Abstract
Circadian rhythms are 24-hour internal biological cycles that play an important role in metabolism, and their disruption has been implicated in the development of diseases such as diabetes mellitus type 2, obesity, coronary artery disease, hypertension, and metabolic syndrome. This phenomenon is illustrated by increased rates of risk factors for cardiovascular disease in night shift workers. Race, sex, and age are factors that play a role in circadian rhythms and metabolic disorders. The focus of this review article is to assess the link between circadian rhythm physiology and metabolic disorders from a race, sex, and age perspective. Black Americans were noted to have shorter free-running circadian periods, or tau, increased cortisol levels, and poorer sleep habits compared to white Americans, possibly contributing to increased rates of obesity, hypertension, and hyperlipidemia. Women were also noted to have shorter tau, increased levels of proinflammatory gut bacteria, and reduced sleep quality compared to men, possibly leading to higher rates of obesity, metabolic syndrome, hypertension (in postmenopausal women), and nonalcoholic fatty liver disease. Older people were noted to have decreased expression of anti-inflammatory clock genes compared to younger people, possibly leading to increased rates of obesity, diabetes, hyperlipidemia, and hypertension. Groups that are at a higher risk for metabolic disorders such as black Americans, women, and the elderly may have internal time keeping systems that place them at a higher risk for developing abnormal hormonal and/or inflammatory pathways.
Keywords: Circadian Rhythms, Metabolic Syndrome, Obesity, Hypertension
Introduction
Circadian rhythms are an integral part of healthy biological functioning. They affect sleep-wake cycles, hormone release, body temperature, and metabolism.1 A typical circadian rhythm in humans has a period of about 24 hours with diurnal variations in sunlight.2 It is synchronized to external environmental cues, known as zeitgebers, but can also run independently of them.3 The independent endogenous circadian period is sometimes denoted as tau, which persists even in the absence of zeitgebers.4 In humans, experiments with blind people have measured average tau to be a little over 24 hours.5 The suprachiasmatic nucleus (SCN) of the hypothalamus regulates the biological clock.6 It operates autonomously on a schedule close to 24 hours (tau) but also senses zeitgebers from light intake via ocular photoreceptors, nutrient ingestion, and temperature.7 This central clock thereby coordinates external cues with its own internal time keeping system to influence neuronal and hormonal pathways, activating peripheral clocks to carry out metabolic processes (Figure 1A).8 Specific genes have been identified in regulating peripheral clocks, including Clock and Bmal1. These genes encode the proteins Per and Cry, which have important roles in numerous metabolic pathways including glucose production, cholesterol regulation, fatty acid oxidation, and insulin secretion.9 These proteins are also tightly regulated by negative feedback mechanisms, in which their buildup during the daytime inhibits further production at nighttime. 10
Figure 1.
(A) The central pacemaker is entrained by the light/dark cycle. Metabolic homeostasis is dependent on the phase match between the social and environmental clocks. Metabolic dysfunction happens when there is a phase mismatch between social and environmental rhythms. Library of Science & Medical Illustrations were utilized in part to create this figure. https://creativecommons.org/licenses/by-nc-sa/4.0. (B) Representation of the effect of circadian rhythm disturbances on body systems. Sleep disturbances among shift workers are due to a desynchronization between the light-dark phase ultimately affecting different body systems. Library of Science & Medical Illustrations were utilized in part to create this figure. https://creativecommons.org/licenses/by-nc-sa/4.0.
Disruption of circadian rhythms has been shown to lead to metabolic disorders in mouse models.11 Studies on circadian dysregulation utilizing gene mutations or food restriction have demonstrated detrimental effects on metabolism leading to a variety of illnesses.12 In clinical medicine, abnormal circadian rhythms have been implicated in the development of obesity, diabetes mellitus type 2, coronary artery disease, thrombosis, inflammatory disorders, and metabolic syndrome.13 Metabolic syndrome refers to the multiple metabolic derangements linked to obesity and insulin resistance with a high risk of diabetes mellitus and cardiovascular disease.14 Circadian disruption is thought to be linked to weight gain and obesity by improper timing of meals. It is possible that eating during habitual sleep time limits opportunities for energy expenditure.15 In regard to diabetes, experimental data have shown that circadian disruption impairs pancreatic beta cell function and insulin sensitivity, and this combined effect of reduced insulin levels and penetrance results in abnormal glucose tolerance and an increased risk for progression to diabetes.16 Disruption of regular circadian cycles can also increase the risk of developing cardiometabolic disorders, as is evidenced by increased rates of risk factors for cardiovascular disease in shift workers (Figure 1B).17
Studies show that the prevalence of metabolic disorders varies depending on race, sex, or age.1,18,19 Black Americans and Hispanics are at an increased risk for developing hypertension, hyperlipidemia, and obesity compared to white Americans.20, 21, 22 Women are more likely to develop obesity and metabolic syndrome relative to men, and postmenopausal women are more likely to develop hypertension.23, 24, 25 Older people are at a higher risk for developing obesity, diabetes, hyperlipidemia, and hypertension than younger people.19 While these studies note that certain ethnicities, genders, or age groups are more prone to developing certain diseases, they rarely comment on possible differences in circadian rhythm physiology, let alone linking these differences to metabolic disease. This review article will explore potential mechanisms by which race, sex, or age variations in circadian rhythms and response to disruption may influence susceptibility to metabolic disorders.
Race, sex, and age are intertwined and sometimes produce mixed patterns in metabolism.26,27 For example, the prevalence of metabolic syndrome is higher in black women than that in white women, but the opposite pattern is seen in men.26 Advanced age also has different effects on circadian rhythms in men vs women, with core body temperature fluctuating less in older men vs younger men but unchanged in women of different ages.27 Men go to bed later than women before 40 years old, but earlier after this age.28 This complex interplay makes studying the effects of race, sex, and age on circadian rhythms and metabolic disorders all the more nuanced.
