Abstract
Cannabis sativa, otherwise known as hemp, is discussed to highlight the various problems and prospects associated with its use as an herbal ingredient. The chemical composition of hemp, with classification based on cannabinoid contents, its biological activities, current global scenarios and legality issues, economic importance, and future prospects, are discussed.
Keywords: Cannabis sativa, cannabinoids, biological activities, cannabis legality, cannabis economy
1. Introduction
For the past 5000 years, hemp (Cannabis sativa L.), a flowering annual herbaceous crop plant, has been utilized in agriculture. Native to Asia, it is now widely distributed throughout North America, Europe, and Africa [1]. Three species of cannabis were originally recognized: Cannabis sativa, Cannabis indica, and Cannabis ruderalis. However, these species are now recognized as variations of Cannabis sativa (var. sativa, var. indica, and var. ruderalis). Currently, C. sativa strains are divided into three main categories: industrial hemp, CBD-type hemp, and medical cannabis (high THC) [2,3]. Globally, 147 million people have been reported to consume cannabis. This high level of cannabis consumption is partly attributed to its intoxicative effect [4]. Many people who are only aware of cannabis as a recreational narcotic fail to recognize its huge social, industrial, and economic significance. Hemp, or cannabis, is a versatile crop; in the past, people have planted hemp for its seeds, fibers, essential oils, and medical properties [1]. Despite being a diverse plant with both medical and industrial uses, C. sativa is a controversial plant. It is popularly grown worldwide for illicit drug usage (marijuana). During the past few decades, there have been deliberate measures (outlawing) put in place to curb the widespread recreational use of cannabis because of its psychoactive Tetrahydrocannabinol (THC) content. This has significantly and unjustly hurt the hemp industry. Worldwide, hemp seeds and inflorescence are widely used as dietary sources and ingredients in food supplements. Rich in physiologically active, nonpsychoactive cannabinoids, hemp inflorescence contains cannabidiol (CBD), cannabinol (CBN), and cannabigerol (CBG), which have strong activity against reactive oxygen species (ROS) as antioxidants (Figure 1); thus, they may exhibit anticonvulsant, anxiolytic, and spasmolytic properties, among others [5]. With its deliciously nutty flavor, hempseed is an excellent source of fiber, minerals, vitamins, and vital fatty acids. It also contains important amino acids, which are found in the highly digestible protein albumin and edestin [6]. Most hempseed oils (>90%) contain polyunsaturated fatty acids [7], which are well known for their protective properties against inflammatory disorders, cancer, and cardiovascular disease [8,9]. Since 2017, hemp has also been used to make beer. This resulted from the federal ban on marijuana and the relaxation of regulatory barriers pertaining to the use of cannabis plant portions other than the seed, which contains the psychoactive Δ9-Tetrahydrocannabinol (Δ9-THC) [10]. This review provides a comprehensive analysis of Cannabis sativa as a useful herbal ingredient in food and medicine, and its biological properties, legality issues, and economic prospects.
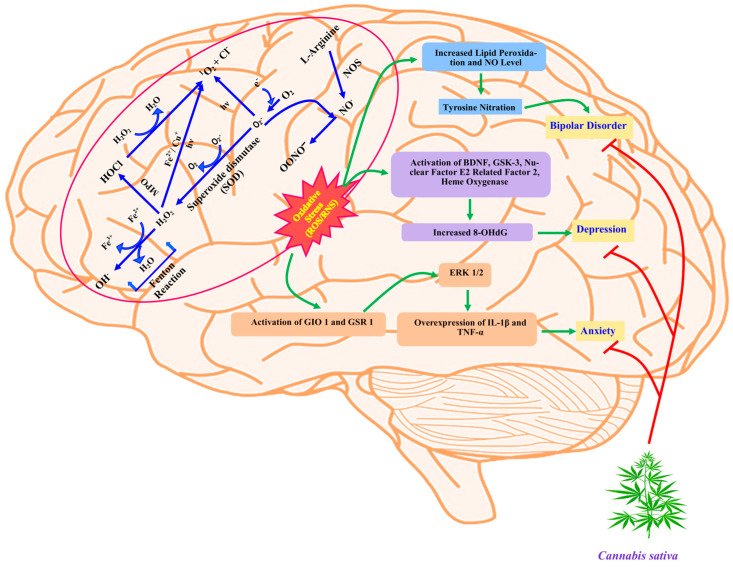
Figure 1.
Schematic representation illustrating how the production of free radicals (ROS/RNS) affects biological systems by causing cellular stress and even resulting in various mental illnesses, as well as how Cannabis sativa may help to treat these. [HOCl: Hypochlorous acid; H2O2: Hydrogen peroxide; 1O2: Singlet oxygen; O2.−: Superoxide; OH•: Hydroxyl radical; OONO−: Peroxynitrite; NO.: Nitric oxide; Fe2+: Iron ion; Cu2+: Copper ion; Cl−: Chlorine ion; ROS: Reactive oxygen species; RNS: Reactive nitrogen species; NOS: Nitric oxide synthase; MPO: Myeloperoxidase; BDNF: Brain-derived neurotrophic factor; GSK-3: Glycogen synthase kinase 3; 8-OHdG: 8-hydroxy2-deoxyguanosine; ERK: Extracellular signal-regulated kinases; IL-1β, interleukin 1 beta; TNF α: Tumor necrosis factor α].
2. Methodology
The study involved an open (no date restriction) literature review of work published on C. sativa concerning its use as an herbal ingredient, using various scientific search engines such as Google Scholar, Mendeley Scopus, PubMed, and Science Direct. The following inclusive criteria were used in the study: Cannabis sativa, hemp, or Cannabis (its active ingredients and cannabinoid contents), and medicinal values, non-medicinal values, biological activities, and economic prospects.
3. Industrial Uses and Processing of Cannabis sativa or Hemp
Hemp industries use known hemp cultivars and other plant components to obtain the necessary components and the desired end products [11]. Three distinct parts of hemp are used: fibers, seeds, and flowers [3]. Industrial hemp uses seeds and fibers, while CBD-type hemp uses flowers only [12].
3.1. Hemp Fiber
Approximately half of the world’s fiber hemp supply is produced in China, where the industrial hemp fiber market is dominant [3]. Cannabis sativa is heavily fertilized to prevent blooming and promote taller stalks for hemp fibers. It is then harvested similarly to hay [3,11]. The optimum density for this form of hemp is 32–37 plants/m2 [13]. Plants are kept in storage until the stalks dry out, at which point the stalks are processed by dividing the tough, woody inside of the hurd from the outer fibrous exterior of the bast fibers [3,11]. While the hard fibers can be processed into building materials like particle board and hempcrete, the bast fibers can be used to make products like textiles, rope, or paper [3].
3.2. Hemp Seed
The plant is grown similarly to that of fiber for hemp seeds. In a dense seeding arrangement, male and female plants are deliberately sown close to one another [3]. For optimal pollination potential, male plants can be surrounded by female plants. To prevent seed dispersal, harvesting must be conducted rapidly [3]. Hemp oil can be extracted by pressing the dehulled seeds [3]. Thereafter, hemp seeds can be prepared for sale as protein powder, flour, hemp oil, roasted snacks, and/or animal feed [14,15].
3.3. Hemp Flowers or CBD-Type Hemp
Hemp with CBD is produced and harvested distinctly. The chemical makeup of hemp changes from tissue to tissue throughout the plant [11]. The female flowers contain significant quantities of CBD and terpenes, which are the desirable ingredients for hemp with CBD [11,16,17,18]. To optimize flower production, plants are widely separated; their heights range from 1.2 to 2.4 m [3]. A distance of 0.9–1.2 m is suitable spacing between hemp plants that are CBD-type [3]. The necessary components must subsequently be extracted from the adult blossoms by thoroughly drying them [3,11]. The cannabinoids are decarboxylated when the flowers are dried [11]. A variety of techniques, such as alcohol extraction, supercritical CO2, or liquid hydrocarbons, can be used to extract their compounds [3]. The steps required for alcohol extraction include chilling the ethanol, pumping it into a container containing C. sativa plant material, and letting it soak. After the alcohol has been removed from the extract through distillation, a mixture of cannabinoids is left behind that can be used in various applications or suspended in oils. Carbon dioxide is pressurized and placed in a chamber along with the plant material using supercritical CO2. By removing the CO2, the distillate can be identified, yielding the purest chemical profile. The last method for using liquefied hydrocarbons is to mix plant material with compressed low molecular weight hydrocarbons, and then the hydrocarbons are evaporated. The liquefied hydrocarbon process is well known for requiring appropriate safety measures because the hydrocarbons involved are flammable. Product makers in the region favor supercritical CO2 and alcohol extractions out of these three techniques [19,20].
4. Hemp Chemical Composition
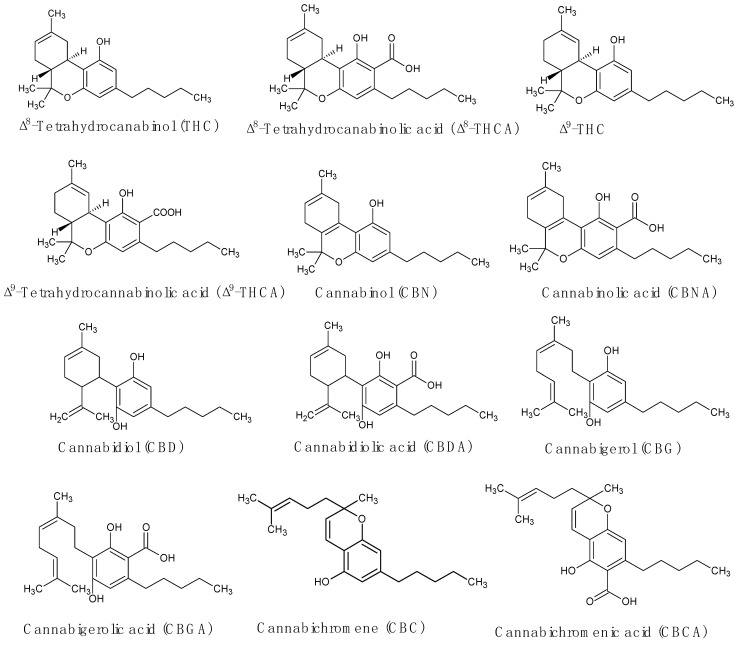
Hemp has about 600 distinct chemical components [11]. Among these, almost 180 are cannabinoids, making up a significant portion of the identified metabolites [21]. According to Głodowska and Łyszcz [21], several of these compounds exist in decarboxylated and acidic forms. THC, CBD, CBN, CBG, and cannabichromene (CBC), as illustrated in Figure 2, are the primary cannabinoids found in hemp [21]. THC and CBD are the two most well-known cannabinoids.
