Abstract
Background
Eosinophilic esophagitis is a chronic inflammatory disorder of the esophagus. This real-world study used patient and physician surveys to describe the clinical characteristics and disease burden of eosinophilic esophagitis—overall and in a subgroup of patients with dysphagia despite treatment.
Methods
Data analyzed in this study were collected in 2020 from US and EU patients with eosinophilic esophagitis. Eligible patients were aged ≥ 12 years with a diagnosis of eosinophilic esophagitis, had an esophageal count of ≥ 15 eosinophils/high-power field at diagnosis, and were currently prescribed treatment for eosinophilic esophagitis.
Results
Overall, 1001 patients were included, of whom 356 (36%) had dysphagia despite treatment. Demographics and clinical characteristics were similar in both populations. The severity of eosinophilic esophagitis was mild in more patients overall (69%) versus those with dysphagia despite treatment (48%). Patient disease history was similar in both populations, with some exceptions: common patient-reported symptoms were dysphagia (70% and 86%) and heartburn/acid reflux (55% and 49%), and common physician-reported symptoms were dysphagia (75% and 91%) and food impaction (46% and 52%). Treatment history was similar in both populations; overall, the most common treatments were proton pump inhibitors (83%) and topical corticosteroids (51%). Patients reported slightly more days with symptoms, higher impacts on activities of daily living, and slightly higher anxiety or depression in the dysphagia-despite-treatment population versus the overall population.
Conclusions
Eosinophilic esophagitis presents severe symptoms and comorbidities that substantially impact patients’ well-being and quality of life. Greater awareness of and novel treatments for eosinophilic esophagitis are needed.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12876-024-03334-4.
Keywords: Eosinophilic esophagitis, Inflammatory disease, Real-world evidence, Health-related quality of life, Disease burden
Introduction
Eosinophilic esophagitis (EoE) is a chronic, immune-mediated, inflammatory disease of the esophagus [1, 2]. Symptoms of EoE include dysphagia, food impaction, abdominal pain, nausea, and vomiting [1, 2]. Although it does not appear to limit the life expectancy of patients [3], clinical observations indicate that EoE is associated with substantial patient burden and impacts to their health-related quality of life (HRQOL) [4, 5].
The diagnosis of EoE is often performed histologically, with the presence of eosinophils on biopsy, indicating inflammation of esophageal tissue [6]. The current standard of care for EoE often relies on elemental and/or elimination diets and esophageal dilations, in addition to medications, such as proton pump inhibitors and swallowed topical steroids (i.e., budesonide, currently approved in the European Union [EU] [7] and Canada [8] only), to manage the disease [9, 10]. However, patient HRQOL is especially impacted among those with more severe symptoms, histologically active disease, or extensive dietary restrictions [11, 12]; indeed, many patients continue to experience symptoms related to EoE despite current standard-of-care treatments [5]. A few biologic therapies have recently been investigated for EoE, though dupilumab is the only agent in this class currently approved to treat EoE [13]. Although several studies have collected survey data on the patient impact of EoE [3, 5, 11, 12, 14–17], patient-reported evidence is still limited regarding EoE symptoms, EoE disease burden, and EoE impact on patient well-being and activities of daily living. Moreover, social media studies in EoE suggest an unmet patient need for accurate, reliable sources of information about the disease, barriers to treatment, and shared decision-making [18, 19].
The objectives of this real-world study using patient-reported survey responses were to describe the demographics, clinical characteristics, and disease burden of patients with EoE and to locate and highlight any data gaps in patient versus physician perspectives to gain a better understanding of the proper management of patients with EoE.
Methods
Study design
This study used real-world data from the Adelphi Real World Disease Specific Programmes™, which are impartial, multinational, cross-sectional physician and patient surveys that provide data related to real-world clinical practices for a range of chronic health conditions, including EoE [20]. The data analyzed in this study were collected from patients consulting for routine care in the latter half of 2020 from across the United States (US) and 5 European countries (EU5): France, Germany, Italy, Spain, and the United Kingdom (UK). In addition to a physician screener and physician survey, physicians were asked to complete patient record forms on patient characteristics and treatment; these same patients were invited to fill out a voluntary patient self-completion questionnaire. Data reported in this study were collected at the time of the survey (e.g. demographic information) and previous recorded medical history (e.g. duration of treatment); although some data are related to specific time points (e.g. previous month, previous 12 months), there were no follow-up data collected in this study (Supplementary Fig. 1).
Eligible patients for this study were aged ≥ 12 years with a biopsy-confirmed physician diagnosis of EoE, an esophageal count of ≥ 15 eosinophils/high-power field (eos/hpf), and a current prescribed treatment for EoE. Patients were eligible for inclusion in this study regardless of the amount of time for which they had physician-diagnosed EoE.
