Abstract
Background
Arteriovenous malformations (AVMs) are abnormal direct connections between arterial and venous systems, without an interposed capillary bed. This permits high-flow arteriovenous shunting, which precipitates structural changes in the afferent and efferent vessels, namely arterial smooth muscle hyperplasia and thinning of venous walls. Patients with intracranial AVMs typically present with a haemorrhage, headache or seizure. Treatment is either via medical management aimed at control of seizures, headache and blood pressure, or interventional via surgical, radiation or radiologically guided embolisation.
Case description
We report the case of a woman in her early 40s presenting with a tonic-clonic seizure against a background of a 31-year history of migraine and an 18-month history of tremors in her right arm. The clinical examination was remarkable for an extremely loud cranial bruit and a right homonymous hemianopia. Imaging diagnosed an 8 cm Martin-Spetzler grade V intracranial arteriovenous malformation in her left parietal lobe, which was deemed unsuitable for operative or radiotherapy-based intervention.
Conclusion
The patient was managed through observation and relatively good control of her breakthrough seizures was achieved through the addition of brivaracetam to her lamotrigine and carbamazepine-based therapy, six years after her initial presentation.
LEARNING POINTS
Arteriovenous malformations may go undetected for decades.
The presence of a cranial bruit is an important sign and tool in diagnosing an intracranial arteriovenous malformation. The presence of a homonymous hemianopia, of which a patient may be unaware, helps to localise an intracranial lesion.
The risks of operative or radiological intervention must be balanced with the risks of haemorrhage or refractory seizure when adopting a strategy for the treatment of an intracranial arteriovenous malformation. Good control of epileptic symptoms in those deemed not suitable for operative or radiological intervention can be achieved by careful titration of anti-seizure medications.
Keywords: Arteriovenous malformation, Martin-Spetzler, epilepsy, cranial bruit, homonymous hemianopia
CASE DESCRIPTION
We report the case of a right-handed woman in her early 40s who presented to our emergency department after collapsing with loss of consciousness. She felt preceding weakness and on recovery, she experienced clonic right arm and bilateral leg movements but denied incontinence and had no evidence of tongue biting on examination. Her past medical history was positive for a thirty-one-year history of migraines and an eighteen-month history of tremors and twitches in her right arm. She denied prior drug allergies and was prescribed only simple analgesia as required. She did not smoke tobacco, drink alcohol or use illicit substances, and lived independently with her husband. No family history of seizures, or other intracranial or neurological abnormalities were reported. On examination, her blood pressure was 111/70 mmHg, heart rate was 99 beats per minute, temperature was 36.2°C, respiratory rate was 16 breaths per minute and oxygen saturations were 98% on room air. Respiratory and abdominal examinations were unremarkable. A neurological examination revealed a resting tremor in her right arm and a right homonymous hemianopia. A cardiovascular examination was remarkable for an extremely loud bruit, radiating throughout the praecordium, which could be traced to a point of maximum volume over the patient’s left posterior skull.
A full blood count, coagulation profile, renal profile, and blood glucose and thyroid function tests were within normal limits. Liver function tests were mildly elevated (alanine aminotransferase (ALT) of 34 IU/l, gamma-glutamyl transferase (GGT) of 66 U/l and aspartate aminotransferase (AST) of 36 IU/l) and subsequently normalised. A bone profile was remarkable for a mild corrected hypocalcaemia of 2.1 mmol/l, which subsequently normalised. The patient’s chest X-ray, urinalysis, and electrocardiogram and cardiac monitoring were unremarkable. A brain computerized tomography (CT) scan with contrast (Fig. 1, Video 1) was reported as showing a large arteriovenous malformation measuring 8×5×5 cm in the posterior left parietal lobe, extending from the vertex almost to the tentorium cerebelli.
Figure and Video 1.