Figure 2.
Some cannabinoids from Cannabis sativa.
4.1. Tetrahydrocannabinol
Tetrahydrocannabinol, also known as Δ9-THC, is thought to be the most studied cannabinoid [21]. Cannabis sativa has long been prohibited because of its psychotropic qualities. In contrast to CBD-type strains that have less than 0.3% THC, medical cannabis (high THC) strains typically have around 10% THC [21]. The acidic form of THC that is biosynthesized and more frequently found in living plants is called tetrahydrocannabinolic acid (THCA). Due to the double bond between the ninth and tenth carbon, it is known as Δ9-THC [22]. Many studies have been conducted to determine the medicinal benefits of Δ9-THC, which has been shown to have potential as an appetite stimulant, painkiller, and antiemetic [23]. Mueller et al. [24] claim that Δ9-THC is detrimental to cognitive function, particularly in young individuals. It has been demonstrated that Δ9-THC influences memory, cognitive speed, and learning [24]. Memory impairment, paranoia, disorientation, and dizziness are among the negative effects that Δ9-THC is more likely to cause than CBD. However, when Δ9-THC and CBD are mixed, CBD usually counteracts the cognitive symptoms of pure Δ9-THC [25].
4.2. Cannabidiol
Cannabidiol (CBD) is one of the most significant cannabis molecules in medicine. Anxiety, depression, multiple sclerosis, post-traumatic stress disorder (PTSD), schizophrenia, and dementia are among the psychiatric and neurologic conditions for which CBD has been demonstrated to show a considerable level of effectiveness [18,21,25,26,27]. It has been reported to exhibit antimicrobial, anti-inflammatory, and antioxidant activities [18,26,27]. Certain malignancies, diabetes, and seizures in epilepsy are known to be manageable with CBD [18,25,26]. It has been demonstrated to promote collagen crosslinking, which improves bone fracture healing [28]. According to Whyte et al. [29], treating mice with pure CBD decreased bone resorption. Epidolex, a pure form of CBD, is an FDA-approved pharmaceutical for the treatment of seizures in Lennox–Gastaut syndrome and Dravet syndrome [30].
4.3. Other Cannabinoids
Cannabis sativa contains other cannabinoids that may have medicinal and other applications. Cannabigerol (CBG), cannabigerolic acid (CBGA), and cannabidiolic acid (CBDA) are chemical precursors of CBD that are included in goods’ total CBD content and are known to have marked biological effects such as anti-inflammatory and antibacterial activities [18]. Young cannabis plants are the ones that typically contain CBC, while the older ones contain CBN [26]. According to Andre [26], the former is known for its sedative, anti-inflammatory, antibacterial, and antifungal properties, while the latter is a sedative byproduct of THC breakdown [26].
4.4. Terpenes
Terpenes may be metabolized either primarily or secondarily. According to Iseppi et al. [31], plants require primary metabolites to survive, but they also require secondary metabolites to flourish in the ecosystem. Hemp contains about 100 different types of terpenes [27,31]. These terpenes give it many fragrant qualities and can affect the medicinal qualities of the CBD-type cannabis [27,31]. They can also interact with the neurotransmitter receptors and increase the permeability of the blood–brain barrier. Thus, terpenes have been demonstrated to function in concert, enhancing the therapeutic potential of the whole-hemp products [21]. Monoterpenes and sesquiterpenes are the two primary terpene subtypes found in hemp [31]. Monoterpenes are compounds with ten carbons, including β-myrcene, pinene, terpinolene, ocimene, and limonene. β-caryophyllene is the most prevalent sesquiterpene [31]. The sedative effects of hemp are believed to be attributed to β-myrcene. Pellati et al. [17] reported that β-myrcene also possesses anti-inflammatory and anxiolytic characteristics. There is anti-proliferative activity in β-caryophyllene [17,31]. According to a study by Głodowska and Łyszcz, [21], terpinolene has antibacterial properties. It was found to inhibit the growth of Gram-positive and Gram-negative bacteria as well as the fungus yeast.
5. Biological Activity of Cannabis sativa Compounds
Studies with C. sativa have shown several biological activities, such as antibacterial, anticoagulant, antidiabetic, anticancer, anti-inflammatory, analgesic, and neuroprotective activities, as well as activities against neuropathic pain.
5.1. Antimicrobial Activity
It has been documented that essential oils and organic extracts of various C. sativa components exhibit antibacterial action against a variety of microbes. Nevertheless, the level of antibacterial activity differs between cultivars [32], depending on the portion of the plant utilized, the manner of extraction, and the kind of extract. The hydroalcoholic extract from the seed was tested against a variety of Gram-positive and -negative bacteria, including Bifidobacterium breve, B. longum, Salmonella typhimurium, Enterobacter aerogenes, Enterococcus faecalis, Limosilactobacillus reuteri, Lacticaseibacillus paracasei, and Levilactobacillus brevis. The results obtained indicated a low level of antibacterial activity, with minimum inhibitory concentrations (MICs) greater than 1 mg/mL [33]. Anjum’s study examined the antibacterial properties of cannabis leaves by testing four extracts prepared with acetone, chloroform, ethanol, and water against three different bacterial strains: Pseudomonas aeruginosa, Staphylococcus aureus, and Escherichia coli [34]. The four extracts displayed commonalities in the results, with an up to 19 mm zone of inhibition (ZI). Furthermore, the findings reported by Manosroi et al. [35] demonstrated the effectiveness of ethanolic extract against Streptococcus mutans as an antibacterial agent, with a ZI value of 1.33 ± 0.58 mm. Furthermore, the antibacterial activity of the essential oil from the aerial portion gave MIC values of 64 and 38 mg/mL, respectively, against Helicobacter pylori and Klebsiella pneumonia, respectively, while the volatile terpenes of cannabis exhibited varying degrees of activity in relation to the targeted bacterial strains. Additionally, a more moderate inhibitory activity was observed against E. coli, P. aeruginosa, and Bacillus subtilis, with a MIC of 1.2 mg/mL [36]. In another study, antibacterial activity was observed against Micrococcus luteus and S. aureus, with a MIC of 4.7 mg/mL for both strains. Numerous biological studies have demonstrated that volatile terpenes of cannabis are potent antibacterial agents [37,38,39].
5.2. Anticoagulant Activity
It has been proposed that anticoagulant plants serve as herbal treatments that may help to find novel therapeutic agents to treat disorders associated with thrombosis. To ascertain the potential anti-prothrombotic impact of cannabis leaf metabolites, blood coagulation tests were carried out, focusing on the three primary cannabinoids: THC, CBD, and CBN [40]. It was revealed that two cannabinoids, THC and CBN, displayed considerable IC50 values of 87 and 83 μg/mL. THC exhibited the maximum activity, measuring 1.79 mg/mL, while CBN showed lower activity, as indicated by a high IC50 value of 9.89 mg/mL. To ascertain the clotting times, this study also included an in vivo test conducted on overweight rats. Consequently, the rats given cannabis showed a 50% efficiency rate with clotting that was twice as high as the control groups, indicating that cannabinoids may have a useful anticoagulant effect.
5.3. Antidiabetic Activity
Diabetes is a complicated metabolic disease that progresses over time and is defined by unusually high blood glucose levels. Another name for this illness is hyperglycemia [41]. Ninety percent of those affected globally have type 2 diabetes, which is defined as non-insulin-dependent diabetes [41]. The World Health Organization (WHO) states that diabetes is a chronic illness brought on by insufficient insulin production by the pancreas or improper insulin utilization by the body. The hormone insulin is responsible for controlling blood sugar levels. The standard method for assessing antidiabetic activity is to quantify the suppression of amylase [42]. Typically produced by the pancreas, this enzyme is essential in raising blood sugar levels because it breaks down dietary carbohydrates like starch into simple monosaccharides in the digestive tract. This is followed by additional α-glucosidase degradation into glucose, which is absorbed and enters the bloodstream. Thus, blocking the actions of the enzymes α-amylase and α-glucosidase might impede the breakdown of carbohydrates, postpone the absorption of glucose, and ultimately lower blood glucose levels [43]. With a value of 3.77 mmol ACAE/g oil, the study by Zengin et al. [44] demonstrated that the essential oils from the cannabis aerial portion demonstrated antidiabetic activities against the α-glucosidase enzyme. Additionally, this essential oil was tested against α-amylase; however, no noteworthy results were seen.
5.4. Anticancer Activity
Globally, cancer continues to be a leading cause of morbidity and mortality. Currently, traditional methods including radiotherapy, chemotherapy, and surgery are used to treat cancer, leaving affected individuals with adverse health impacts [45]. In view of these challenges, there have been deliberate attempts to explore cannabis and other natural resources for less toxic and more potent anticancer agents. In vitro and in vivo experiments on a variety of cancer cell lines, including breast [46], prostate [47], cervix [48], brain [49], colon [50], and leukemia/lymphoma [51], have shown the unambiguous efficacy of various cannabis-derived chemicals. Numerous in vivo and in vitro investigations have shown how phytocannabinoids affect the growth of tumors. According to these findings, at concentrations ranging from 5 to 65 µm, some cannabinoids, like Δ9-THC and CBD, cause apoptosis and limit proliferation in a variety of cancer cell lines [46,52,53,54,55,56,57]. Additionally, combining specific phytocannabinoids enhanced the anticancer potential of cannabis products. For instance, research by Armstrong et al. [52] showed that melanoma cell mortality was higher when CBD and Δ9-THC were combined than when Δ9-THC was used alone. Phytochemicals found in the cannabis plant, particularly cannabinoids, have little differential activity and are generally non-selective in their effects on cancer cells compared to normal cells. As a result, scientists are interested in synthesizing lead compounds based on a molecule’s natural backbone and extracting bioactive phytochemicals from cannabis that have strong anticancer effects.
