Abstract
Background
The aim of this study was to examine the impact of different stages of dengue infection on immune cell counts among dengue patients and to compare them with cases of non-dengue febrile illness.
Methods
The recruited patients were divided into two groups: the first group served as a control (n = 55), representing non-dengue febrile illness, and the second group was identified as dengue febrile illness (n = 149), which was further divided into three groups based on infection stage. Blood samples were collected from the selected patients and subjected to blood cell component analysis. To find IgG and IgM as well as the dengue virus non-structural antigen-1 (NS1), an immunochromatographic test (ICT) kit was utilized. Additionally, a hematological analyzer was used to determine complete blood cell counts (CBC). Data was thoroughly analyzed using Graph Pad Prism 6 software. The differences in means of different groups were calculated by applying the student's t-test.
Results
The findings revealed the presence of severe leucopenia and thrombocytopenia at stages 1 and 2, accompanied by lymphopenia at stage 1. Group comparisons indicated that only teenagers exhibited a significantly lower white blood cell count compared to older individuals, while no significant differences were observed in lymphocytes, platelets, and monocytes across all age groups. Comparing different age groups of normal individuals to dengue-infected patients, the results unveiled that leucopenia was most severe in adults, followed by teenagers and children, with no significant difference in the elderly. Furthermore, adults showed the greatest degree of thrombocytopenia, followed by teens and kids, with the elderly showing the greatest degree of thrombocytopenia. Adults and teens showed extreme neutrophilia, whereas young children and the elderly showed no discernible abnormalities. Elderly patients experienced a marked decrease in monocyte count, a phenomenon not observed in other age groups.
Conclusion
In conclusion both, leucopenia & thrombocytopenia, are most severe in stages 1 and 2, whereas neutrophilia & lymphopenia are predominantly severe in stage 1. These results imply that the consequences associated with dengue infection are more severe in the early stages and tend to ameliorate as the patient progresses toward recovery.
Keywords: Infection control, Dengue infection, Hematological parameters, Immune cell count, Leucopenia, Thrombocytopenia
1. Introduction
Dengue fever is caused by an arthropod-borne virus called dengue virus (DENV) which includes a total of four serotypes i.e., DEN-1, DEN-2, DEN-3, and DEN-4. The virus, also known as arbovirus, is a member of the genus Flavivirus and family Flaviviridae [1,2]. Every continent on the planet has been impacted by dengue-related illnesses, with the exception of Antarctica. Currently, dengue is a problem in over 120 countries, and 2.5 billion people worldwide might contract the sickness [3]. In some of the places, the disease has attained the state of endemic inducing significant health complications in the population. Dengue hemorrhagic fever (DHF), the most serious kind of illness, is caused by dengue fever [4]. Sometimes, however, dengue fever leads to a potentially fatal complication, also known as dengue shock syndrome (DSS). The clinical presentation in both adults and children varies and is dependent upon the severity of the infection, immune status, age as well as genetic background [5].
In Southeast Asia, Africa, and America, the dengue virus and its serotypes infect over 50 million people yearly, and about 50,000 of those cases result in hospitalization for DHF or DSS [6]. Pakistan is among the top-ranked nations and one of the most dengue-affected regions; the disease is most common in the post-monsoon season [7]. Lahore is reported to be one of the worst hit region in 2011 with 14,000 cases and 300 fatalities from dengue [8]. A study reported that out of a total of 1691 asymptomatic dengue infections, 756 were in Karachi, 391 in Islamabad and 228 cases were reported in Peshawar [9]. In the past, dengue infections have also been reported in the Swat and Mansehra districts of the KPK province with high morbidity and mortality rates [10].
The escalating rate of dengue infections underscores the critical importance of early diagnosis and case confirmation, serving not only to facilitate etiological investigations and clinical interventions but also to advance disease control measures. As a result, several diagnostic techniques have been created and put into use to identify dengue infections. These approaches include the detection of the whole dengue virus, which is accomplished by methods like cell culture and immunofluorescence, as well as the identification of viral antigens, which is usually done by ELISA. Additionally, tests such as the complement fixation test (CF) and the hemagglutination inhibition essay (HI) are used to search for anti-dengue virus antibodies. Additionally, the detection of viral nucleic acids is made possible by real-time reverse transcription-polymerase chain reaction (RT-PCR). Building upon these fundamental principles, various commercially available diagnostic kits are now employed to swiftly diagnose dengue infections [[11], [12], [13], [14]].
It is noteworthy that the efficacy of detection depends on the specific phase of illness and the individual's infection status. For four to five days in the early stages, the virus can be found in serum, plasma, circulating blood cells, and other organs [15,16]. But it's crucial to recognize that, even if these techniques are priceless, it might take hours or even days for detection, considered as some of the advantages [5]. According to a prior study, dengue infection alters blood cells and modifies hematological parameters [17]. Leukopenia is one of the hematological changes resulting in a count less than 2 × 103/μL while thrombocytopenia may occur in 88 % of the dengue patients [18]. It was also found that platelet count also alters with the severity of the dengue infection with an increased hematocrit. Furthermore, reports of a rise in liver enzymes and a change in the coagulation profile have also been made [19]. Therefore, as a matter of public health, it is crucial to diagnose and identify dengue infection during its acute phase. It might be useful to doctors and other healthcare professionals in identifying individuals and keeping a close eye out for dengue fever symptoms. Moreover, occurrences of such infectious diseases in large population will cause economic burden on both patients as well as healthcare authorities. Hence, a comprehensive and complete study was required to evaluate all hematological parameters to assess their association with the infection status and the impact of various age and sex groups on them. This study is being done for the first time in northern region of Pakistan that has ethnic population from different strata of society. The main objective of the present study was to evaluate the total hematological parameters in patients visiting for dengue diagnosis to relate the findings as a screening marker and based on this, to identify/separate the dengue patients from non-dengue fever for their better therapeutic interventions.
