Abstract
Background:
Anemia occurs in majority of patients with chronic kidney disease despite adequate dialysis and iron replete status. This study was done to evaluate the effects of lactoferrin with or without iron supplementation for the treatment of anemia in patients with chronic kidney disease (CKD).
Materials and Methods:
In this prospective, observational, single-center, single-arm pilot study, adult patients aged >18 years, having stage 5 CKD (estimated glomerular filtration rate [eGFR] <15 ml/min/1.73 m2), and who had anemia (hemoglobin [Hb] <10 g/dl; transferrin saturation [Tsat] >20%) were included. Patients were treated with 100 mg of oral lactoferrin twice a day for one month with or without iron supplementation. Patients had been on stable erythropoietin doses for ≥1 month prior to inclusion in the study. We report on the improvement in Hb levels and effect on inflammatory markers from baseline at four weeks.
Results:
A total of 46 CKD patients having anemia were included. Patients had a mean age of 39.3 years, and a majority were men (69.6%). Improvement in the mean (SD) Hb level (g/dl) was observed from baseline (8.18 [1.19]) to Week 2 (8.54 [1.57]), which attained significance at Week 4 (8.96 [1.93]; P < 0.001; mean difference: −0.76; 95% confidence interval [CI]: −1.291 to − 0.2383). The improvement in Hb was higher in women than in men (P = 0.48) and in patients receiving lactoferrin with iron supplementation than in those receiving lactoferrin alone (P = 0.14). There was a non-significant decrease in the erythrocyte sedimentation rate (P = 0.14) and a non-significant increase in C-reactive protein (P = 0.54) level.
Conclusion:
Oral lactoferrin therapy was effective in improving hemoglobin levels in patients with advanced CKD and anemia. The effects of lactoferrin therapy on inflammatory markers remain uncertain.
Keywords: Anemia, chronic kidney disease, CKD, hemoglobin, inflammation, iron, lactoferrin
Introduction
Chronic kidney disease (CKD) is a progressive condition with a high prevalence rate of >10% of the global population accounting for >840 million individuals.1 Anemia is amongst the most common complications of CKD and its reported prevalence is twice (15.4%) of that found in the general population (7.6%).2,3 In India, the national level data on anemia in CKD is scarce, but individual studies have reported a prevalence of anemia in 40%– 80% of CKD patients.4,5
Anemia in CKD generally develops when the kidney function deteriorates, leading to a decrease in glomerular filtration rate (GFR) to <60 ml/min.6 CKD progression leads to increased prevalence of anemia, ranging from 8.4% at stage 1 to 53.4% at stage 5.7 Anemia is associated with poor quality of life, increased risk of infections, hospitalization, and, possibly, rapid CKD progression and increased mortality.8 In India, CKD patients generally present at an advanced stage of the disease due to lack of community-based screening programs and awareness, late referral to a nephrologist, and financial constraints.5,9,10 Furthermore, CKD patients who have diabetes and older people are more prone to developing anemia.3,11
The pathophysiology of anemia in CKD is multifactorial, of which deficiency of endogenous erythropoietin (EPO), decreased iron intake and utilization, increased hepcidin levels, and altered homeostasis due to inflammation are the main factors.7,12–15 The kidneys are the main source of erythropoietin (EPO), and a deficiency of EPO is considered as a principal cause of anemia in CKD.16 An increase in serum hepcidin levels due to decreased renal clearance and associated increase in IL-6 levels in CKD leads to iron sequestration in macrophages and subsequently causes iron-restricted erythropoiesis that lead to anemia. Furthermore, iron deficiency and suppression of erythropoiesis by inflammatory cytokines in case of acute inflammation are other causes of anemia in CKD patients.16–18
The therapeutic armamentarium for anemia in CKD includes blood transfusion, recombinant EPO, erythropoiesis-stimulating agents (ESAs), and hypoxia-inducible factor–prolyl hydroxylase (HIF-PH) inhibitors.2,14,19 Blood transfusions have several disadvantages including iron overload, transfusion reactions, risk of infections, and lung damage.20 Studies have reported worsening of cardiovascular outcomes with ESAs in patients with CKD.18,21,22 Intravenous (IV) or oral iron therapy, with or without ESAs, has shown to be effective in hemoglobin corrections in CKD patients.23 However, iron overload is a major drawback and can cause multiple organ failure in patients with renal diseases.24,25 In CKD patients, iron homeostasis should be maintained for achieving effective treatment of anemia.25 The HIF-PH inhibitors have differences in the selectivity of individual HIF-PH inhibitors and may possibly exert off-target effects.26
Lactoferrin, a nonheme iron-binding protein, in combination with iron has demonstrated to be effective in treating anemia in people with chronic diseases, in pregnant people, in children, and in cancer patients.27–29 There is a scarcity of literature on the effects of lactoferrin on hemoglobin improvement in adult CKD patients. Hence, this pilot study was conducted to evaluate the efficacy and safety of lactoferrin in correcting hemoglobin in adult CKD patients with anemia.