Assessments and statistical analyses
Assessments in this study included those for patient demographics, disease history, clinical characteristics, treatment pathways, patient symptom burden, and HRQOL measures (EuroQol 5-dimension 3-level [EQ-5D-3 L] assessment). Data for this study are summarized descriptively. Continuous variables are summarized by means, standard deviation, and medians (interquartile range and range). Categorical variables are reported as counts and percentages.
Study conduct and ethics
The study was conducted according to standard operating procedures. Research was conducted as a survey in accordance with the amended Declaration of Helsinki, adhering to the ICC/ESOMAR International Code on Market, Opinion and Social Research and Data Analytics, the international code on observational research, and HIPAA (Health Insurance Portability and Accountability Act of 1996) guidelines. The survey was submitted to a central international review board (Western Institutional Review Board) and found to be exempt from ethics requirements on 23 July 2020 (Study Number: 1-1328144-1).
Human ethics and consent to participate declarations
Not applicable.
Results
Patient demographics and clinical characteristics
This study focused on an overall population of patients with EoE, as well as a subgroup of those patients who had dysphagia despite treatment, as indicated by the diagnosing physician. Demographics and clinical characteristics were physician-reported, and HRQOL measures were patient-reported; the sources of all other data are indicated as follows with the associated data.
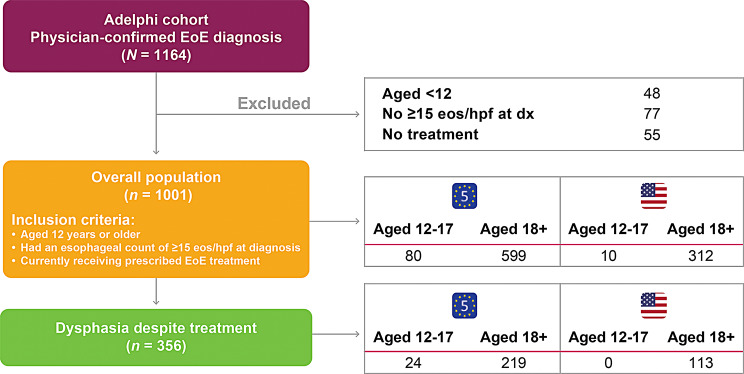
Overall, there were 1001 patients included in this study, of which 356 (36%) had dysphagia despite treatment (Fig. 1). Demographics were generally similar between the overall and dysphagia-despite-treatment populations (Table 1). In the overall population, the mean (SD) age was 36.4 (14.58) years. Most patients were male (66%) and working full-time (58%), without caregiver help (90%). Clinical characteristics were also similar between the two patient populations (Table 2). The most common comorbidities among patients in the overall population were allergic rhinitis (n = 242, 24%) and asthma (n = 238, 24%). There were some differences in clinical characteristics in the overall and dysphagia-despite-treatment populations: 58% (n = 84/145) and 65% (n = 33/51) of patients, respectively, had ≥ 15 eos/hpf at the most recent endoscopy with biopsy and the current severity of EoE was mild in more patients in the overall group (n = 693, 69% vs. n = 171, 48%).
Fig. 1.
Patient population flowchart. EoE, eosinophilic esophagitis; eos/hpf, eosinophils per high-power field
Table 1.
Baseline demographics
| Characteristic | Overall (N = 1001) |
Dysphagia despite treatment (N = 356) |
|---|---|---|
| Age at screening (years), mean (SD) | 36.4 (14.58) | 36.8 (14.49) |
| Male sex, n (%) | 657 (66) | 236 (66) |
| BMI (kg/m2), mean (SD) | 24.3 (3.63) | 24.5 (3.56) |
| Employment status, n (%) | (N = 973) | (N = 341) |
| Working full-time | 563 (58) | 193 (57) |
| Working part-time | 78 (8) | 29 (9) |
| On long-term sick leave | 7 (1) | 3 (1) |
| Homemaker | 48 (5) | 15 (4) |
| Student | 207 (21) | 67 (20) |
| Retired | 43 (4) | 16 (5) |
| Unemployed | 27 (3) | 18 (5) |
| On long-term leave/homemaker/retired/unemployed owing to EoE, n (%) | (N = 121) | (N = 50) |
| Yes | 5 (4) | 3 (6) |
| No | 116 (96) | 47 (94) |
| Caregiver status, n (%) | ||
| Yes | 91 (10) | 28 (8) |
| No | 858 (90) | 309 (92) |
BMI, body mass index
Table 2.
