Abstract
Wastewater-based epidemiology (WBE) and wastewater surveillance have become a valuable complementary data source to collect information on community-wide exposure through the measurement of human biomarkers in influent wastewater (IWW). In WBE, normalization of data with the de facto population that corresponds to a wastewater sample is crucial for a correct interpretation of spatio-temporal trends in exposure and consumption patterns. However, knowledge gaps remain in identifying and validating suitable de facto population biomarkers (PBs) for refinement of WBE backestimations.
WBE studies that apply de facto PBs (including hydrochemical parameters, utility consumption data sources, endo- and exogenous chemicals, biological biomarkers and signalling records) for relative trend analysis and absolute population size estimation were systematically reviewed from three databases (PubMed, Web of Science, SCOPUS) according to the PRISMA guidelines. We included in this review 81 publications that accounted for daily variations in population sizes by applying de facto population normalization.
To date, a wide range of PBs have been proposed for de facto population normalization, complicating the comparability of normalized measurements across WBE studies. Additionally, the validation of potential PBs is complicated by the absence of an ideal external validator, magnifying the overall uncertainty for population normalization in WBE. Therefore, this review proposes a conceptual tier-based cross-validation approach for identifying and validating de facto PBs to guide their integration for i) relative trend analysis, and ii) absolute population size estimation. Furthermore, this review also provides a detailed evaluation of the uncertainty observed when comparing different de jure and de facto population estimation approaches. This study shows that their percentual differences can range up to ±200%, with some exceptions showing even larger variations.
This review underscores the need for collaboration among WBE researchers to further streamline the application of de facto population normalization and to evaluate the robustness of different PBs in different socio-demographic communities.
Keywords: De facto population, Population biomarker, Dynamic population normalization, Wastewater-based surveillance, Uncertainty analysis
Graphical abstract

1. Introduction
Wastewater-based epidemiology (WBE) is a multidisciplinary tool for the evaluation of population lifestyle habits and health-related aspects, such as disease spread and exposure to chemicals (1,2). The data extracted from wastewater analysis can be used as complementary information source to traditional approaches commonly used to measure human exposure (e.g., surveys, clinical data). Moreover, the COVID-19 pandemic has demonstrated its potential for surveillance of infectious diseases and significantly accelerated WBE as a surveillance method. WBE comprises the measurement of human biomarkers concentrations (i.e., metabolic excretion products including parent compounds and metabolites) in influent wastewater (IWW) to investigate human exposure to and/or consumption of different chemicals and pathogens at a population level.
Within WBE, measured concentrations in IWW (from ng/L to μg/L range or gene copies/L) of biomarkers are converted to population-normalized mass loads (PNML, expressed in mg/day/1000 inhabitants or copies/day/1000 inhabitants) by multiplying with the daily wastewater flow rate (L/day) and normalizing this mass load with the population size present in the wastewater treatment plant (WWTP) catchment area at that corresponding day (3). This population normalization enables the comparison of exposure/consumption patterns across different locations (both at a national and international level) and different time points and, therefore, PNMLs serve as a proxy for exposure/consumption at a population level. However, WBE has some intrinsic uncertainties due to the different parameters involved in the back-calculation of PNMLs, among which the estimation of the accurate population size is one of the major uncertainties due to the dynamic character of a WWTP catchment area (4).
Most IWW samples are collected over a 24-h period with frequent sampling intervals, making it necessary to estimate the number of people present within the catchment area over the same timeframe. Until now, most WBE applications that investigated spatio-temporal patterns have used fixed (de jure) population numbers in their back-calculations (5–8). In most cases, residential population numbers (i.e., number of people living within the boundaries of the WWTP catchment area) are provided by the wastewater treatment facilities and can be estimated in different ways, such as the census population of the whole city, households connected to the WWTP area, the total wastewater flow rate and/or design capacity of the WWTP (9). With the design capacity of the WWTP, it is difficult to assess if less people contributed to the wastewater or if the WWTP operated above design capacity to receive wastewater from more people than expected (10). Moreover, this number is often chosen to account for future population increases.
Therefore, several attempts have been made to refine census data. For example, Tscharke et al. applied an alternative approach which overlayed the WWTP catchment population against available geo-referenced census population data in geospatial software (9). The major advantage of the census approach is that WWTP catchment areas can be assessed in terms of socio-demographics, including age, income, and education level. A limitation is that it only captures a static snapshot of the population and usually only acquires a specific portion of the entire population covered by the WWTP (10,11). The WWTP and administrative boundaries may also vary. Additionally, the frequency that national census is conducted may vary substantially across different countries, and significant gaps between successive census takings can occur. The de jure population may not always be appropriate to normalize mass loads (ML) because population estimates may be outdated and transient changes (e.g., births, deaths, resident changes) are not fully considered. It is also likely that administrative regions for de jure populations do not completely coincide with the geographical area of a WWTP. Nevertheless, these models could support the calibration of other population estimation methodologies.
Since de jure population numbers rely on the fact that the population size remains constant over time, it is impossible to verify if observed fluctuations in PNML are due to variations in population size (e.g., work-travel commuting, week-weekend differences, seasonal changes) or changes in consumption/exposure or a combination of both. Castiglioni et al. and Thomas et al. estimated that the use of de jure population numbers contributed to up to 55% of the overall uncertainty associated with WBE (4,12), but an updated uncertainty analysis is needed. This is especially challenging with significant socio-economic disruptions, such as observed during the COVID-19 pandemic (13–15). Hence, it would be more appropriate to use dynamic (de facto) population numbers to account for the dynamics of people in and out the catchment area. For WBE investigations, de facto population is considered more suitable for standardization as it represents more closely the actual population of a WWTP catchment area at a specific time (16). As demonstrated by Thomaidis et al. and Boogaerts et al., relative changes in population size could heavily impact the socio-demographic composition of the population served by the WWTP (14,17). The use of the de facto population in back-calculations can significantly reduce the uncertainty associated with population normalization in WBE and, therefore, it is necessary to identify and validate different approaches to establish the de facto population size. This is especially important considering the major increase in the number of WBE investigations that report per capita data using de jure population (9).
Nowadays, different approaches for the estimation of the de facto population have been proposed, utilizing various population biomarkers (PBs) (14,18–20). The inherent diversity in methodologies leads to discrepancies among these approaches. Consequently, when comparing results derived from different population estimation methods, the margin of error is magnified. Validating new dynamic PBs is challenging with the absence of a golden standard. The use of an external validator with intrinsic uncertainties can sometimes yield artificially high correlations when employing classic correlation approaches (e.g., Pearson or Spearman), potentially resulting in incorrect conclusions about the biomarker’s applicability.
This review documents the current application of de facto population normalization to refine WBE back-calculations. A review of the analytical methods to detect and quantify PBs in IWW is not within the scope of this review and is given elsewhere (21). The primary objectives of this study are:
-
(i)
to evaluate the different approaches to account for relative changes in WWTP population size,
-
(ii)
to give insight in the research required for the application of de facto population normalization in WBE, considering feasibility by proposing a tier-based approach,
-
(iii)
to perform an uncertainty analysis for absolute population size estimation based on different de facto population markers.
2. Literature search and eligibility criteria
This literature review was done in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines from 2020 (22). These PRISMA guidelines enable an evidence-based approach for reporting in systematic reviews and meta-analyses and was applied to ensure the transparency of the literature search. Different electronic search engines were used: PubMed, Web of Science, and SCOPUS. These engines were queried as illustrated in Table S1. The search strategies combined different search terms and/or keywords regarding: (i) wastewater or wastewater-based epidemiology, and (ii) population markers, population estimation or population dynamics. The initial screening based on abstract and title subjects was executed by two investigators, prior to a full-text screening against the inclusion and exclusion criteria. Conflicts with eligibility conditions were removed after mutual consent. Citation tracking and searches in author’s bibliographies were conducted to track down additional references.
We included (i) WBE studies that applied dynamic population estimates for the normalization of PNMLs and (ii) WBE studies that investigated daily variations in population sizes. Studies that only applied static population estimates were excluded.
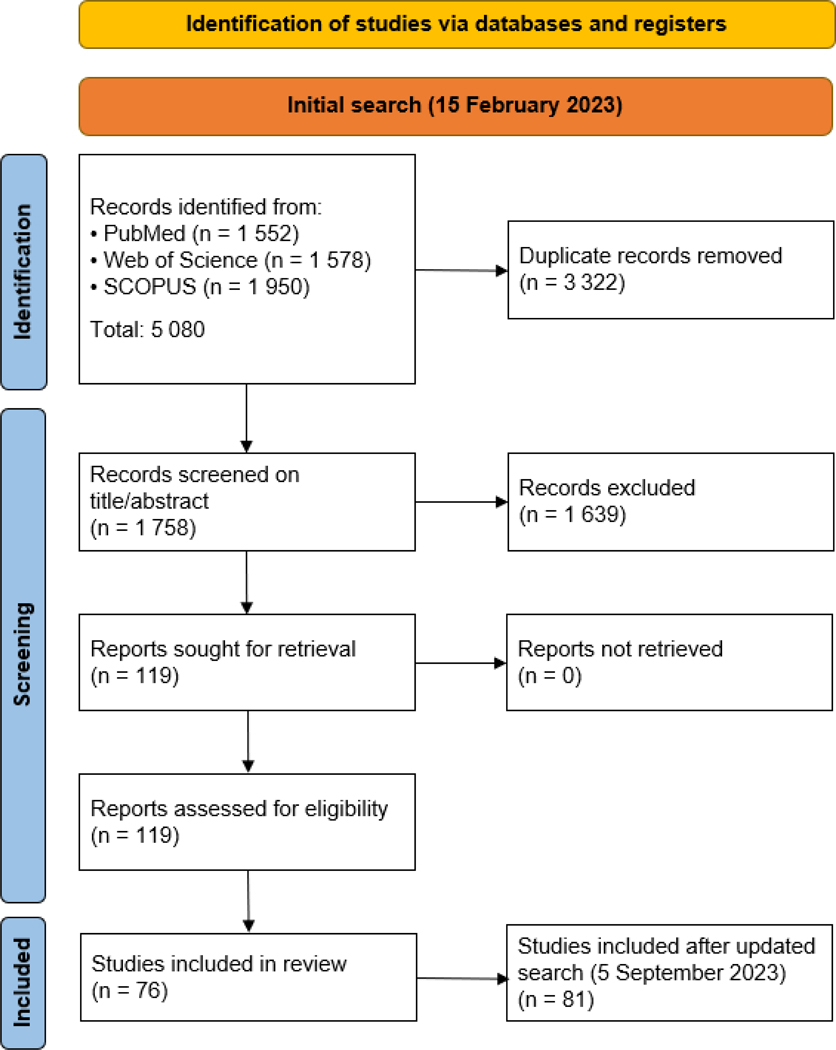
An initial search was performed on February 15, 2023 and updated on September 5, 2023 with 5 newly found records. A total of 5080 records were retrieved after executing the search strategies (Table S1). All citations were imported in the reference manager Zotero, and duplicates were manually removed (N = 3322) with the integrated deduplication tool. After deduplication, 1758 entries were screened on title and abstract content from which another 1639 records were excluded. After full-text screening against the eligibility criteria, a total of 81 citations were included in this systematic literature review, published between 2012 and 2023 as illustrated in Figure S1. Figure 1 summarizes the results of this systematic search as a PRISMA flow diagram. Table S2 summarizes all currently available WBE applications that apply de facto population proxies for the calculation of PNMLs.
Figure 1:
Preferred reporting items for systematic reviews and meta-analyses flow diagram
3. Current state on the application of de facto population proxies in WBE
At this point, the majority of studies applied a limited selection of potential PBs in specific WWTPs without testing their capability to capture population changes or only evaluated their use with small sample sizes (i.e., only one datapoint per WWTP site) (23–25). While a large geographical spread was covered in some WBE studies (25–27), only a handful of studies explored large temporal datasets (10,13,14,28). The low temporal diversity might also be reflected in the coefficients of determination (r2) reported in this section. Therefore, there is a need to investigate the application of dynamic population proxies by using larger temporal datasets. Caution is advised in interpreting coefficients of determination, as there is no ideal calibrator available for validating the de facto population. Consequently, the intrinsic uncertainty of the external validator could result in artificially high correlations. Coefficients of determination are only reported in case of statistical significance (p < 0.05). Table 1 gives a summary of the advantages and disadvantages of the different dynamic population proxies.
Table 1:
Overview of strengths and limitations of different types of dynamic population data
| Dynamic population proxy | Strengths | Limitations |
|---|---|---|
| Hydrochemical parameters (e.g., chemical oxygen demand) | • Routinely measured at the WWTP (no extra analysis required) • Time-efficient and low-cost methods |
• Can originate from non-human sources (e.g., industrial discharge) |
| Utility consumption (electricity and drinking water) | • Used by all members of society • Time-efficient and low-cost methods |
• Subject to large seasonal fluctuations • Not applicable in areas with large industrial activities • Large inter-individual variability in consumption • Usage not directly linked to discharge to IWW (electricity) |
| Exogenous chemical biomarkers | • Measured within the same sample as biomarkers of interest • Metabolites of exogenous compounds are specific for human excretion |
• Affected by cultural and dietary habits • Subject to seasonal fluctuations • Parent compounds not always specific for human excretion • Some of these biomarkers may possess low-moderate in-sewer and/or in-sample stability • Development of analytical methods can be challenging • Different sample handling, instrumentation and storage conditions might be required as for biological biomarkers |
| Endogenous chemical biomarkers | • Measured within the same sample as biomarkers of interest • Specific for human excretion |
• Some of these biomarkers may possess low-moderate in-sewer and/or in-sample stability • Development of analytical methods can be challenging • Different sample handling, instrumentation and storage conditions might be required as for biological biomarkers • Applicability of these population biomarkers may be site specific due to inter-individual differences |
| Biological biomarkers | • Measured within the same sample as biomarkers of interest | • Some of these biomarkers may possess low-moderate in-sewer and/or in-sample stability • Different sample handling, instrumentation and storage conditions might be required as for chemical biomarkers • Applicability of these population biomarkers may be site specific (e.g., PMMoV influenced by pepper-based dietary habits) • Not always specific for human excretion |
| Signalling records (e.g., mobile phone data) | • High frequency measurements • Mobile devices used by the majority of global population • Time-efficient methods |
• Uncertainty related to extrapolation parameters (e.g., devices per person, market share, zone probability) • Not always representative for all population demographics (e.g., coverage older people) • High cost • Not always possible to isolate data from specific regions • Usage not directly linked to discharge to IWW |
3.1. Hydrochemical parameters
Wastewater flow rate, chemical oxygen demand (COD), biochemical oxygen demand (BOD), ammonia (NH3), ammonium (NH4-N), total nitrogen (TN), total Kjeldahl Nitrogen (TKN) total phosphorus (TP), total suspended solids (TSS) and volatile suspended solids (VSS) in IWW are hydrochemical parameters routinely measured by WWTP facilities for various purposes, such as measuring water treatment efficiency. Unlike other parameters, such as endogenous biomarkers mentioned in section 3.3, these biomarkers are not unique to human activity or metabolism. Other factors, such as industrial discharge, agricultural activities and biodegradable substances can also influence these hydrochemical parameters (29). However, given the ease of obtaining this data, they hold potential both in terms of feasibility and cost-effectiveness for use in de facto population estimation. An overview of the different studies that employed hydrochemical parameters as dynamic population proxy can be found in Table 2.