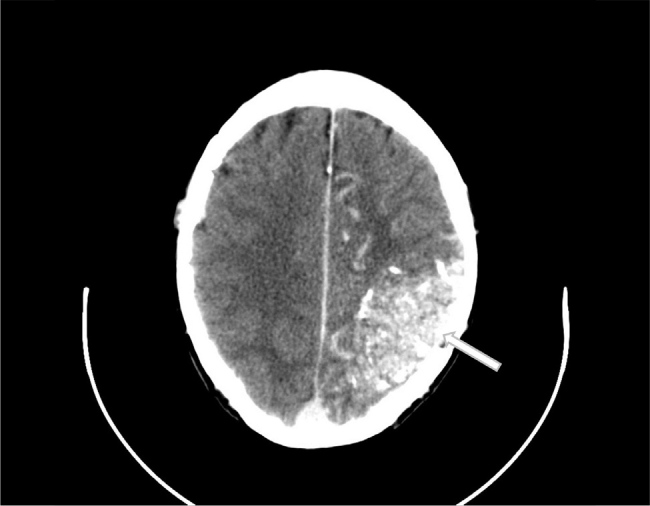
Axial CT brain with contrast showing a large intracranial arteriovenous malformation (8×6×5 cm) in the posterior left parietal lobe. It extends from the vertex almost to the tentorium cerebelli and is associated with large, calcified draining veins. The associated transverse sinuses are shown to be markedly enlarged. No significant mass effect or midline shift is shown. There is additionally some marked vascular indentation shown at the inner diploe of the skull consistent with a large chronic arterial supply and venous drainage.
She was subsequently transferred to a tertiary centre where she underwent a magnetic resonance imaging (MRI) brain, MRI cerebral angiography (Fig. 2, Video 2) and a CT cerebral angiogram (Fig. 3, Video 3), which diagnosed a left parietal arteriovenous malformation of Spetzler-Martin grade V.
Figure and Video 2.
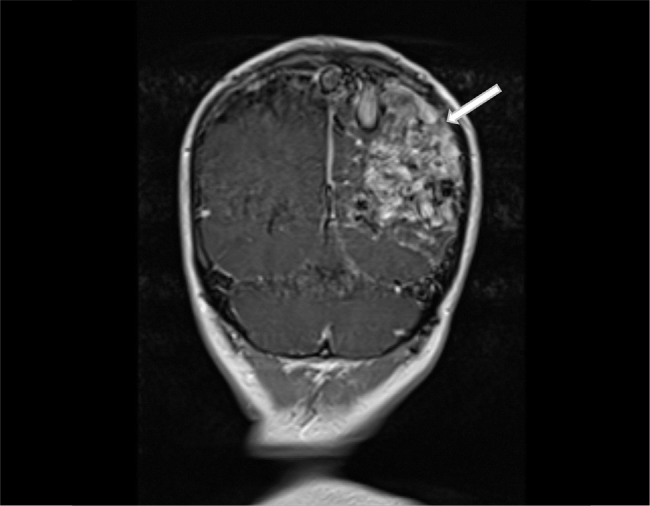
Coronal MRI brain showing a large intracranial arteriovenous malformation in the left parietal lobe of Spetzler-Martin Grade V. The vascular nidus is shown to extend into the left temporal and occipital lobes and to be of 8 cm in anteroposterior diameter, 4 cm in transverse diameter and 5 cm in depth. Extensive gliosis of the adjacent parenchyma is shown in addition to evidence of venous hypertension and congestion in the cortical draining veins.
Figure and Video 3.
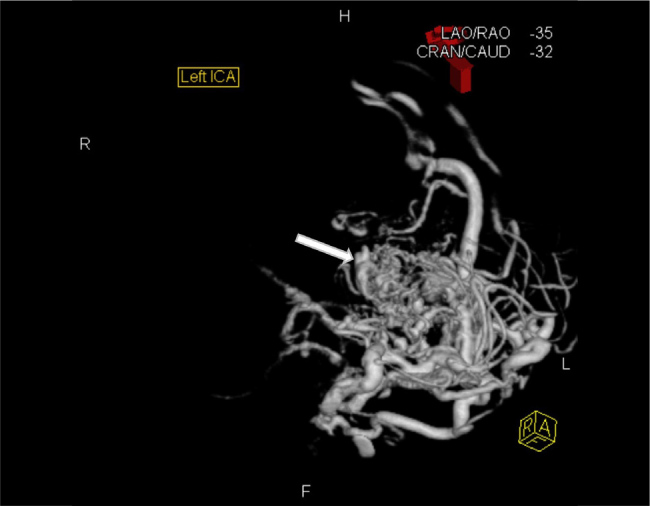
Left internal carotid angiogram showing a large intracranial arteriovenous malformation in the left parietal lobe of Spetzler-Martin grade V. It shows arterial supply from the left anterior cerebral artery, right cavernous carotid artery, left middle cerebral artery, left external carotid artery, left posterior cerebral artery and left anterior inferior cerebellar artery. It additionally shows both superficial and deep venous drainage and aneurysmal pedicles from both the left anterior cerebral artery and left posterior cerebral artery.