2. Materials & methods
2.1. Patient Selection criteria
The study enrolled patients exhibiting symptoms of fever ranging from 38.5 °C to 41.4 °C, along with one or more of the following criteria: 1) Joint pain, 2) Rash, 3) Myalgia, 4) Headache, 5) Retro-ocular pain, 6) Abdominal pain, 7) Hemorrhagic manifestations, and 8) Conjunctival congestion [5]. Patients presenting with fever and showing slight positive results for malaria or immunochromatographic positivity for malaria, as well as those with urinary tract infections, enteric fever, liver abscesses, or pneumonitis, were excluded from the study. All enrolled patients were categorized into two distinct groups: the non-dengue febrile illness group, which served as the control group, and the dengue febrile illness group, further subdivided into Stage 1, Stage 2, and Stage 3 groups.
2.2. Ethical consideration
The ethical committee of the Department of Microbiology at Hazara University Mansehra, Pakistan, approved the study (Approval No: F. HU/Ethical/2015/01), which was carried out in compliance with the Declaration of Helsinki. Each participant received a briefing regarding the study's purpose in their native tongue and was free to ask any questions they had about it. They were ensured confidentiality and anonymity, and those who consented to participate were recruited for the study.
2.3. Sample collection and processing
This study was designed as a randomized controlled trial. Blood samples were collected from a reputable diagnostic facility in the northern region of Pakistan. A total of 149 blood samples were obtained from patients suspected of having dengue, who had visited the center for diagnosis. Additionally, 55 blood samples were collected from patients with non-dengue febrile illness to serve as a control group.
For hematological analysis, each blood sample was divided into 2 mL (mL) sterile EDTA tubes and 3 mL gel vials for serum separation. Every sample was appropriately labeled with patient information. Dengue hemorrhagic fever or dengue fever was suspected in all individuals. Demographic information, including age, gender, prior history of dengue fever, duration of fever, treatments received, and various clinical symptoms such as high fever, skin rash, gum bleeding, hematuria (bleeding in stool or urine), and mucosal bleeding, was obtained through interviews. All collected data was meticulously documented along with patient information, and the samples were stored according to established laboratory protocols.
2.4. Complete blood count (hematological analysis)
Hematological analysis, including hemoglobin, hematocrit, total leukocyte count, differential leukocyte count, and platelet count assessments, was conducted using blood samples from the EDTA tubes. A Hematology Analyzer (Mindray BC 5000, China) was employed for this purpose.
2.5. Analysis of dengue antigen and antibodies
Blood samples from clot tubes were used for immunochromatographic testing (ICT). After centrifugation, serum was separated into new tubes for the detection of NS1 antigen, IgG, and IgM antibodies against the dengue virus in the blood. ICT kits from SD Bioline and Humasis (Korea), were employed for this purpose.
The following criteria were applied to categorize dengue patients based on the infection stage.
-
1.
A positive NS1 test indicated the initial stage of infection.
-
2.
Ns1+IgM or IgM positivity only indicated a chronic stage of infection.
-
3.
The detection of IgG positivity suggested a latent stage of infection.
2.6. Statistical analysis
Data analysis was conducted utilizing GraphPad Prism 6 software. Given the comparison of parameters from two independent groups, we employed the Student's unpaired t-test. Significance levels were interpreted as follows: p* <0.05 (significant), p** < 0.01 (more significant), p*** < 0.001 (most significant), and p**** < 0.0001 (extremely significant).
3. Results
The primary objective of this investigation was to explore the hematological profile of various immune cells throughout distinct phases of infection in dengue-infected individuals. Furthermore, we performed a comparative study of the hematological profiles of dengue-infected persons in various age and gender groups, and generated a control group of 55 samples of non-dengue individuals (dengue-negative individuals). Within this control group, 30 were male, and 25 were female. Among them, 9 samples belonged to children (<10 years of age), 10 to teenagers (aged 11–19), 29 to adults (aged 20–45), and 7 to the elderly (>45 years).
In the cohort of 149 dengue-infected patients, 94 were male, and 55 were female. Age-wise, there were 14 pediatric patients, 28 teenagers, 95 adults, and 12 elderly individuals. The total number of dengue-infected patients was 149. Within this group, 97 individuals tested positive for NS1 antigen (indicating Stage 1 of infection), 38 tested positive for both NS1 antigen and IgM antibodies or IgM antibodies alone (reflecting Stage 2 or the chronic stage), and 14 were solely positive for IgG antibodies (representing Stage 3). Stage 3 specifically encompassed subjects who had recently cleared a prior infection; hence, they tested negative for NS1 and positive for IgG antibodies.
3.1. Comparison of total WBC and platelet counts (PC) between patients with non-denguefebrile illness and dengue-infected patients at various infection stages
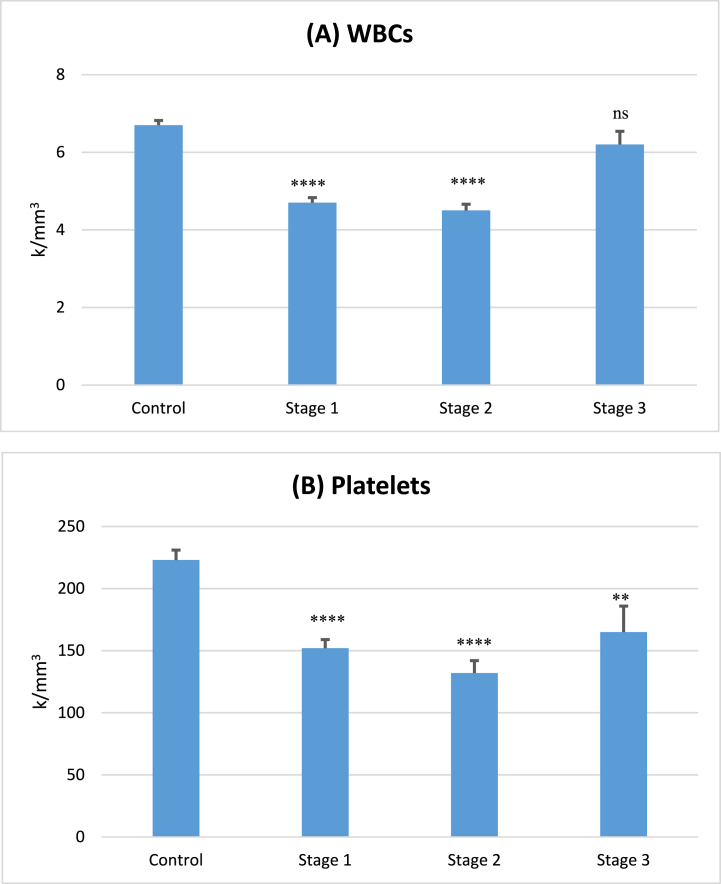
We compared the total WBC counts (all types i.e., Neutrophils, Eosinophils, etc.), and PC between normal subjects and dengue-infected patients during three distinct stages of dengue infection (see Fig. 1). In comparison with patients with non-dengue febrile illness, an extremely significant decrease (p < 0.0001) in WBC count was observed during Stage 1 and Stage 2 of the infection. However, when the infection entered stage 3, the level of difference between both groups shifted to a “non-significance” level i.e., p > 0.05, suggesting a recovery of WBC counts as the infection wanes.