Role of Race in Circadian Disruption and Metabolic disorder
Before discussing the effects of race, it is important to note that race is a complex topic that is inevitably linked to confounding factors and bias. Broad racial categories such as “black”, “white”, “Asian”, or “Hispanic” tend to be monolithic and oftentimes are poorly defined with respect to geographical location and/or socioeconomic factors. For a more comprehensive analysis, both within-group and between-group differences should be analyzed to better appreciate the effect of race on circadian and metabolic disruption. For example, a study of race may actually be a study of underlying socioeconomic and historical differences among groups of people, and frequently if those variables are controlled, the effect of race is no longer statistically significant.1 One previous study of over 10,000 people of West African descent found that African Americans were twice as likely to develop hypertension as West Africans. Rates of obesity and salt intake varied consistently with the prevalence of disease, with higher rates of both factors noted in African Americans. This suggests that environmental rather than genetic factors drive differences.29
Race, Metabolic Disorder, and the Circadian Link
There are studies that show African Americans and Hispanic Americans are at a higher risk of developing risk factors for cardiovascular disease such as hypertension, hyperlipidemia, and obesity.20, 21, 22 A 2009 US National Health and Nutrition Examination Survey showed that Hispanic American males had the highest prevalence of low-density lipoprotein-cholesterol, followed by black Americans and then by white Americans.20 A 2001 Behavioral Risk Factor Surveillance Study showed that obesity, defined as body mass index (BMI) ≥30, was the highest in Black Americans (34.8%) and Native Americans (34.3%), while lowest in Asian Americans (4.8%).21 Nonwhite American groups are also at an increased risk for mortality from cardiovascular disease. One study showed that African Americans had the highest age-adjusted cardiovascular disease mortality in the United States in 2010, followed by white Americans, Hispanic Americans, and then East Asian Americans.22
The degree to which such disparity is attributed to differences in circadian rhythms is less well documented with few studies that evaluate this hypothesis. A healthy circadian rhythm includes an evening blood pressure (BP) drop or “dip” of around 10% or more.30 However, there is a correlation between patients who have “nondipping” circadian BP (<10% fall in average BP from day to evening) and cardiovascular morbidity.30 In a study by Sherwood et al,30 sleep quality was worse in “nondipping” patients than that in patients with standard circadian BP drop, and African Americans were found to have decreased sleep efficiency. Another study on BP “dipping” by Hyman et al31 showed that black Americans and Hispanic Americans are less likely to be “nondippers” than white Americans. Lack of this natural decrease in nocturnal BP has been associated with a worse prognosis from hypertension including more severe end-organ damage.32 In addition to the “dipping” phenomenon, differences in the timing of heart attacks in people of distinctive ethnic backgrounds further support the link between circadian rhythms and metabolic disorder. López et al33 studied 340 acute myocardial infarction patients and found that British Caucasians and Indo-Asians were more likely to have heart attacks between midnight and noon, whereas the converse pattern was true for Mediterranean Caucasians. This study suggests that there are possible differences in circadian rhythms among 3 separate ethnic groups leading to the timing of acute myocardial infarction.
Possible Mechanisms for Race Differences in Circadian Rhythms Impacting Metabolic Disorders
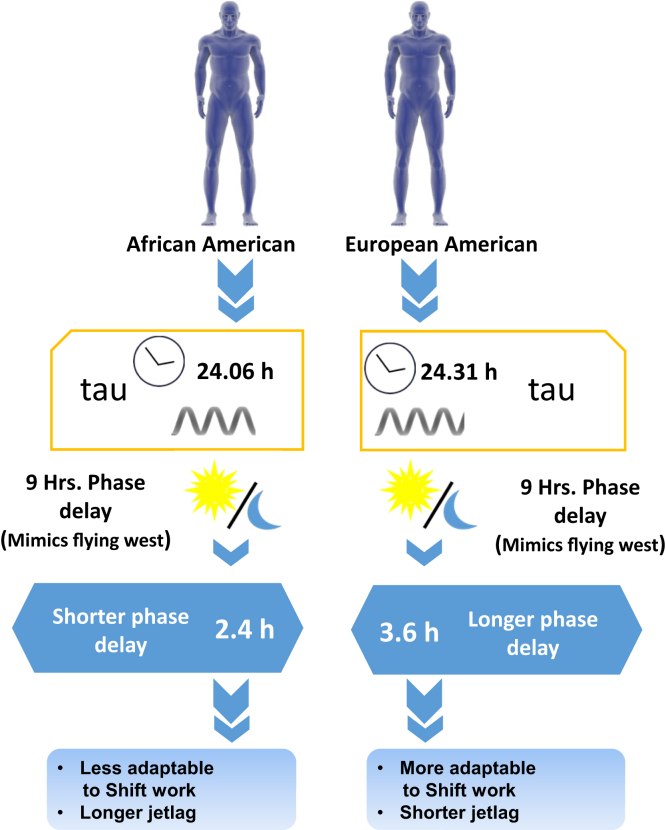
One possible mechanism by which race differences in circadian rhythms affect metabolism might be observed in the adaptation differences to night shift work. Eastman et al compared the circadian rhythms of African Americans to that of European Americans. Of the 23 African Americans and 22 European Americans studied, European Americans, on average, had a statistically significant longer free-running circadian period, or tau, than African Americans by over 0.2 hours (24.31 hours vs 24.06 hours, respectively).34 A significant correlation was found between longer tau at baseline and more extensive phase delays upon circadian disruption; more phase delays mean being able to fall asleep and wake up later, which is adaptive for night shift work. When these subjects underwent a 9-hour phase delay of the light-dark, sleep-wake, and meal schedule, which mimics flying west or sleeping during the day following night shifts, European Americans were found to have more phase delays than African Americans (3.6 hours vs 2.4 hours, respectively).34 African Americans were conversely found to have shorter tau and consequently more minor phase delays.34 This translates to, on average, longer jet lags upon westward travel and more difficulty adapting to night shift work (Figure 2). It is hypothesized that these variations in tau arose from differences in migration patterns out of East Africa tens of thousands of years ago. Those who migrated to Europe had to adapt to a seasonally changing photoperiod, whereas those who migrated to West Africa found themselves around the equator where the photoperiod is more constant throughout the year.34 It is possible that a longer circadian period developed in Europeans as an adaptation to changing photoperiods because it could help hunters at the time track dawn.35
Figure 2.
tau = free-running circadian period. Effect of race on circadian rhythm patterns. African-Americans were reported to display a shorter tau than European-Americans by approximately 0.25 hours. This leads to a shorter phase delay on average for African-Americans by approximately 1.2 hours when subjects underwent a 9-hour phase delay of the light-dark/sleep-wake cycle (mimics flying west). Shorter phase delays are associated with longer jet lag and are less adaptive for shift work. Library of Science & Medical Illustrations were utilized in part to create this figure. https://creativecommons.org/licenses/by-nc-sa/4.
