Abstract
Introduction:
We performed this study in order to investigate the impact of liver cirrhosis (LC) on the difficulty of minimally invasive liver resection (MILR), focusing on minor resections in anterolateral (AL) segments for primary liver malignancies.
Methods:
This was an international multicenter retrospective study of 3,675 patients who underwent MILR across 60 centers from 2004 to 2021.
Results:
1312 (35.7%) patients had no cirrhosis, 2118 (57.9%) had Child A cirrhosis and 245 (6.7%) had Child B cirrhosis. After propensity score matching (PSM), patients in Child A cirrhosis group had higher rates of open conversion (p = 0.024), blood loss >500 mls (p = 0.001), blood transfusion (p < 0.001), postoperative morbidity (p = 0.004), and in-hospital mortality (p = 0.041). After coarsened exact matching (CEM), Child A cirrhotic patients had higher open conversion rate (p = 0.05), greater median blood loss (p = 0.014) and increased postoperative morbidity (p = 0.001). Compared to Child A cirrhosis, Child B cirrhosis group had longer postoperative stay (p = 0.001) and greater major morbidity (p = 0.012) after PSM, and higher blood transfusion rates (p = 0.002), longer postoperative stay (p <0.001), and greater major morbidity (p = 0.006) after CEM. After PSM, patients with portal hypertension experienced higher rates of blood loss >500 mls (p = 0.003) and intraoperative blood transfusion (p = 0.025).
Conclusion:
The presence and severity of LC affect and compound the difficulty of MILR for minor resections in the AL segments. These factors should be considered for inclusion into future difficulty scoring systems for MILR.
Keywords: laparoscopic liver, laparoscopic hepatectomy, minimally-invasive liver, minimally-invasive hepatectomy, cirrhosis, difficulty score
Introduction
Minimally invasive liver resection (MILR) has been increasingly adopted worldwide due to its widely reported short-term benefits such as lower blood loss, reduced post-operative morbidity and shorter hospitalisation stay, with no compromise in oncologic outcomes compared to the traditional approach [1,2]. Despite its many purported advantages, it remains a technically challenging undertaking requiring expertise in both liver resection as well as minimally invasive surgery. As such, consensus guidelines emphasize the need for stepwise progression along the learning curve in order to ensure patient safety [3,4]. To this end, several Difficulty Scoring Systems (DSS) [5] have been developed to facilitate preoperative objective stratification of surgical complexity, so as to enable appropriate case selections for surgeons according to their expertise level, proper risk counselling of patients, as well as fair audit of surgical outcomes [5–9].
Primary liver malignancies often arise on a background of liver cirrhosis (LC), which compounds the difficulty of resection [10]. The cirrhotic liver parenchyma is often stiff and less amenable to transection, and bleeding tendency is increased especially when there is concomitant portal hypertension, thrombocytopenia and coagulopathy[11]. Patients are also at greater risk of post-operative complications such as post-hepatectomy liver failure and death[12]. A recent multicentre study demonstrated that LC was associated with adverse outcomes in laparoscopic liver resections (LLR) [13]. Not surprisingly, a recent survey of surgeons also revealed that most surgeons considered LC has a major factor impacting the outcomes of LLR [14]. Nonetheless to date, none of the DSS have considered the presence of cirrhosis in the calculation of the final difficulty score [5]. In the Iwate score only 1 difficulty point is accorded for the presence Child-Pugh B cirrhosis, but no points are given for Child-Pugh A cirrhosis. To date, the presence of portal hypertension is not included in any DSS, likely because it was traditionally considered a contraindication for LR.
Although, previous studies had demonstrated the detrimental impact of LC on the outcomes of LLR, these studies were associated with several limitations such as including all types/ extents of LLR including difficult and simple resections and including various pathologies such as liver metastases and benign tumors which could potentially confound the study result [15]. Some authors had also reported that the impact of LC on the outcomes of LLR correlated with the extent and difficulty of LLR [16]. LC resulted in poorer outcomes in patients undergoing more complex [Institut Mutualiste Montsouris (IMM) II/III] but not simpler IMM I LLR [16].
Hence, with these previous limitations in mind, we performed this matched-controlled study to analyze the impact of the presence and severity of LC as well as portal hypertension (PHT) on the difficulty of MILR focusing specifically on patients with primary liver malignancies undergoing minor resection in the anterolateral segments.
Methods
This was a retrospective review of 9064 patients who underwent minor MILR of the anterolateral segments (segment 2/3/4b/5/6) across 60 international centers between 2004–2021. Of these, 4,363 MILR were performed for hepatocellular carcinoma, cholangiohepatoma or intrahepatic cholangiocarcinoma. All institutions obtained their respective approvals according to their local center’s requirements. This study was approved by the Singapore General Hospital Institution Review Board and the need for patient consent was waived. The deidentified data were collected in the individual centers. These were collated and analyzed centrally at the Singapore General Hospital.
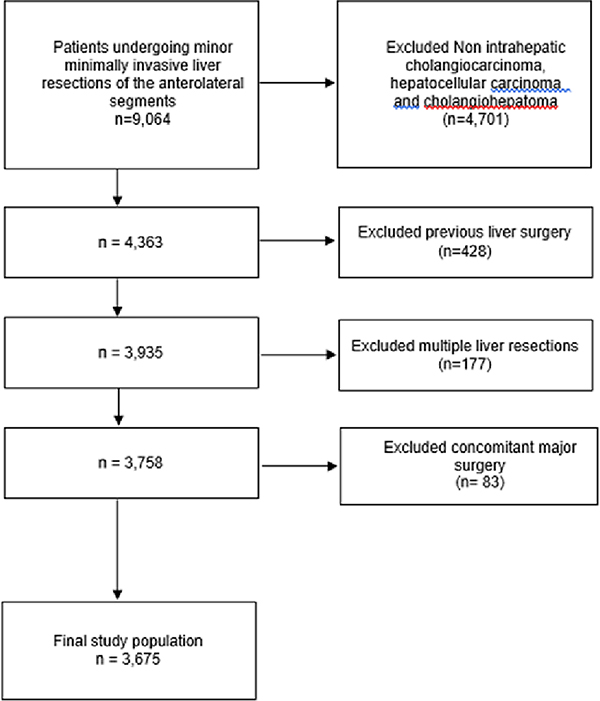
Only patients who underwent totally laparoscopic or robotic liver resections were included. Hand-assisted or laparoscopic-assisted cases were excluded. 428 patients who had previous liver surgery were excluded. Of the remaining 3935 patients, 177 with concomitant multiple liver resections were excluded and a further 83 patients who underwent concomitant major operations such as bilio-enteric anastomoses, colectomies, stoma reversal, gastrectomies, splenectomies and vascular resections were excluded. Finally, 3675 cases were included in this study. Of note, patients who underwent concomitant minor operations such as hernia repair, local ablation and hilar lymph node dissection were included.
Definitions
Liver resections were defined according to the 2000 Brisbane classification[17]. Anterolateral segments included segments 2/3/4b/5/6. Only minor resections were included and these were classified as segmentectomy or wedge/partial resections. Notably, left lateral sectionectomies were excluded. Traditional major resections classified as resection of 3 or more segments were excluded. Additionally, right anterior and right posterior sectionectomies were also considered as major resections in this study and excluded [18,19]. Diameter of the largest lesion was used in the cases of multiple tumors.
Difficulty of resections were graded according to the Iwate score [9]. Post-operative complications were classified according to the Clavien-Dindo classification and recorded for up to 30 days or during the same hospitalization [20]. Clinically significant portal hypertension (PHT) was defined as the presence of ascites, varices or splenomegaly with a platelet count of less than 100 × 109/L [15, 21]. Information on the presence of splenomegaly was obtained from an individual center’s preoperative radiologic reports.
Statistical analyses
Propensity score matching (PSM) [22] and Coarsened Exact Matching (CEM) [23] were used to estimate the effect of varying degrees of liver cirrhosis on minimally invasive minor anterolateral hepatectomy. For propensity score matching, the propensity score was estimated with logistic regression. The factors used in calculating the propensity score are the baseline variables stated in Table 1 – age at operation, gender, robotic or laparoscopic approach, previous abdominal operations, year of operation, ASA status, tumor type, tumor size, multifocality of tumor, concomitant minor operations or hilar lymph node dissection, type of resection, Iwate score and Iwate difficulty grade. For comparison in patients with or without portal hypertension, an additional variable of Child Pugh level was used in the matching. A random-effects parameter was also included in the model to account for between center variations.
Table 1.
