Summary
Background
Canadian Arctic communities have experienced sustained syphilis transmission, with diagnoses rates 18-times higher than the national average. Remoteness from laboratory facilities leads to delays between syphilis screening and treatment, contributing to onward transmission. Rapid diagnostic tests can eliminate treatment delays via testing at the point-of-care. This study aims to describe syphilis diagnostic gaps and to estimate the impact of introducing rapid diagnostic tests at the point-of-care on syphilis transmission.
Methods
To assess the population-level impact of deploying rapid diagnostic tests, an individual-based model was developed using detailed surveillance data, population surveys, and a prospective diagnostic accuracy field study. The model was calibrated to syphilis diagnoses (2017–2022) from a community of approximately 1,050 sexually active individuals. The impacts of implementing rapid diagnostic tests using whole blood (sensitivity: 92% for infectious and 81% for non-infectious syphilis; specificity: 99%) from 2023 onward was calculated using the annual median fraction of cumulative new syphilis infections averted over 2023–2032.
Findings
The median modeled syphilis incidence among sexually active individuals was 44 per 1,000 in 2023. Males aged 16–30 years exhibited a 51% lower testing rate than that of their female counterparts. Maintaining all interventions constant at their 2022 levels, implementing rapid diagnostic tests could avert a cumulative 33% (90% credible intervals: 18–43%) and 37% (21–46%) of new syphilis infections over 5 and 10 years, respectively. Increasing testing rates and contact tracing may enhance the effect of rapid diagnostic tests.
Interpretation
Implementing rapid diagnostic tests for syphilis in Arctic communities could reduce infections and enhance control of epidemics. Such effective diagnostic tools could enable rapid outbreak responses by providing same-day testing and treatment at the point-of-care.
Funding
Canadian Institutes of Health Research.
Keywords: Syphilis, Rapid diagnostic tests, Sexual health, Mathematical modelling, Inuit health
Research in context.
Evidence before this study
Based on data from the World Health Organization (WHO), a steady global rise in syphilis cases has been documented over the last decade, with over 7 million new cases reported in 2020. In Canada, annual infectious syphilis incidence statistics highlighted the disproportionate burden among Indigenous people, reaching up to 18 times the national average diagnosis rate in some regions. The reasons for this burden are complex —including prejudices, injustices, and other structural barriers— but treatment delays play a role and the use of rapid diagnostic tests could help alleviate some of these issues. We performed a PubMed search (from database inception to 2024-03-24), using the following terms: syphilis AND (Indigen∗ OR “First Nations” OR “Aborig∗”) AND (“rapid diagnos∗” OR RDT OR test∗ OR screen∗). The uncovered literature found this issue to be prevalent worldwide. More importantly, the high syphilis burden has resulted in increased cases of congenital syphilis, a condition that can have fatal consequences. Although syphilis can be effectively treated with penicillin once detected, the remoteness of some Indigenous communities poses challenges to timely diagnosis and treatment, elevating the risk of individuals being lost to follow-up after testing. With the development of rapid diagnostic tests, it is possible to diagnose and treat syphilis cases at the point-of-care, eliminating the gap between testing, reception of the positive result, and treatment. In the most recent systematic review available, Zhang et al. showed high sensitivity and specificity of rapid diagnostic tests of syphilis: pooled sensitivity of the treponemal component were 93% (95% CI: 86–97%) and specificity of 98% (95% CI: 96–99%); pooled sensitivity for non-treponemal component at 90% (95% CI: 82–95%) and specificity at 97% (95% CI: 92–99%). Additionally, the CDC Laboratory Recommendations for Syphilis Testing, released in early 2024, underscored the necessity of evaluating the effect of rapid diagnostic tests at the point-of-care in settings lacking regular medical services. However, the population-level impact of implementing those tests on syphilis transmission remains to be quantified.
Added value of this study
We quantified the population-level effect of deploying rapid diagnostic tests at the point-of-care in remote Indigenous communities of Nunavik (Canada) on syphilis transmission. Using a detailed mathematical model, different interventions and scenarios were examined and compared to a modeled counterfactual scenario without RDT. These results will provide support to the implementation of rapid diagnostic tests in remote communities where barriers to healthcare remain.
Implications of all the available evidence
Implementing rapid diagnostic tests for syphilis in Arctic communities could reduce the number of syphilis infections and enhance control of those epidemics. These effective diagnostic tools can enable rapid outbreak responses by providing same-day testing and treatment directly at the point-of-care. Additionally, rapid diagnostic tests provide a flexible tool to reach populations with unmet testing and treatment needs of syphilis.
Introduction
Syphilis is a preventable and treatable sexually transmitted infection that can be acquired through direct contact with lesions during sexual contact.1 If left untreated, it poses substantial health risks, especially in the case of vertical transmission (congenital syphilis) which can have devastating consequences on the fetus.2 Over the past decade, a consistent upsurge in syphilis cases has been documented globally, with over 7 million reported cases worldwide in 2020, with no signs of abatement.3, 4, 5
Indigenous populations often bear a disproportionately high burden of syphilis.6 In Canada, the reported incidence of infectious syphilis among Indigenous people in 2021 soared to 556 per 100,000 person in Nunavik —the northern third of the province of Québec, located above the 55th parallel.7 This is 18 times the average rate in Canada for the same year.8 Similar trends are also observed in other Indigenous territories across Canada.9, 10, 11 The high syphilis burden, especially among women, in these communities is of particular concern given their high birth rates (twice the national birth rate).12,13 Surges in cases of congenital syphilis are already observed as a consequence.14,15
Despite responses from communities and public health authorities to curb syphilis epidemics in these isolated communities, transmission persists. Potential contributors to the high levels of transmission include, among others, diagnostic and treatment delays.16 These delays stem from the lack of local laboratory capacity. Collected specimens must be sent to distant laboratories —up to 1,900 kilometers south— for diagnosis. The typical resulting delay between sample collection and analysis is 6–19 days.17 The time between reception of the result and treatment administration adds to this lag (e.g., challenges in reaching individuals).18 Lengthy response delays increase the likelihood of loss to follow-up (LTFU) for those found to be positive for syphilis.
