Abstract
Breast conserving treatment typically involves surgical excision of tumor and adjuvant radiotherapy targeting the breast area or tumor bed. Accurately defining the tumor bed is challenging and lead to irradiation of greater volume of healthy tissues. Preoperative stereotactic body radiotherapy (SBRT) which target tumor may solves that issues. We conducted a systematic literature review to evaluates the early toxicity and cosmetic outcomes of this promising treatment approach. Secondary we reviewed pathological complete response (pCR) rates, late toxicity, patient selection criteria and radiotherapy protocols. We retrieved literature from PubMed, Scopus, Web of Science, Cochrane, ScienceDirect, and ClinicalTrials.gov. The study adhered to the PRISMA 2020 guidelines. Ten prospective clinical trials (7 phase II, 3 phase I), encompassing 188 patients (aged 18–75 years, cT1-T3 cN0-N3 cM0, primarily with ER/PgR-positive, HER2-negative status,), were analyzed. Median follow-up was 15 months (range 3–30). Treatment involved single-fraction SBRT (15-21Gy) in five studies and fractionated (19.5–31.5Gy in 3 fractions) in the rest. Time interval from SBRT to surgery was 9.5 weeks (range 1–28). Acute and late G2 toxicity occurred in 0–17% and 0–19% of patients, respectively, G3 toxicity was rarely observed. The cosmetic outcome was excellent in 85–100%, fair in 0–10% and poor in only 1 patient. pCR varied, showing higher rates (up to 42%) with longer intervals between SBRT and surgery and when combined with neoadjuvant systemic therapy (up to 90%). Preoperative SBRT significantly reduce overall treatment time, enabling to minimalize volumes. Early results indicate excellent cosmetic effects and low toxicity.
Graphical abstract
Keywords: Breast cancer, Breast neoplasm, Breast tumor, Stereotactic ablative body radiotherapy, Stereotactic body radiation therapy, SBRT, SABR, Preoperative, Neoadjuvant, Tumor bed
Background
The standard of care for breast cancer treatment is postoperative radiotherapy following breast-conserving surgery. However, accurately defining the tumor bed in this context is challenging and often leads to the irradiation of a greater volume of healthy tissues. Preoperative stereotactic body radiotherapy (SBRT) offers a promising alternative by enabling precise targeting of the tumor itself, as opposed to postoperative approaches that focus on the tumor bed. The precision of preoperative SBRT allows for a decreased target volume, reducing early treatment toxicity and enhancing pathologic complete response rates (pCR). Current data from cohort phase I and II trials provide valuable insights into early toxicity. Additionally, the paradigms of treatment sequences are evolving, with neoadjuvant treatments, such as systemic therapy, gaining prominence in the management of breast cancer. Therefore, determining the optimal timing for radiotherapy and understanding its impact on pCR and toxicity are key areas for future research. Given these advancements, SBRT may also become a standard neoadjuvant treatment option.
Introduction
In 2020, approximately 2.3 million cases of breast cancer were diagnosed in women worldwide and approximately 685,000 deaths were recorded. In Central and Eastern Europe, about 160 000 new cases were recorded. Out of 185 in 157 countries, breast cancer was the most frequently diagnosed cancer. It is estimated that by 2040 the number of diagnosed breast cancers will increase by more than 40%, i.e. by approximately 3 million cases per year [1]. A systematic review by Dafni et al. shows that Poland, together with Bulgaria, Slovakia and Romania, has been showing an increasing tendency in the incidence of breast cancer since 2010 compared to other countries in Central and Eastern Europe [2]. Prognosis and targeted treatment are tailored to the breast cancer biological subtype [3–5]. The treatment of early-stage breast cancer is based on an individual approach that depends on the molecular characteristics of the tumor, the stage, general condition and patient preferences, as recommended in the ESMO (European Society for Medical Oncology) and NCCN (National Comprehensive Cancer Network) guidelines [6, 7]. The classic treatment management for early breast cancer includes systemic therapy (if indicated), breast-conserving surgery, and then radiotherapy to the entire breast ± regional lymph nodes ± boost to the tumor bed, or partial breast irradiation (PBI) in selected low risk cases. Despite its effectiveness, this treatment concept can be associated with a certain degree of toxicity in individual cases, which could have a negative impact on quality of life in the perspective of long-term survival, which is why new solutions are being sought [8, 9]. Changing the classical sequence of up-front surgery in the treatment of breast cancer has been already applied in the setting of systemic therapy. Neoadjuvant chemotherapy is used in case of locally advanced disease to obtain higher rates of pCR which results in higher rates of breast preservation and nowadays is the mainstay of the treatment for many patients [10, 11]. A similar concept of neoadjuvant radiotherapy was introduced over the last 10 years, however it has not yet gained widespread acceptance. This approach could have several advantages: better tumor volume identification, possible tumor downstaging with improved rates of breast preservation rates, improved cosmesis, shortening of treatment time, reduction of complication rates in patients requiring breast reconstruction. A growing number of reports indicate that preoperative radiotherapy for early-stage breast cancer significantly improves disease-free survival (DFS), and more importantly, results in a lower risk of side effects and better cosmetic outcomes [12]. One of the most promising modalities of radiotherapy is stereotactic ablative radiotherapy (SABR), which involves the delivery of a very high dose of radiation to the tumor volume with high precision using one or several fractions administered usually in 1 to 10 days [13]. SABR is a method used preoperatively that could potentially also eliminate or be an alternative to the subsequent surgical intervention, in well selected patients. It is an important goal in terms of patients' quality of life, but also makes it possible to reduce the health care costs and the time required for hospitalization and convalescence after treatment [14].
Based on these data, we conducted a systematic literature review to evaluate the outcomes of this promising treatment approach. We assessed toxicity, cosmetic outcomes, pathological complete response (pCR) rates, patient selection criteria, and radiotherapy protocols.
Materials and methods
Search strategy
We conducted a systematic review according to the Population, Intervention, Control, Outcome, Study Design (PICOS) method which is shown in Table 1. We followed the PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement.
Table 1.
Study design according to the Population, Intervention, Control, Outcome, Study Design (PICOS) method
| Population | Patients treated with preoperative stereotactic body radiotherapy (SBRT) for breast cancer |
|---|---|
| Intervention | SBRT to the primary tumor (fraction dose ≥ 5 Gy) |
| Control | Not applicable (the data will be pooled from single arm trials) |
| Outcome |
Primary: early toxicity, cosmesis Secondary: rates of pathologic complete response (pCR), late toxicity |
| Study design | Any retrospective or prospective original studies describing clinical outcomes of patients treated with preoperative SBRT for primary breast cancer |
We searched five databases, namely PubMed, Scopus, Web of Science, Cochrane, ScienceDirect and ClinicalTrials.gov registry. An additional evaluation was conducted via citation searching from selected articles. Two blinded authors independently performed searches using the keywords: (breast cancer or breast neoplasm or breast tumor) and (stereotactic ablative body radiotherapy or stereotactic body radiation therapy or SBRT or SABR) and (neoadjuvant or preoperative or induction). In case of discrepancies, the third author verified the search. We identified potential studies and exported them to a reference management program (Mendeley Desktop) for inclusion based on title and abstract, and then the full article. The research involved an analysis of all studies published up to December 22, 2023.
Selection criteria
The inclusion criteria were as follows: (1) retrospective and prospective clinical trials with published results and (2) studies published in the English language.
Exclusion criteria were: (1) radiotherapy performed in postoperative setting only, (2) preoperative irradiation of the entire breast (3) non unifocal breast tumour (3) presence of distance metastases, (4) lack of access to the full text of the manuscript, (5) studies without results and unclear results (6) case reports, (7) review papers, (8) study protocols.
Data extraction
The extracted data consisted of the author, type of study, sample size, main endpoints, inclusion criteria, radiotherapy technique, contouring schema for gross tumor volume (GTV), clinical target volume (CTV) and planning target volume (PTV), dose and fractionation regimen, concomitant systemic treatment, doses to organs at risk (OARs), time from SBRT to surgery, rates of pCR, early and late toxicity, cosmesis analysis.
Results
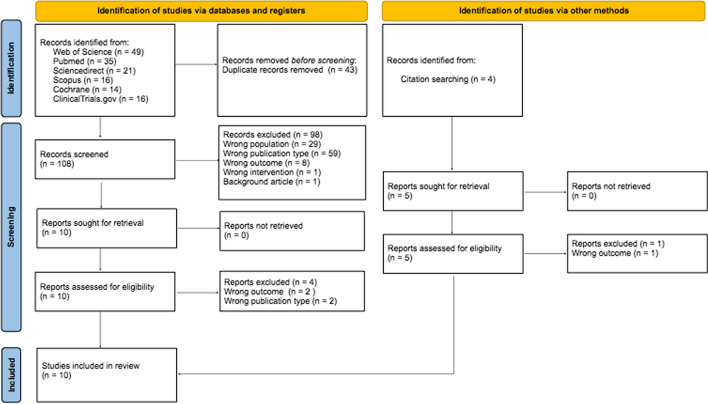
During the initial database search found a total of 151 papers (49- Web of Science, 35—PubMed, 21- ScienceDirect, 16- Scopus, 14—Cochrane, 16- Clinicaltrials.gov). Before screening we have deleted 43 duplicates. In the next step, we excluded titles and abstracts that did not meet the inclusion and exclusion criteria (98 articles). We identified further 5 papers by searching for citations from the included articles. The full text of the qualified articles was analyzed and a selection was made, after which 5 papers were excluded. Finally, 10 primary studies were included in the systematic review process (two studies on same cohort). Figure 1 shows the PRISMA 2020 flowchart with screening results. The list and characteristics of the included studies and their outcomes is shown on the Table 2. All 10 included studies have a prospective design. Two studies were a phase I dose escalation studies, one phase I feasibility study. Seven reports were phase II studies. All of the included studies were single arm and non-randomized [15–24]. The number of included patients was 188 and ranged from 6 to 36 among studies. Median follow up time was 15 (3–30) months.
Fig. 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 flow diagram for the literature selection process
Table 2.
