Abstract
Patient: Female, 42-year-old
Final Diagnosis: Cutaneous metastasis of breast cancer
Symptoms: Mass
Clinical Procedure: —
Specialty: Surgery
Objective:
Unusual clinical course
Background:
Breast cancer (BC) is the most common malignant disease in females and one of the leading causes of death worldwide. Its treatment plan includes a long-term follow-up and close surveillance, as recurrence is a well-acknowledged concern. BC can recur either locally or as a metastasis, and skin metastasis is a common complication in advanced breast cancer patients. It can present as a skin nodule, plaque, or erythematous lesion, and can be difficult to distinguish from benign skin conditions. The risk of skin metastasis is higher in patients with inflammatory BC. Treatment of such a complex condition is even more challenging, with poor prognosis. Here, we report a case of a 42-year-old woman with stage 4 luminal A BC who had soft tissue recurrence.
Case Report:
A 42-year-old woman with a history of left-sided BC diagnosed and treated 10 years ago presented with multiple soft tissue masses mimicking abscesses at the right lower middle of the back, bilateral thighs, and back of the neck, in the last 6 months, the largest measuring 8×10 cm. The masses were found to be metastatic BC that had spread to the skin and lungs. Because it was invasive ductal carcinoma with positive ER and PR receptors, she was started on hormonal treatment and chemotherapy.
Conclusions:
This case report highlights the importance of follow-up in patients with a history of BC, as the cancer can recur and spread many years after treatment.
Key words: Breast Neoplasms, Medical Oncology, Neoplasm Metastasis, Surgical Oncology
Introduction
Breast cancer (BC) is the most common malignancy among women globally and the second leading cause of cancer-related death worldwide. One of the most concerning aspects in BC is metastasis. Although breast cancer typically spreads to lymph nodes, bone, liver, and lungs, there have been reports of BC metastasis to less common sites [1].
Brain metastases from BC is considered rare. However, because of the improved diagnosis and a longer illness course, the incidence rate has progressively increased. Brain metastasis is more common in BC subgroups such as HER2-positive and triple-negative breast cancer [2]. BC metastases to the ovaries is also uncommon, but it is clinically significant since it can mimic main ovarian cancers, making the diagnosis difficult. Abdominal discomfort, bloating, and increased abdominal girth are common clinical manifestations. It is common in advanced BC and is associated with a poor prognosis [3,4]. Gastric metastasis is rare. It often presents as a gastric ulcer or gastrointestinal hemorrhage. Invasive lobular carcinoma is more commonly associated with gastric metastases than invasive ductal carcinoma [5].
BC skin metastasis, also known as cutaneous metastasis, occurs in around 20% of all advanced BC patients. It usually presents as a skin nodule, plaque, or erythematous lesion, and its clinical appearance can be similar to that of benign skin conditions.
A review of the literature showed that the incidence of cutaneous metastases varies between 0.7% and 10.0% for all types of carcinomas. Likewise, a more recent meta-analysis [6] showed that the overall incidence was 5.3%. Women with breast cancer are more likely than those with other malignancies to develop metastatic cutaneous lesions, followed by lung, colon, rectal, renal, ovarian, and bladder cancer [6].
Because of the existence of dermal lymphatic invasion, inflammatory BC has a greater risk of skin metastases [5,7]. Metastases often present as skin-colored nodules on the anterior chest wall, typically from lymphatic extension to the overlying skin. Adenocarcinoma has the greatest risk of skin metastases among the histologic subtypes of breast cancer, ranging from 77% to 82% of cases. BC has been linked to a variety of well-described clinical manifestations, including carcinoma erysiploides, carcinoma telangiectoides, carcinoma en cuirasse, and alopecia neoplastica [8–10].
This case report emphasizes the importance of follow-up and surveillance in patients with a history of BC, as the cancer can recur and spread many years after treatment. Early detection of recurrence essential for patient survival and prognosis.
Case Report
Our patient was a 42-year-old woman with a history of left-sided BC diagnosed treated 10 years ago outside our institution, who previously underwent left lumpectomy followed by adjuvant chemotherapy, radiotherapy, and hormonal therapy for 5 years. She presented to the outpatient department with multiple masses at the right lower middle of the back, bilateral thighs, and back of the neck. The masses had been present for 6 months and were getting bigger, especially the back mass, which was had serous discharge. She also reported a history of weight loss, decreased appetite, and subjective fever.