Materials and Methods
Study design and population
This was a prospective, observational, single-center, single-arm pilot study of patients having CKD with anemia and who received treatment with lactoferrin at the outpatient department of the Post Graduate Institute of Medical Education and Research (PGIMER), Chandigarh, Punjab, India. The study (CTRI/2020/04/024746) was conducted between October 2021 and October 2022.
Adult patients older than 18 years of age and with stage 5 CKD and anemia were included in this study. Both group of patients who were receiving dialysis and who were not receiving dialysis were included. Stage 5 CKD was diagnosed as estimated GFR (eGFR) <15 ml/min/1.73 m2 based on the Kidney Disease Improving Global Outcomes (KDIGO) guidelines 2012.30 CKD patients were included in the study if their Hb levels were less than 10 gm/dl and transferrin saturation (Tsat) was >20%. Patients were excluded if they had experienced blood loss in the last three months, had hematological malignancy, primary hematological diseases that cause anemia, an ongoing infection, hepatitis B, C, or HIV, were receiving systemic immunosuppressive therapy, and were pregnant/lactating.
Treatment
The patients were administered lactoferrin (tab. Ferronemia, La Renon Healthcare Pvt. Ltd., India) at a dose of 100 mg twice a day for one month. Patients could be administered iron supplementation at the investigator’s discretion. All the patients included in this study were on stable doses of erythropoietin therapy, ranging from 133 to 166 IU/kg once per week for at least one month prior to inclusion in the study. None of the patients were on HIF-PH inhibitors. Vitamin B12 and folate levels were tested at screening, and if their levels were found to be deficient, they were given the appropriate treatment for one month and then were included in the study.
Study assessments
The patients were evaluated for medical history and laboratory investigations, including Hb, serum iron, serum ferritin, total iron-binding capacity (TIBC), hepcidin, and inflammatory markers including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), IL1, IL 6, IL 8, IL 10, IL12 and TNF Alpha. The study endpoint included improvement in Hb level from baseline at four weeks. The patient’s data, including age, gender, etiology of CKD, dialysis status, estimated GFR (eGFR), and medical history, were noted.
Sample size and statistical analysis
This was a pilot study, and no formal sample size calculation was performed. This study included 46 adult patients of CKD stage 5 who were diagnosed with anemia. The data was collected, and appropriate statistical analysis was performed using GraphPad Prism version 9. Demographic and baseline characteristics are summarized using descriptive statistics. Categorical variables are summarized with frequency and percentage. Continuous variables are summarized with count, mean, standard deviation, etc. One-way repeated measures ANOVA was applied to multiple comparisons of Hb levels from baseline to Weeks 2 and 4. A P- value of <0.05 was considered significant.
Ethics statement
This pilot study was reviewed and approved by the institutional ethics committee (approval number: IEC-08/2019-1344). The study was conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and in accordance with the International Conference on Harmonization’s Good Clinical Practice guidelines (ICH-GCP). All participants provided written and informed consent.
Results
A total of 46 patients having stage 5 CKD were included in the study. The mean (SD) age of the patients was 39.3 (13.0) and ranged from 18 to 66 years. The majority (69.6%) of patients in our study were men. The mean BMI was 21.47 kg/m2, which ranged from 12.2 to 32.9 kg/m2. The majority (84.78%) of patients were undergoing dialysis. Of all patients, 39.13% received lactoferrin along with an iron supplementation. Seven of these patients were on 100 mg of intravenous iron every two weeks and 11 were on oral iron. The average dose of elemental iron was 25.71 ± 7.13 mg/day. Table 1 provides the demographics and baseline characteristics of study patients.