Characteristic of selected studies for systematic review
| Guidolin et. al. [15] | Tiberi et. al. [16] | Liveringhouse et. al. [17] | Meattini et. al. [18] | Horton et. al. [19] | Bondiau et. al. [20] | Vasmel JE et. al. [21], [22] | Guanglei Chen et. al. [23] | De Caluwe et. al. [24] | |
|---|---|---|---|---|---|---|---|---|---|
| Type of study | Phase II SIGNAL trial, prospective, single arm, non randomized | Phase II, prospective, single arm, nonrandomized | Phase II, prospective, single arm, nonrandomized | Phase II ROCK trial, prospective, single arm, non randomized | Phase I, dose escalation, prospective, single arm, nonrandomized | Phase I, dose escalation, prospective, single arm, non randomized | Phase II, prospective, single arm, non randomized | Phase II, prospective, single arm, non randomized | Phase I Neo- Checkray Trial, prospective, single arm, non randomized |
| Number of participants | 27 | 10 | 20 | 22 | 32 | 25 | 36 | 10 | 6 |
| Main endpoints | Feasibility, toxicity, cosmesis | Radiologic and pathologic response, toxicity | Radiologic and pathologic response | Pathologic response, toxicity, cosmesis | Radiologic response, toxicity, cosmesis | Toxicity, cosmetics, pathologic response, establishing maximum tolerable dose in combination with neoadjuvant therapy | Pathologic and radiologic response, toxicity, cosmesis | Pathologic complete resposne, objective resposne rate, residual cancer burden 0-I, safety | Feasibility of delivering SBRT, toxicity, cosmesis |
| Inclusion criteria TNM |
Postmenopausal, unifocal, ≤ 20 mm, unifocal, ER positive, cN0 In initial ultrasound distance ≥ 2 cm between the lesion and the skin and the lesion and the chest wall |
≥ 65 years, ≤ 20 mm, unifocal, ER positive, HER 2 negative, cN0, Grade1-2 | ≥ 50 years, Unifocal, ≤ 20 mm, ER positive, HER 2 negative, cN0 | ≥ 50 years, ER/PR positive, HER 2 negative unifocal, ≤ 25 mm, cN0 | ≥ 55 years, ER/PR positive, HER 2 negative, ≤ 20 mm, low/intermediate grade, no LVSI, DCIS ≤ 2cm, cN0 | ≥ 18 years, cT1-T3, cN0-N1, unifocal, not suitable for BCT | ≥ 50 years, unifocal, ≤ 20 mm, ER positive, HER2 negative, cN0 or ≥ 70 years, unifocal, ≤ 30 mm, ER positive, HER2 negative, cN0 | 18–75 years, TNBC, cT2-T3N0-N2 | ≥ 18 years, Luminal B, Ki67 ≥ 15% or G3, MammaPrint high risk, cT2-3N0 or cT1b-3N1-3, size ≥ 1.5 cm |
| Contouring |
GTV + 5 mm = CTV CTV + 5 mm = PTV |
GTV + 10 mm = CTV CTV + 10 mm = PTV |
GTV + 15 mm = CTV CTV + 3 mm = PTV |
GTV + 15 mm = CTV CTV + 3 mm = PTV |
GTV + 15 mm = CTV CTV + 3 mm = PTV |
GTV + 5 mm = CTV CTV + 2 mm = PTV |
GTV + 20 mm = CTV CTV + 3 mm = PTV |
GTV, margins NR | GTV + margin NR |
| Concurrent systemic therapy | – | – | – | – | – | 3 cycles of docetaxel (100 mg/m [2]), SBRT on 3 consecutive days during the second cycle but not in the chemotherapy days, 3 FEC cycles (fluorouracil 500 mg/m2, epirubicine 100 mg/m [2], and cyclophosphamide 500 mg/m [2]). Cycles q3w | Six patients (17%) -neoadjuvant endocrine started after NA-PBI | 8 cycles of adebrelimab (20 mg/kg every 3 weeks). SBRT at the second cycle every other day, 6 cycles of nab-paclitaksel (125 mg/m [2] on 1st and 8th day) and carboplatin (area uder curve 6 mg/ml per min on 1st day) every 3 weeks since 3rd cycle |
12 cycles of paclitaxel 80 mg/m [2] weekly then 4 cycles q2w dose dense doxorubicin-cyclophosphamide (60 mg/m [2] and 600 mg/m [2] respectively); then 5 administrations of durvalumab 1500 mg Every 5 weeks for five administrations q4w and 4 administrations oleclumab 3000 mg q2w for followed by q4w for 3 administrations SBRT given in week 4–5, followed by the week 5 systemic treatment |
| MRI coregistered to planning CT | + | – | + | + | + | + | + | + | + |
| System and Technique | LINAC/VMAT | LINAC/IMRT | LINAC/VMAT | Cybernife | LINAC/IMRT | Cyberknife | LINAC/VMAT | LINAC/IMRT | LINAC/VMAT |
| Dose fractionation schemas | 21 Gy- 1 fraction | 20 Gy-1 fraction | 28.5 Gy- 3 fractions, 9.5 Gy daily | 21 Gy- 1 fraction | 15 Gy (n = 8), 18 Gy (n = 8), 21 Gy (n = 16) in 1 fraction | 19.5 Gy (n = 3), 22.5 Gy (n = 3), 25.5 Gy, (n = 6), 28.5 Gy (n = 7), 31.5 Gy (n = 6)- 3 fractions q2d | 20 Gy for GTV, 15 Gy for CTV- SIB—1 fraction | 24 Gy- 3 fractions q2d | 24 Gy- 3 fractions n consecutive days |
| Positioning | Surgical clip, cone-beam CT prone | Surgical clip, cone-beam CT supine | Surgical clip, cone-beam CT supine | Supine | Surgical clip, cone-beam CT prone | Supine | Surgical clip, cone-beam CT supine | NR | NR |
| Time from SBRT to surgery | 1 week | 11–13 weeks | 6–8 weeks | 2 weeks | 10 days | 19–23 weeks | 6 months (n = 15), 8 months (n = 21) | 21–23 weeks | 16–20 weeks |
| pCR | NR | pCR—0 (0%), pPR- 80% (n = 8) | 0 (0%) | 2 (9%) | NR | 9 (36%) | 15 (42%), 5 (33%) after 6 months, 10 (48%) after 8 months, 12 (33%) near pCR | 9 (90%) | 2 (33.3%), 2 (33.3%) near pCR |
| Scales used to evaluate toxicity | CTCAE v. 4.03 | CTCAE | CTCAE v. 4.0 | RTOG/EORTC | CTCAE | NR | CTCAE v. 4.03 | CTCAE v. 5.0 | CTCAE v. 5.0 |
| Acute and late toxicity related to SBRT | Acute—3 weeks postop.- 0 ≥ G2, Late—6 month postop.-1 (3.7%) G2 wound infection, 1 year postop.—No ≥ G2 | Acute- 0 ≥ G2, Late- NR | Acute- 4 to 5 weeks after SBRT- G2- toxicity – breast pain – 1 (5%) and erythema – 1 (5%), no G3, Late: patients without postoperative WBRT (n = 16), G2 -3 (19%)-skin induration, breast pain, atrophy), G3—1wound complication. patients after WBRT: skin induration G2- 2 (50%), seroma G2 -1 (25%), atrophy G2 -1 (25%), G3 toxicity in 2: abscess -1 (25%), breast pain -1 (25%) |
Acute (7–30 days after SBRT)—breast oedema- G2—1 (4.55%), Acute (30 days after SBRT) – breast induration G2 -1 (4,5%) Late (6–12 months after SBRT) G2 -1 (4,5%) |
Acute G2: Wound infection – 1 (3%), breast pain 2 (6%), dermatitis 3 (10%) Late: fibrosis G2 -3 (10%), G3 -1 (3%), breast atrophy G2 -2 (6%), G3 – 1 (3%), breast pain G2 -2 (6%), dermatitis G2 1 (3%), infection G2 1 (3%), seroma G2 1 (3%), hyperpigmentation G2 1 (3%) |
Acute—G2 skin erythema- 1 in 22.5 Gy cohort and 1 in 25.5 Gy cohort, G3 dermatologic erythema- 1 on 7 (14%) in 28,5 Gy cohort, Late- 0% |
Before BCS r 6–8 months after SBRT: G2– chest pain- 1 (3%), breast pain- 1 (3%), breast edema- 1 (3%) Late (12 months from SBRT) G2 – breast pain 2 (6%), chest pain -1 (3%), wound infection 5 (15%) Late G3—1 (3%) transient wound infection Late (18 months after BCS): n = 0 |
Related to sbrt Acute- 0 (0%) Late- 0 (0%) |
Acute < G2- 1 (16.7%) breast pain Late- 0 (0%) |
| Cosmesis |
Modified Harvard–Harris Cosmetic Scale-Physician-rated (good or excellent)-100% (baseline), 93% (3 weeks), 96% (6 months), 92% (1 year postoperatively) Patients rated –(good or excellent)-96% (baseline), 93% (3 weeks), 92% (6 months), 96% (1 year postoperatively) No significance difference from the baseline, |
EORTC scale—NR | Overall physician- reported—„good”- 17 (85%), „fair”- 2 (10%), „poor”- 1 (5%) |
NSABP B-39/RTOG 0413 cosmesis evaluation scale After 6 and 12 months: 28 (100%) -good/excellent |
NR | 94% of the patients very satisfied, satisfied, or not unsatisfied | NR | Excellent in 4 (66.6%) and good in 2 (33.3%) | |
| Median Follow-up time | 12 months | 3 months | 14 months | 18 months | 23 months | 30 months | 21 months | 13 months | 25.5 months |
| Adjuvant EBRT | none | none | In 4 patients (20%) with ypN + | In 2 patients | In 3 patients (6%) | All but 2 who refused | none | NR | NR |
e2d—Every 2 days, q2w – Every 2 weeks, BCT- Breast conserving treatment, GTV- Gross tumor volume, CTV- Clinical target volume, PTV- Planning target volume, CTCAE—Common terminology criteria for adverse events, NR- Not reported, EORTC—European organisation for research and treatment of cancer, RTOG—Radiation therapy oncology group, pCR- Pathological complete response, LINAC—Linear accelerator, IMRT—Intensity modulated radiation therapy, VMAT—Volumetric arc therapy
Qualification criteria
The main qualification criterion for study inclusion was tumor diameter determined by magnetic resonance imaging (MRI) in all but one study by Tiberi et al. where computed tomography (CT) only was used [15–24]. Four studies included patients with a tumor size of ≤ 20 mm [15–17, 19]. Vasmel et al. also used this criterion for patients ≥ 50 years but for patients ≥ 70 years tumors up to 30 mm were also included [21, 22]. In the ROCK trial patients with tumors up to 25 mm were treated [18]. Older patients who were at least 50 years old were eligible for preoperative radiotherapy [15–19, 21, 22]. Bondieu et al. qualified patients aged 18 years and older. Their cohort also included patients with cT2-3 tumors and node positive ( cN1) disease. A special feature of this study is that all patients of their cohort were not suitable for breast conserving surgery (BCS) [20]. Nine studies included low risk luminal A-like subtypes with ER or ER/PgR positive HER-2 negative and unifocal tumors [15–22, 24]. The inclusion criteria of the study of Horton et al. were absence of lymphovascular invasion (LVSI), ductal carcinoma in situ (DCIS) ≤ 2cm and low to intermediate grade tumors (G1-2) [19]. In contrast, Chen et al. included only triple negative breast cancer (TNBC) patients [23]. In this trial, inclusion criteria were patients aged 18–75 years with tumors ≥ 20 mm, mostly cT2 but also cT3, and cN0-N2 according to TNM [23]. In the Neocheck trial patients with cT2-3 cN0 and cT1b-3 cN1-3, with a tumor size of at least 15 mm were qualified [24]. Moreover, further inclusion criteria were a high-risk score in the MammaPrint genomic expression profiling test, high grade (G3) or a Ki67 of ≥ 15% [24]. In the SIGNAL trial an additional criterion for inclusion was a distance of ≥ 2 cm from the tumor to the skin and chest wall [15].
Target volumes definition and radiotherapy modalities
Gross tumor volume (GTV) was based on the planning CT with co-registered MRI [15, 17–24]. Tiberi et al. used only a planning CT only for GTV contouring [16]. The CTV was formed by adding an isotropic margin to the GTV which differed between the studies: 5 mm [15, 20], 10 mm [16], 15 mm [18–21, 22]. Liveringhouse et al. and De Caluwe et al. didn’t report the details regarding the magnitude of the CTV margins [17, 24], while Chen et al. didn’t include any information if any margins were added to the GTV [23]. The chest wall and pectoralis major muscle were excluded from the CTV in all studies, where CTV information was available. In the analysis of Meattini et al. and Bondiau et al. PTV were generated by adding 2–3 mm margin to the CTV [18, 20]. In their studies SBRT was performed on a Cyberknife system [18, 20]. PTV for conventional linear accelerators (LINACs) based SBRT was generated by adding 3 mm [19, 21, 22] or 5 mm [15] to the CTVs. Tiberi et al. used larger CTV-PTV margin which was 10 mm [16]. All studies used an intensity modulated radiation therapy (IMRT) or volumetric arc therapy (VMAT) techniques which were performed on Cyberknife [18, 20] or LINACs. [15–17, 19, 21–24].