Physical examination revealed a morbidly obese woman with ECOG 1 of physical activity. Physical examination revealed a 4×8 cm neck mass located in the midline, associated with fluctuation, hotness, tenderness, and neck stiffness. Also, there was another mass in the lower back in the midline, measuring approximately 8×10 cm which was accompanied with fluctuation, erythema, hotness, and mild tenderness. Upon further exposure, bilateral small masses about 1–2 cm were barely palpated in the thighs. Breast examination was notable for a 2×2 cm firm, mobile, right-sided mass in the upper outer quadrant. The axillae were normal. Multiple skin abscesses were initially suspected, so she was admitted for further investigations and treatment.
Laboratory test results were within normal ranges. Serology studies were sent to rule out underlying infection such as HIV and tuberculosis, but cultures were negative. Hb A1C was sent and was diagnostic for unrecognized diabetes mellitus. A CT scan revealed a large posterior neck subcutaneous hypodensity with predominant internal fluid density and peripheral thick irregular enhancement (Figure 1). It measured about 8 cm at maximum transverse diameter. This was abutting the trapezius muscle, with no clear muscle invasion (Figure 2). A CT chest scan also revealed a heterogenous left upper lobe soft tissue mass lesion attached to the anterior pleura, associated with a 1-cm left hilar node, and bilateral indeterminant pulmonary nodules measuring 1–2 mm were also seen. An abdominal/pelvic wall CT scan showed a right paramedian posterior lower chest and upper abdominal wall subcutaneous mass measuring 8.0×6.5×8.0 cm, predominantly fluid density, with thick irregular peripheral and enhancing wall abutting the right paraspinal muscle (Figures 3, 4).
Figure 1.
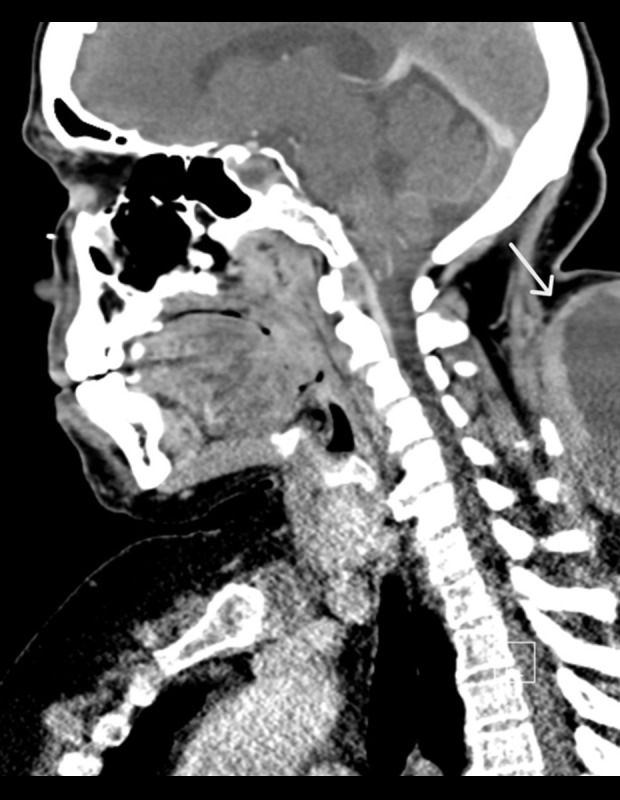
Sagittal CT posterior neck, showing partially visualized subcutaneous paraspinal thick rim enhancing fluid density collection (white arrow).
Figure 2.