5.5. Anti-Inflammatory and Analgesic Activities
Numerous constituents of cannabis have demonstrated potent anti-inflammatory properties. Cannabis seeds have been reported to reduce inflammation, particularly when applied to primary human monocytes treated with lipopolysaccharide (LPS). The seeds lowered TNF-α gene expression and IL-6 gene secretion. Moreover, cannabinoids demonstrated considerable anti-inflammatory action by inhibiting the synthesis of pro-inflammatory cytokines and chemokines and may be useful in treating inflammation-related illnesses [58,59]. Any procedure whose main goal is to lessen pain is referred to as an analgesic action. This can refer to not only medication but also any other technique intended to produce analgesia, or the elimination of pain perception [60]. Since opioids are the only treatment available for severe pain, cannabis has been identified as a much-needed substitute for opioids in terms of its analgesic properties [61]. Cannabinoids from cannabis function as opioid-sparing drugs, combining the benefits of opioids with the ability to reduce dosages and side effects associated with long-term opioid therapy [62]. Therefore, it is worthwhile to give careful thought to the prudent use of cannabis-based medications to relieve patients who are experiencing excruciating pain.
5.6. Neuroprotective Activity
The terpenes and cannabinoids of cannabis have also been shown to have neuroprotective qualities. In Pheochromocytoma-12 cells (PC-12), the neuroprotective properties of 17 substances found in C. sativa aerial parts were assessed. These compounds included ferulic acid, (E)-methyl-p-hydroxycinnamate, and p-hydroxybenzaldehyde, which also showed additional protective benefits against H2O2-induced damage [63]. Furthermore, Landucci et al. [64] demonstrated that suitable concentrations of CBD or CBD/THC ratios can represent a viable therapeutic intervention in the treatment of post-ischemic neuronal death. Di Giacomo et al. [65] reported the neuroprotective and neuromodulating effects induced by CBD and CBG in rat Hypo-E22 cells and isolated hypothalamus. A different study by Esposito et al. [66] emphasized the significance of CBD as a potentially useful novel medication that can lessen amyloid-induced neuroinflammatory reactions. Additionally, utilizing immuno-histochemistry analysis, the work by Perez et al. [67] detailed the neuronal counts of both motor and sensory neurons following CBD treatment. The results obtained indicated a 30% improvement in synaptic preservation on the spinal cord for the group receiving CBD treatment, indicating a moderate neuroprotective impact.
5.7. Neuropathic Chronic Pain Management
Positive (somatosensory function gain) and negative (somatosensory function loss) sensory sensations and indicators are commonly associated with neuropathic pain (NP) [68]. Either peripheral or central NP disorders can lead to chronic NP. The most recent classification of peripheral neuropathy NP was released by the International Association for the Study of Pain (IASP); the subtypes of chronic peripheral NP that fall under this category are trigeminal neuralgia (TN), chronic NP following peripheral nerve injury, painful polyneuropathy, post-herpetic neuralgia, and painful radiculopathy [69]. There are three subcategories of chronic central NP: chronic central NP linked to multiple sclerosis (MS), chronic central NP related to spinal cord injury (SCI) or brain injury, and chronic central post-stroke pain [69]. Twenty patients with chronic fibromyalgia pain were included in experimental randomized placebo-controlled four-way crossover research conducted in the Netherlands to examine the analgesic benefits of inhaled therapeutic cannabis. In the trial, three cannabis strains totaling 100 mg were administered in a single day: Bedrocan (22.4 mg THC, 1 mg CBD), Bediol (13.4 mg THC, 17.8 mg CBD), and Bedrolite (18.4 mg CBD, 1 mg THC). A placebo was also administered. The tolerance to pressure pain threshold was significantly increased by Bedrocan and Bediol. The cannabis strain with high levels of THC and CBD (Bediol) showed the most significant impact. The analgesic effects of CBD in combination with a very low amount of THC (Bedrolite, which primarily comprises CBD) were not better than those of a placebo. This outcome is different from trials where individuals with chronic pain reported positive effects from CBD treatment; these effects were likely connected to better mood, anxiety, and sleep patterns [70].
A prospective non-randomized single-arm clinical trial that took place in Italy in 2018 examined data from 338 patients who suffered from radiculopathy, headaches, fibromyalgia, and different types of neuropathic pain [71]. For a period of 12 months, they were given a daily dosage of 5–40 mg of THC (many of the individuals needed 10 mg), which is equivalent to 28–210 mg of cannabis floss with 19% THC and 1% CBD. At follow-up appointments, the participants’ level of discomfort was assessed. Of the patients, 77 discontinued the study because there was minimal benefit, and 33 quit because of adverse events (AEs), which may have been brought on by the high proportion of THC in Bedrocan (19%). According to this study, cannabis treatment is beneficial in lowering pain intensity using a visual analog scale (VAS), chronic pain-related disability, and the anxiety and depression that follow, as measured by the Hospital Anxiety and Depression Scale (HADS), all without producing severe adverse events [71].
Table 1 summarizes the biological activities of different cannabinoids found in Cannabis sativa.
Table 1.
Therapeutic applications of cannabinoids found in C. sativa.
| Compounds | Biological Application | References |
|---|---|---|
| Δ9-Tetrahydro-cannabinol (Δ9-THC) | Anti-inflammation, antianalgesic, antioxidant, anti-ulcers, antipruritic | [72,73,74] |
| Δ9-THCA | Anticancer and analgesic—stimulates CB1-dependent signaling in the N18TG2 neuroblastoma cell line | [75] |
| Cannabidiol (CBD) | Antianxiety, antinausea, antiarthritic, antipsychotic, anti-inflammatory, and immunomodulatory properties, antidiabetes, atherosclerosis, treatment of alzheimer disease, hypertension, metabolic syndrome, ischemia–reperfusion injury, depression, and neuropathic pain. |
[76,77,78] |
| CBDA | Rescued memory deficits and reduced amyloid-beta and Tau pathology in an Alzheimer’s disease-like mouse model | [79] |
| Cannabinol (CBN) | Sedative, antibiotic, anticonvulsant, anti-inflammatory | [80] |
| CBNA | Antioxidant property due to free radical scavenging activity | [81] |
| Cannabichromene (CBC) | Anti-inflammatory/analgesic, antidepressant | [82,83] |
| CBCA | Rapid antibacterial activity against methicillin-resistant Staphylococcus aureus | [84] |
| Cannabigerol (CBG) | Antioxidant, anti-inflammatory, analgesic effect | [74,82] |
| CBGA | Exhibits antioxidant activity manifested in its ability to scavenge free radicals, to prevent the oxidation process, and to reduce metal ions | [85] |
6. Global Scenario and Legality of Cannabis
The hemp industry, which promotes the growth, processing, and use of hemp and its products, has been encouraged and sustained in recent years by creative applications of hemp materials and the growing concern for the sustainable development of the agricultural bioeconomy, especially in Canada and Europe [86]. In the 1990s, hemp farming was legalized in both the EU and Canada. By 2008, the output of hemp in Canada had risen rapidly, following some initial market adjustments. Currently, Canada produces more than 20,000 hectares of hemp each year under license, and they are almost all for seed [86]. Europe has seen a rapid expansion in hemp farming, with acreage rising from 8000 ha in 2011 to over 33,000 ha in 2016 [87]. The primary growing regions in Europe include Romania, France, the Netherlands, and the Baltic States. Nonetheless, many other European nations have begun or increased their hemp farming, mostly to produce hemp used for both fiber and seeds. The shift in the rapidly growing hemp seed market and the decreasing quality fiber requirements for novel biomaterials are driving dual-purpose hemp production in Europe [88]. Cannabis is becoming more and more popular, with the global cannabis market having a net worth of USD 14.5 billion in 2018. Now that more than 50 nations have legalized cannabis in some capacity for medical purposes and 6 more have legalized cannabis for recreational use, this market is predicted to reach USD 107.67 billion by 2025 [89]. Each country has different laws governing the possession, distribution, and cultivation of cannabis, as well as the conditions under which it can be used medicinally and how it should be taken.
With cannabis legalization in these countries and more countries to pass bills legalizing its use, issues relating to health risk, abuse, and other psychosocial problems abound. Although overdoses of cannabis do not easily result in the respiratory depression that is often associated with opioid use, a significant number of deaths from cardiovascular diseases and hyperemesis syndrome have been reported to accompany the sustained, heavy use of cannabis [90]. Furthermore, the non-medical use of cannabis has been reported to increase the risk of motor vehicle injuries when used at 1–3 h before driving, while it also increases the propensity to produce babies with low birth weights when used habitually during pregnancy [90,91]. Additionally, adolescents who use cannabis are more likely than adults to develop addiction problems [92]. Coffey and Patton have reported that habitual hemp use may contribute to cognitive impairment [93], early dropping out of school [94], the use of other illicit drugs, the development of schizophrenia and affective disorders [95], and suicidal thoughts [96]. Other problems associated with cannabis dependence include anxiety, depression, psychotic symptoms, and adverse gastrointestinal symptoms [97,98].
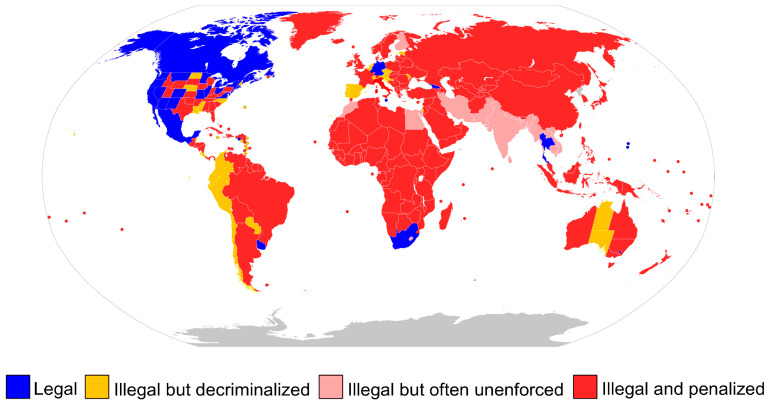
Three United Nations treaties—the 1968 Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances, the 1971 Convention on Psychotropic Substances, and the 1961 Single Convention on Narcotic Drugs—regulate these practices in many countries [99,100]. Under the Single Convention, cannabis was reclassified in 2020 from a Schedule I and IV drug to a Schedule I only substance (with Schedules IV, I, II, and III being the tightest-controlled to least) [101,102]. Although cannabis is classified as a Schedule I substance under the treaty, some countries may permit its medical use, even though it is highly addictive and has a high potential for abuse [102]. Although a few countries have decriminalized the use of cannabis by making simple possession of it an infraction that is treated like a minor traffic offense, many nations forbid its recreational use. Some countries in the Middle East and the Far East have much harsher laws, such as those that punish even minor amounts of possession with years in prison [103]. Canada, Georgia, Germany, Luxembourg, Malta, Mexico, South Africa, Thailand, and Uruguay, together with 24 states, 3 territories, and the District of Columbia in the United States, and the Australian Capital Territory in Australia, are among the nations that have legalized cannabis for recreational purposes (Figure 3).
