Abstract
Chronic liver disease (CLD) is a leading health problem impacting the quality of life globally. China shares a major global burden of CLD—including alcoholic liver disease, nonalcoholic fatty liver disease/metabolic dysfunction-associated fatty liver disease, and drug-induced liver injury, except for chronic viral hepatitis. Several exogenous toxins or endogenous metabolic insults trigger hepatic pathology toward steatosis, inflammation, and fibrosis, which, if left untreated, may culminate in liver cirrhosis. Oxidative stress is a common pathomechanism underlying all phenotypes of toxic liver injury; thus, these may be brought under a unified entity, viz. toxic liver disease (TLD). Therefore, a common strategy to treat TLD is to use antioxidants as hepatoprotective agents. The cornerstone for treating fatty liver disease is lifestyle modification, diet, exercise, and behavioral therapy, along with the limited use of pharmacological agents. Available preclinical and clinical evidence indicates that silymarin is a hepatoprotective agent with established antioxidant, anti-inflammatory, antifibrotic effects. An international expert panel of clinicians was convened to discuss combining alcoholic liver disease, nonalcoholic fatty liver disease/metabolic dysfunction-associated fatty liver disease, drug-induced liver injury, and liver cirrhosis under the single definition of TLD, based on the shared pathologic mechanism of oxidative stress. The panel highlighted the significance of silymarin as an antioxidant treatment for TLD.
Keywords: Alcoholic Fatty Liver Disease, Nonalcoholic Associated Fatty Liver Disease, Metabolic Dysfunction-Associated Fatty Liver Disease, Drug-Induced Liver Injury, Silymarin
Introduction
Chronic liver disease (CLD) is one of the leading global health problems impacting the quality of life of patients. Of note, CLD affects around 1.5 billion people globally.1 China has the highest burden of CLD.2 According to the Global Burden of Disease (2017), 2.14 million deaths have occurred due to liver diseases.3 The disability-adjusted life-years and years of life lost due to CLD globally were 1.6% and 2.1%, respectively. However, disability-adjusted life-years and years of life lost due to CLD in the World Health Organization South East Asia region were 2.2% and 3.0%, respectively.4 Exogenous and endogenous toxins, including alcohol, drugs, metabolic dysregulations, and industrial or environmental toxins, cause toxic liver diseases (TLDs). These toxins cause pronounced structural and functional changes in hepatocytes, leading to liver damage.5,6 Based on etiology, toxic liver damage can be associated with diverse liver diseases—including drug-induced liver injury (DILI), nonalcoholic fatty liver disease (NAFLD)/metabolic dysfunction-associated fatty liver disease (MAFLD), alcoholic liver disease (ALD), liver fibrosis, and cirrhosis. Recently, NAFLD has been redefined as MAFLD manifesting as hepatic steatosis in addition to 1 of the 3 criteria, that is, overweight/obesity, type II diabetes mellitus, or metabolic dysregulation.7 Another relevant subtype of FLD is the toxicant-associated fatty liver disease, which is the outcome of toxicant exposure, including environmental and industrial contaminants. Toxicant-associated steatohepatitis (TASH), which is a severe form of toxicant-associated fatty liver disease, is manifested in the form of hepatic steatosis, inflammatory infiltrate, and, in some cases, fibrosis.8,9
Clinically, DILI, NAFLD/MAFLD, and ALD result in inflammation and fibrosis and, if left untreated, lead to liver cirrhosis and even liver failure.10, 11, 12 Oxidative stress (OS) is a key mechanism for hepatotoxicity and the pathogenesis of TLD due to the accumulation of reactive oxygen species (ROS), leading to structural and functional impairment of the liver.13 The change in OS markers such as catalase, superoxide dismutase (SOD), glutathione peroxidase, coenzyme Q, malondialdehyde, and nonenzymatic electron receptors such as glutathione14, 15, 16 promotes an imbalance in endogenous antioxidant molecules, viz. glutathione S-transferase, heme oxygenase-1, and nicotinamide adenine dinucleotide phosphate.17 Lipid peroxidation (LPO) causes hepatocyte damage and the release of proinflammatory cytokines.15 Covalent bonding of macromolecules (eg, protein, DNA) with metabolically activated intermediates may lead to the formation of protein adducts, which may further contribute to hepatic inflammation and disease progression.14,18 Hence, there is a need to restore the oxidative balance by various mechanisms, such as by inhibition of CYP2E1 and attenuation of ROS generation.19
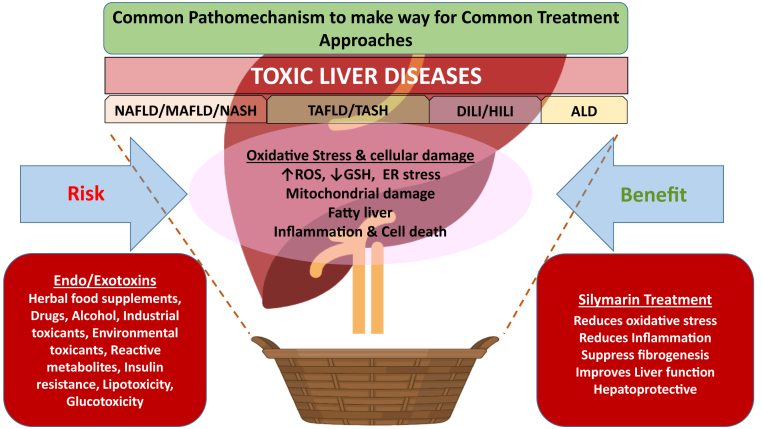
As different liver toxicities share OS as a common disease denominator, they can be grouped under the basket description of TLDs, creating a robust rationale for a shared treatment approach (Figure). OS can be countered with clinically safe and tolerable antioxidant molecules in addition to pharmacological and nonpharmacological interventions. Antioxidants obliterate ROS or may activate endogenous antioxidant pathways, such as the nuclear factor erythroid 2–related factor 2 pathway, and may lower the severity of liver fibrosis.15 By minimizing OS, these agents meaningfully impede clinical progression and serve as hepatoprotective agents.
Figure.
Common oxidative stress mechanism underlies hepatocellular damage by several exogenous and endogenous toxins. Merging clinical phenotypes of fatty liver disease (NAFLD/MAFLD, NASH, DILI, ALD) into a single basket entity, toxic liver diseases (TLDs), based on common pathomechanism will allow clinicians to evaluate the benefits of silymarin as an antioxidant and potential hepatoprotective agent.
The limited treatment options for TLD justify the need for safe alternatives, such as antioxidant therapy. Antioxidants started during the early stages of the disease may prevent disease progression.20, 21, 22 Also, there is no international consensus on the use of antioxidants as a treatment for TLD. Regarding silymarin, although its role as an antioxidant is well recognized, there is a lack of consensus on the dose, treatment duration, and disease stage at which treatment is initiated. Thus, an international advisory panel was convened on December 5, 2020, to understand the merits of combining DILI, NAFLD/MAFLD, nonalcoholic steatohepatitis (NASH), ALD under the basket concept of TLD, and to discuss available evidence and generate consensus statements on the use of silymarin for the clinical management of TLD. The deliberations of the expert panel focused on defining TLD and its scope, understanding the role of OS in TLD, delineating the benefits of silymarin as an antioxidant, and arriving at consensus statements about silymarin as supportive treatment.
Exploring the Pathogenetic Mechanism of Different Phenotypes of TLD
The panel successfully elucidated the diseases falling under the TLD category because of the common underlying pathogenetic mechanism, that is, OS. The respective diseases, their discrete characteristics, epidemiology/prevalence, clinical manifestations (Table 1), and their diagnoses (Table 2) have been illustrated.
Table 1.