A CT thorax, abdomen and pelvis with contrast failed to demonstrate extracranial vascular malformations.
Her case was discussed with a neurosurgical and neurological team at a tertiary centre, who advised that her AVM was too complex for surgical or radiotherapy-based intervention, with a significant risk of precipitating haemorrhage, thrombosis, oedema or local brain trauma. Instead, she was treated with medical anti-seizure therapy. She was administered a loading dose of intravenous phenytoin and then commenced on oral phenytoin and levetiracetam. She experienced breakthrough myoclonic jerking of her right arm. This was treated with an escalation of her levetiracetam and phenytoin dosing. Persistent symptoms subsequently precipitated an alteration of her therapy to carbamazepine and gabapentin, to good effect.
Her intervening years have been marked by tremor, an intermittent stammer and recurrent focal aware motor seizures, which primarily centred on her right upper limb. She presented a further seven times to our emergency department over the following six years with episodic generalised tonic-clonic seizures. Neuroimaging (Fig. 4) remained unchanged at each presentation, and she was cycled through a variety of anti-seizure medications including levetiracetam, phenytoin, gabapentin, clobazam, lamotrigine and carbamazepine.
Figure 4.
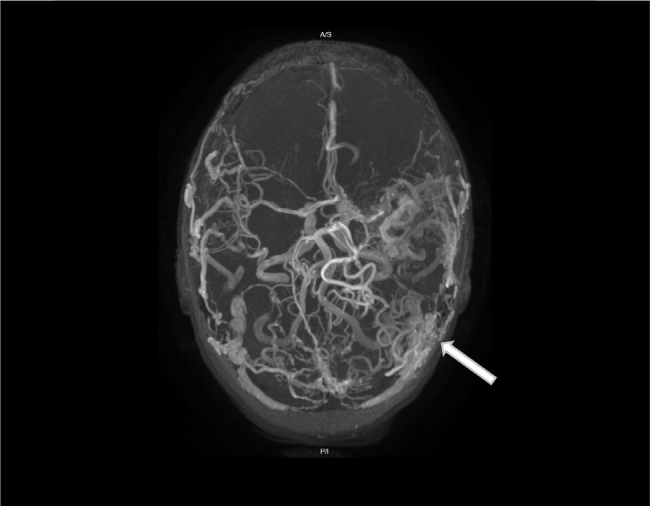
MRI brain and MRI cerebral angiography 3-D axial performed six years after Figure 2 and Video 2, showing a large intracranial arteriovenous malformation in the left parietal lobe with no significant change in size, gliosis of adjacent neural parenchyma or evidence of venous hypertension.
She continued to suffer breakthrough seizure activity until, six years post her initial presentation, she was commenced on brivaracetam to complement her other therapies of lamotrigine and carbamazepine. This addition significantly decreased the frequency, severity and duration of her seizures.
DISCUSSION
Arteriovenous malformations are abnormal direct connections between arterial and venous systems without an interposed capillary bed[1]. This permits high-flow arteriovenous shunting, which precipitates structural changes in the afferent and efferent vessels, namely arterial smooth muscle hyperplasia and thinning of venous walls.
Intracranial AVMs are classified using the Spetzler-Martin grading system (Table 1). The patient’s AVM scored the maximum of five for size, eloquent site location and deep venous drainage. Cerebral AVMs are commonly identified either incidentally on brain imaging, or after presentation with headache, seizure or haemorrhage.
Table 1.
