Abstract
Purpose
Anti SARS-CoV-2 vaccination initially showed high effectiveness in preventing COVID-19. However, after the surge of variants of concern, the effectiveness dropped. Several studies investigated if this was related to the decrease of the humoral response over time; however, this issue is still unclear. The aim of this study was to understand whether SARS-CoV-2 anti-S IgG levels can be used to predict breakthrough infection risk and define the timing for further booster doses administration.
Method
Within the framework of the ORCHESTRA Project, over 20,000 health workers from 11 European centers were enrolled since December 2020. We performed two Cox proportional hazards survival analyses regarding pre-Omicron (from January to July 2021) and Omicron (December 2021–May 2022) periods. The serological response was classified as high (above the 75th percentile), medium (25th-75th), or low (< 25th).
Results
Seventy-four (0.33%) and 2122 (20%) health workers were infected during the first and second periods, respectively. Both Cox analyses showed that having high anti-S titer was linked to a significantly lower risk of infection as compared to having medium serological response [HR of high vs medium anti-S titer = 0.27 (95% CI 0.11–0.66) during the first phase, HR = 0.76 (95% CI 0.62–0.93) during the second phase].
Conclusion
Vaccine effectiveness wanes significantly after new variants surge, making anti-S titer unsuitable to predict optimal timing for further booster dose administration. Studies on other immunological indicators, such as cellular immunity, are therefore needed to better understand the mechanisms and duration of protection against breakthrough infection risk.
Supplementary Information
The online version contains supplementary material available at 10.1007/s15010-024-02189-x.
Keywords: Healthcare workers, COVID-19, SARS-CoV-2, SARS-CoV-2 anti-S IgG, COVID-19 vaccination, Breakthrough infections, Omicron variant
Introduction
In November 2019, a new coronavirus was detected in China. After the rapid spread worldwide, WHO classified the SARS-CoV-2 infection as a global pandemic on 11 March 2020, characterized by high mortality among vulnerable categories. As a consequence, enormous efforts have been made to produce an effective vaccine against the virus, and, starting from December 2020, the vaccine administration began in the European population, prioritizing the high-risk groups, including the elderly and the health workers (HW). General population’s phase 3 trials and observational studies among HW showed very high effectiveness against SARS-CoV-2 infection and COVID-19 severity during the first few months after a full vaccination course [1–4]. However, since the onset and the spread of variants of concern (VOC), protection against infection diminished significantly. In particular, after the emergence of the Omicron variant (OV) in November 2021, and the subsequent increase in the number of new cases, the WHO recommended the administration of a booster dose, with the aim of restoring the immune response and reducing the risk of infection. A previous study involving the ORCHESTRA project Cohorts, showed that this intervention reduced the chance of being infected and the COVID-19 severity during the OV period, although the effectiveness was significantly reduced when compared to previous variants [5]. A significant reduction in symptoms after OV SARS-CoV-2 infection among boostered HW towards not-boostered ones was also reported in a study carried out by Kohler et al. [6]. To understand the role of humoral response in the waning of vaccine protection, several studies investigated the capacity of vaccines to trigger anti-S antibody production among general and HW populations and the correlation between the antibody titer and the risk of breakthrough infection (BI) [7–10]. However, this issue remains unclear. Indeed, although more than 99% of the HW belonging to the ORCHESTRA project developed an antibody titer above the positivity threshold, remaining positive 6 and 9 months after the second dose [11–13], they still had BI during the period of the pre-Omicron variants (POV), and even more during the OV, despite the effect of the third dose on antibody titer [1, 5, 14].
The aims of this study were to understand whether, based on data derived from a large population of HW belonging to different European cohorts, the assessment of SARS-CoV-2 anti-S IgG levels can be used as a predictor of:
BI risk during different VOC periods.
timing for further booster doses administration.
Methods
Study design and setting
We used data from a multicentre retrospective cohort study of HW (including physicians and nurses, as well as technicians, other HW, and administrative workers) within the Horizon 2020 ORCHESTRA research project [15].
Data were collected from different centres in Italy (Bari, Bologna, Brescia, Modena, Padova, Perugia, Trieste, and Verona), Spain (Northern Barcelona region), Romania, and Slovakia.
Participants were enrolled from 27 December 2020. The follow-up ended on 31 May 2022. Data on sociodemographic characteristics such as sex, age, and job title, as well as COVID-19 PCR testing and vaccination status (including date of vaccination, number of doses, and type of vaccine administered) were obtained from medical surveillance records, ad-hoc questionnaires and/or ongoing loco-regional databases. All data were collected in a pseudoanonymized format, managed using the REDCap electronic data capture tools hosted at the Italian Interuniversity Consortium CINECA and undergone extensive data harmonization.
This study followed the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) reporting guidelines and was approved by the Italian Medicine Agency (AIFA) and the Ethics Committee of the Italian National Institute of Infectious Diseases (INMI) Lazzaro Spallanzani. The study was also approved by the local ethical boards.
Case definition and inclusion criteria
A participant was categorized as infected if tested positive on at least one nasopharyngeal swab during the study period Only HW who performed one or more serological tests were involved in the analysis. For not infected HW, we considered the latest sample performed during the timeframe, while for the infected ones, the sample closest to the date of the PCR positivity. Only serology tests performed at least 14 days before the infection were evaluated.
Only HW who completed the first vaccination course were included in the he first analysis, focused on the POV (Phase 1 analysis, from January 2021 to July 2021).
The second analysis, focused on OV period (December 2021–May 2022), included only HW who were vaccinated with a booster dose.