Clinical characteristics
| Characteristic | Overall (N = 1001) |
Dysphagia despite treatment (N = 356) |
|---|---|---|
| Comorbidities in ≥ 5% of patients, n (%) | ||
| Asthma | 238 (24) | 83 (23) |
| Mild | 133 (56) | 49 (59) |
| Moderate | 93 (39) | 31 (37) |
| Severe | 12 (5) | 3 (4) |
| Allergic rhinitis | 242 (24) | 75 (21) |
| Anxiety | 141 (14) | 68 (19) |
| Atopic dermatitis | 78 (8) | 29 (8) |
| Depression | 57 (6) | 29 (8) |
| None of the above | 376 (38) | 123 (35) |
| Oral allergy syndrome | 62 (6) | 20 (6) |
| Peanut allergy | 61 (6) | 29 (8) |
| Peak eos/hpf at most recent endoscopy with biopsy, n (%) | (N = 145) | (N = 51) |
| < 6 | 23 (16) | 4 (8) |
| 6–15 | 38 (26) | 14 (27) |
| ≥ 15 | 84 (58) | 33 (65) |
| Current EoE severity, n (%) | ||
| Mild | 693 (69) | 171 (48) |
| Moderate | 269 (27) | 156 (44) |
| Severe | 39 (4) | 29 (8) |
EoE, eosinophilic esophagitis; eos/hpf, eosinophils per high-powered field
Patient disease and treatment history
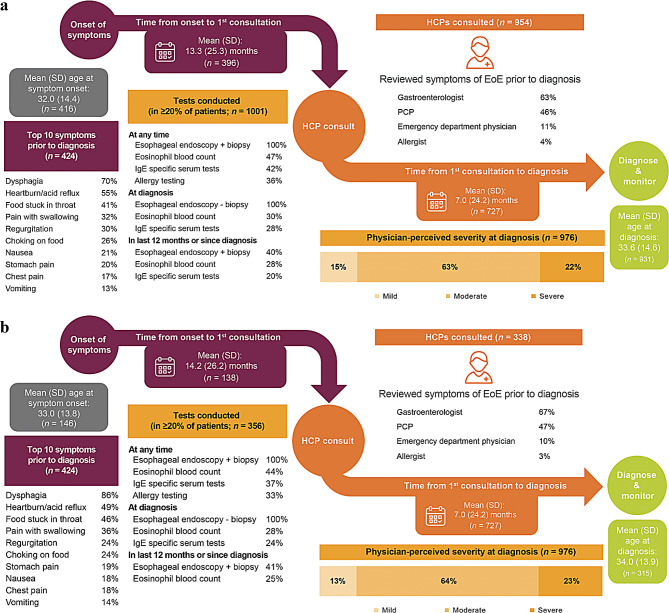
Patient disease history was similar in the overall and dysphagia-despite-treatment populations, respectively, with a few exceptions (Fig. 2). The most common patient-reported symptoms (N = 424 and N = 148) prior to diagnosis were dysphagia (70% and 86%) and heartburn/acid reflux (55% and 49%). The most common physician-reported symptoms (N = 972 and N = 341) were dysphagia (75% and 91%), food impaction (46% and 52%), reflux (45% and 40%), heartburn (32% and 25%), regurgitation (26% and 24%), choking on food (24% and 21%), and chest pain (22% and 24%), indicating differences between patient and physician reports. In the overall population, the mean (SD) time since the onset of symptoms to the first consultation was > 1 year (13.3 [25.26] months); patients were diagnosed on average within 1 year after the first consultation (mean [SD]: 7.0 [24.2] months), at which time 22% of patients had severe EoE. Symptoms were usually reviewed by a gastroenterologist (63%) prior to diagnosis, followed by primary care physicians (46%) and emergency department physicians (11%). Patient disease history was similar in populations categorized by region (US and EU5; Supplementary Table 1).
Fig. 2.
Patient journey from the onset of EoE symptoms to diagnosis (a) overall and (b) in patients with dysphagia despite treatment. EoE, eosinophilic esophagitis; HCP, health care provider; PCP, primary care provider
Treatment history did not differ between the overall and dysphagia-despite-treatment populations (Table 3). In the overall population, the most common current treatments (physician-reported) were proton pump inhibitors (83%) and topical corticosteroids (51%), with approximately 2 medications prescribed per patient. Most patients were very compliant or fully compliant with their medication treatment plan (patient-reported; 75%), and the main reason for noncompliance was forgetting to take the medication (58%). Similar proportions in both populations have had to adhere to an elimination diet (patient-reported), with most patients at least moderately compliant with their elimination diet. Additionally, in the overall population, 20% (196/991) of patients underwent dilation owing to EoE (physician-reported); the mean (SD) number of dilations since diagnosis was 1.7 (1.31; N = 185).
Table 3.