The Clinical Features, Etiology, and Distinctive Features of Different Phenotypes of CLD
| Phenotype | Clinical features | Etiology | Epidemiology/prevalence (global and China, wherever applicable) | Distinctive features |
|---|---|---|---|---|
| DILI | It is manifested in the form of elevated liver enzymes, hepatitis, hepatocellular necrosis, cholestasis, fatty liver, and liver cirrhosis.11 DILI is categorized as intrinsic and idiosyncratic. Intrinsic DILI is dose related and occurs in patients with drug exposure within a short period. Idiosyncratic DILI is not dose related, occurs in a smaller population of drug-exposed patients, and variability in onset delays.23 The different categories of DILI are hepatocellular injury, cholestatic injury, hepatocellular-cholestatic mixed injury, and vascular injury based on the site of injury.24 | Pharmacological agents, complementary and alternative medicines including traditional Chinese medicine, and herbal/dietary supplements are the causative agents in DILI.25,26,27 Drug liver toxicity is limited to antibiotics, mainly represented by antitubercular drugs.24,25,28,29 Herbal medicines may also cause liver injury, although the actual composition of the herbal preparation may remain unclear, particularly in multicompound products.24,25,28,29 | DILI has a high incidence rate in China (23.8 per 100,000 inhabitants).25 Large differences in the epidemiology of DILI between Western and Eastern countries have been reported.30 | The development of jaundice or occasionally acute liver failure with coagulopathy and encephalopathy in the presence of jaundice is distinctive in DILI. Fibrosis, granulomatous hepatitis, and nodular regenerative hyperplasia are often present.23 |
| NAFLD/MAFLD | It is primarily asymptomatic comprising several clinical conditions, including steatosis, steatohepatitis, fibrosis, and cirrhosis.31 IR is an important parameter playing a vital role. There are 2 phenotypes of NAFLD, viz. nonalcoholic fatty liver (NAFL or simple fatty liver) and nonalcoholic steatohepatitis (NASH). Nonalcoholic fatty liver indicates the presence of steatosis only, whereas NASH represents steatosis with lobular inflammation and ballooning.32 | NAFLD manifests as excessive liver fat in the absence of secondary causes and significant alcohol consumption.32,33,34 The fat accumulates in the liver in the absence of alcohol consumption (alcohol intake: <20 g/d for female, <30 g/d for male) or any other secondary cause.34 | As of 2017, there were 882 million cases of NAFLD worldwide, with a prevalence rate of 10.9%, majorly seen in the Middle East and North America. The prevalence of NAFLD in China is in the range of 6.3%–27% with a higher occurrence in males than in females and in urban areas vs rural areas. The total number of cases of NASH in China in 2016 was 32.61 million.1,32,35, 36, 37, 38, 39, 40 | The distinct morphological features of NAFLD/MAFLD are large droplet steatosis, ballooning, lobular inflammation. Perisinusoidal fibrosis occurs at the end stages.41 Hepatic steatosis is accompanied by metabolic dysfunctions.42 Delayed diagnosis and intervention may lead to accumulation of diverse exogenous and endogenous hepatotoxic entities that lead to TLD and fibrosis via multiple biochemical pathways.43 |
| For MAFLD, metabolic dysfunctions such as overweight/obesity, type 2 diabetes mellitus additionally take prominence.44 Diverse endogenous or exogenous molecular mediators can result in multiple metabolic syndromes.7 In addition, given the increased risk of cardiovascular events in the NAFLD/MAFLD population, concomitant comorbidities such as viral hepatitis or ALD might worsen the prognosis of such patients.44 | In a meta-analysis by Liu et al, the global prevalence of MAFLD was demonstrated to be 50.7%, 19.7%, and 57.5% in patients with obesity, patients with type II diabetes mellitus, and patients with metabolic syndrome, respectively.36 | |||
| ALD | It is manifested as manifesting as simple steatosis to steatohepatitis and cirrhosis.45,46 Hepatic inflammation, necrosis, apoptosis, and fibrosis occur due to cytokine and oxidative stress cascade involving interactions between Kupffer’s cells, myofibroblasts, and endothelial cells.46,47 | Alcohol is the causative agent. The daily alcohol consumption of 20 g in females and 30 g in males along with clinical or biological alterations might be indicative of liver injury.48 | The prevalence of ALD globally was 26 million as reported by global disease burden, 2018. However, an increase in prevalence was observed in China, from 2.27% in 2000 to 8.74% in 2015, with a higher occurrence noted in males.45 The higher mortality with ALD is due to its detection at a much later stage.19,49 | ALD occurs even in the absence of clinical or biological manifestations and thereby is often diagnosed at later stages.19 The features of ALD include macrovesicular or mixed-type steatosis, hepatocellular injury with ballooning, lobular inflammation.48 |
| Liver fibrosis | Liver fibrosis is manifested in the form of excess collagen due to new fiber formation and leads to the accumulation of extracellular matrix in the liver parenchyma.50 | It is the sequel of other phenotypes of CLD. | - |
ALD, alcoholic liver disease; CLD, chronic liver diseases; DILI, drug-induced liver injury; MAFLD, metabolic associated fatty liver disease; NAFLD, nonalcoholic fatty liver disease.
Table 2.
Diagnostic and Screening Criteria of Different Phenotypes of CLD
| DILI | The CIOMS has laid down the guidelines for the evaluation of the (1) hepatocellular injury, ALT ≥3 ULN and R ≥ 5; (2) cholestatic injury, alkaline phosphatase (ALP) ≥2 ULN and R ≤2; (3) and hepatocellular-cholestatic mixed injury, ALT ≥3 ULN, ALP ≥2 ULN and 2 < R < 5.24,51 The general diagnosis of DILI is primarily based on the elevation in serum ALT, ALP, GGT, and total bilirubin.26 Drug-induced autoimmune hepatitis diagnosis is based on serology, genetic test, and liver biopsy whenever possible whereas drug-induced secondary sclerosing cholangitis on MRCP or ERCP.23 |
| NAFLD/MAFLD | Liver biopsy is considered the gold standard for differentiating the phenotypes of NAFLD/MAFLD (NAFL and NASH) and their progression (fibrosis and cirrhosis),32,34 nonetheless, noninvasive preliminary diagnostic evaluation includes imaging to determine steatosis and evaluation of conventional liver biochemistry, in addition to fasting blood glucose, total blood count, hemoglobin A1c, and OGTT.10,21 To assess the presence of steatosis, FLI, SteatoTest, and NAFLD liver fat score are determined.32,52 Cytokeratin-18 fragment biomarker is currently being used to assess the extent of inflammation.32,53 In case NAFLD and NASH progress to fibrosis, NAFLD fibrosis score, fibrosis 4 calculator, AST/ALT ratio index, ELF panel, Fibrometer, Fibrotest, Hepascore, NAFLD activity score, in conjunction with imaging techniques such as TE, MRE, and shear wave elastography are used.10,54 |
| In the case of normal-weight individuals with hepatic steatosis and not having type II diabetes, the diagnostic criteria include the presence of at least 2 of the following metabolic conditions: | |
| (1) risk factors identifying the metabolic syndromes (example specific cut-off points for the waist circumference, blood pressure ≥130/85 mm Hg, plasma triglycerides ≥150 mg/dL, plasma HDL-cholesterol <40 mg/dL for men and <50 mg/dL for women, prediabetes, that is, fasting glucose levels 100–125 mg/dL, or 2-h postload glucose levels 140–199 mg/dL, hemoglobin A1c 5.7%–6.4%). | |
| (2) homeostatic assessment, that is, determine the HOMA-IR score ≥2.5; | |
| (3) high plasma high sensitivity-C-reactive protein level >2 mg/L. | |
| Abdominal ultrasonography is usually performed in clinical practice; however, CAP determination using VCTE and MRS, or MRI-PDFF, are also used to quantify liver fat.44,55 The FLI ultrasonographic fatty liver indicator and APRI are scoring systems to determine steatohepatitis and fibrosis.44,42 LSM by VCTE is widely preferred over biopsy in the Asia–Pacific region.42 | |
| Furthermore, physicians also prefer to wait for at least 5 y to perform a liver biopsy postdiagnosis of elevated liver function tests; additionally, if the body mass index of the patient is ≥25 kg/m2, there is an increased risk of progression to NASH.10,21,56 | |
| ALD | The general diagnosis of ALD involves the use of a series of questionnaire.19,46 The screening procedure is either invasive, such as liver biopsy for evaluating the degree of steatosis and fibrosis; or noninvasive, such as TE or quantification of biological markers encompassing GGT, serum ALT, serum AST, MCV, and %CDT.19,48 In case of advanced fibrosis or cirrhosis, serum albumin, prothrombin time, INR, serum bilirubin levels, platelets, or white blood cell counts should be analyzed followed by endoscopy.48 |
| Liver fibrosis | The LSM and TE are noninvasive diagnostic tools for determining fibrosis.57 Other tests such as Fibrotest, Fibrometer, FIB-4, NFS, and ELF are also performed.48 |
ALD, alcoholic liver disease; ALP, alkaline phosphatase; ALT, alanine aminotransferase; APRI, AST-to-platelet ratio index; AST, aspartate aminotransferase; CAP, controlled attenuation parameter; CDT, carbohydrate-deficient transferrin; CIOMS, Council for International Organizations of Medical Sciences; CLD, chronic liver diseases; DILI, drug-induced liver injury; ELF, enhanced liver fibrosis; ERCP, endoscopic retrograde cholangiopancreatography; FIB-4, fibrosis-4, index; FLI, fatty liver index; GGT, gamma-glutamyl transpeptidase; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment-estimated insulin resistance; INR, international normalized ratio; LSM, liver stiffness measurement; MAFLD, metabolic associated fatty liver disease; MCV, mean corpuscular volume; MRCP, magnetic resonance cholangiopancreatography; MRE, magnetic resonance elastography; MRI, magnetic resonance imaging; MRI-PDFF, magnetic resonance imaging proton density fat fraction; MRS, magnetic resonance spectroscopy; NAFL, nonalcoholic fatty liver; NAFLD, nonalcoholic fatty liver disease; NASH, nonalcoholic steatohepatitis; NFS, NAFLD, fibrosis score; OGTT, oral glucose tolerance test; TE, transient elastography; ULN, upper limit of normal; VCTE, vibration-controlled transient elastography.