Details on the HW selection process are presented in Fig. S1and S2.
Statistical analysis
Continuous variables were summarized using median with InterQuartile Range (IQR) whereas categorical variables were described by frequencies, and percentages.
Methods of measurement of the antibody levels varied between the included centres and in the time periods, therefore different analytical methods were used for different cohorts. Due to the different limits of detection presented in the serology test samples, cohort-specific Tobit regression models have been used to predict right-censored serology measurements in Brescia, Bologna, and Perugia cohorts, and to predict both left and right-censored serology measurements in Slovakia cohort. Information on sex, age, number of COVID-19 vaccine doses, and previous COVID-19 infection were used as predictive variables in the Tobit regression models. We then log-transformed the results of all the cohorts included in the study and divided them by the cohort-specific standard deviation (SD). Standardized serological measurements were obtained, allowing comparison across cohorts within the study population. The same approach was used in previous analyses within ORCHESTRA Project [11, 13, 14].
Two multiple-record stratified time-to-event analyses (also called survival analyses) using the technique of Cox Proportional Hazards (PH) regression were performed afterwards, one focusing on the POV period (Phase 1 analysis) and one focusing on the times of OV (Phase 2 analysis). The date of SARS CoV-2 infection is set to be the failure event of the survival models and the time variable is calculated from the date of the serology measurement. The exit time corresponds to when the failure event occurs or, in the case of not infected HW, to the end of the model-specific timeframe.
Cohorts to which HW belong and the number of days between the last vaccine dose and the time of the serology measurement (< 60 days; 60–89 days; ≥ 90 days for the first model, and < 60 days; 60–89 days; 90–119 days; ≥ 120 days for the second one) have been used as stratified variables. Sex, age group (categories: 18–29; 30–39; 40–49; 50–59; ≥ 60), job title (Physicians, Nurses, Technicians, Administratives, Other HW), previous infections (No previous infection; Before 1st vaccine dose; After 1st vaccine dose) and the degree of serological response (categories: High, above or equal to the 75th percentile of the overall standardized serology results distribution; Low, below or equal to the 25th percentile of the overall standardized serology results distribution; Medium, between the 25th and 75th percentile) have been used as model covariates. Other variables such as the type of vaccination administrated were not included in the model since they were redundant and/or not informative. Robust variance estimators for the variance–covariance matrix of the coefficients (and hence the reported standard errors) were adopted to adjust for the clustered structure of data.
The Kaplan–Meier (KM) estimate [16] was used to graphically compare survival over time and the equality of survival functions was tested using the log-rank test. PH assumption was tested based on Schoenfeld residuals. Survival model results are expressed as hazard ratio (HR) and corresponding 95% confidence intervals (CI).
Statistical analysis was performed using Stata® software 17.0 (StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC).
Results
Description of participants
A total of 22,293 and 10,612 HW were included in the first analysis focused on the POV period (Phase 1 analysis) and in the second analysis on the OV (Phase 2 analysis), respectively. The distribution of HW within the different cohorts is shown in Table 1.
Table 1.
Distribution of the health workers included in pre-Omicron and Omicron periods analyses, by Cohort
| Cohort | Pre-Omicron period | Omicron period | ||||
|---|---|---|---|---|---|---|
| Total (N = 22,293) frequency (%) | Not infected HW (N = 22,219) frequency (%) | Infected HW (N = 74) frequency (%) | Total (N = 10,612) frequency (%) | Not infected HW (N = 8,490) frequency (%) | Infected HW (N = 2,122) frequency (%) | |
| Italy—Bari | 345 (1.5) | 341 (1.5) | 4 (5.4) | – | – | – |
| Italy—Bologna | 6,503 (29.2) | 6,488 (29.2) | 15 (20.3) | 743 (7.0) | 650 (7.7) | 93 (4.4) |
| Italy—Brescia | 6,294 (28.2) | 6,282 (28.3) | 12 (16.2) | 4,816 (45.4) | 4,265 (50.2) | 551 (26.0) |
| Italy—Modena | 1,418 (6.4) | 1,406 (6.3) | 12 (16.2) | – | – | – |
| Italy—Padova | – | – | – | 604 (5.7) | 124 (1.5) | 480 (22.6) |
| Italy—Perugia | 1,781 (8.0) | 1,780 (8.0) | 1 (1.3) | 250 (2.4) | 247 (2.9) | 3 (0.1) |
| Italy—Trieste | 4,328 (19.4) | 4,315 (19.4) | 13 (17.6) | 677 (6.4) | 505 (5.9) | 172 (8.1) |
| Italy—Verona | 879 (4.0) | 876 (4.0) | 3 (4.1) | 2,961 (27.9) | 2,153 (25.4) | 808 (38.1) |
| Romania | 450 (1.0) | 450 (1.0) | – | 148 (1.4) | 148 (1.7) | – |
| Slovakia | 217 (2.0) | 217 (2.0) | – | 235 (2.2) | 223 (2.6) | 12 (0.6) |
| Spain—Barcelona | 78 (0.3) | 64 (0.3) | 14 (18.9) | 178 (1.7) | 175 (2.1) | 3 (0.1) |
The median age of participants was 48 years (IQR 36–56) in Phase 1 analysis and 49 years (IQR 38–56) in Phase 2. The majority of participants were female in both periods (69% and 74%, respectively). Socio-demographic and laboratory characteristics of the study population are reported in Table 2
Table 2.