Treatment history
| Characteristic, n (%) | Overall (N = 1001) |
Dysphagia despite treatment (N = 356) |
|---|---|---|
| Physician-reported | ||
| Current prescribed pharmacological treatments | ||
| Topical corticosteroids | 507 (51) | 212 (60) |
| Oral corticosteroids | 86 (9) | 30 (8) |
| Systemic corticosteroids | 2 (< 1) | 2 (1) |
| Other corticosteroids – not specified | 115 (11) | 46 (13) |
| Proton pump inhibitor | 831 (83) | 299 (84) |
| Biologic therapy | 32 (3) | 7 (2) |
| Antihistamine | 152 (15) | 48 (13) |
| Latency reversal agents | 66 (7) | 18 (5) |
| Number of treatments currently prescribed, mean (SD) | 1.8 (0.85) | 1.9 (0.81) |
| Details of dilations due to EoE | ||
| Undergone dilation? | (N = 991) | (N = 349) |
| Yes | 196 (30) | 90 (26) |
| No | 795 (70) | 259 (74) |
| Number of dilations since diagnosis | (N = 185) | (N = 84) |
| Mean (SD) | 1.5 (0.83) | 1.8 (1.72) |
| Patient-reported | ||
| Compliance with prescribed treatment | (N = 417) | (N = 144) |
| Fully compliant | 184 (44) | 52 (36) |
| Very compliant | 128 (31) | 46 (32) |
| Moderately compliant | 92 (22) | 36 (25) |
| Slightly compliant | 12 (3) | 9 (6) |
| Not at all compliant | 1 (< 1) | 1 (1) |
| Reasons for lack of compliance | (N = 204) | (N = 79) |
| I don’t think medication is needed for my EoE | 15 (7) | 8 (10) |
| I have concerns or fears about taking steroids | 42 (21) | 16 (20) |
| I only need to take medication when symptoms get worse and/or when at risk of getting worse | 33 (16) | 12 (15) |
| I do not like to be reliant on my medication | 66 (32) | 29 (37) |
| I do not feel instant results | 30 (15) | 15 (19) |
| Taking medication interferes with my lifestyle | 22 (11) | 11 (14) |
| My EoE medication is not a high priority compared with other medications | 8 (4) | 6 (8) |
| The number of times I have to take medication is not convenient | 12 (6) | 7 (9) |
| I forget to take my medication | 118 (58) | 44 (56) |
| I have experienced side effects | 7 (3) | 4 (5) |
| I am concerned about the long-term safety of treatments | 32 (16) | 15 (19) |
| The cost of the medication is too high | 13 (6) | 8 (10) |
| Other | 1 (< 1) | 0 |
| Details on diet | ||
| Foods eliminated | (N = 416) | (N = 142) |
| Yes, currently | 124 (30) | 124 (37) |
| Not currently but have in the past | 107 (26) | 107 (18) |
| No, never | 185 (44) | 185 (45) |
| Compliance with elimination diet | (N = 223) | (N = 76) |
| Fully | 43 (19) | 21 (28) |
| Very | 72 (32) | 10 (26) |
| Moderately | 87 (39) | 26 (34) |
| Slightly | 18 (8) | 8 (11) |
| Not at all | 3 (1) | 1 (1) |
EoE, eosinophilic esophagitis
Patient symptom burden
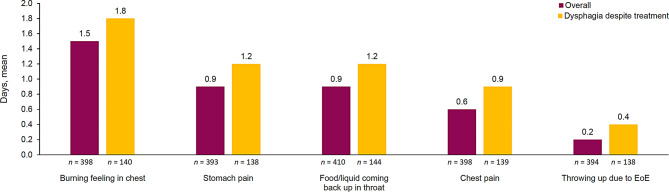
On average, patients in the dysphagia-despite-treatment population reported slightly more days per week with symptoms versus the overall population (Fig. 3), with patients experiencing a range of different symptoms, from mild to severe. In the dysphagia-despite-treatment and overall populations, respectively, patients experienced an average of 1.8 versus 1.5 days with a burning feeling in the chest; 1.2 versus 0.9 days either with food or liquid coming back up into the throat or with stomach pain; and ≤ 1 day of chest pain or throwing up due to EoE in both populations. Similar results were observed by breakdown of US and EU5 populations (Supplementary Fig. 2); however, the mean number of days in which patients experienced chest pain and stomach pain were, on average, notably larger in the EU5 populations versus the US populations.
Fig. 3.
Seven-day symptom burden
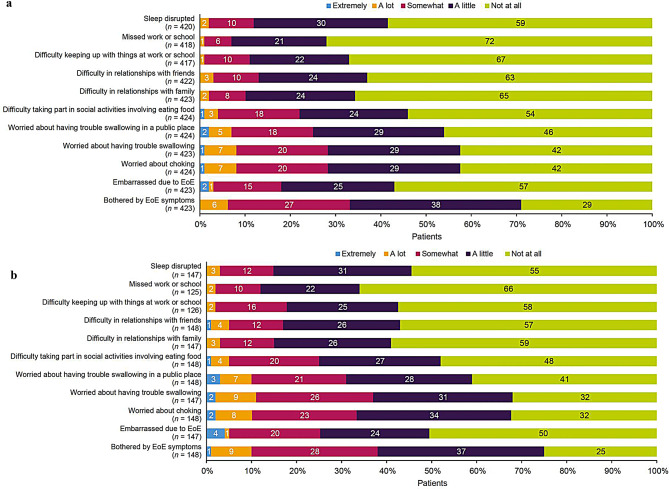
Patients reported higher 7-day impacts on activities of daily living in the dysphagia-despite-treatment versus the overall population (Fig. 4), with patients in both populations experiencing impacts on their sleep, eating habits, and social lives at least a little or a lot each week. In the overall versus dysphagia-despite-treatment populations, respectively, patients reported being at least a little worried about choking (58% vs. 68%); at least a little worried about having trouble swallowing (58% vs. 68%), in a public place as well (54% vs. 59%); and at least a little difficulty taking part in social activities involving food (46% vs. 52%). Additionally, 37% versus 43% of patients in the overall versus dysphagia-despite-treatment populations, respectively, reported at least a little difficulty in relationships with friends, and 35% versus 41% reported at least a little difficulty in relationships with family. These trends were similar in the breakdown by US and EU5 populations (Supplementary Fig. 3 and Supplementary Fig. 4, respectively).