Drug-Induced Liver Injury
The accumulation of ROS, endoplasmic reticulum stress (ERS), depletion of adenosine triphosphate, and inhibition of mitochondrial respiratory chain contribute to DILI.58 Such exogenous hepatic insults beyond a critical threshold may lead to activation of c-Jun N-terminal kinase pathways in hepatocytes, leading to impaired antioxidant mechanisms and cell death.11,25,59,60 Of note, OS and mitochondrial stress change mitochondrial membrane permeability, leading to DNA damage and cellular toxicity.11 A major challenge associated with DILI is the recurrence of liver damage upon readministration of the drug previously causing DILI.60
Nonalcoholic Fatty Liver Disease and MAFLD
The accumulation of fat or triglycerides in hepatocytes due to increased uptake of fatty acids from abdominal adipose tissue, or increased de novo lipid synthesis, or reduced fatty acid oxidation, leads to excessive ROS generation and LPO.61, 62, 63 Notedly, malondialdehyde and 4-hydroxynonenal are the 2 toxic entities generated by LPO,64 which along with immune cells, endothelial cells, stellate cells, and Kupfer cells, may damage hepatocytes. Immune cells show specific receptors for such injured hepatocytes, such as damage-associated molecular patterns or pathogen-associated molecular patterns.65 Activation of these receptors triggers the synthesis and release of inflammatory and profibrogenic mediators in injured hepatocytes. The cellular and molecular cross-talk between stressed or damaged hepatocytes, activated macrophages, and stellate cells drives matrix protein deposition—leading to the synthesis of scar tissue in the liver.66 The 2-hit hypothesis suggests the first core hit to be peripheral insulin resistance (IR) accompanied by obesity and other metabolic syndromes. The second hit might be an imbalance between the generation of ROS and antioxidant mechanisms leading to LPO and mitochondrial dysfunction. The multiple parallel hits model suggests that many mechanisms might act simultaneously, resulting in hepatic inflammation and ERS.43,67, 68, 69 An imbalance between ROS production and antioxidant mechanisms inhibits the mitochondrial respiratory chain and membrane sodium channels.43 The excessive oxidants produced via renin-angiotensin hyperactivity, mitochondrial dysfunction, dyslipidemia, and ERS lead to diminished glucose transport in skeletal muscles, decreased insulin secretion, and vascular dysfunction via activation of stress-activated serine kinases.70 The circulation of excess fatty acids leads to fatty acid accumulation in liver and adipose tissue, causing IR.68
Alcoholic Liver Disease
Alcohol impairs lipid metabolism and antioxidant mechanisms and promotes fat accumulation either by inhibiting mitochondrial β-oxidation or enhancing fatty acid synthesis.19,47,71 The role of OS in fibrosis was confirmed in patients with ALD and NAFLD.72 This evidence suggests that alcohol acts as an external agent promoting toxic liver injury by disturbing the delicate balance between ROS generation and active antioxidant mechanisms.
Liver Fibrosis
Liver fibrosis is the intermediate, partially reversible serious sequel of pathologic change in patients with different phenotypes of CLD, including ALD, NAFLD/MAFLD, DILI, and viral infections, such as viral hepatitis.50 A meta-analysis conducted by Singh et al73 of paired liver biopsy studies clearly indicated that progression to hepatic fibrosis is much faster in patients with NASH compared with patients with NAFLD. Therefore, early diagnosis of these phenotypes can mitigate the risk of progression to cirrhosis and reduce hepatic toxicity pre-emptively.2
As the prevalence of CLD is growing, a higher burden of cirrhosis or liver failure and hepatocellular carcinoma remains imminent if no early interventions are initiated. As liver transplantation is a leading indication for MAFLD/NASH,74 it causes a huge burden on health care resources. Slowing the progression of TLD can reduce the socioeconomic burden, especially if treatment with a hepatoprotective agent targeting common underlying mechanisms is initiated early on.
Consensus Statement on the Toxic Liver Disease Spectrum
The expert panel reviewed the current evidence and acknowledged the extensive liver damage caused by diverse endogenous or exogenous toxic agents that leads to DILI, NAFLD/MAFLD, and ALD. The panel reached a consensus on the justification to bring the previously listed diseases under a unifying entity called “toxic liver diseases,” based on common mechanisms driven by diverse endogenous and exogenous hepatotoxic agents.
OS as a Common Underlying Mechanism and Significant in the Management of TLD
Of note, OS is one of the early key mediators of hepatic injury driving disease progression.34 Hepatocellular injury by ROS is due to obstruction of mitochondrial respiratory enzymes along with the deactivation of glyceraldehyde-3-phosphate dehydrogenase. The ROS produced lead to LPO and tissue damage, which activate cytokine generation by resident macrophages. Local soluble mediators and cytokines lead to the infiltration of more immune cells, which further enhances tissue OS and subsequent fibrosis.43,75 Overnutrition and lack of exercise cause the expansion of adipose tissue, resulting in the secretion of proinflammatory cytokines, resulting in steatosis and fibrosis.68 Macrophages/Kupffer cells secrete proinflammatory cytokines and undergo M1 or M2 activation and are implicated in the pathogenesis of steatosis and activation of fibrogenesis.68 The damage-associated molecular patterns that are released from damaged hepatocytes activate the intrinsic immune response, including the release of cytokines, such as tumor necrosis factor (TNF)-alpha, and in turn activate death receptors such as TNF-R.14 Hepatic stellate cells also contribute to the generation of excessive ROS.76
It has been shown that the antioxidant mechanism is compromised in DILI, leading to the development of intensified OS, which ultimately culminates in mitochondrial damage and cell death. Of note, DILI also leads to change in cell functionality, causing an augmentation in ROS levels or impeding detoxification mechanisms, paving the way for overall OS.14,77 In terms of NAFLD/MAFLD, lipotoxicity aggravates hepatic inflammation, leading to activation of abnormal adipokines causing hepatocyte stress.43 In addition, the polymorphism in superoxide dismutase 2 has been related to the severity of disease progression.78 In MAFLD/NAFLD, fat accumulation leads to IR and inflammation via the activation of Kupffer cells. Sirtuins79 and nuclear factor erythroid 2–related factor 2 redox balance regulators are also involved in oxidative damage in both ALD and NAFLD.16,80 In the case of ALD, OS and carcinogenic metabolites of alcohol and inflammation might aggravate DNA mutations leading to neoplastic alteration of hepatocytes to hepatocellular carcinoma.19 Preclinical studies have established the role of OS in LPO and in redox imbalance causing hepatic damage.81, 82, 83 Of note, the OS in DILI, ALD, and NAFLD/MAFLD evaluated in different animal models was driven by mitochondrial dysfunction, impairment of hepatic function, and inflammation.84, 85, 86 As OS impacts the normal functioning of the liver, there is an urgent need to mitigate OS by establishing a promising therapeutic approach.
Consensus Statement on the Role of OS
The expert panel reviewed current evidence and agreed that OS is a common underlying pathophysiological mechanism driving diverse clinical phenotypes grouped under TLDs. The panel reached a consensus that during the early stages of TLDs, targeting OS in hepatocytes with a hepatoprotective agent may be clinically effective.