Fractionation schemes
Two included phase I trials were dose escalation protocols [19, 20]. Horton et al. used a single fraction regimen and escalated the dose in three cohorts of patients. Fifteen Gy in 8 patients, 18 Gy in 8 patients and 21 Gy in 16 patients were prescribed [19]. Bondiau et al. used 3 fractions, in every other day regime, and escalated the dose in 5 cohorts consisting of 3 patients each. 19.5 Gy, 22.5 Gy, 25.5 Gy, 28.5 Gy and 31.5 Gy were used [20]. Guidolin et al. and Tiberi et al. prescribed 20 Gy in single fraction [15, 16]. In the ROCK trial 21 Gy in one fraction was administered [18]. Vasmel et al. used two dose levels with a simultaneous integrated boost technique (SIB) consisting of 20 Gy to the GTV and 15 Gy to the CTV [21, 22]. Liveringhouse et al. used a fractionated regimen and administered 28.5 Gy in 3 daily fractions [17]. When et al. used 24 Gy in 3 fractions administered every other day [23]. In the NeoCheck trial also 24 Gy in 3 fractions were used but patients were irradiated every day [24].
Dose constraints to organs at risk (OARs)
Dose constrains for individual OARs varied between publications. Table 3 summarizes them along with the dose fractionation schemes that were used. In the studies by Guanglei Chen et Al. and De Caluwe et al. [23, 24] guidelines for dose constrains were not included.
Table 3.
Dose constraints to organs at risk (OARs) used in included studies
| Guidolin et al. [15] | Tiberi et al. [16] | Liveringhouse et al. [17] | Meattini ett, al. [18] | Horton et.al. [19] | Bondiau et al. [20] | Vasmel JE et al. [21, 22] | |
|---|---|---|---|---|---|---|---|
| Dose fractionation schedule | 21 Gy /1 fraction | 20 Gy / 1 fraction | 28,5 Gy / 3 fractions daily | 21 Gy / 1 fraction | 15 Gy (n = 8), 18 Gy (n = 8), 21 Gy (n = 16.) / 1 fraction | 19,5 Gy (n = 3), 22,5 Gy (n = 3), 25,5 Gy, 9 (n = 6), 28,5 Gy (n = 7), 31,5 Gy (n = 6)- 3 fractions every other day | 20 Gy for GTV, 15 Gy for CTV- SIB / 1 fraction |
| OAR | |||||||
|
Breast Uninvolved ipsilateral Contralateral |
V10,5 Gy ≤ 50% V20 Gy ≤ 47% Dmax < 21 Gy Dmax < 1Gy |
Dmax ≤ 3 Gy |
V15Gy ≤ 25%; V25 Gy ≤ 10% V10 Gy < 7%, V5 Gy < 10% |
V10.5Gy < 60% V22Gy < 35% < 1Gy |
< 50% of the breast volume should receive 50% or more of the prescribed dose < 35% of the breast volume should receive prescription doese < 15% of the prescribed dose at any point |
NR |
Ratio PTVCTV to ipsilateral breast volume < 25% Dmean < 5Gy |
|
Lung Total Ipsilateral Contralateral |
V11 Gy ≤ 35% V7,5 Gy ≤ 15% V1,7 Gy ≤ 15% |
D10% ≤ 6 Gy D25% ≤ 2 Gy |
V9 Gy < 5% V9 Gy < 3% |
V7Gy < 1000 cm3 < 1Gy |
V7Gy < 1000 cm3 | V5 Gy < 5 cm3 |
V7.8Gy ≤ 5% Dmean < 3.6 Gy |
| Heart |
Dmax < 22 Gy (Point dose) V3 Gy < 5 cm3 V16 Gy ≤ 15% |
Right breast D5% ≤ 1 Gy Left breast D5% ≤ 3 Gy |
Dmean ≤ 2 Gy V3Gy ≤ 10 cm3 |
V3Gy < 5 cm3 |
Dmax < 5 Gy V3 Gy < 5 cm3 |
NR |
V2.8Gy ≤ 10% V4.7Gy ≤ 5% |
| Thyroid | Dmax < 1,1 Gy (Point dose) |
D100% ≤ 0,6 Gy Dmax < 3 Gy |
Dmax1,2 Gy (4% prescription) | - | Dmax < 3Gy (15% prescription) | - | - |
| Skin | V18,3 Gy < 5 cm3 |
D10cm3 ≤ 20 Gy Dmax ≤ 21 Gy |
D10cm3 ≤ 15 Gy |
V10Gy < 10 cm3 V20Gy < 1 cm3 |
evaluated on a case by case | V15 Gy < 10 cm3 | D1cc < 16 Gy |
| Chest wall |
V10 Gy < 10 cm3 V16 Gy < 2 cm3 |
D1cm3 ≤ 21 Gy Dmax ≤ 22 Gy |
V28.5 Gy ≤ 30 cm3 | V10Gy < 10 cm3 | - | V15 Gy < 10 cm3 | D16.3Gy < 20cc |
DYY cm3 < XX Gy is the maximal dose to YY cm3 in the volume of the OAR that receives the highest doses. DMax is the near-point maximum dose, VXX Gy is the percentage volume of the organ receiving a dose of XX Gy or higher
Timing of surgery after SBRT
The time interval from SBRT to surgery was different in all of the included studies. The median time from SBRT to surgery was 9.5 weeks and it ranged from 1 to 28 weeks [15–24]. In 3 studies the timing between SBRT and surgery was very short, with only 1–2 weeks [15, 18, 19]. Two analyses used longer intervals of 6–13 weeks [16, 17]. However, over half of the included studies waited 16–28 weeks until surgery [20–24].
Concurrent systemic therapy
In three trials SBRT were tested with concomitant systemic therapy [20, 23, 24]. As already mentioned in the inclusion criteria all of this studies included patients with higher risk or more advanced disease compared to other trials. In the study of Bondiau et al. neoadjuvant chemotherapy included 3 cycles of docetaxel and 3 cycles FEC (5-fluorouracil, epirubicin, cyclophosphamide) with SBRT given during the second chemotherapy cycle but not on the same day as chemotherapy [20]. The systemic therapy regimen in the Chen trial consisted of 8 cycles of selective PD-L1 inhibitor – adebrelimab and 6 cycles of nab-paclitaxel and carboplatin, where SBRT was performed during the second cycle of immunotherapy [23]. The regimen used in the De Caluwe trial consisted of 19 weeks used paclitaxel, cyclophosphamide-doxorubicin, anti-PD-L1 antibody durvalumab and anti-CD73 antibody oleclumab with SBRT given in week 4–5. In the Vasmel trial 6 (17%) patients initiated neoadjuvant endocrine treatment after SBRT according to national guidelines [21].
Acute and late toxicity profile related to SBRT
One of the important objectives of preoperative early breast cancer SBRT trials was to assess cosmetic effects and mostly acute toxicity due to the usually short follow- up time. In the study by Vasmel et al. late G2 toxicity, asseses 12 months from SBRT involved breast pain (n = 2), chest pain (n = 1) and wound infection (n = 5) and one G3 wound infection [21]. Bondiau et al. reported no late treatment-related toxicities after 30 months. [20] Similarly no late toxicities were reported in Chen and De Caluve papers [23, 24]. In the study by Horton et al. late breast fibrosis was reported in 71% (mostly G1 in 56%) and atrophy in 20% of patients but no dose correlation analysis was performed. Early toxicity was mild including breast pain, dermatitis and breast edema [19]. In the study by Tiberi et al. six patients (60%) had G1 dermatitis in the irradiated area. No other significant post-radiotherapy complications were reported [16]. In the Liveringhouse trial, 3 patients who did not receive postoperative whole-breast RT (n = 16) experienced G2 toxicity (19%-skin induration, breast pain, atrophy) and one case of G3 toxicity of wound complication which required a re-excision lumpectomy. In 4 patients who received additional postoperative whole-breast RT, the following G2 toxicity was detected: skin induration in 2 patients (50%), seroma in one (25%), atrophy in one patient (25%), G3 toxicity in 2 patients: abscess (n = 1), and breast pain (n = 1). In 7 included studies with late toxicity reports G3 were reported in 2 patients who did not receive postoperative whole breast radiotherapy(WBRT). Details of early and late toxicity are shown in Table 2.
Cosmetic effect assessment
The cosmetic outcome after preoperative SBRT was reported in 6 selected studies. In the trial of Guidolin et al. cosmetic assessment was performed by patients and physicians using the Harvard-Harris cosmetic scale [15]. There was no significant change in cosmetic compared to the baseline at 3 weeks and 6 months from surgery [15]. Meattini et al. used the BCCT.core software for cosmetic effect analysis and reported that 21 of 22 (95,4%) patient had “good” to “excellent” results, 1 patient (4.6%) reported a “fair” result after preoperative SBRT [18]. Horton et al. used the NSABP B-39/RTOG 0413 cosmetic evaluation scale. After 12 months 28 (100%) patients reported “good” or “excellent” cosmetic effects after SABR [19]. In the trial of Vasmel et al. 94% of the patients were very satisfied, satisfied, or not unsatisfied after preoperative stereotactic radiotherapy [21].
In Liveringhouse et al. cosmesis was rated “good” in 17 (85%), “fair” in 2 (10%), and “poor” in 1 (5%) patient, but in patients receiving additional postoperative RT (n = 4), cosmesis was “good” in 3 and “poor” in 1 patient [17]. Of all patients (n = 6) reported in the De Caluve trial 4 had “excellent” and 2 had a “good” cosmetic outcome, but out of 4 patients who received BCS, 3 were assessed as “excellent” and 1as “good”. [24] Bondiau et al. reported that overall 94% of patients were very satisfied and satisfied with the cosmetic outcome [20].
Pathological response
In 8 studies pCR was analyzed [16–18, 20–24] In two [15, 19] no pathological response was assessed. In four of the single fraction SBRT studies pCR were ranged between 0%, 9%,and 42% [16, 18, 20, 21]. The studies by Vasmel et al. showed highest pCR rates among those included: 15 (42%) in total, 5 of 15 (33%) after first 6 months, 10 of 21 (48%) after 8 months and 12 (33%) near pCR (< 10% residual tumor cells), in 7 patients (19%) partial response (10%-50% residual tumor cells) and in 2 (6%) stable disease (> 50% residual tumor cells with features of response to treatment) have been reported. There were no patients without evidence of response. Neoadjuvant endocrine therapy was administered in 6 patients of whom pCR was reached in 2 patients, near pCR in 3 patients and a partial response(PR) in one patient [21, 22]. In this study the time interval from SBRT to surgery was also the longest, 6 months in 15 patients and 8 months in 21 patients [21]. Bondiau et al. reported 36% pCR in the whole cohort. It was very heterogenic, with 67% at a dose level of 25,5 Gy, 43% at 28,5 Gy, 33% at 31,5 Gy with no pCR cases in the 19,5 Gy and 22,5 Gy dose levels groups [20]. Residual cancer burden (RCB) according to Symmans et al. was measured in the De Caluve trial were 2 of 6 patients (33.3%) had a complete pathological response (RCB 0), two (33.3%) had a near pCR (RCB1) one (16.7%) a moderate residual disease (RCB 2) and one (16.7%) an extensive residual disease (RCB 3) [24]. The same measure was used in the Chen et al. trial showing pCR (RCB 0) in 9 patients (90%) and near pCR (RCB 1) in one patient (10%) [23]. Liveringhouse et al., reported no case of pCR and the median RCB was 30% with 2 patients with a 80% residual cancer burden whereas all other patients had ≤ 50% residual cellularity after stereotactic fractionated radiotherapy, 3- fraction scheme, and after median of 49 days from SBRT to breast conserving surgery (BCT) [21].