Numerous studies have shown that shift work, especially at night, negatively affects health.36, 37, 38 A Brazilian study of 211 workers showed that night workers had higher rates of cardiovascular risk and hypertension than day workers.39 In a large cohort study of over 7800 individuals in Britain, night workers were found to have increased BMI, abdominal circumference, and C-reactive protein compared to nonshift workers even after adjusting for diet and exercise confounders.40 Di Lorenza et al41 showed that obesity was significantly higher in shift workers relative to day workers (20.0% vs 9.7%, respectively). A systematic review by Canuto et al42 noted a positive association between shift work and metabolic syndrome. Pan et al43 found a linear association between years of rotating shift work and risk of developing diabetes. A review study showed that shift work and poor sleep were correlated with increased incidence and severity of inflammatory bowel disease.44
Circadian dysregulation plays a central role in the increased risk of metabolic disorders in shift workers.45 There is evidence that disrupted sleep can decrease leptin and increase ghrelin levels, effectively promoting hunger.46 The daily cortisol rhythm can be reversed with shift work, leading to increased insulin resistance.36 Given the significant effect of night shift work on the development of metabolic disorders and, additionally, that African-Americans may have increased difficulty adapting to night shift work, methods to overcome phase delays such as proper timing of the light-dark phase and melatonin could be impactful for African Americans.34
The circadian rhythm also affects the hypothalamic-pituitary-adrenal axis, which impacts hormone levels. Cortisol is a hormone under the control of the circadian rhythm, with levels rising at night and peaking within the first hour after awakening.47 Cortisol is directly related to the stress response, and prolonged high cortisol levels lead to Cushing syndrome, a disease process associated with a constellation of symptoms including weight gain, diabetes, hypertension, muscle weakness, and poor bone health.48 Sleep deprivation can also lead to higher cortisol levels. One study of 149 Chinese servicemen who underwent 24 hours of sleep deprivation showed that cortisol levels were significantly higher after deprivation than before (503.5 nmol/L vs 482.3 nmol/L, respectively).49 One community-based study demonstrated that black and Hispanic Americans have flatter cortisol slopes, which goes against the natural morning-peaking pattern discussed earlier.50 Flat diurnal cortisol slopes are generally maladaptive and have been associated with complications such as chronic fatigue syndrome, rheumatoid arthritis, and poor mental and physical health.51,52 Similarly, Hajat et al53 showed that black Americans had lower levels of early AM cortisol, less steep early decline, and flatter late-day declines than white Americans.
The hypothalamic-pituitary-adrenal axis also has many downstream targets, one of them being blood vessel stiffness. Kim et al54 showed that abnormal sleep duration and poor self-reported sleep quality were linked to a higher pulse wave velocity, a measure of arterial stiffness. Blacks are almost twice as likely to report short sleep and over 60% more likely to report long sleep than whites.55 In a study done in Brazil of 1427 participants, people of African descent had the highest pulse wave velocity compared to that of mixed black and Caucasian persons, people of Caucasian descent, and Amerindians.56 Thus, it is possible that higher rates of hypertension in people of African descent are related to the direct effects of poor sleep habits on arterial stiffness.
Role of Sex in Circadian Disruption and Metabolic Disorder
Sex and Metabolic Disorders
Energy metabolism is known to differ based on sex. Women typically have a higher proportion of body fat relative to men starting with puberty and lasting until menopause.57 When controlled for the BMI and age, males have higher degrees of liver fat, visceral fat, and upper extremity mass than females.58,59 Globally, women have higher rates of obesity than men with exceptions.23 This is likely mediated by differences in sex hormones, namely estrogen, as higher levels of aromatized estrogen have been found in overweight or obese men.18 Obesity rates are increasing particularly in women of childbearing age; 1 in 5 women in Western countries are obese at time of conception.60,61
Metabolic syndrome and sex differences have been studied in numerous countries with mixed results.24,62, 63, 64 In China, Jiang et al24 found in a cross-sectional survey that the incidence of metabolic syndrome increases with age and female gender. In a study by Janssen et al,62 it was found that the rate of metabolic syndrome increases notably during the perimenopausal and early postmenopausal years (odds ratio = 1.45 and 1.24, respectively; both P < .001). The same study concluded that the testosterone predominance in menopause may play a role in the increased risk of metabolic syndrome (independent of cardiovascular disease).62 Meng et al63 in a cross-sectional, community-based investigation found that postmenopausal females have a higher incidence of metabolic syndrome than men of the same age. Hattori et al evaluated over 3100 Japanese persons with metabolic syndrome and measured waist circumference, BP, blood sugar, and other factors. Men were found to have a higher prevalence of metabolic syndrome (23.3 vs 8.75%, P < .001) and premetabolic syndrome (21.2 vs 10.2%, P < .001).64
Sex differences in hypertension have been studied worldwide. In the United States, until the age of 45 years, males have a higher prevalence of hypertension. Between ages 46 and 64 years, women and men have similar prevalence, while women above the age of 65 years have a higher prevalence of hypertension than men.25 Physiologically, estradiol may have antihypertensive affects by promoting nitric oxide production and decreasing the angiotensin II type 1 receptor.65,66 Women also have increased self-reported differences in their BP regulation. Ganzeboom et al67 showed in a survey that more women in their early 20s reported at least an episode of orthostasis intolerances (ie, fainting) when compared to men (50 vs ∼25%).