Comparison between baseline characteristics of MILR if the anterolateral segments in Childs A cirrhosis v non-cirrhosis
| Entire unmatched cohort | 1:1 PSM (nearest neighbour matching) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| All (N = 3430) | Child A Cirrhosis (N = 2118) | Non-cirrhosis (N = 1312) | P-value | Child A Cirrhosis (N = 991) | Non-cirrhosis (N = 991) | P-value (paired) | Child A Cirrhosis (N = 518) | Non-cirrhosis (N = 518) | P-value (paired) | |
|
| ||||||||||
| Median age (IQR), yrs | 64.00 [55.00, 72.00] | 63.00 [55.00, 71.00] | 64.00 [55.00, 73.00] | 0.122 | 63.00 [55.00, 70.79] | 64.00 [55.00, 72.00] | 0.445 | 64.00 [56.00, 71.00] | 64.00 [56.00, 71.00] | 0.878 |
|
| ||||||||||
| Male sex, n (%) | 2468 (72.0) | 1567 (74.0) | 901 (68.7) | 0.001 | 721 (72.8) | 717 (72.4) | 0.880 | 424 (81.9) | 424 (81.9) | NA |
|
| ||||||||||
| Median BMI (IQR) | 24.60 [22.21, 27.44] | 24.70 [22.24, 27.52] | 24.41 [22.20, 27.25] | 0.076 | 24.60 [22.32, 27.62] | 24.30 [22.03, 26.99] | 0.072 | 24.60 [22.21, 27.00] | 24.20 [22.10, 26.82] | 0.338 |
|
| ||||||||||
| Robotic, n (%) | 400 (11.7) | 221 (10.4) | 179 (13.6) | 138 (13.9) | 128 (12.9) | 24 (4.6) | 24 (4.6) | |||
| Laparoscopic, n (%) | 3030 (88.3) | 1897 (89.6) | 1133 (86.4) | 0.005 | 853 (86.1) | 863 (87.1) | 0.555 | 494 (95.4) | 494 (95.4) | NA |
|
| ||||||||||
| Previous abdominal surgery, n (%) | 670 (20.2) | 399 (19.8) | 271 (20.8) | 0.508 | 194 (19.6) | 202 (20.4) | 0.694 | 63 (12.2) | 63 (12.2) | NA |
|
| ||||||||||
| Year of surgery, n (%) | ||||||||||
| 2004–2009 | 193 (5.6) | 132 (6.2) | 61 (4.6) | 61 (6.2) | 53 (5.3) | 19 (3.7) | 19 (3.7) | |||
| 2010–2015 | 968 (28.2) | 617 (29.1) | 351 (26.8) | 279 (28.2) | 279 (28.2) | 127 (24.5) | 127 (24.5) | |||
| 2016–2021 | 2269 (66.2) | 1369 (64.6) | 900 (68.6) | 0.028 | 651 (65.7) | 659 (66.5) | 0.325 | 372 (71.8) | 372 (71.8) | NA |
|
| ||||||||||
| ASA score, n (%) | ||||||||||
| 1/2 | 2356 (68.7) | 1454 (68.7) | 902 (68.8) | 688 (69.4) | 682 (68.8) | 391 (75.5) | 391 (75.5) | |||
| 3/4 | 1071 (31.3) | 662 (31.3) | 409 (31.2) | 0.987 | 303 (30.6) | 309 (31.2) | 0.807 | 127 (24.5) | 127 (24.5) | NA |
|
| ||||||||||
| Tumor type, n (%) | ||||||||||
| HCC | 3026 (88.4) | 1989 (94.0) | 1037 (79.5) | 926 (93.4) | 923 (93.1) | 514 (99.2) | 514 (99.2) | |||
| ICC/ cholangiohepatoma | 396 (11.6) | 128 (6.0) | 268 (20.5) | <0.001 | 65 (6.6) | 68 (6.9) | 0.815 | 4 (0.8) | 4 (0.8) | NA |
|
| ||||||||||
| Median tumor size, mm (IQR) | 29.00 [20.00, 40.00] | 26.00 [20.00, 37.00] | 30.00 [21.00, 45.00] | <0.001 | 30.00 [20.00, 40.00] | 30.00 [20.00, 40.00] | 0.564 | 25.00 [20.00, 33.00] | 25.00 [19.00, 35.00] | 0.417 |
|
| ||||||||||
| Multiple tumors, n (%) | 302 (8.8) | 206 (9.7) | 96 (7.3) | 0.019 | 68 (6.9) | 75 (7.6) | 0.597 | 9 (1.7) | 9 (1.7) | NA |
|
| ||||||||||
| Wedge/partial, n (%) | 2004 (58.4) | 1319 (62.3) | 685 (52.2) | 554 (55.9) | 558 (56.3) | 314 (60.6) | 314 (60.6) | |||
| Segmentectomy, n (%) | 1426 (41.6) | 799 (37.7) | 627 (47.8) | <0.001 | 437 (44.1) | 433 (43.7) | 0.888 | 204 (39.4) | 204 (39.4) | NA |
|
| ||||||||||
| Concomitant minor surgery excluding cholecystectomy, n (%) | 113 (3.3) | 86 (4.1) | 27 (2.1) | 0.002 | 22 (2.2) | 21 (2.1) | 1.000 | 2 (0.4) | 2 (0.4) | NA |
|
| ||||||||||
| Hilar lymph node dissection, n (%) | 112 (3.3) | 9 (0.4) | 103 (7.9) | <0.001 | 9 (0.9) | 13 (1.3) | 0.453 | 1 (0.2) | 1 (0.2) | NA |
|
| ||||||||||
| Median Iwate difficulty score, [IQR](range) | 4.00 [3.00, 6.00] (1, 9) | 4.00 [2.00, 6.00] (1, 9) | 4.00 [3.00, 7.00] (1, 9) | <0.001 | 4.00 [3.00, 6.00] (1, 9) | 4.00 [3.00, 6.00] (1, 9) | 0.870 | 4.00 [3.00, 6.00] (1, 8) | 4.00 [3.00, 6.00] (1, 8) | NA |
|
| ||||||||||
| Iwate difficulty, n (%) | ||||||||||
| Low | 1533 (44.7) | 1053 (49.7) | 480 (36.6) | 418 (42.2) | 406 (41.0) | 249 (48.1) | 249 (48.1) | |||
| Intermediate | 1253 (36.6) | 758 (35.8) | 495 (37.8) | 364 (36.7) | 383 (38.6) | 182 (35.1) | 182 (35.1) | |||
| High | 642 (18.7) | 306 (14.5) | 336 (25.6) | <0.001 | 209 (21.1) | 202 (20.4) | 0.856 | 87 (16.8) | 87 (16.8) | NA |
For PSM of comparison of Child A cirrhotic against non-cirrhotic liver in Table 1 and 2, patients of one strata were matched 1:1, using nearest neighbour matching without replacement or discard, utilizing logit link, to patients of the other strata. To improve matching, a small caliper was used to achieve good balance of < 0.1 across all variables after matching. During matching, any patient with missing data in any of the variables used for matching would be discarded. Similar methodology was employed for PSM comparison in table 3–6, comparing Child A to Child B, and cirrhosis with portal hypertension to cirrhosis without portal hypertension.
Table 2.
Comparison between perioperative outcomes of MILR in Child’s A cirrhosis vs non-cirrhosis
| Entire unmatched cohort | 1:1 PSM (nearest neighbour) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| All (N = 3430) | Childs A Cirrhosis (N = 2118) | Non-cirrhosis (N = 1312) | P-value | Childs A Cirrhosis (N = 991) | Non-cirrhosis (N = 991) | P-value (paired) | Childs A Cirrhosis (N = 518) | Non-cirrhosis (N = 518) | P-value (paired) | |
| Open conversion, n (%) | 183 (5.3) | 127 (6.0) | 56 (4.3) | 0.035 | 62 (6.3) | 39 (3.9) | 0.024 | 32 (6.2) | 18 (3.5) | 0.050 |
| Median operating time (IQR), min | 190.00 [135.00, 253.75] | 190.00 [134.00, 250.00] | 195.00 [140.00, 260.00] | 0.062 | 192.00 [130.00, 260.00] | 190.00 [135.00, 252.00] | 0.479 | 190.00 [135.00, 250.00] | 185.00 [130.00, 245.50] | 0.532 |
| Median blood loss (IQR), ml | 150.00 [50.00, 300.00] | 150.00 [50.00, 300.00] | 150.00 [50.00, 300.00] | 0.538 | 170.00 [50.00, 350.00] | 150.00 [50.00, 300.00] | <0.001 | 150.00 [50.00, 300.00] | 100.00 [50.00, 255.00] | 0.014 |
| Blood loss > 500 mls, n (%) | 343 (10.4) | 232 (11.4) | 111 (8.8) | 0.018 | 132 (13.9) | 81 (8.5) | 0.001 | 51 (10.3) | 44 (8.7) | 0.380 |
| Intraoperative blood transfusion, n (%) | 216 (6.3) | 157 (7.4) | 59 (4.5) | 0.001 | 77 (7.8) | 39 (3.9) | <0.001 | 33 (6.4) | 22 (4.2) | 0.178 |
| Pringle maneuver applied, n (%) | 1626 (48.0) | 942 (45.1) | 684 (52.8) | <0.001 | 474 (48.5) | 495 (50.6) | 0.295 | 258 (50.2) | 257 (50.1) | 1.000 |
| Median postoperative stay, d (IQR), mean (SD) | 5.00 [4.00, 7.00], 6.56 (6.80) | 5.00 [4.00, 7.00], 6.74 (7.47) | 5.00 [4.00, 7.00], 6.28 (5.53) | 0.001 | 5.00 [4.00, 7.00], 6.87 (9.51) | 5.00 [4.00, 7.00], 6.31 (4.81) | 0.114 | 6.00 [4.00, 8.00], 6.84 (5.68) | 5.00 [4.00, 7.00], 6.55 (5.10) | 0.104 |
| Postoperative morbidity, n (%) | 517 (15.1) | 349 (16.5) | 168 (12.8) | 0.004 | 162 (16.4) | 116 (11.7) | 0.004 | 84 (16.3) | 48 (9.3) | 0.001 |
| Major morbidity (Clavien-Dindo grade> 2), n (%) | 120 (3.5) | 75 (3.6) | 45 (3.4) | 0.940 | 36 (3.6) | 30 (3.0) | 0.525 | 17 (3.3) | 15 (2.9) | 0.855 |
| Reoperation, n (%) | 12 (0.3) | 10 (0.5) | 2 (0.2) | 0.147 | 7 (0.7) | 1 (0.1) | 0.077 | 3 (0.6) | 1 (0.2) | 0.617 |
| 30-day readmission, n (%) | 65 (1.9) | 45 (2.1) | 20 (1.5) | 0.260 | 24 (2.4) | 17 (1.7) | 0.349 | 9 (1.7) | 6 (1.2) | 0.606 |
| 30-day mortality, n (%) | 6 (0.2) | 6 (0.3) | 0 (0.0) | 0.089 | 2 (0.2) | 0 (0.0) | 0.480 | 1 (0.2) | 0 (0.0) | 1.000 |
| In-hospital mortality, n (%) | 12 (0.3) | 12 (0.6) | 0 (0.0) | 0.005 | 6 (0.6) | 0 (0.0) | 0.041 | 1 (0.2) | 0 (0.0) | 1.000 |
| 90-day mortality, n (%) | 11 (0.3) | 11 (0.5) | 0 (0.0) | 0.009 | 4 (0.4) | 0 (0.0) | 0.134 | 1 (0.2) | 0 (0.0) | 1.000 |
Table 3.
