Abstract
Aims
This study aims to assess the applicability of the mitral regurgitation (MR) proportionality concept in patients with atrial functional mitral regurgitation (aFMR) treated with transcatheter edge‐to‐edge repair (M‐TEER). We hypothesized that patients with disproportionate MR (higher MR relative to left ventricular size) would exhibit different outcomes compared to those with proportionate MR, despite undergoing M‐TEER.
Methods and results
We retrospectively analysed 98 patients with aFMR from the EuroSMR registry who underwent M‐TEER between 2008 and 2019. Patients met criteria for aFMR (normal indexed left ventricular end‐diastolic volume [LVEDV], preserved left ventricular ejection fraction [LVEF] ≥ 50% without regional wall motion abnormalities, and structurally normal mitral valves). We excluded patients with missing effective regurgitant orifice area (EROA) or LVEDV data. The primary endpoint was 2‐year mortality, with an EROA/LVEDV ratio employed to differentiate disproportionate from proportionate MR. Procedural success and baseline characteristics were analysed, and multivariate Cox proportional hazards models were used to identify mortality predictors. The mean patient age was 79 ± 7.3 years, with 68.8% female, and 79% had a history of atrial fibrillation. The mean EROA was 0.27 ± 0.14 cm2, and LVEDV was 95.6 ± 33.7 mL. Disproportionate MR was identified with an EROA/LVEDV ratio >0.339 cm2/100 mL. While procedural success was similar in both groups, disproportionate MR was associated with a numerically higher estimate of systolic pulmonary artery pressures (sPAP) and rates of NYHA ≥III and TR ≥ 3+. Disproportionate MR had a significant association with increased 2‐year mortality (P < 0.001). The EROA/LVEDV ratio and tricuspid annular plane systolic excursion (TAPSE) were independent predictors of 2‐year mortality (EROA/LVEDV: HR: 1.35, P = 0.010; TAPSE: HR: 0.85, P = 0.020).
Conclusions
This analysis introduces the MR proportionality concept in aFMR patients and its potential prognostic value. Paradoxically, disproportionate MR in aFMR was linked to a 1.35‐fold increase in 2‐year mortality post‐M‐TEER, emphasizing the importance of accurate preprocedural FMR characterization. Our findings in patients with disproportionate MR indicate that a high degree of aFMR with high regurgitant volumes may lead to aggravated symptoms, which is a known contributor to increased mortality following M‐TEER. These results underline the need for further research into the pathophysiology of aFMR to inform potential preventative and therapeutic strategies, ensuring optimal patient outcomes.
Keywords: atrial functional MR, MR proportionality, transcatheter mitral valve repair, valvular heart disease
Introduction
Patients with chronic functional mitral regurgitation (FMR) undergoing transcatheter edge‐to‐edge repair (M‐TEER) represent a heterogeneous group in which both evaluation of FMR severity and characterization of FMR subentities are challenging. To explain the discrepancies in outcomes of recent randomized clinical trials and to better understand the observed benefit of M‐TEER over guideline‐directed medical therapy (GDMT) in one of these trials, a novel conceptual framework integrating the severity of FMR with left ventricular (LV) volumes and ejection fraction (LVEF) has been introduced. It is hypothesized that patients with ‘disproportionate MR’—more MR in a smaller LV (despite similar LVEF)—benefit more from a M‐TEER procedure, for example, with less heart failure hospitalizations and lower mortality.
Methods
Therefore, we retrospectively analysed a cohort of 98 patients with aFMR from the EuroSMR registry that underwent M‐TEER between 2008 and 2019. Patients were considered to have aFMR according to the most recent recommendations (normal indexed LV end‐diastolic volume, preserved LVEF (LVEF ≥ 50%) without regional wall motion abnormalities, structurally normal mitral valve leaflets and dilated left atria). 2 Patients with missing values for effective regurgitant orifice area (EROA) or LV end‐diastolic volume (LVEDV) were excluded (77 included and 21 excluded patients did not differ in respect to clinical baseline characteristics, Tables S1 and S2 ). Procedural success (MR ≤ 2+) and 2‐year mortality according to EROA/LVEDV ratio were assessed.