Current Treatment Approaches
The panel evaluated available treatment options and the extent to which they target OS. The therapeutic strategy is to treat the liver condition along with the associated metabolic syndrome.34 Currently, the mainstay of treatment is intensive lifestyle modification, including diet, exercise, weight loss, and behavioral therapy in patients with TLD including NAFLD/MAFLD and ALD.34,87 For DILI, the drug or causative agent should be discontinued.23 No specific pharmacological agent has been approved to treat NAFLD/MAFLD. Orlistat, bariatric surgery, angiotensin receptor blockers, statins, fibrates, antidiabetic drugs, glucagon-like peptide-1, and thiazolidinediones might be considered personalized treatments for NAFLD/MAFLD.44, 87, 88 Based on limited data, certain relapse-prevention medications (acamprosate, baclofen) are being considered in ALD.46 The EASL guidelines have established the use of N-acetylcysteine, corticosteroids, granulocyte-colony-stimulating factor, pentoxifylline, and anti-TNF agents for ALD.48 Certain studies have also revealed the beneficial aspects of nutritional therapy for liver diseases.46 As reversing NASH and liver fibrosis with existing treatment modalities is difficult, it becomes imperative to protect the liver from OS, inflammation, and fibrosis using a hepatoprotective agent. Vitamin E has been used as a hepatoprotective agent; however, its long-term safety is questionable.89 No such agent is available that can offer a synergistic approach in treating the different phenotypes of TLD by targeting OS. Lifestyle modification,90 correcting metabolic syndrome,91,92 exercise, diet, weight loss,93,94 and the use of antioxidants can target OS. Early intervention with antioxidant therapy can prevent or improve TLD.
Consensus Statement on Treatment Options
The panel, after reviewing the evidence, recognized the need for early interventions that can counter OS for delaying the onset or slowing the progression of TLD.
Silymarin as a Therapeutic
The panel evaluated the antioxidant properties of silymarin and its potential use in TLD. Silymarin derived from the dried seeds and fruits of Silybum marianum has been used as a natural herbal product for centuries because of its flavonolignan content (silybin is the most biologically active).22,95,96 The hydrophobic nature of crude silymarin extract impacts oral bioavailability, which necessitates the inclusion of solubilizing agents in formulations.21,97 Rapid and extensive hepatic biotransformation and enterohepatic circulation followed by biliary excretion have been reported for silymarin.21,22,97 Bioavailability and potential of silymarin accumulation in the liver and plasma in physiological amounts after oral administration have been demonstrated.73 The phenolic group in silymarin interacts with the ROS generated during hepatic metabolism to form a stable, nontoxic compound. Silymarin acts as a free-radical scavenger, reduces OS, and works by modulating liver function enzymes. Silymarin inhibits the activation of fibrogenic stellate cells, restricts the release of proinflammatory mediators by hepatic macrophages, and provides clinical benefit to the injured liver.21,97 Various preclinical and in vitro studies have established silymarin as an anti-inflammatory, antifibrotic, antioxidant agent, and modulator of IR,98, 99, 100, 101, 102, 103 establishing the basis for silymarin as a hepatoprotective agent with clinical potential.22 Moreover, silymarin possesses low toxicity and is devoid of any embryotoxic potential.104
Clinical Evidence Supporting the Use of Silymarin
In patients treated with silymarin, a statistically significant reduction in serum alanine aminotransferase and aspartate aminotransferase levels was reported in a meta-analysis conducted by Avelar et al.105 Similarly, Saller et al106 reviewed studies that suggested a supportive role of silymarin in treating liver cirrhosis. The consensus formulated by Cheng Jun et al. also advocates the use of silymarin as a potential hepatoprotective agent.107 In patients receiving long-term drug treatment, levels of alanine aminotransferase, aspartate aminotransferase, gamma-glutamyl transpeptidase, alanine phosphatase, total bilirubin, along with the quality of life, are improved after 2 months of treatment with silymarin.108 When used prophylactically, silymarin also reduced the risk of DILI and improved LFT in patients receiving anti-TB drugs109 with a concomitant reduction in SOD decline, suggesting a contribution of antioxidant properties driving risk reduction as evident in a double-blind randomized placebo-controlled trial on patients with DILI.110 Notably, an improvement in 4-year survival was observed in patients with ALD and cirrhosis at a dose of 140 mg 3 times a day.111 Silymarin treatment was effective in lowering elevated liver enzymes in patients with ALD and cirrhosis as established from a double-blind controlled trial112 and in patients with NAFLD.113,114 A randomized double-blinded, placebo-controlled trial on patients with NAFLD suggested significant benefits from the use of silymarin in the amelioration of liver aminotransferases without specific side effects.115 In a randomized double-blind placebo-controlled multicenter Phase II trial, standardized 420 mg and 720 mg silymarin preparations were tested on patients with NASH without cirrhosis, wherein histologic improvement ≥2 points in NAFLD activity score were observed in silymarin-treated groups.116 Similarly, randomized trials and double-blind trials on patients with NAFLD and chronic ALD, respectively, using silymarin-expressed restoration of SOD levels in the lymphocytes and erythrocytes117 decreased gamma-glutamyl transpeptidase levels, fatty liver index, and NAFLD-FS indices118 and significant improvement in metabolic markers, OS, endothelial dysfunction, and worsening of disease.119
Higher-than-recommended doses of silymarin have also been used and demonstrated to be safe and tolerable as established in a randomized, double-blind, placebo-controlled trial. The use of 700 mg/d silymarin for 48 weeks in patients with NASH exhibited a significant reduction in fibrosis and improvement in liver biochemistry.120 Similar beneficial effects of silymarin are observed at higher doses in patients with alcoholic cirrhosis121 and in improving the associated metabolic endpoints and IR in patients with cirrhotic diabetes.122 The lower dose of silymarin (<140 mg 3 times a day) also caused a notable decrease in liver enzymes either as a single formulation123 or as a combinatorial approach in improving liver damage.124,125 Overall, based on all clinical evidence, silymarin was found to be safe and well tolerated; thus, it can be considered a beneficial treatment for TLD.
Silymarin as a Treatment for TLD
The key target for TLD is OS, and because silymarin is safe, well tolerated, and has limited interactions with other drugs, its antioxidant properties can be leveraged clinically. Silymarin decreases OS and may be an effective treatment option for patients with TLD. As per accumulating clinical evidence, silymarin can be used as an adjunct or supportive therapy during the early stages of TLD. In the case of DILI, where there are limited treatment options, silymarin may be safer than corticosteroids, which have several adverse effects.24 The Chinese Society of Hepatology and the Chinese guidelines for ALD also recommend the use of silymarin in mild liver inflammation, as an antioxidant, antifibrotic, and hepatoprotective agent.126,57 It is the opinion of the panel of experts that patients with fatty liver with and without metabolic syndrome (especially younger patients with stronger OS), who have a higher risk of progressing to NASH and cirrhosis, are good candidates for early treatment with silymarin. As per current Chinese NAFLD guidelines, patients with NAFLD suspected of having NASH and progressive liver fibrosis may benefit from hepatoprotective drugs such as silymarin. Silymarin may be used as a hepatoprotective drug in NAFLD/MAFLD for at least 12 months if serum LFT has not decreased significantly after 6 months of treatment.89 Patients with chronic viral hepatitis may also benefit from improved liver function, leveraging the hepatoprotective properties of silymarin, without affecting antiviral efficacy.107 A synergistic approach is usually preferred where silymarin is used along with lifestyle modification, providing a requisite motivational drive to patients to be compliant with therapy. The tailored formulation of silymarin needs to be standardized as per good manufacturing practice and herbal standards for ensuring the reliability and reproducibility of clinical outcomes. Therefore, the most common dosage for silymarin as established in the literature is 420 mg/d; however, it lies within the range of 140 mg/d (the lower level) to 1080 mg/d (the highest level), preferably twice or thrice daily.105
Consensus Statement on Silymarin
The expert panel agreed on the safety, tolerability, and clinical benefits of silymarin as an antioxidant, when initiated as early treatment and used for prolonged durations with careful monitoring, and as a pharmacological tool in the management of TLDs either alone or concomitantly with other medications.