Fig. 4.
Seven-day impact on activities of daily living (a) overall and (b) in patients with dysphagia despite treatment.a EoE, eosinophilic esophagitis. aNumbers may not sum to 100% owing to rounding
There were some differences in the most troublesome symptoms (physician-reported) between the two populations studied. In the overall (N = 528) and dysphagia-despite-treatment (N = 281) populations, respectively, the top troublesome symptoms were dysphagia (23% vs. 43%), food impaction (17% vs. 23%), reflux (13% vs. 5%), and heartburn (11% vs. 6%; Table 4). The mean (SD) patient EQ-5D-3 L utility scores were 0.929 (0.11; N = 390) versus 0.912 (0.13; N = 139) in the overall versus dysphagia-despite-treatment populations, respectively. The mean (SD) EQ-5D visual analog scale (VAS) scores, however, were similar in the overall population versus the dysphagia-despite-treatment population, respectively (83.3 [12.3], N = 377 vs. 81.2 [13.6], N = 135). The symptom burden data breakdown by US and EU5 populations can be found in Supplementary Table 2.
Table 4.
Symptom burden
| Characteristic, n (%) | Overall (N = 1001) |
Dysphagia despite treatment (N = 356) |
|---|---|---|
| Physician-reported | ||
| Top 10 most troublesome symptoms to patient, n (%) | N = 528 | N = 281 |
| Dysphagia | 121 (23) | 121 (43) |
| Food impaction | 89 (17) | 65 (23) |
| Reflux | 71 (13) | 15 (5) |
| Heartburn | 58 (11) | 17 (6) |
| Choking on food | 36 (7) | 16 (6) |
| Food-related anxiety (e.g. fear of impaction) | 34 (6) | 14 (5) |
| Regurgitation | 23 (4) | 8 (3) |
| Stomach pain | 15 (3) | 2 (1) |
| Nausea | 8 (2) | 3 (1) |
| Vomiting | 6 (1) | 0 |
| Patient-reported | ||
| EQ-5D VAS score, mean (SD) | N = 377 | N = 135 |
| 83.3 (12.26) | 81.2 (13.59) | |
| EQ-5D-3 L score, mean (SD) | N = 390 | N = 139 |
| 0.929 (0.11) | 0.912 (0.13) | |
EoE, eosinophilic esophagitis; EQ-5D VAS, EuroQol 5-dimension visual analog scale; EQ-5D-3 L, EuroQol 5-dimension 3-level version
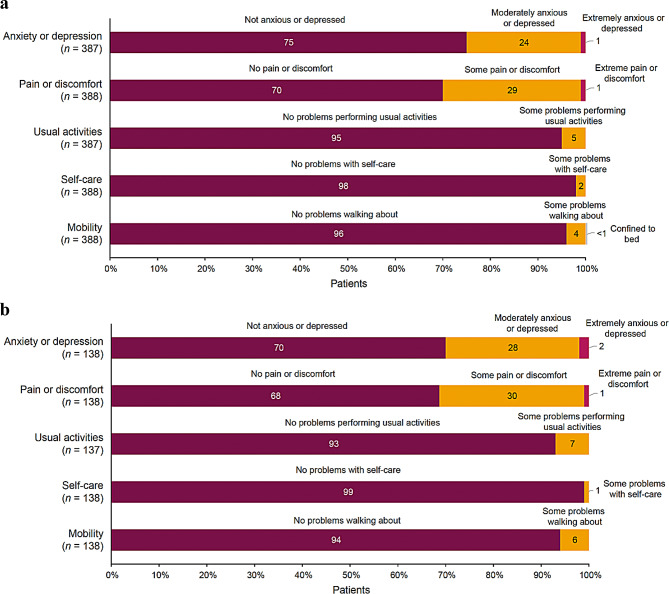
Outcomes from the EQ-5D-3 L utility score indicate slightly higher anxiety or depression in the dysphagia-despite-treatment population versus the overall population, with 30% versus 25% reporting at least moderate anxiety or depression (Fig. 5). Of note, 30% and 32% of patients in the overall and dysphagia-despite-treatment populations, respectively, reported at least moderate pain or discomfort. Outcomes by breakdown into US and EU5 populations generally follow similar trends as well (Supplementary Fig. 5 and Supplementary Fig. 6, respectively); however, greater proportions of patients in the EU5 experienced pain or discomfort and had at least some problems with self-care in the overall population versus the dysphagia-despite-treatment population.