Rapid diagnostic tests for syphilis are designed to facilitate immediate clinical decision-making during one encounter. When used at the point-of-care, it presents an opportunity to reduce the delays between consultation, diagnosis, and treatment.19 Moreover, rapid diagnostic tests require minimal training of health personnel. A recent multisite prospective field diagnostic accuracy study in an intended-use setting in Nunavik and Nunavut reported that frontline sexual health nurses could accurately identify untreated patients with presumed infectious syphilis using a dual antigen syphilis rapid diagnostic tests (the STAR study).17 Successful implementation of rapid diagnostic tests for syphilis screening in antenatal settings to reduce time to treatment has already been demonstrated in several countries,20, 21, 22, 23 along with qualitative investigations in outpatients and rural clinic settings.24 However, the potential population-level impact of deploying rapid diagnostic tests at the point-of-care on syphilis transmission in isolated communities is unknown.25 Furthermore, other epidemic drivers of sustained transmission, including heterogeneity in sexual behaviors and unmet prevention needs among these mostly Indigenous communities have yet to be understood.
In this study, we 1) describe diagnostic gaps and transmission drivers for syphilis and 2) assess the fraction of new syphilis infections that would be averted using rapid diagnostic tests at the point-of-care over a 10-year time horizon. We do so by developing, parameterizing, and calibrating a detailed individual-based mathematical model of syphilis transmission, leveraging strong local surveillance, programmatic, and survey data.
Methods
Model overview
An individual-based model (IBM) was developed to simulate syphilis transmission in a high-burden Arctic community in northern Québec, Canada (exact location redacted to preserve anonymity). The choice of an IBM enables the integration of complex interactions between biological epidemic drivers, sexual networks, and interventions such as contact tracing.26,27 Additionally, it can incorporate a high degree of behavioral heterogeneities and examine different “what if” scenarios.
The model was implemented using a modular coding structure with 4 main components: demographic processes (births, aging, migration, deaths), sexual behaviors (e.g., regular/casual partnership formation and dissolution), syphilis transmission and disease progression, and interventions (i.e., testing, contact tracing, and treatment). The demographic processes were set to match the population's age and sex profiles from census data. The model was initiated with 1,050 sexually active syphilis naïve heterosexual individuals aged 14–60 years old in 2016 (in line with demographic information from the modeled community), one year before some of the first large outbreaks were detected by public health authorities.28 A comprehensive description of model and parameters can be found in Supplementary Methods (see Supplementary Figures S1–S8 and Supplementary Tables S1–S10).
Sexual networks and behaviors
Sexual behaviors were parametrized based on data from the population-based QANUILIRPITAA? 2017 Nunavik Inuit Health survey among local residents in Nunavik which included a module on sexual health and behaviors.29 Specifically, we used information on the reported number of sexual partners in the last 12 months, partnership types (regular/casual), and condom use at the last sexual encounter. At the beginning of each calendar year, individuals are assigned a number of partners based on their sexual activity levels at model entry (low = 0–1 partner; medium = 2–3 partners; high = 4 or more partners). The time of partnership formation was determined simultaneously. To balance the number of sexual partners by sex, women look for partners according to an age mixing matrix taken from the general Canadian population.30 Sexual mixing by activity levels is calibrated and considered to vary between totally assortative and proportionate mixing.31 Individuals can only have one concurrent regular partnership (average duration of 2 years); nevertheless, there is no limit on the number of casual partnerships (duration of 1 day).32 Each partnership is assigned a number of daily sex acts (all types), that depends on partnership type,33,34 and a probability of condom use at each sexual intercourse that differs by sexual activity levels.
Syphilis transmission and progression
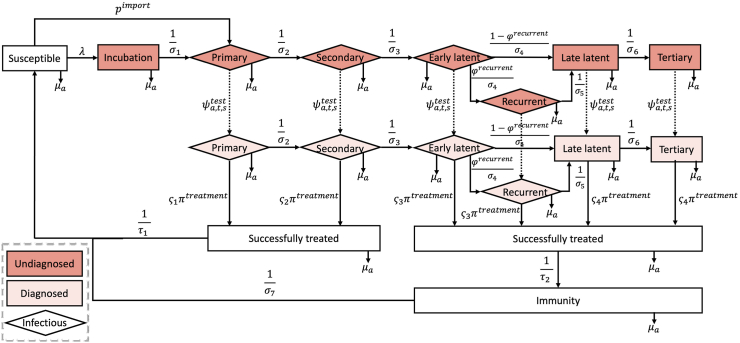
Following syphilis acquisition, individuals can progress through the following stages: incubation, primary, secondary, early latent, late latent, and tertiary stage (Fig. 1). About 25% of those in the early latent stage may relapse back to the secondary stage before going into the late latent stage.35 Only cases treated later than secondary stage can gain temporary immunity against reinfections.36 We do not consider syphilis mortality in this model given the extremely low likelihood of death from syphilis in adults.
Fig. 1.