Figure 3.
The legality of consuming cannabis globally for recreational purposes [104].
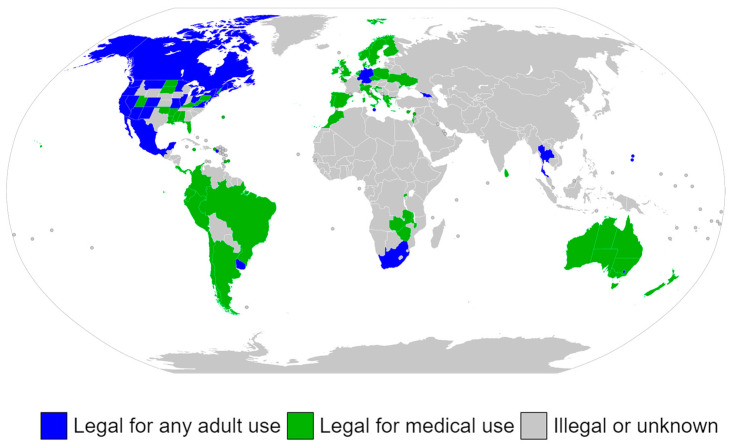
Three countries—Canada, Thailand, and Uruguay—as well as all subnational U.S. jurisdictions that have legalized possession of cannabis apart from Virginia and Washington, D.C., have permitted the commercial selling of cannabis for recreational use. Many nations have also embraced a policy of lax policing, most notably the Netherlands, where the sale of cannabis is allowed in establishments with a license [105]. Albania, Argentina, Australia, Barbados, Brazil, Canada, Chile, Colombia, Costa Rica, Croatia, Cyprus, Czech Republic, Denmark, Ecuador, Finland, Georgia, Germany, Greece, Ireland, Israel, Italy, Jamaica, Lebanon, Luxembourg, Malawi, Malta, Mexico, the Netherlands, New Zealand, North Macedonia, Norway, Panama, Peru, Poland, Portugal, Rwanda, Saint Vincent and the Grenadines, San Marino, South Africa, Spain, Sri Lanka, Switzerland, Thailand, Ukraine, the United Kingdom, Uruguay, Vanuatu, Zambia, and Zimbabwe are among the nations that have legalized cannabis for medical purposes (Figure 4). Others have more stringent regulations that only permit the use of specific medications made from cannabis, like Epidiolex, Marinol, and Sativex [106]. While the use of cannabis for medical purposes is now permitted in 38 states, 4 territories, and the District of Columbia in the U.S., it is still illegal at the federal level [106,107].
Figure 4.
The legalization of cannabis globally for therapeutic purposes [106].
7. Cannabis as a Food or Beverage
7.1. Cannabis or Hemp Food
For decades, hemp has been a valuable food source for humans. Hemp leaves, sprouts, and flowers can be eaten fresh in salads and juices [108]. The most common part of the hemp plant to be eaten is the seed. Three to six weeks after the female flower is fertilized, the real seed, an achene, matures. It is protected by a hard, inconspicuous pericarp [108]. With some notable variations among different genotypes, hemp seeds include roughly one-fourth proteins, one-fourth carbohydrates, and one-third fats. They also give 500–600 Kcal/100 g [15]. The fats found in hemp seeds are polyunsaturated. Seven different hemp seed cultivars were studied, and the results showed that “Finola” had the highest concentration of α- and γ-linolenic acids and the lowest concentration of oleic acid and saturated fatty acids like palmitic and stearic acids. The cultivars were “Bialobrzeskie”, “Felina 32”, “Tygra 75”, “Futura 27”, “Santhica”, “Fedora 17”, and “Finola” [15]. Hemp seed proteins are rich in arginine, an important amino acid with advantageous cardiovascular effects, and a good source of cystine and methionine, two amino acids that contain sulfur. Growing data suggest that hydrolyzed hemp seed proteins have antihypertensive properties, which may be mediated by the suppression of renin and the angiotensin-converting enzyme [109]. This justification led to the development of a human experiment investigating hemp protein powder as a dietary intervention for hypertension [109]. Additionally, hemp seeds can be used to create oil and flour with beneficial nutritional qualities. When hemp flour was combined with starch to generate gluten-free bread, the flavor and color palatability improved, along with the nutritional value, which included higher amounts of proteins and fiber [109]. The nutritious value of hemp flour crackers has also increased [110]. The most popular edible hemp derivative is hemp seed oil, which has potential applications in the cosmetics sector as a sun lotion because of its high vitamin E concentration (100 mg/100 mL) and ability to absorb UV radiation [111]. Hemp seed oil has a nice, nutty flavor with a slightly bitter aftertaste. It can be used in cooking instead of olive oil, which has been shown to have cardiovascular health benefits [112].
7.2. Hemp Beer
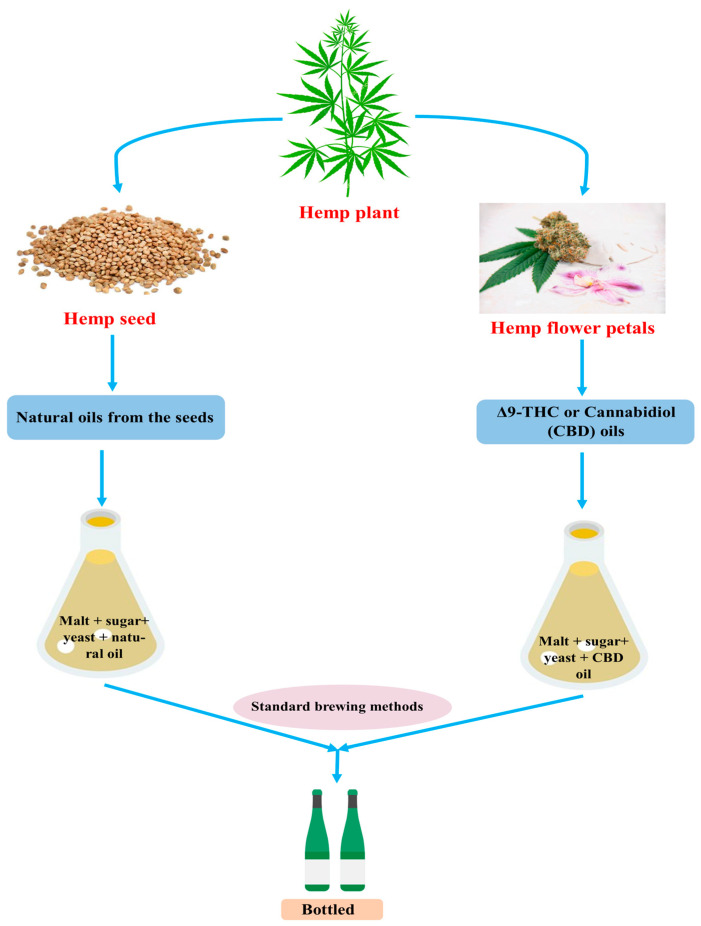
In early 2017, hemp or cannabis-infused alcohol became popular. The federal ban on marijuana and the relaxation of regulatory barriers for cannabis plant parts other than the seed, which contains psychoactive Δ9-THC, led to this outcome [10]. Conventional brewing methods are used, together with the addition of hemp or cannabis. Whole and hulled hemp seeds, hemp seed oil, and hemp seed flour are foods derived from the hemp plant and are used as flavorings in beer [113]. There are no indications that the trend of cannabis-infused beer businesses blending their products will soon come to an end. Indeed, the availability of hemp beer and cannabis-infused beer has contributed to the rise in the popularity of craft beer. Hemp beer and cannabis-infused beer, also known as CBD- or Δ9-THC-infused beer, are two types of cannabis beer made from marijuana or hemp. Hemp beer uses natural oils from seeds to enhance its flavor, while CBD-infused beers use the standard brewing method with CBD oil (Figure 5). However, beers infused with THC are not alcoholic, as it is against the law to combine alcohol and THC, limiting their alcohol content to 0.5% by volume [10]. Due to their oily and hydrophobic nature, α-acids and cannabis are both poorly soluble in water, making it challenging to directly incorporate them into drinks. On the other hand, the α-acids undergo isomerization through boiling in a mildly acidic solution, resulting in iso-α-acids that possess an organoleptic bitter taste and are more soluble. Encapsulation, microemulsions, biopolymer nanoparticles, and filled hydrogel beads are the components of the colloidal delivery method for cannabis oil. These mechanisms guarantee cannabis distribution to the user by stabilizing the compounds against oxidation in a water-based beverage [114]. There are beers with Δ9-THC infusions available that have an alcohol-by-volume (ABV) of less than 0.5% and an Δ9-THC concentration ranging from 5 to 10 mg [115]. In contrast to Δ9-THC beers, CBD-infused beers include 10 milligrams of CBD and range in alcohol content from 3.5 to 6% ABV. When consumers consume CBD beers, they report “elevating” and “naturally calming” feelings, but THC beer causes intoxication, euphoria, lethargy, and occasionally cravings. The daily consumption of foods containing hemp should not amount to more than 12 μg of Δ9-THC per kilogram of body weight, as advised by the German Federal Institute for Health Protection of Consumers and Veterinary Medicine. This estimate indicates that 5 μg/kg of THC is present in beverages, both alcoholic and non-alcoholic [116].
Figure 5.
Hemp beer and cannabis-infused beer preparation.
8. Economic Importance of Cannabis
Cannabis sativa remains one of humanity’s useful natural resources because of its multipurpose applications in medicine, food, agriculture, and biotechnology. In the early 1990s, the Emerald Triangle region of Northern California was so popular for producing medical cannabis that the plant became the major driver of their economy, dominating other business activities, such as livestock and dairy production and lumbering [117]. By 1996, California had enacted the Compassionate Use Act, which was the first to permit legal access to and use of botanical cannabis for medicinal purposes under physician supervision [118]. With this, the state of California’s revenue generation through cannabis was USD 4 billion from USD 5.7 billion in sales in 2021 and has improved substantially to USD 5.1 billion in taxable sales by the end of 2023 [119]. The United States collected nearly USD 3 billion in marijuana revenue in 2022, while nationwide legalization could generate USD 8.5 billion annually for all states [119]. Similarly, Canada has generated about CAD 11 billion in sales since cannabis was legalized for recreational use in the country in 2018, with the industry contributing CAD 43.5 billion to the country’s GDP [120]. Australia remains in the early phase of development due to the export of cannabis only being legalized in 2018. The market potential in Australia is extremely high, with at least 200,000 patients eligible for medical cannabis, so the legal cannabis market is expected to increase from the fifty-two million Australian Dollars (AUD 52 m) that was achieved in 2018 to AUD 1.2 bn by 2025, with the potential to create over 10,000 new jobs across the horticultural, pharmaceutical, and medical industries [121].