Comparison between baseline characteristics of MILR in 2118 Childs A vs 245 Childs B cirrhosis
| Entire unmatched cohort | 1:1 PSM (nearest neighbour matching) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| All (N = 2363) | Childs A (N =2118) | Childs B (N = 245) | P-value | Childs A (N = 215) | Childs B (N = 215) | P-value (paried) | Childs A (N = 191) | Childs B (N = 191) | P-value (paired) | |
|
| ||||||||||
| Median age (IQR), yrs | 63.00 [55.00, 71.00] | 63.00 [55.00, 71.00] | 61.00 [51.00, 69.00] | 0.002 | 59.00 [52.10, 68.00] | 61.00 [51.00, 70.00] | 0.791 | 61.00 [53.00, 69.00] | 60.00 [51.00, 68.95] | 0.068 |
|
| ||||||||||
| Male sex, n (%) | 1760 (74.5) | 1567 (74.0) | 193 (78.8) | 0.121 | 172 (80.0) | 171 (79.5) | 1.000 | 156 (81.7) | 156 (81.7) | NA |
|
| ||||||||||
| BMI | 24.70 [22.28, 27.50] | 24.70 [22.24, 27.52] | 24.94 [22.48, 27.33] | 0.873 | 24.16 [22.05, 26.65] | 24.77 [22.43, 27.12] | 0.275 | 24.72 [23.01, 27.19] | 24.40 [22.15, 26.97] | 0.113 |
|
| ||||||||||
| Robotic, n (%) | 260 (11.0) | 221 (10.4) | 39 (15.9) | 36 (16.7) | 33 (15.3) | 23 (12.0) | 23 (12.0) | |||
| Laparoscopic, n (%) | 2103 (89.0) | 1897 (89.6) | 206 (84.1) | 0.013 | 179 (83.3) | 182 (84.7) | 0.787 | 168 (88.0) | 168 (88.0) | NA |
|
| ||||||||||
| Previous abdominal surgery, n (%) | 440 (19.5) | 399 (19.8) | 41 (17.2) | 0.385 | 33 (15.3) | 38 (17.7) | 0.603 | 27 (14.1) | 27 (14.1) | NA |
|
| ||||||||||
| Year of surgery, n (%) | ||||||||||
| 2004–2009 | 148 (6.3) | 132 (6.2) | 16 (6.5) | 14 (6.5) | 14 (6.5) | 11 (5.8) | 11 (5.8) | |||
| 2010–2015 | 702 (29.7) | 617 (29.1) | 85 (34.7) | 71 (33.0) | 70 (32.6) | 65 (34.0) | 65 (34.0) | |||
| 2016–2021 | 1513 (64.0) | 1369 (64.6) | 144 (58.8) | 0.174 | 130 (60.5) | 131 (60.9) | 0.873 | 115 (60.2) | 115 (60.2) | NA |
|
| ||||||||||
| ASA score, n (%) | ||||||||||
| 1/2 | 1621 (68.7) | 1454 (68.7) | 167 (68.2) | 138 (64.2) | 147 (68.4) | 135 (70.7) | 135 (70.7) | |||
| 3/4 | 740 (31.3) | 662 (31.3) | 78 (31.8) | 0.918 | 77 (35.8) | 68 (31.6) | 0.342 | 56 (29.3) | 56 (29.3) | NA |
|
| ||||||||||
| Tumor type, n (%) | ||||||||||
| HCC | 2225 (94.2) | 1989 (94.0) | 236 (96.3) | 213 (99.1) | 213 (99.1) | 191 (100.0) | 191 (100.0) | |||
| ICC/ cholangiohepatoma | 137 (5.8) | 128 (6.0) | 9 (3.7) | 0.174 | 2 (0.9) | 2 (0.9) | 1.000 | 0 (0.0) | 0 (0.0) | NA |
|
| ||||||||||
| Median tumor size, mm (IQR) | 26.00 [20.00, 38.00] | 26.00 [20.00, 37.00] | 30.00 [20.00, 45.00] | 0.026 | 28.00 [20.00, 45.00] | 30.00 [20.00, 45.00] | 0.839 | 30.00 [20.00, 43.50] | 30.00 [20.00, 45.00] | 0.259 |
|
| ||||||||||
| Multiple tumors, n (%) | 230 (9.7) | 206 (9.7) | 24 (9.8) | 1.000 | 15 (7.0) | 17 (7.9) | 0.855 | 5 (2.6) | 5 (2.6) | NA |
|
| ||||||||||
| Wedge/ partial liver resection, n (%) | 1444 (61.1) | 1319 (62.3) | 125 (51.0) | 99 (46.0) | 116 (54.0) | 97 (50.8) | 97 (50.8) | |||
| Segmentectomy, n (%) | 919 (38.9) | 799 (37.7) | 120 (49.0) | 0.001 | 116 (54.0) | 99 (46.0) | 0.068 | 94 (49.2) | 94 (49.2) | NA |
|
| ||||||||||
| Concomitant minor surgery excluding cholecystectomy, n (%) | 100 (4.2) | 86 (4.1) | 14 (5.7) | 0.289 | 10 (4.7) | 12 (5.6) | 0.823 | 6 (3.1) | 6 (3.1) | NA |
|
| ||||||||||
| Hilar lymph node dissection, n (%) | 10 (0.4) | 9 (0.4) | 1 (0.4) | 1.000 | 2 (0.9) | 1 (0.5) | 1.000 | 0 (0.0) | 0 (0.0) | NA |
|
| ||||||||||
| Median Iwate difficulty score excluding Child score, [IQR] (range) | 4.00 [2.00, 6.00] (1, 9) | 4.00 [2.00, 6.00] (1, 9) | 4.00 [2.00, 6.00] (1, 8) | 0.046 | 4.00 [3.00, 6.00] (1, 8) | 4.00 [2.00, 6.00] (1, 8) | 0.293 | 4.00 [3.00, 6.00] (1, 8) | 4.00 [3.00, 6.00] (1, 8) | NA |
|
| ||||||||||
| Iwate difficulty exclude Child score, n (%) | ||||||||||
| Low | 1157 (49.0) | 1055 (49.8) | 102 (41.6) | 87 (40.5) | 94 (43.7) | 80 (41.9) | 80 (41.9) | |||
| Intermediate | 859 (36.4) | 760 (35.9) | 99 (40.4) | 79 (36.7) | 81 (37.7) | 71 (37.2) | 71 (37.2) | |||
| High | 346 (14.6) | 302 (14.3) | 44 (18.0) | 0.043 | 49 (22.8) | 40 (18.6) | 0.507 | 40 (20.9) | 40 (20.9) | NA |
For CEM, continuous variables were coarsened using an automatic binning algorithm based on Sturge’s rule into bins. Patients were 1:1 matched using with nearest neighbour matching without replacement within each stratum, any unmatched units in the stratum would be dropped. This methodology was applied to all 3 CEM models.