Table 1:
Demographics and baseline characteristics
| Parameters | Values |
|---|---|
| Age in years, mean(SD), range | 39.3 (13.0), 18–66 |
| Gender, n (%) | |
| Male | 32 (69.6) |
| Female | 14 (30.4) |
| Diabetes present, n (%) | 7 (15.22) |
| Hypertension present, n (%) | 39 (84.78) |
| Dialysis, n (%) | 39 (84.78) |
| Serum iron, µg/dl, mean (SD) | 102.73 (66.62) |
| BMI, kg/m2, mean (SD) | 21.47 (21.99) |
| Hb, g/dl, mean (SD) | 8.18 (1.19) |
| TIBC, µg/dl, mean (SD) | 241.20 (57.80) |
| Tsat, %, mean (SD) | 40.53 (20.86) |
| Ferritin, ng/ml, mean (SD) | 954.20 (783.75) |
| Vitamin B12, pg/ml, mean (SD) | 1217.56 (699.06) |
| Serum folate, ng/ml, mean (SD) | 13.93 (8.46) |
| ESR, ml/h, mean (SD) | 71.38 (27.12) |
| CRP, mg/dl, mean (SD) | 8.36 (13.44) |
| TNF-α, pg/ml, mean (SD) | 0.31 (1.3) |
| IL-12p70, pg/ml, mean (SD) | 0.17 (0.9) |
| IL-10, pg/ml, mean (SD) | 0.2 (0.9) |
| IL-6, pg/ml, mean (SD) | 7.75 (10.9) |
| IL-1, pg/ml, mean (SD) | 0.18 (0.9) |
| IL-8, pg/ml, mean (SD) | 26.26 (29.4) |
| Hepcidin, ng/dl, mean (SD) | 182.6 (89.62) |
| Iron treatment, n (%) | 18 (39.13) |
BMI=Body mass index, CRP=C-reactive protein, ESA=Erythropoietin stimulating agent, ESR=Erythrocyte sedimentation rate, Hb=Hemoglobin, IL=Interleukin, SD=Standard deviation, TIBC=Total iron-binding capacity, Tsat=Transferrin saturation, TNF=Tumor necrosis factor
Improvement in hemoglobin
After oral lactoferrin therapy, improvement in the mean (SD) Hb level (g/dl) was observed from baseline (8.18 [1.19]) to Week 2 (8.54 [1.57]), which attained significance in Week 4 (8.96 [1.93]; P < 0.001; mean difference: −0.76; 95% confidence interval [CI]: −1.291 to − 0.2383) [Figure 1].
Figure 1:
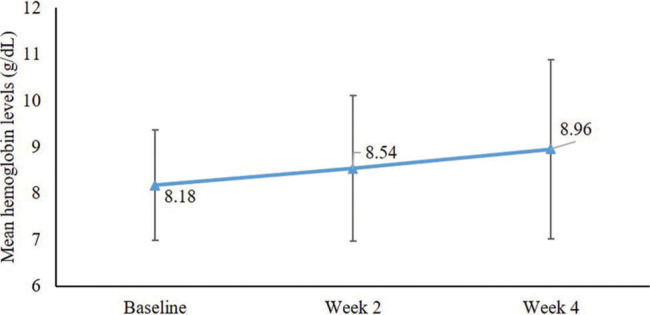
Improvement in hemoglobin levels after lactoferrin therapy.
The improvement in mean Hb level was higher in women than in men from baseline at four weeks (0.99 vs. 0.67 g/dl), though it was not significant (P = 0.48). In patients receiving iron supplementation along with lactoferrin versus those who were receiving lactoferrin alone, the improvement in mean Hb level was higher from baseline to four weeks (1.10 vs. 0.55 g/dl; P = 0.14) [Figure 2].
Figure 2:
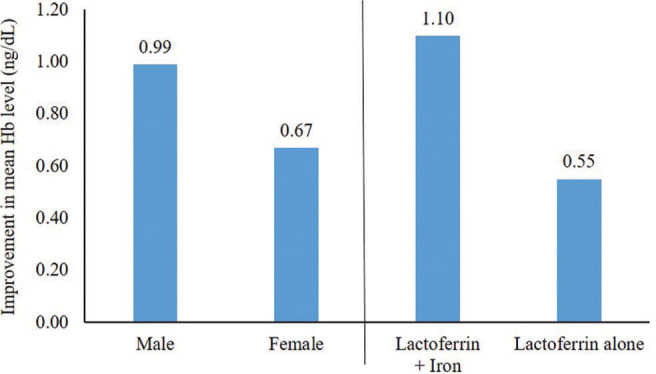
Improvement in hemoglobin levels after lactoferrin therapy from baseline to Week 4 in subgroups based on gender and patients receiving iron supplementation.