Fig. 1.
(A) Total white blood cell and (B) platelet counts across various phases of dengue infection.
The data is displayed as means ± SEM. Statistical analysis was performed using a two-tailed Student's unpaired t-test. Significant differences are denoted as follows: (p < 0.0001), ** (p < 0.01), ns (p > 0.05).
Moreover, during stages 1–2 of the infection, an extremely significant decrease (p < 0.0001) in the platelet counts was also evident between patients with non-dengue febrile illness and infected groups (Fig. 1). At Stage 3, though the difference between the control and infected group remained significant (p = 0.001), a noticeable reduction in PC was evident as the p values changed from “extremely significant” in stages 1–2 to “more significant” (p**** to p**) at stage 3 (Table 1). This decrease suggests a gradual recovery of platelet count during the convalescent stage of the infection.
Table 1.
Comparison of White Blood Cell and Platelets counts in different groups.
| Groups | WBC Count (k/mm3) | Platelet Counts (k/mm3) |
|---|---|---|
| Control | 6.7 ± 0.12 | 223 ± 8 |
| Stage 1 | 4.7 ± 0.13 | 152 ± 7 |
| Stage 2 | 4.5 ± 0.16 | 132 ± 10 |
| Stage 3 | 6.2 ± 0.34 | 165 ± 21 |
3.2. Analysis of effect of dengue infection stages on differential WBC counts
We analyzed the proportions of neutrophils, lymphocytes, monocytes, and eosinophils among total white blood cells (WBCs) during various stages of dengue infection (depicted in Fig. 2).
-
•
Neutrophils: Compared to the control group, the percentage of neutrophils exhibited an extremely significant increase (p < 0.0001) at stage 1 of the infection (Fig. 2). Subsequently, as the disease progressed and entered to next stages i.e., 2&3, this percentage decreased resulting in no statistically significant differences between control and infected groups.
-
•
Lymphocytes: Contrary to neutrophils, the lymphocyte's percentage exhibited a significant decrease at first two stages (p < 0.0001 for stage 1 & p < 0.05 for stage 2, respectively). However, once the patient reached stage 3 of the infection, the level of significance between the control and the infected groups returned to normal levels i.e., no significant difference (Fig. 2).
-
•
Monocytes: Monocyte counts showed a distinct pattern compared to above both WBCs. During the first stage of infection, monocyte counts decreased significantly (p < 0.05) (Fig. 2). In subsequent stage/stage 2, the percentage of monocytes increased and reached at the “no significant” level compared to the control group. Finally, upon attaining stage 3, the percentage of monocytes significantly increased (p < 0.05) and surpassed the control group differential counts.
-
•
Eosinophils: In contrast to all other WBCs discussed above, the Eosinophil counts showed a consistent pattern of percentage increase right from the first stage to the recovery/last phase (Fig. 2). However, in statistical comparison to the control group, the increase remained within the non-significant levels (p > 0.05).
Fig. 2.
Percentage profiles of differential WBCs (neutrophils, lymphocytes, monocytes, and eosinophils) across various phases of dengue infections.
Data is represented as means ± SEM. Statistical analysis was conducted using a two-tailed Student's unpaired t-test. Significant differences are indicated as follows: **** (p < 0.0001), * (p < 0.05). The absence of * on stage 1, stage 2, and stage 3 bars indicates a non-significant comparison with the control.
3.3. Gender-based analysis of immune cells counts
We conducted a comparative analysis of various immune cell parameters, including total white blood cell (WBC) and platelet counts, as well as the percentages of neutrophils, lymphocytes, monocytes, and eosinophils, between male and female dengue-infected patients (as illustrated in Fig. 3).
Fig. 3.
Hematological profile variations based on gender in dengue-infected patients.
The data is presented as means ± SEM. Statistical analysis was performed using a two-tailed Student's unpaired t-test. * (p < 0.05), ns (not significant).
Among these parameters, statistically significant differences were observed in white blood cell counts between the patients of both genders. Specifically, the WBC counts of female patients were significantly lower when compared to their male counterparts (p = 0.0044). On the other hand, there were no statistically significant variations in platelet counts, neutrophil, lymphocyte, monocyte, and eosinophil percentages between male and female patients.
This analysis sheds light on gender-based variations in immune cell profiles, highlighting a noteworthy discrepancy in white blood cell counts among male and female dengue-infected patients.
3.4. Comparison of immune cells across age groups in dengue-infected patients
We further analyzed various immune cell parameters among different age groups (Fig. 4). Our analysis revealed that white blood cell counts were significantly higher in adults when compared to teenagers (p = 0.0474). When comparing the other age categories, no discernible variations were found. Platelet counts showed no significant variation when comparing the four age groups using a Student t-test. In terms of neutrophil counts, we observed a significant increase in adults when compared to children (p = 0.0370), while no significant differences were noted in comparisons with the other age groups. Similarly, no statistically significant differences were identified when comparing age groups for lymphocyte and monocyte counts. Interestingly, eosinophil counts were significantly lower in adults when compared to children (p = 0.0330), but no statistical differences were observed in comparisons with the other age groups.
Fig. 4.
Hematological profile variations based on age groups in dengue-infected patients.