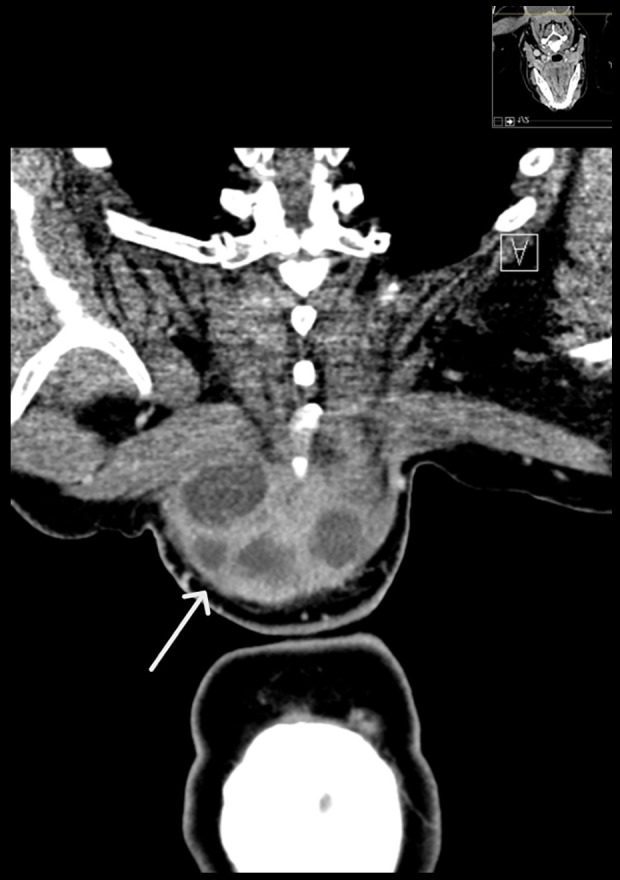
Coronal CT posterior neck, showing soft tissue mass with multiple internal fluid densities/necrosis and peripheral thick irregular enhancement (white arrow), abutting the trapezius muscle with no clear muscle invasion.
Figure 3.
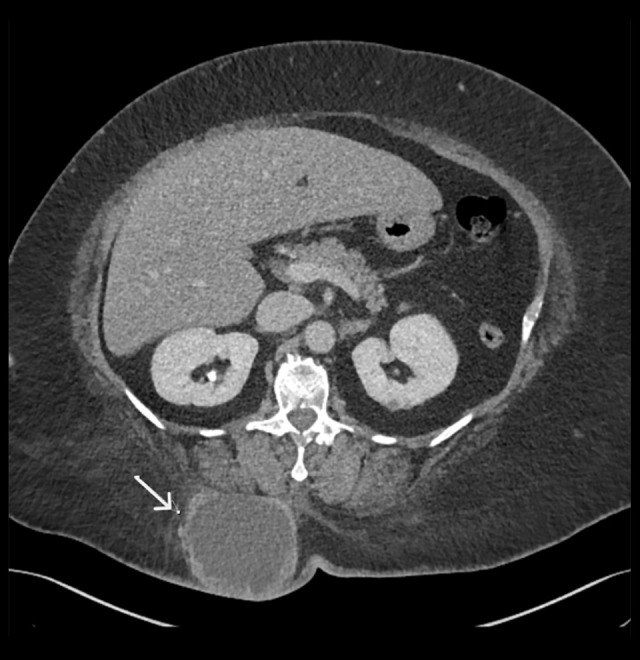
Axial CT abdomen showing a subcutaneous right paramedian posterior lower chest and upper abdominal wall mass (white arrow), with thick enhancing wall, central necrosis, and overlying skin thickening.
Figure 4.
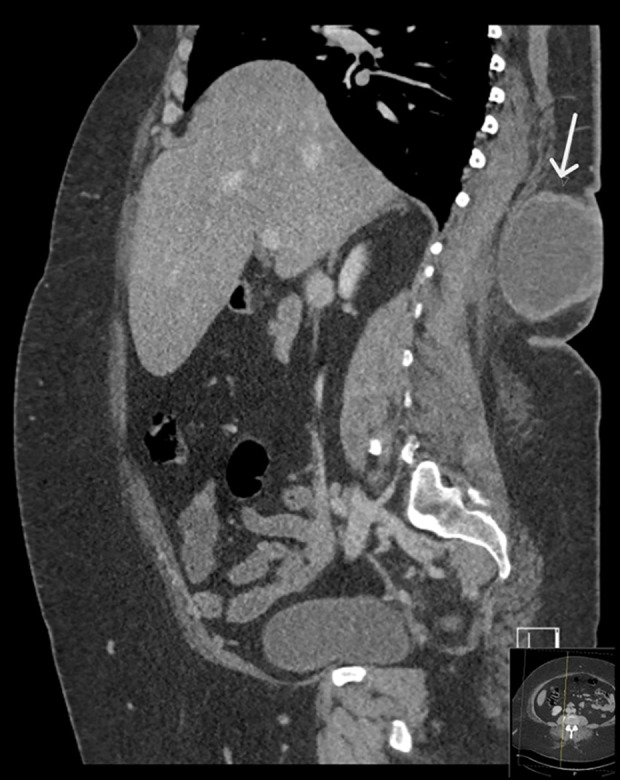
Sagittal CT abdomen showing a subcutaneous right paramedian posterior lower chest and upper abdominal wall mass (white arrow), with thick enhancing wall and central necrosis.