Conclusion
As the burden of TLD is increasing, it is critical to develop treatments that can target key pathophysiological mechanisms. OS as a common target can bring together NAFLD/MAFLD, ALD, DILI, NASH, and liver cirrhosis under the single basket entity of TLD and can be managed with an antioxidant. Silymarin as an antioxidant and hepatoprotective agent is safe and has reasonable clinical efficacy in TLD, as per the label. Hence, silymarin treatment can be initiated as early as possible, continued for at least 3–6 months or longer with periodic monitoring of liver function, alongside other pharmacological and nonpharmacological interventions, for TLD.
Acknowledgments
Authors' Contributions:
All authors contributed equally toward study conception and design, analysis, interpretation of data, drafting of article, and revising it critically for intellectual content. All authors were involved in the final approval for submission and agree to be accountable for all aspects of the work.
Footnotes
Conflicts of Interest: The authors disclose no conflicts.
Funding: No funding was received by any of the authors for the development of this publication. The medical writing and editing for this manuscript were supported by Viatris.
Ethical Statement: The study did not require the approval of an institutional review board.
Writing Assistance: The authors wish to thank Bushra Nabi and Lakshman Puli of IQVIA, India, for their writing and editing support.
Contributor Information
Anton Gillessen, Email: anton.gillessen@rub.de.
Xiaoyuan Xu, Email: xiaoyuanxu6@163.com.
References
- 1.James S.L., Abate D., Abate K.H., et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789–1858. doi: 10.1016/S0140-6736(18)32279-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu X., Xu J. Calling a stage-based treatment model for chronic liver diseases in China mainland. Ann Hepatol. 2020;19:585–589. doi: 10.1016/j.aohep.2019.09.007. [DOI] [PubMed] [Google Scholar]
- 3.Paik J.M., Golabi P., Younossi Y., et al. Changes in the global burden of chronic liver diseases from 2012 to 2017: the growing impact of NAFLD. Hepatology. 2020;72:1605–1616. doi: 10.1002/hep.31173. [DOI] [PubMed] [Google Scholar]
- 4.Asrani S.K., Devarbhavi H., Eaton J., et al. Burden of liver diseases in the world. J Hepatol. 2019;70:151–171. doi: 10.1016/j.jhep.2018.09.014. [DOI] [PubMed] [Google Scholar]
- 5.Jaeschke H., Gores G.J., Cederbaum A.I., et al. Mechanisms of hepatotoxicity. Toxicol Sci. 2002;65:166–176. doi: 10.1093/toxsci/65.2.166. [DOI] [PubMed] [Google Scholar]
- 6.Valeeva E.T., Mukhammadiyeva G.F., Bakirov A.B. Polymorphism of glutathione S-transferase genes and the risk of toxic liver damage in petrochemical workers. Int J Occup Environ Med. 2020;11:53–58. doi: 10.15171/ijoem.2020.1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Targher G., Byrne C.D. From nonalcoholic fatty liver disease to metabolic dysfunction-associated fatty liver disease: is it time for a change of terminology? Hepatoma Res. 2020;6:64. [Google Scholar]
- 8.Wahlang B., Beier J.I., Clair H.B., et al. Toxicant-associated steatohepatitis. Toxicol Pathol. 2013;41:343–360. doi: 10.1177/0192623312468517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Armstrong L.E., Guo G.L. Understanding environmental contaminants' direct effects on non-alcoholic fatty liver disease progression. Curr Environ Health Rep. 2019;6:95–104. doi: 10.1007/s40572-019-00231-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Leoni S., Tovoli F., Napoli L., et al. Current guidelines for the management of non-alcoholic fatty liver disease: a systematic review with comparative analysis. World J Gastroenterol. 2018;24:3361–3373. doi: 10.3748/wjg.v24.i30.3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ye H., Nelson L.J., Gomez Del Moral M., et al. Dissecting the molecular pathophysiology of drug-induced liver injury. World J Gastroenterol. 2018;24:1373–1385. doi: 10.3748/wjg.v24.i13.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sepanlou S.G., Safiri S., Bisignano C., et al. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5:245–266. doi: 10.1016/S2468-1253(19)30349-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cichoz-Lach H., Michalak A. Oxidative stress as a crucial factor in liver diseases. World J Gastroenterol. 2014;20:8082–8091. doi: 10.3748/wjg.v20.i25.8082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Villanueva-Paz M., Morán L., López-Alcántara N., et al. Oxidative stress in drug-induced liver injury (DILI): from mechanisms to biomarkers for use in clinical practice. Antioxidants (Basel) 2021;10:390. doi: 10.3390/antiox10030390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vuppalanchi R., Juluri R., Bell L.N., et al. Oxidative stress in chronic liver disease: relationship between peripheral and hepatic measurements. Am J Med Sci. 2011;342:314–317. doi: 10.1097/MAJ.0b013e31821d9905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li S., Tan H.Y., Wang N., et al. The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci. 2015;16:26087–26124. doi: 10.3390/ijms161125942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Boyer-Diaz Z., Morata P., Aristu-Zabalza P., et al. Oxidative stress in chronic liver disease and portal hypertension: potential of DHA as nutraceutical. Nutrients. 2020;12:2627. doi: 10.3390/nu12092627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.2020. Drug-induced liver injury (DILI): Current status and future directions for drug development and the post-market setting. A consensus by a CIOMS Working Group. Geneva, Switzerland: Council for International Organizations of Medical Sciences (CIOMS), [Google Scholar]
- 19.Avila M.A., Dufour J.F., Gerbes A.L., et al. Recent advances in alcohol-related liver disease (ALD): summary of a Gut round table meeting. Gut. 2020;69:764–780. doi: 10.1136/gutjnl-2019-319720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Polimeni L., Del Ben M., Baratta F., et al. Oxidative stress: new insights on the association of non-alcoholic fatty liver disease and atherosclerosis. World J Hepatol. 2015;7:1325–1336. doi: 10.4254/wjh.v7.i10.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hashem A., Shastri Y., Al Otaibi M., et al. Expert opinion on the management of non-alcoholic fatty liver disease (NAFLD) in the Middle East with a focus on the use of silymarin. Gastroenterol Insights. 2021;12:155–165. [Google Scholar]
- 22.Gillessen A., Schmidt H.H. Silymarin as supportive treatment in liver diseases: a narrative review. Adv Ther. 2020;37:1279–1301. doi: 10.1007/s12325-020-01251-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu; Clinical Practice Guideline Panel: Chair; Panel Members. EASL clinical practice guidelines: drug-induced liver injury. J Hepatol 2019;70:1222–1261. [DOI] [PubMed]
- 24.Yu Y.C., Mao Y.M., Chen C.W., et al. CSH guidelines for the diagnosis and treatment of drug-induced liver injury. Hepatol Int. 2017;11:221–241. doi: 10.1007/s12072-017-9793-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shen T., Liu Y., Shang J., et al. Incidence and etiology of drug-induced liver injury in mainland China. Gastroenterology. 2019;156:2230–2241.e11. doi: 10.1053/j.gastro.2019.02.002. [DOI] [PubMed] [Google Scholar]
- 26.Chen Y., Wang C., Yang H., et al. Epidemiology of drug- and herb-induced liver injury assessed for causality using the updated RUCAM in two hospitals from China. Biomed Res Int. 2021;2021:8894498. doi: 10.1155/2021/8894498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wai C.T., Tan B.H., Chan C.L., et al. Drug-induced liver injury at an Asian center: a prospective study. Liver Int. 2007;27:465–474. doi: 10.1111/j.1478-3231.2007.01461.x. [DOI] [PubMed] [Google Scholar]