Data is presented as means ± SEM. Statistical analysis was conducted using a two-tailed Student's unpaired t-test. Values sharing the same alphabet letters indicate no statistical difference, whereas values with differing alphabet letters are significantly distinct.
3.5. Age-based analysis of hematological profiles in dengue-infected patients and control
We conducted a comparative assessment of the hematological profiles of different immune cells in dengue-infected patients across various age groups when compared to their respective control groups (Fig. 5). The purpose of this investigation was to determine whether there were any notable differences in hematological parameters between the immune cell counts in different age groups and the equivalent control groups.
Fig. 5.
Comparative Hematological Profiles of Dengue Patients and Corresponding patients with non-dengue febrile illness Age Groups.
Data is represented as means ± SEM. Statistical analysis was conducted using a two-tailed Student's unpaired t-test. Significance levels are indicated as follows: **** (p < 0.0001), *** (p < 0.001), ** (p < 0.01), * (p < 0.05), ns (not significant).
Except for the elderly, the WBC counts were significantly lower in patients than their respective patients with non-dengue febrile illness groups (Fig. 5). The “extremely significant” results were observed in adults (p**** < 0.0001), “most significant” in the teens category (***p = 0.0002) while “significant* (p* = 0.0145) in the children category when compared with their respective patients with non-dengue febrile illness groups.
Similarly, in comparison to their patients with non-dengue febrile illness age fellows, platelet counts exhibited a significant decrease in patients of all age categories (p = 0.0334, 0.0004, 0.0001, and 0.0226 among children, teens, adults, and elderly, respectively). Neutrophil counts, on the other hand, displayed significantly increased levels in the patients categorized under teen and adults groups (p = 0.0489 & 0.012, respectively), but without any significant differences in children and the elderly (p > 0.05), when compared to their respective control groups (Fig. 5).
For lymphocytes, the teenager and adult patient groups showed a significant decrease (p = 0.0388 & 0.0009, respectively) while the other two age groups i.e., children and elderly showed no significant differences in comparison to their health age groups. Regarding monocytes, every age group—aside from the elderly exhibited non-significant variation in the number of patients when compared to their respective normal age groups. Similarly, regarding eosinophils, no patient age group exhibited any noteworthy variations when compared to the corresponding control groups (Fig. 5).
4. Supplementary results
We also examined the levels of hemoglobin (Hb) and hematocrit (HCT) in dengue-infected patients and compared them to those in normal individuals.
4.1. Comparison of hemoglobin (Hb) and hematocrit (HCT) between normal individuals and dengue-infected patients at different stages of dengue infection
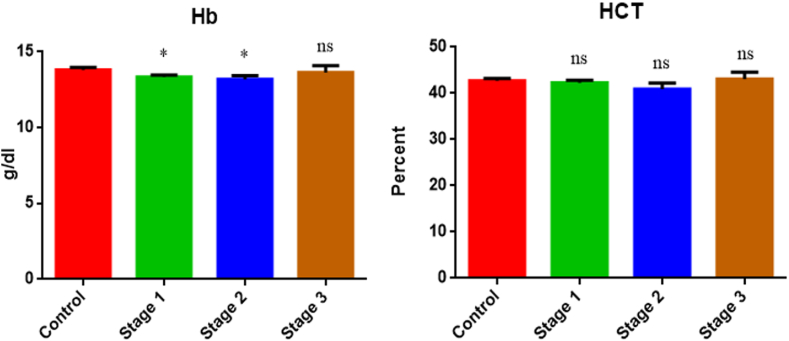
Our analysis of hemoglobin levels revealed a significant difference in Stages 1 and 2 (0.0370 and 0.0302, respectively) while Stage 3 exhibited no significant difference (as illustrated in Fig. 6). In contrast, when comparing hematocrit levels (HCT), no significant differences were observed across all three stages.
Fig. 6.
Comparative analysis of hemoglobin (Hb) and hematocrit (Hct) in different stages of dengue-infected patients compared to normal individuals.
4.2. Comparison of hemoglobin (Hb) and hematocrit (HCT) between normal and Dengue-Infected Males and Females
We analyzed hemoglobin (Hb) and hematocrit (HCT) levels in both dengue-infected and normal individuals, separately for males and females.
When comparing Hb levels in male dengue patients with those in normal males, we observed a more significant difference (0.0076) (as depicted in Fig. 7). Conversely, when comparing Hb levels in female dengue patients with those in normal females, no significant difference was observed. In terms of hematocrit (HCT) levels, our analysis revealed no significant differences when comparing normal and dengue-infected females and males.
Fig. 7.
Comparative analysis of hemoglobin (Hb) and hematocrit (Hct) in normal and dengue-infected males and females.
4.3. Comparison of hemoglobin (Hb) and hematocrit (HCT) across various age groups of dengue-infected patients
We conducted an analysis of hemoglobin (Hb) and hematocrit (HCT) levels among different age groups of dengue-infected patients to explore potential variations. Our examination of Hb levels showed significant differences in the children-teens and children-adults age groups (0.0145 and 0.0127, respectively), as depicted in Fig. 8. No other age groups exhibited significant differences in Hb levels. Likewise, in the case of HCT, the children-teens group displayed a more significant difference (0.0064), and the children-adults group showed a significant difference (0.0137). Other age groups did not, however, show statistically significant variations in HCT levels.
Fig. 8.
Comparative analysis of hemoglobin (Hb) and hematocrit (Hct) across various age groups.
4.4. Comparison of hemoglobin (Hb) and hematocrit (HCT) Between Normal Individuals and Dengue-infected patients across various age groups
Our analysis of Hb and HCT levels in both normal individuals and dengue-infected patients across different age groups did not reveal any significant differences (as illustrated in Fig. 9).
Fig. 9.
Comparative analysis of hemoglobin (Hb) and hematocrit (Hct) between normal individuals and dengue-infected patients across various age categories.
5. Discussion
The global impact of dengue on public health continues to be a significant concern. In Pakistan, since 2005, dengue has been spreading rapidly during the monsoon season, establishing itself as an endemic disease throughout the country [7]. The primary aim of this study was to assess individuals with suspected dengue who sought diagnosis at the study center. Our study concentrated on the hematological changes in distinct blood components, namely immune cell counts, throughout different phases of dengue illness. These counts were assessed among different demographic groups, including gender and age, and were compared to immune cell counts in patients with non-dengue febrile illness.