Several partnerships and collaborations exist between Australian and Canadian companies. However, Canada struggles to meet the demand in a rapidly expanding recreational cannabis market and is thus seeking new suppliers [121]. Many Canadian entrepreneurs and investors have been streaming into Lesotho and a few other African countries where cannabis strains are present in abundance. Lesotho is a lower middle-income country, with a population of 2 million people, a per capita gross domestic product (GDP) of USD 999.7 as of 2022, and just USD 750 million in its internal reserve as of January 2024, which is also close to the country’s annual revenue base [122]. Lesotho has been largely growing cannabis since approximately the 1550s through illegal means and unlawfully using it for both medicinal and recreational purposes. It was only in 2017 that Lesotho started licensing cannabis companies and regulating the cultivation of cannabis for medical purposes [123]. So far, the cannabis economy in Lesotho is yet to be fully exploited due to cultural beliefs, stigmatization, and policy limitations [123]. The neighboring South Africa has a framework that legalizes the medical and recreational use of cannabis [124]. The country has put in place policies and recent regulatory changes which, for instance, limit the THC level in cannabis to 0.2% to prevent the misuse of cannabis and circumvent regulations around cultivation [125]. With the South African government’s plan to industrialize the sector by enacting a “Cannabis for Private Purposes Bill”, among other policies still in the pipeline, the hemp sector is projected to create over 130,000 new jobs and bring in more revenue to the country [125].
Therefore, more effort needs to be put in place for phase decriminalization, proper monitoring, and the regulation of cannabis among the top-producing countries. Through this, there would be numerous economic implications for states and their constituents, including raising tax revenues; generating economic activity such as new businesses, jobs, and income; and potentially altering migration patterns and demands for real estate. Overall, the continued growth and expansion of the cannabis market are almost certain because of the increasing demand for health-promoting, eco-friendly, and sustainable hemp products. These verifiable facts provide a formidable template for manufacturers, governments, and investors to tap into this lucrative and rapidly evolving market segment to help achieve economic stability for national development.
9. Conclusions and Future Prospects
It has been highlighted through this study that Cannabis sativa, though with restricted use in many countries because of its abusive and/or illicit use, can be a useful herbal ingredient due to its unique chemical diversity and notable biological properties. It has also been shown that the beneficial and/or detrimental effects of cannabis are dependent on the quality and quantity of its secondary metabolites, notably its cannabinoid contents (CBDs, THCs, CBGs, CBCs, and CBNs). With more countries legalizing cannabis cultivation and use, the public is becoming more aware of the negative psycho-social impact of cannabis and its usefulness in medicine, agriculture, food, and industrialization. Therefore, to fully harness the bioresources offered through cannabis for food, health and wellbeing, industrialization, and economic growth, there is a need for the routine scientific standardization of cannabis cultivars, with adequate monitoring, evaluation, regulation, and support.
In the future, it is our expectation that the hemp industry will not be limited to health or recreational use but will find applications as a valuable natural resource for cosmetics, textiles, and energy productions. There is, therefore, a need for many more countries to legalize cannabis and create a regulatory framework for the inclusion of CBD- and Δ9-THC-based cannabis. This will pave the way for the development of a wide range of cannabis- and/or hemp-infused products. Interestingly, this trend has already begun, such that CBD-, Δ9-THC-, and hemp oil products now feature prominently in the catalog of pharmaceutical products, contributing significantly to health and wellbeing as well as economic growth.
Acknowledgments
The authors acknowledge the support of the African Medicinal Flora and Fauna Research Niche, Walter Sisulu University (WSU), and the APC funding by the Directorate of Research Development and Innovation, WSU, South Africa.
Author Contributions
Conceptualization, A.O.O. (Ayodeji O. Oriola) and P.K.; software, P.K.; formal analysis, A.O.O. (Ayodeji O. Oriola) and P.K.; writing—original draft preparation, P.K. and A.O.O. (Ayodeji O. Oriola); writing—review and editing, A.O.O. (Adebola O. Oyedeji); supervision, A.O.O. (Adebola O. Oyedeji). All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
This study was funded by the National Research Foundation (NRF) Cannabis Grant UID137963.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Dingha B., Sandler L., Bhowmik A., Akotsen-Mensah C., Jackai L., Gibson K., Turco R. Industrial hemp knowledge and interest among North Carolina organic farmers in the United States. Sustainability. 2019;11:2691. doi: 10.3390/su11092691. [DOI] [Google Scholar]
- 2.Brighenti V., Pellati F., Steinbach M., Maran D., Benvenuti S. Development of a new extraction technique and HPLC method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L. (hemp) J. Pharm. Biomed. Anal. 2017;143:228–236. doi: 10.1016/j.jpba.2017.05.049. [DOI] [PubMed] [Google Scholar]
- 3.Johnson R. Defining Hemp: A fact sheet. Congr. Res. Serv. 2019;R44742:1–12. [Google Scholar]
- 4.World Health Organization Cannabis. 2019. [(accessed on 30 November 2019)]. Available online: https://www.who.int/teams/mental-health-and-substance-use/alcohol-drugs-and-addictive-behaviours/drugs-psychoactive/cannabis.
- 5.Mechoulam R., Parker L.A., Gallily R. Cannabidiol: An overview of some pharmacological aspects. J. Clin. Pharmacol. 2002;42:11S–19S. doi: 10.1002/j.1552-4604.2002.tb05998.x. [DOI] [PubMed] [Google Scholar]
- 6.Callaway J.C. Hempseed as a nutritional resource: An overview. Euphytica. 2004;140:65–72. doi: 10.1007/s10681-004-4811-6. [DOI] [Google Scholar]
- 7.Chen T., He J., Zhang J., Zhang H., Qian P., Hao J., Li L. Analytical characterization of hempseed (seed of Cannabis sativa L.) oil from eight regions in China. J. Diet. Suppl. 2010;7:117–129. doi: 10.3109/19390211003781669. [DOI] [PubMed] [Google Scholar]
- 8.Roche H.M. Unsaturated fatty acids. Proc. Nutr. Soc. 1999;58:397–401. doi: 10.1017/S002966519900052X. [DOI] [PubMed] [Google Scholar]
- 9.Saini R.K., Keum Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018;203:255–267. doi: 10.1016/j.lfs.2018.04.049. [DOI] [PubMed] [Google Scholar]
- 10.Cortez D. Craft beer and marijuana cohesiveness is possible how one can learn from the others regulatory madness. Ohio State Businees Law J. 2018;12:159–196. [Google Scholar]
- 11.Davidson M., Reed S., Oosthuizen J., O’Donnell G., Gaur P., Cross M., Dennis G. Occupational health and safety in cannabis production: An Australian perspective. Int. J. Occup. Environ. Health. 2018;24:75–85. doi: 10.1080/10773525.2018.1517234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rupasinghe H.P.V., Davis A., Kumar S.K., Murray B., Zheljazkov V.D. Industrial hemp (Cannabis sativa subsp. sativa) as an emerging source for value-added functional food ingredients and nutraceuticals. Molecules. 2020;25:4078. doi: 10.3390/molecules25184078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Deng G., Du G., Yang Y., Bao Y., Liu F. Planting Density and Fertilization Evidently Influence the Fiber Yield of Hemp (Cannabis sativa L.) Agronomy. 2019;9:368. doi: 10.3390/agronomy9070368. [DOI] [Google Scholar]
- 14.Krüger M., van Eeden T., Beswa D. Cannabis sativa cannabinoids as functional ingredients in snack foods—Historical and developmental aspects. Plants. 2022;11:3330. doi: 10.3390/plants11233330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Irakli M., Tsaliki E., Kalivas A., Kleisiaris F., Sarrou E., Cook C.M. Effect οf genotype and growing year on the nutritional, phytochemical, and antioxidant properties of industrial hemp (Cannabis sativa L.) seeds. Antioxidants. 2019;8:491. doi: 10.3390/antiox8100491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.De Meijer E.P., Bagatta M., Carboni A., Crucitti P., Moliterni V.C., Ranalli P., Mandolino G. The inheritance of chemical phenotype in Cannabis sativa L. Genetics. 2003;163:335–346. doi: 10.1093/genetics/163.1.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pellati F., Brighenti V., Sperlea J., Marchetti L., Bertelli D., Benvenuti S. New Methods for the comprehensive analysis of bioactive compounds in Cannabis sativa L. (Hemp) Molecules. 2018;23:2639. doi: 10.3390/molecules23102639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brighenti V., Licata M., Pedrazzi T., Maran D., Bertelli D., Pellati F., Benvenuti S. Development of a new method for the analysis of cannabinoids in honey by means of high-performance liquid chromatography coupled with electrospray ionisation-tandem mass spectrometry detection. J. Chromatogr. 2019;1597:179–186. doi: 10.1016/j.chroma.2019.03.034. [DOI] [PubMed] [Google Scholar]
- 19.Arendt S. Sourcing Natural Ingredients. Everything about Hemp. Wander; Frederick, MD, USA: 2019. [Google Scholar]
- 20.Boffman J. CBD 101: The Basics and Product Navigation. Everything about Hemp. Thriv Nutraceuticals; Frederick, MD, USA: 2019. pp. 1–4. [Google Scholar]
- 21.Głodowska M., Łyszcz M. Cannabis sativa L. and its antimicrobial properties—A review. [(accessed on 30 July 2024)];Strona. 2016 :77–82. Available online: https://www.researchgate.net/publication/317185536_Cannabis_sativa_L_and_its_antimicrobial_properties_-_A_review. [Google Scholar]
- 22.Pertwee R.G. Handbook of Experimental Pharmacology. Volume 168. Springer; Berlin/Heidelberg, Germany: 2005. Pharmacological Actions of Cannabinoids. [DOI] [PubMed] [Google Scholar]
- 23.Oza M., William B., Gummadidala P.M., Dias T., Omebeyinje M.H., Chen L., Mitra C., Jesmin R., Chakraborty P., Sajish M., et al. Acute and short-term administrations of delta-9- tetrahydrocannabinol modulate major gut metabolomic regulatory pathways in C57BL/6 mice. Sci. Rep. 2019;9:10520. doi: 10.1038/s41598-019-46478-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mueller R.L., Ellingson J.M., Bidwell L.C., Bryan A.D., Hutchison K.E. Are the Acute Effects of THC Different in Aging Adults? Brain Sci. 2021;11:590. doi: 10.3390/brainsci11050590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Freeman T.P., Hindocha C., Green S.F., Bloomfield M.A. Medicinal use of cannabis-based products and cannabinoids. Br. Med. J. 2019;1141:1–7. doi: 10.1136/bmj.l1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Andre C.M., Hausman J., Guerriero G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016;7:19. doi: 10.3389/fpls.2016.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pavlovic R., Nenna G., Calvi L., Panseri S., Borgonovo G., Giupponi L., Cannazza G., Giorgi A. Quality traits of cannabidiol oils: Cannabinoids content, terpene fingerprint and oxidation stability of European commercially available preparations. Molecules. 2018;23:1230. doi: 10.3390/molecules23051230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Skřivan M., Englmaierová M., Vít T., Skřivanová E. Hempseed increases gamma-tocopherol in egg yolks and the breaking strength of tibias in laying hens. PLoS ONE. 2019;14:e0217509. doi: 10.1371/journal.pone.0217509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Whyte L.S., Ryberg E., Sims N.A., Ridge S.A., Mackie K., Greasley P.J., Ross R.A., Rogers M.J. The putative cannabinoid receptor GPR55 affects osteoclast function in vitro and bone mass in vivo. Proc. Natl. Acad. Sci. USA. 2009;106:16511–16516. doi: 10.1073/pnas.0902743106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Food and Drug Administration (FDA) FDA Regulation of Cannabis and Cannabis-Derived Products, Including Cannabidiol (CBD). Public Health Focus, Food and Drug Administration. [(accessed on 24 July 2024)];2021 Available online: https://www.fda.gov/news-events/public-health-focus/fda-regulation-cannabis-and-cannabis-derived-products-including-cannabidiol-cbd#farmbill.