Fig. 5.
EQ-5D-3L utility domain responses (a) overall and (b) in patients with dysphagia despite treatment.a EQ-5D-3L, EuroQol 5-dimension 3-level. aNumbers may not sum to 100% owing to rounding
Discussion
This real-world study evaluated clinical characteristics, treatment history, and symptom burden in patients with EoE. Although the average time between the first healthcare provider consultation to EoE diagnosis was approximately half the time between the onset of symptoms to the first healthcare provider consultation, the high standard deviation in both of the populations we evaluated indicates significant wait time. In addition, by the time patients were diagnosed with EoE, a proportion of patients may have progressed to severe disease. Treatment history data, which were collected prior to the approval of dupilumab for EoE [13], were similar between both populations. Regardless of the congruent treatment histories, results from this study further demonstrate that EoE substantially impacts patients’ HRQOL and activities of daily living, which includes negative effects on their social, mental, and emotional well-being. Patients with dysphagia despite treatment experience higher symptom and HRQOL burden with dysphagia itself and anxiety or depression per EQ-5D-3 L responses, further underscoring the urgent need for novel, targeted treatment for patients with EoE. Moreover, because there were few notable differences in the overall versus dysphagia-despite-treatment populations, treatment of EoE patients warrants close follow-up and open discussions around symptoms to ensure the disease is well-managed.
In addition to the higher symptom burden experienced among patients with dysphagia despite treatment, outcomes from this study show that there is some discordance between patients and physicians regarding what each of them reports as being burdensome or worrisome. The most troublesome symptoms per physicians were dysphagia and food impaction; however, for patients, the most troublesome symptoms were food/liquid coming back up in the throat, stomach pain, and a burning feeling in the chest, and more than 50% of patients reported being worried about trouble when swallowing or choking. These differences reflect physicians’ lack of awareness of patients’ fears and how they might affect their lives.
Despite this discrepancy in awareness of patients’ fears, the current patient-reported assessment does not capture these anxieties. For example, the high EQ-5D-3 L VAS scores (indicating good health) and similar EQ-5D-3 L outcomes on usual activity, self-care, and mobility in both the overall and dysphagia-despite-treatment populations indicate that patients in both of these cohorts could lead a normal life; however, many impacts are not reflected in the current assessments. Social media studies also suggest a remaining unmet need for accurate sources of information about EoE [18, 19]. Although patients affected by chronic illnesses may often consult social media and patient-to-patient forums, there remains an unmet need for reliable information, owing to the lack of physician-verified claims and data. Given that EoE was first reported in the late 1960s [21] to 1970s [22, 23], few qualitative studies have been conducted that assess patient well-being through HRQOL [15–17], and outlets like social media may offer a wealth of evidence-based knowledge outside of the clinical setting that provides valuable insight into how patients actually perceive their EoE. Patients with EoE often have anxiety about eating owing to fear of choking, with the added psychosocial impact of not being able to eat normally and the resulting anxiety and/or embarrassment [12, 24]. Presenting symptoms may vary and make it challenging for a physician to diagnose properly. Supplementing physicians’ knowledge and understanding from the patient perspective is therefore crucial in order to make proper diagnoses and treatment recommendations. Patients may also view their disease as a series of distinct episodes that warrant management with as-needed strategies (e.g. chewing slowly and carefully); these behavioral modifications may therefore mask the impact of their symptoms when consulting their physicians, resulting in possible delays in diagnosis and treatment. While physicians are focused on diagnosing EoE to initiate pharmaceutical intervention as early as possible, per a recent qualitative analysis of three publicly accessible electronic health forums hosting EoE communities, many patients who recognized EoE as a chronic disease have voiced their preference for using dietary approaches over medications as a more permanent solution without reliance on lifelong medication use [19].
It is interesting to further note some slight differences in the overall versus dysphagia-despite-treatment poulations when stratified by location (i.e., US and EU5 countries). The types of tests conducted varied, with higher proportions of patients in the EU5 countries undergoing eosinophil blood count tests, IgE-specific serum tests, and allergy testing compared with patients in the US. The time from onset to first consultation was on average longer in the US compared with EU5 in both overall and dysphagia-despite-treatment populations. Higher patient-reported EQ-5D VAS scores and generally lower 7-day symptom burden were reported in the US compared with EU5 in both populations as well. Taken together, these results suggest that there are differences in healthcare system outputs coupled with variations in the EoE experience in the US compared with EU5 countries, which may impact how EoE is managed in these different regions.