Results
The mean age was 79 ± 7.3 years with 68.8% female and 79% showed a history of atrial fibrillation. The mean EROA was 0.27 ± 0.14 cm2, LVEDV was 95.6 ± 33.7 mL and median mitral annular diameter was 36 (33–41.5; 27 mm is considered normal) mm. At the time of procedure, 93.5% of patients reported NYHA‐class ≥III. ROC analysis revealed an EROA/LVEDV ratio of 0.00339 cm2/mL as the best discriminatory cut‐off for 2‐year mortality (EROA/LVEDV >0.339 cm2/100 mL defining ‘disproportionate MR’), which deviates from the cut‐off in HFrEF‐FMR. Besides EROA and LVEDV baseline and echocardiographic characteristics as well as procedural success rates (MR ≤ 2+: 80.0% vs. 84.3%, P = 0.67 for disproportionate vs. proportionate MR) were comparable in both groups. In patients with disproportionate aFMR, estimates of systolic pulmonary artery pressures (sPAP: 49.4 ± 13.3 mmHg vs. 44.3 ± 13.8 mmHg, P = 0.13) and the rates of NYHA ≥III (93% vs. 83%, P = 0.109) and TR ≥ 3 + (28.0% vs. 15.7%, P = 0.2) were numerically higher. Disproportionate MR was associated with increased risk of 2‐year mortality (P < 0.001, Figure 1 ). Multivariate Cox proportional hazard model identified the EROA/LVEDV and TAPSE as the only independent predictors for 2‐year mortality (EROA/LVEDV: hazard ratios [HR]: 1.35, 95% CI: 1.1–1.7, P = 0.010; TAPSE: HR: 0.85, 95% CI: 0.8–0.9, P = 0.020).
Figure 1.
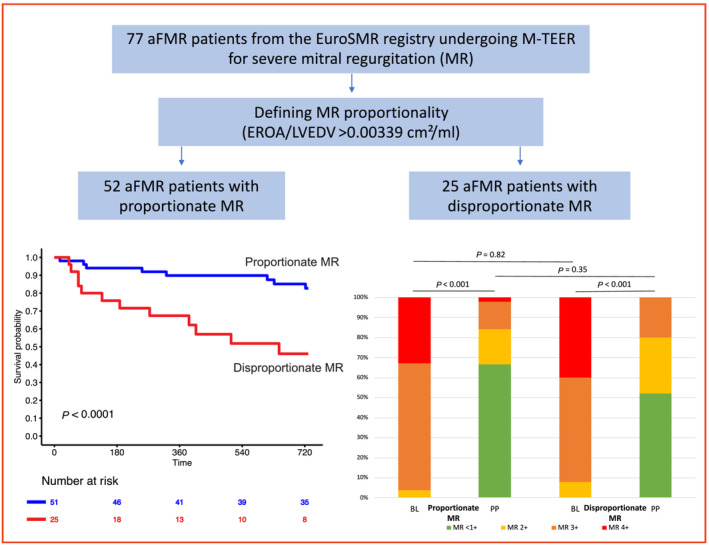
MR proportionality in patients with aFMR. This figure demonstrates a study flow‐chart (above). Below, a Kaplan–Meier curve shows 2‐year survival following M‐TEER according to MR proportionality and the according NYHA class development at follow‐up.
Aims
Although MR proportionality may have prognostic value in patients with heart failure with reduced ejection fraction (HFrEF) and FMR, 1 its predictive role in M‐TEER‐treated HFrEF‐FMR patients remains subject of debate. Additionally, it is unclear whether the proportionality concept is applicable to all M‐TEER‐treated FMR aetiologies, such as those with atrial functional mitral regurgitation (aFMR), which was the primary objective of this study.