Effects of lactoferrin therapy on inflammation
The mean (SD) ESR (ml/h) decreased from baseline (74.22 [28.60]) to Week 4 (67.58 [34.31]), though the difference was not significant (P = 0.14). The mean (SD) CRP (mg/dl) increased from baseline (8.23 [13.16]) at four weeks (11.71 [24.63]) but this difference was not significant (P = 0.54). At baseline, the mean (SD) IL-6 (pg/ml) and TNF-α (pg/ml) levels were 7.75 (10.9) and 0.31 (1.3), respectively. The post-treatment levels of these biomarkers were not available and hence could not be presented.
Hepcidin levels and improvement in hemoglobin
The increase in Hb was not associated with baseline hepcidin levels. The baseline hepcidin levels were categorized in three quartiles as <115, 115–218, and 218–260 ng/ml. At four weeks, there were no significant differences in mean Hb levels observed among hepcidin quartiles (P = 0.388).
Safety
One patient developed diarrhea two days after starting treatment with lactoferrin. However, it did not improve on stopping lactoferrin and responded to antibiotics. This patient was excluded from the study. No other major or minor adverse effects were observed.
Discussion
This pilot study demonstrates that lactoferrin therapy was effective in increasing the Hb levels in patients with CKD. A significant (P < 0.001) increase in Hb level was reported from baseline to four weeks with daily oral lactoferrin therapy with or without iron supplementation. Also, greater improvements, though non-significant, in mean Hb level from baseline at four weeks in women versus men and in patients receiving lactoferrin with iron supplementation versus those receiving lactoferrin alone were reported.
Lactoferrin is a natural nonheme iron-binding cationic glycoprotein belonging to the transferrin family. Lactoferrin has 300 times higher affinity for iron versus transferrin, and it can retain iron at a pH <4, such as in the gastrointestinal tract or inflammatory lesions.31 Lactoferrin binds to two iron ions and enhances the absorption of iron. Lactoferrin enters the intestinal cell through its own receptor and releases the iron there which is transported to the circulation via transferrin.32 Lactoferrin is a promising option for correction of anemia and has demonstrated greater effectiveness and no major and minor side effects, except diarrhea in correcting hematological parameters and decreasing serum IL-6 levels.32,33 Lactoferrin exerts hematological properties through the regulation of hepcidin ferroportin axis, downregulating inflammation through decrease in IL-6 and increasing the systemic iron reserve.34 Lactoferrin also demonstrates reno-protective effects in CKD through reduction of inflammation, oxidative stress, apoptosis and renal fibrosis, and induction of autophagy and mitochondrial biogenesis in the kidneys.35,36 In human kidney proximal tubular cells, lactoferrin induces autophagy through activating the AMP-activated protein kinase (AMPK) and inhibiting the Akt/mTOR pathways.36
In patients with CKD, the level of proinflammatory cytokines such as IL-6 and TNF-α are elevated.37 The increase in the levels of inflammatory markers such as IL-6 in patients with CKD is observed due to oxidative stress, chronic inflammation, fluid overload, and decreased clearance of IL-6 associated with impaired renal function.38 An increased level of IL-6 is reported to be involved in iron homeostasis disorders.39 Lactoferrin downregulates inflammation through decrease in IL-6 and increases the systemic iron reserve.34 It is hence emerging as a potent regulator of iron and inflammatory homeostasis.39
The efficacy of lactoferrin in improving Hb levels has been evaluated mainly in anemia in those with chronic diseases, those who are pregnant, in children, and in cancer patients. In a meta-analysis of 8 studies in patients with iron deficiency, anemia, pregnant or non-pregnant women, Zhao et al.40 observed that lactoferrin therapy had more beneficial effects on serum iron (P < 0.00001), ferritin (P = 0.003) and Hb level (P < 0.00001) as compared to ferrous sulfate. El Amrousy et al.32 evaluated the efficacy of lactoferrin in iron-deficiency anemia in children with inflammatory bowel disease and concluded that lactoferrin was an effective treatment in improving hemoglobin and that it showed fewer side effects compared to oral elemental iron. Furthermore, lactoferrin is associated with better gastrointestinal tolerance and fewer side effects than ferrous sulfate.40
Lactoferrin, through its IL-6 downregulating properties, can modulate hepcidin and ferroportin synthesis. In a multicenter, comparative, interventional study, lactoferrin increased total serum iron and decreased the inflammatory markers IL-6 and hepcidin to a greater extent than ferrous sulfate did in pregnant or non-pregnant women with anemia or anemia of inflammation.34 In the current study, based on the different quartiles of baseline hepcidin levels, no significant differences for Hb improvement were observed. The effects of lactoferrin therapy on inflammation remained uncertain in this study as there was a non-significant decrease in mean ESR, whereas an increase in CRP was observed. Most of the patients were on dialysis. Factors that could have caused increased CRP are dialysis itself, presence of dialysis catheters, and chronic kidney disease per se. TNF-α level was very low in a majority of the patients in this study. However, TNF-α levels were found to be elevated in patients with CKD as opposed to healthy individuals.41 In our study, improvements in inflammatory markers were not certain; however, there was improvement in Hb levels. It indicates that improvement in inflammation either is not significant with lactoferrin or a higher dose of lactoferrin is required to reduce inflammation in CKD. Interestingly, a few of the studies on pregnant women with anemia used a high daily dose of lactoferrin at 250 mg/day versus 100 mg b.i.d. in our study.40,42,43 Future studies are warranted to evaluate the effects of lactoferrin on inflammatory markers.
