Xue X Huang J Yang Z Xu P Wang M, et al. SCP2 mediates the transport of lipid hydroperoxides to mitochondria in chondrocyte ferroptosis. Cell Death Discov 2023;9:234. doi: 10.1038/s41420-023-01522-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Qiu Y Yao J Li L Xiao M Meng J Huang X, et al. Machine learning identifies ferroptosis-related genes as potential diagnostic biomarkers for osteoarthritis. Front Endocrinol (Lausanne) 2023;14:1198763. doi: 10.3389/fendo.2023.1198763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xia L, Gong N. Identification and verification of ferroptosis-related genes in the synovial tissue of osteoarthritis using bioinformatics analysis. Front Mol Biosci 2022;9:992044. doi: 10.3389/fmolb.2022.992044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang X Liu T Qiu C Yu S Zhang Y Sheng Y, et al. Characterization and role exploration of ferroptosis-related genes in osteoarthritis. Front Mol Biosci 2023;10:1066885. doi: 10.3389/fmolb.2023.1066885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yang T Yang G Wang G Jia D Xiong B Lu X, et al. Bioinformatics identification and integrative analysis of ferroptosis-related key lncRNAs in patients with osteoarthritis. Biosci Rep 2023;43:BSR20230255. doi: 10.1042/bsr20230255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jing X Lin J Du T Jiang Z Li T Wang G, et al. Iron overload is associated with accelerated progression of osteoarthritis: The role of DMT1 mediated iron homeostasis. Front Cell Dev Biol 2021;8:594509. doi: 10.3389/fcell.2020.594509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Liu H Deng Z Yu B Liu H Yang Z Zeng A, et al. Identification of SLC3A2 as a potential therapeutic target of osteoarthritis involved in ferroptosis by integrating bioinformatics, clinical factors and experiments. Cells 2022;11:3430. doi: 10.3390/cells11213430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lv Z Han J Li J Guo H Fei Y Sun Z, et al. Single cell RNA-seq analysis identifies ferroptotic chondrocyte cluster and reveals TRPV1 as an anti-ferroptotic target in osteoarthritis. EBioMedicine 2022;84:104258. doi: 10.1016/j.ebiom.2022.104258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhao C Sun G Li Y Kong K Li X Kan T, et al. Forkhead box O3 attenuates osteoarthritis by suppressing ferroptosis through inactivation of NF-κB/MAPK signaling. J Orthop Translat 2023;39:147–162. doi: 10.1016/j.jot.2023.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang S Li W Zhang P Wang Z Ma X Liu C, et al. Mechanical overloading induces GPX4-regulated chondrocyte ferroptosis in osteoarthritis via Piezo1 channel facilitated calcium influx. J Adv Res 2022;41:63–75. doi: 10.1016/j.jare.2022.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Miao Y Chen Y Xue F Liu K Zhu B Gao J, et al. Contribution of ferroptosis and GPX4’s dual functions to osteoarthritis progression. EBioMedicine 2022;76:103847. doi: 10.1016/j.ebiom.2022.103847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sun K Hou L Guo Z Wang G Guo J Xu J, et al. JNK-JUN-NCOA4 axis contributes to chondrocyte ferroptosis and aggravates osteoarthritis via ferritinophagy. Free Radic Biol Med 2023;200:87–101. doi: 10.1016/j.freeradbiomed.2023.03.008. [DOI] [PubMed] [Google Scholar]
- 71.Wen Z Xia G Liang C Wang X Huang J Zhang L, et al. Selective clearance of senescent chondrocytes in osteoarthritis by targeting excitatory amino acid transporter protein 1 to induce ferroptosis. Antioxid Redox Signal 2023;39:262–277. doi: 10.1089/ars.2022.0141. [DOI] [PubMed] [Google Scholar]