- 31.Iseppi R., Brighenti V., Licata M., Lambertini A., Sabia C., Messi P., Pellati F., Benvenuti S. Chemical characterization and evaluation of the antibacterial activity of essential oils from fibre-type Cannabis sativa L. (Hemp) Molecules. 2019;24:2302. doi: 10.3390/molecules24122302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Novak J., Zitterl-Eglseer K., Deans S.G., Franz C.M. Essential oils of different cultivars of Cannabis sativa L. and their antimicrobial activity. Flavour Fragr. J. 2001;16:259–262. doi: 10.1002/ffj.993. [DOI] [Google Scholar]
- 33.Frassinetti S., Gabriele M., Moccia E., Longo V., Di Gioia D. Antimicrobial and antibiofilm activity of Cannabis sativa L. seeds extract against Staphylococcus aureus and growth effects on probiotic Lactobacillus spp. LWT. 2020;124:109149. doi: 10.1016/j.lwt.2020.109149. [DOI] [Google Scholar]
- 34.Anjum M. 35. Evaluation of antimicrobial activity and ethnobotanical study of Cannabis sativa L. Pure Appl. Biol. 2018;7:706–713. doi: 10.19045/bspab.2018.70088. [DOI] [Google Scholar]
- 35.Manosroi A., Chankhampan C., Kietthanakorn B.O., Ruksiriwanich W., Chaikul P., Boonpisuttinant K., Sainakham M., Manosroi W., Tangjai T., Manosroi J. Pharmaceutical and cosmeceutical biological activities of hemp (Cannabis sativa L. var. sativa) leaf and seed extracts. Chiang Mai J. Sci. 2019;46:180–195. [Google Scholar]
- 36.Nafis A., Kasrati A., Jamali C.A., Mezrioui N., Setzer W., Abbad A., Hassani L. Antioxidant activity and evidence for synergism of Cannabis sativa (L.) essential oil with antimicrobial standards. Ind. Crops Prod. 2019;137:396–400. doi: 10.1016/j.indcrop.2019.05.032. [DOI] [Google Scholar]
- 37.El Hamdaoui A., Msanda F., Boubaker H., Leach D., Bombarda I., Vanloot P., El Aouad N., Abbad A., Boudyach E., Achemchem F. Essential oil composition, antioxidant and antibacterial activities of wild and cultivated Lavandula mairei Humbert. Biochem. Syst. Ecol. 2018;76:1–7. doi: 10.1016/j.bse.2017.11.004. [DOI] [Google Scholar]
- 38.Prabuseenivasan S., Jayakumar M., Ignacimuthu S. In vitro antibacterial activity of some plant essential oils. BMC Complement. Altern. Med. 2006;6:1–8. doi: 10.1186/1472-6882-6-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.El Yaagoubi M., Mechqoq H., Ortiz S., Cavaleiro C., Lecsö-Bornet M., Pereira C.G., Rodrigues M.J., Custódio L., El Mousadik A., Picot L. Chemical composition and biological screening of the essential oils of Micromeria macrosiphon and M. arganietorum (Lamiaceae) Chem. Biodivers. 2021;18:e2100653. doi: 10.1002/cbdv.202100653. [DOI] [PubMed] [Google Scholar]
- 40.Coetzee C., Levendal R.-A., Van de Venter M., Frost C. Anticoagulant effects of a cannabis extract in an obese rat model. Phytomedicine. 2007;14:333–337. doi: 10.1016/j.phymed.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 41.Xiong W.T., Gu L., Wang C., Sun H.-X., Liu X. Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J. Ethnopharmacol. 2013;150:935–945. doi: 10.1016/j.jep.2013.09.027. [DOI] [PubMed] [Google Scholar]
- 42.Agarwal P., Gupta R. Alpha-amylase inhibition can treat diabetes mellitus. Res. Rev. J. Med. Health Sci. 2016;5:1–8. [Google Scholar]
- 43.Kajaria D., Tripathi J., Tripathi Y.B., Tiwari S. In-vitro α-amylase and glycosidase inhibitory effect of ethanolic extract of antiasthmatic drug—Shirishadi. J. Adv. Pharm. Technol. Res. 2013;4:206. doi: 10.4103/2231-4040.121415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zengin G., Menghini L., Di Sotto A., Mancinelli R., Sisto F., Carradori S., Cesa S., Fraschetti C., Filippi A., Angiolella L. Chromatographic analyses, in vitro biological activities, and cytotoxicity of Cannabis sativa L. essential oil: A multidisciplinary study. Molecules. 2018;23:3266. doi: 10.3390/molecules23123266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kar P., Banerjee S., Saleh-E-In M.M., Anandraj A., Kormuth E., Pillay S., Al-Ghamdi A.A., Ali M.A., Lee J., Sen A., et al. β-sitosterol conjugated silver nanoparticle-mediated amelioration of CCl4-induced liver injury in Swiss albino mice. J. King Saud Univ. Sci. 2022;34:102113. doi: 10.1016/j.jksus.2022.102113. [DOI] [Google Scholar]
- 46.Ligresti A., Moriello A.S., Starowicz K., Matias I., Pisanti S., De Petrocellis L., Laezza C., Portella G., Bifulco M., Di Marzo V. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. J. Pharmacol. Exp. Ther. 2006;318:1375–1387. doi: 10.1124/jpet.106.105247. [DOI] [PubMed] [Google Scholar]
- 47.Sarfaraz S., Afaq F., Adhami V.M., Malik A., Mukhtar H. Cannabinoid receptor agonist-induced apoptosis of human prostate cancer cells LNCaP proceeds through sustained activation of ERK1/2 leading to G1 cell cycle arrest. J. Biol. Chem. 2006;281:39480–39491. doi: 10.1074/jbc.M603495200. [DOI] [PubMed] [Google Scholar]
- 48.Lukhele S.T., Motadi L.R. Cannabidiol rather than Cannabis sativa extracts inhibit cell growth and induce apoptosis in cervical cancer cells. BMC Complement. Altern. Med. 2016;16:335. doi: 10.1186/s12906-016-1280-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Marcu J.P., Christian R.T., Lau D., Zielinski A.J., Horowitz M.P., Lee J., Pakdel A., Allison J., Limbad C., Moore D.H. Cannabidiol enhances the inhibitory effects of Δ9-tetrahydrocannabinol on human glioblastoma cell proliferation and survival: Cannabinoid synergy inhibits glioblastoma cell growth. Mol. Cancer Ther. 2010;9:180–189. doi: 10.1158/1535-7163.MCT-09-0407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cianchi F., Papucci L., Schiavone N., Lulli M., Magnelli L., Vinci M.C., Messerini L., Manera C., Ronconi E., Romagnani P. Cannabinoid receptor activation induces apoptosis through tumor necrosis factor-mediated ceramide de novo synthesis in colon cancer cells. Clin. Cancer Res. 2008;14:7691–7700. doi: 10.1158/1078-0432.CCR-08-0799. [DOI] [PubMed] [Google Scholar]
- 51.McKallip R.J., Lombard C., Fisher M., Martin B.R., Ryu S., Grant S., Nagarkatti P.S., Nagarkatti M. Targeting CB2 cannabinoid receptors as a novel therapy to treat malignant lymphoblastic disease. Blood J. Am. Soc. Hematol. 2002;100:627–634. doi: 10.1182/blood-2002-01-0098. [DOI] [PubMed] [Google Scholar]
- 52.Armstrong J.L., Hill D.S., McKee C.S., Hernandez-Tiedra S., Lorente M., Lopez-Valero I., Anagnostou M.E., Babatunde F., Corazzari M., Redfern C.P. Exploiting cannabinoid-induced cytotoxic autophagy to drive melanoma cell death. J. Investig. Dermatol. 2015;135:1629–1637. doi: 10.1038/jid.2015.45. [DOI] [PubMed] [Google Scholar]
- 53.Borrelli F., Pagano E., Romano B., Panzera S., Maiello F., Coppola D., De Petrocellis L., Buono L., Orlando P., Izzo A.A. Colon carcinogenesis is inhibited by the TRPM8 antagonist cannabigerol, a Cannabis-derived non-psychotropic cannabinoid. Carcinogenesis. 2014;35:2787–2797. doi: 10.1093/carcin/bgu205. [DOI] [PubMed] [Google Scholar]
- 54.Galanti G., Fisher T., Kventsel I., Shoham J., Gallily R., Mechoulam R., Lavie G., Amariglio N., Rechavi G., Toren A. D9-Tetrahydrocannabinol inhibits cell cycle progression by downregulation of E2F1 in human glioblastoma multiforme cells. Acta Oncol. 2008;47:1062–1070. doi: 10.1080/02841860701678787. [DOI] [PubMed] [Google Scholar]
- 55.Galve-Roperh I., Sánchez C., Cortés M.L., del Pulgar T.G., Izquierdo M., Guzmán M. Anti-tumoral action of cannabinoids: Involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation. Nat. Med. 2000;6:313–319. doi: 10.1038/73171. [DOI] [PubMed] [Google Scholar]
- 56.McAllister S.D., Murase R., Christian R.T., Lau D., Zielinski A.J., Allison J., Almanza C., Pakdel A., Lee J., Limbad C. Pathways mediating the effects of cannabidiol on the reduction of breast cancer cell proliferation, invasion, and metastasis. Breast Cancer Res. Treat. 2011;129:37–47. doi: 10.1007/s10549-010-1177-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Solinas M., Massi P., Cinquina V., Valenti M., Bolognini D., Gariboldi M., Monti E., Rubino T., Parolaro D. Cannabidiol, a non-psychoactive cannabinoid compound, inhibits proliferation and invasion in U87-MG and T98G glioma cells through a multitarget effect. PLoS ONE. 2013;8:e76918. doi: 10.1371/journal.pone.0076918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Klein T.W. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat. Rev. Immunol. 2005;5:400–411. doi: 10.1038/nri1602. [DOI] [PubMed] [Google Scholar]