Sex and Circadian Rhythm
Studies in mice have shown that the differences in the circadian rhythm between males and females are driven by gonadal hormones via the SCN.2 Male SCNs have more androgen receptors.68 This leads to morphological differences in the SCNs between male and female mice, including volume, number of synapses, and action potential threshold.69 When male mice underwent gonadectomy, effectively cutting off their androgen production, they were found to have more extended circadian periods. Treatment with testosterone or dihydrotestosterone effectively restored the circadian periods to normal.70
In humans, a month-long study of melatonin and temperature rhythms in 52 women and 105 men found that tau was significantly shorter in the women than that in the men (24.09 hours and 24.19 hours, respectively).71 This may explain why women are more likely to self-report a preference for mornings and typically go to bed earlier and wake up earlier than men.72
Menopause has been implicated with low sleep quality.73 Nocturnal hot flashes related to menopause were found to behave in a circadian pattern, typically peaking in the afternoon.74 A study of over 6000 women of Latin American origin found that over half the study group suffered from a sleep disorder. Postmenopausal status was directly related to sleep disorders.75
Possible Mechanisms for Sex Differences in the Circadian Rhythm Impacting Metabolic Disorder
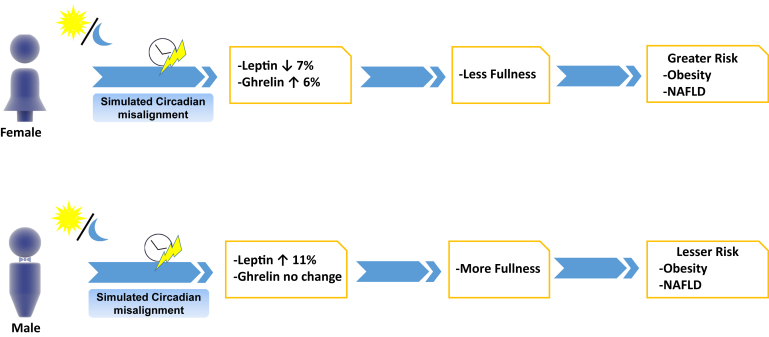
Qian et al showed that men and women adapt differently to shift work. In their study, 6 women and 8 men underwent 2 8-day lab protocols that simulated circadian alignment and misalignment. Upon misalignment, it was found that women had a ∼7% decrease in leptin and an ∼8% increase in ghrelin, a hunger hormone. Men, however, were found to have an ∼11% increase in leptin and no significant change in ghrelin.76 Females, on average, reported reduced fullness during the experiment, suggesting that women who experience frequent shift work may be at an elevated risk of developing obesity compared to men (Figure 3).
Figure 3.
Effect of sex on circadian rhythm patterns. Women were reported to have lower satiety and higher hunger hormones than men after undergoing 2 8-day lab protocols that simulated circadian alignment and misalignment. This leads to reduced fullness for women on average, increasing the risk for obesity and development of NAFLD. Library of Science & Medical Illustrations were utilized in part to create this figure. https://creativecommons.org/licenses/by-nc-sa/4.0.
Women are at an increased risk for nonalcoholic fatty liver disease (NAFLD), which is closely associated with insulin resistance, metabolic syndrome, and diabetes mellitus type 2.77 In a study of over 34,000 participants, women had a higher prevalence of NAFLD than men (22.9% vs 18.3%, respectively); a higher prevalence was noted in all age groups.78 Wang et al79 showed that among women, obesity and postmenopausal state significantly increased the risk of developing NAFLD and nonalcoholic steatohepatitis. Therapies targeted at improving circadian alignment could be especially beneficial for women.
The gut microbiome is influenced by the circadian rhythm, with fluctuations of certain bacteria in the intestinal tract.80,81 Disruption of intestinal circadian rhythms by irregular eating schedules can raise levels of proinflammatory bacteria and lower levels of protective bacteria, resulting in metabolic disorders.82 The gut microbiome differs notably between males and females. Bae et al2 showed that in men, Bacteroides in the gut decreases as the BMI increases; however, there was no change in Bacteroides prevalence with changes in the BMI in women. Women are known to be more susceptible to type 1 diabetes mellitus, so the higher prevalence of Bacteroides in obese women may play a role in developing such disease. This is supported by studies of mice, in which castration of male mice led to a gut microbiome more like that of female mice, subsequently increasing their rate of type 1 diabetes.2
Role of Age in Circadian Disruption and Metabolic Disorder
The Effects of Advanced Age on Metabolism and Circadian Rhythms
It is well known that aging is associated with a myriad of health problems. As a person grows older, there is a near-linear decrease in the basal metabolic rate, resulting in decreasing skeletal musculature and increasing percentage of fat tissue.83 This may explain why older people are at an increased risk for developing metabolic disorders such as obesity, diabetes, hyperlipidemia, and hypertension.19 Kuk et al studied metabolic syndrome using the Third National Health and Nutrition Examination Survey. They found that metabolic syndrome is present in 26% of patients younger than 65 years old and 55% of patients who are least 65 years old.84
Circadian rhythms also change with advancing age. Older adults commonly experience advancement of timing of their sleep to earlier hours. They also report increased complaints associated with sleep, including a more significant number of nighttime awakenings and early morning awakenings.85 Adolescents, conversely, display later chronotypes with a peak in ‘lateness’ at around 19 years old.28 Such a shift to earlier bedtime and awakening would suggest that older people possibly have shorter tau on average. However, the length of the circadian period was minimally affected by age in studying melatonin rhythms of 6 blind men across 10 years; interestingly, there was a small but significant increase in tau during the decade.86 The same finding was reproduced a year later by Duffy and Czeisler, who compared the circadian periods of 13 healthy older men and women to that of 9 young adults who participated in a similar study. Duffy and Czeisler employed a constant routine protocol in which subjects behaved uniformly for 40 hours, helping to limit confounding zeitgeibers such as food intake that may not have been controlled for in the study with blind men. They found that the intrinsic period was not statistically different between the older and younger study participants, suggesting that tau does not explain earlier awakening observed in the elderly.87 Rather, there is evidence suggesting that a reduction in sleep quality may better account for this. Dijk et al88 showed that older people have an increase in frequency of awakenings and more interrupted non-REM sleep.