Schematic diagram of the individual-based model's structure for the natural history of syphilis and its testing and treatment cascade. Where is the age-specific all-cause mortality rate; is the individual's probability of infection; is the probability of importing syphilis from another community; is duration of each syphilis disease stage; is the proportion of early latent cases that progress through the recurrent stage; is the probability of testing for syphilis, which varies by age, time, and sex; is the sensitivity of the conventional syphilis testing assay for each disease stage k; is the probability that treatment is successful; and is the duration of treatment for patients initiated at different syphilis disease stage.
Susceptible individuals can be infected by a partner with infectious syphilis (cases at primary, secondary, and recurrent stage). The probability of transmission is a function of the number of sex acts and whether a condom is used. As per Canadian guidelines, we assumed that people diagnosed with syphilis will not have condomless intercourse until treatment is completed for both partners. In addition, individuals in the high sexual activity group may acquire syphilis outside of the community (imported cases).
Syphilis testing and linkage to treatment
Syphilis can be diagnosed through routine testing, mass screening events, follow-up test after syphilis treatment, and contact tracing activities. Parameters for this module were informed by the Nunavik syphilis screening database (which records all syphilis tests performed in Nunavik, along with socio-demographic information), data collected from the STAR study,18 and local health providers' knowledge. The first two diagnosis paths are captured by assigning a time-varying daily probability of testing based on the individual's age and sex that mimics the population's heterogenous test-seeking behaviors. The conventional testing assay used to diagnose syphilis is a combined treponemal-specific enzyme immunoassay and rapid plasma reagin.37 It has a sensitivity that varies by disease stage: 94.5% (primary stage), 100% (secondary, early latent, and recurrent stage), and 98.5% (late latent and tertiary stage).38 Its specificity is 100%.39
A delay between specimen collection and reporting of results to local practitioners is assigned to each test. It is sampled from the empirical distribution (e.g., delays directly sampled from the Nunavik syphilis screening database) of the time delays between specimen collection and analysis. For individuals with a positive syphilis test, 90% of those diagnosed cases will be linked to care and receive treatment within 7 days after the positive results (that is, we assumed 10% LTFU). Further, 10% of treated cases will experience treatment failure and continue disease progression. The duration of treatment for those diagnosed at, and prior to early latent stage, is 1 day while for the remaining stages, it will last for 21 days. Cases treated prior to late latent stage will attend 3 follow-up tests at 3, 6, and 12 months; whilst those receiving treatment later will be followed at 12 and 24 months.37 We assumed that there is a 50% probability of adherence to follow-up tests.
Furthermore, syphilis being a reportable disease, sexual contacts of cases from the preceding 3 months will be traced and tested at different probabilities that depend on partnership type and the infection stage of the index case (e.g., partners of those at primary and secondary stages are more likely to be traced). Additionally, a proportion of partners identified through contact tracing will receive treatment at specimen collection (hereafter, “proportion of partner treated onsite”) regardless of the results, allowing people to refuse the treatment without a positive result.
Rapid diagnostic tests
We modeled the use of rapid diagnostic tests (Chembio DPP Syphilis Screen & Confirm Assay) using uncentrifuged whole blood as a diagnostic tool at the point-of-care. Rapid diagnostic tests are being administered in parallel to the standard-of-care serum tests. That is, everyone tested for syphilis will be tested using rapid diagnostic tests at the time of specimen collection along with the conventional laboratory-based tests. In this case, individuals with reactive rapid diagnostic test will be immediately linked to care and treated onsite without LTFU. Cases missed by rapid diagnostic tests (false negatives) will be picked up by the conventional testing algorithm and receive the treatment with the regular delay and probability of LTFU. The diagnostic accuracy of rapid diagnostic tests was based on estimates obtained in the STAR study conducted in the region using whole blood specimens: we set the treponemal component of the rapid diagnostic tests' sensitivity at 92% for infectious syphilis, 81% for non-infectious (approximated using overall sensitivity), and the specificity at 99%.17
Syphilis prevention gaps and impacts of rapid diagnostic tests
First, we examined the demographic characteristics of individuals that acquired and transmitted syphilis in relation to their syphilis testing rates (by age, sex, and sexual activity levels), as well as their estimated time between infection and diagnosis using the IBM. Second, we estimated the impact of implementing rapid diagnostic tests from 2023 onwards on syphilis transmission. This was assessed while maintaining the same testing, contact tracing, LTFU rates, and proportion of partners treated onsite as those in 2022. In addition, we elaborated several complementary scenarios considering the following factors: testing rates at 75%, 125%, 150% of the average testing rate in 2022; contact tracing rates ranging from no tracing, 50% of the observed 2022 rate, and up to 100% tracing; higher rapid diagnostic tests sensitivity set at 100% for infectious and 95% for non-infectious syphilis, as observed for the non-treponemal component using serum sample17; LTFU when diagnosed using regular testing with 70% and 80% cases being treated within 7 days after positive result reporting; and 50% and 100% of partners treated onsite.
For all scenarios, the annual fraction of cumulative number of syphilis infections averted by comparing the rapid diagnostic tests scenarios above to the current standard-of-care (counterfactual):
where t represents the calendar year; and and denote the cumulative number of syphilis infections with and without rapid diagnostic tests, respectively. To improve our counterfactual comparisons, the number of syphilis infections in the scenario without rapid diagnostic tests was computed over the same sexual network as the one with rapid diagnostic tests (i.e., removing the stochasticity due to demographic processes and partnerships formation and dissolution).
