Abstract
Leptospirosis is a bacterial zoonotic disease of major One Health significance and public health impact globally, with a wide host range including mammals, cetaceans and herpetofauna. This study aimed to determine Leptospira seroprevalence, risk factors for seroreactivity and prevalence of urinary Leptospira shedding among domestic cats in Hong Kong.
Microagglutination testing of 22 Leptospira serovars from 20 serogroups was performed on 738 sera from outdoor free-roaming “community” cats (n = 391) and privately-owned (n = 347) cats. Urine from 268 community cats was tested for pathogenic Leptospira DNA by qPCR targeting lipL32. Potential risk factors associated with exposure were assessed using logistic regression.
Overall Leptospira seroprevalence was 9.35%. Of 14 serogroups detected, Javanica (4.3%), Djasiman (2.3%) and Australis (1.5%) were most common. Seroreactivity was significantly higher among community (13.3%) than privately-owned cats (4.9%; OR 2.98 [95% CI 1.68–5.25], P < 0.001), especially to Javanica (7.65% of community cats versus 0.58% of privately-owned cats (P < 0.001). Antibody titres to all serogroups ranged from 1:100 to 1:6400 (median 1:200) and were highest for Javanica (median 1:800).
Leptospira DNA was detected in urine from 12/268 community cats (4.48%; median load 6.42 × 102 copies/mL urine; range 1.40 × 101–9.63 × 104). One in three seroreactive community cats with paired urine and blood samples had leptospiruria. After adjusting for source, none of breed, sex, neuter status, age, district rodent infestation rate, serum alanine transaminase or creatinine values were associated with seroreactivity.
Cats in Hong Kong are exposed to a diversity of Leptospira serogroups and can shed Leptospira silently in urine. The higher seroprevalence among outdoor free-roaming community cats highlights the importance of environmental drivers in leptospirosis transmission and risks of exposure for sympatric human populations. Gloves should be worn when handling feline urine to minimise the risk of zoonotic transmission from subclinically infected cats.
Keywords: Feline, Leptospiruria, Leptospira, Microagglutination testing, Serology, Prevalence
Highlights
-
•
Cats in Hong Kong are exposed to a wide variety of Leptospira serovars, most commonly Javanica.
-
•
Around 4.5% of healthy domestic cats tested shed leptospire DNA in urine.
-
•
Community cats had an Odds Ratio of 2.98 [95% CI: 1.68–5.25] for Leptospira seropositivity compared to privately-owned cats.
-
•
Gloves should be worn when handling feline urine, even from healthy cats.
1. Introduction
Leptospirosis is a zoonotic disease of profound One Health significance and public-health impact globally. It is caused by bacterial spirochetes from the genus Leptospira which can infect humans, dogs, cats and most other terrestrial mammals as well as cetaceans and herpetofauna [1]. The estimated disease burden in 2015 of 1 million human cases resulting in 60, 000 deaths annually, is at risk of dramatic surges due to more frequent climate-change associated flooding events, especially in countries of lower socioeconomic status [2,3].
Leptospira transmission primarily occurs when mucous membranes or abraded skin come into direct contact with urine from an infected reservoir host, or with contaminated soil or water. Less commonly, Leptospira transmission occurs from being bitten by or ingesting an infected animal [4]. Acute clinical disease (leptospirosis) is most frequently reported in dogs and humans [2], whereas subclinical (asymptomatic) infections occur in many animals, including cats.
Leptospira are classified by genomic differences into 64 species that form two pathogenic [P1 (pathogenic), P2 (intermediately pathogenic)] and two saprophytic (S1, S2) subclades [5]. P1 species frequently associated with leptospirosis in humans and animals include L. interrogans, L. kirschneri, L. noguchii and L. borgpetersenii [6]. Leptospira can also be classified by their outer lipopolysaccharide antigens into >300 pathogenic serovars [5]. Strains from the same serovar may belong to different genetic species [7]. Antigenically related serovars are grouped into 30 serogroups, which can be identified using the microagglutination test (MAT).
For some Leptospira serovars infections in animal reservoirs are almost always subclinical and urinary shedding is persistent, including serovars Icterohaemorrhagiae in rodents, Hardjo in cattle and Canicola in dogs [8]. Rodents are an important infection reservoir in urban environments and can also persistently shed many other Leptospira serovars. [9]. Predation of rodents is an additional route of Leptospira transmission for cats.
In Hong Kong human leptospirosis is a notifiable disease with 1 to 8 cases reported annually from 2012 to 2022 [10]. While the prevalence of Leptospira infection in farmed pigs in Hong Kong is negligible [11], conditions are favourable for leptospirosis outbreaks due to high annual rainfall [12,13]. The proximity of rodents to humans in densely populated urban areas creates an environment further conducive to Leptospira transmission. Rodent Leptospira seroprevalence in Mainland China, which borders Hong Kong, is estimated to be 15.9% [14].
An integrated One-Health approach is critical to inform public health policy to minimise the risk of leptospirosis regionally and within certain occupation groups, such as veterinarians and farmers. Since feline Leptospira infections are always subclinical and urinary shedding occurs for up to 8 months, an increased understanding of the epidemiological role of cats in Leptospira carriage and shedding is desirable. [15].
Hong Kong has a sizable population of outdoor, free-roaming street cats, also known as “community cats”, whose human contact is limited to intermittent feeding by the public. Although exact numbers are not known, the Hong Kong SPCA has neutered >75, 000 community cats since the year 2000. In comparison, most privately-owned cats live alongside their owners in high-rise apartments with no outdoor access. The distinct cat populations and environmental conditions in Hong Kong present a unique opportunity to investigate the role of cats in Leptospira transmission and the impact of lifestyle and environment on feline Leptospira exposure.
The aims of this study were to determine Leptospira seroprevalence, risk factors for seropositivity and the prevalence of urinary Leptospira shedding among cats in Hong Kong.