Fine-needle aspiration of the neck mass showed atypical cells, so the patient was taken to the operating room for incision and drainage. A moderate amount of serous fluid came out, and the cavity wall was thick and hard. An incisional biopsy of the neck mass was done, which revealed metastatic mammary (ductal) carcinoma positive for ER in 91–100% and PR in 71–80% of tumor cells, but negative HER-2 (Luminal A). The lung lesion was also biopsied and was positive for adenocarcinoma, consistent with metastatic BC.
Right breast mammography revealed a round lobulated dense breast lesion measuring 2.5×2.6×2.6 cm, with no suspicious calcification. Mildly prominent axillary lymph nodes were noted. The left side was unremarkable apart from post-surgical changes and clips. Breast ultrasound was consistent with these findings as well. Staging work-up showed no CT evidence of intra- abdominopelvic metastasis.
Diagnosis
Stage 4 breast cancer, Luminal A.
Treatment Plan
The patient was started on anti-hormone therapy and capecitabine based on immunohistochemistry findings after discussion of the Breast Tumor Board. Follow-up after 3 months showed regressing tumor and stable lung metastasis.
Discussion
BC is the most common cancer in women worldwide. In 2020, there were about 2.26 million new cases of BC, accounting for 11.7% of all cancer cases in women [11], making for approximately 8 million women alive who were diagnosed with breast cancer in the last 5 years. BC accounts for 0.5–1% of cancers in men [12].
BC is the most common cancer among women in the Kingdom of Saudi Arabia, where, in the last decade, it has accounted for 14.8% of all cancer cases in women and 8.5% of all cancer-related deaths. According to the Ministry of Health of Saudi Arabia, 2022, over 50% of breast cancer cases are detected at a later stage, compared to only 20% in other wealthy countries. As a result, Saudi Arabia has a higher mortality rate and higher annual treatment costs for BC than the United States across all stages of the disease [12]. Data on breast cancer incidence and patterns in the Saudi population are scarce, and this is also true in our city, Jeddah, in the western region of Saudi Arabia. A 2019 retrospective study by Alotaibi et al included 3 tertiary hospitals in Jeddah and showed that two-thirds of BC patients were diagnosed at advanced stage [13]. This is higher than the internationally reported figures and is also higher than for other regions in the country, such as the eastern province. Unfortunately, patients with advanced stage are more prone to have greater morbidity and lower survival rates compared to early-diagnosed cases [14]. The most common types of cancer that metastasize to the skin are breast, colon, lung, ovary, and bladder cancer, with an overall prevalence of 5.3% [12].
Skin metastasis typically appears late in the course of the disease, after the primary cancer has been diagnosed [15]. It is estimated that 0.7–9% of patients with cancer will develop skin metastasis at some point during their treatment [15,16]. The prevalence of cutaneous metastases has increased as cancer survival rates have increased, and therapy options have become more diverse. Because of the wide range in clinical course, identifying both the skin cancer and the original tumor can be challenging [17]. Metastasis can be the earliest indicator of relapse after cancer treatment and can be a predictor of poor survival. It is more prevalent among the elderly. A skin biopsy should be taken in cancer patients with early or rapid onset, prolonged healing, bleeding, or vascular appearance of lesions that do not disappear following therapy [15]. The scalp, navel, chest wall, and abdomen are the most common locations of metastasis, and they occur on the chest and abdomen in 75% of women. BC is the most prevalent primary malignancy in women, and it tends to spread later to the anterior thoracic wall [16,18].
Although the clinical manifestation of metastasis can differ, it is frequently characterized by suddenly appearing, normochromic or brownish solid nodules. Lesions can either be associated with pain or not, and they may initially spread abruptly before stabilizing. They may appear sclerotic or inflammatory, and they may be single or numerous. They sometimes retract the skin [15,19]. Metastatic breast cancer that has spread extensively to the skin can resemble cellulitis or an armored breastplate (en cuirasse pattern) [16]. A previous study in 2000 reported that skin lesions from BC metastasis are often multiple, hard, dark, and painless nodules that suddenly appear and are about 2 cm in size. However, the appearance of the lesions can vary depending on the histological type of the cancer, such as erysipelas (redness and swelling of the skin), papules (small, raised bumps on the skin), widespread sclerosis (thickening and hardening of the skin), or eczema (red, itchy rash) [18]. Papules and/or nodules were the most common presentation, accounting for 80% of cases. Telangiectasic carcinoma ranked second with 11.2%, followed by erysipeloid carcinoma with 3%, “en cuirasse” carcinoma with 3%, alopecia neoplastica with 2%, and a zosteriform pattern with 0.8%. Rarely, cutaneous metastases appear as the initial sign of malignancy [14]. BC can spread to the skin either directly, by hematologic or lymphatic dissemination, or through surgical implantation [19].