Model calibration
An Approximate Bayesian Computation Sequential Monte Carlo (ABC-SMC) algorithm40 was used to select a total of 100 posterior parameter sets that meets the tolerance criteria that best reproduced the observed annual number of sex-stratified syphilis diagnoses in the community from 2017 to 2022. In addition, we performed cross-validation of the following modeled outcomes with the surveillance and laboratory databases: proportion of reinfections, proportion of cases diagnosed at infectious stage, and sex-stratified annual testing rates. Detailed calibration algorithm and cross-validation outcomes are summarized in Appendix A. The median and 90% credible interval (CrI) of the 100 calibrated parameter sets, which were each based on the median of 20 stochastic simulations, are reported.
The model has a time step of 1 day and it was coded in R version 4.3.1 using a C++ back-end, implemented with the Rcpp library.41 The reporting of this manuscript follows the CDC Emerging Infectious Diseases—Mathematical Modeling Guidelines: Editorial criteria for mathematical, economic, and statistical manuscripts.42
Ethical approval
This study has been approved by the Research Institute of the McGill University Health Centre (#2020-5834). The QANUILIRPITAA? 2017 Nunavik Inuit Health Survey received ethical approval from the Centre de recherche du CHU de Québec, Université Laval (REB 2016-2499, 2016-2499-21), and McGill University (REB 20-04-034).
Role of funding source
This work was funded by the Canadian Institutes of Health Research (CIHR; award PJT-162262). YX received a Doctoral Research Award from CIHR. MM-G's research program is supported by a Canada Research Chair (Tier II) in Population Health Modeling. CPY holds a Clinical Research Scholar career award from the Fonds de recherche du Québec-Santé (FRQS). The funders had no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript.
Results
Model fits
The calibrated model was able to replicate the observed annual number of diagnosed syphilis cases by age and sex in the community from 2017 to 2022 (Fig. 2). In terms of in-sample validation, the mean absolute error of the model was 6.8 cases overall (6.0 cases for females, 1.6 for males, 2.9 for 14–22-year-olds, 1.8 for 23–30-year-olds, and 5.5 for 31–60-year-olds). The cross-validation targets indicated a good alignment between the simulated and observed values (Supplementary Figures S6–S8).
Fig. 2.
Observed (red dots) and modeled (lines) number of syphilis diagnoses (1st row) overall, by sex, and (2nd row) by broad age groups in the study community located in Nunavik (Québec, Canada). The observed number of cases are from the Nunavik syphilis surveillance database and are used as a calibration outcome. The lines correspond to the median of the simulations and the shaded areas to the 90% credible intervals.
Description of the syphilis epidemic
Surveillance data
From 2017 to 2022, a total of 269 syphilis cases were diagnosed in the community among individuals aged ≥14 years (approximately 1,000 to 1,200 individuals). The diagnosis rate peaked at 52 per 1,000 people in 2020 with sustained high incidence over 40 per 1,000 during the following 2 years. The diagnosis rates for women were consistently higher than those among men (Fig. 3). Although the vast majority of the cases were concentrated among those aged 20–44 years, the proportion of cases occurring among 14–19-year-olds increased over time. Testing data in 2022 suggest that young males aged 15–29 years old test for syphilis at rates that are 50% lower than females of the same age group. Testing positivity was higher among males across all age groups. Those aged 30–44 years had the highest testing positivity across sex (4.2% [55/1,303] for females and 4.6% [38/824] for males). Notably, the positivity rate among the youngest (14–19 years) and oldest (45–60 years) increased over time for both sexes. An average delay of 7 days (range: 3–41 days) between specimen collection and positive results reporting, were observed.
Fig. 3.
Number of diagnosed syphilis cases by sex, broad age groups, and year (2017–2022) in the study community located in Nunavik (Québec, Canada). Data source: Nunavik syphilis surveillance database.
Prevention gaps estimated from the individual-based model
Assuming all factors unchanged, the model projected an incidence of 44 (90% CrI: 15–66) per 1,000 sexually active people in 2023. The model suggested that males aged 15–29 years disproportionally contribute to syphilis transmissions: they constitute 24% of the population but transmit close to 40% of infections that occurred over 2017–2022. Additionally, 44% (90% CrI: 40–49%) of transmissions were between casual partners. Individuals with more than 1 partner in the last 12 months accounted for 74% (90% CrI: 67–86%) of the total number of transmission events despite constituting 30–40% of the population. The high sexual activity group (10% of the population) contributed to most of transmission events (81%, 90% CrI: 75–89%) but acquired proportionally fewer new infections (47%, 90% CrI: 37–67%). The average time from infection to diagnosis was 165 days (90% CrI: 156–182 days) over 2017–2022 but varied between sex: from 159 days (90% CrI: 156–182 days) among women to 174 days (90% CrI: 164–191 days) among men. Our analysis suggested that importation of cases from outside the community is important in sustaining transmission: on average we estimated 4–5 imported cases every year since 2018 in the modeled community. At the end of 2022, 22% (90% CrI: 29–45%) of individuals in the high sexual activity population were estimated to be infected with or to have developed temporary immunity to Treponema pallidum.
Potential impact of implementing rapid diagnostic tests
Maintaining all interventions constant at their 2022 levels (testing rate, contact tracing rate, and have 90% of the cases being treated with 7 days after diagnosis), implementing rapid diagnostic tests using whole blood samples as a screening tool prior to laboratory testing could avert a cumulative 33% (90% CrI: 18–43%) of new syphilis infections over 5 years (Fig. 4). This proportion increases to 37% (90% CrI: 21–46%) over 10 years. If rapid diagnostic tests were to be performed using serum sample (higher sensitivity), the impact of rapid diagnostic tests would not be meaningfully affected. Lowering the proportion of individuals with a reactive rapid diagnostic test treated to 95% did not impact the result. To isolate the effect of rapid diagnostic tests on reducing the gap between infection and diagnosis from contact tracing (because finding more new cases results in more contact tracing), a scenario without partner tracing was examined. In this scenario, implementing rapid diagnostic tests reduces the time to diagnosis by 20 days.