2. Materials and methods
2.1. Ethics statement
Ethical approvals for this study were granted by the Animal Ethics Committee of City University of Hong Kong, approval numbers A-0478, A-0709 and A-0696. Licenses for sampling community cats were granted by The Government of the Hong Kong SAR, Department of Health, license numbers 20–164 to 20–179 and 22–6 to 22–8.
2.2. Sample collection
2.2.1. Community cats
Whole blood was collected from free-roaming, outdoor community cats presented to a trap-neuter-return program from January to May 2021 and January to August 2022. Serum was separated and stored at −80 °C until batch testing. Urine samples were also collected between January to August 2022 by manual bladder expression or cystocentesis. Urine (2.5 mL) was mixed with phosphate buffered saline (PBS, 1 mL) to prevent degradation of Leptospira DNA [7]. PBS-buffered urine samples were stored at 4 °C for up to 48 h or at −80 °C until DNA extraction. Signalment (age, breed, sex, neuter status) and capture location of each cat was recorded. The mean of monthly district rodent infestation rates, reported by the Food and Environmental Hygiene Department [16], was calculated to give annual district rodent rates between January 2020 to June 2022. Samples from a subset of 90 community cats were submitted to the CityU Veterinary Diagnostic Laboratory (VDL) to determine serum creatinine values.
2.2.2. Privately-owned cats
Residual diagnostic sera (>0.5 mL) were obtained from cats presented to CityU Veterinary Medical Centre (VMC) or from VDL between January 2020 and April 2022. Signalment, serum alanine transferase (ALT) and creatinine values were recorded where available.
A flow chart depicting recruitment and processing was created using draw.io (draw.io AG, Zurich, Switzerland).
2.3. Microagglutination testing of sera
Sera underwent MAT against a panel of 22 Leptospira serovars from 20 serogroups at the World Health Organisation Collaborating Centre for Reference and Research on Leptospirosis, Queensland Health Forensic and Scientific Services, Brisbane, Australia (Table 1). Serovars were selected to represent a wide range of serogroups, informed by serovars present in neighbouring regions [[17], [18], [19], [20], [21]]. Sera were tested at dilutions from 1:50 to 1:6400. MAT titres ≥1:100 were considered positive (reactive), in accordance with World Organisation for Animal Health guidelines [22].
Table 1.
List of Leptospira strains representing 22 Leptospira serovars used for MAT in this study, and their respective serogroup and species [41].
| Species | Serogroup | Serovar | Strain |
|---|---|---|---|
| L. borgpetersenii | Ballum | Ballum | Mus 127 |
| L. borgpetersenii | Javanica | Javanica | Veldrat Batavia 46 |
| L. borgpetersenii | Tarassovi | Tarassovi | Perepelitsin |
| L. borgpetersenii | Mini | Mini | Sari |
| L. interrogans | Australis | Australis | Ballico |
| L. interrogans | Autumnalis | Autumnalis | Akiyami A |
| L. interrogans | Bataviae | Bataviae | Swart |
| L. interrogans | Canicola | Canicola | Hond Utrecht IV |
| L. interrogans | Icterohaemorrhagiae | Copenhageni | M20 |
| L. interrogans | Djasiman | Djasiman | Djasiman |
| L. interrogans | Sejroe | Hardjo | Hardjoprajitno |
| L. interrogans | Hebdomadis | Hebdomadis | Hebdomadis |
| L. interrogans | Pomona | Pomona | Pomona |
| L. interrogans | Pyrogenes | Pyrogenes | Salinem |
| L. interrogans | Serjoe | Saxkoebing | Mus 24 |
| L. interrogans | Australis | Pohnpei | PRK12 |
| L. kirschneri | Cynopteri | Cynopteri | 3522C |
| L. kirschneri | Grippotyphosa | Grippotyphosa | Moskva V |
| L. noguchii | Panama | Panama | CZ 214 |
| L. santarosai | Shermani | Shermani | 1342 K |
| L. weilii | Celledoni | Celledoni | Celledoni |
| L. weilii | Sarmin | Sarmin | Sarmin |
2.4. Detection of Leptospira DNA in urine
2.4.1. DNA extraction
Refrigerated and frozen PBS-buffered urine samples were centrifuged at 6000 xg for 10 mins and at 13,000 x g for 5 mins, respectively. Supernatant was discarded except for 200 μL which was used to resuspend the pellet. Samples were stored at 4 °C for up to 24 h before DNA extraction using the QIAamp DNA Mini Kit (Qiagen GmbH, Hilden, Germany) with the following modification; for each reaction, 2 μL of VetMAX Xeno™ DNA Control (Thermo Fisher Scientific, Austin, United States of America) was added to the lysis solution, serving as an exogenous extraction internal positive control (IPC). DNA was eluted in 80 μL of Buffer AE (Qiagen GmbH). DNA concentration (ng/μL) was measured with the NanoDrop™ OneC Microvolume UV–Vis Spectrophotometer (Thermo Fisher Scientific) and DNA extracts were kept at 4 °C for up to 48 h or at -80 °C before PCR.
2.4.2. Internal positive control
To confirm integrity of extracted DNA, the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) housekeeping gene was amplified by PCR [23]. Samples testing negative for GAPDH were tested for the IPC by qPCR using the VetMAX™ Xeno™ IPC assay (Thermo Fisher Scientific). A DNA extract obtained from 200 μL of deionized-water containing 2 μL XENO DNA IPC was used as no-inhibition DNA control during each run. A sample was deemed inhibited when a shift in Ct value of >1.5 cycles was observed when compared to the no-inhibition DNA control. Samples failing to amplify the XENO DNA IPC were excluded from further analyses.