After matching, balance was checked via standardized mean difference across the covariates, with a threshold of 0.1 being indicative of tight match. Love plot of each match’s covariate balance was plotted and presented below (Supplementary data S1–S6).
For unpaired comparisons of frequencies of categorical variables, Chi squared was used. For the unpaired comparisons of median values and interquartile ranges, Mann-Whitney U test was used, and for the comparisons of mean values and standard deviations, one-way test was used. For paired sample tests, McNemar’s test was used for categorical variables and Wilcoxon Signed-Rank test was used for continuous. The statistical analyses were performed with RStudio version 1.4.1717, R version 4.1.0.
Results
A total of 3675 patients were included for final analysis (Figure 1). Of these, 1312 had no cirrhosis, 2118 had Child A cirrhosis and 245 patients had Child B cirrhosis.
Figure 1.
Selection of patients meeting study inclusion criteria.
Non-cirrhotic vs. Child A cirrhosis patients
A total of 3430 patients were enrolled in this cohort, including 2118 patients with Child A cirrhosis and 1312 patients without cirrhosis. The baseline characteristics are shown in Table 1. We conducted PSM and CEM to offset potential confounding factors. Before matching, gender (p = 0.001), year of surgery (p = 0.028), tumor type (p < 0.001), median tumor size (p < 0.001), multiple tumours (p = 0.019), surgical type (p < 0.001), concomitant minor surgery (p = 0.002), hilar lymph node dissection (p < 0.001), median difficulty score (p < 0.001), and IWATE difficulty score (p < 0.001) were statistically different between two groups. After 1:1 PSM, 991 matched pairs were included for further analysis. All the baseline characteristics of the two groups reached statistical equilibrium. Similarly, after 1:1 CEM matching, 518 matched pairs were included for further analysis and all baseline indicators reached equilibrium.
The perioperative outcomes are summarized in Table 2. In the entire unmatched cohort, the Child A cirrhosis group had a higher rate of open conversion (6.0% VS 4.3%, p = 0.035), blood loss > 500 mls (11.4% VS 8.8%, p = 0.018), intraoperative blood transfusion (7.4% VS 4.5%, p = 0.001), rate of pringle maneuver application (p < 0.001), median postoperative stay (p = 0.001), postoperative morbidity (p = 0.004), in-hospital mortality (p = 0.005), and 90-day mortality (p = 0.009) compared to non-cirrhotics. After 1:1 PSM, rate of open conversion (p = 0.024), median blood loss (p < 0.001), blood loss >500 mls (p = 0.001), intraoperative blood transfusion (p < 0.001), postoperative morbidity (p = 0.004), and in-hospital mortality (p = 0.041) were higher in the Child A cirrhosis group. In the 1:1 CEM cohort, the Child A cirrhosis group had higher rate of open conversion (p = 0.05), greater median blood loss (p = 0.014) and increased postoperative morbidity (p = 0.001).
Child A vs. Child B cirrhosis patients
In the entire unmatched cohort (2118 Child A group versus 245 Child B), there were some differences in median age, proportion of robotic versus laparoscopic approach, extent of surgery and median Iwate difficulty score excluding Child score (Table 3). After 1:1PSM and 1:1 CEM, 215 and 191 pairs of patients were included for further comparison, respectively. All baseline differences disappeared in the matched patient cohorts.
Table 4 summarizes the perioperative outcomes. After 1:1PSM, the Child B cirrhosis group had a longer postoperative stay (p = 0.001) and a higher rate of major morbidity (p = 0.012). In the 1:1CEM cohort, Child B patients had higher rates of blood transfusion (p = 0.002), longer postoperative stay (p < 0.001), and greater major morbidity (p = 0.006).
Table 4.
Comparison between perioperative outcomes of MILR in Child’s A vs B cirrhosis
| Entire unmatched cohort | 1:1 PSM (nearest neighbour) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| All (N = 2363) | Childs A (N=2118) | Childs B (N = 245) | P-value | Childs A (N = 215) | Childs B (N = 215) | P-value (paried) | Childs A (N = 191) | Childs B (N = 191) | P-value (paired) | |
| Open conversion, n (%) | 138 (5.8) | 127 (6.0) | 11 (4.5) | 0.419 | 10 (4.7) | 8 (3.7) | 0.803 | 10 (5.2) | 9 (4.7) | 1.000 |
| Median operating time (IQR), min | 190.00 [135.00, 250.00] | 190.00 [134.00, 250.00] | 201.50 [144.00, 260.00] | 0.164 | 200.00 [143.00, 269.50] | 194.00 [143.00, 260.00] | 0.469 | 200.00 [145.00, 258.00] | 200.50 [149.25, 260.00] | 0.737 |
| Median blood loss (IQR), ml | 150.00 [50.00, 300.00] | 150.00 [50.00, 300.00] | 150.00 [50.00, 350.00] | 0.078 | 200.00 [52.50, 400.00] | 150.00 [60.00, 350.00] | 0.738 | 150.00 [100.00, 350.00] | 180.00 [84.50, 400.00] | 0.482 |
| Blood loss > 500 mls, n (%) | 270 (11.9) | 232 (11.4) | 38 (15.9) | 0.056 | 26 (12.4) | 32 (15.2) | 0.568 | 26 (13.9) | 32 (17.2) | 0.451 |
| Intraoperative blood transfusion, n (%) | 200 (8.5) | 157 (7.4) | 43 (17.6) | <0.001 | 29 (13.5) | 37 (17.2) | 0.332 | 15 (7.9) | 37 (19.4) | 0.002 |
| Pringle maneuver applied, n (%) | 1045 (44.8) | 942 (45.1) | 103 (42.9) | 0.571 | 93 (44.3) | 97 (46.2) | 0.766 | 94 (50.3) | 82 (43.6) | 0.213 |
| Median postoperative stay, d (IQR), mean (SD) | 6.00 [4.00, 8.00], 7.05 (7.60) | 5.00 [4.00, 7.00], 6.74 (7.47) | 7.00 [5.00, 13.00], 9.66 (8.21) | <0.001 | 6.00 [4.00, 10.00], 9.07 (18.02) | 7.00 [5.00, 13.00], 9.53 (7.11) | 0.001 | 6.00 [4.00, 8.00], 7.09 (6.42) | 8.00 [5.00, 13.00], 9.70 (7.67) | <0.001 |
| Postoperative morbidity, n (%) | 408 (17.3) | 349 (16.5) | 59 (24.1) | 0.004 | 40 (18.6) | 50 (23.3) | 0.282 | 37 (19.4) | 45 (23.6) | 0.374 |
| Major morbidity (Clavien-Dindo grade> 2), n (%) | 100 (4.2) | 75 (3.6) | 25 (10.2) | <0.001 | 7 (3.3) | 20 (9.3) | 0.012 | 4 (2.1) | 18 (9.4) | 0.006 |
| Reoperation, n (%) | 12 (0.5) | 10 (0.5) | 2 (0.8) | 0.358 | 0 (0.0) | 2 (0.9) | 0.480 | 0 (0.0) | 0 (0.0) | NA |
| 30-day readmission, n (%) | 50 (2.1) | 45 (2.1) | 5 (2.0) | 1.000 | 4 (1.9) | 4 (1.9) | 1.000 | 7 (3.7) | 3 (1.6) | 0.343 |
| 30-day mortality, n (%) | 10 (0.4) | 6 (0.3) | 4 (1.6) | 0.014 | 1 (0.5) | 3 (1.4) | 0.617 | 0 (0.0) | 2 (1.0) | 0.480 |
| In-hospital mortality, n (%) | 14 (0.6) | 12 (0.6) | 2 (0.8) | 0.650 | 1 (0.5) | 1 (0.5) | 1.000 | 0 (0.0) | 2 (1.0) | 0.480 |
| 90-day mortality, n (%) | 16 (0.7) | 11 (0.5) | 5 (2.0) | 0.019 | 2 (0.9) | 3 (1.4) | 1.000 | 0 (0.0) | 2 (1.0) | 0.480 |
Cirrhotic patients (all Child grades) without vs. with PHT
The entire unmatched cohort contained 626 patients with cirrhosis and PHT, and 1701 patients with cirrhosis but without PHT (Table 5). After 1:1 PSM and 1:1 CEM, 450 and 290 matched pair were included in each group, respectively, and all baseline characteristics were balanced (Table 5).
Table 5.