Spetzler-Martin grading system for intracranial arteriovenous malformations.
| Size of nidus | |
|---|---|
| Small (<3 cm) | 1 |
| Medium (3–6 cm) | 2 |
| Large (>6 cm) | 3 |
| Eloquence of adjacent brain | |
| Non-eloquent | 0 |
| Eloquent | 1 |
| Venous drainage | |
| Superficial veins only | 0 |
| Deep veins | 1 |
The epileptogenic mechanisms of sensitively located intracranial AVMs are thought to pertain to[2]:
Ischaemia and hypoperfusion of adjacent neural tissue due to arteriovenous shunting, vascular steal phenomenon, venous hypertension and the loss of cerebral circulation autoregulation;
The gliosis of surrounding brain tissue secondary to prior occult haemorrhage or local haemosiderin deposition as a result of the extravasation of red blood cells through pathologically dilated leaky capillaries;
The mass effect of larger AVMs, which may cause chronic neural irritation and associated remodelling.
Treatment is either via observation with medical management centred on control of headache, blood pressure and seizure activity, or is interventional and is centred on the use of one or a combination of surgical resection, stereotactic radiosurgery or embolisation. AVMs that have presented with haemorrhage have an annual rate of subsequent haemorrhage four times higher than those of AVMs that presented otherwise. Accordingly, these lesions are generally treated to reduce the risk of future events[3].
There is no absolute consensus on which lesions should be subjected to intervention. A large multicentre, randomised prospective trial (the ARUBA trial), the results of which favoured an observation approach, was heavily criticised for selection bias, a short period of follow-up and a lack of subgroup analysis[4]. Further studies have suggested that the risk of morbidity and mortality increases with Martin-Spetzler grade, and that the key to achieving good outcomes is the selection of appropriate patients – in terms of prior haemorrhage, size, location and co-morbidities – in a multidisciplinary centre with a high volume of procedures[5]. Microsurgery is generally more appropriate for smaller lesions, whereas stereotactic radiotherapy is more appropriate for inaccessible lesions and those associated with sensitive cerebral structures.
This patient complained of a long history of migrainous headache with aura and presented with generalised seizure-like activity. A visual deficit, in terms of a right homonymous hemianopia, was demonstrated on examination. Her persistent intermittent right arm twitching may represent focal motor seizures caused in part by the mechanisms described above, including vascular steal, venous hypertension and parenchymal gliosis. She achieved reasonable symptom control with medical anti-seizure therapy.
Footnotes
Conflicts of Interests: The Authors declare that there are no competing interests.
Patient Consent: Written informed consent has been obtained from the concerned patient permitting publication of their clinical history.
REFERENCES
- 1.Lawton MT, Rutledge WC, Kim H, Stapf C, Whitehead KJ, Li DY, et al. Brain arteriovenous malformations. Nat Rev Dis Primers. 2015;1:15008. doi: 10.1038/nrdp.2015.8. [DOI] [PubMed] [Google Scholar]
- 2.Turjman F, Massoud TF, Sayre JW, Viñuela F, Guglielmi G, Duckwiler G. Epilepsy associated with cerebral arteriovenous malformations: a multivariate analysis of angioarchitectural characteristics. AJNR Am J Neuroradiol. 1995;16:345–350. [PMC free article] [PubMed] [Google Scholar]
- 3.Kim H, Al-Shahi Salman R, McCulloch CE, Stapf C, Young WL MARS Coinvestigators. Untreated brain arteriovenous malformation: patient-level meta-analysis of hemorrhage predictors. Neurology. 2014;83:590–597. doi: 10.1212/WNL.0000000000000688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mohr JP, Parides MK, Stapf C, Moquete E, Moy CS, Overbey JR, et al. International ARUBA investigators. Medical management with or without Interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicentre, non-blinded, randomised trial. Lancet. 2014;383:614–621. doi: 10.1016/S0140-6736(13)62302-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Javadpour M, Al-Mahfoudh R, Mitchell PS, Kirollos R. Outcome of microsurgical excision of unruptured brain arteriovenous malformations in ARUBA-eligible patients. Br J Neurosurg. 2016;30:619–622. doi: 10.1080/02688697.2016.1181153. [DOI] [PubMed] [Google Scholar]