Table 2:
Overview of different studies that employed hydrochemical parameters as dynamic population proxy
| PB | Daily per capita emission estimation | Coefficient of determination [external validator] | Reference |
|---|---|---|---|
| Wastewater flow | 251 L/day/inhabitant | Not applicable | Zheng et al.2019 (30) |
| 251 L/day/inhabitant | Not applicable | Wang et al.2021 (49) | |
| 300 L/day/inhabitant | Not applicable | Verhagen et al. 2023 (50) | |
| 300 L/day/inhabitant | Not applicable | Tscharke et al. 2019 (9) | |
| 167 L/day/inhabitant | r2 = 0.93 [census data] | Hou et al. 2021 (29) | |
| Chemical oxygen demand (COD) | 110 g/day/inhabitant (51) | Not applicable | Di Marcantonio et al. 2022 (35) |
| 136 g/day/inhabitant (11,52,53) | 640 000 +/− 98 000 people vs 600 000 from census location 1 and 1 700 000 +/− 1250 000 vs. 2 500 000 from census | Bijlsma et al. 2016 (37) | |
| 120 g/day/inhabitant | Not applicable | Schenk et al. 2023 (54) | |
| 128 g/day/inhabitant | No reliable correlation with BOD, TN, TP | Rico et al. 2017 (19) | |
| 43.9 g/day/inhabitant | r2COD = 0.87 [cotinine mass load] | Zheng et al. 2019 (30) | |
| 43.9 g/day/inhabitant (55) | Not Applicable | Song et al. 2020 (42) | |
| 43.9 g/day/inhabitant (55) | Not Applicable | Zhang et al. 2019 (41) | |
| Biological oxygen demand (BOD) | 5.9 g/day/inhabitant | RSD 37% (n=7) [census data] | Croft et al. 2020 (38) |
| Not applicable | r2BOD = 0.94 [census data], wet weather and r2TSS = 0.96 [census data], dry weather | Hoar et al. 2014 (46) | |
| 60 g/day/inhabitant (11,52,53) | 730 000 +/− 120 000 people vs 600 000 from census location 1 and 1 850 000 +/− 180 000 vs. 2 500 000 from census | Bijlsma et al. 2016 (37) | |
| Not applicable | r2BOD = 0.96 [census data] | Li et al. 2022 (45) | |
| 50.7 g/day/inhabitant | Population estimates differed from other values (e.g., mobile phone data) and therefore assumed as high uncertainty | Sim et al. 2023 (47) | |
| 60 g/day/inhabitant | No reliable correlation with COD, TN, TP | Rico et al. 2017 (19) | |
| Ammonia (NH3) | Not applicable | r2NH3 = 0.91 [census data], both for wet and dry weather | Hoar et al. 2014 (46) |
| Ammonium (NH4-N) | Not applicable | Not specified but strong correlation with BE and THC-COOH | Been et al. 2014 (20) |
| Not applicable | Not specified, but viral loads adjusted for ammonia were consistent with trends for 3 out of 4 pandemic waves | Xie et al. 2022 (23) | |
| 9.5 g/day/inhabitant | r2NH4N = 0.91 [census data] | Zhang et al. 2019 (41) | |
| 8.1 g/day/inhabitant | Not applicable | Benaglia et al. 2020 (56) | |
| 6 g/day/inhabitant (55) | Not applicable | Shao et al. 2021 (57) | |
| 6 g/day/inhabitant (55) | Not applicable | Wang et al. 2019 (31) | |
| 6 g/day/inhabitant, considering daily variation and lower industry input surveyed from the WWTP operators | r2NH4N = 0.99 [WWTP expert judgement population] | Zheng et al. 2017 (55) | |
| 8.6 g/day/inhabitant, mean of 6.5 and 10.7 | Not applicable | Aberi et al. 2021 (58) | |
| 6 g/day/inhabitant (55) | r2NH4N = 0.42 [census data of all WWTPs], r2NH4N = 0.68 [census data of all WWTPs with WWTPS that process industrial wastewater removed] | Cong et al. 2021 (43) | |
| 6.9 g/day/inhabitant (20) | not mentioned, but average population for both investigated catchment areas determined using ammoniacal nitrogen load were 16% lower and 3% higher. | Bowers et al. 2021 (44) | |
| 6 g/day/inhabitant (55) | Not applicable | Saththasivam et al. 2021 (59) | |
| 6.9 g/day/inhabitant (20) | Not specified | O’Rourke et al. 2020 (60) | |
| 6.9 g/day/inhabitant (20) | RSD 18% (n=7) [census data] | Croft et al. 2020 (38) | |
| 8 g/day/inhabitant | Not applicable | Schenk et al. 2023 (54) | |
| 6 g/day/inhabitant | Not applicable | Zheng et al. 2019 (30) | |
| 6 g/day/inhabitant (55) | r2Population model = 0.91 [census data], r2NH4N = 0.88 [cotinine mass load] | Song et al. 2020 (42) | |
| 6 g/day/inhabitant (55) | Not applicable | Zhang et al. 2019 (41) | |
| Total nitrogen (TN) and total Kjeldahl Nitrogen (TKN) | 12.6 – 13.4 g/day/inhabitant (TKN) | r2TN = 0.89 [census data], wet weather and r2TN = 0.91 [census data], dry weather | Hoar et al. 2014 (46) |
| 12 g/day/inhabitant (TN) | Not applicable | Schenk et al. 2023 (54) | |
| Not applicable (TKN) | r2TN = 0.98 [census data] | Li et al. 2022 (45) | |
| 10.6 g/day/inhabitant (TN) | Population estimates differed from other values (e.g., mobile phone data) and therefore assumed as high uncertainty | Sim et al. 2023 (47) | |
| 12.5 – 14 g/day/inhabitant (n=4) (TN) | No improvement in the strength of the relationship between clinical case numbers and COVID-19 in IWW | Isaksson et al. 2022 (15) | |
| 8.5 g/day/inhabitant (TN) | r2 = 0.79 [census data] | Yaniv et al. 2021 (48) | |
| 10 g/day/inhabitant (TN) | No reliable correlation with BOD, COD,TP | Rico et al. 2017 (19) | |
| Total phosphorus (TP) | 1.7 g/day/inhabitant (40) | RSD 21% (n=7) [census data] | Croft et al. 2020 (38) |
| 1.2 g/day/inhabitant | Population estimates differed from other values (e.g., mobile phone data) and therefore assumed as high uncertainty | Sim et al. 2023 (47) | |
| 1.2–2.1 g/day/inhabitant (n=4) | No improvement in the strength of the relationship between clinical case numbers and COVID-19 in IWW | Isaksson et al. 2022 (15) | |
| 1.7 g/day/inhabitant | No reliable correlation with BOD, COD, TN | Rico et al. 2017 (19) | |
| 0.8 g/day/inhabitant (55) | r2TP = 0.79 [cotinine mass load] | Zheng et al. 2019 (30) | |
| 0.8 g/day/inhabitant (55) | Not Applicable | Song et al. 2020 (42) | |
| 0.8 g/day/inhabitant (55) | Not Applicable | Zhang et al. 2019 (41) | |
| Total suspended solids (TSS) | Not applicable | r2TSS = 0.78 [census data], wet weather and r2TSS = 0.71 [census data], dry weather | Hoar et al. 2014 (46) |
| Not applicable | r2TSS = 0.83 [census data] | Li et al. 2022 (45) | |
| Volatile suspended solids (VSS) | Not applicable | r2VSS = 0.85 [census data] | Li et al. 2022 (45) |
3.1.1. Wastewater flow rate
Zheng et al. and Wang et al. employed methods to calculate real-time population estimates based on wastewater flow rate and daily per capita wastewater discharge rates. Discharge rates were estimated at an average of 251 ± 18 L per day per inhabitant. These estimates were derived from daily tap water usage data and the de jure population (30,31). Nevertheless, both wastewater flow and discharge rate are inherently linked to temporal, geographical and cultural differences. Hou et al. correlated population estimated based on wastewater flow rate with the census population and found a high correlation (r2 = 0.93) (29). However, they observed large variations between the absolute estimates of both population types (see Section 4.2.) (29,32).
3.1.2. Chemical and biological oxygen demand
COD, the amount of oxygen that is needed for the oxidation of organic matter present in IWW and BOD, the oxygen amount consumed by micro-organisms to decompose organic matter, could be a useful population biomarker since they represent organic matter produced by each individual contributor of a WWTP (33,34). Multiple WBE studies have applied COD and BOD measurements as possible PBs since they are easy to measure (13,19,35–38). Some studies even performed a comparative analysis between different hydrochemical parameters including COD, BOD, NH4-N, TN and TP for population normalization (13,19,38), but no consensus was found regarding the most suitable standardization. For example, some studies found a good agreement between census-based population numbers compared to COD and BOD measurements in non-industrial locations (19,36,37,39), while substantial disparity between these estimates was found in industrial sites (13,19,36). Other WBE studies found that COD-derived values tended to overestimate the population size (9,26,40), but considered this bias to be acceptable. These findings indicate that these parameters are not applicable in every location, but that the use of COD as PB is limited to catchment areas that receive primarily wastewater from residential areas. Additionally, BOD could by biased by the presence of substances that might cause the inhibition of the oxidizing bacteria (33).
3.1.3. Ammonia and ammonium
Both NH3 and NH4-N can be measured in IWW as indirect markers of urine, primarily originating from the hydrolysis of urea. NH4-N has been proposed as a more reliable population marker compared to conventional hydrochemical parameters (e.g., BOD, COD, TN and TP) because it is believed to be less influenced by non-human sources (20,41–47). Different studies showed that NH4-N concentrations were sensitive to weekly and seasonal population fluctuations resulting from commuting patterns (e.g., tourism) (20,31). Moreover, Been et al. assumed that non-human sources such as biodegradable substances, agricultural activities, industrial discharge should remain constant over time (20,29), but no detailed investigation was provided. For example, seasonal changes could occur due to differences in industrial and agricultural activities. Additionally, different studies have compared population normalization based on NH4-N with static population figures (41–43), and discrepancies were found. For example, Zhang et al. and Song et al. found good correlations between de facto population based on NH4-N and the census population (r2 = 0.91 in both studies) (41,42), while Cong et al. only found only a moderate correlation (r2 = 0.41) (43). In the latter study, the correlation coefficient improved after removing WWTPs that processed industrial wastewater (r2 = 0.68). They suggested that NH4-N was a suitable PB, but only if domestic wastewater production accounted for ≥ 90% of the total wastewater load. In addition, Zheng et al. also found a relationship (r2 = 0.99) between the NH4-N corrected population and the estimates based on the design capacity of the WWTP (30). However, it is crucial to note that the majority of these studies employ only a single data point per WWTP for comparison with census data, whereas multiple data points per WWTP are needed to make the correlation more robust. Furthermore, Bowers and Subedi et al. and Zhang et al. found that the NH4-N adjusted population size were higher compared to census data (41,44), potentially due to the presence of commuters, tourists and other people not counted by the census.
3.1.4. Other hydrochemical parameters
Some WBE studies conducted a comparative analysis between different hydrochemical parameters to test their applicability as population markers. However, no consensus was found between these different studies, indicating that the utility of these measurements should be assessed in each individual WWTP catchment area. In addition to the aforementioned parameters, TN, TKN, TP, TSS and VSS were also examined. For example, Li et al. reported good correlations between TKN-, BOD-, TSS-, and VSS-derived population sizes and census data (r2 > 0.69), with the highest correlation coefficient reported for TKN (r2 = 0.96) (45). However, Hoar et al. found significant differences between the census-based populations and the NH3-, TKN-, TSS-, and BOD-derived population estimates, particularly during wet weather conditions (46). These disparities were relatively minor in the case of ammonium-based nitrogen, which was in line with the observations from Croft et al. (38). Similarly, Sim et al. found poor correlations between the BOD/TN/TP-based population and mobile phone estimates (47). Other studies tested the effect of population normalization on the relationship between clinical case numbers and COVID-19 infections, but factors such as inclusion of asymptomatic transmitters with WBE and differences in testing frequency complicate this comparison (15,48).
3.2. Utility consumption: drinking water and electricity
Given that drinking water and electricity are commodities used by virtually all members of society, they possess the potential to reflect population size. Assuming that each individual consumes both water and electricity, Hou et al. calculated per capita domestic water consumption by utilizing annual water supply and consumption data obtained from local water authorities in various catchment areas (32). However, this data was considered unreliable due to variations in statistical criteria related to water management among different cities. For instance, some cities included the water consumption of tertiary and construction industries within domestic water consumption. Baz-Lomba et al. investigated drinking water production and electricity consumption as potential population proxies (61), and although they appeared suitable during model calibration, annual estimates indicated bias associated with different seasons and population habits. Currently, a higher electricity consumption during the winter period is not taken into account, which could lead to discrepancies especially in regions with strong seasonal variations. Although these parameters showed some potential, it is clear that the variability between different seasons and the lack of measurement consensus (e.g., separation of domestic and industrial origin) make these biomarkers not suitable for dynamic population estimation.