Conclusions
This analysis investigates for the first time the concept of MR proportionality in a cohort of aFMR patients. The most common approach to quantify MR by Doppler echocardiography is the calculation of the EROA, which is derived from the proximal isovelocity surface area (PISA) method. This method focuses on pathologies at the level of the mitral valve, albeit the critical contribution of LV volume and function to the hemodynamic severity of MR are neglected. Facing these challenges of MR quantification, a novel framework termed MR proportionality has been proposed. 3 Although this hypothesis helps to explain the different outcomes of the two large randomized trials in HFrEF, whether it is applicable to all MR etiologies remains unknown. In aFMR patients, LV anatomy and function are by definition sustained while atrial fibrillation and/or HFpEF are the causes of severe symptomatic MR. Despite its different pathophysiology, current guidelines do not emphasize the need to differentiate aFMR from ventricular FMR which is usually HFrEF‐associated. 4 Although the prognostic relevance of MR proportionality might be less clear in HFrEF‐FMR patients, it seems to be of relevance in aFMR patients due to the paradoxically increased mortality. In our cohort, a disproportionate aFMR was associated with 1.35‐fold higher 2‐year mortality after M‐TEER. The reasons for this paradoxical mortality increase remain unclear, considering that baseline parameters did not differ between groups. However, it might be speculated that a higher regurgitant volume will increase pulmonary congestion resulting in higher pulmonary artery pressures with higher rates of significant TR leading to more severe heart failure related symptoms. Estimates of sPAP, and the rates of NYHA ≥III and TR ≥ 3 + demonstrated a trend towards higher values in disproportionate aFMR. This finding indicates that a high degree of aFMR with high regurgitant volumes may lead to aggravated symptoms which is a known contributor to increased mortality following M‐TEER. 4 This finding appears to be of particular relevance: First, the recently proposed results of improved outcomes of M‐TEER treated patients with disproportionate predominantly ventricular FMR do not seem to be applicable to aFMR patients with preserved LV function. This highlights aFMR as a pathophysiologically distinct aetiology and the need for a detailed preprocedural FMR characterization. Second, the prognostic relevance of heart failure related symptoms is underlined, which is an aetiology‐independent outcome predictor.
Despite these results, a reduction of regurgitant volume by M‐TEER in an aFMR patient with larger LVEDV has shown to be of clinical benefit as well in the largest available registry on M‐TEER treated aFMR patients. 4 However, additional studies are needed to better understand the pathophysiology of aFMR, which could lead to potential preventative and improved therapeutic measures for the benefit of patients. 2
Conflict of interest
J. Hausleiter received speaker honoraria from and serves as consultant for Abbott Vascular and Edwards Lifesciences. C. Iiliadis has received travel support by Abbott and consultant honoraria by Abbott and Edwards Lifesciences. Daniel Kalbacher receives personal fees from Abbott Medical, Edwards Lifesciences and PiCardia Ltd. Ralph Stephan von Bardeleben has received Institutional grants and served as speaker to Abbott Vascular and Edwards Lifesciences. Trials unpaid to Abbott Vascular, Edwards Lifesciences, Lifetec, Medtronic, NeoChord. All other authors report no relevant conflicts of interest in the context of this manuscript.
Funding
None.
Supporting information
Table S1. Baseline characteristics of patients.
Table S2. Echocardiographic Characteristics of Patients.
Acknowledgements
Open Access funding enabled and organized by Projekt DEAL.
Doldi, P. M. , Stolz, L. , Kassar, M. , Kalbacher, D. , Petronio, A. S. , Butter, C. , von Bardeleben, R. S. , Iliadis, C. , Grayburn, P. , and Hausleiter, J. (2024) Paradox of disproportionate atrial functional mitral regurgitation and survival after transcatheter edge‐to‐edge repair. ESC Heart Failure, 11: 2447–2450. 10.1002/ehf2.14789.
References
- 1. Bartko PE, Hülsmann M, Goliasch G. The authors reply: proportionality of functional mitral regurgitation: questioning the model's fundamentals and its applicability in clinical practice. J Am Coll Cardiol Img 2020;13:1458. doi: 10.1016/j.jcmg.2019.11.026 [DOI] [PubMed] [Google Scholar]
- 2. Farhan S, Silbiger JJ, Halperin JL, Zhang L, Dukkipati SR, Vogel B, et al. Pathophysiology, echocardiographic diagnosis, and treatment of atrial functional mitral regurgitation: JACC state‐of‐the‐art review. J Am Coll Cardiol 2022;80:2314‐2330. doi: 10.1016/j.jacc.2022.09.046 [DOI] [PubMed] [Google Scholar]
- 3. Grayburn PA, Sannino A, Packer M. Proportionate and disproportionate functional mitral regurgitation: a new conceptual framework that reconciles the results of the MITRA‐FR and COAPT trials. JACC Cardiovasc Imaging 2019;12:353‐362. doi: 10.1016/j.jcmg.2018.11.006 [DOI] [PubMed] [Google Scholar]
- 4. Doldi P, Stolz L, Orban M, Karam N, Praz F, Kalbacher D, et al. Transcatheter mitral valve repair in patients with atrial functional mitral regurgitation. JACC Cardiovasc Imaging 2022;15:1843‐1851. doi: 10.1016/j.jcmg.2022.05.009 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Baseline characteristics of patients.
Table S2. Echocardiographic Characteristics of Patients.