Socio-demographic and laboratory characteristics of Health Workers included in the study
| Variable | Pre-Omicron period | Omicron period | ||||||
|---|---|---|---|---|---|---|---|---|
| Total (N = 22,293) frequency (%) | Not infected HW (N = 22,219) frequency (%) | Infected HW (N = 74) frequency (%) | Stratified log-rank test, P value (*) | Total (N = 10,612) frequency (%) | Not infected HW (N = 8,490) frequency (%) | Infected HW (N = 2,122) frequency (%) | Stratified log-rank test, P value (*) | |
| Sex | ||||||||
| Female | 15,373 (69.0) | 15,322 (69.0) | 51 (69.0) | 0.113 | 7,775 (73.3) | 6,209 (73.1) | 1,566 (73.8) | 0.269 |
| Male | 6,920 (31.0) | 6,897 (31.0) | 23 (31.0) | 2,837 (26.7) | 2,281 (26.9) | 556 (26.2) | ||
| Age | ||||||||
| 18–29 | 2,060 (9.2) | 2,051 (9.2) | 9 (12.2) | 0.726 | 1,058 (10.0) | 757 (8.9) | 301 (14.2) | 0.002 |
| 30–39 | 5,103 (22.9) | 5,090 (22.9) | 13 (17.6) | 1,928 (18.2) | 1,479 (17.4) | 449 (21.2) | ||
| 40–49 | 4,967 (22.3) | 4,944 (22.3) | 23 (31.1) | 2,473 (23.3) | 1,933 (22.8) | 540 (25.4) | ||
| 50–59 | 7,098 (31.8) | 7,077 (31.8) | 21 (28.3) | 3,899 (36.7) | 3,225 (38.0) | 674 (31.8) | ||
| ≥ 60 | 3,065 (13.8) | 3,057 (13.8) | 8 (10.8) | 1,254 (11.8) | 1,096 (12.9) | 158 (7.4) | ||
| Job title | ||||||||
| Physicians | 6,143 (27.6) | 6,120 (27.5) | 23 (31.1) | 0.073 | 2,308 (21.8) | 1,806 (21.2) | 502 (23.7) | < 0.001 |
| Nurses | 8,180 (36.7) | 8,144 (36.7) | 36 (48.6) | 4,025 (37.9) | 3,080 (36.3) | 945 (44.5) | ||
| Technicians | 2,274 (10.2) | 2,271 (10.2) | 3 (4.1) | 1,108 (10.4) | 915 (10.8) | 193 (9.1) | ||
| Administrative | 1,610 (7.2) | 1,607 (7.2) | 3 (4.1) | 1,096 (10.3) | 949 (11.2) | 147 (6.9) | ||
| Other HW | 4,086 (18.3) | 4,077 (18.4) | 9 (12.1) | 2,075 (19.6) | 1,740 (20.5) | 335 (15.8) | ||
| Previous infections | ||||||||
| No | 22,288 (100) | 22,219 (100) | 69 (93.2) | < 0.001 | 9,754 (91.9) | 7,809 (92.0) | 1,945 (91.7) | 0.204 |
| Before 1st vaccine dose | 4 (0.0) | – | 4 (5.4) | 810 (7.6) | 681 (8.0) | 129 (6.1) | ||
| After 1st vaccine dose | 1 (0.0) | – | 1 (1.4) | 48 (0.5) | – | 48 (2.2) | ||
| Serological response | ||||||||
| High | 5,573 (25.0) | 5,544 (24.9) | 29 (39.2) | 0.016 | 2,734 (25.8) | 2,644 (31.1) | 90 (4.2) | 0.041 |
| Medium | 11,144 (50.0) | 11,121 (50.1) | 23 (31.1) | 5,227 (49.2) | 3,799 (44.8) | 1,428 (67.3) | ||
| Low | 5,576 (25.0) | 5,554 (25.0) | 22 (29.7) | 2,651 (25.0) | 2,047 (24.1) | 604 (28.5) | ||
(*) Log-rank to compare the survival distributions of the groups defined by the different values of the categorical variables
COVID-19 infections and serology measurements
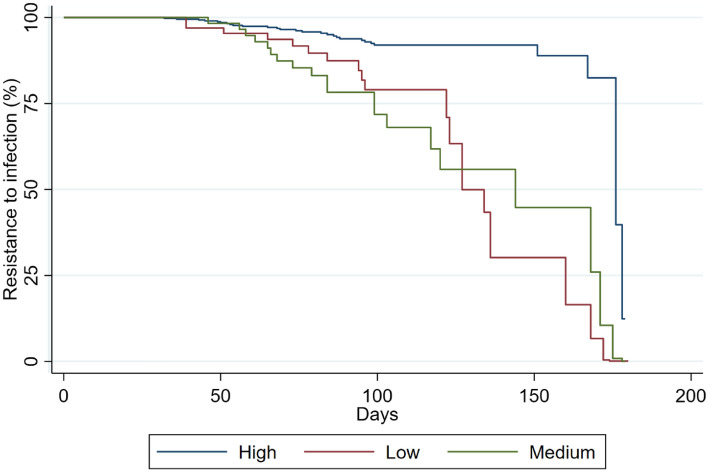
A total of 74 (0,3%) HW were infected during Phase 1 analysis. The median time-to-event (where event should be understood as the SARS-CoV-2 infection) was 104 days (IQR 26–129). The survival function is shown in Fig. 1. The median number of days elapsed between the second vaccine dose and the serology test was 100 days (IQR 60–127). The median standardized serology measurement was 6.25 (IQR 5.48–7.18).
Fig. 1.