2.4.3. Detection of leptospiral lipL32 gene
To detect pathogenic Leptospira spp. qPCR targeting lipl32 was performed [24,25]. A pMK-T plasmid containing the lipL32 fragment was synthesized (Thermo Fisher Scientific). After in-house transformation and cloning into competent E. coli JM109 Strain (Promega) cells, plasmid DNA was purified and quantified. A standard curve from 108 to 101 target copies per reaction was generated using serial tenfold dilutions of the lipL32 plasmid. Plasmid dilutions were prepared in Tris–EDTA (TE) with 5 μg/mL salmon sperm DNA (Thermo Fisher Scientific).
To identify a 123-bp fragment of the lipL32 gene, forward lipL32-F (5’-CTGTGATCAACTATTACGGATA-3′) and reverse lipL32-R (5’-GAACTCCCATTTCAGCGAT-3′) primers, together with lipL32-P TaqMan probe (6-FAM-5’-AAAGCCAGGACAAGCGCCG-3’-BHQ1) were used [24]. Each PCR reaction contained 200 nM of the lipL32 primers and probe in a final volume of 25 μL (iQ Supermix; Bio-Rad Pacific Limited, Hong Kong SAR, China). Template DNA (10 μL) was added to each PCR reaction, with a final template concentration ranging from 4 to 100 ng per reaction. The amplification was performed on a Bio-Rad CFX96 Touch with a thermal cycling profile of 95 °C for 3 min, followed by 42 cycles of denaturation at 95 °C for 10 s and annealing-extension at 60 °C for 30 s.
No-template negative controls contained nuclease-free water and 5 μg/mL of salmon sperm DNA in TE. Samples and standards were run in triplicate and results were reported as mean values. A sample was considered positive if at least 3 target DNA copies per reaction were detected in at least two of three replicates.
Data analysis for lipL32 qPCR was performed with CFX Maestro Software. Validation parameters of R2 ≥ 0.98 for assay linearity and 90 to 110% for PCR efficiency per run were used.
2.4.4. Statistical analysis
Analyses were conducted using Stata v18 (StataCorp LLC, College Station, Texas, USA). The proportions of positive samples of tested serovars (“seroprevalence”) were graphed and compared between the two sources of cats using tests of proportions. Serum ALT and creatinine levels were categorized as “elevated” or “normal” based on reference intervals. Scatter plots comparing mean district-level rodent rate and the percentage of seropositive cats per district for all serogroups and specifically for serogroup Javanica were created.
Univariable associations between Leptospira seropositivity and each independent variable of interest (source, sex, age, breed, neuter status, serum ALT and creatinine values) were evaluated using simple logistic regression. Age was categorized into three groups (<12, 12–83, and > 83 months) to enable meaningful comparisons. Odds ratios (OR) and their corresponding 95% confidence intervals (95% CI) were estimated. Independent variables with a conservative P < 0.2 were considered for inclusion in a multivariate logistic regression model [26]. Pairwise correlations between the independent variables with P < 0.2 were assessed using Chi-square tests and where collinearity was present, a theoretical causal web guided the selection of the most meaningful variable(s) for the final multivariable model.
3. Results
3.1. Study population
The final study population comprised 773 cats, of which 426 were community cats and 347 were privately-owned (Fig. 1). Sera from 738 cats underwent MAT, while 268 urine samples from community cats were tested for Leptospira DNA. Paired urine qPCR and serum MAT results were available for 233 community cats. Serum creatinine and ALT results were available for 437 and 297 cats, respectively.
Fig. 1.
Flow chart describing sample recruitment and refinement of the study population, which comprised of community cats and privately-owned cats. Samples excluded due to “inhibition on qPCR” contained PCR inhibitors that prevented DNA amplification.
Community cats were all domestic shorthairs (DSH), were 0.5 to 9 (median 2) years-old and comprised of 48% males and 52% females, with 97.4% sexually-intact and 2.6% neutered. Privately-owned cats were aged 0.5 to 21 years (median 10.6), with 62.2% males and 89.8% cats were neutered. Of 27 breeds represented, the most common were DSH (40.9%), British shorthair (19.9%) and Exotic shorthair (10.1%).
3.2. Seroprevalence
Overall Leptospira seroprevalence was 9.3% (69/738) with MAT titres of 1:100–1:6400 (median 1:200). Community cats were more likely to be seroreactive (13.3%, 52/391) than privately-owned cats (4.9%, 17/347) (OR = 2.98, P < 0.001). Seroreactivity was detected against 15 serovars from 14 serogroups (Table 2), most commonly serogroups/serovars Javanica (4.3%, 32/738), Djasiman (2.3%, 17/738) and Australis (1.5%, 11/738).
Table 2.
Frequency distribution of antibody titres against each serovar of Leptospira spp. by microscopic agglutination test (MAT) in sera from cats of Hong Kong.
| Serovar⁎ | Antibody Titre |
Total | ||||||
|---|---|---|---|---|---|---|---|---|
| 1:100 | 1:200 | 1:400 | 1:800 | 1:1600 | 1:3200 | 1:6400 | ||
| Javanica | 4 | 5 | 7 | 6 | 4 | 3 | 3 | 32 |
| Djasiman | 14 | 3 | 0 | 0 | 0 | 0 | 0 | 17 |
| Australis | 6 | 4 | 0 | 0 | 0 | 0 | 0 | 10 |
| Bataviae | 2 | 2 | 1 | 1 | 1 | 0 | 0 | 7 |
| Pohnpei | 3 | 0 | 0 | 1 | 0 | 0 | 0 | 4 |
| Hebdomadis | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 3 |
| Mini | 0 | 1 | 0 | 0 | 2 | 0 | 0 | 3 |
| Celledoni | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| Sarmin | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 2 |
| Autumnalis | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Canicola | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Pomona | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Ballum | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Copenhageni | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Pyrogenes | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Total | 36 | 18 | 10 | 8 | 7 | 4 | 4 | 87⁎⁎ |
Serovars Cynopteri, Grippotyphosa, Hardjo, Panama, Saxkoebing, Shermani and Tarassovi were tested but not included in the table as no antibodies were detected against them.