Fig. 4.
Annual fractions of cumulative syphilis infections averted 2023–2032 by implementing rapid diagnostic tests since 2023 in the study community located in Nunavik (Québec, Canada). Dots represent the median and the error bars the 90% credible intervals.
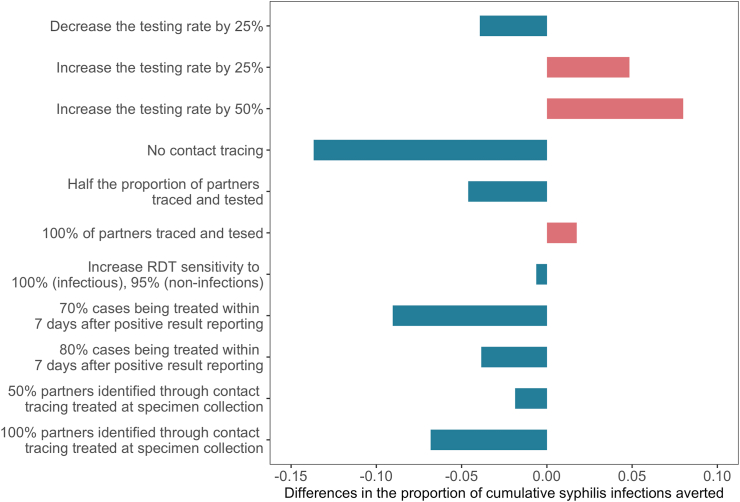
With all other factors unchanged, increasing testing rates may contribute to important reduction in the fraction of cumulative syphilis infections averted (Fig. 5). By increasing the 2022 syphilis testing rate by 50% from 2023 onwards, an additional 8% and 9% cumulative syphilis infections can be averted over 5 and 10 years, respectively. On the other hand, decreasing the probability of contact tracing will cancel out the effects. Notably, if there's no contact tracing, the fraction averted will be 14%-points lower compared to the current main scenario. Increasing LTFU for cases diagnosed with regular testing and treating more partners onsite have a negative impact on the effect of rapid diagnostic tests. The negative effects observed for lowering loss to follow-up, increasing rapid diagnostic tests sensitivity, and treating more partners on site may be explained by the decreased susceptible population.
Fig. 5.
Differences between fractions of cumulative syphilis infections averted under different scenarios with varying contact tracing rate and testing rate, as compared to the fraction with the same testing rates and probability of contact tracing in 2022. The x-axis represents the absolute variation (in percentage-points) in the averted fraction of syphilis infections over 5 years.
Discussion
To curb syphilis transmission in remote Arctic communities, where access to and resources for healthcare are limited, it is essential to improve the early detection and treatment of individuals with syphilis.16 Rapid diagnostic tests can bridge this gap by informing clinical decisions at the point-of-care. Using a detailed model of syphilis transmission in a community in Nunavik (Quebec, Canada) with high syphilis incidence, this study suggests that implementing rapid diagnostic tests as a screening tool may avert 1 in 3 of the cumulative syphilis infections over the next five years. The impact of rapid diagnostic tests is further improved if overall testing rates are increased, as well as with enhanced partner contact tracing.
Our results highlight the potential of rapid diagnostic tests for syphilis outbreak responses in isolated communities. Rapid diagnostic tests can be used as a screening tool by non-laboratorians, in complementarity to the use of conventional laboratory assays. Although previous studies considered the “acceptable trade-off” of lower sensitivities of rapid diagnostic tests using whole blood sample in clinical settings for early diagnosis and treatment at the point-of-care,43 our results indicate a minimal impact of increasing the sensitivity of rapid diagnostic tests on population-level transmission of syphilis. This can be explained by rapid diagnostic tests already achieving a high sensitivity for infectious syphilis when using whole blood samples and the fact that any false negative rapid diagnostic tests results would be confirmed positives with the conventional testing assays.
Although the effect of rapid diagnostic tests may be offset through empiric onsite treatment of partners of cases, it is rarely feasible to implement completely.44 Given its high diagnostic accuracy, rapid diagnostic tests are an important tool to inform correct clinical diagnosis, to avoid unnecessary treatment of uninfected individuals, and to detect cases that are not suspected on the basis of contact tracing investigations.45 On the other hand, the deployment of rapid diagnostic tests should not be misconstrued as an endorsement for weakening contact tracing efforts. Both earlier detection and treatment of cases, contact tracing, and rapid diagnostic tests should complement each other to better curb syphilis transmission. Rapid diagnostic tests are a flexible tool that requires minimal training and laboratory capacity and can be extended to non-traditional settings to reach individuals that could face barriers accessing healthcare.
Increasing syphilis testing rates is pivotal for outbreak control.46 The strategy of implementing rapid diagnostic tests should be accompanied by renewed efforts to increase testing rates, as exemplified by the estimated 5–6 months delay between infection and diagnosis, particularly among groups with high positivity rates. Our study highlights the missed intervention opportunities in several groups in our region: 1) individuals aged 30–44 years should be tested more often as they had the highest positivity; 2) males younger than 30 years old as they tested less often but transmit more; and 3) people with a recent travel history and several sexual partners.