Kaplan–Meier survival curves by antibody levels among health workers involved in the pre-Omicron period analysis
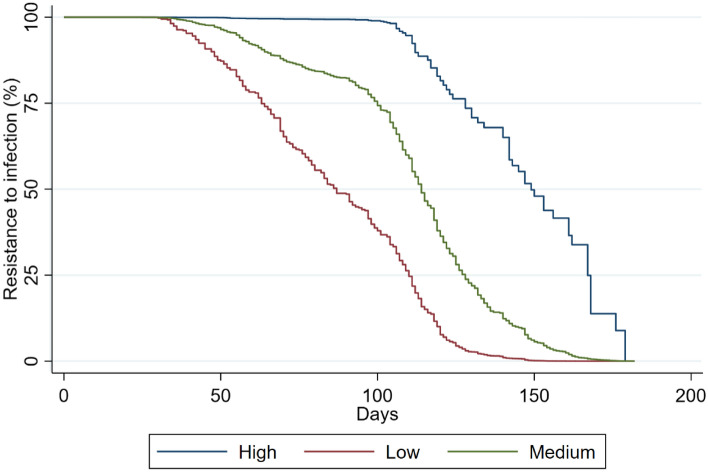
Regarding Phase 2 analysis, 2122 (20%) HW had SARS-CoV-2 infection. The median survival time was 85 days (IQR 43–94). The survival function is shown in Fig. 2. The median number of days after the full vaccination course completion and the serology test was 85 days (IQR 49–117). The median standardized serology measurement was 11.55 (IQR 10.21–12.42).
Fig. 2.
Kaplan–Meier survival curves by antibody levels among health workers involved in the Omicron period analysis
Survival analyses and factors associated with the risk of COVID-19 infection
Analysing the KM survival curves related to antibody levels of HW involved in Phase 1 analysis (Fig. 1), we observed that HW with higher anti-S titers (blue curve) had higher disease-free survival probability throughout the period than HW who had a medium-level serological response (green curve).
In the Cox PH model, HW with a high serological response presented a HR for BI of 0.27 [95% CI 0.11–0.66], when compared to HW with a medium-level serological response. Moreover, the stratified log-rank test for equality of survivor functions showed a significant difference between the three levels of serological response (P = 0.016). The test of the PH assumption based on Schoenfeld residuals revealed no evidence that the PH assumption has been violated (P = 0.918). A borderline significant HR was observed for female sex (HR = 0.59, 95% CI 0.34–1.01). Further details are presented in Table 3.
Table 3.
Cox proportional hazards survival analysis of the pre-Omicron period
| HR | 95% CI | P value | |
|---|---|---|---|
| Sex (Reference: male) | |||
| Female | 0.59 | 0.34–1.01 | 0.056 |
| Age (Reference: 18–29) | |||
| 30–39 | 0.91 | 0.34–2.49 | 0.859 |
| 40–49 | 1.06 | 0.44–2.50 | 0.911 |
| 50–59 | 0.94 | 0.36–2.44 | 0.902 |
| ≥ 60 | 0.53 | 0.17–1.65 | 0.272 |
| Job title (Reference: physicians) | |||
| Nurses | 1.18 | 0.59–2.35 | 0.644 |
| Technicians | 0.35 | 0.09–1.32 | 0.120 |
| Administrative | 0.32 | 0.07–1.49 | 0.147 |
| Other HCWs | 0.61 | 0.23–1.61 | 0.321 |
| Previous infections (Reference: no previous infections) | |||
| Before 1st vaccine dose | 1.22 | 0.20–6.19 | 0.894 |
| After 1st vaccine dose | |||
| Serological response (Reference: medium) | |||
| High | 0.27 | 0.11–0.66 | 0.004 |
| Low | 1.03 | 0.42–2.52 | 0.947 |
In the Phase 2 analysis, we found evidence of reduced risk of COVID-19 infection in three job titles when compared to physicians: nurses [HR = 0.84, 95% CI 0.74–0.95], administrative workers [HR = 0.72, 95% CI 0.60–0.86] and other working categories of HW [HR = 0.78, 95% CI 0.67–0.90]. A previous infection after the first vaccine dose entailed as well a reduced risk of COVID-19 infection, as compared to HW who had no previous infections [HR = 0.76, 95% CI 0.59–0.97]. Results of the Cox PH model also showed a strong relationship between a high-level serological response and a decreasing risk of COVID-19 infection, when compared to HW with medium-level serological response [HR = 0.76, 95% CI 0.62–0.93]. The KM survival curves related to the three different levels of serological response (Fig. 2) showed not only that HW with higher antibody levels (blue curve) presented higher survival probability at each time than HW, who had a medium-level serological response (green curve), but also that HW with lower antibody levels (red curve) presented lower survival probability at each time than HW who had a medium-level serological response (green curve).
Moreover, the stratified log-rank test for equality of survivor functions showed a significant difference between the three different levels of serological response (P = 0.041), as well as between the five age groups (P = 0.002) and the five job titles (P ≤ 0.001). The test of the PH assumption based on Schoenfeld residuals revealed no evidence that the PH assumption has been violated (P = 0.054). Further details are presented in Table 4.
Table 4.