Clock genes have been implicated in controlling circadian rhythms and their expression changes with advancing age. Bmal1 is a clock gene that has been shown to have anti-inflammatory properties. Nguyen et al89 showed that knockout of this gene in mice leads to impaired rhythmic cycling of inflammatory monocytes, predisposing the mice to pathologies associated with acute and chronic inflammation. It is hypothesized that decreased expression of Bmal1 with aging leads to a greater risk for developing metabolic disorders in the elderly. One study of hamsters showed that aging decreases Bmal1 expression in both the SCN and other brain regions but not in muscle.90 Other clock genes may also have anti-inflammatory properties such as Cry. Hashiramoto et al91 showed that knockout Cry mice have higher levels of inflammatory markers including tumour necrosis factor alpha, interleukin 1 beta, interleukin 6, and matrix metalloproteinase-3, contributing to worsening joint swelling and arthritis. It would be interesting to see if expression of Cry also decreases with aging.
Summary
The effects of race, sex, and age on circadian rhythms and response to circadian disruption are a driving force behind differences in metabolism and the prevalence of the metabolic disorder. In general, Americans of African descent, women, and the elderly are at increased susceptibility to disease, the etiology of which may be due to underlying differences in circadian physiology. Black Americans and females, on average, have shorter tau relative to white Americans and males, respectively. This is associated with more minor phase delays and may contribute to increased difficulty adjusting to night shift work. Older people have preserved tau but significant advancement of their sleep-wake cycle, which also has problems.
Further study of circadian rhythms is warranted in other racial/ethnic groups, as most current literature focuses on differences between blacks and whites. If, for example, Asians and Hispanics were included in a tau study and differences were still observed among the groups, it would provide further evidence that genetics rather than socioeconomics causes variation in circadian periods. Lastly, future research directions should be aimed at studying potential lifestyle modifications and pharmacologic therapies to mitigate circadian disruption and effect on metabolic disorders. These therapies could potentially be helpful in reducing the prevalence of NAFLD in women.
Acknowledgments
Authors' Contributions:
All authors were involved in data collection, writing the manuscript, and editing the manuscript.
Footnotes
Conflicts of Interest: The authors disclose no conflicts. Vinod K. Rustgi is a member of the Board of Editors. Their paper was handled in accordance with our conflict of interest policy. See https://www.ghadvances.org/content/authorinfo#conflict_of_interest_policy for full details.
Funding: The authors report no funding.
Ethical Statement: The study did not require the approval of an institutional review board.
References
- 1.Egan K.J., Knutson K.L., Pereira A.C., et al. The role of race and ethnicity in sleep, circadian rhythms and cardiovascular health. Sleep Med Rev. 2017;33:70–78. doi: 10.1016/j.smrv.2016.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bae S.A., Fang M.Z., Rustgi V., et al. At the interface of lifestyle, behavior, and circadian rhythms: metabolic implications. Front Nutr. 2019;6:132. doi: 10.3389/fnut.2019.00132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vitaterna M., Takahashi J., Turek F. Overview of circadian rhythms. Alcohol Res Health. 2001;25:85–93. [PMC free article] [PubMed] [Google Scholar]
- 4.Smith M.R., Burgess H.J., Fogg L.F., et al. Racial differences in the human endogenous circadian period. PLoS One. 2009;4 doi: 10.1371/journal.pone.0006014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sack R.L., Lewy A.J., Blood M.L., et al. Circadian rhythm abnormalities in totally blind people: incidence and clinical significance. J Clin Endocrinol Metab. 1992;75:127–134. doi: 10.1210/jcem.75.1.1619000. [DOI] [PubMed] [Google Scholar]
- 6.Asher G., Sassone-Corsi P. Time for food: the intimate interplay between nutrition, metabolism, and the circadian clock. Cell. 2015;161:84–92. doi: 10.1016/j.cell.2015.03.015. [DOI] [PubMed] [Google Scholar]
- 7.Freedman M.S. Regulation of mammalian circadian behavior by non-rod, non-cone, ocular photoreceptors. Science. 1999;284:502–504. doi: 10.1126/science.284.5413.502. [DOI] [PubMed] [Google Scholar]
- 8.Saini C., Suter D.M., Liani A., et al. The mammalian circadian timing system: synchronization of peripheral clocks. Cold Spring Harb Symp Quant Biol. 2011;76:39–47. doi: 10.1101/sqb.2011.76.010918. [DOI] [PubMed] [Google Scholar]
- 9.Imai S.-I. Nicotinamide phosphoribosyltransferase (Nampt): a link between NAD biology, metabolism, and diseases. Curr Pharm Des. 2009;15:20–28. doi: 10.2174/138161209787185814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhao E., Tait C., Minacapelli C., et al. Circadian rhythms, the gut microbiome, and metabolic disorders. Gastro Hep Adv. 2021;1:P93–P105. [Google Scholar]
- 11.Sahar S., Sassone-Corsi P. Regulation of metabolism: the circadian clock dictates the time. Trends Endocrinol Metab. 2012;23:1–8. doi: 10.1016/j.tem.2011.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Le Minh N. Glucocorticoid hormones inhibit food-induced phase-shifting of peripheral circadian oscillators. EMBO J. 2001;20:7128–7136. doi: 10.1093/emboj/20.24.7128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Maury E., Ramsey K.M., Bass J. Circadian rhythms and metabolic syndrome: from experimental genetics to human disease. Circ Res. 2010;106:447–462. doi: 10.1161/CIRCRESAHA.109.208355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Laaksonen D.E. Metabolic syndrome and development of diabetes mellitus: application and validation of recently suggested definitions of the metabolic syndrome in a prospective cohort study. Am J Epidemiol. 2002;156:1070–1077. doi: 10.1093/aje/kwf145. [DOI] [PubMed] [Google Scholar]