Two strengths of this study are noteworthy: the study’s inclusion of large, robust numbers of patients and its objective and impartial nature. Both clinical and subjective variables are captured, with real-life patient-reported outcomes. Consistent methodology was used in all regions, allowing for true cross-country comparisons. Given that the study was descriptive in nature, there was no set hypothesis prior to data collection, providing flexibility in outcomes studied and potentially eliminating bias. A limitation is that this disease-specific study may ignore the presence of multiple chronic conditions. In addition, the patient surveys were voluntary: not all patients completed the questionnaire, and these data represent only the population of patients who did.
EoE is a multisymptomatic, chronic allergic inflammatory disease of the esophagus that can greatly hinder a patient’s overall well-being and quality of life, especially related to diet and eating, despite standard-of-care treatments. In addition to the need for novel treatments, there is also a substantial need for greater awareness of the condition, including the burdensome symptoms and disease journey faced by many patients with EoE, to encourage earlier diagnosis and closer management of the disease.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
The authors thank Lucy Earl (Adelphi Real World) and Anamaria Brailean (AstraZeneca) for their support in conducting this study. Medical writing support was provided by Laura Weber, PhD, CMPP, and Dan Jackson, PhD, CMPP, from Citrus Scientific, a Citrus Health Group, Inc., company (Chicago, Illinois), which was funded by AstraZeneca (Cambridge, UK) in accordance with Good Publication Practice (GPP 2022) guidelines.
Abbreviations
- EoE
Eosinophilic esophagitis
- HRQoL
Health-related quality of life
- EU
European Union
- UK
United Kingdom
- Eos/hpf
Eosinophils/high-power field
- EQ-5D-3L
EuroQol 5-dimension 3-level
- ICC/ESOMAR
International Code on Market/European Society for Opinion and Market Research
- HIPAA
Health Insurance Portability and Accountability Act of 1996
- SD
Standard deviation
- VAS
Visual analog scale
Author contributions
All authors were involved in all aspects of the study and met all authorship requirements.
Funding
This study was funded by AstraZeneca (Cambridge, UK). The Adelphi Real World EoE Disease Specific Programme™ used in this analysis was funded and collected independently by Adelphi Real World.
Data availability
This study was an analysis of secondary data accessed from a database of cross-sectional patient and physician survey data collected as part of the Adelphi Real World’s EoE Disease Specific Programme (DSP)™, and as such, the data that support the findings of this study are available from Adelphi. However, restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Adelphi.
Declarations
Ethics approval and consent to participate
The study was conducted according to standard operating procedures. Research was conducted as a survey in accordance with the amended Declaration of Helsinki, adhering to the ICC/ESOMAR International Code on Market, Opinion and Social Research and Data Analytics, the international code on observational research, and HIPAA (Health Insurance Portability and Accountability Act of 1996) guidelines. The survey was submitted to a central international review board (Western Institutional Review Board) and found to be exempt from ethics requirements on 23 July 2020 (Study Number: 1-1328144-1).
Consent for publication
Not applicable.
Competing interests
Yes - X.X., J.K., R.K., E.G., and H.S.-F. are or were employees of AstraZeneca at the time of this study and may own stock. J.S. is an employee of Adelphi Real World, which received funding from AstraZeneca to conduct this analysis.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Dellon ES, Gonsalves N, Hirano I, et al. ACG clinical guideline: evidenced based approach to the diagnosis and management of esophageal eosinophilia and eosinophilic esophagitis (EoE). Am J Gastroenterol. 2013;108(5):679–92. 10.1038/ajg.2013.71 [DOI] [PubMed] [Google Scholar]
- 2.Lucendo AJ, Molina-Infante J, Arias A, et al. Guidelines on eosinophilic esophagitis: evidence-based statements and recommendations for diagnosis and management in children and adults. United Eur Gastroenterol J. 2017;5(3):335–58. 10.1177/2050640616689525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pokrzywinski RM, Harding G, Brooks A, Goodwin B, Williams J. Documenting the journey of patients with eosinophilic esophagitis and the impact of the disease on patients and their caregivers: a cross-sectional, qualitative research study. Adv Ther. 2020;37(10):4458–78. 10.1007/s12325-020-01463-2 [DOI] [PubMed] [Google Scholar]
- 4.Jensen ET, Kappelman MD, Martin CF, Dellon ES. Health-Care utilization, costs, and the burden of disease related to eosinophilic esophagitis in the United States. Am J Gastroenterol. 2015;110(5):626–32. 10.1038/ajg.2014.316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bredenoord AJ, Patel K, Schoepfer AM, et al. Disease Burden and Unmet need in Eosinophilic Esophagitis. Am J Gastroenterol. 2022;117(8):1231–41. 10.14309/ajg.0000000000001777 [DOI] [PubMed] [Google Scholar]
- 6.O’Shea KM, Aceves SS, Dellon ES, et al. Pathophysiology of Eosinophilic Esophagitis. Gastroenterology. 2018;154(2):333–45. 10.1053/j.gastro.2017.06.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jorveza. (budesonide) [package insert]. Freiburg, Germany: Dr. Falk Pharma GmbH; 2022.