Inflammation has been reported to increase the risk of cardiovascular events and subsequent mortality.As an example high CRP levels that occur in inflammation in patients on hemodialysis have been reported as a predictor of cardiovascular death.44 Improvement in inflammation improves cardiovascular outcomes and mortality.45 We did not find a consistent decrease in inflammatory markers in our study; it indicates that the dose of lactoferrin used in this study may not improve mortality outcomes. It is possible that a larger dose for a prolonged duration is needed to improve inflammation and inflammatory markers in CKD. If so, lactoferrin may improve outcomes in CKD, beyond improvement in anemia and this may retard decline in glomerular filtration rate and reduce mortality.
El-Farsy et al.29 recently demonstrated the efficacy and safety of lactoferrin for anemia correction in children with anemia and CKD stages 1 to 4. The authors reported a significant increase in Hb levels within a month of lactoferrin therapy, which is consistent with our findings where significant increase in Hb levels were reported four weeks after lactoferrin therapy in adult patients had been initiated. However, the researchers did not report the effects of lactoferrin on inflammatory markers in these patients who had renal anemia.29
Patients who have CKD are generally treated with IV iron rather than oral iron formulations as the efficacy of oral iron supplements in these patients can be hampered because of limited gastrointestinal absorption of iron due to elevated circulating concentrations of hepcidin.46 The current study has reported benefits of combining lactoferrin with iron, and studies evaluating these effects with IV iron also are warranted.
Renal anemia is more prevalent in diabetic CKD patients than in non-diabetic CKD patients, as reported in the Prevalence of Anaemia in Early Renal Insufficiency (PAERI) study (52.7 vs 39.4%; P < 0.01).47 In the current study, 15.2% of patients having CKD and anemia were suffering from diabetes. Limitations to our study include the following: this was a pilot study design without randomization and with a short follow-up duration. The details pertaining to hepcidin (key mediator of iron metabolism), ferritin, transferrin, iron status, and adverse events at follow-up were not available and could hence not be analyzed in this pilot study. Prior to conducting a larger study with a longer follow-up, in this pilot study, we intended to assess if lactoferrin was effective at all in improving anemia in CKD and whether the response to anemia was dependent on baseline hepcidin and inflammatory markers. A study with a larger number of patients and longer follow-up is ongoing and we shall report the changes in iron indices and hepcidin on follow-up. We intended to estimate the effect size of lactoferrin, if any, in this study. Based on these results, we are conducting a randomized study with a parallel non-lactoferrin arm. Also, the benefits from improvement in anemia, such as improvement in quality of life, reduced risk of infections and hospitalization, and avoidance of delay in vascular access creation due to severe anemia, could not be assessed in this pilot due to the short study duration. We plan to address these limitations in our larger ongoing study.
Conclusion
This pilot study demonstrates that lactoferrin therapy was effective in improving hemoglobin levels in CKD stage 3–5 patients with anemia. Largescale randomized clinical trials are required to better elicit these findings. Furthermore, studies are warranted to evaluate the association between lactoferrin and inflammatory markers, which are known to aggravate anemia in patients with CKD. Overall, lactoferrin, with its reno-protective properties, could be a suitable add-on therapy irrespective of EPO, ESA, or HIF-PH inhibitors patients with renal anemia, though these effects need to be evaluated in future studies.
Acknowledgments
The authors thank the study participants without whom the study would not have been completed.
Funding Statement
Financial support and sponsorship
This pilot study was partially funded by the Indian Council of Medical Research (ICMR project ID 2020-9417). The lactoferrin used in the study was tab. Ferronemia of La Renon Healthcare Pvt. Ltd., Ahmedabad, India.