As antibodies against multiple Leptospira serovars were detected in some cats, the total number of antibody titres reported (n = 87) is greater than the number of seropositive cats detected (n = 69).
The most frequently detected serogroup/serovar in community cats was Javanica (7.65%) and in privately-owned cats was Djasiman (2.59%) (Fig. 2). Exposure to Javanica was significantly higher in community cats (7.65%, 30/392) than privately-owned (0.57%, 2/347) (P < 0.001). Half of the cats seroreactive for serogroup Javanica (16/32) had titres ≥1:800.
Fig. 2.
Seroprevalence of the three most common serogroups of Leptospira spp. (Javanica, Djasiman and Australis) detected from 391 community and 347 privately-owned cats in Hong Kong. ‘*’ indicates a statistically significant difference in the seroprevalence of serogroup Javanica between the two sources of cats (P < 0.001).
Antibodies to 2–5 serogroups were detected in 11/69 (15.9%) seropositive cats (Supplementary Materials 1). Three cats had antibodies against both serovars of serogroup Australis (Australis and Pohnpei), while three others were reactive to serogroups Mini and Hebdomadis. Serogroup Javanica had the highest antibody titre in 5/11 (45.5%) cats reactive to multiple serogroups.
3.3. Urinary shedding of Leptospira
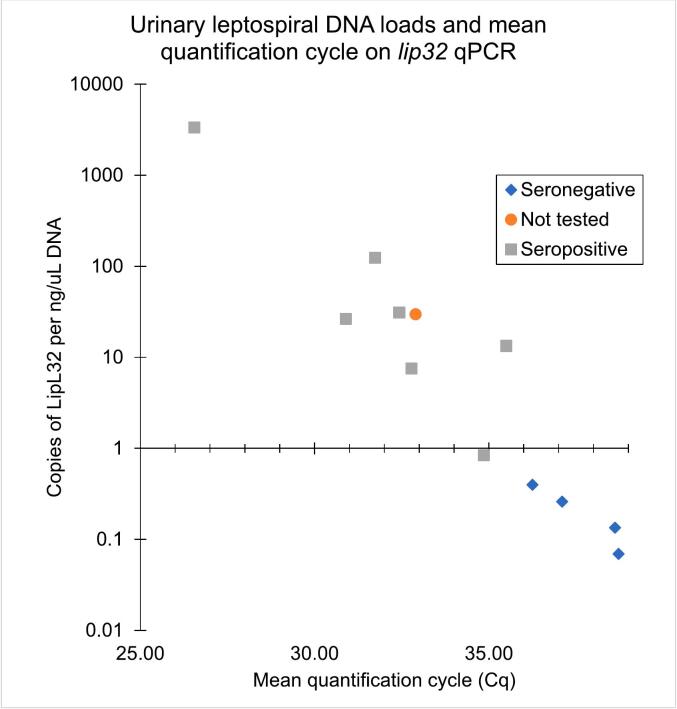
Leptospira DNA was detected in urine of 12/268 (4.48%) community cats (Table 3), with a median of 6.41 × 102 copies/mL of urine and median cycle quantification (Ct) value of 33.9. All cats with >102 copies of lipL32/mL of urine were seroreactive (Fig. 3).
Table 3.
Signalment and test results for 12 community cats from Hong Kong with Leptospira DNA detected in their urine by lipL32 qPCR.
| Study ID | Sex | Neuter status | Age (months) | qPCR Ct value | qPCR copies/mL urine⁎ | Reciprocal MAT titre | MAT positive serovar |
|---|---|---|---|---|---|---|---|
| E | Male | Entire | 24 | 26.55 | 9.63 × 104 | 3200 1600 |
Hebdomadis, Mini |
| N | Female | Entire | 24 | 31.74 | 4.48 × 103 | 100 | Bataviae |
| O | Female | Neutered | 72 | 32.43 | 2.38 × 103 | 400 | Javanica |
| P | Male | Entire | 36 | 32.90 | 2.09 × 103 | Not tested | Not tested |
| Q | Male | Entire | 36 | 30.90 | 9.86 × 102 | 400 | Javanica |
| R | Male | Entire | 48 | 35.50 | 1.52 × 103 | 200 | Bataviae |
| S | Female | Entire | 12 | 32.78 | 2.81 × 102 | 200 | Bataviae |
| T | Male | Entire | 36 | 34.86 | 2.98 × 102 | 200 | Javanica |
| U | Male | Entire | 60 | 36.25 | 7.84 × 101 | Negative | Negative |
| V | Male | Entire | 6 | 37.10 | 3.78 × 101 | Negative | Negative |
| W | Female | Entire | 24 | 38.62 | 1.40 × 101 | Negative | Negative |
| X | Male | Entire | 36 | 38.72 | 1.45 × 101 | Negative | Negative |
Copies per reaction were converted to copies per mL urine considering volumes of urine used for DNA extraction, eluate and DNA template per reaction.
Fig. 3.
Scatter plot of leptospiral urinary DNA loads in copies of leptospiral gene lipL32 per ng/ μL DNA, and mean quantification cycle (Ct) on quantitative PCR for cats shedding Leptospira in urine (n = 12), with different markers representing serological status.
Of the community cats with paired sera and urine, 10.7% (25/233) were positive by at least one test. Seven (28.0%) were seroreactive and shedding lipL32, 14 (56%) were seroreactive but not shedding, while four (16%) were seronegative and had leptospiruria (16%, 4/25). A third of seroreactive cats (7/21) were shedding Leptospira DNA in urine, with seroreactivity to serogroups Javanica, Bataviae Hebdomadis and/or Mini (Table 3).