5.1. Stage-wise comparison
The findings of this study uncovered pronounced leucopenia (a low WBC count) in stages 1 and 2, associated with IgM-positive individuals. Notably, the WBC count returned to normal in stage 3 for dengue-infected individuals. This occurrence of leucopenia in stages 1 and 2 can be attributed to bone marrow suppression triggered by the dengue virus. As evidence from previous studies, bone marrow suppression inhibits the synthesis of several hematological components that might alter the infection fighting process of the host system [20]. The recovery of normal WBC counts in stage 3 suggests viral clearance and the transition to the recovery stage. This observation is consistent with recent studies [20,21] which reported leucopenia in 51 % (<4000 cells/mm3) and 52.3 % of dengue-infected individuals, respectively. Similarly, Butt et al. [22], found that 52.8 % of individuals with dengue experienced leucopenia, aligning closely with our findings. Multiple studies have reported leucopenia as a characteristic consequence of dengue infection, with a substantial proportion of patients displaying reduced WBC counts [23,24].
Our study also brought to light extreme thrombocytopenia (a low platelet count) in stages 1 and 2 of dengue-infected patients, with platelet counts rebounding in stage 3. The mechanism behind thrombocytopenia in the earlier stages is rooted in bone marrow suppression leading to decreased platelet production [25]. Alternatively, immune-mediated mechanisms can contribute to platelet destruction and the formation of antigen-antibody complexes [26]. The hemorrhagic fever that is one of the complications of dengue fever is attributed to such immune-mediated mechanisms. Estimating the level of thrombocytes NS1 antigen in patients might provide significant data on the immunological role of a disease [27]. The increase in platelet count observed in stage 3 is indicative of viral clearance, and patients subsequently enter the recovery stage. An association between dengue infection and thrombocytopenia has been documented in previous studies. Mehta et al. (2016) [28] noted thrombocytopenia in 74 % of IgM-positive patients out of a total of 200. Another study by Tathe et al. (2013) [29] demonstrated that out of 56 NS1-positive cases, 70.27 % of antibody-positive patients displayed thrombocytopenia. The results of the study conducted by Dhir et al. (2015) [30] also established a significant (p-value = 0.0332) association of thrombocytopenia to the positivity of specific antibodies. Kulkarni et al. (2011) reported similar results where 79.2 % of NS1-positive cases exhibited thrombocytopenia [31].
Notably, a high neutrophil count (neutrophilia) was found in stage 1 in this investigation. This high count in stage 2 eventually returning to normal. But when the illness moved closer to the recovery/convalescent stage, this normal level count drastically decreased, resulting in neutropenia/low neutrophil count. Although no authentic data is available on this phenomenal change of neutrophils across various stages of infection, a previous study conducted on the neutrophil counts in children and adults, reported that during infection, both age groups showed neutropenia (18.8 % of children & and 29.5 % of adults), as well as neutrophilia (12.5 % children & 12 % adults) [32]. A similar study published by Jameel et al. (2012) established that dengue illness leads to low granulocyte counts. The major type of granulocytes facing this reduction are neutrophils [26]. This count reduction ultimately leads to a lowering in total leukocyte counts. Furthermore, for lymphocyte counts, this study revealed that severe lymphopenia occurs at the initial stage of infection. However, as the infection shifts to the next stage, the severity of lymphopenia is reduced. Once the recovery stage is attained, the lymphocyte counts become normal. Additionally, this study showed that stage 1 had low monocyte numbers, stage 2 had normal monocyte counts, and stage 3 had higher monocyte counts. All three phases showed similar eosinophil counts, with stage 3 showing a little rise.
A previous study, conducted by Rashmi (2015) [24] investigated the effects of dengue infection on differential WBC counts. The author analyzed the mixed cell fractions and reported an increase in differential count in more than fifty percent of patients. Further, in contrast to a couple of previous studies which reported either eosinophilia or eosinopenia [32,33] the eosinophil counts in this study did not change during the disease progression. However, as Benachinmardi et al. (2013) reported eosinophilia in a very low number of patients (six only) [32], the reason for this increase may solely not be associated with dengue as other underlying conditions i.e., allergies may also play a role in their count levels. Similarly, the Nayar et al. (2019) study is a generalist examination that would have overlooked significant dengue-associated features, even though it was carried out on many viruses and revealed eosinopenia during acute stages [33].
5.2. Gender-wise comparison
Our study findings indicated more severe leucopenia in female dengue patients compared to males. Although a significant difference in total WBCs of males and females was observed (decrease in females), the platelets and all individual immune cell counts remained “non-significant” between both genders (Fig. 3). This implies that there was no statistically significant difference in the responses of males and females patients to infection. On the other hand, it was also clear that leucopenia, anemia, and a few other characteristics were somewhat significantly associated with the female gender (Figs. 3 and 7). Even though the prevalence of dengue was greater in men, prior research has consistently shown higher incidences of severe dengue sickness in women. This is explained by the overall low WBC levels. It was noted that 69 % of females had thrombocytopenia, while 64 % of males exhibited the same condition [[34], [35], [36]]. Saha et al. (2014) demonstrated severe leucopenia in 27.9 % of females compared to 20.3 % of males [23]. Their study also showed more severe thrombocytopenia in 27.3 % of females and 41 % of males, with moderate thrombocytopenia observed in 38 % of both genders.
5.3. Age-wise comparison
We conducted a comparative analysis of immune cell counts among dengue patients of different age groups, as well as between the infected and patients with non-dengue febrile illness of corresponding age groups i.e., infected children vs normal children of the same age group, etc. (Fig. 4, Fig. 5). Among the patients of different age groups, platelet and lymphocyte counts were statistically “non-significant”. On the other hand, a “significant” leukophilia and neutrophilia was evident in adult groups, while a “significant” lowering of the count was also observed for eosinophils in the same age group (Fig. 4). As reported in early studies, excess neutrophils and leucophils could contribute in inflammatory process during their mechanism while acting against a infection causing microbe [23,35].