Footnotes
How to cite this article: Kekan K, Divyaveer S, Kashyap M, Premkumar M, Zohmangaihi D, Mallik N, et al. Effectiveness of Lactoferrin in the Treatment of Anemia in Chronic Kidney Disease: A Single-Center Pilot Study. Indian J Nephrol. 2024;34:222–7. doi: 10.4103/ijn.ijn_13_23
Conflicts of interest
There are no conflicts of interest.
References
- 1.Kovesdy CP. Epidemiology of chronic kidney disease: An update 2022. Kidney Int Suppl (2011) 2022;12:7–11. doi: 10.1016/j.kisu.2021.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gafter-Gvili A, Schechter A, Rozen-Zvi B. Iron deficiency anemia in chronic kidney disease. Acta Haematologica. 2019;142:44–50. doi: 10.1159/000496492. [DOI] [PubMed] [Google Scholar]
- 3.Stauffer ME, Fan T. Prevalence of anemia in chronic kidney disease in the United States. PLoS One. 2014;9:e84943. doi: 10.1371/journal.pone.0084943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zaawari A, Tejaswani KL, Davina GD, Singanvani A. Prevalence of anemia among chronic kidney disease patients in India: A single-centre study. Int J Basic Clin Pharmacol. 2022;11:404. doi: 10.18203/2319-2003.ijbcp20222135. [DOI] [Google Scholar]
- 5.Singh AK, Farag YM, Mittal BV, Subramanian KK, Reddy SR, Acharya VN et al. Epidemiology and risk factors of chronic kidney disease in India-results from the SEEK (Screening and Early Evaluation of Kidney Disease) study. BMC Nephrol. 2013;14:114. doi: 10.1186/1471-2369-14-114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hsu CY, McCulloch ME, Curhan GC. Epidemiology of anemia associated with chronic renal insufficiency among adults in the United States: Results from the Third National Health and Nutrition Examination Survey. J Am Soc Nephrol. 2002;13:504–10. doi: 10.1681/ASN.V132504. [DOI] [PubMed] [Google Scholar]
- 7.Portolés J, Martín L, Broseta JJ, Cases A. Anemia in chronic kidney disease: From pathophysiology and current treatments, to future agents. Front Med. 2021;8:642296. doi: 10.3389/fmed.2021.642296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.KDOQI; National Kidney Foundation KDOQI clinical practice guidelines and clinical practice recommendations for anemia in chronic kidney disease. Am J Kidney Dis. 2006;47(Suppl 3):S11–145. doi: 10.1053/j.ajkd.2006.03.011. [DOI] [PubMed] [Google Scholar]
- 9.Jafar TH, Ramakrishnan C, John O, Tewari A, Cobb B, Legido-Quigley H et al. Access to CKD care in rural communities of India: A qualitative study exploring the barriers and potential facilitators. BMC Nephrol. 2020;21:26. doi: 10.1186/s12882-020-1702-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dhanorkar M, Prasad N, Kushwaha R, Behera M, Bhaduaria D, Yaccha M et al. Impact of early versus late referral to nephrologists on outcomes of chronic kidney disease patients in Northern India. Int J Nephrol. 2022;2022:4768540. doi: 10.1155/2022/4768540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fishbane S, Spinowitz B. Update on anemia in ESRD and earlier stages of CKD: Core curriculum 2018. Am J Kidney Dis. 2018;71:423–35. doi: 10.1053/j.ajkd.2017.09.026. [DOI] [PubMed] [Google Scholar]
- 12.Portolés J, Gorriz JL, Rubio E, de Alvaro F, García F, Alvarez-Chivas V et al. The development of anemia is associated to poor prognosis in NKF/KDOQI stage 3 chronic kidney disease. BMC Nephrol. 2013;14:2. doi: 10.1186/1471-2369-14-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Babitt JL, Eisenga MF, Haase VH, Kshirsagar AV, Levin A, Locatelli F et al. Controversies in optimal anemia management: Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int. 2021;99:1280–95. doi: 10.1016/j.kint.2021.03.020. [DOI] [PubMed] [Google Scholar]
- 14.Martínez-Castelao A, Cases A, Coll E, Bonal J, Galceran JM, Fort J et al. C.E.R.A. administered once monthly corrects and maintains stable hemoglobin levels in chronic kidney disease patients not on dialysis: The observational study MICENAS II. Nefrologia. 2015;35:80–6. doi: 10.3265/Nefrologia.pre2014.Sep.12588. [DOI] [PubMed] [Google Scholar]