- 59.Nagarkatti P., Pandey R., Rieder S.A., Hegde V.L., Nagarkatti M. Cannabinoids as novel anti-inflammatory drugs. Future Med. Chem. 2009;1:1333–1349. doi: 10.4155/fmc.09.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wood C., Duparc N., Leblanc V., Cunin-Roy C. L’hypnose et la douleur. Médecine Clin. Pour Les Pédiatres. 2004;11:40–44. [Google Scholar]
- 61.Munch G. Ph.D. Thesis. Université de Lorraine; Lorraine, France: 2015. Le Cannabis, les deux Versants: Drogue et Medicament. [Google Scholar]
- 62.Jeannin C. Master’s Thesis. University of Liege; Liege, Belgium: 2020. Évaluation et Prise en Charge de la Douleur chez le lapin de Compagnie: Comment les Optimiser en l’état Actuel des Connaissances? [Google Scholar]
- 63.Li J., Wang G., Qin Y., Zhang X., Wang H.-F., Liu H.W., Zhu L.J., Yao X.S. Neuroprotective constituents from the aerial parts of Cannabis sativa L. subsp. sativa. RSC Adv. 2020;10:32043–32049. doi: 10.1039/D0RA04565A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Landucci E., Mazzantini C., Lana D., Davolio P.L., Giovannini M.G., Pellegrini-Giampietro D.E. Neuroprotective effects of cannabidiol but not Δ9-tetrahydrocannabinol in rat hippocampal slices exposed to oxygen-glucose deprivation: Studies with cannabis extracts and selected cannabinoids. Int. J. Mol. Sci. 2021;22:9773. doi: 10.3390/ijms22189773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.di Giacomo V., Chiavaroli A., Recinella L., Orlando G., Cataldi A., Rapino M., Di Valerio V., Ronci M., Leone S., Brunetti L. Antioxidant and neuroprotective effects induced by cannabidiol and cannabigerol in rat CTX-TNA2 astrocytes and isolated cortexes. Int. J. Mol. Sci. 2020;21:3575. doi: 10.3390/ijms21103575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Esposito G., Scuderi C., Savani C., Steardo Jr L., De Filippis D., Cottone P., Iuvone T., Cuomo V., Steardo L. Cannabidiol in vivo blunts β-amyloid induced neuroinflammation by suppressing IL-1 and iNOS expression. Br. J. Pharmacol. 2007;151:1272–1279. doi: 10.1038/sj.bjp.0707337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Perez M., Benitez S.U., Cartarozzi L.P., Del Bel E., Guimaraes F.S., Oliveira A.L. Neuroprotection and reduction of glial reaction by cannabidiol treatment after sciatic nerve transection in neonatal rats. Eur. J. Neurosci. 2013;38:3424–3434. doi: 10.1111/ejn.12341. [DOI] [PubMed] [Google Scholar]
- 68.Gierthmühlen J., Baron R. Neuropathic Pain. Semin. Neurol. 2016;36:462–468. doi: 10.1055/s-0036-1584950. [DOI] [PubMed] [Google Scholar]
- 69.Szok D., Tajti J., Nyári A., Vécsei L. Therapeutic approaches for peripheral and central neuropathic pain. Behav. Neurol. 2019;2019:8685954. doi: 10.1155/2019/8685954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Van De Donk T., Niesters M., Kowal M., Olofsen E., Dahan A., Van Velzen M. An experimental randomized study on the analgesic effects of pharmaceutical-grade cannabis in chronic pain patients with fibromyalgia. Pain. 2019;160:860–869. doi: 10.1097/j.pain.0000000000001464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Poli P., Crestani F., Salvadori C., Valenti I., Sannino C. Medical cannabis in patients with chronic pain: Effect on pain relief, pain disability, and psychological aspects. A prospective non-randomized single arm clinical trial. Clin. Ter. 2018;169:e102–e107. doi: 10.7417/T.2018.2062. [DOI] [PubMed] [Google Scholar]
- 72.Russo E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011;163:1344–1364. doi: 10.1111/j.1476-5381.2011.01238.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rahn E.J., Hohmann A.G. Cannabinoids as pharmacotherapies for neuropathic pain: From the bench to the bedside. Neurotherapeutics. 2009;6:713–737. doi: 10.1016/j.nurt.2009.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Deiana S. Potential medical uses of cannabigerol: A brief overview. In: Preedy V.R., editor. Handbook of Cannabis and Related Pathologies. Academic Press; Cambridge, MA, USA: 2017. pp. 958–967. [Google Scholar]
- 75.Eldeeb K., Leone-Kabler S., Howlett A. Comparison of Δ9-tetrahydrocannabinolic acid A (THCA-A) and Delta-9-tetrahydrocannabinol (THC) in neuronal cell functions. J. Pharmacol. Exp. Ther. 2023;385:551. [Google Scholar]
- 76.Russo E.B. Cannabidiol claims and misconceptions. Trends Pharmacol. Sci. 2017;38:198–201. doi: 10.1016/j.tips.2016.12.004. [DOI] [PubMed] [Google Scholar]
- 77.Burstein S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorg. Med. Chem. 2015;23:1377–1385. doi: 10.1016/j.bmc.2015.01.059. [DOI] [PubMed] [Google Scholar]
- 78.Ujváry I., Hanuš L. Human metabolites of cannabidiol: A review on their formation, biological activity, and relevance in therapy. Cannabis Cannabinoid Res. 2016;1:90–101. doi: 10.1089/can.2015.0012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kim J., Choi P., Park Y.T., Kim T., Ham J., Kim J.C. The cannabinoids, CBDA and THCA, rescue memory deficits and reduce amyloid-beta and Tau pathology in an Alzheimer’s disease-like mouse model. Int. J. Mol. Sci. 2023;24:6827. doi: 10.3390/ijms24076827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.McPartland J.M., Russo E.B. Cannabis and cannabis extracts: Greater than the sum of their parts? J. Cannabis Ther. 2001;1:103–131. doi: 10.1300/J175v01n03_08. [DOI] [Google Scholar]
- 81.Muscarà C., Smeriglio A., Trombetta D., Mandalari G., La Camera E., Grassi G., Circosta C. Phytochemical characterization and biological properties of two standardized extracts from a non-psychotropic Cannabis sativa L. cannabidiol (CBD)-chemotype. Phytother. Res. 2021;35:5269–5281. doi: 10.1002/ptr.7201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hartsel J.A., Eades J., Hickory B., Makriyannis A. Cannabis sativa and hemp. In: Gupta R.C., editor. Nutraceuticals. Academic Press; Cambridge, MA, USA: 2016. pp. 735–754. [Google Scholar]
- 83.El-Alfy A.T., Ivey K., Robinson K., Ahmed S., Radwan M., Slade D., Khan I., ElSohly M., Ross S. Antidepressant-like effect of Δ9-tetrahydrocannabinol and other cannabinoids isolated from Cannabis sativa L. Pharmacol. Biochem. Behav. 2010;95:434–442. doi: 10.1016/j.pbb.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Galletta M., Reekie T.A., Nagalingam G., Bottomley A.L., Harry E.J., Kassiou M., Triccas J.A. Rapid antibacterial activity of cannabichromenic acid against methicillin-resistant Staphylococcus aureus. Antibiotics. 2020;9:523. doi: 10.3390/antibiotics9080523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Dawidowicz A.L., Olszowy-Tomczyk M., Typek R. CBG, CBD, Δ9-THC, CBN, CBGA, CBDA and Δ9-THCA as antioxidant agents and their intervention abilities in antioxidant action. Fitoterapia. 2021;152:104915. doi: 10.1016/j.fitote.2021.104915. [DOI] [PubMed] [Google Scholar]
- 86.Cherney J., Small E. Industrial hemp in North America: Production, politics and potential. Agronomy. 2016;6:58. doi: 10.3390/agronomy6040058. [DOI] [Google Scholar]
- 87.Carus M., Reinders M., Baňas B., Frank B., Kruse D., Sfrija D. The Cologne Declaration on Industrial Hemp. European Industrial Hemp Association (EIHA); Hürth, Germany: 2017. [(accessed on 30 July 2024)]. Available online: https://eiha.org/media/2017/07/17-06-06%20EIHA%20Cologne%20Declaration_final.pdf. [Google Scholar]
- 88.Amaducci S., Errani M., Venturi G. Response of hemp to plant population and nitrogen fertilization. Ital. J. Agron. 2002;6:103–111. [Google Scholar]
- 89.AheadIntel Cannabis Market Report and Patent Landscape Analysis-2025. 2020. [(accessed on 11 July 2020)]. Available online: https://www.aheadintel.com/cannabis-market/
- 90.Hall W., Stjepanović D., Caulkins J., Lynskey M., Leung J., Campbell G., Degenhardt L. Public health implications of legalising the production and sale of cannabis for medicinal and recreational use. The Lancet. 2019;394:1580–1590. doi: 10.1016/S0140-6736(19)31789-1. [DOI] [PubMed] [Google Scholar]