Our results indicate a lower effectiveness of rapid diagnostic tests with an increase in LTFU for regular testing. This can seem counterintuitive since LTFU is a significant factor contributing to delayed treatment. The projected epidemics under different LTFU scenarios shows that, without rapid diagnostic tests and a higher LTFU, the epidemics plateaued at high incidence rates and a higher proportion of cases are treated at later stages (Supplementary Figures S9–S11). This plateauing can be explained by increased immunity against syphilis when treated late and depletion of susceptible in the high sexual activity group. This means that increasing LTFU leads to reductions in syphilis incidence (and potential risk of clinical complications), a perverse effect of screening programs that has been documented before.35,47,48
The results from this study should be interpreted considering the following main limitations. First, we assumed case importation in the high sexual activity group only and that importation risk would remain constant in the future. Second, we made additional assumptions regarding the number of sexual partners being reported and subsequently traced. However, we calibrated these parameters and conducted sensitivity analyses to examine the robustness of these assumptions. Further, there is a high level of stochasticity in our model given the small, modeled population size of a little more than 1,000 individuals, even if we improved counterfactual comparisons by matching simulations to the same sexual network. Finally, our results may only be generalizable to similar settings with comparatively long delays between specimen collection and acting on the positive results.
Strengths of our study included the detailed demographic and behavioral data from the study population, surveillance and laboratory testing data, the use of rapid diagnostic tests accuracy data obtained from the field work in the same population, and incorporation of past interventions. This enabled us to model the complex sexual and healthcare seeking behaviors, and to obtain reliable estimates of the effectiveness of rapid diagnostic tests. Moreover, the adoption of an individual-based model allowed us to incorporate the heterogeneities in sexual and testing behaviors which increases the accuracy of our estimates. Furthermore, our work assessed the impact of conventional interventions on the population-level impact of implementing rapid diagnostic tests at the point-of-care.
Conclusion
Introducing rapid diagnostic tests as a diagnostic tool for syphilis in similar Arctic communities could cut close to one third of all new syphilis infections over the next 5 years. Requiring minimal training, rapid diagnostic tests has the required flexibility to be deployed into non-traditional testing settings and reach groups with unmet syphilis testing needs that disproportionately contribute to transmission (e.g., males 15–29 years). Such effective diagnostic tools can enable rapid outbreak responses by providing same-day testing and treatment at the point-of-care. It is crucial to maintain complementary interventions such as contact tracing to maximize the effect of rapid diagnostic tests.
Contributors
YX, CPY, and MMG conceived of and designed this modeling study, with inputs from all co-authors. YX, CC, CPY, and MMG collected the information required to develop, parameterize, and calibrate the model. YX performed the literature search, conducted data analyses, and drafted the manuscript. CC, VM, AS, BS, ML, DG, TW, FX, RB, JST, CPY, and MMG interpreted results, supported data curation, critically reviewed and edited the article. YX, CC, CPY, and MMG assessed and verified the underlying data reported in the manuscript.
Data sharing statement
Access to the syphilis surveillance data can be requested from the Nunavik Regional Board of Health and Social Services (NRBHSS). Data from the QANUILIRPITAA? 2017 survey belongs to the community and requests must be sent to the QANUILIRPITAA? 2017 Data Management Committee. The remaining information is publicly available.
Declaration of interests
MM-G's research program is supported by Canadian Institutes of Health Research (CIHR) and a Canada Research Chair (Tier II) in Population Health Modeling. Both fundings are grant to McGill University. YX's work is supported by the CIHR Doctoral Research Award. CPY holds a Clinical Research Scholar career award from the Fonds de recherche du Québec-Santé (FRQS) and reports the following relationships: Independent Data Monitoring Committee (IDMC) for Medicago Inc. 2021-2022 and InventVacc Biologicals Inc. in 2024. WHO AMR Diagnostic Initiative; Technical working group member (TWG) 2023; WHO Typhoid Diagnostic Reference Panel (TyDReP) 2024-; Voting Member on Committee to Advise on Tropical Medicine and Travel (CATMAT), Public Health Agency of Canada (PHAC). These relationships have no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript.
Acknowledgements
We acknowledge all Nunavimmiut QANUILIRPITAA? 2017 survey participants and all of those who planned and carried out this survey. The list of all people who contributed to this effort can be found in the Acknowledgements section of thematic reports available on the NRBHSS website (http://nrbhss.ca/en/nrbhss/public-health/nunavik-health-surveys/qanuilirpitaa-2017). We are grateful to the survey's Data Management Committee for their collaboration throughout this project. The use of the QANUILIRPITAA? 2017 Nunavik-owned data was approved on April 5th, 2023.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.lana.2024.100845.