- 15.Hayat A, Haria D, Salifu MO. Erythropoietin stimulating agents in the management of anemia of chronic kidney disease. Patient Prefer Adherence. 2008;2:195–200. doi: 10.2147/PPA.S2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Babitt JL, Lin HY. Mechanisms of anemia in CKD. J Am Soc Nephrol. 2012;23:1631–4. doi: 10.1681/ASN.2011111078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wojtaszek E, Glogowski T, Malyszko J. Iron and chronic kidney disease: Still a challenge. Front Med (Lausanne) 2020;7:565135. doi: 10.3389/fmed.2020.565135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Batchelor EK, Kapitsinou P, Pergola PE, Kovesdy CP, Jalal DI. Iron deficiency in chronic kidney disease: Updates on pathophysiology, diagnosis, and treatment. J Am Soc Nephrol. 2020;31:456–68. doi: 10.1681/ASN.2019020213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cheng HW, Chan KY, Lau HT, Man CW, Cheng SC, Lam C. Use of erythropoietin-stimulating agents (ESA) in patients with end-stage renal failure decided to forego dialysis: Palliative perspective. Am J Hosp Palliat Care. 2017;34:380–4. doi: 10.1177/1049909115624653. [DOI] [PubMed] [Google Scholar]
- 20.Fox KM, Yee J, Cong Z, Brooks JM, Petersen J, Lamerato L et al. Transfusion burden in non-dialysis chronic kidney disease patients with persistent anemia treated in routine clinical practice: A retrospective observational study. BMC Nephrol. 2012;13:5. doi: 10.1186/1471-2369-13-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Singh AK, Szczech L, Tang KL, Barnhart H, Sapp S, Wolfson M et al. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085–98. doi: 10.1056/NEJMoa065485. [DOI] [PubMed] [Google Scholar]
- 22.Pfeffer MA, Burdmann EA, Chen CY, Cooper ME, de Zeeuw D, Eckardt KU et al. A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019–32. doi: 10.1056/NEJMoa0907845. [DOI] [PubMed] [Google Scholar]
- 23.Hörl WH. Iron therapy for renal anemia: How much needed, how much harmful? Pediatr Nephrol. 2007;22:480–9. doi: 10.1007/s00467-006-0405-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nashwan AJ, Yassin MA, Mohamed Ibrahim MI, Abdul Rahim HF, Shraim M. Iron overload in chronic kidney disease: Less ferritin, more T2*MRI. Front Med (Lausanne) 2022;9:865669. doi: 10.3389/fmed.2022.865669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sulaikha S, Koorikat S, MT NB, K ST, P A, Midhun KP. Lactoferrin, a promising molecule for the treatment of anemia of chronic kidney disease. J Drug Deliv Ther. 2019;9:610–2. [Google Scholar]
- 26.Nagano N. Selectivity of HIF-PH inhibitors: Concerns regarding possible off-target effects. Clin Exp Nephrol. 2021;25:1047–8. doi: 10.1007/s10157-021-02070-3. [DOI] [PubMed] [Google Scholar]
- 27.Artym J, Zimecki M, Kruzel ML. Lactoferrin for prevention and treatment of anemia and inflammation in pregnant women: A comprehensive review. Biomedicines. 2021;9:898. doi: 10.3390/biomedicines9080898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Macciò A, Madeddu C, Gramignano G, Mulas C, Sanna E, Mantovani G et al. Efficacy and safety of oral lactoferrin supplementation in combination with rHuEPO-beta for the treatment of anemia in advanced cancer patients undergoing chemotherapy: Open-label, randomized controlled study. Oncologist. 2010;15:894–902. doi: 10.1634/theoncologist.2010-0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.El-Farsy M, El-Hakim I, Al-Arian R. Role of oral lactoferrin as a source of iron supplementation in correction of anemia in pediatric patients with chronic kidney disease stages 2-4. J Egypt Soc Nephrol Transplantat. 2022;22:193–9. doi: 10.4103/jesnt.jesnt_22_21. [DOI] [Google Scholar]
- 30.Ahemii K. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int. 2012;2:279. [Google Scholar]
- 31.Rosa L, Cutone A, Lepanto MS, Paesano R, Valenti P. Lactoferrin: A natural glycoprotein involved in iron and inflammatory homeostasis. Int J Mol Sci. 2017;18:1985. doi: 10.3390/ijms18091985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.El Amrousy D, El-Afify D, Elsawy A, Elsheikh M, Donia A, Nassar M. Lactoferrin for iron-deficiency anemia in children with inflammatory bowel disease: A clinical trial. Pediatr Res. 2022;92:762–6. doi: 10.1038/s41390-022-02136-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Paesano R, Berlutti F, Pietropaoli M, Goolsbee W, Pacifici E, Valenti P. Lactoferrin efficacy versus ferrous sulfate in curing iron disorders in pregnant and non-pregnant women. Int J Immunopathol Pharmacol. 2010;23:577–87. doi: 10.1177/039463201002300220. [DOI] [PubMed] [Google Scholar]