- 15.Noh J. The effect of circadian and sleep disruptions on obesity risk. J Obes Metab Syndr. 2018;27:78–83. doi: 10.7570/jomes.2018.27.2.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mason I.C., Qian J., Adler G.K., et al. Impact of circadian disruption on glucose metabolism: implications for type 2 diabetes. Diabetologia. 2020;63:462–472. doi: 10.1007/s00125-019-05059-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Reutrakul S., Knutson K.L. Consequences of circadian disruption on cardiometabolic health. Sleep Med Clin. 2015;10:455–468. doi: 10.1016/j.jsmc.2015.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kautzky-Willer A., Harreiter J., Pacini G. Sex and gender differences in risk, pathophysiology and complications of type 2 diabetes mellitus. Endocr Rev. 2016;37:278–316. doi: 10.1210/er.2015-1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sohail S., Yu L., Bennett D.A., et al. Irregular 24-hour activity rhythms and the metabolic syndrome in older adults. Chronobiol Int. 2015;32:802–813. doi: 10.3109/07420528.2015.1041597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mozaffarian D., Benjamin E.J., Go A.S., et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation. 2015;131:e29–e322. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 21.Wang Y., Beydoun M.A. The obesity epidemic in the United States--gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiol Rev. 2007;29:6–28. doi: 10.1093/epirev/mxm007. [DOI] [PubMed] [Google Scholar]
- 22.National Center for Health Statistics (US) Health United States, 2013: with special feature on prescription drugs. National Center for Health Statistics (US); Hyattsville: 2014. [PubMed] [Google Scholar]
- 23.Kanter R., Caballero B. Global gender disparities in obesity: a review. Adv Nutr. 2012;3:491–498. doi: 10.3945/an.112.002063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang B., Zheng Y., Chen Y., et al. Age and gender-specific distribution of metabolic syndrome components in East China: role of hypertriglyceridemia in the SPECT-China study. Lipids Health Dis. 2018;17:92. doi: 10.1186/s12944-018-0747-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ramirez L.A., Sullivan J.C. Sex differences in hypertension: where we have been and where we are going. Am J Hypertens. 2018;31:1247–1254. doi: 10.1093/ajh/hpy148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moore J.X., Chaudhary N., Akinyemiju T. Metabolic syndrome prevalence by race/ethnicity and sex in the United States, national health and nutrition examination survey, 1988-2012. Prev Chronic Dis. 2017;14:E24. doi: 10.5888/pcd14.160287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hood S., Amir S. The aging clock: circadian rhythms and later life. J Clin Invest. 2017;127:437–446. doi: 10.1172/JCI90328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fischer D., Lombardi D.A., Marucci-Wellman H., et al. Chronotypes in the US – influence of age and sex. PLoS One. 2017;12 doi: 10.1371/journal.pone.0178782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cooper R., Rotimi C., Ataman S., et al. The prevalence of hypertension in seven populations of West African origin. Am J Public Health. 1997;87:160–168. doi: 10.2105/ajph.87.2.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sherwood A., Hill L.K., Blumenthal J.A., et al. The effects of ambulatory blood pressure monitoring on sleep quality in men and women with hypertension: dipper vs. nondipper and race differences. Am J Hypertens. 2019;32:54–60. doi: 10.1093/ajh/hpy138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hyman D.J., Ogbonnaya K., Taylor A.A., et al. Ethnic differences in nocturnal blood pressure decline in treated hypertensives. Am J Hypertens. 2000;13:884–891. doi: 10.1016/s0895-7061(00)00279-x. [DOI] [PubMed] [Google Scholar]
- 32.Fagher B., Valind S., Thulin T. End-organ damage in treated severe hypertension: close relation to nocturnal blood pressure. J Hum Hypertens. 1995;9:605–610. [PubMed] [Google Scholar]
- 33.López F., Lee K.W., Marín F., et al. Are there ethnic differences in the circadian variation in onset of acute myocardial infarction? Int J Cardiol. 2005;100:151–154. doi: 10.1016/j.ijcard.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 34.Eastman C.I., Tomaka V.A., Crowley S.J. Circadian rhythms of European and African-Americans after a large delay of sleep as in jet lag and night work. Sci Rep. 2016;6:36716. doi: 10.1038/srep36716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Eastman C.I., Tomaka V.A., Crowley S.J. Sex and ancestry determine the free-running circadian period. J Sleep Res. 2017;26:547–550. doi: 10.1111/jsr.12521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Scheer F.A.J.L., Hilton M.F., Mantzoros C.S., et al. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc Natl Acad Sci U S A. 2009;106:4453–4458. doi: 10.1073/pnas.0808180106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wong P.M., Hasler B.P., Kamarck T.W., et al. Social jetlag, chronotype, and cardiometabolic risk. J Clin Endocrinol Metab. 2015;100:4612–4620. doi: 10.1210/jc.2015-2923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gu F., Han J., Laden F., et al. Total and cause-specific mortality of U.S. nurses working rotating night shifts. Am J Prev Med. 2015;48:241–252. doi: 10.1016/j.amepre.2014.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pimenta A.M., Kac G., Campos E Souza R.R., et al. Night-shift work and cardiovascular risk among employees of a public university. Rev Assoc Méd Bras (1992) 2012;58:168–177. [PubMed] [Google Scholar]
- 40.Thomas C., Power C. Shift work and risk factors for cardiovascular disease: a study at age 45 years in the 1958 British birth cohort. Eur J Epidemiol. 2010;25:305–314. doi: 10.1007/s10654-010-9438-4. [DOI] [PubMed] [Google Scholar]
- 41.Di Lorenzo L., de Pergola G., Zocchetti C., et al. Effect of shift work on body mass index: results of a study performed in 319 glucose-tolerant men working in a Southern Italian industry. Int J Obes. 2003;27:1353–1358. doi: 10.1038/sj.ijo.0802419. [DOI] [PubMed] [Google Scholar]
- 42.Canuto R., Garcez A.S., Olinto M.T.A. Metabolic syndrome and shift work: a systematic review. Sleep Med Rev. 2013;17:425–431. doi: 10.1016/j.smrv.2012.10.004. [DOI] [PubMed] [Google Scholar]
- 43.Pan A., Schernhammer E.S., Sun Q., et al. Rotating night shift work and risk of type 2 diabetes: two prospective cohort studies in women. PLoS Med. 2011;8:e1001141. doi: 10.1371/journal.pmed.1001141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Canakis A., Qazi T. Sleep and fatigue in IBD: an unrecognized but important extra-intestinal manifestation. Curr Gastroenterol Rep. 2020;22:8. doi: 10.1007/s11894-020-0746-x. [DOI] [PubMed] [Google Scholar]
- 45.Brum M.C.B., Filho F.F.D., Schnorr C.C., et al. Shift work and its association with metabolic disorders. Diabetol Metab Syndr. 2015;7:45. doi: 10.1186/s13098-015-0041-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Taheri S., Lin L., Austin D., et al. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004;1:e62. doi: 10.1371/journal.pmed.0010062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Elverson C.A., Wilson M.E. Cortisol: circadian rhythm and response to a stressor. Newborn Infant Nurs Rev. 2005;5:159–169. [Google Scholar]
- 48.Thau L, Gandhi J, Sharma S. Physiology, Cortisol. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing. 2022. https://www.ncbi.nlm.nih.gov/books/NBK538239/. Accessed April 7, 2022.