Discussion
The use of radiotherapy as a main neoadjuvant treatment or in combination with chemotherapy in a preoperative setting represents a newly intensively researched approach in early-stage breast cancer treatment [12, 25, 26]. The results of studies using moderate hypofractionation, as a preoperative treatment, showed good cosmetic effect and local control (LC), which encouraged researchers to look for shorter and more intensive treatment regimens [27, 28].
To the best of our knowledge, the above systematic review represents the most up-to-date compilation of studies evaluating the feasibility of preoperative PBI with the use of SBRT limited to 5 fractional doses [14, 29–31]. For the above review 10 studies were included (in 7 studies SBRT was used as the only neoadjuvant treatment modality, in 3 of them SBRT was used with concomitant systemic therapy [20, 23, 24]. In two studies published recently, SBRT was performed preoperatively with immune-chemotherapy [23, 24]. The main advantages and disadvantages of preoperative radiotherapy in breast cancer were summarized in Table 4.
Table 4.
Advantages and disadvantages of preoperative PBI. [32]
| Advantages of preoperative radiotherapy | Disadvantages of preoperative radiotherapy |
|---|---|
|
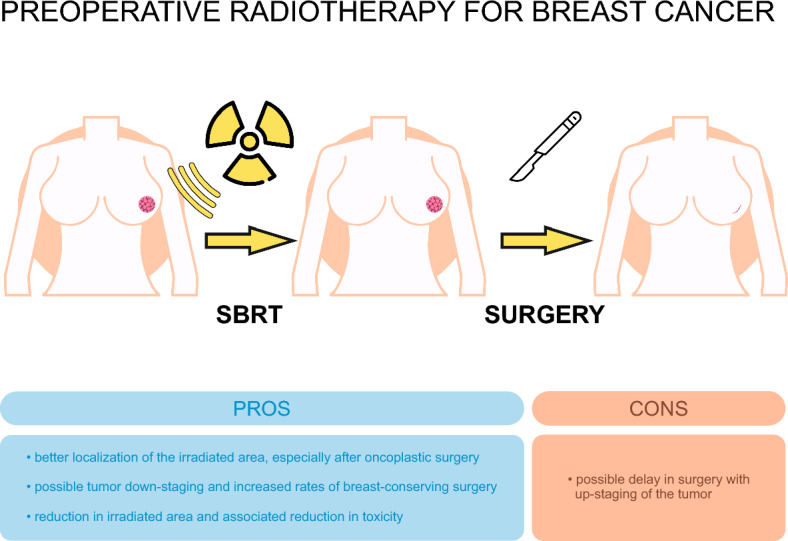
Better localization of the irradiated area, better visibility of the primary tumor—possibility to insert tracers near the tumor during biopsy of the lesion, which minimizes the risk of geographic error; Possible tumor down-staging and increased rates of breast-conserving surgery; Preoperative radiotherapy reduces the problem of the possible technical treatment planning challenges after oncoplastic surgery Reduction in irradiated area and associated reduction in toxicity. With preoperative radiation therapy, we irradiate the tumor volume with a margin. Surgery is performed after radiation therapy, so the area of the breast receiving the highest dose of radiation is removed, which can lead to limited fibrosis and good cosmetic outcomes; Better oxygenation of tumor tissues which determines a better therapeutic effect of radiotherapy than in tissues undergoing surgical intervention; In favorable cases of breast cancer, postoperative WBRT may be omitted if preoperative PBI has been applied. Generation of an abscopal effect, which is not the case with postoperative radiotherapy; o Reduction in number of radiotherapy sessions |
Possible delay in performing surgery due the duration or complications after radiation therapy-this problem does not apply to stereotactic radiation therapy performed in the shortest possible time, Upstaging of the tumor, positive margins after surgery, changing the receptor profile to a less favorable one which requires more aggressive treatment regimens like: irradiating the whole breast, nodal areas or chemotherapy |
SBRT is a method of external beam radiotherapy that allows highly precise delivery of a high dose of radiation to a limited target area with a large dose drop around the target. The biological efficacy of SBRT is not only based on the mechanism of DNA damage at the molecular level, but also on additional effects at the tissue and cellular level (damaging the vasculature and cell membranes), as well as on the abscopal effect of enhancing the anti-tumor immune response due to the action of high doses of radiotherapy on the cancerous tumor, which produces a therapeutic effect beyond the irradiated field [33–35].
Preoperative radiotherapy has become the standard treatment for rectal cancer and sarcoma. In the case of breast cancer, it is currently the subject of clinical trials. Beyond the improved target visualization and reduced risk of tumor cell dissemination during surgery, preoperative radiotherapy offers multiple radiobiological advantages. This include better oxygenation of non-operating tissues and immune-priming as outlined by Brackstone et al. [36] Additionally preoperative radiotherapy may significantly enhance immune responses against tumors. It can transform tumor into personalized in situ vaccine, teaching the immune system to recognize and combat cancer, what cannot be observed after irradiation of tumor bed [37, 38]. This may be important in eradicating subclinical diseases and distant micrometastases, potentially leading to long-lasting immune memory against future tumors [37–39]. In this context, it's noteworthy that two studies in this review specifically explored the combination of SBRT and immunotherapy [23, 23].
Target volume definition
Stereotactic radiotherapy employed in radical breast cancer treatment can be utilized postoperatively as a boost after WBRT or PBI [40, 41]. Due to its targeted approach, it requires meticulous target definition during treatment planning and consistent positioning accuracy to minimize geographic error risk. In post-operative setting, surgical clips are required to accurately identify the tumor bed. For the increasingly popular oncoplastic procedures, the location of the tumor bed poses a major challenge for the radiation oncologists and need collaborative target determination with surgeon.
Despite guidelines for contouring the tumor bed in PBI, challenges in delineation remain. The main issues include: [42, 43].
Extent and location: often, the contoured area is disproportionately large compared to the original tumor or located differently than indicated by pre-surgery examination and imaging studies [44].
Scattered clips (in 43 -73% of patients undergoing surgery, clips on postoperative CT imaging were visualized outside the original quadrant of the tumor) [45, 46].
Preoperative tumor delineation on CT scans leads to less inter-observer variability compared to postoperative delineation of tumor bed [47]. The target volume, the tumor, can be demarcated by implanting fiducial markers around it during the biopsy. The potential for fusion of imaging studies, such as magnetic resonance imaging (MRI) with planning computed tomography (CT) scans, is instrumental in accurately determining the tumor's exact extent. Compared to CT scans MRI better visualize irregularities and spikes in the tumor [48]. Vasmel et al. published consensus on contouring primary breast tumors on MRI in the setting of neoadjuvant PBI in trials [49]. In all but one study MRI was co-registered with CT scans for planning radiotherapy [16]. The GTV in Tiberi et al. study was defined as the primary tumor based on physical exam, CT scans, and breast ultrasonography [16].
The technical aspects of neoadjuvant PBI have been elegantly highlighted by Zerella et al. in recently published narrative review [50].
Preoperative PBI implementation techniques
The implementation of radiotherapy can be performed in prone or supine position using various immobilization systems. Diagnostic contrast-enhanced MRI is typically performed in prone position, which makes it easier to perform image fusion when realizing radiotherapy in an analogous position. When treating patients in the supine position, the solution may be to perform deformable fusion with diagnostic MRI images or to perform another MRI scan in the supine position like in the ongoing CRYSTAL study [51]. Widespread clinical introduction of the MR-LINAC (a linear accelerator (LINAC) with integrated MR scanner can be particularly helpful in the delivery of radiotherapy for breast cancer [52].
Comparison between PBI and WBRT
Comparison of postoperative WBRT and PBI show no differences in overall survival (OS) with favorable toxicity profile for the PBI technique in well selected low risk early breast cancer patients [53, 54]. In extended follow-up periods (up to 15 years), PBI may result in a marginally higher rate of local recurrence(LR) compared to WBRT [55]. However, advancements in systemic treatments, the integration of adjuvant therapies, and enhancements in local treatments have reduced local recurrence rates in breast cancer by half [56]. Consequently, there are emerging considerations for omitting postoperative radiotherapy in certain cases [7]. Within this context, PBI could emerge as an optimal balance. It is noteworthy that none of the analyzed studies reported tumor progression which is opposite to postoperative PBI where mature data are available.
Toxicity profile
By limiting the irradiated volume, dose to OARs can be significantly reduced, which is particularly important for the heart, the left anterior descending coronary artery (LAD), lung or contralateral breast, as it reduces the risk of secondary malignances and the incidence of cardiac and lung complications [57, 58]. The volume of the irradiated area for preoperative PBI is smaller than postoperatively which may translate into reduced toxicity in the long term follow-up.
The early toxicity profile of preoperative SBRT in breast cancer seems to be favorable. Among studies selected for the review, in terms of G3 acute toxicity events, only one erythema (14%) from 28.5 Gy cohort in Bondiau study was reported [20]. The late G3 toxicity was reported in one patient (2.8%) in Vasmel et al. trial and in one (6.3%) in Liveringhouse trial [17, 21].
PBI performed postoperatively is associated with better cosmetic outcome and fewer late radiation toxicities than WBRT. However, no direct comparative studies exist on the toxicity of stereotactic PBI when applied either preoperatively or postoperatively. Notably, the levels of both early and late toxicities are reportedly low [59, 60].
Comprehensive data on the late toxicity of preoperative SBRT used as PBI is still limited, necessitating a longer period of observation.In the above analysis, the longest follow up-25.5 months was reported by De Caluwe et al. During this time no late effect was reported [24].
It is also important to mention that addition of SBRT to immunochemotherapy does not increase the percentage of grade 3 or higher adverse effects (AEs) observed after immunochemotherapy [23, 24].
The optimal SBRT dose guidelines in preoperative setting
There is no established fractionation scheme for preoperative SBRT. In the above study, 1 and 3 fractional regimens were analyzed. The highest total dose of 21 Gy was given in one fraction in the SIGNAL study. In the 3 fractional regimens, the highest total dose was 31.5 Gy. In the currently ongoing dose escalation study the highest prescribed dose is 38 Gy/1fr [61].
The optimal time gap from radiotherapy to surgery
Notable in the above systematic review is the lack of a standardized radiotherapy dose and time interval between the end of radiotherapy and the performance of surgery. In the studies reviewed, the time varies from 1 week to 8 months. In ongoing ABLATIVE-2 study, the time between radiation therapy and surgery it is 12 months [62]. This is the longest time interval used in the analyzed completed and ongoing studies of preoperative radiotherapy in breast cancer [9].
The pCR rate following preoperative SBRT seems to be positively correlated with the duration between radiotherapy and surgery in a cohort of early-stage breast cancer patients possessing favorable prognostic factors. Vasmel JE et al. [21, 22] reported 33% pCR after 6 months, and 48% pCR after 8 months from radiotherapy to BCS among early stage breast cancer patients with favorable risk factors. The above observation prompts us to look for a group of patients in whom surgery can be abandoned as in rectal cancer [63].
Bondiau et al. [20] analyzed cases of patients ineligible for BCS who received preoperative radiotherapy in combination with chemotherapy achieving a pCR of 36% after 21–24 weeks. Guanglei Chen et al. [23] in a group of patients with TNBC breast cancer using radiotherapy in combination with chemo-immunotherapy (ICI: adebrelimab) achieved higher pCR rates: 90% after 21–23 weeks. In analogous group of patients (TNBC breast cancer patient who are candidates to neoadjuvant chemotherapy and received immunochemotherapy without preoperative radiotherapy) KEYNOTE522 study pCR: 58–64,8% were achieved [64]. It is speculated that preoperative SBRT exerts synergy with immunochemotherapy.