- 72.Mo Z, Xu P, Li H. Stigmasterol alleviates interleukin-1beta-induced chondrocyte injury by down-regulatingsterol regulatory element binding transcription factor 2 to regulateferroptosis. Bioengineered 2021;12:9332–9340. doi: 10.1080/21655979.2021.2000742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhou X Zheng Y Sun W Zhang Z Liu J Yang W, et al. D-mannose alleviates osteoarthritis progression by inhibiting chondrocyte ferroptosis in a HIF-2α-dependent manner. Cell Prolif 2021;54:e13134. doi: 10.1111/cpr.13134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Pan Z He Q Zeng J Li S Li M Chen B, et al. Naringenin protects against iron overload-induced osteoarthritis by suppressing oxidative stress. Phytomedicine 2022;105:154330. doi: 10.1016/j.phymed.2022.154330. [DOI] [PubMed] [Google Scholar]
- 75.Wang X, Liu Z, Peng P, Gong Z, Huang J, Peng H. Astaxanthin attenuates osteoarthritis progression via inhibiting ferroptosis and regulating mitochondrial function in chondrocytes. Chem Biol Interact 2022;366:110148. doi: 10.1016/j.cbi.2022.110148. [DOI] [PubMed] [Google Scholar]
- 76.Yan J, Feng G, Ma L, Chen Z, Jin Q. Metformin alleviates osteoarthritis in mice by inhibiting chondrocyte ferroptosis and improving subchondral osteosclerosis and angiogenesis. J Orthop Surg Res 2022;17:333. doi: 10.1186/s13018-022-03225-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Xu C Ni S Xu N Yin G Yu Y Zhou B, et al. Theaflavin-3,3′-digallate inhibits erastin-induced chondrocytes ferroptosis via the Nrf2/GPX4 signaling pathway in osteoarthritis. Oxid Med Cell Longev 2022;2022:3531995. doi: 10.1155/2022/3531995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.He Q Yang J Pan Z Zhang G Chen B Li S, et al. Biochanin A protects against iron overload associated knee osteoarthritis via regulating iron levels and NRF2/System xc-/GPX4 axis. Biomed Pharmacother 2023;157:113915. doi: 10.1016/j.biopha.2022.113915. [DOI] [PubMed] [Google Scholar]
- 79.Chen R Ying C Zou Y Lin C Fu Q Xiang Z, et al. Sarsasapogenin inhibits YAP1-dependent chondrocyte ferroptosis to alleviate osteoarthritis. Biomed Pharmacother 2023;168:115772. doi: 10.1016/j.biopha.2023.115772. [DOI] [PubMed] [Google Scholar]
- 80.Sun J, Zhang Y, Wang C, Ruan Q. Kukoamine A protects mice against osteoarthritis by inhibiting chondrocyte inflammation and ferroptosis via SIRT1/GPX4 signaling pathway. Life Sci 2023;332:122117. doi: 10.1016/j.lfs.2023.122117. [DOI] [PubMed] [Google Scholar]
- 81.Li S He Q Chen B Zeng J Dou X Pan Z, et al. Cardamonin protects against iron overload induced arthritis by attenuating ROS production and NLRP3 inflammasome activation via the SIRT1/p38MAPK signaling pathway. Sci Rep 2023;13:13744. doi: 10.1038/s41598-023-40930-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gong Z, Wang Y, Li L, Li X, Qiu B, Hu Y. Cardamonin alleviates chondrocytes inflammation and cartilage degradation of osteoarthritis by inhibiting ferroptosis via p53 pathway. Food Chem Toxicol 2023;174:113644. doi: 10.1016/j.fct.2023.113644. [DOI] [PubMed] [Google Scholar]
- 83.Yang Z Jiang W Xiong C Shang J Huang Y Zhou X, et al. Calcipotriol suppresses GPX4-mediated ferroptosis in OA chondrocytes by blocking the TGF-β1 pathway. Cytokine 2023;171:156382. doi: 10.1016/j.cyto.2023.156382. [DOI] [PubMed] [Google Scholar]
- 84.Ruan Q, Wang C, Zhang Y, Sun J. Brevilin A attenuates cartilage destruction in osteoarthritis mouse model by inhibiting inflammation and ferroptosis via SIRT1/Nrf2/GPX4 signaling pathway. Int Immunopharmacol 2023;124(Pt B):110924. doi: 10.1016/j.intimp.2023.110924. [DOI] [PubMed] [Google Scholar]
- 85.Wan Y, Shen K, Yu H, Fan W. Baicalein limits osteoarthritis development by inhibiting chondrocyte ferroptosis. Free Radic Biol Med 2023;196:108–120. doi: 10.1016/j.freeradbiomed.2023.01.006. [DOI] [PubMed] [Google Scholar]