A comparison between immune cell counts of the infected age group and the comparable patients with non-dengue febrile illness age group revealed that WBC and platelet counts were “significantly” lower in the infected individuals (Fig. 5). Both conditions i.e., leukopenia and thrombocytopenia, showed a continuity in their severity pattern from children to adults. Leukopenia was significantly less evident in elderly patients. A previous study conducted by Khan and Hasan (2012) found that patients aged ≤15 years exhibited severe thrombocytopenia in 2.0 % of cases and moderate thrombocytopenia in 39.6 % of cases [36]. The same study reported that among patients of age range from 16 to 50 years, thrombocytopenia was severe in ∼2 % of patients while a considerable number (42.9 %) had a moderate form of this condition. When the increase in age (>50 years), though the severity of the condition percent of patients remained consistent with the above age group (∼2 %), the moderate levels of the condition increased and approximately 47 % of patients exhibited moderate thrombocytopenia. Another study, conducted to determine the levels of immune cells in patients of different age groups concluded that leucopenia was more frequent (62.5 %) in the patients grouped under12 years of age, while it decreased (37 %) in patients who were designated as “over 12 years” age group [32]. However, there was no discernible difference in thrombocytopenia amongst the aforementioned groups.
Neutrophils followed a completely different pattern. Exclusively, all age groups showed neutrophilia, though statistically at “non-significant” levels in children and elder groups. Lymphocyte reduction (lymphopenia) was also statistically at “non-significant” levels in children and aged patients but was ‘significant in the other two groups i.e., teens, and adults. A study conducted by Benachinmardi et al. (2013) showed an equal percentage of neutrophilia (12.5 %) in children and adults while the lymphopenia was 34.4 % and 59 %, respectively [32].
A significant lowering in monocyte counts was only found in aged patients while all other groups have no significant difference to their corresponding patients with non-dengue febrile illness. Finally, on statistical parameters, the eosinophil counts in all age groups remained “non-significant” when compared with their normal patients with non-dengue febrile illness age groups. These results correspond to the Benachinmardi et al. (2013) which reported that most patients had normal eosinophil counts (only 6 adults with slight eosinophilia) [32].
5.4. Future implications and limitations
In summary, both leucopenia and thrombocytopenia conditions might have extreme severity at the initial stages (1&2) of dengue infection. In addition, neutrophilia and lymphopenia might be pronounced in stage 1, but not in subsequent stages. Among gender groups, females could be more prone to severe leucopenia conditions while teens might have similar sensitivity for this condition when compared with elderly/aged patients. Neutrophilia condition appears to be more significant in the category of adults than children, while the inverse has been observed for eosinophil counts (significantly lower counts in adults than children). These findings collectively suggest that dengue patients might face severe complications during the early phases (first and second stages) of infection, which gradually subside as patients progress toward the recovery stage [37]. The findings of the study are important since such type of research is being conducted for the first time in northern region of Pakistan. This information could be useful for the healthcare providers in designing the strategies for reducing the complications of dengue infection.
Although the research analyzed the important association of age, gender and infection status with hematological parameters in dengue patients, but the data represented 204 subjects. Inclusion of more dengue sufferers is necessary to detect precise influence of these parameters on the population of a region. Furthermore, there could be other variables such as diet, race, occupation and comorbid conditions that might play a significant role in fighting the infectious diseases. Therefore, a comprehensive research involving more number of patients with detailed analysis of various confounding factors is essential for determining the accurate association of hematological parameters including atypical lymphocytes analysis in dengue infection.
A longitudinal study tracking patients over time would offer valuable insights into hematological changes, temporal patterns, and causal relationships in dengue infection. Including this aspect in future research can aid in developing more effective management and public health strategies. Further, inclusion of genetic predisposition and molecular markers to explain the gender differences in the hematological responses could provide new avenues for personalized medicine.
6. Conclusion
The study revealed that the hematological parameters might be influenced at different stages of dengue infection in a study population. The serum levels of leucocytes, thrombocytes, lymphocytes and monocytes appears to be low at early stage, and attained normality as the disease progressed. Besides, these variations were found to be prominent in female gender. The data could be useful for the healthcare providers in designing the therapeutic interventions based on the stages of the dengue infection, which might reduce the complications in patients. However, a comprehensive research involving more patients including other variables are essential for establishing exact association between hematological parameters and severity of dengue infection.
Funding
This study received financial support through the Researchers Supporting Project, under grant number RSP2024R115, at King Saud University, Riyadh, Saudi Arabia. The authors would like to express their appreciation to AlMaarefa University, Riyadh, Saudi Arabia, for their generous financial assistance in conducting this research.
Data availability
Data included in article/supp.material/referenced in the article.
CRediT authorship contribution statement
Faiza Zeb: Writing – original draft, Conceptualization. Kashif Syed Haleem: Visualization, Validation, Data curation. Mansour Almuqbil: Writing – review & editing, Resources, Methodology, Funding acquisition. Maliha Rashid: Visualization, Methodology, Formal analysis. Wajid Hussain: Writing – original draft, Visualization, Project administration. Farhana Maqbool: Writing – original draft, Validation, Software. Isfahan Tauseef: Visualization, Methodology, Conceptualization. Laila Jafri: Visualization, Validation, Conceptualization. Basheerahmed Abdulaziz Mannasaheb: Writing – review & editing, Software, Resources. Syed Arif Hussain: Visualization, Resources, Conceptualization. Mohammed Sharique Ahmed Quadri: Visualization, Project administration, Methodology, Investigation. Amro Mohammed Sawadi Khormi: Writing – original draft, Visualization, Conceptualization. Syed Mohammed Basheeruddin Asdaq: Writing – review & editing, Supervision, Data curation, Conceptualization.
Declaration of competing interest
Nil.
Acknowledgement
The authors wish to extend their gratitude to King Saud University, Riyadh, Saudi Arabia, for the support provided under the Researchers Supporting Project with grant number RSP2024R115. We also acknowledge the kind financial support from AlMaarefa University, Riyadh, Saudi Arabia, which was instrumental in enabling this research.
Contributor Information
Faiza Zeb, Email: isfi_qau@yahoo.com.
Kashif Syed Haleem, Email: Kashifhaleem18@outlook.com.