Appendix A. Supplementary data
References
- 1.Syphilis – CDC detailed fact sheet. Centers for Disease Control and Prevention (CDC) https://www.cdc.gov/std/syphilis/stdfact-syphilis-detailed.htm
- 2.WHO guidelines approved by the guidelines review committee. 2016. WHO guidelines for the treatment of treponema pallidum (syphilis)https://www.who.int/publications/i/item/9789241549714 [PubMed] [Google Scholar]
- 3.U.S . Centers for disease control and prevention (CDC) 2023. STI epidemic showed no signs of slowing in 2021 – cases continued to escalate.https://www.cdc.gov/media/releases/2023/s0411-sti.html [Google Scholar]
- 4.Gonorrhoea and syphilis at record levels in 2022. UK Health Security Agency; 2023. https://www.gov.uk/government/news/gonorrhoea-and-syphilis-at-record-levels-in-2022#:∼:text=infectious%20syphilis%20diagnoses%20increased%20to,with%20sexually%20transmitted%20infections%20(%20STIs%20) [Google Scholar]
- 5.STI: incident cases of syphilis in 15-49 year olds (in millions) World Health Organization; 2023. https://www.who.int/data/gho/data/indicators/indicator-details/GHO/incident-cases-of-syphilis-in-individuals-(in-millions) [Google Scholar]
- 6.Cardoso A.M., Caldas A.D.R., Oliveira E.S., et al. Maternal and congenital syphilis in indigenous peoples: a scoping review of the worldwide literature. Int J Equity Health. 2023;22(1):84. doi: 10.1186/s12939-023-01890-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Portrait des infections transmissibles sexuellement et par le sang (ITSS) au Québec: année 2021 et projections 2022. 2023. https://www.inspq.qc.ca/en/node/651816
- 8.Chlamydia, gonorrhea and infectious syphilis in Canada: 2021 surveillance data update. Public Health Agency of Canada (PHAC); 2023. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/chlamydia-gonorrhea-infectious-syphilis-2021-surveillance-data.html [Google Scholar]
- 9.Singh A.E., Kulleperuma K., Begin J., et al. Lessons from management of syphilis in Nunavut, Canada, 2012-2020. Can Commun Dis Rep. 2022;48(2-3):102–110. doi: 10.14745/ccdr.v48i23a08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Syphilis Government of Saskatchewan. https://www.saskatchewan.ca/residents/health/diseases-and-conditions/sexually-transmitted-infections-services/syphilis
- 11.Benoit P., Tennenhouse L., Lapple A., et al. Congenital syphilis re-emergence in winnipeg, manitoba. Can Commun Dis Rep. 2022;48(2-3):89–94. doi: 10.14745/ccdr.v48i23a06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Table 13-10-0418-01 Crude birth rate, age-specific fertility rates and total fertility rate (live births) Statistics Canada; 2023. https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310041801 [Google Scholar]
- 13.Nunavik in figures. 2020. https://www.nunivaat.org/doc/document/2021-09-13-01.pdf
- 14.Battams N. The Vanier Institute of the Family; 2023. Fertility rate in Canada fell to (Another) record low in 2022.https://vanierinstitute.ca/fertility-rate-in-canada-fell-to-another-record-low-in-2022/#:∼:text=According%20to%20new%20data%20published,of%20the%20COVID%2D19%20pandemic [Google Scholar]
- 15.Duhaime G., Caron A., Everett K. 2020. Nunavik in figures 2020; p. 64.https://www.nunivaat.org/doc/document/2021-09-13-01.pdf [Google Scholar]
- 16.Public health risk profile: infectious syphilis outbreaks and re-emergence of congenital syphilis in Canada. 2023. https://www.canada.ca/en/public-health/services/emergency-preparedness-response/rapid-risk-assessments-public-health-professionals/risk-profile-infectious-syphilis-outbreaks-emergence-congenital-syphilis.html
- 17.Caya C., Singh A.E., Serhir B., et al. Rapid diagnostic testing for syphilis in Arctic communities (the STAR study): a multisite prospective field diagnostic accuracy study in an intended-use setting. Clin Microbiol Infect. 2023;29(10):1335.e1–1335.e7. doi: 10.1016/j.cmi.2023.06.013. [DOI] [PubMed] [Google Scholar]
- 18.Caya C., Maheu-Giroux M., Xia Y., et al. Stopping syphilis transmission in Arctic communities through rapid diagnostic testing: the STAR study protocol. PLoS One. 2022;17(9) doi: 10.1371/journal.pone.0273713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Phang Romero Casas C., Martyn-St James M., Hamilton J., Marinho D.S., Castro R., Harnan S. Rapid diagnostic test for antenatal syphilis screening in low-income and middle-income countries: a systematic review and meta-analysis. BMJ Open. 2018;8(2) doi: 10.1136/bmjopen-2017-018132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Myer L., Wilkinson D., Lombard C., Zuma K., Rotchford K., Karim S.S. Impact of on-site testing for maternal syphilis on treatment delays, treatment rates, and perinatal mortality in rural South Africa: a randomised controlled trial. Sex Transm Infect. 2003;79(3):208–213. doi: 10.1136/sti.79.3.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ansbro E.M., Gill M.M., Reynolds J., et al. Introduction of syphilis point-of-care tests, from pilot study to national programme implementation in Zambia: a qualitative study of healthcare workers' perspectives on testing, training and quality assurance. PLoS One. 2015;10(6) doi: 10.1371/journal.pone.0127728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Garcia P.J., Carcamo C.P., Chiappe M., et al. Rapid syphilis tests as catalysts for health systems strengthening: a case study from Peru. PLoS One. 2013;8(6):e66905. doi: 10.1371/journal.pone.0066905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sweeney S., Mosha J.F., Terris-Prestholt F., et al. The costs of accessible quality assured syphilis diagnostics: informing quality systems for rapid syphilis tests in a Tanzanian setting. Health Policy Plan. 2014;29(5):633–641. doi: 10.1093/heapol/czt049. [DOI] [PubMed] [Google Scholar]