Cox proportional hazards survival analysis of the Omicron period
| HR | 95% CI | P value | |
|---|---|---|---|
| Sex (Reference: male) | |||
| Female | 0.98 | 0.89–1.09 | 0.766 |
| 30–39 | 1.04 | 0.88–1.21 | 0.669 |
| 40–49 | 0.91 | 0.78–1.05 | 0.208 |
| 50–59 | 0.89 | 0.77–1.03 | 0.117 |
| ≥ 60 | 0.86 | 0.71–1.04 | 0.119 |
| Job title (Reference: physicians) | |||
| Nurses | 0.84 | 0.74–0.95 | 0.008 |
| Technicians | 0.85 | 0.72–1.00 | 0.063 |
| Administrative | 0.72 | 0.60–0.86 | 0.000 |
| Other HCWs | 0.78 | 0.67–0.90 | 0.001 |
| Previous infections (Reference: no previous infections) | |||
| Before 1st vaccine dose | 1.03 | 0.85–1-25 | 0.739 |
| After 1st vaccine dose | 0.76 | 0.59–0.97 | 0.025 |
| Serological response (Reference: medium) | |||
| High | 0.76 | 0.62–0.93 | 0.009 |
| Low | 1.03 | 0.91–1.18 | 0.621 |
Discussion
The cumulative incidence of BI among vaccinated HW belonging to the ORCHESTRA Project was significantly higher during the spread of the OV, as compared to previous VOC periods (0.33% vs 20%). These results are in line with data on the general population, confirming that OV has increased contagiousness even among individuals who were vaccinated with a booster dose [17]. Furthermore, the same trend was shown in the study of Asamoah-Boaheng et al., which investigated the incidence of SARS-CoV-2 infection among 1000 paramedics in Canada. Indeed, they found a BI incidence of 1.5% and 13%, before and after the Omicron era, respectively [18].
The reduced effectiveness of vaccines against the new variants is due to the occurrence of mutations at the level of the surface protein S, the protein toward which vaccines stimulate the humoral response [19]. As a consequence, a reduction in protection of anti-S IgG levels toward infection risk can be expected.
Indeed, our study reported that, during POV, HW with a serological level of anti-S IgG above the 75th percentile had a 73% reduced risk of BI than those who had an antibody titer between the 25th and 75th percentile. On the other hand, low responders (below the 25th percentile) did not have a significantly increased risk. Similar results were found both in the general and HW populations. Aldridge et al. evaluated the effect of 1 Log unit increase on BI risk among 9244 individuals of the Virus watch Cohort. They found a HR of 0.85 and reported a lower risk of BI in subjects with an anti-S IgG level above the 75th percentile if compared to subjects who had an antibody level below the 25th, by day 20 of follow-up [20]. Seekircher et al. analyzed the protective role of anti-S IgG in the Austrian population. During the 6-month follow-up, they reported a diminished risk of BI in individuals with higher levels of anti-S IgG. In particular, having twice the immunological parameter was related to a HR of 0.72 [21]. An inverted correlation between anti-S RBD IgG levels and risk of BI was found also by Smoot et al., in a study that involved 2139 nursing home residents and staff during the Delta surge [22]. However, not all studies have detected a correlation between antibodies and the risk of BI. Indeed, Yang et al. investigated this aspect among 551 HW who underwent anti-S1 RBD titration from March to October 2021. The antibody geometric mean titer did not differ between HW who subsequently had a BI and those who were not infected. On the other hand, authors reported that only 1 out of 57 HW had a BI less than 10 weeks after the full vaccination. Almost all of them, therefore, occurred after this period, when the antibody titer had decreased by 55%. These data seem to suggest that the onset of BI was possible only after a significant reduction in the antibody level [23].
Considering the OV period, our survival analysis revealed that a high antibody level (above the 75th percentile) was protective against BI after the booster dose when compared to a medium response (HR = 0.76). Again, the lowest titer was not correlated with a higher risk. Several studies showed the same effect. Indeed, Asamoah-Boaheng et al. found a reduction in the risk of BI of 20% among paramedics [18]. The study of Mohlendick et al. involved 1391 boostered HW belonging to the University Hospital Essen. The results demonstrated a twofold increased risk of BI among HW with an anti-S1 RBD titer lower than 2816.0 BAU/ml when compared to HW who had an antibody level above this cut-off (OR = 2.12; CI 1.24–3.58) during the period between November 2021 and March 2022 [24]. Barda et al. used antibody levels before the 4th dose administration and observed BI incidence while OV was prevalent in Israel, up to 6 months after the second booster dose. Among the 1098 HW involved, an anti-S IgG level > 700 BAU/mL was related to a 35% BI risk reduction. Furthermore, a tenfold increase in IgG levels decreased the risk of BI by more than 50% [25]. Gilboa et al. investigated if a higher peak of anti-S RBD against SARS-CoV-2 after booster dose was protective against infection in a Cohort of 2865 naïve HW. They found that infected HW during the OV period had a lower peak than those not infected (2659 BAU/mL vs 3107 BAU/mL; ratio of means = 0.86) [26]. A previous study, carried out in the framework of the ORCHESTRA project, confirmed this effect. Indeed, they reported that a tenfold increase of anti-S IgG led to an OR of the risk of BI equal to 0.71. In addition, HW with an anti-S titer above 2000 BAU had an OR of 0.52 and 0.34 when compared to HW with a titer lower than 500 and 1024, respectively. On the other hand, the antibody level was not related to the virus infectivity [27].
As in the POV period, some studies did not detect a protective effect of antibody titer. Indeed, among the 527 HW involved in the study of Dodge et al., although subjects who were infected by OV had lower anti-S IgG titers before positivity than negative ones, the difference was not statistically significant [28]. In the same way, even Santoro et al. reported that there was no correlation between anti-S RBD levels and OV infection in a similar sample (487 HW) [29].