- 49.Song H.T., Sun X.Y., Yang T.S., et al. Effects of sleep deprivation on serum cortisol level and mental health in servicemen. Int J Psychophysiol. 2015;96:169–175. doi: 10.1016/j.ijpsycho.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 50.DeSantis A.S., Adam E.K., Hawkley L.C., et al. Racial and ethnic differences in diurnal cortisol rhythms. Psychosom Med. 2015;77:6–15. doi: 10.1097/PSY.0000000000000131. [DOI] [PubMed] [Google Scholar]
- 51.Kuras Y.I., Assaf N., Thoma M.v., et al. Blunted diurnal cortisol activity in healthy adults with childhood adversity. Front Hum Neurosci. 2017;11:574. doi: 10.3389/fnhum.2017.00574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Adam E.K., Quinn M.E., Tavernier R., et al. Diurnal cortisol slopes and mental and physical health outcomes: a systematic review and meta-analysis. Psychoneuroendocrinology. 2017;83:25–41. doi: 10.1016/j.psyneuen.2017.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hajat A., Diez-Roux A., Franklin T.G., et al. Socioeconomic and race/ethnic differences in daily salivary cortisol profiles: the multi-ethnic study of atherosclerosis. Psychoneuroendocrinology. 2010;35:932–943. doi: 10.1016/j.psyneuen.2009.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim C.W., Chang Y., Zhao D., et al. Sleep duration, sleep quality, and markers of subclinical arterial disease in healthy men and women. Arterioscler Thromb Vasc Biol. 2015;35:2238–2245. doi: 10.1161/ATVBAHA.115.306110. [DOI] [PubMed] [Google Scholar]
- 55.Kingsbury J.H., Buxton O.M., Emmons K.M., et al. Sleep and its relationship to racial and ethnic disparities in cardiovascular disease. Curr Cardiovasc Risk Rep. 2013;7 doi: 10.1007/s12170-013-0330-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.de Lima Santos P.C.J., de Oliveira Alvim R., Ferreira N.E., et al. Ethnicity and arterial stiffness in Brazil. Am J Hypertens. 2011;24:278. doi: 10.1038/ajh.2010.244. [DOI] [PubMed] [Google Scholar]
- 57.Wu B.N., O'Sullivan A.J. Sex differences in energy metabolism need to be considered with lifestyle modifications in humans. J Nutr Metab. 2011;2011:391809. doi: 10.1155/2011/391809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Power M.L., Schulkin J. Sex differences in fat storage, fat metabolism, and the health risks from obesity: possible evolutionary origins. Br J Nutr. 2008;99:931–940. doi: 10.1017/S0007114507853347. [DOI] [PubMed] [Google Scholar]
- 59.Nielsen S., Guo Z., Johnson C.M., et al. Splanchnic lipolysis in human obesity. J Clin Invest. 2004;113:1582–1588. doi: 10.1172/JCI21047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.King J.C. Maternal obesity, metabolism, and pregnancy outcomes. Annu Rev Nutr. 2006;26:271–291. doi: 10.1146/annurev.nutr.24.012003.132249. [DOI] [PubMed] [Google Scholar]
- 61.Zera C., McGirr S., Oken E. Screening for obesity in reproductive-aged women. Prev Chronic Dis. 2011;8:A125. [PMC free article] [PubMed] [Google Scholar]
- 62.Janssen I. Menopause and the metabolic syndrome: the study of women's health across the nation. Arch Intern Med. 2008;168:1568–1575. doi: 10.1001/archinte.168.14.1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Meng Z., Liu M., Zhang Q., et al. Gender and age impacts on the association between thyroid function and metabolic syndrome in Chinese. Medicine. 2015;94:e2193. doi: 10.1097/MD.0000000000002193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hattori T., Konno S., Munakata M. Gender differences in lifestyle factors associated with metabolic syndrome and preliminary metabolic syndrome in the general population: the Watari Study. Intern Med. 2017;56:2253–2259. doi: 10.2169/internalmedicine.8578-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kim J.M., Kim T.H., Lee H.H., et al. Postmenopausal hypertension and sodium sensitivity. J Menopausal Med. 2014;20:1–6. doi: 10.6118/jmm.2014.20.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Song J.J., Ma Z., Wang J., et al. Gender differences in hypertension. J Cardiovasc Transl Res. 2020;13:47–54. doi: 10.1007/s12265-019-09888-z. [DOI] [PubMed] [Google Scholar]
- 67.Ganzeboom K.S., Colman N., Reitsma J.B., et al. Prevalence and triggers of syncope in medical students. Am J Cardiol. 2003;91:1006–1008. doi: 10.1016/s0002-9149(03)00127-9. A8. [DOI] [PubMed] [Google Scholar]
- 68.Fernandez-Guasti A., Kruijver F.P.M., Fodor M., et al. Sex differences in the distribution of androgen receptors in the human hypothalamus. J Comp Neurol. 2000;425:422–435. doi: 10.1002/1096-9861(20000925)425:3<422::aid-cne7>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 69.Bailey M., Silver R. Sex differences in circadian timing systems: implications for disease. Front Neuroendocrinol. 2014;35:111–139. doi: 10.1016/j.yfrne.2013.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Iwahana E., Karatsoreos I., Shibata S., et al. Gonadectomy reveals sex differences in circadian rhythms and suprachiasmatic nucleus androgen receptors in mice. Horm Behav. 2008;53:422–430. doi: 10.1016/j.yhbeh.2007.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Duffy J.F., Cain S.W., Chang A.M., et al. Sex difference in the near-24-hour intrinsic period of the human circadian timing system. Proc Natl Acad Sci U S A. 2011;108 Suppl 3(Suppl 3):15602–15608. doi: 10.1073/pnas.1010666108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Adan A., Natale V. Gender differences in morningness–eveningness preference. Chronobiol Int. 2002;19:709–720. doi: 10.1081/cbi-120005390. [DOI] [PubMed] [Google Scholar]