A skin biopsy is always required to diagnose skin metastases. In most cases with cutaneous metastases, the primary cancer has become locally advanced beyond surgical management [20,21]. Treatment options include chemotherapy, radiation, and hormone therapy, based on immunohistochemistry characteristics [22,23]. Furthermore, local radiation can be considered in patients with large ulcerating breast tumors, in addition to debridement and dressing. Unfortunately, the prognosis is usually poor [24].
Studies indicate that many [6] individuals with inflammatory breast cancer and [25] even early-stage triple-negative breast cancer have a recurrence rate of 40–50%. Current guidelines recommend annual mammography after treatment, but there should be individualized extended surveillance to monitor for late recurrence [26,27].
Conclusions
In this case, a patient with metastatic BC presented with multiple new skin masses. The patient had previously been treated for left-sided BC 10 years prior, and the new masses were all found to be metastatic BC. She has begun chemotherapy and hormonal treatment, and her disease was responding at 3-monts follow-up.
This report highlights the importance of long follow-up for patients with a history of BC. Even after many years, the cancer can recur and spread to other parts of the body. Patients should be aware of the signs and symptoms of recurrence, and they should see their doctor right away if they experience any new or concerning symptoms.
Footnotes
Ethics Statement
This study involving a human participant was reviewed and approved by the Ethics Committee of King Abdulaziz University Hospital.
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
References:
- 1.Wang R, Zhu Y, Liu X, et al. The Clinicopathological features and survival outcomes of patients with different metastatic sites in stage IV breast cancer. BMC cancer. 2019;19(1):1–2. doi: 10.1186/s12885-019-6311-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Martin AM, Cagney DN, Catalano PJ, et al. Brain metastases in newly diagnosed breast cancer: A population-based study. JAMA Oncology. 2017;3(8):1069–77. doi: 10.1001/jamaoncol.2017.0001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kondi-Pafiti A, Kairi-Vasilatou E, Iavazzo C, et al. Metastatic neoplasms of the ovaries: A clinicopathological study of 97 cases. Arch Gynecol Obstet. 2011;284:1283–88. doi: 10.1007/s00404-011-1847-4. [DOI] [PubMed] [Google Scholar]
- 4.Kutasovic JR, McCart Reed AE, Males R, et al. Breast cancer metastasis to gynaecological organs: A clinicopathological and molecular profiling study. J Pathol Clin Res. 2019;5(1):25–39. doi: 10.1002/cjp2.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hong J, Kim Y, Cho J, et al. Clinical features and prognosis of breast cancer with gastric metastasis. Oncol Lett. 2019;17(2):1833–41. doi: 10.3892/ol.2018.9754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stewart RL, Updike KL, Factor RE, et al. A multigene assay determines risk of recurrence in patients with triple-negative breast cancer. Cancer Res. 2019;79(13):3466–78. doi: 10.1158/0008-5472.CAN-18-3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gan EY, Chio MT, Tan WP. A retrospective review of cutaneous metastases at the National Skin Centre Singapore. Australas J Dermatol. 2015;56(1):1–6. doi: 10.1111/ajd.12194. [DOI] [PubMed] [Google Scholar]
- 8.Conner KB, Cohen PR. Cutaneous metastasis of breast carcinoma presenting as alopecia neoplastica. South Med J. 2009;102(4):385–89. doi: 10.1097/SMJ.0b013e31819b0c26. [DOI] [PubMed] [Google Scholar]
- 9.Silverman D, Ruth K, Sigurdson ER, et al. Skin involvement and breast cancer: Are T4b lesions of all sizes created equal? J Am Coll Surg. 2014;219(3):534–44. doi: 10.1016/j.jamcollsurg.2014.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Strickley JD, Jenson AB, Jung JY. Cutaneous metastasis. Hematol Oncol Clin North Am. 2019;33(1):173–97. doi: 10.1016/j.hoc.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 11.Gradishar WJ, Moran MS, Abraham J, et al. NCCN Guidelines® Insights: Breast cancer, Version 4.2023: Featured updates to the NCCN guidelines. J Natl Compr Canc Netw. 2023;21(6):594–608. doi: 10.6004/jnccn.2023.0031. [DOI] [PubMed] [Google Scholar]