- 28.Liu Y., Zhan S.P., Song L., et al. Drug-induced liver injury: clinical and etiologic features at a large tertiary teaching hospital in China. Med Sci Monit. 2020;26 doi: 10.12659/MSM.919435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Biswas A., Santra S., Bishnu D., et al. Isoniazid and rifampicin produce hepatic fibrosis through an oxidative stress-dependent mechanism. Int J Hepatol. 2020;2020 doi: 10.1155/2020/6987295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hayashi P.H., Fontana R.J. Clinical features, diagnosis, and natural history of drug-induced liver injury. Semin Liver Dis. 2014;34:134–144. doi: 10.1055/s-0034-1375955. [DOI] [PubMed] [Google Scholar]
- 31.Brown G.T., Kleiner D.E. Histopathology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Metabolism. 2016;65:1080–1086. doi: 10.1016/j.metabol.2015.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.European Association for the Study of the Liver (EASL) European Association for the Study of Diabetes (EASD) European Association for the Study of Obesity (EASO) EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388–1402. doi: 10.1016/j.jhep.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 33.Cotter T.G., Rinella M. Nonalcoholic fatty liver disease 2020: the state of the disease. Gastroenterology. 2020;158:1851–1864. doi: 10.1053/j.gastro.2020.01.052. [DOI] [PubMed] [Google Scholar]
- 34.Chalasani N., Younossi Z., Lavine J.E., et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–357. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 35.Sarin S.K., Kumar M., Eslam M., et al. Liver diseases in the Asia-Pacific region: a Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol Hepatol. 2020;5:167–228. doi: 10.1016/S2468-1253(19)30342-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Estes C., Anstee Q.M., Arias-Loste M.T., et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016-2030. J Hepatol. 2018;69:896–904. doi: 10.1016/j.jhep.2018.05.036. [DOI] [PubMed] [Google Scholar]
- 37.Bedossa P., Poitou C., Veyrie N., et al. Histopathological algorithm and scoring system for evaluation of liver lesions in morbidly obese patients. Hepatology. 2012;56:1751–1759. doi: 10.1002/hep.25889. [DOI] [PubMed] [Google Scholar]
- 38.Bedossa P., Consortium F.P. Utility and appropriateness of the fatty liver inhibition of progression (FLIP) algorithm and steatosis, activity, and fibrosis (SAF) score in the evaluation of biopsies of nonalcoholic fatty liver disease. Hepatology. 2014;60:565–575. doi: 10.1002/hep.27173. [DOI] [PubMed] [Google Scholar]
- 39.Ge X., Zheng L., Wang M., et al. Prevalence trends in non-alcoholic fatty liver disease at the global, regional and national levels, 1990-2017: a population-based observational study. BMJ Open. 2020;10 doi: 10.1136/bmjopen-2019-036663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu J., Ayada I., Zhang X., et al. Estimating global prevalence of metabolic dysfunction-associated fatty liver disease in overweight or obese adults. Clin Gastroenterol Hepatol. 2022;20:e573–e582. doi: 10.1016/j.cgh.2021.02.030. [DOI] [PubMed] [Google Scholar]
- 41.Lazebnik L.B., Radchenko V.G., Golovanova Е.V., et al. Non-alcoholic fatty liver disease: clinic, diagnostics, treatment. Exp Clin Gastroenterol. 2017;2:22–37. [Google Scholar]
- 42.Eslam M., Newsome P.N., Sarin S.K., et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol. 2020;73:202–209. doi: 10.1016/j.jhep.2020.03.039. [DOI] [PubMed] [Google Scholar]
- 43.Yang J., Fernandez-Galilea M., Martinez-Fernandez L., et al. Oxidative stress and non-alcoholic fatty liver disease: effects of omega-3 fatty acid supplementation. Nutrients. 2019;11:872. doi: 10.3390/nu11040872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Eslam M., Sarin S.K., Wong V.W., et al. The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol Int. 2020;14:889–919. doi: 10.1007/s12072-020-10094-2. [DOI] [PubMed] [Google Scholar]
- 45.Gao Y.G.B., Niu J. The emerging alcoholic liver disease in China. North Am J Med Sci. 2016;9:55–58. [Google Scholar]
- 46.Crabb D.W., Im G.Y., Szabo G., et al. Diagnosis and treatment of alcohol-associated liver diseases: 2019 practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2020;71:306–333. doi: 10.1002/hep.30866. [DOI] [PubMed] [Google Scholar]
- 47.Seitz H.K., Bataller R., Cortez-Pinto H., et al. Alcoholic liver disease. Nat Rev Dis Primers. 2018;4:16. doi: 10.1038/s41572-018-0014-7. [DOI] [PubMed] [Google Scholar]
- 48.European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu; European Association for the Study of the Liver. EASL clinical practice guidelines: management of alcohol-related liver disease. J Hepatol 2018;69:154–181. [DOI] [PubMed]
- 49.Shah N.D., Ventura-Cots M., Abraldes J.G., et al. Alcohol-related liver disease is rarely detected at early stages compared with liver diseases of other etiologies worldwide. Clin Gastroenterol Hepatol. 2019;17:2320–2329.e12. doi: 10.1016/j.cgh.2019.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sánchez-Valle V., C Chavez-Tapia N., Uribe M., et al. Role of oxidative stress and molecular changes in liver fibrosis: a review. Curr Med Chem. 2012;19:4850–4860. doi: 10.2174/092986712803341520. [DOI] [PubMed] [Google Scholar]
- 51.Chalasani N.P., Maddur H., Russo M.W., et al. ACG clinical guideline: diagnosis and management of idiosyncratic drug-induced liver injury. Am J Gastroenterol. 2021;116:878–898. doi: 10.14309/ajg.0000000000001259. [DOI] [PubMed] [Google Scholar]
- 52.Fedchuk L., Nascimbeni F., Pais R., et al. Performance and limitations of steatosis biomarkers in patients with nonalcoholic fatty liver disease. Aliment Pharmacol Ther. 2014;40:1209–1222. doi: 10.1111/apt.12963. [DOI] [PubMed] [Google Scholar]
- 53.Cusi K., Chang Z., Harrison S., et al. Limited value of plasma cytokeratin-18 as a biomarker for NASH and fibrosis in patients with non-alcoholic fatty liver disease. J Hepatol. 2014;60:167–174. doi: 10.1016/j.jhep.2013.07.042. [DOI] [PubMed] [Google Scholar]
- 54.Kaswala D.H., Lai M., Afdhal N.H. Fibrosis assessment in nonalcoholic fatty liver disease (NAFLD) in 2016. Dig Dis Sci. 2016;61:1356–1364. doi: 10.1007/s10620-016-4079-4. [DOI] [PubMed] [Google Scholar]
- 55.Hernaez R., Lazo M., Bonekamp S., et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology. 2011;54:1082–1090. doi: 10.1002/hep.24452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kwok R., Choi K.C., Wong G.L., et al. Screening diabetic patients for non-alcoholic fatty liver disease with controlled attenuation parameter and liver stiffness measurements: a prospective cohort study. Gut. 2016;65:1359–1368. doi: 10.1136/gutjnl-2015-309265. [DOI] [PubMed] [Google Scholar]
- 57.Xu X.Y., Ding H.G., Li W.G., et al. Chinese guidelines on the management of liver cirrhosis (abbreviated version) World J Gastroenterol. 2020;26:7088–7103. doi: 10.3748/wjg.v26.i45.7088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.David S., Hamilton J.P. Drug-induced liver injury. US Gastroenterol Hepatol Rev. 2010;6:73–80. [PMC free article] [PubMed] [Google Scholar]
- 59.Han D., Dara L., Win S., et al. Regulation of drug-induced liver injury by signal transduction pathways: critical role of mitochondria. Trends Pharmacol Sci. 2013;34:243–253. doi: 10.1016/j.tips.2013.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Garcia-Cortes M., Robles-Diaz M., Stephens C., et al. Drug induced liver injury: an update. Arch Toxicol. 2020;94:3381–3407. doi: 10.1007/s00204-020-02885-1. [DOI] [PubMed] [Google Scholar]
- 61.Koo S.H. Nonalcoholic fatty liver disease: molecular mechanisms for the hepatic steatosis. Clin Mol Hepatol. 2013;19:210–215. doi: 10.3350/cmh.2013.19.3.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Macdonald G.A., Bridle K.R., Ward P.J., et al. Lipid peroxidation in hepatic steatosis in humans is associated with hepatic fibrosis and occurs predominately in acinar zone 3. J Gastroenterol Hepatol. 2001;16:599–606. doi: 10.1046/j.1440-1746.2001.02445.x. [DOI] [PubMed] [Google Scholar]
- 63.George J., Pera N., Phung N., et al. Lipid peroxidation, stellate cell activation and hepatic fibrogenesis in a rat model of chronic steatohepatitis. J Hepatol. 2003;39:756–764. doi: 10.1016/s0168-8278(03)00376-3. [DOI] [PubMed] [Google Scholar]