Comparing the effect of antibody titer in the two periods, our data showed that while it still had a protective role in both periods, its effectiveness was significantly reduced after the onset of OV. Indeed, the risk reduction dropped from −73 to −24%. Asamoah-Boaheng et al. reported a decline from −35 to −20%, before and after the OV period [18]. The reduction in the protective effect was also reported by Smoot et al., who showed an inverse correlation only during the Delta variant period, while no correlation after the onset of the OV [22]. A previous study from one of the cohorts belonging to ORCHESTRA Project (Brescia), evaluated the effect of the SARS-CoV-2 anti-S antibody level on the risk of infection in 4824 HW. It revealed that the number of infections caused by the OV were noticeably higher (12 ×) than those caused by POV during a similar follow-up period (7–9 months). Furthermore, the number of SARS-CoV-2 infections among HW with higher serological response was not-neglectable (5.6% during a 6–8 months period) [30].
These findings supported the hypothesis that, rather than referring BI to the limited effectiveness of anti-SarS-CoV-2 vaccinations, the observation of new infection cases (BI), and even their increase during the phases of the pandemic following the first wave when vaccination was not yet available, can be referred to the occurrence of new variants including Omicron.
Such information is relevant, in order to appraise the development of the COVID-19 pandemic, both for the governmental institutions and the general public. In fact, the occurrence of new cases of infection in the vaccinated contributed to the poor trust of the public to vaccination campaigns.
These data seem to confirm that, although a higher anti-S titer is a predictor of the risk of BI even during VOC periods it cannot be used to exclude the risk of being infected by SARS-CoV-2 and, therefore, being infectious. Similarly, antibody levels in vaccinated HW is not likely to be a useful parameter to evaluate the timing for further booster dose. Therefore, it is a priority to investigate other possible indicators of protection that may have a stronger and longer relation with the risk of BI, such as cellular immunity.
Our study has some limitations.
First of all, subjects were classified as infected by POV or OV based only on the date of PCR positivity. Indeed, no laboratory data on strain type were available and some cases could therefore be misclassified. Furthermore, serological assays were performed with different methods among cohorts. To analyze the data jointly, antibody levels were normalized on a logarithmic scale, using a method already applied in previous studies of the ORCHESTRA project [11–14]. This prevents the interpretation of our results along a unique scale of anti-S level measurements. In most cohorts, the measurement of anti-S level was idiosyncratic rather than following a fixed schedule. Moreover, HW participating in the POV and OV periods were slightly different, regarding age and proportion of women (69.0% vs 73.3%), physicians (27.6% vs 21.8%), administrative staff (7.2% vs 10.3%) and, as expected, previously infected (0% vs 8.1%). However, multivariable survival analysis was adjusted for these variables to prevent/minimize confounding bias. Finally, clinical data were available only in a subset of HW, and the analysis of the correlation between anti-S levels and symptomatic or asymptomatic infections was therefore not performed.
Our study has also some strengths.
To the best of our knowledge, this study is the one exploring the correlation between antibodies and BI risk with the highest number of HW involved. Another strength is that our study is multicentric, including 11 different cohorts belonging to 4 European countries. In addition, our study investigated the effect of anti-S IgG on BI risk in two different periods with a follow-up of about 6 months, enabling to compare trends in different pandemic eras, in contrast to the majority of previous studies, that analyzed only one period.
Conclusion
Our data confirmed that a higher anti-S level was protective against infection during the POV period, as well as during the Omicron surge, among a large population of vaccinated HW, belonging to different European Cohorts. However, the protection was not complete and waned significantly after the emergence of new variants; therefore, the anti-S titer is not suitable as the predictor of the timing for further booster dose administration. Studies on cellular immunity against SARS-CoV-2 are therefore needed to better understand the mechanisms and duration of protection vs the risk of BI.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The Verona group thanks the General Management, Medical Management, and all personnel of the Units of Occupational Health, Laboratory Medicine and Microbiology of the University Hospital of Verona and all personnel of the Unit of Epidemiology and Medical Statistics, University of Verona, for their constant support and contributions. The Padova group would like to sincerely thanks the General Management, Medical Management, all personnel of Units of Occupational Health and Laboratory Medicine and Microbiology for their collaboration and valuable contributions. The authors and working group of the Northern Barcelona cohort would like to sincerely thank the participants for their effort and selfless involvement in the ProHEpiC-19 cohort study. Also, they want to thank the Management Department, Primary Care Directorate, and the Directorate of the Clinical Laboratory of the Metropolitan North for the facilities they have given for the project. In addition, we deeply thank the technical staff of Irsi Caixa for processing the samples. We thank “CERCA Programme/Generalitat de Catalunya” for institutional support. The Modena group thanks the General Management and the personnel of the Microbiology Laboratory of the University Hospital of Modena for their constructive collaboration and active contribution. The Slovakian group thanks: Zora Kľocová Adamčáková, Regional Authority of Public Health, Health promotion Dpt., Banská Bystrica, 97556; Eleonóra Fabiánová, Dpt. Occupational Health, Regional Authority of Public Health , Banská Bystrica, 97556; Jana Holčíková, Occupational Medicine Clinic, University Hospital, Bratislava, 83105; Alena Koščálová, Infectology Clinic, University Hospital, Bratislava, 83105; Zuzana Sirotná , Laboratory Dpt., Public Health Authority of the Slovak Republic, Bratislava, 826 45; Marianna Mrázová, Public Health Institute, St. Elizabeth University of Health and Social Work, 81106 Bratislava; Daniela Križanová, Hygiene Dpt., National Institute for Cardiovascular Diseases, 833 48 Bratislava 37, Slovakia; Lenka Palcová, Dpt. of Science and Research, Central Military Hospital, Ružomberok, 03426, Slovakia; Oto Osina Occupational Medicine Clinic, University Hospital, Martin, 03659, Slovakia.