Similarly, high percentage of pCR were achieved in the PEARL study pCR at 74% in TNBC after SBRT: 24Gy/3fr and: with anti-PD(L)1—pembrolizumab in addition to neoadjuvant chemotherapy (NAC) [65].
Pathologic CR was achieved in less than 10% of cases in studies where the time gap from the end of radiotherapy to surgery was less than 13 weeks [16–19].
Markers of response to preoperative SBRT
In addition to determining the optimal time gap from radiotherapy to surgery translating into the highest pCR rates, research is underway to identify markers indicative of response to preoperative treatment. Bosma et al. analyzed gene expression patterns among 77 patients who received preoperative radiotherapy (10 × 4 Gy in 10 days or 5 × 6 Gy in 5 days) and a lumpectomy 6 weeks thereafter and observed no or very limited response in 22 patients. Clinically significant differences in gene expression between patients with and without response to radiotherapy were not identified. However, the authors found, by comparing samples before and after radiotherapy, that genes involved in p53 signaling, TNFA1 signaling, apoptosis, epithelial mesenchymal transition, and inflammatory response were upregulated, and genes involved in mitotic spindle, G2M checkpoint, and E2F targets were downregulated [66]. High Ki-67 before NAC was a predictor for pCR in breast cancer patients. Ongoing trials may explore whether this approach will be effective in preoperative radio-chemotherapy [67]. In ABLATIVE trial Vasmel et al. revealed that after preoperative PBI the number of Tumor -Infiltrating Lymphocytes (TILs) in tumor tissue decreased, but no differences in numbers of pre irradiation TILs between responders and non-responders were observed [68]. Radiological, biomolecular or genomic biomarkers are searched to identify the group of good responders to radiotherapy for whom surgery could be omitted. In ongoing trials, the Ki67 levels, gene expression and circulating target free DNA are being analyzed as potential predictors of local control response. At the same time, there are reports of the possibility of omitting adjuvant radiotherapy in a group of patients over 65 with early-stage breast cancer with favorable prognostic factors [69]. The trend in de-escalation of breast cancer treatment translating into a reduction in toxicity has been strong in recent years. Perhaps in the future, patients with favorable prognostic factors will be able to choose to use SBRT or BCS as their sole treatment modality.
New technologies
New image-guided radiotherapy (IGRT) technologies are emerging including intra-fractional tracking of tracers placed in the tumor/tumor bed using Cyberknife (Accuray, Palo Alto CA), magnetic resonance-based linear accelerators (MR LINACS), or the Gamma-pod equivalent of Gammaknife for breast cancer treatment which are being used in more trials of preoperative stereotactic radiotherapy [52, 70–72].
Currently ongoing trials
The Table 5 summarizes currently ongoing trials using preoperative stereotactic radiotherapy in the treatment of breast cancer.
Table 5.
Ongoing trials on preoperative PBI with SBRT
| ClinicalTrials.gov ID/ Study status | Inclusion criteria | Study plan | Primary/secondary endpoints | |
|---|---|---|---|---|
| Feasibility Study of Stereotactic Body Radiotherapy for Early Breast Cancer (ARTEMIS) [73] | NCT02065960/ Unknown status | Age ≥ 70 years; invasive carcinoma, G I/II, ER and PR positive, HER2 negative Stage I (T1 N0 M0) on clinical and radiological assessment with MRI of the breasts and axillary ultrasound; Candidate for BCS; | SBRT: 40 Gy in 5 fractions delivered every other day,followed by BCS |
Primary: Feasibility Secondary: Acute and late Radiation Toxicity, pCR, Ipsilateral Breast Tumour Recurrence, DFS |
| Preoperative Single-Fraction Radiotherapy in Early Stage Breast Cancer [74] | NCT02482376/ Active, not-recruiting | Age ≥ 60 years or 50–59 with a low Oncotype score (0–17); Clinical T1N0M0 invasive carcinoma or DCIS < or equal to 2cm; ER positive; HER2 negative; Candidate for BCS; | SBRT: 21Gy followed by breast conserving surgery |
Primary: Physician reported cosmesis outcomes Secondary: Ki-67 as a measure of tumor response;Patient reported cosmesis outcomes; LC; Assess the impact of RT on: gene expression, circulating cell free DNA |
| CMP-001 and Pre-operative Stereotactic Body Radiation Therapy (SBRT) in Early Stage Triple Negative Breast Cancer (TNBC) [75] | NCT04807192/ recruiting | Age ≥ 18 years; TNBC, cT1-2, at least 5 mm, cN0-1 cM0); ECOG: 0–1; planned BCS or mastectomy; No planned neoadjuvant chemotherapy/endocrine therapy or other anticancer therapy; Presence of measurable disease in the breast; Primary tumor accessible to injections and biopsy. The lesion to be injected should be confined in a single irradiation volume that does not result in more than 30% of the whole breast; The injected tumor should be located at least 5 mm from the skin or pectoral muscle |
Arm 1: SBRT: 1 × 8 Gy at D1 Arm 2: CMP-001 (4 sequential administrations of CMP001 at Day 1 (SC), Day 5 (± 1) (IT), Day 9 (± 1) (IT) and Day 16 (± 1) (IT) + SBRT 1 × 8 Gy at D1 |
Increase in sTILs, Toxicity of CMP-001 combined with SBRT, pCR, minimal residual cancer, Ki-67 levels, OS, DFS,event-free survival (EFS) |
| Study of Stereotactic Radiotherapy for Breast Cancer [76] | NCT03043794/ recruiting | Age ≥ 50 years; Invasive ductal carcinoma; T1 N0; Clearly demarcated tumor on magnetic resonance imaging (MRI); ER and/or PR ≥ 10%; HER2 negative; Candidate for BCS + SLNB; | SBRT: 21 Gy followed by standard of care surgery | RCB, pCR, treatment-related toxicity, cosmetic outcome, time to recurrence, QoL |
|
Breast Cancer Study of Stereotactic Body Radiation Therapy (SBRT) Combined Neoadjuvant Treatment (Study to Evaluate the Efficacy and Safety of Stereotactic Body Radiation Therapy (SBRT) Combined Neoadjuvant Treatment for Patients With Triple-negative and Hormone Receptor-positive, HER2-negative Breast Cancer) [23, 77 ] |
NCT05132790/ Active, not-recruiting | Age: 18–75 years; invasive adenocarcinoma of the breast; TNBC patients HER2-neu 0–1 + by IHC or FISH-negative; ER-positive and HER2-negative breast cancer; tumor ≥ 2 cm (mammogram, breast ultrasound, breast MRI); Any nodal status; ECOG 0 -1 |
Experimental: TNBC (SHR-1316 at a dose 20mg/kg q3w): Combination of SBRT and SHR1316, followed by SHR1316 plus nab-paclitaxel and carboplatin or cisplatin Experimental: HER2-/HR + BC (SHR6390 at a dose of 150mg orally, daily): SHR6390 plus exemestane with/without ovarian function suppression/ablation (OFS) after SBRT |
pCR, RCB, objective response rate (ORR), AE,serious adverse events (SAE) |
| Stereotactic Image-Guided Neoadjuvant Ablative Radiation Then Lumpectomy (SIGNAL 2) [78] | NCT02212860/ Active, not-recruiting | Age ≥ 50 years and postmenopausal; Tumor size < 3 cm; ER positive; Invasive ductal carcinoma or other favorable subtypes (lobular, medullary, papillary, colloid, mucinous, or tubular); cN0; Surgical expectation that a > 2 mm margin can be obtained; Lesion 1 cm or greater from the skin surface; surgery within 14–20 days of radiation therapy; Able to tolerate the prone position | Stereotactic image-guided neoadjuvant ablative radiation (randomized to 1 × 21 Gy or 3 × 10 Gy) followed by lumpectomy | Immune priming, angiogenesis, proliferation, apoptosis, hypoxia, invasion markers level, toxicity |
| Preoperative Stereotactic Ablative Body Radiotherapy (SABR) for Early-Stage Breast Cancer [79] |
NCT03137693/ terminated |
Age ≥ 50 years; Invasive adenocarcinoma of breast; marker clip in breast; Unifocal tumor = < 2 cm based on contrast-enhanced prone-breast MRI; cN0; candidate for BCS; Able to tolerate the prone position; target lesion at least 10 mm from skin defined on MRI; ER positive; Her-2 negative; No received or planned fof neoadjuvant chemotherapy prior to SABR or surgery; ECOG 0–2 |
SABR: 28.5 Gy in 3 fractions BCS 6–8 weeks after |
pCR |
| SABR-CaRe in Early Stage Breast Cancer [80] | NCT04959474/ recruiting | Age ≥ 40 years (with TNBC ≥ 50 years); DCIS or invasive breast cancer; BMI > = 21 at time of enrollment; KPS: 70 – 100; Tumor size = < 3.0 cm; Gross disease may be multifocal as long as the total extent of tumor, gross and microscopic, occupies a volume with greatest dimension = < 3 cm; Patient is not being considered for preoperative chemotherapy |
Arm I: standard dietary recommendations, SABR every other day for 5 fr, surgery within 4–12 weeks, surgical resection with SLNB) Arm II: caloric restriction diet, SABR every other day for 5 fr, surgery within 4–12 weeks, surgical resection with SLNB |
Percent reduction in cellularity of breast tumor |
|
Preoperative Boost Associated With Neoadjuvant Chemotherapy in Luminal B Breast Cancer IBISCO TRIAL [81] |
NCT05673304/ recruiting | Age ≥ 18 years; Luminal B BC, cT1-2; Indication for neoadjuvant chemotherapy | SBRT as an anticipated boost with a total dose of 24 Gy (8 Gy × 3 fractions QD) within 2 weeks from the start of neoadjuvant chemotherapy | pCR, acute/late toxicity |
| Three Fraction Radiation to Induce Immuno-Oncologic Response (TRIO) [82] | NCT03978663/ recruiting | Age ≥ 18 years; locally advanced breast cancer defined as stages IIB-III (excluding inflammatory breast cancer). Stage IIA is eligible for TNBC and HER2-positive breast cancers; Invasive mammary carcinoma of any subtype excluding lobular, sarcomatous, or metaplastic subtypes, or with lobular features; Plan to be treated with neoadjuvant chemotherapy; M0 | SBRT: 3 fractions administered as an anticipated boost prior to neoadjuvant chemotherapy | pCR, Immune priming, surgical wound healing, LRR |