- 64.Ayala A., Muñoz M.F., Argüelles S. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev. 2014;2014:360438. doi: 10.1155/2014/360438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Heymann F., Tacke F. Immunology in the liver--from homeostasis to disease. Nat Rev Gastroenterol Hepatol. 2016;13:88–110. doi: 10.1038/nrgastro.2015.200. [DOI] [PubMed] [Google Scholar]
- 66.Tacke F., Weiskirchen R. Non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH)-related liver fibrosis: mechanisms, treatment and prevention. Ann Transl Med. 2020;9:729. doi: 10.21037/atm-20-4354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tilg H., Moschen A.R. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology. 2010;52:1836–1846. doi: 10.1002/hep.24001. [DOI] [PubMed] [Google Scholar]
- 68.Kitade H., Chen G., Ni Y., et al. Nonalcoholic fatty liver disease and insulin resistance: new insights and potential new treatments. Nutrients. 2017;9:387. doi: 10.3390/nu9040387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Xian Y.X., Weng J.P., Xu F. MAFLD vs. NAFLD: shared features and potential changes in epidemiology, pathophysiology, diagnosis, and pharmacotherapy. Chin Med J (Engl) 2020;134:8–19. doi: 10.1097/CM9.0000000000001263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Henriksen E.J. In: Bioactive food as dietary interventions for diabetes. 2nd ed. Watson R.R., Preedy V.R., editors. Academic Press; 2019. Chapter 1 - role of oxidative stress in the pathogenesis of insulin resistance and type 2 diabetes; pp. 3–17. [Google Scholar]
- 71.Shinde A., Ganu J., Naik P., et al. Oxidative stress and antioxidative status in patients with alcoholic liver disease. Biomed Res. 2012;23:105–108. [Google Scholar]
- 72.Das K.S., Balakrishnan V., Mukherjee S., et al. Evaluation of blood oxidative stress-related parameters in alcoholic liver disease and non-alcoholic fatty liver disease. Scand J Clin Lab Invest. 2008;68:323–334. doi: 10.1080/00365510701673383. [DOI] [PubMed] [Google Scholar]
- 73.Singh S., Allen A.M., Wang Z., et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol. 2015;13:643–654.e1-9. doi: 10.1016/j.cgh.2014.04.014. quiz e39-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mathurin P., Lucey M.R. Liver transplantation in patients with alcohol-related liver disease: current status and future directions. Lancet Gastroenterol Hepatol. 2020;5:507–514. doi: 10.1016/S2468-1253(19)30451-0. [DOI] [PubMed] [Google Scholar]
- 75.Novo E., Busletta C., Bonzo L.V., et al. Intracellular reactive oxygen species are required for directional migration of resident and bone marrow-derived hepatic pro-fibrogenic cells. J Hepatol. 2011;54:964–974. doi: 10.1016/j.jhep.2010.09.022. [DOI] [PubMed] [Google Scholar]
- 76.Luangmonkong T., Suriguga S., Mutsaers H.A.M., et al. Targeting oxidative stress for the treatment of liver fibrosis. Rev Physiol Biochem Pharmacol. 2018;175:71–102. doi: 10.1007/112_2018_10. [DOI] [PubMed] [Google Scholar]
- 77.Iorga A., Dara L., Kaplowitz N. Drug-induced liver injury: cascade of events leading to cell death, apoptosis or necrosis. Int J Mol Sci. 2017;18:1018. doi: 10.3390/ijms18051018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Nobili V., Donati B., Panera N., et al. A 4-polymorphism risk score predicts steatohepatitis in children with nonalcoholic fatty liver disease. J Pediatr Gastroenterol Nutr. 2014;58:632–636. doi: 10.1097/MPG.0000000000000279. [DOI] [PubMed] [Google Scholar]
- 79.Ding R.B., Bao J., Deng C.X. Emerging roles of SIRT1 in fatty liver diseases. Int J Biol Sci. 2017;13:852–867. doi: 10.7150/ijbs.19370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang Y.K., Wu K.C., Klaassen C.D. Genetic activation of Nrf2 protects against fasting-induced oxidative stress in livers of mice. PLoS One. 2013;8 doi: 10.1371/journal.pone.0059122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Korolczuk A., Caban K., Amarowicz M., et al. Oxidative stress and liver morphology in experimental cyclosporine A-induced hepatotoxicity. Biomed Res Int. 2016;2016:5823271. doi: 10.1155/2016/5823271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lee T.Y., Chang H.H., Lo W.C., et al. Alleviation of hepatic oxidative stress by Chinese herbal medicine Yin-Chen-Hao-Tang in obese mice with steatosis. Int J Mol Med. 2010;25:837–844. doi: 10.3892/ijmm_00000412. [DOI] [PubMed] [Google Scholar]
- 83.Kamimura S., Gaal K., Britton R.S., et al. Increased 4-hydroxynonenal levels in experimental alcoholic liver disease: association of lipid peroxidation with liver fibrogenesis. Hepatology. 1992;16:448–453. doi: 10.1002/hep.1840160225. [DOI] [PubMed] [Google Scholar]
- 84.Zhao Q.Y., Ge L.H., Zhang K., et al. Assessment of mitochondrial function in metabolic dysfunction-associated fatty liver disease using obese mouse models. Zool Res. 2020;41:539–551. doi: 10.24272/j.issn.2095-8137.2020.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Roghani M., Kalantari H., Khodayar M.J., et al. Alleviation of liver dysfunction, oxidative stress and inflammation underlies the protective effect of ferulic acid in methotrexate-induced hepatotoxicity. Drug Des Devel Ther. 2020;14:1933–1941. doi: 10.2147/DDDT.S237107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kolota A., Glabska D., Oczkowski M., et al. Oxidative stress parameters in the liver of growing male rats receiving various alcoholic beverages. Nutrients. 2020;12:158. doi: 10.3390/nu12010158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mehta S.R. Advances in the treatment of nonalcoholic fatty liver disease. Ther Adv Endocrinol Metab. 2010;1:101–115. doi: 10.1177/2042018810379587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Katsiki N., Mikhailidis D.P., Mantzoros C.S. Non-alcoholic fatty liver disease and dyslipidemia: an update. Metabolism. 2016;65:1109–1123. doi: 10.1016/j.metabol.2016.05.003. [DOI] [PubMed] [Google Scholar]
- 89.National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association. Fatty Liver Expert Committee, Chinese Medical Doctor Association Guideline of prevention and treatment for nonalcoholic fatty liver disease: a 2018 update. Mod Med Health. 2019;20:163–173. [Google Scholar]
- 90.Yadav R.K., Ray R.B., Vempati R., et al. Effect of a comprehensive yoga-based lifestyle modification program on lipid peroxidation. Indian J Physiol Pharmacol. 2005;49:358–362. [PubMed] [Google Scholar]
- 91.Roberts C.K., Sindhu K.K. Oxidative stress and metabolic syndrome. Life Sci. 2009;84:705–712. doi: 10.1016/j.lfs.2009.02.026. [DOI] [PubMed] [Google Scholar]
- 92.Devaraj S., Wang-Polagruto J., Polagruto J., et al. High-fat, energy-dense, fast-food-style breakfast results in an increase in oxidative stress in metabolic syndrome. Metabolism. 2008;57:867–870. doi: 10.1016/j.metabol.2008.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rector R.S., Warner S.O., Liu Y., et al. Exercise and diet induced weight loss improves measures of oxidative stress and insulin sensitivity in adults with characteristics of the metabolic syndrome. Am J Physiol Endocrinol Metab. 2007;293:E500–E506. doi: 10.1152/ajpendo.00116.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Roberts C.K., Won D., Pruthi S., et al. Effect of a short-term diet and exercise intervention on oxidative stress, inflammation, MMP-9, and monocyte chemotactic activity in men with metabolic syndrome factors. J Appl Physiol (1985) 2006;100:1657–1665. doi: 10.1152/japplphysiol.01292.2005. [DOI] [PubMed] [Google Scholar]
- 95.Javed S., Kohli K., Ali M. Reassessing bioavailability of silymarin. Altern Med Rev. 2011;16:239. [PubMed] [Google Scholar]
- 96.Abenavoli L., Capasso R., Milic N., et al. Milk thistle in liver diseases: past, present, future. Phytother Res. 2010;24:1423–1432. doi: 10.1002/ptr.3207. [DOI] [PubMed] [Google Scholar]