Mansour Almuqbil, Email: mmetwazi@ksu.edu.sa.
Maliha Rashid, Email: maliharashid78@gmail.com.
Wajid Hussain, Email: wajidhust.edu@yahoo.com.
Farhana Maqbool, Email: fairy_es11@yahoo.com.
Isfahan Tauseef, Email: isfahan@hu.edu.pk.
Basheerahmed Abdulaziz Mannasaheb, Email: bmannasaheb@mcst.edu.sa.
Syed Arif Hussain, Email: pulmoarif@gmail.com.
Mohammed Sharique Ahmed Quadri, Email: mquadri@mcst.edu.sa.
Amro Mohammed Sawadi Khormi, Email: Amr1411@hotmail.com.
Syed Mohammed Basheeruddin Asdaq, Email: sasdag@um.edu.sa, sasdaq@gmail.com.
References
- 1.Park K.B., Patnaik H.H., Kim T.-Y., Jo Y.H., Kim N.-Y., Yang S.-C., Lee W.-G., Lee H.-I., Cho S.-H., Han Y.S. Current trends in large-scale viral surveillance methods in mosquitoes. Entomol. Res. 2020;50:292–308. [Google Scholar]
- 2.Atif M., Raheel U., Alam F., Arshad H., Baloch F. Serotyping of dengue virus from deadly outbreaks of Pakistan. J Hum Virol Retrovirol. 2016;3:92–96. [Google Scholar]
- 3.Jing Q., Wang M. Dengue epidemiology. Global Health Journal. 2019;3:37–45. [Google Scholar]
- 4.Wang W.-H., Urbina A.N., Chang M.R., Assavalapsakul W., Lu P.-L., Chen Y.-H., Wang S.-F. Dengue hemorrhagic fever – a systemic literature review of current perspectives on pathogenesis, prevention and control. J. Microbiol. Immunol. Infect. 2020 doi: 10.1016/j.jmii.2020.03.007. [DOI] [PubMed] [Google Scholar]
- 5.WORLD HEALTH ORGANIZATION . new edition. 2019. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control. [PubMed] [Google Scholar]
- 6.Guzman M.G., Halstead S.B., Artsob H., Buchy P., Farrar J., Gubler D.J., Hunsperger E., Kroeger A., Margolis H.S., Martínez E., Nathan M.B., Pelegrino J.L., Simmons C., Yoksan S., Peeling R.W. Dengue: a continuing global threat. Nat. Rev. Microbiol. 2010;8:S7–S16. doi: 10.1038/nrmicro2460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jahan F. Dengue fever (DF) in Pakistan. Asia Pac. Fam. Med. 2011;10:1. doi: 10.1186/1447-056X-10-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zameer S., Soomro S., Baig S., Rubeen R., Sharafat S. Seasonal variation and Seroprevalence of dengue fever in Karachi, Pakistan. Journal of Muhammad Medical College, Mirpurkhas. 2017;8:12–15. [Google Scholar]
- 9.Rafique I., Saqib M.A.N., Munir M.A., Qureshi H., Taseer I.-U.-H., Iqbal R., Ahmed W., Akhtar T., Rizwanullah Asymptomatic dengue infection in adults of major cities of Pakistan. Asian Pac. J. Tropical Med. 2017;10:1002–1006. doi: 10.1016/j.apjtm.2017.09.013. [DOI] [PubMed] [Google Scholar]
- 10.Suleman M., Faryal R., Aamir U.B., Alam M.M., Nisar N., Sharif S., Shaukat S., Khurshid A., Angez M., Umair M., Mujtaba G., Sufian M.M., Arshad Y., Rehman L., Zaidi S.S.Z. Dengue outbreak in Swat and Mansehra, Pakistan 2013: an epidemiological and diagnostic perspective. Asian Pac. J. Tropical Med. 2016;9:380–384. doi: 10.1016/j.apjtm.2016.03.010. [DOI] [PubMed] [Google Scholar]
- 11.Sekaran S.D., Ew C.L., Kantesh B., Appana R., Subramaniam G. Evaluation of a dengue NS1 capture ELISA assay for the rapid detection of dengue. J Infect Developing Countries. 2007;1:182–188. [Google Scholar]
- 12.Kulkarni R., Modak M., Gosavi M., Wani D., Mishra A.C., Arankalle V.A. Comparative assessment of commercial enzyme-linked immunosorbent assay & rapid diagnostic tests used for dengue diagnosis in India. Indian J. Med. Res. 2020;151:71. doi: 10.4103/ijmr.IJMR_613_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mcbride W.J.H. Evaluation of dengue NS1 test kits for the diagnosis of dengue fever. Diagn. Microbiol. Infect. Dis. 2009;64:31–36. doi: 10.1016/j.diagmicrobio.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 14.Kumarasamy V., Wahab A.A., Chua S., Hassan Z., Mohamad M., Chua K. Evaluation of a commercial dengue NS1 antigen-capture ELISA for laboratory diagnosis of acute dengue virus infection. J. Virol Methods. 2007;140:75–79. doi: 10.1016/j.jviromet.2006.11.001. [DOI] [PubMed] [Google Scholar]
- 15.WORLD HEALTH ORGANIZATION . Geneva: WHO; 1997. A Comprehensive Report of Dengue Activity in Kolkata, India–a Four-Year Profile A Comprehensive Report of Dengue Activity in Kolkata, India–a Four-Year Profile. 2012. Dengue hemorrhagic fever: diagnosis, treatment, prevention and control. [Google Scholar]
- 16.Putnak J.R., Kanesa-Thasan N., Innis B.L. A putative cellular receptor for dengue viruses. Nat. Med. 1997;3:828–829. doi: 10.1038/nm0897-828. [DOI] [PubMed] [Google Scholar]
- 17.Chaloemwong J., Tantiworawit A., Rattanathammethee T., Hantrakool S., Chai-Adisaksopha C., Rattarittamrong E., Norasetthada L. Useful clinical features and hematological parameters for the diagnosis of dengue infection in patients with acute febrile illness: a retrospective study. BMC hematology. 2018;18:1–10. doi: 10.1186/s12878-018-0116-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ageep A.K., Malik A.A., Elkarsani M.S. Clinical presentations and laboratory findings in suspected cases of dengue virus. Saudi Med. J. 2006;27:1711. [PubMed] [Google Scholar]