- 24.Marks M., Esau T., Asugeni R., et al. Point-of-care tests for syphilis and yaws in a low-income setting - a qualitative study of healthcare worker and patient experiences. PLoS Negl Trop Dis. 2018;12(4) doi: 10.1371/journal.pntd.0006360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Papp J.R., Park I.U., Fakile Y., Pereira L., Pillay A., Bolan G.A. CDC laboratory Recommendations for syphilis testing, United States, 2024. MMWR Recomm Rep. 2024;73(1):1–32. doi: 10.15585/mmwr.rr7301a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.White P.J. Mathematical models in infectious disease epidemiology. Infect Dis. 2017;1:49–53.e1. doi: 10.1016/b978-0-7020-6285-8.00005-8. [DOI] [Google Scholar]
- 27.Roche B., Duboz R. Individual-based models for public health. 2017;37:347–365. doi: 10.1016/bs.host.2017.08.008. [DOI] [Google Scholar]
- 28.Nunavik Health and Social Services perspective Nunavik regional board of health and social services. https://www.cerp.gouv.qc.ca/fileadmin/Fichiers_clients/Documents_deposes_a_la_Commission/P-106.pdf
- 29.QANUILIRPITAA? 2017 Nunavik Inuit health survey sexual and reproductive health report. 2017. https://nrbhss.ca/sites/default/files/health_surveys/Sexual_and_Reproductive_Health_report_en.pdf
- 30.Brisson M. HPV-advice technical appendix. http://www.marc-brisson.net/HPVadvise.pdf
- 31.Easterly C.W., Alarid-Escudero F., Enns E.A., Kulasingam S. Revisiting assumptions about age-based mixing representations in mathematical models of sexually transmitted infections. Vaccine. 2018;36(37):5572–5579. doi: 10.1016/j.vaccine.2018.07.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hui B.B., Gray R.T., Wilson D.P., et al. Population movement can sustain STI prevalence in remote Australian indigenous communities. BMC Infect Dis. 2013;13:188. doi: 10.1186/1471-2334-13-188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gray R.T., Hoare A., McCann P.D., et al. Will changes in gay men's sexual behavior reduce syphilis rates? Sex Transm Dis. 2011;38(12):1151–1158. doi: 10.1097/OLQ.0b013e318238b85d. [DOI] [PubMed] [Google Scholar]
- 34.Tuite A.R., Fisman D.N., Mishra S. Screen more or screen more often? Using mathematical models to inform syphilis control strategies. BMC Public Health. 2013;13(1):1–9. doi: 10.1186/1471-2458-13-606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Garnett G.P., Aral S.O., Hoyle D.V., Cates W., Jr., Anderson R.M. The natural history of syphilis. Implications for the transmission dynamics and control of infection. Sex Transm Dis. 1997;24(4):185–200. doi: 10.1097/00007435-199704000-00002. [DOI] [PubMed] [Google Scholar]
- 36.Magnuson H.J., Thomas E.W., Olansky S., Kaplan B.I., De Mello L., Cutler J.C. Inoculation syphilis in human volunteers. Medicine (Baltimore) 1956;35(1):33–82. doi: 10.1097/00005792-195602000-00002. [DOI] [PubMed] [Google Scholar]
- 37.Institut national d’excellence en santé et en services sociaux. 2016. Pharmacological treatment STBBI - syphilis.https://www.inesss.qc.ca/fileadmin/doc/INESSS/Outils/Guides_ITSS/INESSS_STBBI_Guide_Syphilis_EN.pdf [Google Scholar]
- 38.Park I.U., Fakile Y.F., Chow J.M., et al. Performance of treponemal tests for the diagnosis of syphilis. Clin Infect Dis. 2019;68(6):913–918. doi: 10.1093/cid/ciy558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cantor A., Nelson H.D. 2016. M D, al e. Table 3 sensitivity and specificity of commonly used syphilis tests. presented at: screening for syphilis in Nonpregnant adolescents and adults: systematic review to update the 2004 US preventive services task force recommendation.https://www.ncbi.nlm.nih.gov/books/NBK368468/table/ch1.t3/ [PubMed] [Google Scholar]
- 40.Toni T., Welch D., Strelkowa N., Ipsen A., Stumpf M.P. Approximate Bayesian computation scheme for parameter inference and model selection in dynamical systems. J R Soc Interface. 2009;6(31):187–202. doi: 10.1098/rsif.2008.0172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Eddelbuettel D., François R. Rcpp: seamless R and C++ integration. J Stat Software. 2011;40:1–18. [Google Scholar]
- 42.Mathematical modeling guidelines: editorial criteria for mathematical, economic, and statistical manuscripts. CDC Emerging Infectious Disease Journal; 2016. https://wwwnc.cdc.gov/eid/page/mathematical-modeling-guidelines [Google Scholar]
- 43.Zhang Y., Goh S.M., Mello M.B., et al. Improved rapid diagnostic tests to detect syphilis and yaws: a systematic review and meta-analysis. Sex Transm Infect. 2022;98(8):608–616. doi: 10.1136/sextrans-2022-055546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Antimicrobial resistance. World Health Organization; 2023. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance#:∼:text=AMR%20is%20a%20natural%20process,in%20humans%2C%20animals%20and%20plants [Google Scholar]
- 45.Herring A., Ballard R., Mabey D., Peeling R.W., WHO/TDR Sexually Transmitted Diseases Diagnostics Initiative Evaluation of rapid diagnostic tests: syphilis. Nat Rev Microbiol. 2006;4(12 Suppl):S33–S40. doi: 10.1038/nrmicro1563. [DOI] [PubMed] [Google Scholar]
- 46.Natoli L., Guy R.J., Shephard M., et al. Public health implications of molecular point-of-care testing for chlamydia and gonorrhoea in remote primary care services in Australia: a qualitative study. BMJ Open. 2015;5(4) doi: 10.1136/bmjopen-2014-006922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Syphilis among gay, bisexual, two-spirit and other men who have sex with men: a resource for population-specific prevention. Public Health Agency of Canada; 2015. https://www.canada.ca/en/public-health/services/infectious-diseases/sexual-health-sexually-transmitted-infections/syphilis-resource-population-specific-prevention.html [Google Scholar]
- 48.Pourbohloul B., Rekart M.L., Brunham R.C. Impact of mass treatment on syphilis transmission: a mathematical modeling approach. Sex Transm Dis. 2003;30(4):297–305. doi: 10.1097/00007435-200304000-00005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.