Author contributions
Conceptualization: SP, PB, GS, MGLM; methodology: GS, MGLM, GV, MD, MA, GD; formal analysis: MD, MA, GD; writing—original draft preparation: GS, MD; funding acquisition: SP, PB; supervision: SP, PB. All authors performed data collection, read, reviewed, edited, and approved the final manuscript.
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement. The ORCHESTRA project is funded by the European Commission, Horizon 2020 Program, Grant Agreement No. 101016167. The Verona and Oviedo cohorts were also supported by the Regional Health Authority (Azienda Zero), Veneto Region, Italy, and by the Health Research Institute of Asturias (ISPA), Spain, respectively. The Northern Barcelona cohort has been funded by the regional Ministry of Health of the Generalitat de Catalunya (Spain) (Called COVID-19-PoC SLT16_04). The funding sources had no role in the writing of the manuscript or the decision to submit it for publication. No author has been paid to write this article by a pharmaceutical company or other agency.
Data availability
The datasets generated during the current study are not accessible to the public due to the inclusion of sensitive information necessitating compliance with data protection statutes and guidelines. Appropriate forms of data sharing can be arranged after a substantiated request to the last author.
Declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Ethics approval
The research was performed following the 1964 Declaration of Helsinki standards and its later amendments. The study was approved (No.436 14 October 2021) by the Italian Medicine Agency (AIFA) and the Ethics Committee of the Italian National Institute of Infectious Diseases (INMI) Lazzaro Spallanzani. All the cohorts collected loco-regional ethical approvals.
References
- 1.Porru S, Monaco MGL, Spiteri G, Carta A, Pezzani MD, Lippi G, et al. SARS-CoV-2 breakthrough infections: incidence and risk factors in a large european multicentric cohort of health workers. Vaccines (Basel). 2022;10:1193. 10.3390/vaccines10081193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Porru S, Spiteri G, Monaco MGL, Valotti A, Carta A, Lotti V, et al. Post-vaccination SARS-CoV-2 infections among health workers at the university hospital of Verona, Italy: a retrospective cohort survey. Vaccines (Basel). 2022;10:272. 10.3390/vaccines10020272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Angel Y, Spitzer A, Henig O, Saiag E, Sprecher E, Padova H, et al. Association between vaccination with BNT162b2 and incidence of symptomatic and asymptomatic SARS-CoV-2 infections among health care workers. JAMA. 2021;325:2457–65. 10.1001/jama.2021.7152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med. 2021;384:403–16. 10.1056/NEJMoa2035389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Porru S, Monaco MGL, Spiteri G, Carta A, Caliskan G, Violán C, et al. Incidence and determinants of symptomatic and asymptomatic SARS-CoV-2 breakthrough infections after booster dose in a large european multicentric cohort of health workers-ORCHESTRA project. J Epidemiol Glob Health. 2023;13:577–88. 10.1007/s44197-023-00139-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kohler P, Babouee Flury B, Güsewell S, Egger T, Leal O, Brucher A, et al. Clinical symptoms of SARS-CoV-2 breakthrough infection during the Omicron period in relation to baseline immune status and booster vaccination-a prospective multicentre cohort of health professionals (SURPRISE study). Influenza Other Respir Viruses. 2023;17:e13167. 10.1111/irv.13167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kared H, Wolf A-S, Alirezaylavasani A, Ravussin A, Solum G, Tran TT, et al. Immune responses in Omicron SARS-CoV-2 breakthrough infection in vaccinated adults. Nat Commun. 2022;13:4165. 10.1038/s41467-022-31888-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Feikin DR, Higdon MM, Abu-Raddad LJ, Andrews N, Araos R, Goldberg Y, et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: results of a systematic review and meta-regression. Lancet. 2022;399:924–44. 10.1016/S0140-6736(22)00152-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Evans JP, Zeng C, Carlin C, Lozanski G, Saif LJ, Oltz EM, et al. Neutralizing antibody responses elicited by SARS-CoV-2 mRNA vaccination wane over time and are boosted by breakthrough infection. Sci Transl Med. 2022;14:eabn8057. 10.1126/scitranslmed.abn8057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haveri A, Solastie A, Ekström N, Österlund P, Nohynek H, Nieminen T, et al. Neutralizing antibodies to SARS-CoV-2 Omicron variant after third mRNA vaccination in health care workers and elderly subjects. Eur J Immunol. 2022;52:816–24. 10.1002/eji.202149785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Visci G, Zunarelli C, Mansour I, Porru S, De Palma G, Duval X, et al. Serological response after SARS-CoV2 vaccination in healthcare workers: a multicenter study. Med Lav. 2022;113:e2022022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collatuzzo G, Visci G, Violante FS, Porru S, Spiteri G, Monaco MGL, et al. Determinants of anti-S immune response at 6 months after COVID-19 vaccination in a multicentric European cohort of healthcare workers - ORCHESTRA project. Front Immunol. 2022;13:986085. 10.3389/fimmu.2022.986085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Collatuzzo G, Lodi V, Feola D, De Palma G, Sansone E, Sala E, et al. Determinants of anti-S immune response at 9 months after COVID-19 vaccination in a multicentric european cohort of healthcare workers-ORCHESTRA Project. Viruses. 