| Neo-adjuvant Chemotherapy Combined With Stereotactic Body Radiotherapy to the Primary Tumour + /- Durvalumab, + /- Oleclumab in Luminal B Breast Cancer: (Neo-CheckRay) [83] | NCT03875573/ recruiting | Age ≥ 18 years; luminal B breast cancer subjects candidate for neo-adjuvant chemotherapy |
Arm I: Experimental: Chemotherapy and pre-operative radiation therapy (boost dose) 3 × 8 Gy Arm II: Experimental: like Arm I plus durvalumab Arm III: Experimental: like Arm I plus durvalumab plus oleclumab |
valuation of the immune related or radiation therapy related toxicity, Evaluation of the feasibility of the primary surgery, Phase II: Demonstration of the tumour response in arms 2 or 3 versus arm 1 |
|
GammaPod Dose Escalation Radiation for Early Stage Breast Cancer (GCC 1926- Phase Ib Dose Escalation of Single-Fraction Preoperative Stereotactic Partial-Breast Irradiation for Early-Stage Breast Cancer) [84] |
NCT04234386/ recruiting | Patients > 45 years; unifocal visible on a CT scan tumor; N0; ER positive; HER2 negative; invasive ductal carcinoma; candidates for BCS; Tumor must not involve the overlying skin or underlying chest wall; Greatest tumor dimension is < 3 cm based on US. MR imaging measurements can be included only if performed before the biopsy; patients weigh < 150 kg; Patients must be able to lie prone position; no lymphovascular invasion on biopsy |
single-fraction radiation dose delivered with the GammaPod as a PBI before a lumpectomy Dose Level 1: 21 Gy Dose Level 2: 24 Gy Dose Level 3: 27 Gy Dose Level 4: 30 Gy |
Establish the single-fraction radiation dose (MTD), Incidence of Dose-Limiting Toxicities (DLTs), pCR, ipsilateral LRR |
|
Single Fraction Preoperative Radiotherapy for Early Stage Breast Cancer |
NCT04679454/ recruiting | Histologically proven unifocal adenocarcinoma; T1-T2; tumor size up to 2.5 cm; cN0; Age ≥ 18 years old; ECOG 0–2; Planned BCS |
phase I dose escalation study to identify the maximum tolerated dose (MTD) delivering 18 Gy, 21 Gy and 24 Gy in single fraction phase II study to evaluate the rate of pathological complete response (pCR) when a dose level is chosen according to the results of the previous phase I study Surgical tumor removal will be scheduled after 4–8 weeks after radioablation. Whole breast radiation therapy without boost will be performed after surgery |
Identify the maximum tolerated dose (MTD), pCR, chronic toxicity, Cosmetic outcomes, Post surgery complications, DFS, Local relapse, OS, breast cancer specific survival (BCSS) |
| Single-dose Preoperative Partial Breast Irradiation in Low-risk Breast Cancer Patients (ABLATIVE-2) [86] | NCT05350722/ recruiting | WHO ≤ 2; Age ≥ 50 years; unifocal cT1N0 breast cancer on MMG, ultrasound and MRI; Patients with an indication for chemotherapy or HER2-targeted therapy are not eligible. Patients with an indication for endocrine therapy are eligible; Tumor size as assessed on MRI; grade 1–2; Non-lobular invasive histological type carcinoma; LCIS or (non-extensive) DCIS is accepted; ER positive; HER2 negative; cN0 | PBI with a single dose of 20Gy/15Gy on the GTV and CTV respectively | pCR 12 months after radiotherapy |
| A Phase I Dose Escalation Study of Single Fraction Ablative Pre-operative Partial Breast (S-PBI) for Early Stage Breast Cancer [61] | NCT04040569 /recruiting | Age > / = 18 years old; cT1-T2cN0; Tumor must be unifocal, not involve the overlying skin; must be visible on CT scan and/or preferably marked with clip(s) in tumor; ER/PR positive, Her2 negative | Preoperative single dose escalation study: 30Gy/1fr; 34Gy/1fr; 38Gy/1fr | Dose escalation studyLC, acute/late toxicity/ cosmetic outcomes |
| Phase 2 Surgical Excision vs Neoadjuvant Radiotherapy + Delayed Surgical Excision of Ductal Carcinoma (NORDIS) [87] | NCT03909282/recruiting | Core needle biopsy demonstrating DCIS; Mammographic or MRI non-mass lesion measuring 4 cm or less in greatest dimension; ER/PR positive or negative DCIS; HER2 positive, negative or unknown DCIS; biopsy marker placed within the tumor bed confirmed on post biopsy imaging and evidence of residual radiographic abnormality; Placement of Savi scout optical reflectance marker in tumor bed area as a wireless guide for surgery and for neoRT treatment planning is preferred; Planned lumpectomy. ECOG: 0, 1, or 2 |
Active Comparator: Surgical excision of ductal carcinoma Experimental: Neoadjuvant partial breast irradiation delivered once a day for 5 days before surgery. The planned daily dose is 6 Gy |
pCR correlation of DCIS subtype with pCR assessment pre and post therapy of tumor grade, atypia, necrosis |
Conclusion
Preoperative SBRT is a promising treatment option for breast cancer patients. The precise localization of the irradiated area (targeting the tumor rather than the tumor bed) is a notable feature. Early treatment toxicity is reported to be relatively low. However, the existing data are derived from phase I and II trials focusing on small patient cohorts with limited follow-up, leading to a gap in information regarding late complications or long-term efficacy. Currently, MRI appears to be the optimal imaging modality for planning preoperative SBRT. While various fractionation schemes of SBRT are being explored, there is a lack of comparative studies among them. The key issue seems to be to determine the optimal time interval between the applied radiotherapy and surgery. Given the observed increase in percentage of clinical and pathological CR with the time between radiotherapy and surgery, it seems possible, in future, to select patients who obligatorily require surgical treatment and those in whom surgical treatment can be safely omitted.
Abbreviations
- BCS
Breast conserving surgery
- BCT
Breast conserving treatment
- cN0
Clinically node negative
- CT
Computed tomography
- CTCAE
Common terminology criteria for adverse events
- CTV
Clinical target volume
- DCIS
Ductal carcinoma in situ
- DFS
Disease-free survival
- EBRT
External beam radiation therapy
- ECOG
Eastern cooperative oncology group
- EORTC
European organisation for research and treatment of cancer
- ER
Estrogen receptor
- FISH
Fluorescence in situ hybridization
- GTV
Gross tumor volume
- HER2
Human epidermal growth factor receptor 2
- IMRT
Intensity modulated radiation therapy
- Ki67
A marker for proliferation
- LC
Local control
- LINAC
Linear accelerator
- LVSI
Lymphovascular space invasion
- MRI
Magnetic resonance imaging
- NA-PBI
Neoadjuvant partial breast irradiation
- NSABP
National surgical adjuvant breast and bowel project
- NR
Not reported
- OARs
Organs at risk
- OS
Overall survival
- pCR
Pathological complete response
- PgR
Progesterone receptor
- PRISMA
Preferred reporting items for systematic reviews and meta-analyses
- PTV
Planning target volume
- QoL
Quality of life
- RCB
Residual cancer burden
- RTOG
Radiation therapy oncology group
- SABR
Stereotactic ablative body radiotherapy
- SBRT
Stereotactic body radiotherapy
- SC
Subcutaneous
- SLNB
Sentinel lymph node biopsy
- TNBC
Triple negative breast cancer
- VMAT
Volumetric modulated arc therapy
- WBRT
Whole breast radiotherapy
- PBI
Partial breast irradiation
- MTD
Maximum tolerated dose
- LRR
Local-regional recurrence
- DLT
Dose-limiting toxicity
- BCSS
Breast cancer-specific survival
- MMG
Mammography
- US
Ultrasound
- RT
Radiotherapy
- CR
Complete response
- AEs
Adverse events
- KPS
Karnofsky performance status
Author contributions
MB: conceptualization, methodology, writing—original draft, formal analysis and data curation, KK-B: writing—original draft and formal analysis, MAZ: writing—review & editing and validation, SC: writing—review & editing, MH: data curation and visualisation, MCL: writing—review & editing, MG: data curation and visualization, AG: data curation, PH: data curation, BAJ-F: writing—review & editing and supervision, JF: writing—review & editing and supervision, ŁK: formal analysis, validation, writing—review & editing and supervision.
Funding
No funding was received for this systemic review.
Availability of data and materials
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and Consent to participate
Not applicable as no new human participants were directly involved in this systemic review.
Competing interests
All authors declare that they have no conflicts of interest to disclose.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Arnold M, Morgan E, Rumgay H, et al. Current and future burden of breast cancer: global statistics for 2020 and 2040. Breast. 2022;66:15–23. 10.1016/j.breast.2022.08.010. 10.1016/j.breast.2022.08.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dafni U, Tsourti Z, Alatsathianos I. Breast cancer statistics in the European Union: incidence and survival across European countries. Breast Care. 2019;14(6):344–53. 10.1159/000503219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Orrantia-Borunda E, Anchondo-Nuñez P, Acuña-Aguilar LE, Gómez-Valles FO, Ramírez-Valdespino CA. Subtypes of Breast Cancer. Breast Cancer. August 2022:31–42. 10.36255/EXON-PUBLICATIONS-BREAST-CANCER-SUBTYPES [PubMed]
- 4.Krishnamurti U, Silverman JF. HER2 in breast cancer: A review and update. Adv Anat Pathol. 2014;21(2):100–7. 10.1097/PAP.0000000000000015. 10.1097/PAP.0000000000000015 [DOI] [PubMed] [Google Scholar]
- 5.Female Breast Cancer Subtypes — Cancer Stat Facts. https://seer.cancer.gov/statfacts/html/breast-subtypes.html. Accessed January 21, 2024.
- 6.Loibl S, Azim HA, Bachelot T, et al. ESMO Expert Consensus Statements on the management of breast cancer during pregnancy (PrBC). Ann Oncol Off J Eur Soc Med Oncol. 2023;34(10):849–66. 10.1016/J.ANNONC.2023.08.001. 10.1016/J.ANNONC.2023.08.001 [DOI] [PubMed] [Google Scholar]
- 7.Dwyer, Mary. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines ® ) Breast Cancer NCCN.org NCCN Guidelines for Patients ® available at www.nccn.org/patients. 2023. https://www.nccn. Accessed January 21, 2024.