3.4. Rodent infestation rate
Annual district rodent infestation rates ranged from 1.6% (Islands) to 5.2% (Yau Tsim Mong) (median 2.9%). No association was observed between rodent infestation rate and Leptospira seroprevalence in community cats (Supplementary Materials 2 and 3).
3.5. Univariable associations
Leptospira seroprevalence in privately-owned cats with elevated ALT values was 4.4% (1/23) compared to 5.8% (15/258) in cats with non-elevated levels. Results from univariable logistic regressions indicated significant association between Leptospira seropositivity and the source, breed, age, and neuter status of the study cats (Table 4). However, there were strong, significant (P < 0.001) correlations between “source” and the other three independent variables. Therefore, as the most important factor which theoretically preceded and dictated the distribution of the other three variables, “source” was chosen for multivariable logistic regression modelling, along with sex (P < 0.2). In multivariable modelling, sex was not significant (P = 0.363) and was removed from the final model. As a result, the final model became equivalent to the simple logistic model presented for source in Table 4. The odds of Leptospira seroreactivity in community cats was 2.98 [95% CI: 1.68–5.25] times higher than that in privately-owned cats in this study (P < 0.001).
Table 4.
Results of logistic regressions assessing univariable associations between seropositivity to Leptospira spp. and independent variables of interest for the 738 studied cats.
| Variable | Categories | No. of seronegative cats | No. of seropositive cats | ORb | 95% CIc | P-value |
|---|---|---|---|---|---|---|
| Source | Privately-owned | 330 | 17 | – | – | – |
| Community | 339 | 52 | 2.98 | 1.68–5.25 | < 0.001d | |
| Breed | Purebred | 194 | 11 | – | – | – |
| Domestic Shorthair (DSH) | 475 | 58 | 2.15 | 1.11–4.19 | 0.024 | |
| Sex | Male | 371 | 32 | – | – | – |
| Female | 298 | 37 | 1.44 | 0.87–2.37 | 0.151 | |
| Neuter status | Neutered | 302 | 14 | – | – | – |
| Entire | 367 | 55 | 3.06 | 1.69–5.53 | < 0.001 | |
| Age⁎ (months) | < 12 | 59 | 5 | – | – | – |
| 12–83 | 360 | 51 | 1.67 | 0.64–4.36 | 0.294 | |
| > 83 | 250 | 13 | 0.61 | 0.21–1.79 | 0.371 | |
| Creatinine (μmol/L) | Normal | 312 | 23 | – | – | – |
| Elevated | 98 | 4 | 0.55 | 0.19–1.64 | 0.286 |
Overall P-value for age = 0.006; follow-up pairwise comparisons (with Bonferroni adjustment) indicated a significant difference between age categories 12–83 months and > 83 months (P = 0.005).
OR Odds Ratio
CI Confidence Interval
Bolded values are statistically signficant
4. Discussion
The overall Leptospira seroprevalence of 9.35% in cats in our study is comparable with global estimates (11.7%) [27] and similar to nearby Taiwan (9.3%) [18]. Leptospira seroprevalence of community (13.3%) and privately-owned cats (4.3%) in Hong Kong was also similar to that in stray (10.7%) and privately-owned cats (5.7%) in Taiwan [18]. We found outdoor community cats were almost three times as likely to be seropositive than privately-owned cats, similar to the findings of a global meta-analysis in which outdoor cats had 2.74-times higher odds of seroreactivity than indoor cats [27]. Whilst we did not collect data on the domicile of privately-owned cats, 90.5% of Hong Kong people live in apartments, thus most privately-owned cats likely have no outdoor access and limited rodent exposure [28]. The few seroreactive privately-owned cats (4.90%) could have been adopted community cats, had outdoor access, or been exposed indoors.
In accordance with findings from meta-analyses that rodent contact is associated with human but not feline Leptospira exposure, we found no relationship between rodent infestation rates and feline seropositivity [27,29]. Although low numbers of cats in some districts and the indirect method used to estimate rodent infestation rates may have hampered our ability to detect an association, environmental exposure to Leptospira is the most plausible explanation for the substantially higher seroreactivity in community cats. Waterlogged soil is an ideal environment for pathogenic Leptospira to replicate, yet is often overlooked as a reservoir of infection [13,30].
Direct comparisons of feline Leptospira seroprevalence between studies is difficult, since MAT titres as low as 1:20 have been considered seropositive [21,31]. Reducing the MAT cut-off from 1:100 to 1:50 in our study would have increased the overall seroprevalence from 9.35% to 17.3%, including 20.7% of community cats and 13.5% of privately-owned cats, largely driven by serogroup Djasiman, which was responsible for 54.2% (52/96) of the 1:50 titres. However, this lower cut-off would have also reduced the odds ratio of infection in community cats from 2.98 to 1.66 (95% CI: 1.10–2.52), suggesting reduced specificity.
In our study there was high Leptospira serovar diversity with 15/22 serovars (68.2%) detected compared to 3/20 (15%), 6/25 (24%) and 7/24 (29.2%) in cats from Malaysia, Vietnam and Thailand, respectively [17,20,21]. However, these results are not directly comparable since serovar panels were not identical.
We detected serogroup Canicola in one cat (1:400) for the first time in a felid in East and Southeast Asia. We also detected serovar Pohnpei, originally isolated from rodents in Micronesia but not tested for before in Asian cats [32]. However, some cats seroreactive for Pohnpei also reacted to serovar Australis in the same serogroup, indicating possible cross-reactivity. Cats seroreactive to serogroup Mini also reacted to the heterologous serogroup Hebdomadis, which can result from antigenic similarities in rfb locus genes [33].