2022;14:2657. 10.3390/v14122657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Collatuzzo G, De Palma G, Violante FS, Porru S, Larese Filon F, Fabianova E, et al. Temporal trends of COVID-19 antibodies in vaccinated healthcare workers undergoing repeated serological sampling: an individual-level analysis within 13 months in the ORCHESTRA cohort. Front Immunol. 2022;13:1079884. 10.3389/fimmu.2022.1079884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tacconelli E, Gorska A, Carrara E, Davis RJ, Bonten M, Friedrich AW, et al. Challenges of data sharing in European Covid-19 projects: a learning opportunity for advancing pandemic preparedness and response. Lancet Reg Health Eur. 2022;21:100467. 10.1016/j.lanepe.2022.100467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81. 10.1080/01621459.1958.10501452 [DOI] [Google Scholar]
- 17.Sarkar A, Omar S, Alshareef A, Fanous K, Sarker S, Alroobi H, et al. The relative prevalence of the Omicron variant within SARS-CoV-2 infected cohorts in different countries: a systematic review. Hum Vaccin Immunother. 2023;19:2212568. 10.1080/21645515.2023.2212568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Asamoah-Boaheng M, Goldfarb DM, Karim ME, OBrien SF, Wall N, Drews SJ, et al. The relationship between anti-spike SARS-CoV-2 antibody levels and risk of breakthrough COVID-19 among fully vaccinated adults. J Infect Dis. 2023;227:339–43. 10.1093/infdis/jiac403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kimura I, Yamasoba D, Nasser H, Ito H, Zahradnik J, Wu J, et al. Multiple mutations of SARS-CoV-2 Omicron BA2 variant orchestrate its virological characteristics. J Virol. 2023;97:e0101123. 10.1128/jvi.01011-23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Aldridge RW, Yavlinsky A, Nguyen V, Eyre MT, Shrotri M, Navaratnam AMD, et al. Sars-cov-2 antibodies and breakthrough infections in the virus watch cohort. Nat Commun. 2022;13:4869. 10.1038/s41467-022-32265-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Seekircher L, Bánki Z, Kimpel J, Rössler A, Schäfer H, Falkensammer B, et al. Immune response after two doses of the BNT162b2 COVID-19 vaccine and risk of SARS-CoV-2 breakthrough infection in Tyrol, Austria: an open-label, observational phase 4 trial. Lancet Microbe. 2023;S2666–5247(23):00107–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Smoot K, Yang J, Tacker DH, Welch S, Khodaverdi M, Kimble W, et al. Persistence and protective potential of SARS-CoV-2 antibody levels after COVID-19 vaccination in a West Virginia Nursing Home Cohort. JAMA Netw Open. 2022;5:e2231334. 10.1001/jamanetworkopen.2022.31334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang SL, Mat Ripen A, Leong CT, Lee JV, Yen CH, Chand AK, et al. COVID-19 breakthrough infections and humoral immune response among BNT162b2 vaccinated healthcare workers in Malaysia. Emerg Microbes Infect. 2022;11:1262–71. 10.1080/22221751.2022.2065936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Möhlendick B, Čiučiulkaitė I, Elsner C, Anastasiou OE, Trilling M, Wagner B, et al. Individuals with weaker antibody responses after booster immunization are prone to omicron breakthrough infections. Front Immunol. 2022;13:907343. 10.3389/fimmu.2022.907343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Barda N, Canetti M, Gilboa M, Asraf K, Indenboim V, Weiss-Ottolenghi Y, et al. The association between prebooster vaccination antibody levels and the risk of severe acute respiratory syndrome coronavirus 2 infection. Clin Infect Dis. 2023;76:1315–7. 10.1093/cid/ciac886 [DOI] [PubMed] [Google Scholar]
- 26.Gilboa M, Regev-Yochay G, Mandelboim M, Indenbaum V, Asraf K, Fluss R, et al. Durability of immune response after COVID-19 booster vaccination and association with COVID-19 omicron infection. JAMA Netw Open. 2022;5:e2231778. 10.1001/jamanetworkopen.2022.31778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gilboa M, Gonen T, Barda N, Cohn S, Indenbaum V, Weiss-Ottolenghi Y, et al. Factors associated with protection from SARS-CoV-2 omicron variant infection and disease among vaccinated health care workers in Israel. JAMA Netw Open. 2023;6:e2314757. 10.1001/jamanetworkopen.2023.14757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dodge MC, Ye L, Duffy ER, Cole M, Gawel SH, Werler MM, et al. Kinetics of SARS-CoV-2 serum antibodies through the alpha, delta, and omicron surges among vaccinated health care workers at a Boston hospital. Open Forum Infect Dis. 2023;10:ofad266. 10.1093/ofid/ofad266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Santoro A, Capri A, Petrone D, Colavita F, Meschi S, Matusali G, et al. SARS-CoV-2 breakthrough infections according to the immune response elicited after mRNA third dose vaccination in COVID-19-Naïve Hospital Personnel. Biomedicines. 2023;11:1247. 10.3390/biomedicines11051247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sansone E, Collatuzzo G, Renzetti S, Ditano G, Bonfanti C, Sala E, et al. The effect of the immunization schedule and antibody levels (Anti-S) on the risk of SARS-CoV-2 infection in a large cohort of healthcare workers in Northern Italy. Vaccines (Basel). 2023;11:746. 10.3390/vaccines11040746 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during the current study are not accessible to the public due to the inclusion of sensitive information necessitating compliance with data protection statutes and guidelines. Appropriate forms of data sharing can be arranged after a substantiated request to the last author.