Comparison between baseline characteristics of MILR in 2361 cirrhosis patients with and without portal hypertension ()
| Entire unmatched cohort | 1:1 PSM (nearest neighbour matching) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| All (N = 2327) | Cirrhosis PHT (N = 626) | Cirrhosis NPHT (N = 1701) | P-value | Cirrhosis PHT (N = 450) | Cirrhosis NPHT (N = 450) | P-value | Cirrhosis PHT (N = 290) | Cirrhosis NPHT (N = 290) | P-value (paired) | |
|
| ||||||||||
| Median age (IQR), yrs | 63.00 [55.00, 71.00] | 63.00 [55.00, 71.00] | 63.00 [55.00, 71.00] | 0.734 | 62.00 [54.00, 70.00] | 64.00 [55.00, 70.00] | 0.217 | 61.00 [54.00, 69.00] | 62.00 [55.00, 69.00] | 0.607 |
|
| ||||||||||
| Male sex, n (%) | 1731 (74.4) | 468 (74.8) | 1263 (74.3) | 0.844 | 339 (75.3) | 335 (74.4) | 0.822 | 230 (79.3) | 230 (79.3) | NA |
|
| ||||||||||
| Median BMI (IQR) | 24.68 [22.22, 27.45] | 25.08 [22.84, 28.00] | 24.50 [22.10, 27.25] | <0.001 | 24.93 [22.50, 27.70] | 25.00 [22.49, 27.68] | 0.826 | 24.80 [22.70, 27.40] | 24.34 [22.16, 27.24] | 0.426 |
|
| ||||||||||
| Robotic, n (%) | 260 (11.2) | 56 (8.9) | 204 (12.0) | 43 (9.6) | 41 (9.1) | 12 (4.1) | 12 (4.1) | |||
| Laparoscopic, n (%) | 2067 (88.8) | 570 (91.1) | 1497 (88.0) | 0.046 | 407 (90.4) | 409 (90.9) | 0.907 | 278 (95.9) | 278 (95.9) | NA |
|
| ||||||||||
| Previous abdominal surgery, n (%) | 436 (19.7) | 142 (22.7) | 294 (18.5) | 0.028 | 98 (21.8) | 97 (21.6) | 1.000 | 36 (12.4) | 36 (12.4) | NA |
|
| ||||||||||
| Childs A, n (%) | 2082 (89.5) | 1581 (92.9) | 501 (80.0) | 392 (87.1) | 398 (88.4) | 280 (96.6) | 280 (96.6) | |||
| Childs B, n (%) | 245 (10.5) | 120 (7.1) | 125 (20.0) | 0.028 | 58 (12.9) | 52 (11.6) | 0.561 | 10 (3.4) | 10 (3.4) | NA |
|
| ||||||||||
| Year of surgery, n (%) | ||||||||||
| 2004–2009 | 144 (6.2) | 48 (7.7) | 96 (5.6) | 33 (7.3) | 37 (8.2) | 10 (3.4) | 10 (3.4) | |||
| 2010–2015 | 692 (29.7) | 173 (27.6) | 519 (30.5) | 121 (26.9) | 118 (26.2) | 92 (31.7) | 92 (31.7) | |||
| 2016–2021 | 1491 (64.1) | 405 (64.7) | 1086 (63.8) | 0.113 | 296 (65.8) | 295 (65.6) | 0.939 | 188 (64.8) | 188 (64.8) | NA |
|
| ||||||||||
| ASA score, n (%) | ||||||||||
| 1/2 | 1604 (69.0) | 393 (62.8) | 1211 (71.3) | 298 (66.2) | 297 (66.0) | 205 (70.7) | 205 (70.7) | |||
| 3/4 | 721 (31.0) | 233 (37.2) | 488 (28.7) | <0.001 | 152 (33.8) | 153 (34.0) | 1.000 | 85 (29.3) | 85 (29.3) | NA |
|
| ||||||||||
| Tumor type, n (%) | ||||||||||
| HCC | 2194 (94.3) | 613 (97.9) | 1581 (93.0) | 441 (98.0) | 440 (97.8) | 288 (99.3) | 288 (99.3) | |||
| ICC/ cholangiohepatoma | 132 (5.7) | 13 (2.1) | 119 (7.0) | <0.001 | 9 (2.0) | 10 (2.2) | 1.000 | 2 (0.7) | 2 (0.7) | NA |
|
| ||||||||||
| Median tumor size, mm (IQR) | 26.00 [20.00, 38.00] | 25.00 [18.00, 35.00] | 27.00 [20.00, 40.00] | <0.001 | 25.00 [18.00, 35.00] | 25.00 [20.00, 35.00] | 0.620 | 23.50 [18.00, 32.00] | 25.00 [19.25, 30.00] | 0.748 |
|
| ||||||||||
| Multiple tumors, n (%) | 228 (9.8) | 50 (8.0) | 178 (10.5) | 0.088 | 35 (7.8) | 32 (7.1) | 0.807 | 4 (1.4) | 4 (1.4) | NA |
|
| ||||||||||
| Wedge/ partial liver resection, n (%) | 1420 (61.0) | 410 (65.5) | 1010 (59.4) | 291 (64.7) | 293 (65.1) | 199 (68.6) | 199 (68.6) | |||
| Segmentectomy, n (%) | 907 (39.0) | 216 (34.5) | 691 (40.6) | 0.008 | 159 (35.3) | 157 (34.9) | 0.942 | 91 (31.4) | 91 (31.4) | NA |
|
| ||||||||||
| Concomitant minor surgery excluding cholecystectomy, n (%) | 95 (4.1) | 31 (5.0) | 64 (3.8) | 0.242 | 22 (4.9) | 21 (4.7) | 1.000 | 5 (1.7) | 5 (1.7) | NA |
|
| ||||||||||
| Hilar lymph node dissection, n (%) | 9 (0.4) | 5 (0.8) | 4 (0.2) | 0.065 | 0 (0.0) | 1 (0.2) | 1.000 | 0 (0.0) | 0 (0.0) | NA |
|
| ||||||||||
| Median Iwate difficulty score, [IQR] (range) | 4.00 [3.00, 6.00] (1, 9) | 4.00 [2.00, 6.00] (1, 9) | 4.00 [3.00, 6.00] (1, 9) | 0.008 | 3.00 [2.00, 6.00] (1, 8) | 3.00 [2.00, 5.00] (1, 9) | 0.704 | 3.00 [2.00, 5.00] (1, 9) | 3.00 [2.00, 5.00] (1, 9) | NA |
|
| ||||||||||
| Iwate difficulty, n (%) | ||||||||||
| Low | 1097 (47.2) | 309 (49.4) | 788 (46.4) | 232 (51.6) | 233 (51.8) | 169 (58.3) | 169 (58.3) | |||
| Intermediate | 841 (36.2) | 236 (37.7) | 605 (35.6) | 152 (33.8) | 164 (36.4) | 97 (33.4) | 97 (33.4) | |||
| High | 388 (16.7) | 81 (12.9) | 307 (18.1) | 0.013 | 66 (14.7) | 53 (11.8) | 0.533 | 24 (8.3) | 24 (8.3) | NA |
Perioperative outcomes are summarised in Table 6. In the 1:1PSM cohort, patients with PHT experienced higher rates of blood loss > 500 mls (p = 0.003) and intraoperative blood transfusion (p = 0.025). There were no differences in other outcomes. After 1:1 CEM, the two groups had no significant differences in outcomes.
Table 6.