3.3. Exogenous and endogenous biomarkers
Human biomarkers, either from exogenous or endogenous origin, are excreted continuously via various human sources (including urine, faeces, saliva) and enter the wastewater system. Therefore, MLs of biomarker in IWW could inform on the total number of contributors and, thus, on the de facto population served by a WWTP catchment area. These PBs can be measured simultaneously with the target biomarkers of interest within daily 24-h composite IWW samples. To be suitable candidates, PBs should be (i) excreted in sufficient amounts, (ii) specific to human metabolism, (iii) stable in IWW, (iv) excreted with limited intra- and inter-individual variability and (v) independent of extraneous variables (e.g., seasonal effects, socio-demographic differences) (10,11,19). This approach can be applied to account for transient and permanent population movements across different periods, including seasonal and daily changes (e.g., migration, tourism, commuting, visits) (11). However, variations in daily urinary excretion could also lead to some uncertainties and could be driven by a population’s health status and dietary intake. A limitation of the use of parent compounds as PBs is that they could be discharged directly in the sewer system (e.g., caffeine from washing of coffee mugs or direct disposal of pharmaceuticals). A summary of the different studies that used exogenous and endogenous biomarkers as dynamic population proxy can be found in Table 3.
Table 3:
Overview of potential endogenous and exogenous population biomarkers (PB) of both chemical and biological origin. Endogenous PB are shown in yellow, exogenous PB in blue and biological PB in green.
| PB | Origin | Stability (reference) | Coefficient of determination [external validator] | Reference for population study |
|---|---|---|---|---|
| 1,4-methylimidazole acetic acid (MIAA) | Histamine metabolite | • Variable in-sample stability between different studies (29,71) | r2MIAA = 0.84 [census data] | Hou et al. 2021 (29) |
| 5-hydroxy-indole acetic acid (5-HIAA) | Serotonin metabolite | • Variable in-sample stability between different studies (16,19,29,64,67) • In-sewer stability is pH-dependent: high in-sewer stability at pH 7 and fully degraded at pH 2 after 12 h (67) |
r25-HIAA = 0.96 [census data] | Hsu et al. 2022 (62) |
| r25-HIAA = 0.82 [census data] | Li et al. 2022 (45) | |||
| r25-HIAA = 0.88 [census data] (using concentrations instead of mass loads) | Chen et al. 2014 (64) | |||
| r25-HIAA = 0.35 [mobile phone data] | Gudra et al. 2022 (70) | |||
| No significant correlation | Pandopulos et al. 2021 (69) | |||
| Not specified, but absolute population estimates were in line with to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Androstenedione | Sex hormone precursor | • Fully degraded within 48 hours in IWW (64) | Not compared with population estimates | Chen et al. 2014 (64) |
| Coprostanol | Cholesterol metabolite | • No data available (64) | Not compared with population estimates | Reynolds et al. 2022 (72) |
| Cortisol | Stress hormone | • Fully degraded within 48 hours in IWW (64) • Low in-sewer stability under rising main and gravity sewer conditions (67) |
Not compared with population estimates | Chen et al. 2014 (64) |
| Creatinine | Creatine metabolite | • Low in-sewer stability (>40% decay) (19,67,68) | r2creatinine = 0.05 [census data] | Hsu et al. 2022 (62) |
| r2creatinine = 0.92 [census data] | Li et al 2022 (74) | |||
| Not specified, but absolute population estimates were lower compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Homovanillic acid (HVA) | Catecholamine metabolite | • Variable in-sample stability between different studies (16,69) | r2HVA = 0.922, but poor correlations in small catchment areas [census data] |
Pandopulos et al. 2021 (69) |
| Vanillylmandelic acid (VMA) | Catecholamine metabolite | • High stability at room temperature and pH 2 for 14 days (69) | r2VMA = 0.908, but poor correlations in small catchment areas [census data] |
Pandopulos et al. 2021 (69) |
| 4-Pyridoxic acid (4-PA) | Vitamin B6 metabolite | • High in sample stability at 4 °C and 27 °C for 48 h (29) | r24-PA = 0.91 [census data] | Hou et al. 2021 (29) |
| Acesulfame | Artificial sweetener | • High in-sample stability at 4 °C for at least 24 h (19,29) and 27 °C for 48 h (29) • No in-sewer degradation within 12 h after spiking under control, rising main and gravity sewer conditions (75,76) |
r2acesulfame = 0.63 [census data] | Gao et al. 2016 (26) |
| r2acesulfame = 0.995 [census data] | O’Brien et al. 2014 (25) | |||
| r2acesulfame = 0.72 [census data] | Shen et al. 2023 (65) | |||
| Aspartame | Artificial sweetener | • No data available | No correlation with population density [census data] | Shen et al. 2023 (65) |
| Atenolol | Beta-blocker | • High in-sample stability at 4 °C for 24 h (19) • No in-sewer degradation within 12 h after spiking under control, rising main and gravity sewer conditions (75,76) |
r2atenolol = 0.82 [census data] | O’Brien et al. 2014 (25) |
| Not specified, but absolute population estimates were higher compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Caffeine | Stimulant | • High in-sample stability for 24 h at 4 °C and 20 °C (36) • Variable in-sewer stability between different studies (75,76) |
Not compared with population estimates | Senta et al. 2015 (36) |
| r2caffeine = 0.77 [census data] | Gao et al. 2016 (26) | |||
| r2caffeine = 0.87 [census data] | O’Brien et al. 2014 (25) | |||
| r2caffeine = 0.97 [census data] | Hsu et al. 2022 (62) | |||
| r2caffeine = 0.79 [census data] | Li et al.2022 (45) | |||
| Not specified, but absolute population estimates were in line with to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Carbamazepine | Anticonvulsant | • High in-sample stability at 4 °C for 24 h (19) • No in-sewer degradation within 12 h after spiking under control, rising main and gravity sewer conditions (75,76) |
No significant correlation with the census population | Gao et al. 2016 (26) |
| r2carbamazepine = 0.85 [census data] | O’Brien et al. 2014 (25) | |||
| Not specified, but absolute population estimates were lower compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Codeine | Opioid | • High in-sample stability for 24 h at 4 °C (19) and 20 °C (77) • Low in-sewer stability within 12 h under rising main and gravity sewer conditions (75) |
r2codeine= 0.91 [census data] | O’Brien et al. 2014 (25) |
| Not specified, but absolute population estimates were lower compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Cotinine | Nicotine metabolite | • High in-sample stability for 24 h at 4 °C (up to 14 days) (19,36,64) • High in-sewer stability for 12 h under control, gravity sewer and rising main conditions (78,79) |
r2cotinine = 0.93 [census data] | Hou et al. 2023 (80) |
| r2cotinine = 0.78 [census data] | Wang et al. 2022 (63) | |||
| r2cotinine = 0.96, after log-transformation [census data] | Chen et al. 2014 (64) | |||
| Furosemide | Diuretic drug | • Low in-sewer stability within 12 h under control, rising main and gravity sewer conditions (75) | r2furosemide= 0.84 [census data] | O’Brien et al. 2014 (25) |
| Gabapentin | Anticonvulsant and analgesic drug | • No in-sewer degradation within 12 h after spiking under control, rising main and gravity sewer conditions (75,76) | r2gabapentin = 0.73 [census data] | Gao et al. 2016 (26) |
| r2gabapentin = 0.97 [census data] | O’Brien et al. 2014 (25) | |||
| Hydrochlorothiazide | Diuretic and antihypertensive drug |
• High in-sample stability at 4 °C for 24 h (19) • Medium in-sewer stability within 12 h after spiking under rising main conditions (75,76) |
r2hydrochlorothiazide = 0.48 [census data] | Gao et al. 2016 (26) |
| r2hydrochlorothiazide = 0.97 [census data] | O’Brien et al. 2014 (25) | |||
| Not specified, but absolute population estimates were higher compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Hydroxycotinine | Nicotine metabolite | • High in-sample stability at 4 °C and 27 °C for 48 h (29) • Variable in-sewer stability in different studies (76,78,79) |
r2hydroxycotinine = 0.66 [census data] | Hou et al. 2023 (80) |
| Not specified, but absolute population estimates were lower compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Ibuprofen | Analgesic and anti-inflammatory drug | • No in-sewer degradation within 12 h after spiking under control, rising main and gravity sewer conditions (75,76) | r2ibuprofen = 0.55 [census data] | Gao et al. 2016 (26) |
| Iopromide | Contrast media | • Low in-sewer stability under control, rising main and gravity sewer conditions (75) | r2iopromide = 0.06 [census data] | Gao et al. 2016 (26) |
| r2opromide = 0.38 [census data] | O’Brien et al. 2014 (25) | |||
| Methylxanthine | Caffeine and theophylline metabolite | • High in-sample stability for 24 h at 4 °C and 20 °C (36) | r2methylxanthine = 0.59 [census data] | Li et al. 2022 (45) |
| Naproxen | Analgesic and anti-inflammatory drug | • High in-sample stability at 4 °C for 24 h (19) • Medium in-sewer stability under control, rising main and gravity sewer conditions (>20% decay after 12 h) (75) |
r2naproxen = 0.92 [census data] | Gao et al. 2016 (26) |
| r2naproxen = 0.91 [census data] | O’Brien et al. 2014 (25) | |||
| Not specified, but absolute population estimates were higher compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Neotame | Artificial sweetener | • No data available | No correlation with population density [census data] | Shen et al. 2023 (65) |
| Norfloxacin | Antibiotic | • Low in-sewer stability within 12 h under rising main and gravity sewer conditions (75) | r2norfloxacin= 0.93 [census data] | O’Brien et al. 2014 (25) |
| Paracetamol | Antipyretic and analgesic | • Low in-sewer stability within 12 h under rising main and gravity sewer conditions (75,76) | Not compared with population estimates | Hou et al. 2023 (80) |
| r2paracetamol = 0.40 [census data] | Gao et al. 2016 (26) | |||
| r2paracetamol = 0.81 [census data] | O’Brien et al. 2014 (25) | |||
| Paraxanthine | Caffeine metabolite | • High in-sample stability for 24 h at 4 °C and 20 °C (36) • High in-sewer stability for 12 h under control and gravity sewer conditions, but fully degraded under rising main conditions (76) |
r2paraxanthine = 0.97 [census data] | Hsu et al. 2022 (62) |
| r2paraxanthine = 0.88 [census data] | Li et al. 2022 (45) | |||
| Salicylic acid | Acetylsalicylic acid metabolite and topical drug | • High in-sample stability at 4 °C for 24 h (19) • Low in-sewer stability within 12 h under control, rising main and gravity sewer conditions (75) |
r2salicylic acid = 0.31 [census data] | Gao et al. 2016 (26) |
| r2salicylic acid = 0.92 [census data] | O’Brien et al.2014 (25) | |||
| Not specified, but absolute population estimates were lower compared to BOD and COD estimates [hydrochemical data] | Rico et al. 2017 (19) | |||
| Theobromine | Alkaloid found in cocoa | • No data available | r2theobromine= 0.85 [census data] | Li et al. 2022 (45) |
| Theophylline | Respiratory drug and caffeine metabolite | • No data available | r2theophylline = 0.90 [census data] | Li et al. 2022 (45) |
| CrAssphage | Human gut microbiophage | • No data available | r2CrAssphage DNA = 0.53 and r2CrAssphage RNA = 0.36, both after log10 transformation [design capacity] | Wilder et al. 2021 (81) |
| PMMoV | Viral pathogen associated with peppers and their processed products | • Not data available for influent wastewater, but stable in other water types for 21 days at 4 °C (e.g., wetland sample) (82) | r2PMMoV= 0.85 [census data] | Hsu et al. 2022 (62) |
3.3.1. Exogenous biomarkers
Different studies have correlated the daily MLs of caffeine and its metabolites (i.e., paraxanthine and methylxanthine) with population size numbers based on census data and generally found strong correlations (r2 > 0.77) (25,26,45,62). Senta et al. also found lower MLs of caffeine biomarkers during the weekend (12–19% less) compared to the workweek, potentially indicating a population decrease, an increase in caffeine consumption during the workweek or combinations (36). It should be noted that the applicability of caffeine may be limited due to large intra- and interindividual differences in the consumption of caffeine products (e.g., coffee, caffeine supplements, energy drinks). Other methylxanthines, theophylline and theobromine, have been suggested as potential PB, showing strong correlation with population census data (r2 > 0.85) (45). However, their applicability was only tested in a single WBE study with a limited sample size.
Several studies indicated that population sizes estimated from cotinine MLs in IWW corresponded well with the number of inhabitants served by the WWTP (r2 ≥ 0.78) (29,63,64). In the same manner, relative changes in the ML of hydroxycotinine were in line with the variations in population size numbers (29). Senta et al. also showed that population estimates derived from cotinine and hydroxycotinine were slightly lower, yet in good agreement with the population number based on BOD and COD measurements (36). However, the absolute cotinine-derived population sizes were underestimated with 45% by Wang et al. compared to the theoretical values based on the design capacity of each WWTP, potentially due to an overestimation with design capacity to allow future population increase and uncertainties in flow measurements, differences in excretion rates of cotinine and variations in nicotine uptake (63). Hou et al. underestimated population numbers using MLs of cotinine and hydroxycotinine (13–70% lower for cotinine and 10–88% for hydroxycotinine) compared to census data in the different WWTPs (29). Similar to caffeine, the applicability of cotinine and hydroxycotine might be complicated by the individual differences in the amounts and types of nicotine-containing products used (e.g., cigarettes, e-cigarettes, vapers, snus).
4-pyridoxic acid (4-PA), a vitamin B6 metabolite, has been applied to back-estimate absolute population sizes by Hou et al (29). Relatively, the ML of 4-PA correlated well with the census population (r24-PA = 0.91), but discrepancies (up to 70%) were found in absolute population estimates (29). They observed that the use of 4-PA contributed up to 50% uncertainty in the back-calculation of the use of different compounds. It should also be noted that 4-PA excretion can be influenced by dietary habits and the intake of vitamin B6 supplements.
Different studies found high correlations between the MLs of the artificial sweetener acesulfame and the population equivalents and population density (expressed in number of inhabitants per km2) (r2 > 0.63) (25,26,65) and a weak positive correlation for sucrose (r2 = 0.20) (65). On the contrary, Shen et al. did not find significant correlations between the MLs of other artificial sweeteners (i.e., aspartame and neotame) and the population size (65). These (artificial) sweeteners have been proposed as potential PBs because of their widespread occurrence in food products. However, their applicability could be limited due to the inter-individual and geographical differences in the intake of artificial sweetener containing food products.