- 19.Phuong C.X.T., Nhan N.T., Kneen R., Thuy P.T.T., Van Thien C., Nga N.T.T., Thuy T.T., Solomon T., Stepniewska K., Wills B. Clinical diagnosis and assessment of severity of confirmed dengue infections in Vietnamese children: is the world health organization classification system helpful? Am. J. Trop. Med. Hyg. 2004;70:172–179. [PubMed] [Google Scholar]
- 20.Ganeshkumar P., Murhekar M.V., Poornima V., Saravanakumar V., Sukumaran K., Anandaselvasankar A., John D., Mehendale S.M. Dengue infection in India: a systematic review and meta-analysis. PLoS Neglected Trop. Dis. 2018;12 doi: 10.1371/journal.pntd.0006618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wichmann O., Stark K., Shu P.-Y., Niedrig M., Frank C., Huang J.-H., Jelinek T. Clinical features and pitfalls in the laboratory diagnosis of dengue in travellers. BMC Infect. Dis. 2006;6:120. doi: 10.1186/1471-2334-6-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Butt N., Abbassi A., Munir S., Ahmad S.M., Sheikh Q.H. Haematological and biochemical indicators for the early diagnosis of dengue viral infection. J Coll Physicians Surg Pak. 2008;18:282–285. [PubMed] [Google Scholar]
- 23.Saha A.K., Chatterjee G., Hazra S.C. Clinicohematological profiles of hospitalized patients with dengue in Kolkata in 2012 epidemic, West Bengal. Iran. J. Med. Sci. 2014;39:471. [PMC free article] [PubMed] [Google Scholar]
- 24.Rashmi M. Haematological and biochemical markers as predictors of dengue infection. Malays. J. Pathol. 2015;37:247. [PubMed] [Google Scholar]
- 25.Hay S.I., Cox J., Rogers D.J., Randolph S.E., Stern D.I., Shanks G.D., Myers M.F., Snow R.W. Climate change and the resurgence of malaria in the East African highlands. Nature. 2002;415:905–909. doi: 10.1038/415905a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rigau-Pérez J.G. The early use of break-bone fever (Quebranta huesos, 1771) and dengue (1801) in Spanish. Am. J. Trop. Med. Hyg. 1998;59:272–274. doi: 10.4269/ajtmh.1998.59.272. [DOI] [PubMed] [Google Scholar]
- 27.De Mel S., Thilakawardana B.U., De Mel P., Clarice C.S.H., Shalindi M., De Mel C., Chandrasena L., Yip C., Yap E.S., Seneviratne S.L., Abeysuriya V. Triple positivity for nonstructural antigen 1, immunoglobulin M and immunoglobulin G is predictive of severe thrombocytopaenia related to dengue infection. J. Clin. Virol. 2020;129 doi: 10.1016/j.jcv.2020.104509. [DOI] [PubMed] [Google Scholar]
- 28.Mehta K.D., Ghediya B., Sheth S., Khandhediya S., Shingala H., Sinha M. Study of correlation between platelet count and serological markers of dengue infection with importance of NS1 Antigen in Western Region of India. Natl. J. Lab. Med. 2016;5:55–59. [Google Scholar]
- 29.Tathe S., Chincholkar V., Kulkarni D., Nilekar S., Ovhal R., Halgarkar C. A study of NS1 antigen and platelet count for early diagnosis of dengue infection. Int. J. Curr. Microbiol. App. Sci. 2013;2:40–44. [Google Scholar]
- 30.Dhir G., Dhir T., Suri V., Dhir D., Khatri K. Hematological and serological test profile in dengue. Dengue Hemorrhagic Fever and Dengue Shock Syndrome in Bathinda Region. 2015 [Google Scholar]
- 31.Kulkarni R., Patil S., Ajantha G., Upadhya A., Kalabhavi A., Shubhada R., Shetty P., Jain P. Association of platelet count and serological markers of dengue infection-importance of NS1 antigen. Indian J. Med. Microbiol. 2011;29:359. doi: 10.4103/0255-0857.90159. [DOI] [PubMed] [Google Scholar]
- 32.Benachinmardi K.K., Panduranga C., Srinivasamurthy V., Burugina S.N., Vani B., Navaneeth B. Haematological profile in acute dengue infection: a study at tertiary care teaching hospital. J. Pharmaceut. Biomed. Sci. 2013;36:1866–1870. [Google Scholar]
- 33.Nayar S., Sriranjani B., Shanmugam P. 2019. Haematological Predictors of Recovery in Dengue Cases. [Google Scholar]
- 34.Kabra S., Jain Y., Pandey R., Singhal T., Tripathi P., Broor S., Seth P., Seth V. Dengue haemorrhagic fever in children in the 1996 Delhi epidemic. Trans. Roy. Soc. Trop. Med. Hyg. 1999;93:294–298. doi: 10.1016/s0035-9203(99)90027-5. [DOI] [PubMed] [Google Scholar]
- 35.Shekhar K.C., Huat O.L. Epidemiology of dengue/dengue hemorrhagic fever in Malaysia—a retrospective epidemiological study 1973-1987. Part I: dengue hemorrhagic fever (DHF) Asia Pac. J. Publ. Health. 1992;6:15–25. doi: 10.1177/101053959300600203. [DOI] [PubMed] [Google Scholar]
- 36.Khan E., Hasan R. vol. 26. Journal of Postgraduate Medical Institute (Peshawar-Pakistan); 2012. (Dengue Infection in Asia; a Regional Concern). [Google Scholar]
- 37.Jameel T., Mehmood K., Mujtaba G., Choudhry N., Afzal N., Paul R.F. Changing haematological parameters in dengue viral infections. J. Ayub Med. Coll. Abbottabad. 2012;24:3–6. [35] KHAN, E. & HASAN, R. 2012. Dengue infection in Asia; a regional concern. Journal of Postgraduate Medical Institute (Peshawar-Pakistan), 26. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data included in article/supp.material/referenced in the article.