Overall, cats in Hong Kong were most commonly exposed to serogroup Javanica (4.33%), which was also the most common serogroup detected among cats in Okinawa island, Japan and in Vietnam [19,20]. By contrast, in Taiwan, which is geographically closer to Hong Kong, Shermani was the most common serogroup (4.2%) detected in cats [18]. It was also the most common cause of human leptospirosis cases there and was detected in cattle, swine and stray dogs [18,34]. However, serogroup Shermani was not detected in any cats in our study. These differences in serogroup frequency among cats in regions neighbouring Hong Kong could be due to multiple differences in Leptospira transmission drivers, such as reservoir species diversity, land use and population density despite relative geographic proximity. Geographically weighted logistic regression models and Bayesian, hierarchical mixed-model frameworks incorporating transmission drivers such as climatic data (e.g. precipitation, temperature), distance to rivers, livestock density (e.g. pigs, cattle), and residential setting (e.g. urban, periurban, rural) and poverty rates, have been used to inform a precision-medicine targeted approach to leptospirosis preparedness in countries such as Fiji [35,36]. Such models would be further strengthened by incorporation of direct measures of Leptospira carriage and shedding in animals, using an enhanced One-Health approach.
Community cats were significantly more likely to be exposed to serogroup Javanica (7.65%) than privately-owned cats (0.58%) in our study. In addition, three of the seven seroreactive cats shedding Leptospira were reactive to serogroup Javanica. The combination of high frequency of exposure at titres to serogroup Javanica and urinary shedding among cats seroreactive to Javanica suggests active circulation of this serogroup among outdoor free-roaming cats in Hong Kong.
In Okinawa island, cats and black rats (Rattus rattus) were infected with the same multilocus sequence type of serovar Javanica, suggesting an infection cycle between rats and cats [19]. Serovar Javanica commonly infects many rodent species including R. rattus, R. exulans, R. lose, R. norvegicus and the greater bandicoot rat (Bandicota indica) in Japan, Taiwan and the Philippines [37]. Although feline Leptospira serosurveillance data in Mainland China have not yet been reported, the two most common serovars identified in rats in China are Icterohaemorrhagiae and Javanica. Leptospira serovars circulating in rodents in Hong Kong have not been investigated. Beyond cats and rodents, Leptospira from serogroup Javanica have been detected in bovine urine, canine blood and soil in India, Malaysia and Japan [[38], [39], [40], [41]] and can cause severe disease in humans [42,43], demonstrating the regional specificity, diversity of infection sources and One Health importance of Leptospira.
The prevalence of leptospiruria in our study (4.48%) was similar to the global point prevalence in cats (3.7%) [27]. While presence of DNA does not indicate organism viability, Leptospira have previously been cultured from feline urine [44].
Neither our study nor Shropshire et al. found a significant relationship between elevated creatinine and Leptospira seroreactivity in cats [45]. Two previous reports investigating association between seroreactivity and chronic kidney disease (CKD) in cats had contradictory results [15,46]. Future studies could use other CKD biomarkers such as serum symmetric diemethylarginine to increase sensitivity of detection [47,48]. While periods of high rainfall are associated with Leptospira outbreaks and environmental contamination [[49], [50], [51]], the effect of seasonality in our study was not evaluated since samples from community and privately-owned cats were not collected year-round.
5. Conclusions
Cats in Hong Kong are exposed to a diverse array of Leptospira serogroups, especially Javanica. The almost three-times higher risk of exposure in free-roaming outdoor community cats, which was not associated with rodent infestation rates, highlights the importance of environmental sources of Leptospira as transmission drivers of leptospirosis. Contact with urine of subclinically infected cats shedding Leptospira is an unrecognised occupational risk for veterinarians and,gloves should be worn when handling feline urine to minimise zoonotic risk.
Funding
This work was supported by internal grants from City University of Hong Kong of one of the authors (VRB), namely a DON_RMG grant (Project No. 9229086) and an SGP grant (Project No. 9380113).
CRediT authorship contribution statement
Wing Yan Jacqueline Tam: Data curation, Formal analysis, Project administration, Software, Writing – original draft. Omid Nekouei: Data curation, Formal analysis, Methodology, Software, Writing – review & editing. Francesca Rizzo: Investigation, Methodology, Validation, Writing – review & editing. Lok See Tiffany Cheng: Formal analysis, Methodology, Writing – review & editing. Yan Ru Choi: Methodology, Project administration, Writing – review & editing. Megan Staples: Formal analysis, Methodology, Writing – review & editing. Stefan Hobi: Investigation, Methodology, Writing – review & editing. Jane Gray: Investigation, Methodology, Resources, Writing – review & editing. Fiona Woodhouse: Investigation, Methodology, Writing – review & editing. Patricia Yi Man Shuen: Investigation, Methodology, Writing – review & editing. Ying Fei Chai: Investigation, Methodology, Writing – review & editing. Julia A. Beatty: Investigation, Methodology, Resources, Supervision, Writing – review & editing. Vanessa R. Barrs: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that there is no conflict of interest regarding the publication of this article. Internal funding from City University of Hong Kong had no influence on the study design, results, interpretation or publication of findings. Generative AI tools were not used in the writing of our manuscript.
Acknowledgments
The authors thank all staff at the SPCA Wan Chai Hong Kong that assisted with sample collection for this project.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.onehlt.2024.100851.
Appendix A. Supplementary data
Table describing the characteristics of cats reactive to more than one Leptospira serovar on MAT.
Supplementary material 2: Scatter plot of rodent infestation rate against seropositivity of community cats caught from each of the 18 administrative districts.
Supplementary material 3: Scatter plot of rodent infestation rate against percentage of community cats exposed to serovar Javanica from each of the 18 administrative districts.
Supplementary material 4: Table of district-level rodent infestation rates and absolute and relative frequency of cats seropositive to all serovars or serovar Javanica specifically per district by MAT.