A linear relationship was found by O’Brien et al. between the MLs of 14 PBs (e.g., atenolol, carbamazepine, paracetamol) and population sizes based on census data with high coefficients of determination (r2 > 0.8; except for iopromide with r2 = 0.38) (25). However, collection of census data only occurs every five years in Australia and, therefore, the association between both variables was only investigated on one given day to obtain de facto population size numbers across 10 WWTPs. Thus, the low temporal diversity could potentially also result in artificially high coefficients of determination. Therefore, more datapoints per location are needed to further test the applicability of this PB model. Gao et al. further tested the applicability of the PB proposed in this study and found significant positive correlations with population estimates based on census data (r2 > 0.31) (26), with the exception of carbamazepine. However, these results were not fully in line with the data shown by Rico et al., who applied hydrochemical parameters to validate the use of these PBs (19). They observed discrepancies between population estimates based on hydrochemical parameters (BOD, COD, TN and TP) and exogenous PB. Population size numbers derived from atenolol, hydrochlorothiazide and naproxen were significantly higher compared to the ones based on hydrochemical data, while those based on salicylic acid, acesulfame, carbamazepine, codeine and hydroxycotinine were lower.
Kasprzyk-Hordern et al. analyzed a wide range of chemical biomarkers (N > 100) in IWW from five different communities with distinct characteristics (e.g., population size, socio-demographic features, industry) on seven consecutive days (24). They found high correlations (r2 > 0.99 on average) between the population equivalents, based on water utility estimates, and the average daily MLs of the PBs under investigation. Additionally, they reported low interday variability in population estimates for a selection of promising PBs, except for one particular location where the estimates increased over the weekend. However, they only measured five cities in one geographical area and for one week, indicating that the applicability of these PBs should be further explored with larger sample sizes and higher spatio-temporal variability.
3.3.2. Endogenous biomarkers
Several studies (N = 12) have attempted to find suitable endogenous biomarkers in IWW that reflect relative changes in population sizes. One of the first biomarkers examined was creatinine, a human metabolite of creatine and phosphorylcreatine, which is produced relatively constantly in the muscles and primarily excreted from the body through the urine (62). It has been routinely used to correct for urine dilution (66). Brewer et al. showed that creatinine-normalized hourly MLs of substances were significantly different compared to non-normalized trends, potentially as the result of population fluxes in and out of a catchment area (66). For example, the estimated population based on creatinine measurements was 14–30% lower during the weekends compared to the residential population. However, independent population figures were unavailable to validate the use of this PB and, therefore, its full potential was not fully explored in this study. While Li et al. found high correlations between the MLs of creatinine and the population sizes based on census data (r2 = 0.92) (45), Hsu et al. found a poor correlation (r2 = 0.05) (62). The results from Hsu et al. were in line with the results found by others (67,68). These findings are likely attributed to the low in-sewer and in-sample stability of creatinine, while they could also be influenced by substantial variation in intra- and interindividual urinary excretion (11,68).
Different neurotransmitter metabolites have been proposed to refine population-normalized WBE estimates (16,69). Pandopulos et al. proposed homovanillic acid (HVA) and vanillylmandelic acid (VMA), metabolites of dopamine, noradrenaline and adrenaline, as PB candidate and found higher concentrations over the duration of a festival period with 50% and 34% increase, respectively (16). They also observed a strong coefficient of determination (r2HVA = 0.922 and r2VMA = 0.908) between their MLs and the de jure population measured at census day in 38 WWTPs (69). However, they reported weaker correlations and higher variability in smaller catchment areas (population < 50 000), suggesting that the applicability of these PBs may be less relevant for estimating dynamics in small populations. Only one datapoint per WWTP was included in this study which means that interpretation of the results has to be done with caution, as mentioned earlier. The mean MLs of HVA and VMA were not significantly different in metropolitan and regional sites. Moreover, Pandopulos et al reported high in-sample stability at room temperature at pH 2 for 14 days for VMA and HVA (69), but the results were not consistent between different studies (16).
Some WBE studies investigated 5-hydroxy-indole acetic acid (5-HIAA), a serotonin metabolite, as a PB. While high correlations between the estimated population size and 5-HIAA MLs were found (19,45,62,64), others studies reported only weak to moderate correlations (16,70). Additionally, in-sewer stability studies reported inconsistent results between different experiments (29,62). For example, some reports showed that 5-HIAA had high in-sewer stability (19,64,67), while others reported poor in-sample stability (16,29).
1,4-methylimidazole acetic acid (MIAA), a histamine metabolite, has been used to calculate absolute population size figures by taking into account the flow rate and daily excretion profile of these compounds (29). Relative trends in ML of MIAA correlated well with the census population (r2MIAA = 0.84) (29). Contrastingly, variations up to 145% were found between the absolute population estimates based on MIAA and census data (29). This study also provided a sensitivity analysis based on Monte Carlo simulations and reported that the use of MIAA as PB contributed up to 34% of the total uncertainty in the back-calculation of the use of different compounds. Hou et al. found that MIAA had a high in-sample stability for 48 h at room temperature. Contrastingly, Choi et al. found that MIAA fully degraded within 12 h under rising main sewer conditions (71).
For the surveillance of SARS-CoV-2 infections using WBE, different fecal indicators have been suggested as potential PB, both from chemical and biological origin (45,72). Coprostanol (5β-cholestan3β-ol) is a cholesterol metabolite that originates from the gut microbial metabolism and makes up around 60% of the overall sterol content in human feces (72,73). Due to its high abundance in the stool, coprostanol has been suggested as a potential PB for WBE studies (72). However, its potential as a PB is limited due to its lipophilic characteristics and its strong affinity to bind to suspended particulate matter (64). Another issue is that coprostanol is excreted by other vertebrates making it difficult to distinguish between human excretion and animal input (73). Reynolds et al. indicated that the coprostanol concentrations in IWW from two Irish WWTPs showed considerable variation (29 – 50% CV). Interpretation is complicated due to absence of flow corrections and independent population data to validate the coprostanol measurements. The applicability of cortisol (a stress hormone) and androstenedione (a sex hormone precursor) as PB was also evaluated by Chen et al., but the study revealed that they were both completely degraded within 48 h in IWW (64).
3.4. Biological population markers
In this review, PB are considered biological if they are measured using molecular assays such as quantitative polymerase chain reaction (qPCR) and/or originate from biological processes such as the interaction between humans and micro-organisms. Different biological PBs have been proposed for population normalization (e.g., PMMoV, CrAssphage, 16S RNA, B-2 macroglobin, human reference gene RNase P and human-specific mitochondrial DNA) (83–86), but only biological PBs that were validated with independent population data (e.g., mobile phone data, hydrochemical population data) were considered for this literature review.
For the surveillance of pathogens in IWW, microbiologists mainly use biological PBs to correct for population dynamics within the catchment area since they lack the expertise and instrumentation to quantify chemical PBs (21). For the same reason, the application of biological PBs for the monitoring of chemical biomarkers in IWW is limited. Samples that are collected for biological and chemical analyses are also generally handled and analyzed in different ways (e.g., different storage conditions, different sample preparation and detection methods), which complicates the simultaneous quantification of biological and chemical biomarkers within the same IWW sample. Furthermore, and in contrast to chemical analysis, control reference standards (e.g., deuterated analyte-own analogues) are unavailable for biological applications, which could lead to variations in matrix inhibition and recovery efficiencies between the target pathogens and the biological PB between different samples (87). Co-concentration of viruses such as porcine coronavirus, to function as external controls during sample extraction for SARS-CoV-2 RNA is a common method, but it is not entirely clear if these viruses fully act in the same manner as SARS-CoV-2 in the wastewater system (81). Furthermore, inter-individual differences in fecal shedding may complicate their use as PB.
PMMoV has been widely adopted as fecal indicator for the normalization of viral concentrations in IWW (e.g., SARS-CoV-2, influenza) (28,62,88). PMMoV is a viral pathogen in Capsicum sp. which has been associated with pepper-based food and their processed products (62). Kitajima et al. showed the potential of PMMoV to track human fecal sources due to its occurrence in pepper-based diets and its high in-sewer stability (89). Additionally, PMMoV was not influenced by seasonal trends (89). Just like SARS-CoV-2, PMMoV is also a positive-sense single-stranded RNA virus, which makes it suitable as an internal control for sample-to-sample variability in wastewater dilution or sample preparation (90). However, PMMoV concentrations can be significantly different between cultures and countries due to differences in pepper-based food products consumption (21,91). In addition, PMMoV may also originate from non-human sources (62).
Even though the use of PMMoV as fecal indicator is widely adopted for WBE of SARS-CoV-2 (28,88,90,92–95) and recommended by the Center for Disease Control for population normalization (62), only a few studies have validated the applicability of this PB based on independent population data, (62). For example, Hsu et al. reported a strong correlation between the ML of PMMoV and the census population (r2 = 0.92). Most studies only measure the correspondence between SARS-CoV-2 RNA measurements in IWW and the number of clinical cases (27,28,90,93,94,96,97), but discrepancies between both data sources may originate from a wide range of factors, such as variability in fecal shedding, inclusion of asymptomatic transmitters with WBE, in-sewer loss of SARS-CoV-2 RNA, differences in geographical resolution or changes in diagnostic testing frequencies (90). Nevertheless, normalization using PMMoV resulted in some studies in higher coefficients of determination between WBE data and the number of positive cases (27,28,91,93,94,97), further showing the suitability of PMMoV as a potential PB. In some locations, normalization by PMMoV reduced or did not significantly affect the strength of the correlation between normalized SARS-CoV-2 RNA loads and COVID-19 incidence rates (72,93,96).
Other biological PB have been proposed for the normalization of biomarker concentrations (81). For example, different studies applied CrAssphage DNA and RNA concentrations to normalize SARS-CoV-2 RNA loads (81,87,94,98). Wilder et al. found a positive correlation between the log10-transformed MLs of CrAssphage DNA and RNA and the log10-transformed population size numbers (r2 = 0.53 and r2 = 0.36, respectively) (81). They also found a weak linear relationship between the ratio of SARS-CoV-2 to CrAssphage RNA and DNA and the number of positive test cases (r2 = 0.27 and r2 = 0.31, respectively), which was in line with the results of Mitranescu et al. and Reynolds et al. (94,98). In addition, the latter study tested the applicability of a bacterial fecal indicator, HF183, by correlating normalized SARS-CoV-2 RNA loads with the number of clinical cases (98). They found positive correlations in one out of two locations, showing some evidence for the utility of HF183 as alternative PB, but more research is needed.
3.5. Signalling records
Monitoring mobile phone usage has been proposed as an alternative approach to measure population density and mobility (14,47,61) and has been considered among the most accurate methods for relative population normalization because of its high frequency (47,61,99). Other studies outside the WBE field also showed a good relationship between mobile network activity and population density (100,101). Different models have been proposed for the estimation of population estimates based on mobile phone activity. For example, Boogaerts et al applied an approach based on passive signalling records. Within this method, anonymized passive mobile device signals to mobile masts are aggregated by the mobile network operator and extrapolated to the population size in a given WWTP. For this purpose, all signalization records of mobile phone devices with cell phone masts overlapping the WWTP catchment area are compiled, excluding machine-to-machine (i.e., automatic sharing of information between multiple devices) and Internet of Things communications (i.e., connection of devices over the internet) (14). A Chinese study used a different methodology based on location-based services (LBS) (99). However, different smartphone applications (e.g., WeChat, QQ) employ LBS with the user’s permission to track the user’s geographical location. These signals can be used to estimate the daily population size within specific spatial areas. A major advantage of mobile phone data is that it enables frequent and spatially accurate measurement of the number of persons within the WWTP catchment area, which allows the normalization of biomarker loads in every sample to an accurate population number (12).
Even though the application of mobile phone data is very useful to monitor relative temporal changes in population sizes, this approach may not always be applicable to estimate absolute population figures, since it is limited by varying cell phone possessions and market share of mobile phone companies (11). Additionally, this approach is also associated with some uncertainties with regards to other extrapolation parameters (e.g., zone probability, contact probability) used to estimate population sizes based on aggregated and anonymized mobile phone signals (14). The contribution of each of these extrapolation parameters to the overall uncertainty in absolute population estimation remains unclear. Additionally, these resources are not easily obtainable and may pose a high cost (61,99). A major limitation of these signalling records is that mobile phone users do not always contribute to an IWW sample. Methodological assumptions (e.g., visit duration and termination) should also be taken into consideration when assessing the applicability of mobile phone data. Inclusion of contributors to the IWW load heavily depends on the time intervals for which mobile phone users are included. For example, Sim et al. considered all mobile phone signals present in small unit cells (2500 m2) on an hourly basis and compiled this information for every WWTP catchment area using a geographical information system (GIS) (47). Contrastingly, Boogaerts et al. only considered mobile device signals that were present within the boundaries of the WWTP for a minimum of two hours (14).
The use of mobile phone data revealed the high degree of intra- and interday, weekly and monthly variability in the number of people present within a WWTP catchment area (12,14,99). For example, the abovementioned Chinese study overlayed population size figures based on LBS data with population estimates based on NH4-N loads (99). This study found a good correspondence in inter- and intraday temporal trends between both data sources in a WWTP in Dalian, China. Additionally, they further applied this approach to a total of 42 WWTPs in Dalian and found a good correlation between the LBS model-based population and the NH4-N population (r2 = 0.78), but also with the population based on the design capacity of the WWTPs (r2 = 0.87). Additionally, Baz-Lomba et al. reported a positive correlation between the time series of the mobile phone data and the NH4-N concentrations with a time lag of four hours. These results revealed that an increase in the mobile phone population was associated with an increase in NH4-N levels four hours later (61). Besides the positive correlation with the NH4-N levels (r2 = 0.88), high coefficients of determination were also found when overlaying drinking water production (r2 = 0.60) and electricity consumption (r2= 0.75).
Similar intra- and interday temporal trends were found by other studies (47,61,70,102). The mean population size was typically higher in metropoles and industrial cities during the workweek compared to the weekend (14,61,99). Contrastingly, Sim et al. found opposite weekly temporal patterns for a seaside city which was characterized by large leisure, entertainment and shopping facilities (47). Here, the weekend population was substantially higher compared to the workweek population due to the influx of tourists. In some locations, they observed no clear week-weekend patterns. This highlights the heterogenicity in population movements in different geographical areas and the need to carefully assess these relative changes for each WWTP separately. Furthermore, Sim et al. noted discrepancies between mobile phone and census population data, particularly in the industrial and business sites. However, census data does not reflect the non-residential active population, i.e., commuters and tourists. Additionally, significant differences were found between mobile phone data and hydrochemical population data (47), but the latter may not always be ideal to estimate population sizes due to industrial discharge within the wastewater system.