- 34.Lepanto MS, Rosa L, Cutone A, Conte MP, Paesano R, Valenti P. Efficacy of lactoferrin oral administration in the treatment of anemia and anemia of inflammation in pregnant and non-pregnant women: An interventional study. Front Immunol. 2018;9:2123. doi: 10.3389/fimmu.2018.02123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zahan MS, Ahmed KA, Moni A, Sinopoli A, Ha H, Uddin MJ. Kidney protective potential of lactoferrin: Pharmacological insights and therapeutic advances. Korean J Physiol Pharmacol. 2022;26:1–13. doi: 10.4196/kjpp.2022.26.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hsu Y-H, Chiu IJ, Lin YF, Chen YJ, Lee YH, Chiu HW. Lactoferrin contributes a renoprotective effect in acute kidney injury and early renal fibrosis. Pharmaceutics. 2020;12:434. doi: 10.3390/pharmaceutics12050434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ishigami J, Taliercio J, Feldman H, Srivastava A, Townsend R, Cohen D et al. Inflammatory markers and incidence of hospitalization with infection in chronic kidney disease: The chronic renal insufficiency cohort study. Am J Epidemiol. 2019;189:433–44. doi: 10.1093/aje/kwz246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Su H, Lei C-T, Zhang C. Interleukin-6 signaling pathway and its role in kidney disease: An update. Front Immunol. 2017;8:405. doi: 10.3389/fimmu.2017.00405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lepanto MS, Rosa L, Paesano R, Valenti P, Cutone A. Lactoferrin in aseptic and septic inflammation. Molecules. 2019;24:1323. doi: 10.3390/molecules24071323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhao X, Zhang X, Xu T, Luo J, Luo Y, An P. Comparative effects between oral lactoferrin and ferrous sulfate supplementation on iron-deficiency anemia: A comprehensive review and meta-analysis of clinical trials. Nutrients. 2022;14:543. doi: 10.3390/nu14030543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lee BT, Ahmed FA, Hamm LL, Teran FJ, Chen CS, Liu Y. S et al. Association of C-reactive protein, tumor necrosis factor-alpha, and interleukin-6 with chronic kidney disease. BMC Nephrol. 2015;16:77. doi: 10.1186/s12882-015-0068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rezk M, Dawood R, Abo-Elnasr M, Al Halaby A, Marawan H. Lactoferrin versus ferrous sulphate for the treatment of iron deficiency anemia during pregnancy: A randomized clinical trial. J Matern Fetal Neonatal Med. 2016;29:1387–90. doi: 10.3109/14767058.2015.1049149. [DOI] [PubMed] [Google Scholar]
- 43.Paesano R, Pietropaoli M, Berlutti F, Valenti P. Bovine lactoferrin in preventing preterm delivery associated with sterile inflammation. Biochem Cell Biol. 2012;90:468–75. doi: 10.1139/o11-060. [DOI] [PubMed] [Google Scholar]
- 44.Selim G, Stojceva-Taneva O, Zafirovska K, Sikole A, Gelev S, Dzekova P et al. Inflammation predicts all-cause and cardiovascular mortality in haemodialysis patients. Prilozi. 2006;27:133–44. [PubMed] [Google Scholar]
- 45.Amdur RL, Feldman HI, Dominic EA, Anderson AH, Beddhu S, Rahman M et al. Use of measures of inflammation and kidney function for prediction of atherosclerotic vascular disease events and death in patients with CKD: Findings from the CRIC study. Am J Kidney Dis. 2019;73:344–53. doi: 10.1053/j.ajkd.2018.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gutiérrez OM. Treatment of iron deficiency anemia in CKD and end-stage kidney disease. Kidney Int Rep. 2021;6:2261–9. doi: 10.1016/j.ekir.2021.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Astor BC, Muntner P, Levin A, Eustace JA, Coresh J. Association of kidney function with anemia: The third national health and nutrition examination survey (1988-1994) Arch Intern Med. 2002;162:1401–8. doi: 10.1001/archinte.162.12.1401. [DOI] [PubMed] [Google Scholar]