- 8.Jorveza. (budesonide) [package insert]. Blainville, Quebec, Canada: AVIR Pharma Inc.; 2022.
- 9.Dellon ES, Liacouras CA, Molina-Infante J, et al. Updated International Consensus Diagnostic Criteria for Eosinophilic Esophagitis: Proceedings of the AGREE Conference. Gastroenterology. 2018;155(4):1022–1033.e10. [DOI] [PMC free article] [PubMed]
- 10.Greuter T, Hirano I, Dellon ES. Emerging therapies for eosinophilic esophagitis. J Allergy Clin Immunol. 2020;145(1):38–45. 10.1016/j.jaci.2019.10.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lucendo AJ, Arias-Gonzalez L, Molina-Infante J, Arias A. Systematic review: health-related quality of life in children and adults with eosinophilic oesophagitis-instruments for measurement and determinant factors. Aliment Pharmacol Ther. 2017;46(4):401–9. 10.1111/apt.14194 [DOI] [PubMed] [Google Scholar]
- 12.Taft TH, Carlson DA, Simons M, et al. Esophageal hypervigilance and symptom-specific anxiety in patients with eosinophilic esophagitis. Gastroenterology. 2021;161(4):1133–44. 10.1053/j.gastro.2021.06.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.FDA Approves First Treatment for Eosinophilic Esophagitis, a Chronic Immune Disorder. 2022; https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-eosinophilic-esophagitis-chronic-immune-disorder
- 14.Mukkada V, Falk GW, Eichinger CS, King D, Todorova L, Shaheen NJ. Health-related quality of life and costs Associated with Eosinophilic Esophagitis: a systematic review. Clin Gastroenterol Hepatol. 2018;16(4):495–503.e8. 10.1016/j.cgh.2017.06.036 [DOI] [PubMed] [Google Scholar]
- 15.Franciosi JP, Hommel KA, DeBrosse CW, et al. Quality of life in paediatric eosinophilic oesophagitis: what is important to patients? Child Care Health Dev. 2012;38(4):477–83. 10.1111/j.1365-2214.2011.01265.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Taft TH, Kern E, Keefer L, Burstein D, Hirano I. Qualitative assessment of patient-reported outcomes in adults with eosinophilic esophagitis. J Clin Gastroenterol. 2011;45(9):769–74. 10.1097/MCG.0b013e3182166a5a [DOI] [PubMed] [Google Scholar]
- 17.Flood EM, Beusterien KM, Amonkar MM, et al. Patient and caregiver perspective on pediatric eosinophilic esophagitis and newly developed symptom questionnaires*. Curr Med Res Opin. 2008;24(12):3369–81. 10.1185/03007990802536900 [DOI] [PubMed] [Google Scholar]
- 18.Thanawala SU, Beveridge CA, Muir AB et al. Hashing out current social media use in eosinophilic esophagitis. Dis Esophagus 2021;34(11):doab059. [DOI] [PMC free article] [PubMed]
- 19.Chang JW, Chen VL, Rubenstein JH, Dellon ES, Wallner LP, De Vries R. What patients with eosinophilic esophagitis may not share with their providers: a qualitative assessment of online health communities. Dis Esophagus 2022;35(6):doab073. [DOI] [PMC free article] [PubMed]
- 20.Anderson P, Benford M, Harris N, Karavali M, Piercy J. Real-world physician and patient behaviour across countries: Disease-Specific programmes - a means to understand. Curr Med Res Opin. 2008;24(11):3063–72. 10.1185/03007990802457040 [DOI] [PubMed] [Google Scholar]
- 21.Kelley ML Jr., Frazer JP. Symptomatic mid-esophageal webs. JAMA. 1966;197(2):143–6. 10.1001/jama.1966.03110020131047 [DOI] [PubMed] [Google Scholar]
- 22.Shiflett DW, Gilliam JH, Wu WC, Austin WE, Ott DJ. Multiple esophageal webs. Gastroenterology. 1979;77(3):556–9. 10.1016/0016-5085(79)90022-2 [DOI] [PubMed] [Google Scholar]
- 23.Landres RT, Kuster GG, Strum WB. Eosinophilic esophagitis in a patient with vigorous achalasia. Gastroenterology. 1978;74(6):1298–301. 10.1016/0016-5085(78)90710-2 [DOI] [PubMed] [Google Scholar]
- 24.Taft TH, Guadagnoli L, Edlynn E. Anxiety and depression in Eosinophilic Esophagitis: a scoping review and recommendations for Future Research. J Asthma Allergy. 2019;12:389–99. 10.2147/JAA.S193045 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
This study was an analysis of secondary data accessed from a database of cross-sectional patient and physician survey data collected as part of the Adelphi Real World’s EoE Disease Specific Programme (DSP)™, and as such, the data that support the findings of this study are available from Adelphi. However, restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Adelphi.