Data availability
All data supporting the conclusions of the study are available in the text, as supplementary material or through accessing the full dataset deposited in the OSF repository here: https://osf.io/r4n6t
References
- 1.Cilia G., et al. Insight into the epidemiology of leptospirosis: a review of Leptospira isolations from “unconventional” hosts. Animals (Basel) 2021;11(1) doi: 10.3390/ani11010191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Costa F., et al. Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl. Trop. Dis. 2015;9(9) doi: 10.1371/journal.pntd.0003898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baharom M., et al. Environmental and occupational factors associated with leptospirosis: a systematic review. Heliyon. 2024;10(1) doi: 10.1016/j.heliyon.2023.e23473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hartmann K., et al. Leptospira species infection in cats: ABCD guidelines on prevention and management. J. Feline Med. Surg. 2013;15(7):576–581. doi: 10.1177/1098612X13489217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vincent A.T., et al. Revisiting the taxonomy and evolution of pathogenicity of the genus Leptospira through the prism of genomics. PLoS Negl. Trop. Dis. 2019;13(5) doi: 10.1371/journal.pntd.0007270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sykes J.E., et al. A global one health perspective on leptospirosis in humans and animals. J. Am. Vet. Med. Assoc. 2022;260(13):1589–1596. doi: 10.2460/javma.22.06.0258. [DOI] [PubMed] [Google Scholar]
- 7.Levett P.N. Leptospirosis. Clin. Microbiol. Rev. 2001;14(2):296–326. doi: 10.1128/CMR.14.2.296-326.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Markey B.K., et al. Second edition. ed. Mosby Elsevier; Edinburgh: 2013. Clinical Veterinary Microbiology. [Google Scholar]
- 9.Dupouey J., et al. Human leptospirosis: an emerging risk in Europe? Comp. Immunol. Microbiol. Infect. Dis. 2014;37(2):77–83. doi: 10.1016/j.cimid.2013.12.002. [DOI] [PubMed] [Google Scholar]
- 10.Protection, C.f.H . In: Number of Notifiable Infectious Diseases by Month in 2022. D.o. Health, editor. Government of the HKSAR; Hong Kong: 2022. [Google Scholar]
- 11.Flay K.J., et al. Absence of serological or molecular evidence of Leptospira infection in farmed swine in the Hong Kong special administrative region. One Health. 2021;13 doi: 10.1016/j.onehlt.2021.100321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sumalapao D., et al. Frequency of typhoon occurrence accounts for the Poisson distribution of human leptospirosis cases across the different geographic regions in the Philippines. Asian Pac J Trop Med. 2019;12(1):38–42. [Google Scholar]
- 13.Yanagihara Y., et al. Leptospira is an environmental bacterium that grows in waterlogged soil. Microbiol. Spectr. 2022;10(2) doi: 10.1128/spectrum.02157-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li J.M., et al. Prevalence of Leptospira in murine in China: a systematic review and meta-analysis. Front. Vet. Sci. 2022:9. doi: 10.3389/fvets.2022.944282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Weis S., et al. Detection of Leptospira DNA in urine and presence of specific antibodies in outdoor cats in Germany. J. Feline Med. Surg. 2017;19(4):470–476. doi: 10.1177/1098612X16634389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Food and Environmental Hygiene Department Rodent Pests and Their Control. 2023. https://www.fehd.gov.hk/english/pestcontrol/risk-pest-rodents.html Rodent. [cited 2023 14 April]; Available from:
- 17.Alashraf A.R., et al. Serological detection of anti-Leptospira antibodies in shelter cats in Malaysia. Top. Companion Anim. Med. 2019;34:10–13. doi: 10.1053/j.tcam.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 18.Chan K.W., et al. Serological and PCR detection of feline Leptospira in southern Taiwan. Vector Borne Zoonotic Dis. 2014;14(2):118–123. doi: 10.1089/vbz.2013.1324. [DOI] [PubMed] [Google Scholar]
- 19.Kakita T., et al. Molecular and serological epidemiology of Leptospira infection in cats in Okinawa Island, Japan. Sci. Rep. 2021;11(1):10365. doi: 10.1038/s41598-021-89872-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mai L.T.P., et al. Leptospira infection among human-close-contact animals in different geographical areas in Vietnam. Sci. Prog. 2021;104(3) doi: 10.1177/00368504211031747. p. 368504211031747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sprissler F., et al. Leptospira infection and shedding in cats in Thailand. Transbound. Emerg. Dis. 2019;66(2):948–956. doi: 10.1111/tbed.13110. [DOI] [PubMed] [Google Scholar]
- 22.Health, W.O.f.A . twelfth edition 2023. World Organization for Animal Health; 2023. Leptospirosis, in Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. [Google Scholar]
- 23.Beatty J.A., et al. Felis catus gammaherpesvirus 1; a widely endemic potential pathogen of domestic cats. Virology. 2014;460-461:100–107. doi: 10.1016/j.virol.2014.05.007. [DOI] [PubMed] [Google Scholar]
- 24.Blanchard S., et al. Quantitative real-time PCR assays for the detection of pathogenic Leptospira species in urine and blood samples in canine vaccine clinical studies: a rapid alternative to classical culture methods. J. Clin. Microbiol. 2021;59(7) doi: 10.1128/JCM.03006-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stoddard R.A., et al. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn. Microbiol. Infect. Dis. 2009;64(3):247–255. doi: 10.1016/j.diagmicrobio.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 26.Dohoo I.R., Martin W., Stryhn H. 2 ed. VER Inc.; Charlottetown: 2009. Veterinary Epidemiologic Research. [Google Scholar]
- 27.Ricardo T., et al. Leptospiral infection in domestic cats: systematic review with meta-analysis. Prev. Vet. Med. 2023;212 doi: 10.1016/j.prevetmed.2023.105851. [DOI] [PubMed] [Google Scholar]
- 28.Census and Statistics Department . 2021 Population Census. Hong Kong Special Administrative Region; Hong Kong: 2021. 2021 population census main results. [Google Scholar]