Comparison between perioperative outcomes of MILR in cirrhosis patients with and without PHT
| Entire unmatched cohort | 1:1 PSM (nearest neighbour) | 1:1 CEM | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| All (N = 2327) | Cirrhosis PHT (N = 626) | Cirrhosis NPHT (N = 1701) | P-value | Cirrhosis PHT (N = 450) | Cirrhosis NPHT (N = 450) | P-value | Cirrhosis PHT (N = 290) | Cirrhosis NPHT (N = 290) | P-value (paired) | |
| Open conversion, n (%) | 137 (5.9) | 49 (7.8) | 88 (5.2) | 0.021 | 38 (8.4) | 25 (5.6) | 0.118 | 25 (8.6) | 13 (4.5) | 0.067 |
| Median operating time (IQR), min | 190.00 [136.00, 252.00] | 180.00 [124.00, 245.00] | 195.00 [140.00, 255.00] | 0.010 | 180.00 [124.50, 250.00] | 185.00 [134.00, 240.00] | 0.872 | 180.00 [124.25, 240.00] | 180.00 [130.00, 243.00] | 0.563 |
| Median blood loss (IQR), ml | 150.00 [50.00, 300.00] | 200.00 [50.00, 350.00] | 110.00 [50.00, 300.00] | <0.001 | 200.00 [100.00, 400.00] | 120.00 [50.00, 300.00] | <0.001 | 200.00 [100.00, 327.50] | 150.00 [50.00, 300.00] | 0.046 |
| Blood loss > 500 mls, n (%) | 262 (11.7) | 89 (14.8) | 173 (10.6) | 0.008 | 70 (15.9) | 39 (9.0) | 0.003 | 36 (13.0) | 34 (12.5) | 0.787 |
| Intraoperative blood transfusion, n (%) | 198 (8.5) | 65 (10.4) | 133 (7.8) | 0.061 | 51 (11.3) | 31 (6.9) | 0.025 | 24 (8.3) | 17 (5.9) | 0.296 |
| Pringle maneuver applied, n (%) | 1033 (45.0) | 281 (45.4) | 752 (44.8) | 0.850 | 199 (44.5) | 186 (41.6) | 0.446 | 127 (43.9) | 113 (39.6) | 0.298 |
| Median postoperative stay, d (IQR), mean (SD) | 6.00 [4.00, 8.00], 7.09 (7.65) | 5.00 [4.00, 8.00], 7.08 (6.96) | 6.00 [4.00, 8.00], 7.09 (7.89) | 0.231 | 5.77 [4.00, 8.00], 7.01 (6.21) | 5.00 [4.00, 7.00], 6.98 (12.59) | 0.141 | 5.00 [4.00, 7.00], 7.04 (7.23) | 5.00 [4.00, 8.00], 6.82 (5.29) | 0.887 |
| Postoperative morbidity, n (%) | 402 (17.3) | 128 (20.5) | 274 (16.2) | 0.017 | 78 (17.4) | 73 (16.3) | 0.721 | 54 (18.6) | 42 (14.5) | 0.201 |
| Major morbidity (Clavien-Dindo grade> 2), n (%) | 98 (4.2) | 33 (5.3) | 65 (3.8) | 0.155 | 23 (5.1) | 15 (3.3) | 0.243 | 11 (3.8) | 10 (3.4) | 1.000 |
| Reoperation, n (%) | 12 (0.5) | 3 (0.5) | 9 (0.5) | 1.000 | 2 (0.4) | 2 (0.4) | 1.000 | 1 (0.3) | 1 (0.3) | 1.000 |
| 30-day readmission, n (%) | 50 (2.2) | 12 (1.9) | 38 (2.2) | 0.754 | 11 (2.4) | 14 (3.1) | 0.663 | 6 (2.1) | 8 (2.8) | 0.789 |
| 30-day mortality, n (%) | 9 (0.4) | 2 (0.3) | 7 (0.4) | 1.000 | 1 (0.2) | 2 (0.4) | 1.000 | 2 (0.7) | 1 (0.3) | 1.000 |
| In-hospital mortality, n (%) | 13 (0.6) | 2 (0.3) | 11 (0.6) | 0.533 | 1 (0.2) | 1 (0.2) | 1.000 | 2 (0.7) | 2 (0.7) | 1.000 |
| 90-day mortality, n (%) | 15 (0.6) | 2 (0.3) | 13 (0.8) | 0.380 | 1 (0.2) | 3 (0.7) | 0.617 | 2 (0.7) | 3 (1.0) | 1.000 |
Discussion
Minor resections (wedge and segmentectomy) in AL segments are often considered the ‘easiest’ technical resections in MILR by almost all existing DSS. Both the Ban DSS [6] and its later modification, the Iwate criteria [9], allocate fewer difficulty points to lesions in superficial, anterolateral segments. In the Hasegawa score, lesions in anterolateral segments 2/3/4 are accorded lower scores (0 points) versus lesions in S5/6 (1 point) and S7/8 (2 points) [24]. The Southampton score assigns 0 difficulty points for minor resections, versus 2 points for technically major and 4 points for anatomically major resections respectively [7]. Finally, in the IMM score, wedge resection and left lateral sectionectomies are considered to be the simplest resections, while anterolateral segmentectomies are intermediate level [8]. As such, these cases are thought to be suitable for novice surgeons in the early stages of their learning curve for LLR.
In this large multicentre study, we found that patient with CP A cirrhosis who underwent minor LLR in the AL segments experienced higher rates of open conversion, blood loss and greater post-operative morbidity compared to patients with no cirrhosis. Although patient cohorts were matched to ensure there were no differences in preoperative baseline characteristics, the group with CP A cirrhosis had inferior perioperative outcomes. This implies that the presence of underlying LC does in fact, increase the complexity of LLR, even for technically ‘easy’ LLRs.
Previous studies had suggested that LC increased the complexity of LLR. Hobeika et al analyzed 282 matched pairs of patients with and without cirrhosis who underwent LLR across 27 centers [13]. The LC group experienced higher rates of severe complications (odds ratio (OR) 1.74, 95 percent confidence interval (CI) 0.92 to 3.41; p = 0.096), PHLF (OR 7.13, 0.91 to 323.10; p = 0.068), and a higher risk of death (OR 5.13, 1.08 to 48.61; p = 0.039) versus non-cirrhotics. Similarly, Goh et al reported a single-center matched study compared MILR in patients with LC versus without, and found that the LC group had greater blood loss, higher blood transfusion rates, increased conversion and postoperative morbidity, as well as longer operation times and postoperative hospital stay [16]. In the aforementioned study, 41% of lesions were located in ‘difficult segments (I, IVa, VII, VIII). Hence, a major difference from our study was that both the Hobeika et al and Goh et al studies contained a mixture of major and minor LLRs. Interestingly, Goh et al found that upon subset analyses, LC only significantly impacted outcomes of more complex resection [Institut Mutualiste Montsouris (IMM) II/III] and not simpler IMM I resections [16].
Hence, in contrast to these previous studies; the present study focused exclusively on “technically simple” minor resections in the AL segments, so as to minimise the confounding effect of tumour location and extent of resection on perioperative outcomes. Furthermore, only patients with primary malignancies were included unlike previous studies [13,16], as a major confounding factor in these studies was the inclusion of patients with other pathologies including benign lesions and colorectal liver metastases which occurred more frequently in the non-cirrhotic cohort. In the present analysis, despite controlling for various factors the LC cohort was associated with significantly poorer outcomes compared to the non-cirrhotic arm. These results suggest that there is room to further refine existing DSS for MILR by incorporating the presence of cirrhosis as a variable into the computation of scores. This will facilitate a more accurate preoperative estimation of technical difficulty, so as to allow appropriate case selection according to the expertise level of individual surgeons.
In the second part of this study, we compared outcomes following LLR for patients with CP B versus CP A. Major morbidity rates were higher in CP B cirrhosis, and this ultimately resulted in longer LOS as well. It is worthwhile to note, however, that intraoperative outcomes (operating time, blood loss, transfusion, conversion rates) were similar. These results were similar to Watanabe’s study [25], where 357 LLR patients were divided into three cohorts as follows: Child-Pugh A (n=280), Child-Pugh B7 (n=42) and Child-Pugh B8/9 (n=35)(22). 14% of patients in the Child-Pugh A cohort underwent anatomic resection while all patients in the advanced cirrhosis group had non-anatomical resections. Patients with Child-Pugh B cirrhosis had significantly more post-operative complications versus Child-Pugh A (P=0.033), with no differences in operative time and blood loss. Furthermore, the Child-Pugh B8/9 group had significantly higher complication rate than other two groups (Child-Pugh A vs. B7 vs. B8/9: 8.2% vs. 9.6% vs. 26%, respectively; p = 0.010). Troisi et al performed a multicenter PSM study for patients with Child-Pugh B cirrhosis who underwent open versus LLR for hepatocellular carcinoma [26]. Amongst the LLR cohort, patients with Child-Pugh B9 developed more complications compared to those with B8 and B7 cirrhosis. From this, we can infer that although the severity of cirrhosis might not directly impact the conduct of surgery, patients with severe liver disease are more systemically unwell and hence they are at greater risk of post-operative complications. One important application of DSS for MILR is to enable appropriate preoperative risk counselling for patients. To this end, the inclusion of the presence of cirrhosis as well as its severity into future DSS would enhance its effectiveness.
Finally, we found that patients with cirrhosis as well as PHT had slightly higher rates of blood loss (P<0.001) and blood transfusion (P=0.025) on PSM compared to their counterparts with no PHT, although the differences did not persist on CEM. This suggests that the MIS approach may partially mitigate the risks of LR in this group of patients, particularly that of bleeding. The magnified laparoscopic view in MILR allows precise dissection of vasculobiliary structures, which may be ligated by surgical clips and energy devices [26], while continuous pneumoperitoneum pressure tamponades venous ooze from the liver surface.
A key strength of this study is that it contains one of the largest multicentre patient cohorts to date, from both Eastern and Western units and focussed on a specific subset of patients undergoing MILR. However, there are several limitations that must be highlighted. The retrospective nature of this study, as well as the inclusion of patients over a relatively long period meant that information and selection biases are inherent despite the utilization of two robust statistical matching techniques. Most cases were performed in high-volume centers and the results may not be readily generalizable. However, the recent move to centralise complex surgical procedures to specialty units may mean that this is a realistic portrayal of surgical practises in the near future. Almost 40% of resections in this study were segmentectomies. As such, the median Iwate score was 4, places them in the ‘intermediate’ difficulty category, hence cases were slightly more complex despite being in the ‘easy’ anterolateral segments. The proportion of patients with CP B cirrhosis and PHT were also relatively small. Furthermore, although the measurement of hepatic venous-portal gradient is the gold standard for the diagnosis of portal hypertension, this invasive method is not routinely used in most centers prior to liver resection. Hence, a simple definition was used for the presence of clinically-significant PHT due to the limitations of this study. Finally, as an international multicenter study, there would be heterogeneity in each individual center’s surgical technique, selection process and postoperative management.
Conclusion
This study demonstrated that the presence and severity of LC affected and compounded the difficulty of LLR even for ‘simple’ minor resections in AL segments, resulting in higher rates of open conversion, blood loss and greater post-operative morbidity. Inclusion of cirrhosis into future DSS would be important for case selection and would allow more accurate benchmarking of cases for surgical audits.