- 12.Nava G, Greer KE, Patterson JW, Lin KY. Metastatic cutaneous breast carcinoma: A case report and review of the literature. Can J Plast Surg. 2009;17(1):25–28. doi: 10.1177/229255030901700105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Waheed N, Hameed M, Alendijani YA, et al. Breast cancer diagnosis and survival among patients diagnosed by a structured community based screening program compared to opportunistic diagnosis: A case control study. Asian Pac J Cancer Prev. 2023;24(3):923–27. doi: 10.31557/APJCP.2023.24.3.923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khatcheressian JL, Hurley P, Bantug E, et al. Breast cancer follow-up and management after primary treatment: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(7):961–65. doi: 10.1200/JCO.2012.45.9859. [DOI] [PubMed] [Google Scholar]
- 15.Wong CY, Helm MA, Kalb RE, et al. The presentation, pathology, and current management strategies of cutaneous metastasis. N Am J Med Sci. 2013;5(9):499–504. doi: 10.4103/1947-2714.118918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Weimann ET, Botero EB, Mendes C, et al. Cutaneous metastasis as the first manifestation of occult malignant breast neoplasia. An Bras Dermatol. 2016;91(5 Suppl. 1):105–7. doi: 10.1590/abd1806-4841.20164572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bordel-Gómez MT, Used-Aznar MM. [Cutaneous metastases from adenocarcinoma of unknown primary origin.] Actas Dermosifiliogr. 2006;97:662–65. doi: 10.1016/s0001-7310(06)73490-1. [in Spanish] [DOI] [PubMed] [Google Scholar]
- 18.Hu SC, Chen GS, Wu CS, et al. Rates of cutaneous metastases from different internal malignancies: Experience from a Taiwanese medical center. J Am Acad Dermatol. 2009;60:379–87. doi: 10.1016/j.jaad.2008.10.007. [DOI] [PubMed] [Google Scholar]
- 19.Mordenti CP, Concetta FK, Cerroni M, Chimenti LS. Cutaneous metastatic breast carcinoma. Acta Dermatoven APA. 2000;9(4):143–48. [Google Scholar]
- 20.Lehman J, Benacci J. Cutaneous metastasis of invasive ductal carcinoma of the breast to an infusaport site. Cutis. 2008;81:223–26. [PubMed] [Google Scholar]
- 21.Henriques L, Palumbo M, Guay MP, et al. Imiquimod in the treatment of breast cancer skin metastasis. J Clin Oncol. 2014;32:e22–25. doi: 10.1200/JCO.2012.46.4883. [DOI] [PubMed] [Google Scholar]
- 22.Varol U, Yildiz I, Alacacioglu A, Uslu R. Anticancer therapy for breast cancer patients with skin metastases refractory to conventional treatments. Asian Pac J Cancer Prev. 2014;15:1885–87. doi: 10.7314/apjcp.2014.15.4.1885. [DOI] [PubMed] [Google Scholar]
- 23.World Health Organization (WHO) Breast cancer [Internet] Mar 13, 2024. Available from: https://www.who.int/news-room/fact-sheets/detail/breast-cancer.
- 24.Shrivastava N, Balasubramanian A. Cutaneous metastasis in breast cancer: A case series. Cureus. 2023;15(6):e40109. doi: 10.7759/cureus.40109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li J, Xia Y, Wu Q, et al. Outcomes of patients with inflammatory breast cancer by hormone receptor- and HER2-defined molecular subtypes: A population-based study from the SEER program. Oncotarget. 2017;8(30):49370–79. doi: 10.18632/oncotarget.17217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Strickley JD, Jenson AB, Jung JY. Cutaneous metastasis. Hematology/Oncology Clinics. 2019;33(1):173–97. doi: 10.1016/j.hoc.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 27.Alotaibi N, Alosaimy R, Al Mushaddak W. Prevalence and determinants of advanced-stage breast cancer in Jeddah tertiary hospitals, Saudi Arabia. International Journal of Medicine in Developing Countries. 2019;3(8):716. [Google Scholar]