- 29.Mwachui M.A., et al. Environmental and behavioural determinants of leptospirosis transmission: a systematic review. PLoS Negl. Trop. Dis. 2015;9(9) doi: 10.1371/journal.pntd.0003843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Casanovas-Massana A., et al. Quantification of Leptospira interrogans survival in soil and water microcosms. Appl. Environ. Microbiol. 2018;84(13) doi: 10.1128/AEM.00507-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Murillo A., et al. Leptospira detection in cats in Spain by serology and molecular techniques. Int. J. Environ. Res. Public Health. 2020;17(5) doi: 10.3390/ijerph17051600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Simms J. Animal leptospirosis in the federated state of Micronesia. Pac. Health Dialog. 1998;1:30–37. [Google Scholar]
- 33.Medeiros E.J.S., et al. Genetic basis underlying the serological affinity of leptospiral serovars from serogroups Sejroe, Mini and Hebdomadis. Infect. Genet. Evol. 2022;103 doi: 10.1016/j.meegid.2022.105345. [DOI] [PubMed] [Google Scholar]
- 34.Chou L.F., et al. Potential impact on kidney infection: a whole-genome analysis of Leptospira santarosai serovar Shermani. Emerg. Microbes Infect. 2014;3(11) doi: 10.1038/emi.2014.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mayfield H.J., et al. Use of geographically weighted logistic regression to quantify spatial variation in the environmental and sociodemographic drivers of leptospirosis in Fiji: a modelling study. Lancet Planet Health. 2018;2(5):e223–e232. doi: 10.1016/S2542-5196(18)30066-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rees E.M., et al. Quantifying the relationship between climatic indicators and leptospirosis incidence in Fiji: a modelling study. PLOS Glob. Public Health. 2023;3(10) doi: 10.1371/journal.pgph.0002400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Koizumi N., et al. Comparative genomic analysis of Leptospira borgpetersenii serogroup Javanica isolated from Rattus species in Southern Japan, Philippines, and Taiwan. Infect. Genet. Evol. 2020;85 doi: 10.1016/j.meegid.2020.104447. [DOI] [PubMed] [Google Scholar]
- 38.Rahman S.A., et al. Detection and characterization of Leptospira spp. in dogs diagnosed with kidney and/or liver disease in Selangor, Malaysia. J. Vet. Diagn. Investig. 2021;33(5):834–843. doi: 10.1177/10406387211024575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Natarajaseenivasan K., et al. Seroprevalence of Leptospira borgpetersenii serovar Javanica infection among dairy cattle, rats and humans in the cauvery river valley of southern India. Southeast Asian J. Trop. Med. Public Health. 2011;42(3) [PubMed] [Google Scholar]
- 40.Saito M., et al. Comparative analysis of Leptospira strains isolated from environmental soil and water in the Philippines and Japan. Appl. Environ. Microbiol. 2013;79(2):601–609. doi: 10.1128/AEM.02728-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alexander A.D., et al. Pathogenic Leptospiras isolated from Malaysian surface waters. Appl. Microbiol. 1975;29(1):30–33. doi: 10.1128/am.29.1.30-33.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Valverde Mde L., et al. Arenal, a new Leptospira serovar of serogroup Javanica, isolated from a patient in Costa Rica. Infect. Genet. Evol. 2008;8(5):529–533. doi: 10.1016/j.meegid.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 43.Shafei M.N., et al. Seroprevalence of leptospirosis among town service workers in Northeastern State of Malaysia. Int. J. Collab. Res. Intern. Med. Public Health. 2012;4(4):395–403. [Google Scholar]
- 44.Dorsch R., et al. Cats shedding pathogenic Leptospira spp.—an underestimated zoonotic risk? PLoS One. 2020;15(10) doi: 10.1371/journal.pone.0239991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shropshire S.B., et al. Evaluation of the Leptospira species microscopic agglutination test in experimentally vaccinated cats and Leptospira species seropositivity in aged azotemic client-owned cats. J. Feline Med. Surg. 2016;18(10):768–772. doi: 10.1177/1098612X15593902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rodriguez J., et al. Serologic and urinary PCR survey of leptospirosis in healthy cats and in cats with kidney disease. J. Vet. Intern. Med. 2014;28(2):284–293. doi: 10.1111/jvim.12287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.International Renal Interest Society. IRIS Staging of CKD; 2023. [Google Scholar]
- 48.Lefebvre H.P. Nephrology and Urology of Small Animals. 2014. Renal function testing; pp. 91–96. [Google Scholar]
- 49.Kupek E., de Sousa Santos Faversani M.C., de Souza Philippi J.M. The relationship between rainfall and human leptospirosis in Florianopolis, Brazil, 1991-1996. Braz. J. Infect. Dis. 2000;4(3):131–134. [PubMed] [Google Scholar]
- 50.Matsushita N., et al. The non-linear and lagged short-term relationship between rainfall and leptospirosis and the intermediate role of floods in the Philippines. PLoS Negl. Trop. Dis. 2018;12(4) doi: 10.1371/journal.pntd.0006331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pappachan M.J., Sheela M., Aravindan K.P. Relation of rainfall pattern and epidemic leptospirosis in the Indian state of Kerala. J. Epidemiol. Community Health. 2004;58(12):1054. doi: 10.1136/jech.2003.018556. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table describing the characteristics of cats reactive to more than one Leptospira serovar on MAT.
Supplementary material 2: Scatter plot of rodent infestation rate against seropositivity of community cats caught from each of the 18 administrative districts.
Supplementary material 3: Scatter plot of rodent infestation rate against percentage of community cats exposed to serovar Javanica from each of the 18 administrative districts.
Supplementary material 4: Table of district-level rodent infestation rates and absolute and relative frequency of cats seropositive to all serovars or serovar Javanica specifically per district by MAT.
Data Availability Statement
All data supporting the conclusions of the study are available in the text, as supplementary material or through accessing the full dataset deposited in the OSF repository here: https://osf.io/r4n6t