Supplementary Material
Acknowledgement
Funding
Dr T. P. Kingham was partially supported by the US National Cancer Institute MSKCC Core Grant number P30 CA008748 for this study
Dr M. Yin was partially funded by the Research Project of Zhejiang Provincial Public Welfare Fund project in the Field of Social development (LGF20H160028)
Dr B.K.P. Goh was partially funded by a grant from the Intuitive Foundation for this work. Any research findings, conclusions, or recommendations expressed in this work are those of the authors and not of the Intuitive Foundation
Collaborators
International robotic and laparoscopic liver resection study group investigators
Nicholas Syn (Yong Loo Lin School of Medicine, National University of Singapore)
Mikel Gastaca (Hepatobiliary Surgery and Liver Transplantation Unit, Biocruces Bizkaia Health Research Institute, Cruces University Hospital, University of the Basque Country, Bilbao, Spain)
Celine De Meyere (Department of Digestive and Hepatobiliary/Pancreatic Surgery, Groeninge Hospital, Kortrijk, Belgium)
Juul Meurs,(Department of Digestive and Hepatobiliary/Pancreatic Surgery, Groeninge Hospital, Kortrijk, Belgium)
Kelvin K. Ng (Division of Hepatobiliary and Pancreatic Surgery, Department of Surgery, Prince of Wales Hospital, The Chinese University of Hong Kong, New Territories, Hong Kong SAR, China)
Kit-Fai Lee (Division of Hepatobiliary and Pancreatic Surgery, Department of Surgery, Prince of Wales Hospital, The Chinese University of Hong Kong, New Territories, Hong Kong SAR, China)
Pavel Tarakanov (Department of Hepato-Pancreato-Biliary Surgery, Moscow Clinical Scientific Center, Moscow, Russia)
Ruslan Alikhanov (Department of Hepato-Pancreato-Biliary Surgery, Moscow Clinical Scientific Center, Moscow, Russia)
Nita Thiruchelvam (Hepatopancreatobiliary Unit, Department of Surgery, Changi General Hospital, Singapore)
Jae Young Jang (Department of General Surgery, CHA Bundang Medical Center, CHA University School of Medicine, Seongnam, Korea)
Masayuki Kojima (Department of Surgery, Fujita Health University School of Medicine, Aichi, Japan)
Jaime Arthur Pirola Kruger (Liver Surgery Unit, Department of Gastroenterology, University of Sao Paulo School of Medicine, Sao Paulo, Brazil)
Fabricio Ferreira Coelho (Liver Surgery Unit, Department of Gastroenterology, University of Sao Paulo School of Medicine, Sao Paulo, Brazil)
Victor Lopez-Lopez (Department of General, Visceral and Transplantation Surgery, Clinic and University Hospital Virgen de la Arrixaca, IMIB-ARRIXACA, El Palmar, Murcia, Spain)
Margarida Casellas I Robert (Hepatobiliary and Pancreatic Surgery Unit, Department of Surgery, Dr. Josep Trueta Hospital, IdIBGi, Girona, Spain)
Roberto Montalti (Department of Clinical Medicine and Surgery, Division of HPB, Minimally Invasive and Robotic Surgery, Federico II University Hospital Naples, Naples, Italy)
Mariano Giglio (Department of Clinical Medicine and Surgery, Division of HPB, Minimally Invasive and Robotic Surgery, Federico II University Hospital Naples, Naples, Italy)
Boram Lee (Department of Surgery, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seoul, Korea)
Mizelle D’Silva, (Department of Surgery, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seoul, Korea)
Hao-Ping Wang Department of Surgery, Chang Gung Memorial Hospital, Kaohsiung
Franco Pascual (Department of Hepatobiliary Surgery, Assistance Publique Hopitaux de Paris, Centre Hepato-Biliaire, Paul-Brousse Hospital, Villejuif, France)
Mansour Saleh (Department of Hepatobiliary Surgery, Assistance Publique Hopitaux de Paris, Centre Hepato-Biliaire, Paul-Brousse Hospital, Villejuif, France)
Shian Yu (Department of Hepatobiliary Surgery, Affiliated Jinhua Hospital, Zhejiang University School of Medicine, Jinhua, China)
Simone Vani (Hepatobiliary Surgery Unit, Fondazione Policlinico Universitario A. Gemelli, IRCCS, Catholic University of the Sacred Heart, Rome, Italy)
Francesco Ardito, (Hepatobiliary Surgery Unit, Fondazione Policlinico Universitario A. Gemelli, IRCCS, Catholic University of the Sacred Heart, Rome, Italy)
Ugo Giustizieri (HPB Surgery, Hepatology and Liver Transplantation, Fondazione IRCCS Istituto Nazionale Tumori di Milano, Milan, Italy)
Davide Citterio (HPB Surgery, Hepatology and Liver Transplantation, Fondazione IRCCS Istituto Nazionale Tumori di Milano, Milan, Italy)
Federico Mocchegiani (HPB Surgery and Transplantation Unit, United Hospital of Ancona, Department of Experimental and Clinical Medicine Polytechnic University of Marche)
Marco Colasanti (Division of General Surgery and Liver Transplantation, San Camillo Forlanini Hospital, Rome, Italy)
Giammauro Berardi (Division of General Surgery and Liver Transplantation, San Camillo Forlanini Hospital, Rome, Italy)
Yoelimar Guzmán (General & Digestive Surgery, Hospital Clínic, Barcelona, Spain)
Kevin P. Labadie (Department of Surgery, University of Washington Medical Center. Seattle, USA)
Maria Conticchio (Unit of Hepato-Pancreatc-Biliary Surgery, “F. Miulli” General Regional Hospital, Acquaviva delle Fonti, Bari, Italy)
Epameinondas Dogeas (Department of Surgery, Division of Hepatobiliary and Pancreatic Surgery, University of Pittsburgh Medical Center, Pittsburgh, PA, USA)
Emanuele F. Kauffmann (Division of General and Transplant Surgery, University of Pisa, Pisa, Italy)
Mario Giuffrida (Hepatobiliary Surgery Unit, Department of Medicine and Surgery, University of Parma, Parma, Italy)
Daniele Sommacale (Department of Digestive and Hepatobiliary and Pancreatic Surgery, AP-HP, Henri-Mondor Hospital, Creteil, France)
Alexis Laurent, (Department of Digestive and Hepatobiliary and Pancreatic Surgery, AP-HP, Henri-Mondor Hospital, Creteil, France)
Paolo Magistri (HPB Surgery and Liver Transplant Unit, University of Modena and Reggio Emilia, Modena, Italy)
Kohei Mishima (Center for Advanced Treatment of Hepatobiliary and Pancreatic Diseases, Ageo Central General Hospital, Saitama, Japan)
Moritz Schmelzle, (Department of Surgery, Campus Charité Mitte and Campus Virchow-Klinikum, Charité-Universitätsmedizin, Corporate Member of Freie Universität Berlin, and Berlin Institute of Health, Berlin, Germany)
Felix Krenzien (Department of Surgery, Campus Charité Mitte and Campus Virchow-Klinikum, Charité-Universitätsmedizin, Corporate Member of Freie Universität Berlin, and Berlin Institute of Health, Berlin, Germany)
Prashant Kadam (Department of Hepatopancreatobiliary and Liver Transplant Surgery, University Hospitals Birmingham NHS Foundation Trust, Birmingham, United Kingdom)
Chung-Ngai Tang (Department of Surgery, Pamela Youde Nethersole Eastern Hospital, Hong Kong SAR, China)
Jacob Ghotbi (The Intervention Centre and Department of HPB Surgery, Oslo University Hospital, Institute of Clinical Medicine, University of Oslo, Oslo, Norway)
Åsmund Avdem Fretland (The Intervention Centre and Department of HPB Surgery, Oslo University Hospital, Institute of Clinical Medicine, University of Oslo, Oslo, Norway)
Fabio Forchino (Department of General and Oncological Surgery. Mauriziano Hospital, Turin, Italy)
Bernardo Dalla Valle (General and Hepatobiliary Surgery, Department of Surgery, Dentistry, Gynecology and Pediatrics University of Verona, GB Rossi Hospital, Verona, Italy)
Alessandro Mazzotta (Department of Digestive, Oncologic and Metabolic Surgery, Institute Mutualiste Montsouris, Universite Paris Descartes, Paris, France)
Phan Phuoc Nghia (Department of Hepatopancreatobiliary Surgery, University Medical Center, University of Medicine and Pharmacy, Ho Chi Minh City, Vietnam)
Francois Cauchy (Department of HPB Surgery and Liver Transplantation, Beaujon Hospital, Clichy, France)
Qu Liu, (Faculty of Hepatopancreatobiliary Surgery, the First Medical Center of Chinese People’s Liberation Army (PLA) General Hospital, Beijing, China)
Yoshikuni Kawaguchi (Hepato-Biliary-Pancreatic Surgery Division, Department of Surgery, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan)
Footnotes
- Dr Goh BK has received travel grants, honorarium and research grants from Johnson and Johnson, Olympus and Intuitive Surgery.