- 8.Shah C, Bauer-Nilsen K, McNulty RH, Vicini F. Novel radiation therapy approaches for breast cancer treatment. Semin Oncol. 2020;47(4):209–16. 10.1053/J.SEMINONCOL.2020.05.003. 10.1053/J.SEMINONCOL.2020.05.003 [DOI] [PubMed] [Google Scholar]
- 9.Jacobson GM, Takita C. Editorial : New Approaches to Breast Cancer Radiotherapy. 2021;11(February):10–2. 10.1056/NEJMoa0906260. 10.1056/NEJMoa0906260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mauri D, Pavlidis N, Ioannidis JPA. Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst. 2005;97(3):188–94. 10.1093/JNCI/DJI021. 10.1093/JNCI/DJI021 [DOI] [PubMed] [Google Scholar]
- 11.Cardoso F, Kyriakides S, Ohno S, et al. Early breast cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up †. Triple-negative breast cancer - Clin results Biomark Anal GeparNuevo study. 2019;30:1194–220. 10.1093/annonc/mdz173. 10.1093/annonc/mdz173 [DOI] [Google Scholar]
- 12.Corradini S, Krug D, Meattini I, et al. Preoperative radiotherapy: A paradigm shift in the treatment of breast cancer? A review of literature. Crit Rev Oncol Hematol. 2019;141:102–11. 10.1016/J.CRITREVONC.2019.06.003. 10.1016/J.CRITREVONC.2019.06.003 [DOI] [PubMed] [Google Scholar]
- 13.Alongi F, Arcangeli S, Filippi AR, Ricardi U, Scorsetti M. Review and uses of stereotactic body radiation therapy for oligometastases. Oncologist. 2012;17(8):1100–7. 10.1634/theoncologist.2012-0092. 10.1634/theoncologist.2012-0092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barry A, Fyles A. Establishing the role of stereotactic ablative body radiotherapy in early-stage breast cancer. Int J Breast Cancer. 2018;2018(1):2734820. 10.1155/2018/2734820. 10.1155/2018/2734820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guidolin K, Yaremko B, Lynn K, Gaede S, Kornecki A, Muscedere G, BenNachum I, Shmuilovich O, Mouawad M, Yu E, Sexton T. Stereotactic image-guided neoadjuvant ablative single-dose radiation, then lumpectomy, for early breast cancer: the SIGNAL prospective single-arm trial of single-dose radiation therapy. Curr Oncol. 2019;26(3):334–40. 10.3747/co.26.4479. 10.3747/co.26.4479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tiberi D, Vavassis P, Nguyen D, Guilbert MC, Simon-Cloutier A, Dubé P, Gervais MK, Sideris L, Leblanc G, Hijal T, Dufresne MP. Tumour response 3 months after neoadjuvant single-fraction radiotherapy for low-risk breast cancer. Curr Oncol. 2020;27(3):155–8. 10.3747/co.27.6059. 10.3747/co.27.6059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liveringhouse CL, Mills MN, Ahmed KA, et al. Phase 2 study of preoperative SABR for early-stage breast cancer: introduction of a novel form of accelerated partial breast radiation therapy. Int J Radiat Oncol Biol Phys. 2023;116(3):611–6. 10.1016/J.IJROBP.2022.12.036. 10.1016/J.IJROBP.2022.12.036 [DOI] [PubMed] [Google Scholar]
- 18.Meattini I, Francolini G, Di Cataldo V, et al. Preoperative robotic radiosurgery for early breast cancer: results of the phase II ROCK trial (NCT03520894). Clin Transl Radiat Oncol. 2022;37:94. 10.1016/J.CTRO.2022.09.004. 10.1016/J.CTRO.2022.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Horton JK, Blitzblau RC, Yoo S, et al. Preoperative single-fraction partial breast radiotherapy – a novel phase i dose-escalation protocol with radiation response biomarkers. Int J Radiat Oncol Biol Phys. 2015;92(4):846. 10.1016/J.IJROBP.2015.03.007. 10.1016/J.IJROBP.2015.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bondiau PY, Courdi A, Bahadoran P, et al. Phase 1 clinical trial of stereotactic body radiation therapy concomitant with neoadjuvant chemotherapy for breast cancer. Int J Radiat Oncol Biol Phys. 2013;85(5):1193–9. 10.1016/j.ijrobp.2012.10.034. 10.1016/j.ijrobp.2012.10.034 [DOI] [PubMed] [Google Scholar]
- 21.Vasmel JE, Charaghvandi RK, Houweling AC, et al. Tumor response after neoadjuvant magnetic resonance guided single ablative dose partial breast irradiation. Int J Radiat Oncol Biol Phys. 2020;106(4):821–9. 10.1016/j.ijrobp.2019.11.406. 10.1016/j.ijrobp.2019.11.406 [DOI] [PubMed] [Google Scholar]
- 22.Vasmel JE, Groot Koerkamp ML, Mandija S, et al. Dynamic contrast-enhanced and diffusion-weighted magnetic resonance imaging for response evaluation after single-dose ablative neoadjuvant partial breast irradiation. Adv Radiat Oncol. 2022;7(2): 100854. 10.1016/J.ADRO.2021.100854. 10.1016/J.ADRO.2021.100854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen G, Gu X, Xue J, Zhang X, Yu X, Zhang Y, Li A, Zhao Y, He G, Tang M, Xing F. Effects of neoadjuvant stereotactic body radiotherapy plus adebrelimab and chemotherapy for triple-negative breast cancer: A pilot study. Elife. 2023;22(12):e91737. 10.7554/eLife.91737. 10.7554/eLife.91737 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.De Caluwe A, Romano E, Poortmans P, et al. First-in-human study of SBRT and adenosine pathway blockade to potentiate the benefit of immunochemotherapy in early-stage luminal B breast cancer: results of the safety run-in phase of the NeoCheckRay trial. J Immunother Cancer. 2023;11(12):1–12. 10.1136/jitc-2023-007279. 10.1136/jitc-2023-007279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koenig JL, Kozak MM, Sabolch A, et al. Use of preoperative radiation therapy in early-stage and locally advanced breast cancer data source. Cureus. 2019. 10.7759/cureus.5748. 10.7759/cureus.5748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lightowlers SV, Boersma LJ, Fourquet A, et al. Preoperative breast radiation therapy: Indications and perspectives. Eur J Cancer. 2017;82:184–92. 10.1016/j.ejca.2017.06.014. 10.1016/j.ejca.2017.06.014 [DOI] [PubMed] [Google Scholar]
- 27.Van Der Leij F, Bosma SCJ, Van De Vijver MJ, et al. First results of the preoperative accelerated partial breast irradiation (PAPBI) trial. Radiother Oncol. 2015;114(3):322–7. 10.1016/j.radonc.2015.02.002. 10.1016/j.radonc.2015.02.002 [DOI] [PubMed] [Google Scholar]
- 28.Nichols E, Kesmodel SB, Bellavance E, et al. Preoperative accelerated partial breast irradiation for early-stage breast cancer: preliminary results of a prospective, phase 2 trial. Int J Radiat Oncol Biol Phys. 2017;97(4):747–53. 10.1016/j.ijrobp.2016.11.030. 10.1016/j.ijrobp.2016.11.030 [DOI] [PubMed] [Google Scholar]
- 29.Ahmed M, Jozsa F, Douek M. A systematic review of neo-adjuvant radiotherapy in the treatment of breast cancer. 10.3332/ecancer.2021.1175 [DOI] [PMC free article] [PubMed]
- 30.Piras A, Sanfratello A, Boldrini L, et al. Stereotactic radiotherapy in early-stage breast cancer in neoadjuvant and exclusive settings: a systematic review. Oncol Res Treat. 2023;46(3):116–23. 10.1159/000528640. 10.1159/000528640 [DOI] [PubMed] [Google Scholar]
- 31.Civil YA, Jonker LW, Groot Koerkamp MPM, et al. Preoperative partial breast irradiation in patients with low-risk breast cancer: a systematic review of literature. Ann Surg Oncol. 2023;30(6):3263–79. 10.1245/S10434-023-13233-9/TABLES/3. 10.1245/S10434-023-13233-9/TABLES/3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li YR, Barry PN. Pre-operative partial breast irradiation: revolutionizing radiation treatment for women with early stage breast cancer. Ann Breast Surg Open Access J Bridge Breast Surg Across World. 2022. 10.21037/abs-20-133. 10.21037/abs-20-133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Brown JM, Carlson DJ, Brenner DJ. The tumor radiobiology of SRS and SBRT: Are more than the 5 Rs involved? Int J Radiat Oncol Biol Phys. 2014;88(2):254–62. 10.1016/j.ijrobp.2013.07.022. 10.1016/j.ijrobp.2013.07.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garau MM. Radiobiology of stereotactic body radiation therapy (SBRT). Rep Pract Oncol Radiother. 2017;22(2):86–95. 10.1016/j.rpor.2017.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Craig DJ, Nanavaty NS, Devanaboyina M, et al. The abscopal effect of radiation therapy. Futur Oncol. 2021;17(13):1683–94. 10.2217/fon-2020-0994. 10.2217/fon-2020-0994 [DOI] [PubMed] [Google Scholar]
- 36.Brackstone M. Preoperative radiotherapy to optimize immune priming in breast cancer: Early results from SIGNAL Trials. Br J Surg. 2022;109(3):237–8. 10.1093/bjs/znab471. 10.1093/bjs/znab471 [DOI] [PubMed] [Google Scholar]
- 37.Formenti SC, Demaria S. Radiation therapy to convert the tumor into an in situ vaccine. Int J Radiat Oncol Biol Phys. 2012;84(4):879–80. 10.1016/j.ijrobp.2012.06.020. 10.1016/j.ijrobp.2012.06.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Golden EB, Marciscano AE, Formenti SC. Radiation therapy and the in situ vaccination approach. Int J Radiat Oncol Biol Phys. 2020;108(4):891–8. 10.1016/j.ijrobp.2020.08.023. 10.1016/j.ijrobp.2020.08.023 [DOI] [PubMed] [Google Scholar]
- 39.Zhang Y, Xu Z, Chen H, Sun X, Zhang Z. Survival comparison between postoperative and preoperative radiotherapy for stage I – III non - inflammatory breast cancer. Sci Rep. 2022. 10.1038/s41598-022-18251-3. 10.1038/s41598-022-18251-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vermeulen S, Cotrutz C, Morris A, et al. Accelerated partial breast irradiation: using the CyberKnife as the radiation delivery platform in the treatment of early breast cancer. Front Oncol. 2011. 10.3389/fonc.2011.00043. 10.3389/fonc.2011.00043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jardel P, Kammerer E, Villeneuve H, Thariat J. Stereotactic radiation therapy for breast cancer in the elderly. 2020;9(Suppl 1):86–96. 10.21037/tcr.2019.07.18. 10.21037/tcr.2019.07.18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yang TJ, Tao R, Elkhuizen PHM, Van Vliet-Vroegindeweij C, Li G, Powell SN. Tumor bed delineation for external beam accelerated partial breast irradiation: A systematic review. Radiother Oncol. 2013;108(2):181–9. 10.1016/j.radonc.2013.05.028. 10.1016/j.radonc.2013.05.028 [DOI] [PubMed] [Google Scholar]
- 43.Van MAM, Elkhuizen PHM, Minkema D, Duppen JC. Multiinstitutional study on target volume delineation variation in breast radiotherapy in the presence of guidelines. Radiother Oncol. 2010;94(3):286–91. 10.1016/j.radonc.2010.01.009. 10.1016/j.radonc.2010.01.009 [DOI] [PubMed] [Google Scholar]
- 44.Nichols EM, Dhople AA, Mohiuddin MM, Flannery TW, Yu CX, Regine WF. Comparative analysis of the post-lumpectomy target volume versus the use of pre-lumpectomy tumor volume for early-stage breast cancer: implications for the future. Int J Radiat Oncol Biol Phys. 2010;77(1):197–202. 10.1016/j.ijrobp.2009.04.063. 10.1016/j.ijrobp.2009.04.063 [DOI] [PubMed] [Google Scholar]
- 45.Kirwan CC, Al Sarakbi W, Loncaster J, Chan HY, Thompson AM, Wishart GC. Tumour bed clip localisation for targeted breast radiotherapy: compliance is proportional to trial-related research activity: tumour bed clip localisation in breast radiotherapy. Eur J Surg Oncol. 2014;40(2):158–62. 10.1016/j.ejso.2013.11.016. 10.1016/j.ejso.2013.11.016 [DOI] [PubMed] [Google Scholar]
- 46.Pezner RD, Tan MC, Clancy SL, Chen YJ, Joseph T, Vora NL. Radiation therapy for breast cancer patients who undergo oncoplastic surgery: localization of the tumor bed for the local boost. Am J Clin Oncol. 2013;36(6):535–9. 10.1097/COC.0B013E318256EFBA. 10.1097/COC.0B013E318256EFBA [DOI] [PubMed] [Google Scholar]
- 47.Van Der Leij F, Elkhuizen PHM, Janssen TM, et al. Target volume delineation in external beam partial breast irradiation: Less inter-observer variation with preoperative-compared to postoperative delineation. Radiother Oncol. 2014;110(3):467–70. 10.1016/j.radonc.2013.10.033. 10.1016/j.radonc.2013.10.033 [DOI] [PubMed] [Google Scholar]