- 86.Guo Z Lin J Sun K Guo J Yao X Wang G, et al. Deferoxamine alleviates osteoarthritis by inhibiting chondrocyte ferroptosis and activating the Nrf2 pathway. Front Pharmacol 2022;13:791376. doi: 10.3389/fphar.2022.791376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Jing X Wang Q Du T Zhang W Liu X Liu Q, et al. Calcium chelator BAPTA‑AM protects against iron overload‑induced chondrocyte mitochondrial dysfunction and cartilage degeneration. Int J Mol Med 2021;48:196. doi: 10.3892/ijmm.2021.5029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhou M Zhai C Shen K Liu G Liu L He J, et al. miR-1 Inhibits the ferroptosis of chondrocyte by targeting CX43 and alleviates osteoarthritis progression. J Immunol Res 2023;2023:2061071. doi: 10.1155/2023/2061071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kong R, Ji L, Pang Y, Zhao D, Gao J. Exosomes from osteoarthritic fibroblast-like synoviocytes promote cartilage ferroptosis and damage via delivering microRNA-19b-3p to target SLC7A11 in osteoarthritis. Front Immunol 2023;14:1181156. doi: 10.3389/fimmu.2023.1181156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Yao Q Wu X Tao C Gong W Chen M Qu M, et al. Osteoarthritis: Pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther 2023;8:56. doi: 10.1038/s41392-023-01330-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Keller SF, Mandell BF. Management and cure of gouty arthritis. Med Clin North Am 2021;105:297–310. doi: 10.1016/j.mcna.2020.09.013. [DOI] [PubMed] [Google Scholar]
- 92.Fatima T McKinney C Major TJ Stamp LK Dalbeth N Iverson C, et al. The relationship between ferritin and urate levels and risk of gout. Arthritis Res Ther 2018;20:179. doi: 10.1186/s13075-018-1668-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Topiwala A Mankia K Bell S Webb A Ebmeier KP Howard I, et al. Association of gout with brain reserve and vulnerability to neurodegenerative disease. Nat Commun 2023;14:2844. doi: 10.1038/s41467-023-38602-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li X He T Yu K Lu Q Alkasir R Guo G, et al. Markers of iron status are associated with risk of hyperuricemia among Chinese adults: Nationwide population-based study. Nutrients 2018;10:191. doi: 10.3390/nu10020191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Shen J Xie Z Zhao T Li Z Yang Q Xi Y, et al. Iron supplementation aggravates joint inflammation in mice with acute gouty arthritis. Int J Rheum Dis 2022;25:962–965. doi: 10.1111/1756-185x.14370. [DOI] [PubMed] [Google Scholar]
- 96.Zhong CS Zeng B Qiu JH Xu LH Zhong MY Huang YT, et al. Gout-associated monosodium urate crystal-induced necrosis is independent of NLRP3 activity but can be suppressed by combined inhibitors for multiple signaling pathways. Acta Pharmacol Sin 2022;43:1324–1336. doi: 10.1038/s41401-021-00749-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Du K Zhou Q Wang Z Mo C Dong W Wei N, et al. Polydatin ameliorates inflammation and oxidative stress associated with MSU-induced gouty arthritis in mice by regulating PPAR-γ and ferritin activation. Life Sci 2023;326:121766. doi: 10.1016/j.lfs.2023.121766. [DOI] [PubMed] [Google Scholar]
- 98.Zhang X Zhang D Jia H Feng Q Wang D Liang D, et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat Med 2015;21:895–905. doi: 10.1038/nm.3914. [DOI] [PubMed] [Google Scholar]
- 99.Ma Y Li W Niu S Zhu X Chu M Wang W, et al. BzATP reverses ferroptosis-induced gut microbiota disorders in collagen-induced arthritis mice. Int Immunopharmacol 2023;124(Pt A):110885. doi: 10.1016/j.intimp.2023.110885. [DOI] [PubMed] [Google Scholar]