- Dr Marino MV is a consultant for CAVA robotics LLC.
- Johann Pratschke reports a research grant from Intuitive Surgical Deutschland GmbH and personal fees or non-financial support from Johnson & Johnson, Medtronic, AFS Medical, Astellas, CHG Meridian, Chiesi, Falk Foundation, La Fource Group, Merck, Neovii, NOGGO, pharma-consult Peterson, and Promedicis.
- Moritz Schmelzle reports personal fees or other support outside of the submitted work from Merck, Bayer, ERBE, Amgen, Johnson & Johnson, Takeda, Olympus, Medtronic, Intuitive.
- Fernando Rotellar reports speaker fees and support outside the submitted work from Integra, Medtronic, Olympus, Corza, Sirtex and Johnson & Johnson.
- Roberto Troisi reports speaker fees and support outside the submitted work from Integra, Stryker, Medtronic, Medistim, MSD.
Publisher's Disclaimer: This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Ciria R, Cherqui D, Geller DA, Briceno J, Wakabayashi G. Comparative Short-term Benefits of Laparoscopic Liver Resection: 9000 Cases and Climbing. Ann Surg [Internet]. 2016. [cited 2022 Jul 15];263(4):761–77. Available from: https://pubmed.ncbi.nlm.nih.gov/26700223/ [DOI] [PubMed] [Google Scholar]
- 2.Goh BKP, Lee SY, Teo JY, Kam JH, Jeyaraj PR, Cheow PC, et al. Changing trends and outcomes associated with the adoption of minimally invasive hepatectomy: a contemporary single-institution experience with 400 consecutive resections. Surg Endosc. 2018. Nov 1;32(11):4658–65. [DOI] [PubMed] [Google Scholar]
- 3.Wakabayashi G, Cherqui D, Geller DA, Buell JF, Kaneko H, Han HS, et al. Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg [Internet]. 2015. Apr 1 [cited 2022 Jul 23];261(4):619–29. Available from: https://pubmed.ncbi.nlm.nih.gov/25742461/ [DOI] [PubMed] [Google Scholar]
- 4.Hilal MA, Aldrighetti L, Dagher I, Edwin B, Troisi RI, Alikhanov R, et al. The Southampton Consensus Guidelines for Laparoscopic Liver Surgery: From Indication to Implementation. Ann Surg. 2018. Jul 1;268(1):11–8. [DOI] [PubMed] [Google Scholar]
- 5.Linn YL, Wu AG, Han HS, et al. Systematic review and meta-analysis of difficulty scoring systems for laparoscopic and robotic liver resections. J Hepatobiliary Pancreat Sci 2022. [DOI] [PubMed] [Google Scholar]
- 6.Ban D, Tanabe M, Ito H, Otsuka Y, Nitta H, Abe Y, et al. A novel difficulty scoring system for laparoscopic liver resection. J Hepatobiliary Pancreat Sci. 2014. Oct 1;21(10):745–53. [DOI] [PubMed] [Google Scholar]
- 7.Halls MC, Berardi G, Cipriani F, Barkhatov L, Lainas P, Harris S, et al. Development and validation of a difficulty score to predict intraoperative complications during laparoscopic liver resection. British Journal of Surgery. 2018. Aug 1;105(9):1182–91. [DOI] [PubMed] [Google Scholar]
- 8.Kawaguchi Y, Fuks D, Kokudo N, Gayet B. Difficulty of Laparoscopic Liver Resection. Ann Surg. 2018. Jan 1;267(1):13–7. [DOI] [PubMed] [Google Scholar]
- 9.Wakabayashi G What has changed after the Morioka consensus conference 2014 on laparoscopic liver resection? Hepatobiliary Surg Nutr 2016; 5(4):281–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hackl C, Schlitt HJ, Renner P, Lang SA. Liver surgery in cirrhosis and portal hypertension. World J Gastroenterol. 2016. Mar 7;22(9):2725–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kabir T, Tan ZZ, Syn NL, Wu E, Lin JD, Zhao JJ, et al. Laparoscopic versus open resection of hepatocellular carcinoma in patients with cirrhosis: Meta-Analysis. British Journal of Surgery. 2022. Jan 1;109(1):21–9. [DOI] [PubMed] [Google Scholar]
- 12.Kabir T, Goh BK. Laparoscopic versus open resection of hepatocellular carcinoma in patients with cirrhosis. Minerva surgery [Internet] 2022. Dec 15 [cited 2022 Dec 25]; Available from: http://www.ncbi.nlm.nih.gov/pubmed/36519820 [DOI] [PubMed] [Google Scholar]
- 13.Hobeika C, Fuks D, Cauchy F, Goumard C, Soubrane O, Gayet B, et al. Impact of cirrhosis in patients undergoing laparoscopic liver resection in a nationwide multicentre survey. Br J Surg [Internet]. 2020. Feb 1 [cited 2022 Jul 15];107(3):268–77. Available from: https://pubmed.ncbi.nlm.nih.gov/31916594/ [DOI] [PubMed] [Google Scholar]
- 14.Halls MC, Cherqui D, Taylor MA, et al. Are the current difficulty scores for laparoscopic liver surgery telling the whole story? An international survey and recommendations for the future. HPB (Oxford) 2017; 20(3):231–236. [DOI] [PubMed] [Google Scholar]
- 15.Lim C, Scatton O, Wu AG, et al. Impact of liver cirrhosis and portal hypertension on minimally invasive limited liver resection for primary liver malignancies in the posterosuperior segments: an international multicenter study. EJSO 2023. in press [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Goh BKP, Syn N, Lee SY, Koh YX, Teo JY, Kam JH, et al. Impact of liver cirrhosis on the difficulty of minimally-invasive liver resections: a 1:1 coarsened exact-matched controlled study. Surg Endosc [Internet]. 2021. Sep 1 [cited 2022 Dec 25];35(9):5231–8. Available from: https://pubmed.ncbi.nlm.nih.gov/32974782/ [DOI] [PubMed] [Google Scholar]
- 17.Strasberg SM. Nomenclature of hepatic anatomy and resections: a review of the Brisbane 2000 system. J Hepatobiliary Pancreat Surg [Internet]. 2005. Oct [cited 2022 Jul 23];12(5):351–5. Available from: https://pubmed.ncbi.nlm.nih.gov/16258801/ [DOI] [PubMed] [Google Scholar]
- 18.Chin KM, Linn YL, Cheong CK, et al. Minimally invasive vs open major hepatectomies for liver malignancies: a propensity score-matched analysis. J Gastrointest Surg 2022;26:1041–53. [DOI] [PubMed] [Google Scholar]
- 19.Goh BK, Wang Z, Koh YX, Lim KI. Evolution and trends in the adoption of laparoscopic liver resection in Singapore: analysis of 300 cases. Ann Acad Med Singap 2021;50:742–50. [PubMed] [Google Scholar]
- 20.Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg [Internet]. 2004. Aug [cited 2021 Dec 7];240(2):205–13. Available from: https://pubmed.ncbi.nlm.nih.gov/15273542/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Coelho FF, Herman P, Kruger JA, et al. Impact of liver cirrhosis and portal hypertenstion on the outcomes of minimally-invasive left lateral sectionectomies for primary liver malignancies. Surgery 2023. in press [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Austin PC. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivariate Behav Res [Internet]. 2011. May [cited 2023 Apr 10];46(3):399. Available from: /pmc/articles/PMC3144483/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blackwell M, Iacus S, King G, Porro G. Cem: Coarsened Exact Matching in Stata. 10.1177/1536867X0900900402 [Internet]. 2009. Dec 1 [cited 2023 Apr 10];9(4):524–46. Available from: https://journals.sagepub.com/doi/10.1177/1536867X0900900402 [DOI] [Google Scholar]
- 24.Hasegawa Y, Wakabayashi G, Nitta H, Takahara T, Katagiri H, Umemura A, et al. A novel model for prediction of pure laparoscopic liver resection surgical difficulty. Surg Endosc. 2017. Dec 1;31(12):5356–63. [DOI] [PubMed] [Google Scholar]
- 25.Watanabe Y, Aikawa M, Kato T, Takase K, Watanabe Y, Okada K, et al. Influence of Child-Pugh B7 and B8/9 cirrhosis on laparoscopic liver resection for hepatocellular carcinoma: a retrospective cohort study. Surg Endosc [Internet]. 2022. [cited 2022 Dec 25]; Available from: https://pubmed.ncbi.nlm.nih.gov/36203111/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Troisi RI, Berardi G, Morise Z, Cipriani F, Ariizumi S, Sposito C, et al. Laparoscopic and open liver resection for hepatocellular carcinoma with Child-Pugh B cirrhosis: multicentre propensity score-matched study. Br J Surg [Internet]. 2021. Mar 12 [cited 2022 Jul 21];108(2):196–204. Available from: https://pubmed.ncbi.nlm.nih.gov/33711132/ [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.