Besides mobile phone data, other types of signalling records have been employed to estimate population sizes. For example, Wright et al. applied daily university affiliate Wi-Fi connections to approximate the campus population (e.g., students and employees) (103). These population figures revealed similar weekly temporal changes compared to the abovementioned studies (12,102), but more research is required to further validate this approach. The applicability of this population proxy might be difficulted by several limitations, including the lack of connectivity in specific areas and the possibility to connect with multiple devices.
3.6. Multi-parameter modelling approaches
To address the limitations associated with single-parameter population estimates (see Table 1), various multi-parameter modelling approaches have been proposed to achieve more reliable and precise population size estimations. It should be noted that not every population study validates the applicability of these multi-parameter models (54,104). In this section, we only focus on those studies that provide a validation with available population data. In a 2014 study, O’Brien et al. developed and validated a model for estimating the de facto population based on the levels of different PBs (25). Their selection of chemical biomarkers was based on two criteria: (i) they were present in sufficiently high concentrations for direct injection in LC-MS/MS, and (ii) their levels correlated well with the population on census day (previously determined by plotting the ML of PBs against census data). This model assumed that the daily MLs of these xenobiotics entering the WWTP were proportional to the contributing population. It was further applied to provide more accurate population-normalized drug use estimates (25). Choi et al. indicated that the included PBs in the multi-compound model should have high in-sewer stability (<10% in-sewer degradation within 24 h) (76). However, this multi-compound model is susceptible to random or systematic effects due to factors, such as direct disposal of parent compounds, variations in excretion rates and daily differences in xenobiotic intake. Gao et al. assessed the applicability of this population model in a Chinese WBE study by comparing model estimates with 2010 census data. For 21 out of 31 catchment areas, the estimated population size based on the population model was within 20% of the census population (26). This study emphasizes the need for re-calibration and validation of existing population models with samples from catchment areas with diverse characteristics. However, it is important to note that the accuracy of census data that was conducted four years before the wastewater sampling, can introduce significant uncertainties when calibrating the population model. Choi et al. indicated that incorporating multiple markers of population size into a single model through Bayesian inference has proven to lead to more precise population size estimations, regardless of whether all the previously mentioned criteria are met (1). In addition, Hou et al. developed a multi-parameter population model based on population biomarkers (i.e., cotinine, 4-PA, MIAA, hydroxycotinine) and flow rate, which was consistent with census data (29). This study indicated a reduction in population-associated uncertainty when using a multi-parameter population model (29).
4. Future insights and strategies to further implement the use of de facto population markers in WBE research
4.1. A conceptual framework for identifying and validating novel population biomarkers
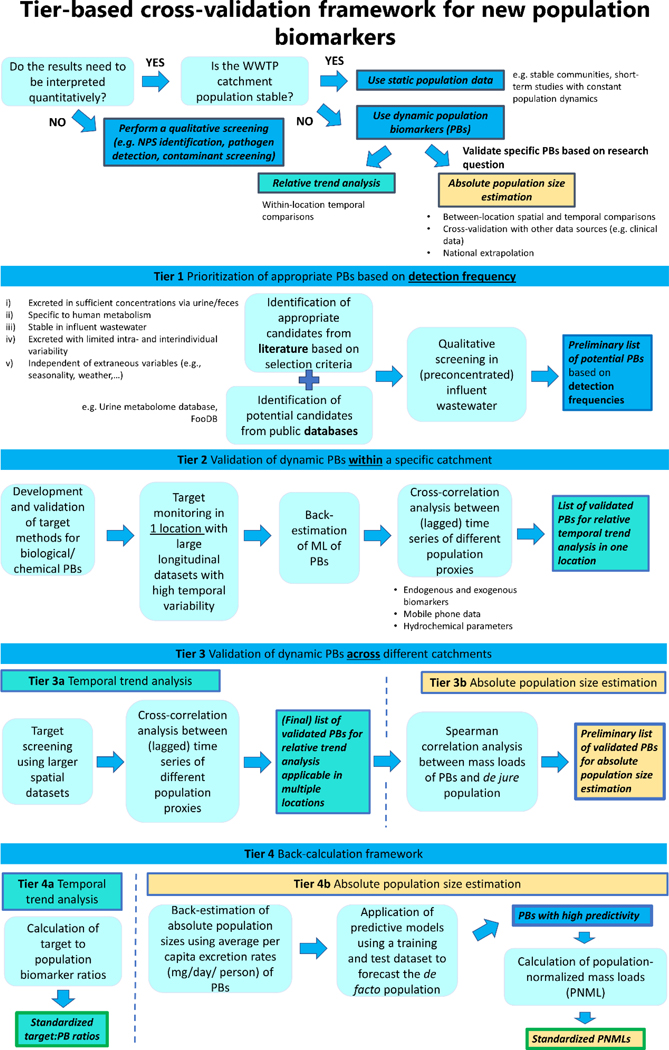
As described in Section 3, it is crucial to evaluate and validate the applicability of (novel) PBs before deploying them in specific and/or multiple locations. In Figure 2, we present a tiered approach to identify and validate promising PB candidates for population normalization across WBE studies. This workflow specifically focuses on integrating endogenous and exogenous PBs (whether chemical or biological) as low-cost population proxies for spatio-temporal trend analysis. Before applying this framework, it is essential to evaluate the necessity for de facto PBs tailored to a specific research question. For qualitative screenings (e.g., NPS identification, pathogen detection, environmental screenings), providing (semi-)quantitative measurements is often not required, and population normalization is thus not needed. Moreover, the population dynamics of each WWTP catchment should be carefully assessed to determine the need for de facto population figures. In certain WWTP catchment areas, the de jure population may remain applicable in short-term case studies where population movements and demographics remain stable over time. It may also be suitable in very large catchment areas less affected by population swings. In addition, the implementation of de facto PBs should also be evaluated in terms of feasibility such as extra costs, analyses and time.
Figure 2:
Theoretical framework for the identification and validation of new dynamic population biomarkers for population normalization
Nevertheless, multiple WBE studies have demonstrated significant variations in underlying population size figures (e.g., week-to-week differences, fluctuations during holiday periods, seasonal changes), highlighting the necessity for de facto PBs. In each wastewater monitoring campaign, PBs should be validated based on the specific research needs, whether aiming for normalization in relative trend analysis or absolute population size estimation. To reliably interpret temporal differences in the PNML of different WBE biomarkers, PBs should accurately capture the relative changes in population numbers. Without de facto population data it is impossible to attribute a change in PNML to alterations in population and/or exposure levels. Even when a potential PB cannot accurately back-estimate the actual population size, the time trend in its mass load may still be predictive for relative changes in the population size. However, back-estimation of the absolute population size is required in case of comparisons between locations, cross-validation with other data sources (e.g., clinical data, pharmaceutical sales data, seizure data) and national extrapolations. For this reason, our tiered approach differentiates between the validation of PBs intended for relative temporal trend analysis and those intended for absolute population size estimation.
The first tier of this framework focusses on the screening of literature and biomarker databases, such as the Urine Metabolome Database and FooDB to identify eligible candidates. During this initial screening, information on these biomarkers will be compiled on their inter- and intraindividual variability in urinary and/or fecal excretion, their in-sewer stability and their human metabolic specificity. Based on the eligibility criteria for PBs, a qualitative screening in (preconcentrated) IWW will be performed against broad suspect lists of potential candidates. A preliminary list of potential PB will be assembled based on their detection frequency in IWW extracts originating from different locations.
In the second tier, dedicated targeted analysis methods will be developed and validated for the selection of prioritized PBs to further evaluate their potential for population normalization. Initially, these assays should be applied to obtain MLs of these PBs in specific locations with large sets of IWW samples, aiming for high temporal variability. In the subsequent step, a cross-correlation analysis should be conducted between the (lagged) time series of different dynamic population markers (MLs of PBs, mobile phone data, hydrochemical parameters, etc.) to assess their overlap in temporal pattern. This tier aims to derive a list of validated PBs that are capable of accounting for relative changes in the underlying population size (e.g., week-weekend effects, seasonal changes) by evaluating the temporal agreement between different PBs. Although hydrochemical parameters may offer a cost-effective solution for cross-validation and are readily available, their applicability as PBs may be limited. In addition to human contribution, the levels in IWW could also be influenced by biodegradable substances, direct discharge from agriculture, industry, and food waste (29). Nevertheless, their ability to account for relative changes in population size should be further explored in this tier, regardless of its applicability for absolute population size estimation. An alternative approach to validate the capability of newly identified PBs to account for relative population changes is to apply dynamic population figures based on mobile phone data. Even though the use of this population proxy for absolute population size estimations may be limited due to different uncertainties (e.g., differences in cell phone possessions, market penetration), evidence supports its applicability to account for relative changes in population sizes (12,14,61,70). For this reason, mobile phone data can be used to validate other (low-cost) population proxies for the correction of daily, weekly, monthly and annual variability within a specific WWTP (70).
To further assess the applicability of the most promising PBs, the target screening should be expanded to also cover a wider spatial variability. Therefore, the third tier further addresses the need to cross-correlate between the time series of different population proxies in multiple locations to obtain a selection of population proxies that are applicable for relative trend analysis in different catchments. So far, the framework mainly focussed on the application of PBs to account for relative changes in population size. However, as discussed before, the use of absolute population sizes may be required in some instances, for example between-location comparisons and/or cross-validation with other data sources. Therefore, a Spearman correlation analysis should also be conducted to investigate the association between the ML of these PBs and the de jure population size. In previous WBE research, MLs of 4-PA, 5-HIAA, acesulfame, atenolol, caffeine, carbamazepine, codeine, cotinine, hydroxycotinine, CrAssphage, furosemide, gabapentin, HVA, MIAA, methylxanthine, naproxen, norfloxacin, paraxanthine, PMMoV, theobromine, theophylline, and VMA already showed moderate to high correlations (r > 0.59) with the daily catchment population size, but their applicability was only investigated in a limited number of studies and days per location. Therefore, more research is needed to further validate the use of these PBs for temporal trend analysis in WBE research.
The final tier of this framework is divided in two different junctions, one focussing on relative trend analysis (i.e., direct approach) and another on absolute population size estimation (i.e., indirect approach). In tier 4a, target-to-PB ratios will be calculated for those PBs with high cross-correlation coefficients, thus showing high temporal similarity with other dynamic population proxies. These ratios can be employed for temporal trend analysis within a given location. In tier 4b, absolute population sizes will be back-estimated taking into account the average per capita excretion rates of these PBs. However, appropriate excretion factors are not always available for this back-estimation or there is no consensus on which excretion factors should be employed. Therefore, future research should aim to optimize these values among different WBE studies. In a subsequent step, predictive modelling (e.g., linear regression models, machine learning, models) will be applied to forecast the absolute population size in different locations. Population estimates originating from different PBs will be compared, together with the de jure population size. PBs with high predictivity will be employed for the calculation of PNMLs. In Section 4.2., we will further discuss the accuracy and precision of different population proxies in estimating the absolute population size. The aim of tier 4 is to identify suitable PBs that can be used in different WBE studies in order to investigate spatio-temporal trends across different locations. Therefore, WBE researchers should collaborate and apply the proposed methodology in different locations to achieve standardization for population normalization.
At this point, the proposed tiered approach mainly considered the application of single parameter models for population normalization. However, the implementation of multi-parameter models should also be considered in future research since the use of a single population proxy may not be universally applicable due to its intrinsic limitations (e.g., cost, reliability, intervariability). The multi-parameter models may further decrease the uncertainty observed with population normalization and could potentially better predict the underlying spatio-temporal patterns in population dynamics within specific WWTPs. This was further evidenced by the different population studies that employed a multi-component model for estimating the daily number of people contributing to the IWW within a given catchment (25,26). In parallel, the robustness of these models should be evaluated in different locations to test whether they can accurately and precisely back-estimate population sizes in areas with different socio-demographic characteristics. Multiple WBE studies already indicated that the PNML of different biomarkers can be significantly impacted by socio-demographic disruptions (14,17). More research is needed to test if a multi-parameter model is able to account for these phenomena or if different parameters should be tested in function of the demography of the catchment area. Nevertheless, even when applying multi-parameter population models, WBE researchers should strive for standardization of these approaches across different studies to streamline data reporting and analysis.
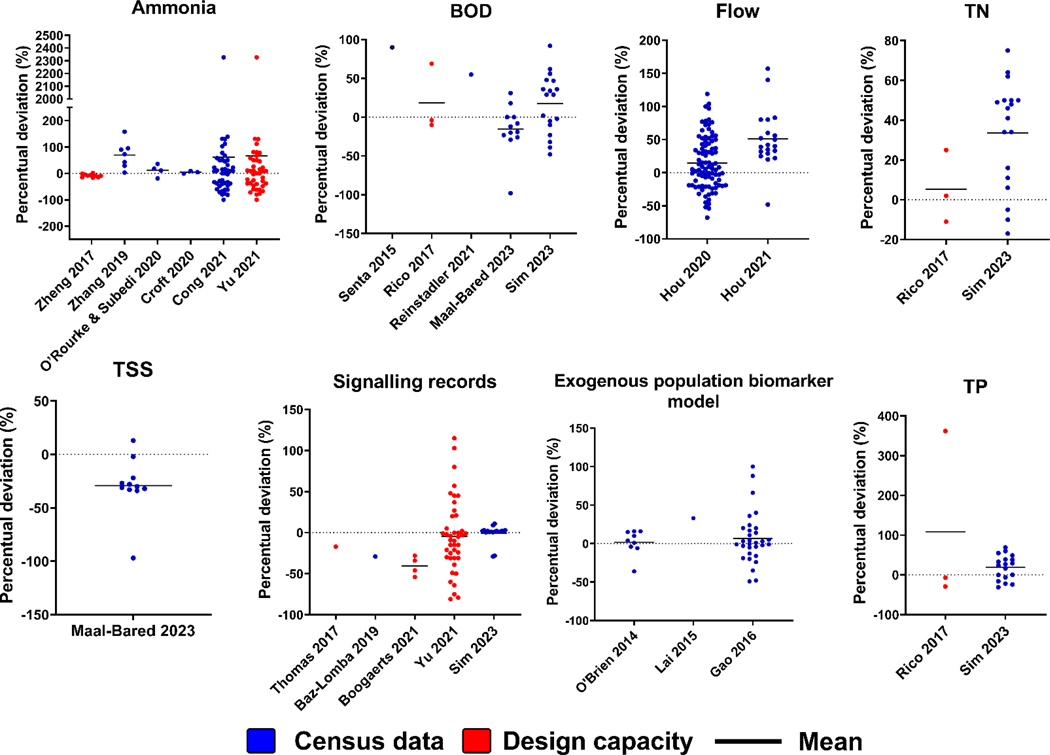
4.2. Accuracy and precision of population proxies for absolute population estimation
While the majority of studies has focused on determining the correlation between MLs of PBs and population sizes to test if they are capable of correcting for population dynamics (19,23,25,26,29,62,64,65,69,70), only a few have reported percentual differences between the de jure and de facto population size (10,19,25,26,29,36). To further assess the overall uncertainty in population size estimation, the percentage deviation between the de jure and de facto population size was calculated based on Equation 1 using all publicly available data. While acknowledging the availability of more sophisticated uncertainty analysis techniques, we opted for a method akin to the one outlined by Castiglioni et al. (4). This choice stems from the necessity of acquiring additional information to furnish more nuanced measurements. The primary objective of this study is to obtain a preliminary understanding of the uncertainty linked with the estimation of absolute population size.