- 73.Pines A. Circadian rhythm and menopause. Climacteric. 2016;19:551–552. doi: 10.1080/13697137.2016.1226608. [DOI] [PubMed] [Google Scholar]
- 74.Freedman R.R. Menopausal hot flashes: mechanisms, endocrinology, treatment. J Steroid Biochem Mol Biol. 2014;142:115–120. doi: 10.1016/j.jsbmb.2013.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Blümel J.E., Cano A., Mezones-Holguín E., et al. A multinational study of sleep disorders during female mid-life. Maturitas. 2012;72:359–366. doi: 10.1016/j.maturitas.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 76.Qian J., Morris C.J., Caputo R., et al. Sex differences in the circadian misalignment effects on energy regulation. Proc Natl Acad Sci U S A. 2019;116:23806–23812. doi: 10.1073/pnas.1914003116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.European Association for the Study of the Liver (EASL) European Association for the Study of Diabetes (EASD) European Association for the Study of Obesity (EASO) EASL–EASD–EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. Diabetologia. 2016;59:1121–1140. doi: 10.1007/s00125-016-3902-y. [DOI] [PubMed] [Google Scholar]
- 78.Summart U., Thinkhamrop B., Chamadol N., et al. Gender differences in the prevalence of nonalcoholic fatty liver disease in the Northeast of Thailand: a population-based cross-sectional study. F1000Res. 2017;6:1630. doi: 10.12688/f1000research.12417.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang Z., Xu M., Hu Z., et al. Prevalence of nonalcoholic fatty liver disease and its metabolic risk factors in women of different ages and body mass index. Menopause. 2015;22:667–673. doi: 10.1097/GME.0000000000000352. [DOI] [PubMed] [Google Scholar]
- 80.Thaiss C.A., Zeevi D., Levy M., et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159:514–529. doi: 10.1016/j.cell.2014.09.048. [DOI] [PubMed] [Google Scholar]
- 81.Liang X., Bushman F.D., FitzGerald G.A. Rhythmicity of the intestinal microbiota is regulated by gender and the host circadian clock. Proc Natl Acad Sci U S A. 2015;112:10479–10484. doi: 10.1073/pnas.1501305112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Voigt R.M., Forsyth C.B., Green S.J., et al. Circadian rhythm and the gut microbiome. Int Rev Neurobiol. 2016;131:193–205. doi: 10.1016/bs.irn.2016.07.002. [DOI] [PubMed] [Google Scholar]
- 83.Shimokata H., Kuzuya F. Aging, basal metabolic rate, and nutrition. Nippon Ronen Igakkai Zasshi. 1993;30:572–576. doi: 10.3143/geriatrics.30.572. [DOI] [PubMed] [Google Scholar]
- 84.Kuk J.L., Ardern C.I. Age and sex differences in the clustering of metabolic syndrome factors: association with mortality risk. Diabetes Care. 2010;33:2457–2461. doi: 10.2337/dc10-0942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Duffy J.F., Zitting K.M., Chinoy E.D. Aging and circadian rhythms. Sleep Med Clin. 2015;10:2457–2461. doi: 10.1016/j.jsmc.2015.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kendall A.R., Lewy A.J., Sack R.L. Effects of aging on the intrinsic circadian period of totally blind humans. J Biol Rhythms. 2001;16:87–95. doi: 10.1177/074873040101600110. [DOI] [PubMed] [Google Scholar]
- 87.Duffy J.F., Czeisler C.A. Age-related change in the relationship between circadian period, circadian phase, and diurnal preference in humans. Neurosci Lett. 2002;318:117–120. doi: 10.1016/s0304-3940(01)02427-2. [DOI] [PubMed] [Google Scholar]
- 88.Dijk D.J., Duffy J.F., Czeisler C.A. Age-related increase in awakenings: impaired consolidation of NonREM sleep at all circadian phases. Sleep. 2001;24:565–577. doi: 10.1093/sleep/24.5.565. [DOI] [PubMed] [Google Scholar]
- 89.Nguyen K.D., Fentress S.J., Qiu Y., et al. Circadian gene Bmal1 regulates diurnal oscillations of Ly6Chi inflammatory monocytes. Science. 2013;341:1483–1488. doi: 10.1126/science.1240636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Duncan M.J., Prochot J.R., Cook D.H., et al. Influence of aging on Bmal1 and Per2 expression in extra-SCN oscillators in hamster brain. Brain Res. 2013;1491:44–53. doi: 10.1016/j.brainres.2012.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hashiramoto A., Yamane T., Tsumiyama K., et al. Mammalian clock gene cryptochrome regulates arthritis via proinflammatory cytokine TNF-α. J Immunol. 2010;184:1560–1565. doi: 10.4049/jimmunol.0903284. [DOI] [PubMed] [Google Scholar]