- 48.Den Hartogh MD, Philippens MEP, van Dam IE, et al. MRI and CT imaging for preoperative target volume delineation in breast-conserving therapy. Radiat Oncol. 2014;9(1):1–9. 10.1186/1748-717X-9-63. 10.1186/1748-717X-9-63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Vasmel JE, Groot Koerkamp ML, Kirby AM, et al. Consensus on contouring primary breast tumors on mri in the setting of neoadjuvant partial breast irradiation in trials. Pract Radiat Oncol. 2020;10(6):e466–74. 10.1016/j.prro.2020.03.011. 10.1016/j.prro.2020.03.011 [DOI] [PubMed] [Google Scholar]
- 50.Zerella MA, Zaffaroni M, Ronci G, et al. A narrative review for radiation oncologists to implement preoperative partial breast irradiation. Radiol Med. 2023;128(12):1553–70. 10.1007/S11547-023-01706-6. 10.1007/S11547-023-01706-6 [DOI] [PubMed] [Google Scholar]
- 51.Zerella MA, Zaffaroni M, Ronci G, et al. Single fraction ablative preoperative radiation treatment for early-stage breast cancer : the CRYSTAL study – a phase I / II clinical trial protocol. BMC Cancer. 2022. 10.1186/s12885-022-09305-w. 10.1186/s12885-022-09305-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Berlangieri A, Elliott S, Wasiak J, Chao M, Foroudi F. Use of magnetic resonance image-guided radiotherapy for breast cancer: a scoping review. J Med Radiat Sci. 2022;69(1):122–33. 10.1002/jmrs.545. 10.1002/jmrs.545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Livi L, Meattini I, Marrazzo L, et al. Accelerated partial breast irradiation using intensity-modulated radiotherapy versus whole breast irradiation: 5-year survival analysis of a phase 3 randomised controlled trial. Eur J Cancer. 2015;51(4):451–63. 10.1016/j.ejca.2014.12.013. 10.1016/j.ejca.2014.12.013 [DOI] [PubMed] [Google Scholar]
- 54.Marta GN, Coles C, Kaidar-Person O, et al. The use of moderately hypofractionated post-operative radiation therapy for breast cancer in clinical practice: A critical review. Crit Rev Oncol Hematol. 2020. 10.1016/J.CRITREVONC.2020.103090. 10.1016/J.CRITREVONC.2020.103090 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.McCormick B, Winter KA, Woodward W, et al. Randomized Phase III trial evaluating radiation following surgical excision for good-risk ductal carcinoma in situ: long-term report from NRG oncology/RTOG 9804. J Clin Oncol. 2021;39(32):3574–82. 10.1200/JCO.21.01083. 10.1200/JCO.21.01083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bouganim N, Tsvetkova E, Clemons M, Amir E. Evolution of sites of recurrence after early breast cancer over the last 20 years: Implications for patient care and future research. Breast Cancer Res Treat. 2013;139(2):603–6. 10.1007/S10549-013-2561-7/METRICS. 10.1007/S10549-013-2561-7/METRICS [DOI] [PubMed] [Google Scholar]
- 57.Ott OJ, Stillkrieg W, Lambrecht U, et al. External-beam-accelerated partial-breast irradiation reduces organ-at-risk doses compared to whole-breast irradiation after breast-conserving surgery. Cancers (Basel). 2023;15(12):1–13. 10.3390/cancers15123128. 10.3390/cancers15123128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zureick AH, Grzywacz VP, Almahariq MF, et al. Dose to the left anterior descending artery correlates with cardiac events after irradiation for breast cancer. Int J Radiat Oncol Biol Phys. 2022;114(1):130–9. 10.1016/j.ijrobp.2022.04.019. 10.1016/j.ijrobp.2022.04.019 [DOI] [PubMed] [Google Scholar]
- 59.Yadav BS, Loganathan S, Sharma SC, Singh R, Dahiya D. Comparison of toxicity and cosmetic outcomes after accelerated partial breast irradiation or whole breast irradiation using 3-dimensional conformal external beam radiation therapy. Adv Radiat Oncol. 2020;5(2):171–9. 10.1016/j.adro.2019.09.005. 10.1016/j.adro.2019.09.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Haussmann J, Budach W, Corradini S, et al. Comparison of adverse events in partial- or whole breast radiotherapy: investigation of cosmesis, toxicities and quality of life in a meta-analysis of randomized trials. Radiat Oncol. 2023;18(1):1–23. 10.1186/s13014-023-02365-7. 10.1186/s13014-023-02365-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Study Details | A Phase I Dose Escalation Study of Single Fraction Ablative Pre-operative Partial Breast (S-PBI) for Early Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT04040569. Accessed January 21, 2024.
- 62.Study Details | Single-dose Preoperative Partial Breast Irradiation in Low-risk Breast Cancer Patients | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05350722. Accessed January 21, 2024.
- 63.Heald RJ, Beets G, Carvalho C. Report from a consensus meeting: response to chemoradiotherapy in rectal cancer – predictor of cure and a crucial new choice for the patient. Color Dis. 2014;16(5):334–7. 10.1111/CODI.12627. 10.1111/CODI.12627 [DOI] [PubMed] [Google Scholar]
- 64.Schmid P, Cortes J, Pusztai L, et al. Pembrolizumab for early triple-negative breast cancer. N Engl J Med. 2020;382(9):810–21. 10.1056/nejmoa1910549. 10.1056/nejmoa1910549 [DOI] [PubMed] [Google Scholar]
- 65.McArthur HL, Shiao S, Karlan S, et al. Abstract PD10–01: The PEARL trial: Pre-operative pembrolizumab with radiation therapy in early stage triple negative breast cancer. Cancer Res. 2022. 10.1158/1538-7445.SABCS21-PD10-01. 10.1158/1538-7445.SABCS21-PD10-01 [DOI] [Google Scholar]
- 66.Bosma SCJ, Hoogstraat M, van der Leij F, et al. Response to preoperative radiation therapy in relation to gene expression patterns in breast cancer patients. Int J Radiat Oncol Biol Phys. 2020;106(1):174–81. 10.1016/j.ijrobp.2019.09.002. 10.1016/j.ijrobp.2019.09.002 [DOI] [PubMed] [Google Scholar]
- 67.Chen X, He C, Han D, et al. 2017 The predictive value of Ki-67 before neoadjuvant chemotherapy for breast cancer: a systematic review and meta-analysis. 13(9):843–857. 10.2217/FON-2016-0420 [DOI] [PubMed]
- 68.Vasmel JE, Vreuls CPH, Manson QF, et al. Clinical investigation tumor-infiltrating lymphocytes in low-risk patients with breast cancer treated with single-dose preoperative partial breast irradiation. Int J Radiat Oncol Biol Phys. 2021. 10.1016/j.ijrobp.2020.12.009. 10.1016/j.ijrobp.2020.12.009 [DOI] [PubMed] [Google Scholar]
- 69.Kunkler IH, Williams LJ, Jack WJL, Cameron DA, Dixon JM. Breast-conserving surgery with or without irradiation in early breast cancer. N Engl J Med. 2023;388(7):585–94. 10.1056/nejmoa2207586. 10.1056/nejmoa2207586 [DOI] [PubMed] [Google Scholar]
- 70.Swamy K. Stereotactic body radiotherapy immunological planning—a review with a proposed theoretical model. Front Oncol. 2022;12(January):1–15. 10.3389/fonc.2022.729250. 10.3389/fonc.2022.729250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Obayomi-Davies O, Kole TP, Oppong B, et al. Stereotactic accelerated partial breast irradiation for early-stage breast cancer: Rationale, feasibility, and early experience using the cyberknife radiosurgery delivery platform. Front Oncol. 2016;6(MAY):1–6. 10.3389/fonc.2016.00129. 10.3389/fonc.2016.00129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yu CX, Mutaf Y, Regine WF, Feigenberg SJ, Nichols EM, Hoban PW. GammaPod: a dedicated device for external beam breast cancer SBRT in the prone position. Int J Radiat Oncol. 2015;93(3):E586–7. 10.1016/j.ijrobp.2015.07.2046. 10.1016/j.ijrobp.2015.07.2046 [DOI] [Google Scholar]
- 73.Study Details | Feasibility Study of Stereotactic Body Radiotherapy for Early Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT02065960?cond=breast cancer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=1. Accessed January 21, 2024.
- 74.Study Details | Preoperative Single-Fraction Radiotherapy in Early Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT02482376?cond=breast can-cer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=2. Accessed January 21, 2024.
- 75.Study Details | CMP-001 and Pre-operative Stereotactic Body Radiation Therapy (SBRT) in Early Stage Triple Negative Breast Cancer (TNBC) | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT04807192?cond=breast can-cer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=3. Accessed January 21, 2024.
- 76.Study Details | Study of Stereotactic Radiotherapy for Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03043794?cond=breast cancer&term=Preoperative&intr=Stereotactic Body Radithapy%2FSBRT&rank=5. Accessed January 21, 2024.
- 77.Study Details | Breast Cancer Study of Stereotactic Body Radiation Therapy (SBRT) Combined Neoadjuvant Treatment | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05132790?cond=breast can-cer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=6. Accessed January 21, 2024.
- 78.Study Details | Stereotactic Image-Guided Neoadjuvant Ablative Radiation Then Lumpectomy | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT02212860?cond=breast cancer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=7. Accessed January 21, 2024.
- 79.Study Details | Preoperative Stereotactic Ablative Body Radiotherapy (SABR) for Early-Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03137693?cond=breast cancer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=9. Accessed January 21, 2024.
- 80.Study Details | SABR-CaRe in Early Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT04959474?cond=breast cancer&term=Preoperative&intr=Stereotactic Body Radiotherapy%2FSBRT&rank=10. Accessed January 21, 2024.
- 81.Study Details | Preoperative Boost Associated With Neoadjuvant Chemotherapy in Luminal B Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05673304?cond=%22Breast Cancer%22 OR %22breast neoplasm%22 OR %22breast tumor%22 &term=preopera-tive OR induction OR neoadiuvant&intr=%22stereotactic ablative body radiotherapy%22 OR %22stereotactic body radiation thera-py%22 OR SBRT OR SABR&rank=10. Accessed January 21, 2024.
- 82.Study Details | Three Fraction Radiation to Induce Immuno-Oncologic Response | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03978663?cond=%22Breast Cancer%22 OR %22breast neoplasm%22 OR %22breast tumor%22 &term=preopera-tive OR induction OR neoadiuvant&intr=%22stereotactic ablative body radiotherapy%22 OR %22stereotactic body radiation thera-py%22 OR SBRT OR SABR&rank=11. Accessed January 21, 2024.
- 83.Study Details | Neo-adjuvant Chemotherapy Combined With Stereotactic Body Radiotherapy to the Primary Tumour +/- Durvalumab, +/- Oleclumab in Luminal B Breast Cancer: | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03875573?cond=%22Breast Cancer%22 OR %22breast neoplasm%22 OR %22breast tumor%22 &term=preopera-tive OR induction OR neoadiuvant&intr=%22stereotactic ablative body radiotherapy%22 OR %22stereotactic body radiation thera-py%22 OR SBRT OR SABR&rank=14. Accessed January 21, 2024.
- 84.Study Details | GammaPod Dose Escalation Radiation for Early Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT04234386?cond=%22Breast Cancer%22 OR %22breast neoplasm%22 OR %22breast tumor%22 &term=preopera-tive OR induction OR neoadiuvant&intr=%22stereotactic ablative body radiotherapy%22 OR %22stereotactic body radiation thera-py%22 OR SBRT OR SABR&rank=16#study-plan. Accessed January 21, 2024.
- 85.Study Details | Single Fraction Preoperative Radiotherapy for Early Stage Breast Cancer | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT04679454. Accessed January 21, 2024.
- 86.Study Details | Single-dose Preoperative Partial Breast Irradiation in Low-risk Breast Cancer Patients | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05350722. Accessed January 20, 2024.
- 87.Study Details | Phase 2 Surgical Excision vs Neoadjuvant Radiotherapy+Delayed Surgical Excision of Ductal Carcinoma | ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT03909282. Accessed January 21, 2024.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.