Not all investigations reported individual values for the de facto population estimates. Therefore, these studies were not included for the uncertainty analysis. In addition, this analysis is limited by the number of available studies that employ specific population proxies. Some PBs are only used in a single WBE study and, therefore, it is difficult to assess their general applicability for absolute population estimation. It should also be noted that this section specifically focuses on absolute population size estimation. As described in Section 4.1., it is possible that certain population proxies are able to describe the underlying relative temporal trends in the de facto population size, but fail to absolutely back-estimate the de facto population number. These proxies can still be employed for the calculation of target to PB ratios in tier 4a of the population framework, but might not be compatible with the predictive models proposed in tier 4b. These two dimensions of PBs should be taken into account when answering specific research questions.
| Equation 1: Estimation of the percentage difference between the de jure and de facto population size |
Table S3 and Figure 3 provide a comparison between the de facto and de jure population size using different population estimation methodologies. Overall, the percentual differences between the de facto and de jure population size ranged between −100 and 200% depending on the applied population normalization approach, except for ammonium (up to 2300%), TP (up to 360%), atenolol (up to 2850%), naproxen (up to 1700%) and hydrochlorothiazide (up to 2400%) which showed larger variations in specific locations. The large percentage deviations for NH4-N and TP could be partially explained by direct discharge through non-human related sources such as industries, detergent use, and agricultural run-off. Hydrochlorothiazide, atenolol and naproxen are prescribed to a specific subset of the population and, therefore, their applicability as PBs could be influenced by the demography of the catchment population. In other words, these PBs may not be entirely representative for the whole catchment population and therefore comparison between different catchment areas is difficult. Absolute population estimation based on 5-HIAA, salicylic acid, acesulfame, atenolol, carbamazepine, codeine, cotinine, creatinine, hydrochlorothiazide, hydroxycotinine, naproxen, MIAA, 4-PA, drinking water and electricity consumption was only done in a few studies. These population estimation approaches require further investigation and will not be discussed in this section.
Figure 3:
Agreement between de facto and de jure population size: a comparison between different population proxies. The percentual deviation is calculated as the difference between the de facto and de jure population relative to the de jure population. BOD = Biological Oxygen Demand, TN = Total Nitrogen, TSS = Total suspended solids, TP = total phosphorous
This comparison highlights the large uncertainties observed with population estimation in WBE in terms of accuracy (i.e., correspondence between the de jure and de facto population size) and precision (i.e., spread in percentual change among the different studies) of potential PB candidates. This comparative analysis shows that most population proxies were not capable of back-estimating the de jure population size (see Figure 3), probably also because the de jure population underestimates the total number of contributors to the IWW. The average percentage deviation was higher than ±20% bias among several studies for most population proxies (10,13,19,29,36,41,43,47,61,96,99,102), with exceptions for NH4-N (30,38,60), BOD (19,47,96), flow rate (32), TN (19), TP (47), the exogenous PB model of O’Brien et al. (25,26) and mobile phone data (12,47,99). In addition, large intra- and inter-study variations were observed among the different locations and time periods. The standard deviation (SD) was 27% for TSS and ranged [10 – 45]% for mobile phone data, [16 – 33]% for the exogenous population model, [32 – 44]% for BOD, [6 – 351]% for ammonia, [30 – 219]% for TP, [40 – 44]% for flow and [18 – 27]% for TN. However, the large differences in SD within different studies could potentially also be explained by variations in number of samples (ranging between 1 and 95). These findings might also further reveal the dynamic nature of the WWTP catchment population and the ineffectiveness of static population figures to account for such population changes. Additionally, the true extent of population fluctuations may be substantially underestimated in previous WBE studies. Moreover, it is difficult to determine whether the percentage deviations are the result of the lack of de jure population to account for population movements, the incapability of dynamic population proxies to accurately back-estimate the actual population size or a combination. Castiglioni et al. and Thomas et al. estimated that employing fixed population numbers accounted for approximately 55% of the total uncertainty linked with WBE (4,12). However, our findings indicate that in certain case studies, this threshold is surpassed. This more in-depth analysis strongly reinforces the substantial and non-negligible uncertainty associated with population estimation.
In addition, it also highlights the need for accurate external validators to calibrate (novel) population proxies and/or models (of Antwerp, Universiteitsplein 1, 2610 Antwerp, Belgium 4.1.). Currently the population model designed by O’Brien et al. shows promising results in back-estimating the catchment population size (25). The mean percentage deviation was only 2% for O’Brien et al. and 7% for Gao et al (25,26). In addition, the variation was also lower compared to other population types (with an SD ranging [16– 33]%). The major advantage of the study of O’Brien et al. is that MLs of PBs of interest are measured on a census day on which the catchment population is accurately surveyed. However, the model of O’Brien et al. was only applied in three different WBE studies with limited data points (i.e., one sample per location). Another limitation of this study was that it mainly focused on calibrating the model in different Australian and Chinese locations, but the dataset did not cover large temporal diversity and geographical areas. Furthermore, the census population in the study of Gao et al. was already outdated, which further complicates this comparison. Therefore, efforts should be made to further refine the applicability of this model and test it in other locations. As described in section 4.1., these efforts should also further investigate the ability of this population model to capture the temporal diversity in population size within a WWTP. For temporal trend analysis, it is highly important that the population model is able to predict these underlying population changes.
Discrepancies among different WBE studies were observed when applying mobile phone data for back-estimating the absolute population size. For example, the population estimates from Baz-Lomba et al. and Boogaerts et al. were respectively 29% and 41% lower compared to the de jure population (61,102), while the estimations of Yu et al., Sim et al. and Thomas et al. were within 20% bias (47,99,105). In addition, a high variability in percentual differences was observed in the study of Yu et al. (N = 42), while the SD was <20% for Boogaerts et al. (N = 4) and Sim et al. (N =18). Even though this population proxy shows a lot of potential to account for relative changes in population size, its applicability for back-estimating the population size should be further explored.
Even though NH4-N has been commonly adopted for population normalization in WBE research, its applicability needs to be evaluated cautiously in specific locations. The absolute population estimates from Cong et al., Zhang et al. and Yu et al. were 60–70% higher compared to the de jure population (41,43,99), while the measurements of Zheng et al., O’Rourke and Subedi et al. and Croft et al. were within 20% bias (38,55,60). In addition, four out of six studies reported a SD higher than 20% percentual change. The results of this comparative analysis also show that the use of other hydrochemical parameters for back-estimation of absolute population sizes might be complicated by the contribution of non-human sources to IWW.
4.3. Lessons learned from the large-scale population disruption during the COVID-19 crisis
Temporal analysis of daily measurements of de facto population markers clearly shows that the population in a catchment area is dynamic, which impacts the calculation and interpretation of PNMLs in WBE drastically. This is further evidenced by the major population disruption in WWTPs observed during the COVID-19 pandemic, when most countries implemented specific SARS-CoV-2 spreading countermeasures such as lockdowns and social restrictions. As a result, population habits and mobility were heavily disrupted. This led to a major change in the demographic composition of the different catchment areas, affecting temporal patterns in the PNMLs of different biomarkers of interest in different locations. The impact of the social measures on the population dynamics was further evidenced by different report (102,106,107), and highlights the need for dynamic population markers in WBE.
For example, Boogaerts et al. employed mobile phone data as a dynamic population proxy for population normalization during the COVID-19 pandemic. This study reported a reduction (up to 50%) in the catchment population size during the lockdown phase (102). They also reported that the static population figure was consistently higher compared to the dynamic population estimate (even before the start of the pandemic). As previously indicated, mobile phone data might not always be capable of back-estimating the absolute population size due to some intrinsic limitations. Nevertheless, this dynamic population proxy shows high promise in capturing relative trends in a WWTP’s catchment population. To our knowledge, Boogaerts et al. is the only study that applied mobile phone data to investigate population changes during the public health crisis (102). Other investigations mainly employed COD for real-time estimation of the catchment population size (13,35), although it has been illustrated that the applicability of COD as PB is limited (see Section 4.1). In contrast to the study of Boogaerts et al., Di Marcantonio et al. did not find any significant differences between the de jure and de facto population size (35). This could also be due to the fact that they used a different experimental design to back-estimate the de facto population (i.e., based on daily COD loads) or that the number of people contributing to the WWTP remained the same before and after the onset of the COVID-19 pandemic. Yet, Reinstadler et al. measured a decrease in the average COD measurement, which would indicate a decrease in population. However, the wastewater flow rate also increased during the same time period and, for this reason, they hypothesized that the catchment population remained stable (13). However, both parameters could be highly impacted by other variables (e.g., weather, industrial activity) and, therefore, this hypothesis should be interpreted with caution.
Summarizing, events that influence the population of a catchment area (e.g., COVID-19 measures) lead to population and socio-demographic changes. For instance, Boogaerts et al. observed a significant decrease in a catchment area with a large student population that ceased commuting due to the measures, while Di Marcantonio et al. observed a status quo in the examined WWTPs (35,102). Therefore, it is important to mention that effects of certain events on the population size are very area specific and depend on the socio-demographics of the catchment area. The COVID-19 pandemic clearly illustrated the necessity to focus on more reliable dynamic population proxies.
5. Conclusions
This review documents the state-of-the-art towards applying de facto population normalization to refine spatio-temporal measurements in WBE. There is a clear need for collaboration among WBE researchers to streamline the implementation of de facto PBs across different socio-demographic communities. In particular, future efforts should aim to optimize the accuracy and precision of de facto population markers to reduce the overall uncertainty in absolute population size estimation. To achieve this, a tiered approach for identifying, integrating and validating dynamic population proxies is proposed, focussing on capturing relative changes in population numbers and back-estimating de facto population sizes. This conceptual framework may assist WBE researchers to overcome the inherent challenges associated with de facto population normalization, such as the lack of definitive validation standards and discrepancies in accuracy and precision of different population types. This tiered approach also highlights the need for integrating time series analysis techniques for cross-validating the use of de facto PBs for relative trend analysis. In addition, this study provides a comparison between de facto and de jure population sizes using various population estimation methods, revealing substantial percentual differences (up to 200%, with some exceptions) and further highlighting the need for standardization in de facto population normalization.
Supplementary Material
Acknowledgements
Tim Boogaerts (project number: 1225924N) and Maarten Quireyns (project number: G060920N) received funding of the Research Foundation – Flanders for conducting this study. L. Bijlsma acknowledges grant RYC2020-028936-I funded by MCIN/AEI/ 10.13039/501100011033 and by “ESF Investing in your future”. D. Burgard is supported by the National Institute on Drug Abuse of the National Institutes of Health under Award Number R15DA051792.