- 91.Gunn J.K., Rosales C.B., Center K.E., Nuñez A., Gibson S.J., Christ C., Ehiri J.E. Prenatal exposure to cannabis and maternal and child health outcomes: A systematic review and meta-analysis. BMJ Open. 2016;6:e009986. doi: 10.1136/bmjopen-2015-009986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Coffey C., Patton G.C. Cannabis use in adolescence and young adulthood: A review of findings from the Victorian Adolescent Health Cohort Study. Can. J. Psychiatry. 2016;6:318–327. doi: 10.1177/0706743716645289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Scott J.C., Slomiak S.T., Jones J.D., Rosen A.F.G., Moore T.M., Gur R.C. Association of cannabis with cognitive functioning in adolescents and young adults: A systematic review and meta-analysis. JAMA Psychiatry. 2018;75:585–595. doi: 10.1001/jamapsychiatry.2018.0335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Stiby A.I., Hickman M., Munafò M.R., Heron J., Yip V.L., Macleod J. Adolescent cannabis and tobacco use and educational outcomes at age 16: Birth cohort study. Addiction. 2015;110:658–668. doi: 10.1111/add.12827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Moore T.H., Zammit S., Lingford-Hughes A., Barnes T.R., Jones P.B., Burke M., Lewis G. Cannabis use and risk of psychotic or affective mental health outcomes: A systematic review. The Lancet. 2007;370:319–328. doi: 10.1016/S0140-6736(07)61162-3. [DOI] [PubMed] [Google Scholar]
- 96.Borges G., Bagge C.L., Orozco R. A literature review and meta-analyses of cannabis use and suicidality. J. Affect. Disord. 2016;195:63–74. doi: 10.1016/j.jad.2016.02.007. [DOI] [PubMed] [Google Scholar]
- 97.Gage S.H., Hickman M., Heron J., Munafò M.R., Lewis G., Macleod J., Zammit S. Associations of cannabis and cigarette use with depression and anxiety at age 18: Findings from the Avon longitudinal study of parents and children. PLoS ONE. 2015;10:e0122896. doi: 10.1371/journal.pone.0122896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bhandari S., Jha P., Lisdahl K.M., Hillard C.J., Venkatesan T. Recent trends in cyclic vomiting syndrome—Associated hospitalizations with liberalization of cannabis use in the state of Colorado. Intern. Med. J. 2018;49:649–655. doi: 10.1111/imj.14164. [DOI] [PubMed] [Google Scholar]
- 99.Habibi R., Hoffman S.J. Legalizing cannabis violates the UN drug control treaties, but progressive countries like Canada have options. Ottawa Law Rev. 2017;49:427. [Google Scholar]
- 100.Bewley-Taylor D., Jelsma M., Rolles S., Walsh J. Cannabis Regulation and the UN Drug Treaties: Strategies for Reform. 2016. [(accessed on 26 April 2024)]. Available online: https://www.tni.org/files/publication-downloads/cannabis_regulation_and_the_un_drug_treaties_june_2016_web_0.pdf.
- 101.Chappell B.U.N. Commission Removes Cannabis from Its Most Strict Drug Control List. NPR. 2020. [(accessed on 30 July 2024)]. Available online: https://www.npr.org/2020/12/02/941283185/u-n-commission-removes-cannabis-from-its-most-strict-drug-control-list#:~:text=Dray%2FGetty%20Images-,The%20U.N.,deems%20cannabis%20a%20controlled%20substance.
- 102.European Monitoring Centre for Drugs and Drug Addiction . European Monitoring Centre for Drugs and Drug Addiction. Volume 2 Office for Official Publications of the European Communities; Luxembourg: 2003. [Google Scholar]
- 103.Powell B. The 7 Countries with the Strictest Weed Laws. High Times. 2018. [(accessed on 26 April 2024)]. Available online: https://web.archive.org/web/20201126132111/https://hightimes.com/guides/countries-strictest-weed-laws/
- 104.Farrelly K.N., Wardell J.D., Marsden E., Scarfe M.L., Najdzionek P., Turna J., MacKillop J. The impact of recreational cannabis legalization on cannabis use and associated outcomes: A systematic review. Subst. Abuse. 2023;17:11782218231172054. doi: 10.1177/11782218231172054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Knottnerus J.A., Blom T., van Eerden S., Mans J.H.H., Mheen D.V., de Neeling J.N.D., Schelfhout D.C.L., Seidell J.C., van Wijk A.H., van Wingerde C.G.K., et al. Cannabis policy in The Netherlands: Rationale and design of an experiment with a controlled legal (‘closed’) cannabis supply chain. Health Policy. 2023;129:104699. doi: 10.1016/j.healthpol.2022.12.007. [DOI] [PubMed] [Google Scholar]
- 106.Baratta F., Pignata I., Ravetto Enri L., Brusa P. Cannabis for medical use: Analysis of recent clinical trials in view of current legislation. Front. Pharmacol. 2022;13:888903. doi: 10.3389/fphar.2022.888903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.National Conference of State Legislatures (NCSL) Medical Use of Cannabis. 2023. [(accessed on 26 April 2024)]. Available online: https://www.ncsl.org/health/state-medical-cannabis-laws.
- 108.Chandra S., Lata H., ElSohly M.A. Cannabis sativa L.—Botany and Biotechnology. Springer; New York, NY, USA: 2017. [Google Scholar]
- 109.Aluko R.E. Hemp Seed (Cannabis sativa L.) Proteins: Composition, Structure, Enzymatic Modification, and Functional or Bioactive Properties. Sustainable Protein Sources. Academic Press; Cambridge, MA, USA: 2017. pp. 121–132. [Google Scholar]
- 110.Radočaj O., Dimić E., Tsao R. Effects of hemp (Cannabis sativa L.) seed oil press-cake and decaffeinated green tea leaves (Camellia sinensis) on functional characteristics of gluten-free crackers. J. Food Sci. 2014;79:318–325. doi: 10.1111/1750-3841.12370. [DOI] [PubMed] [Google Scholar]
- 111.Oomah B.D., Busson M., Godfrey D.V., Drover J.C. Characteristics of hemp (Cannabis sativa L.) seed oil. Food Chem. 2002;76:33–43. doi: 10.1016/S0308-8146(01)00245-X. [DOI] [Google Scholar]
- 112.Rabrenović B.B., Vujasinović V.B. Industrial Hemp. Academic Press; Cambridge, MA, USA: 2022. Industrial hempseed oil and lipids: Processing and properties; pp. 95–124. [DOI] [Google Scholar]
- 113.Small E., Marcus D. Hemp: A new crop with new uses for North America. In: Janick J., Whipkey A., editors. Trends in New Crops and New Uses. ASHS Press; Norfolk, VA, USA: 2002. pp. 284–326. [Google Scholar]
- 114.Marangoni I.P., Marangoni A.G. Cannabis edibles: Dosing, encapsulation, and stability considerations. Curr. Opin. Food Sci. 2019;28:1–6. doi: 10.1016/j.cofs.2019.01.005. [DOI] [Google Scholar]
- 115.Yurasek A.M., Aston E.R., Metrik J. Co-use of alcohol and cannabis: A review. Curr. Addict. Rep. 2017;4:184–193. doi: 10.1007/s40429-017-0149-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.The German Federal Institute for Risk Assessment (BfR) BgVV Recommends Guidance Values for THC (Tetrahydrocannabinol) in Hemp-Containing Foods, Bundesinstitut für Risikobewertung (BfR) 2000. [(accessed on 12 June 2024)]. Available online: https://www.bfr.bund.de/en/presseinformation/2000/07/bgvv_recommends_guidance_values_for_thc__tetrahydrocannabinol__in_hemp_containing_foods-1309.html.
- 117.Harper M.L. A History Cannabis as a Cultural Communication Artifact. Communication Senior Capstones. 2022. 30. [(accessed on 12 July 2024)]. Available online: https://digitalcommons.humboldt.edu/senior_comm/3.
- 118.Bridgeman M.B., Abazia D.T. Medicinal cannabis: History, pharmacology, and implications for the acute care setting. Pharm. Ther. 2017;42:180. [PMC free article] [PubMed] [Google Scholar]
- 119.Cannabis Business Times. 2024. [(accessed on 12 June 2024)]. Available online: https://www.cannabisbusinesstimes.com/news/california-cannabis-market-sales-taxes-billion-2024/#:~:text=Cannabis%20Business%20Times-California%27s%20Adult%2DUse%20Cannabis%20Market%20Eclipsed%20%245.1%20Billion%20in%20%27Taxable,year%2Dover%2Dyear%20decline.
- 120.Baldavoo K., Hassen S. Evaluating the revenue and taxation implications of cannabis legalization in South Africa: Insights from Canada and the United States. Econ. Manag. Sustain. 2024;9:43–52. doi: 10.14254/jems.2024.9-1.3. [DOI] [Google Scholar]
- 121.Roberts S., Kennis M. Special Report on ASX-Listed Cannabis and Hemp Stocks. Pitt Strict Research. 2020. [(accessed on 12 July 2024)]. Available online: https://static1.squarespace.com/static/5af533a312b13fb602fe7d7b/t/5e9e9eada22fda7ad4f840f6/1587453622641/Pitt+Street+Research+Cannabis+and+Hemp+Special+Report+24+March+2020.pdf.
- 122.World Bank. 2023. [(accessed on 12 June 2024)]. Available online: https://www.worldbank.org/en/country/lesotho/overview.
- 123.Thetsane R.M. Envisaging challenges for the emerging medicinal cannabis sector in Lesotho. J. Cannabis Res. 2024;6:23. doi: 10.1186/s42238-024-00229-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Adebisi Y.A., Quazeem Olaoye D. Medical use of cannabis in Africa: The pharmacists’ perspective. Innov. Pharm. 2022;13:10. doi: 10.24926/iip.v13i1.4430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Akhtar S. Ph.D. Thesis. University of Pretoria; Pretoria, South Africa: 2021. Cannabinoid Profile and Regulatory Compliance of Non-Scheduled Cannabinoid-Containing Products in South Africa. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.