- 97.Bijak M. Silybin, a major bioactive component of milk thistle (Silybum marianum L. Gaernt.)-chemistry, bioavailability, and metabolism. Molecules. 2017;22 doi: 10.3390/molecules22111942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Dehmlow C., Erhard J., de Groot H. Inhibition of Kupffer cell functions as an explanation for the hepatoprotective properties of silibinin. Hepatology. 1996;23:749–754. doi: 10.1053/jhep.1996.v23.pm0008666328. [DOI] [PubMed] [Google Scholar]
- 99.Li C.C., Hsiang C.Y., Wu S.L., et al. Identification of novel mechanisms of silymarin on the carbon tetrachloride-induced liver fibrosis in mice by nuclear factor-kappaB bioluminescent imaging-guided transcriptomic analysis. Food Chem Toxicol. 2012;50:1568–1575. doi: 10.1016/j.fct.2012.02.025. [DOI] [PubMed] [Google Scholar]
- 100.Kim M., Yang S.G., Kim J.M., et al. Silymarin suppresses hepatic stellate cell activation in a dietary rat model of non-alcoholic steatohepatitis: analysis of isolated hepatic stellate cells. Int J Mol Med. 2012;30:473–479. doi: 10.3892/ijmm.2012.1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yao J., Zhi M., Gao X., et al. Effect and the probable mechanisms of silibinin in regulating insulin resistance in the liver of rats with non-alcoholic fatty liver. Braz J Med Biol Res. 2013;46:270–277. doi: 10.1590/1414-431X20122551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Jia J.-D., Bauer M., Cho J.J., et al. Antifibrotic effect of silymarin in rat secondary biliary fibrosis is mediated by downregulation of procollagen α1 (I) and TIMP-1. J Hepatol. 2001;35:392–398. doi: 10.1016/s0168-8278(01)00148-9. [DOI] [PubMed] [Google Scholar]
- 103.Gharagozloo M., Velardi E., Bruscoli S., et al. Silymarin suppress CD4+ T cell activation and proliferation: effects on NF-kappaB activity and IL-2 production. Pharmacol Res. 2010;61:405–409. doi: 10.1016/j.phrs.2009.12.017. [DOI] [PubMed] [Google Scholar]
- 104.Fraschini F., Demartini G., Esposti D. Pharmacology of silymarin. Clin Drug Invest. 2002;22:51–65. [Google Scholar]
- 105.de Avelar C.R., Pereira E.M., de Farias Costa P.R., et al. Effect of silymarin on biochemical indicators in patients with liver disease: systematic review with meta-analysis. World J Gastroenterol. 2017;23:5004–5017. doi: 10.3748/wjg.v23.i27.5004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Saller R., Brignoli R., Melzer J., et al. An updated systematic review with meta-analysis for the clinical evidence of silymarin. Forsch Komplementmed. 2008;15:9–20. doi: 10.1159/000113648. [DOI] [PubMed] [Google Scholar]
- 107.Committee of Experts on Silymarin Preparation in Liver Diseases Experts consensus on silymarin preparation in liver diseases. Chin J Hepatol. 2016;8:5–9. [Google Scholar]
- 108.Gillessen A., Herrmann W.A., Kemper M., et al. [Effect of silymarin on liver health and quality of life. Results of a non-interventional study] MMW Fortschr Med. 2014;156 Suppl 4:120–126. doi: 10.1007/s15006-014-3758-3. [DOI] [PubMed] [Google Scholar]
- 109.Tao L., Qu X., Zhang Y., et al. Prophylactic therapy of silymarin (milk thistle) on antituberculosis drug-induced liver injury: a meta-analysis of randomized controlled trials. Can J Gastroenterol Hepatol. 2019;2019:3192351. doi: 10.1155/2019/3192351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Luangchosiri C., Thakkinstian A., Chitphuk S., et al. A double-blinded randomized controlled trial of silymarin for the prevention of antituberculosis drug-induced liver injury. BMC Complement Altern Med. 2015;15:334. doi: 10.1186/s12906-015-0861-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ferenci P., Dragosics B., Dittrich H., et al. Randomized controlled trial of silymarin treatment in patients with cirrhosis of the liver. J Hepatol. 1989;9:105–113. doi: 10.1016/0168-8278(89)90083-4. [DOI] [PubMed] [Google Scholar]
- 112.Salmi H., Sarna S. Effect of silymarin on chemical, functional, and morphological alterations of the liver: a double-blind controlled study. Scand J Gastroenterol. 1982;17:517–521. doi: 10.3109/00365528209182242. [DOI] [PubMed] [Google Scholar]
- 113.Hajiaghamohammadi A.A., Ziaee A., Oveisi S., et al. Effects of metformin, pioglitazone, and silymarin treatment on non-alcoholic fatty liver disease: a randomized controlled pilot study. Hepat Mon. 2012;12 doi: 10.5812/hepatmon.6099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hashemi S.J., Hajiani E., Heydari S.E. A placebo-controlled trial of silymarin in patients with nonalcoholic fatty liver disease. Hepat Mon. 2009;9:265–270. [Google Scholar]
- 115.Anushiravani A., Haddadi N., Pourfarmanbar M., et al. Treatment options for nonalcoholic fatty liver disease: a double-blinded randomized placebo-controlled trial. Eur J Gastroenterol Hepatol. 2019;31:613–617. doi: 10.1097/MEG.0000000000001369. [DOI] [PubMed] [Google Scholar]
- 116.Navarro V.J., Belle S.H., D'Amato M., et al. Silymarin in non-cirrhotics with non-alcoholic steatohepatitis: a randomized, double-blind, placebo controlled trial. PLoS One. 2019;14 doi: 10.1371/journal.pone.0221683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Müzes G., Deák G., Láng I., et al. [Effect of silimarin (Legalon) therapy on the antioxidant defense mechanism and lipid peroxidation in alcoholic liver disease (double blind protocol)] Orv Hetil. 1990;131:863–866. [PubMed] [Google Scholar]
- 118.Aller R., Izaola O., Gómez S., et al. Effect of silymarin plus vitamin E in patients with non-alcoholic fatty liver disease. A randomized clinical pilot study. Eur Rev Med Pharmacol Sci. 2015;19:3118–3124. [PubMed] [Google Scholar]
- 119.Federico A., Dallio M., Masarone M., et al. Evaluation of the effect derived from silybin with vitamin D and vitamin E administration on clinical, metabolic, endothelial dysfunction, oxidative stress parameters, and serological worsening markers in nonalcoholic fatty liver disease patients. Oxid Med Cell Longev. 2019;2019:8742075. doi: 10.1155/2019/8742075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wah Kheong C., Nik Mustapha N.R., Mahadeva S. A randomized trial of silymarin for the treatment of nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol. 2017;15:1940–1949.e8. doi: 10.1016/j.cgh.2017.04.016. [DOI] [PubMed] [Google Scholar]
- 121.Lucena M.I., Andrade R.J., de la Cruz J.P., et al. Effects of silymarin MZ-80 on oxidative stress in patients with alcoholic cirrhosis. Results of a randomized, double-blind, placebo-controlled clinical study. Int J Clin Pharmacol Ther. 2002;40:2–8. doi: 10.5414/cpp40002. [DOI] [PubMed] [Google Scholar]
- 122.Velussi M., Cernigoi A.M., Dapas F., et al. Long-term (23 months) treatment with an anti-oxidant drug (silymarin) is effective on hyperinsulinemia, exogenous insulin need and malondialdehyde levels in cirrhotic diabetic patients. J Hepatol. 1997;26:871–879. doi: 10.1016/s0168-8278(97)80255-3. [DOI] [PubMed] [Google Scholar]
- 123.Solhi H., Ghahremani R., Kazemifar A.M., et al. Silymarin in treatment of non-alcoholic steatohepatitis: a randomized clinical trial. Caspian J Intern Med. 2014;5:9. [PMC free article] [PubMed] [Google Scholar]
- 124.Loguercio C., Federico A., Trappoliere M., et al. The effect of a silybin-vitamin e-phospholipid complex on nonalcoholic fatty liver disease: a pilot study. Dig Dis Sci. 2007;52:2387–2395. doi: 10.1007/s10620-006-9703-2. [DOI] [PubMed] [Google Scholar]
- 125.Sorrentino G., Crispino P., Coppola D., et al. Efficacy of lifestyle changes in subjects with non-alcoholic liver steatosis and metabolic syndrome may be improved with an antioxidant nutraceutical: a controlled clinical study. Drugs R D. 2015;15:21–25. doi: 10.1007/s40268-015-0084-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Li M.Y., Fan J.G., National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association; Fatty Liver Disease Expert Committee, Chinese Medical Doctor Association Guidelines of prevention and treatment for alcoholic liver disease (2018, China) Chin J Hepatol. 2018;26:188–194. [Google Scholar]