6. References
- 1.Choi P, Tscharke B, Donner E, O’Brien J, Grant S, Kaserzon S, et al. Wastewater-based epidemiology biomarkers: Past, present and future. TRAC-TRENDS IN ANALYTICAL CHEMISTRY. 2018. Aug;105:453–69. [Google Scholar]
- 2.Sims N, Kasprzyk-Hordern B. Future perspectives of wastewater-based epidemiology: Monitoring infectious disease spread and resistance to the community level. Environ Int. 2020. Jun;139:105689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baker DR, Barron L, Kasprzyk-Hordern B. Illicit and pharmaceutical drug consumption estimated via wastewater analysis. Part A: Chemical analysis and drug use estimates. Science of The Total Environment. 2014. Jul 15;487:629–41. [DOI] [PubMed] [Google Scholar]
- 4.Castiglioni S, Bijlsma L, Covaci A, Emke E, Hernández F, Reid M, et al. Evaluation of Uncertainties Associated with the Determination of Community Drug Use through the Measurement of Sewage Drug Biomarkers. Environ Sci Technol. 2013. Feb 5;47(3):1452–60. [DOI] [PubMed] [Google Scholar]
- 5.González-Mariño I, Baz-Lomba JA, Alygizakis NA, Andrés-Costa MJ, Bade R, Bannwarth A, et al. Spatio-temporal assessment of illicit drug use at large scale: evidence from 7 years of international wastewater monitoring. Addiction. 2020. Jan;115(1):109–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Been F, Emke E, Matias J, Baz-Lomba JA, Boogaerts T, Castiglioni S, et al. Changes in drug use in European cities during early COVID-19 lockdowns – A snapshot from wastewater analysis. Environment International. 2021. Aug 1;153:106540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bade R, Tscharke BJ, O’Brien JW, Magsarjav S, Humphries M, Ghetia M, et al. Impact of COVID-19 Controls on the Use of Illicit Drugs and Alcohol in Australia. Environ Sci Technol Lett. 2021. Sep 14;8(9):799–804. [DOI] [PubMed] [Google Scholar]
- 8.Wang S, Green H, Wilder M, Du Q, Kmush B, Collins M, et al. High-throughput wastewater analysis for substance use assessment in central New York during the COVID-19 pandemic. ENVIRONMENTAL SCIENCE-PROCESSES & IMPACTS. 2020. Nov 1;22(11):2147–61. [DOI] [PubMed] [Google Scholar]
- 9.Tscharke BJ, O’Brien JW, Ort C, Grant S, Gerber C, Bade R, et al. Harnessing the Power of the Census: Characterizing Wastewater Treatment Plant Catchment Populations for Wastewater-Based Epidemiology. Environ Sci Technol. 2019. Sep 3;53(17):10303–11. [DOI] [PubMed] [Google Scholar]
- 10.Lai FY, Anuj S, Bruno R, Carter S, Gartner C, Hall W, et al. Systematic and day-to-day effects of chemical-derived population estimates on wastewater-based drug epidemiology. Environ Sci Technol. 2015. Jan 20;49(2):999–1008. [DOI] [PubMed] [Google Scholar]
- 11.Daughton CG. Real-time estimation of small-area populations with human biomarkers in sewage. Science of The Total Environment. 2012. Jan;414:6–21. [DOI] [PubMed] [Google Scholar]
- 12.Thomas KV, Amador A, Baz-Lomba JA, Reid M. Use of Mobile Device Data To Better Estimate Dynamic Population Size for Wastewater-Based Epidemiology. Environ Sci Technol. 2017. Oct 3;51(19):11363–70. [DOI] [PubMed] [Google Scholar]
- 13.Reinstadler V, Ausweger V, Grabher AL, Kreidl M, Huber S, Grander J, et al. Monitoring drug consumption in Innsbruck during coronavirus disease 2019 (COVID-19) lockdown by wastewater analysis. Sci Total Environ. 2021. Feb 25;757:144006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Boogaerts T, Bertels X, Pussig B, Quireyns M, Toebosch L, Van Wichelen N, et al. Evaluating the impact of COVID-19 countermeasures on alcohol consumption through wastewater-based epidemiology: A case study in Belgium. Environ Int. 2022. Dec;170:107559. [DOI] [PubMed] [Google Scholar]
- 15.Isaksson F, Lundy L, Hedstrom A, Szekely A, Mohamed N. Evaluating the Use of Alternative Normalization Approaches on SARS-CoV-2 Concentrations in Wastewater: Experiences from Two Catchments in Northern Sweden. ENVIRONMENTS. 2022. Mar;9(3). [Google Scholar]
- 16.Pandopulos AJ, Gerber C, Tscharke BJ, O’Brien J, White JM, Bade R. A sensitive analytical method for the measurement of neurotransmitter metabolites as potential population biomarkers in wastewater. J Chromatogr A. 2020. Feb 8;1612:460623. [DOI] [PubMed] [Google Scholar]
- 17.Thomaidis NS, Gago-Ferrero P, Ort C, Maragou NC, Alygizakis NA, Borova VL, et al. Reflection of Socioeconomic Changes in Wastewater: Licit and Illicit Drug Use Patterns. Environ Sci Technol. 2016. Sep 20;50(18):10065–72. [DOI] [PubMed] [Google Scholar]
- 18.O’Brien JW, Thai PK, Tscharke BJ. Chapter 5 - Population biomarkers for wastewater-based epidemiology. In: Hadi Dehghani M, Karri RR, Rousis N, Gracia-Lor E, editors. Wastewater-Based Epidemiology for the Assessment of Human Exposure to Environmental Pollutants [Internet]. Academic Press; 2023. p. 123–38. Available from: https://www.sciencedirect.com/science/article/pii/B9780443191725000056 [Google Scholar]
- 19.Rico M, Andrés-Costa MJ, Picó Y. Estimating population size in wastewater-based epidemiology. Valencia metropolitan area as a case study. J Hazard Mater. 2017. Feb 5;323(Pt A):156–65. [DOI] [PubMed] [Google Scholar]
- 20.Been F, Rossi L, Ort C, Rudaz S, Delémont O, Esseiva P. Population normalization with ammonium in wastewater-based epidemiology: application to illicit drug monitoring. Environ Sci Technol. 2014. Jul 15;48(14):8162–9. [DOI] [PubMed] [Google Scholar]
- 21.Oloye FF, Xie Y, Challis JK, Femi-Oloye OP, Brinkmann M, McPhedran KN, et al. Understanding common population markers for SARS-CoV-2 RNA normalization in wastewater – A review. Chemosphere. 2023. Aug 1;333:138682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021. Mar 29;372:n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xie Y, Challis J, Oloye F, Asadi M, Cantin J, Brinkmann M, et al. RNA in Municipal Wastewater Reveals Magnitudes of COVID-19 Outbreaks across Four Waves Driven by SARS-CoV-2 Variants of Concern. ACS ES&T WATER. [DOI] [PubMed] [Google Scholar]
- 24.Kasprzyk-Hordern B, Proctor K, Jagadeesan K, Edler F, Standerwick R, Barden R. Human population as a key driver of biochemical burden in an inter-city system: Implications for One Health concept. J Hazard Mater. 2022. May 5;429:127882. [DOI] [PubMed] [Google Scholar]
- 25.O’Brien J, Thai P, Eaglesham G, Ort C, Scheidegger A, Carter S, et al. A Model to Estimate the Population Contributing to the Wastewater Using Samples Collected on Census Day. ENVIRONMENTAL SCIENCE & TECHNOLOGY. 2014. Jan 7;48(1):517–25. [DOI] [PubMed] [Google Scholar]
- 26.Gao J, O’Brien J, Du P, Li X, Ort C, Mueller J, et al. Measuring selected PPCPs in wastewater to estimate the population in different cities in China. SCIENCE OF THE TOTAL ENVIRONMENT. 2016. Oct 15;568:164–70. [DOI] [PubMed] [Google Scholar]
- 27.Bertels X, Hanoteaux S, Janssens R, Maloux H, Verhaegen B, Delputte P, et al. Time series modelling for wastewater-based epidemiology of COVID-19: A nationwide study in 40 wastewater treatment plants of Belgium, February 2021 to June 2022. Science of The Total Environment. 2023. Nov 15;899:165603. [DOI] [PubMed] [Google Scholar]
- 28.Janssens R, Hanoteaux S, Maloux H, Klamer S, Laisnez V, Verhaegen B, et al. SARS-CoV-2 Surveillance in Belgian Wastewaters. Viruses. 2022. Sep 2;14(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hou C, Chu T, Chen M, Hua Z, Xu P, Xu H, et al. Application of multi-parameter population model based on endogenous population biomarkers and flow volume in wastewater epidemiology. Sci Total Environ. 2021. Mar 10;759:143480. [DOI] [PubMed] [Google Scholar]
- 30.Zheng QD, Wang Z, Liu CY, Yan JH, Pei W, Wang Z, et al. Applying a population model based on hydrochemical parameters in wastewater-based epidemiology. Sci Total Environ. 2019. Mar 20;657:466–75. [DOI] [PubMed] [Google Scholar]
- 31.Wang Z, Shao XT, Tan DQ, Yan JH, Xiao Y, Zheng QD, et al. Reduction in methamphetamine consumption trends from 2015 to 2018 detected by wastewater-based epidemiology in Dalian, China. Drug Alcohol Depend. 2019. Jan 1;194:302–9. [DOI] [PubMed] [Google Scholar]
- 32.Hou C, Hua Z, Xu P, Xu H, Wang Y, Liao J, et al. Estimating the prevalence of hepatitis B by wastewater-based epidemiology in 19 cities in China. Sci Total Environ. 2020. Oct 20;740:139696. [DOI] [PubMed] [Google Scholar]
- 33.Hur J, Lee BM, Lee TH, Park DH. Estimation of Biological Oxygen Demand and Chemical Oxygen Demand for Combined Sewer Systems Using Synchronous Fluorescence Spectra. Sensors. 2010;10(4):2460–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Heidrich ES, Curtis TP, Dolfing J. Determination of the Internal Chemical Energy of Wastewater. Environ Sci Technol. 2011. Jan 15;45(2):827–32. [DOI] [PubMed] [Google Scholar]
- 35.Di Marcantonio C, Chiavola A, Gioia V, Frugis A, Cecchini G, Ceci C, et al. Impact of COVID19 restrictions on organic micropollutants in wastewater treatment plants and human consumption rates. Sci Total Environ. 2022. Mar 10;811:152327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Senta I, Gracia-Lor E, Borsotti A, Zuccato E, Castiglioni S. Wastewater analysis to monitor use of caffeine and nicotine and evaluation of their metabolites as biomarkers for population size assessment. Water Res. 2015. May 1;74:23–33. [DOI] [PubMed] [Google Scholar]
- 37.Bijlsma L, Botero-Coy AM, Rincón RJ, Peñuela GA, Hernández F. Estimation of illicit drug use in the main cities of Colombia by means of urban wastewater analysis. Sci Total Environ. 2016. Sep 15;565:984–93. [DOI] [PubMed] [Google Scholar]
- 38.Croft TL, Huffines RA, Pathak M, Subedi B. Prevalence of illicit and prescribed neuropsychiatric drugs in three communities in Kentucky using wastewater-based epidemiology and Monte Carlo simulation for the estimation of associated uncertainties. J Hazard Mater. 2020. Feb 15;384:121306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Vuori E, Happonen M, Gergov M, Nenonen T, Järvinen A, Ketola RA, et al. Wastewater analysis reveals regional variability in exposure to abused drugs and opioids in Finland. Sci Total Environ. 2014. Jul 15;487:688–95. [DOI] [PubMed] [Google Scholar]
- 40.van Nuijs ALN, Mougel JF, Tarcomnicu I, Bervoets L, Blust R, Jorens PG, et al. Sewage epidemiology--a real-time approach to estimate the consumption of illicit drugs in Brussels, Belgium. Environ Int. 2011. Apr;37(3):612–21. [DOI] [PubMed] [Google Scholar]
- 41.Zhang X, Huang R, Li P, Ren Y, Gao J, Mueller JF, et al. Temporal profile of illicit drug consumption in Guangzhou, China monitored by wastewater-based epidemiology. Environ Sci Pollut Res Int. 2019. Aug;26(23):23593–602. [DOI] [PubMed] [Google Scholar]
- 42.Song XB, Shao XT, Liu SY, Tan DQ, Wang Z, Wang DG. Assessment of metformin, nicotine, caffeine, and methamphetamine use during Chinese public holidays. Chemosphere. 2020. Nov;258:127354. [DOI] [PubMed] [Google Scholar]
- 43.Cong ZX, Shao XT, Liu SY, Pei W, Wang DG. Wastewater analysis reveals urban, suburban, and rural spatial patterns of illicit drug use in Dalian, China. Environ Sci Pollut Res Int. 2021. May;28(20):25503–13. [DOI] [PubMed] [Google Scholar]
- 44.Bowers I, Subedi B. Isoprostanes in wastewater as biomarkers of oxidative stress during COVID-19 pandemic. Chemosphere. 2021. May;271:129489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li Y, Miyani B, Zhao L, Spooner M, Gentry Z, Zou Y, et al. Surveillance of SARS-CoV-2 in nine neighborhood sewersheds in Detroit Tri-County area, United States: Assessing per capita SARS-CoV-2 estimations and COVID-19 incidence. Sci Total Environ. 2022. Dec 10;851(Pt 2):158350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hoar C, Li Y, Silverman A. Assessment of Commonly Measured Wastewater Parameters to Estimate Sewershed Populations for Use in Wastewater-Based Epidemiology: Insights into Population Dynamics in New York City during the COVID-19 Pandemic. ACS ES&T WATER. [DOI] [PubMed] [Google Scholar]
- 47.Sim W, Park S, Ha J, Kim D, Oh JE. Evaluation of population estimation methods for wastewater-based epidemiology in a metropolitan city. Sci Total Environ. 2023. Jan 20;857(Pt 1):159154. [DOI] [PubMed] [Google Scholar]
- 48.Yaniv K, Shagan M, Lewis YE, Kramarsky-Winter E, Weil M, Indenbaum V, et al. City-level SARS-CoV-2 sewage surveillance. Chemosphere [Internet]. 2021;283. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85108527978&doi=10.1016%2fj.chemosphere.2021.131194&partnerID=40&md5=6df2e2a12d2f6ceb9d8da3667bd4fd61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang Z, Zheng Q, Gartner C, Chan GCK, Ren Y, Wang D, et al. Comparison of tobacco use in a university town and a nearby urban area in China by intensive analysis of wastewater over one year period. Water Res. 2021. Nov 1;206:117733. [DOI] [PubMed] [Google Scholar]
- 50.Verhagen R, Kaserzon SL, Thomas KV, Mueller JF, Tscharke BJ. Exploring drug consumption patterns across varying levels of remoteness in Australia. Science of The Total Environment. 2023. Dec 10;903:166163. [DOI] [PubMed] [Google Scholar]
- 51.Seeley IH. Wastewater Engineering. In: Seeley IH, editor. Public Works Engineering [Internet]. London: Macmillan Education UK; 1992. p. 160–214. Available from: 10.1007/978-1-349-06927-9_4 [DOI] [Google Scholar]
- 52.Thomas KV, Bijlsma L, Castiglioni S, Covaci A, Emke E, Grabic R, et al. Comparing illicit drug use in 19 European cities through sewage analysis. Science of The Total Environment. 2012. Aug 15;432:432–9. [DOI] [PubMed] [Google Scholar]
- 53.Zessner M, Lindtner S. Estimations of municipal point source pollution in the context of river basin management. Water Sci Technol. 2005;52(9):175–82. [PubMed] [Google Scholar]
- 54.Schenk H, Heidinger P, Insam H, Kreuzinger N, Markt R, Nägele F, et al. Prediction of hospitalisations based on wastewater-based SARS-CoV-2 epidemiology. Sci Total Environ. 2023. Feb 9;162149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zheng QD, Lin JG, Pei W, Guo MX, Wang Z, Wang DG. Estimating nicotine consumption in eight cities using sewage epidemiology based on ammonia nitrogen equivalent population. Sci Total Environ. 2017. Jul 15;590–591:226–32. [DOI] [PubMed] [Google Scholar]
- 56.Benaglia L, Udrisard R, Bannwarth A, Gibson A, Béen F, Lai FY, et al. Testing wastewater from a music festival in Switzerland to assess illicit drug use. Forensic Science International [Internet]. 2020;309. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85080028423&doi=10.1016%2fj.forsciint.2020.110148&partnerID=40&md5=920102feae50b79a719c0667c30c4dc7 [DOI] [PubMed] [Google Scholar]
- 57.Shao XT, Cong ZX, Liu SY, Wang Z, Zheng XY, Wang DG. Spatial analysis of metformin use compared with nicotine and caffeine consumption through wastewater-based epidemiology in China. Ecotoxicol Environ Saf. 2021. Jan 15;208:111623. [DOI] [PubMed] [Google Scholar]
- 58.Aberi P, Arabzadeh R, Insam H, Markt R, Mayr M, Kreuzinger N, et al. Quest for Optimal Regression Models in SARS-CoV-2 Wastewater Based Epidemiology. Int J Environ Res Public Health. 2021. Oct 14;18(20). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Saththasivam J, El-Malah SS, Gomez TA, Jabbar KA, Remanan R, Krishnankutty AK, et al. COVID-19 (SARS-CoV-2) outbreak monitoring using wastewater-based epidemiology in Qatar. Sci Total Environ. 2021. Jun 20;774:145608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.O’Rourke CE, Subedi B. Occurrence and Mass Loading of Synthetic Opioids, Synthetic Cathinones, and Synthetic Cannabinoids in Wastewater Treatment Plants in Four U.S. Communities. Environ Sci Technol. 2020. Jun 2;54(11):6661–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Baz-Lomba JA, Di Ruscio F, Amador A, Reid M, Thomas KV. Assessing Alternative Population Size Proxies in a Wastewater Catchment Area Using Mobile Device Data. Environ Sci Technol. 2019. Feb 19;53(4):1994–2001. [DOI] [PubMed] [Google Scholar]
- 62.Hsu SY, Bayati M, Li C, Hsieh HY, Belenchia A, Klutts J, et al. Biomarkers selection for population normalization in SARS-CoV-2 wastewater-based epidemiology. Water Res. 2022. Sep 1;223:118985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang H, Xu B, Yang L, Huo T, Bai D, An Q, et al. Consumption of common illicit drugs in twenty-one cities in southwest China through wastewater analysis. Sci Total Environ. 2022. Dec 10;851(Pt 1):158105. [DOI] [PubMed] [Google Scholar]
- 64.Chen C, Kostakis C, Gerber JP, Tscharke BJ, Irvine RJ, White JM. Towards finding a population biomarker for wastewater epidemiology studies. Science of The Total Environment. 2014. Jul 15;487:621–8. [DOI] [PubMed] [Google Scholar]
- 65.Shen G, Lei S, Li H, Yu Q, Wu G, Shi Y, et al. Occurrence and removal of four artificial sweeteners in wastewater treatment plants of China. ENVIRONMENTAL SCIENCE-PROCESSES & IMPACTS. 2023. Jan 25;25(1):75–84. [DOI] [PubMed] [Google Scholar]
- 66.Brewer A, Ort C, Banta-Green C, Berset J, Field J. Normalized Diurnal and Between-Day Trends in Illicit and Legal Drug Loads that Account for Changes in Population. ENVIRONMENTAL SCIENCE & TECHNOLOGY. 2012. Aug 7;46(15):8305–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Thai PK, O’Brien JW, Banks APW, Jiang G, Gao J, Choi PM, et al. Evaluating the in-sewer stability of three potential population biomarkers for application in wastewater-based epidemiology. Sci Total Environ. 2019. Jun 25;671:248–53. [DOI] [PubMed] [Google Scholar]
- 68.Thai PK, O’Brien J, Jiang G, Gernjak W, Yuan Z, Eaglesham G, et al. Degradability of creatinine under sewer conditions affects its potential to be used as biomarker in sewage epidemiology. Water Research. 2014. May 15;55:272–9. [DOI] [PubMed] [Google Scholar]
- 69.Pandopulos AJ, Bade R, Tscharke BJ, O’Brien JW, Simpson BS, White JM, et al. Application of catecholamine metabolites as endogenous population biomarkers for wastewater-based epidemiology. Sci Total Environ. 2021. Apr 1;763:142992. [DOI] [PubMed] [Google Scholar]
- 70.Gudra D, Dejus S, Bartkevics V, Roga A, Kalnina I, Strods M, et al. Detection of SARS-CoV-2 RNA in wastewater and importance of population size assessment in smaller cities: An exploratory case study from two municipalities in Latvia. Sci Total Environ. 2022. Jun 1;823:153775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Choi PM, O’Brien JW, Li J, Jiang G, Thomas KV, Mueller JF. Population histamine burden assessed using wastewater-based epidemiology: The association of 1,4‑methylimidazole acetic acid and fexofenadine. Environ Int. 2018. Nov;120:172–80. [DOI] [PubMed] [Google Scholar]
- 72.Reynolds L, Sala-Comorera L, Khan M, Martin N, Whitty M, Stephens J, et al. Coprostanol as a Population Biomarker for SARS-CoV-2 Wastewater Surveillance Studies. WATER. 2022. Jan;14(2). [Google Scholar]
- 73.Gracia-Lor E, Castiglioni S, Bade R, Been F, Castrignanò E, Covaci A, et al. Measuring biomarkers in wastewater as a new source of epidemiological information: Current state and future perspectives. Environment International. 2017. Feb 1;99:131–50. [DOI] [PubMed] [Google Scholar]
- 74.Li L, Mazurowski L, Dewan A, Carine M, Haak L, Guarin TC, et al. Longitudinal monitoring of SARS-CoV-2 in wastewater using viral genetic markers and the estimation of unconfirmed COVID-19 cases. Sci Total Environ. 2022. Apr 15;817:152958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.O’Brien JW, Banks APW, Novic AJ, Mueller JF, Jiang G, Ort C, et al. Impact of in-Sewer Degradation of Pharmaceutical and Personal Care Products (PPCPs) Population Markers on a Population Model. Environ Sci Technol. 2017. Apr 4;51(7):3816–23. [DOI] [PubMed] [Google Scholar]
- 76.Choi PM, Li J, Gao J, O’Brien JW, Thomas KV, Thai PK, et al. Considerations for assessing stability of wastewater-based epidemiology biomarkers using biofilm-free and sewer reactor tests. Sci Total Environ. 2020. Mar 20;709:136228. [DOI] [PubMed] [Google Scholar]
- 77.Boogaerts T, Quireyns M, Covaci A, De Loof H, van Nuijs ALN. Analytical method for the simultaneous determination of a broad range of opioids in influent wastewater: Optimization, validation and applicability to monitor consumption patterns. Talanta. 2021. Sep 1;232:122443. [DOI] [PubMed] [Google Scholar]
- 78.Zheng Q, Eaglesham G, Tscharke BJ, O’Brien JW, Li J, Thompson J, et al. Determination of anabasine, anatabine, and nicotine biomarkers in wastewater by enhanced direct injection LC-MS/MS and evaluation of their in-sewer stability. Sci Total Environ. 2020. Nov 15;743:140551. [DOI] [PubMed] [Google Scholar]
- 79.Banks APW, Lai FY, Mueller JF, Jiang G, Carter S, Thai PK. Potential impact of the sewer system on the applicability of alcohol and tobacco biomarkers in wastewater-based epidemiology. Drug Test Anal. 2018. Mar;10(3):530–8. [DOI] [PubMed] [Google Scholar]
- 80.Hou C, Zhong Y, Zhang L, Liu M, Yan F, Chen M, et al. Estimating the prevalence of hypertension in 164 cities in China by wastewater-based epidemiology. J Hazard Mater. 2023. Feb 5;443(Pt A):130147. [DOI] [PubMed] [Google Scholar]
- 81.Wilder ML, Middleton F, Larsen DA, Du Q, Fenty A, Zeng T, et al. Co-quantification of crAssphage increases confidence in wastewater-based epidemiology for SARS-CoV-2 in low prevalence areas. Water Res X. 2021. May 1;11:100100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rachmadi AT, Kitajima M, Pepper IL, Gerba CP. Enteric and indicator virus removal by surface flow wetlands. Science of The Total Environment. 2016. Jan 15;542:976–82. [DOI] [PubMed] [Google Scholar]
- 83.Steenbeek R, Timmers PHA, van der Linde D, Hup K, Hornstra L, Been F. Monitoring the exposure and emissions of antibiotic resistance: Co-occurrence of antibiotics and resistance genes in wastewater treatment plants. Journal of Water and Health. 2022. Jul 4;20(8):1157–70. [DOI] [PubMed] [Google Scholar]
- 84.El-Malah SS, Saththasivam J, Jabbar KA, K K A, Gomez TA, Ahmed AA, et al. Application of human RNase P normalization for the realistic estimation of SARS-CoV-2 viral load in wastewater: A perspective from Qatar wastewater surveillance. Environ Technol Innov. 2022. Aug;27:102775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Holm RH, Nagarkar M, Yeager RA, Talley D, Chaney AC, Rai JP, et al. Surveillance of RNase P, PMMoV, and CrAssphage in wastewater as indicators of human fecal concentration across urban sewer neighborhoods, Kentucky. FEMS Microbes. 2022. Jan 1;3:xtac003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Yang Z, Anglès d’Auriac M, Goggins S, Kasprzyk-Hordern B, Thomas KV, Frost CG, et al. A novel DNA biosensor using a ferrocenyl intercalator applied to the potential detection of human population biomarkers in wastewater. Environ Sci Technol. 2015. May 5;49(9):5609–17. [DOI] [PubMed] [Google Scholar]
- 87.Sangsanont J, Rattanakul S, Kongprajug A, Chyerochana N, Sresung M, Sriporatana N, et al. SARS-CoV-2 RNA surveillance in large to small centralized wastewater treatment plants preceding the third COVID-19 resurgence in Bangkok, Thailand. Sci Total Environ. 2022. Feb 25;809:151169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mercier E, D’Aoust PM, Thakali O, Hegazy N, Jia JJ, Zhang Z, et al. Municipal and neighbourhood level wastewater surveillance and subtyping of an influenza virus outbreak. Sci Rep. 2022. Sep 22;12(1):15777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kitajima M, Iker BC, Pepper IL, Gerba CP. Relative abundance and treatment reduction of viruses during wastewater treatment processes — Identification of potential viral indicators. Science of The Total Environment. 2014. Aug 1;488–489:290–6. [DOI] [PubMed] [Google Scholar]
- 90.Wu F, Zhang J, Xiao A, Gu X, Lee WL, Armas F, et al. SARS-CoV-2 Titers in Wastewater Are Higher than Expected from Clinically Confirmed Cases. mSystems. 2020. Jul 21;5(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.D’Aoust PM, Mercier E, Montpetit D, Jia JJ, Alexandrov I, Neault N, et al. Quantitative analysis of SARS-CoV-2 RNA from wastewater solids in communities with low COVID-19 incidence and prevalence. Water Research [Internet]. 2021;188. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85094557618&doi=10.1016%2fj.watres.2020.116560&partnerID=40&md5=a6665f7543975110a34962410d84d567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Baldwin WM, Dayton RD, Bivins AW, Scott RS, Yurochko AD, Vanchiere JA, et al. Highly socially vulnerable communities exhibit disproportionately increased viral loads as measured in community wastewater. Environ Res. 2023. Jan 26;222:115351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Perez-Zabaleta M, Archer A, Khatami K, Jafferali MH, Nandy P, Atasoy M, et al. Long-term SARS-CoV-2 surveillance in the wastewater of Stockholm: What lessons can be learned from the Swedish perspective? Sci Total Environ. 2023. Feb 1;858(Pt 3):160023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mitranescu A, Uchaikina A, Kau A, Stange C, Ho J, Tiehm A, et al. Wastewater-Based Epidemiology for SARS-CoV-2 Biomarkers: Evaluation of Normalization Methods in Small and Large Communities in Southern Germany. ACS ES&T WATER. [DOI] [PubMed] [Google Scholar]
- 95.Grube AM, Coleman CK, LaMontagne CD, Miller ME, Kothegal NP, Holcomb DA, et al. Detection of SARS-CoV-2 RNA in wastewater and comparison to COVID-19 cases in two sewersheds, North Carolina, USA. Sci Total Environ. 2023. Feb 1;858(Pt 3):159996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Maal-Bared R, Qiu Y, Li Q, Gao T, Hrudey SE, Bhavanam S, et al. Does normalization of SARS-CoV-2 concentrations by Pepper Mild Mottle Virus improve correlations and lead time between wastewater surveillance and clinical data in Alberta (Canada): comparing twelve SARS-CoV-2 normalization approaches. Sci Total Environ. 2023. Jan 15;856(Pt 1):158964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wolfe M, Archana A, Catoe D, Coffman M, Dorevich S, Graham K, et al. Scaling of SARS-CoV-2 RNA in Settled Solids from Multiple Wastewater Treatment Plants to Compare Incidence Rates of Laboratory-Confirmed COVID-19 in Their Sewersheds. ENVIRONMENTAL SCIENCE & TECHNOLOGY LETTERS. 2021. May 11;8(5):398–404. [DOI] [PubMed] [Google Scholar]
- 98.Reynolds L, Sala-Comorera L, Khan M, Martin N, Whitty M, Stephens J, et al. Coprostanol as a Population Biomarker for SARS-CoV-2 Wastewater Surveillance Studies. WATER. 2022. Jan;14(2). [Google Scholar]
- 99.Yu H, Shao XT, Liu SY, Pei W, Kong XP, Wang Z, et al. Estimating dynamic population served by wastewater treatment plants using location-based services data. Environ Geochem Health. 2021. Nov;43(11):4627–35. [DOI] [PubMed] [Google Scholar]
- 100.Deville P, Linard C, Martin S, Gilbert M, Stevens FR, Gaughan AE, et al. Dynamic population mapping using mobile phone data. Proceedings of the National Academy of Sciences. 2014. Nov 11;111(45):15888–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bachir D, Gauthier V, El Yacoubi M, Khodabandelou G. Using mobile phone data analysis for the estimation of daily urban dynamics. In: 2017 IEEE 20th International Conference on Intelligent Transportation Systems (ITSC). 2017. p. 626–32. [Google Scholar]
- 102.Boogaerts T, Quireyns M, De Prins M, Pussig B, De Loof H, Matheï C, et al. Temporal monitoring of stimulants during the COVID-19 pandemic in Belgium through the analysis of influent wastewater. Int J Drug Policy. 2022. Jun;104:103679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wright J, Driver EM, Bowes DA, Johnston B, Halden RU. Comparison of high-frequency in-pipe SARS-CoV-2 wastewater-based surveillance to concurrent COVID-19 random clinical testing on a public U.S. university campus. Sci Total Environ. 2022. May 10;820:152877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Driver EM, Gushgari A, Chen J, Halden RU. Alcohol, nicotine, and caffeine consumption on a public U.S. university campus determined by wastewater-based epidemiology. Sci Total Environ. 2020. Jul 20;727:138492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Thomas KV, Amador A, Baz-Lomba JA, Reid M. Use of Mobile Device Data To Better Estimate Dynamic Population Size for Wastewater-Based Epidemiology. Environ Sci Technol. 2017. Oct 3;51(19):11363–70. [DOI] [PubMed] [Google Scholar]
- 106.Google. Community Mobility Reports [Internet]. California, US; Available from: https://www.google.com/covid19/mobility/ [Google Scholar]
- 107.Liu Y, Wang Z, Rader B, Li B, Wu CH, Whittington JD, et al. Associations between changes in population mobility in response to the COVID-19 pandemic and socioeconomic factors at the city level in China and country level worldwide: a retrospective, observational study. The Lancet Digital Health. 2021. Jun 1;3(6):e349–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.