Abstract
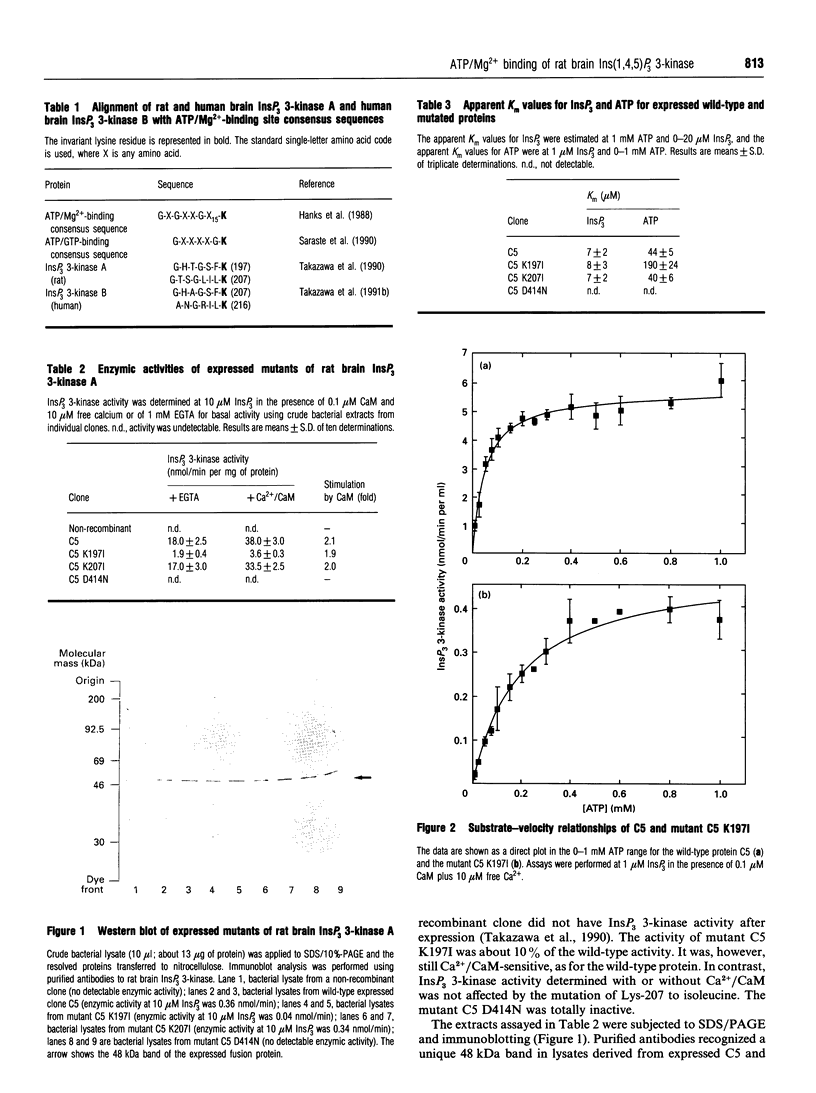
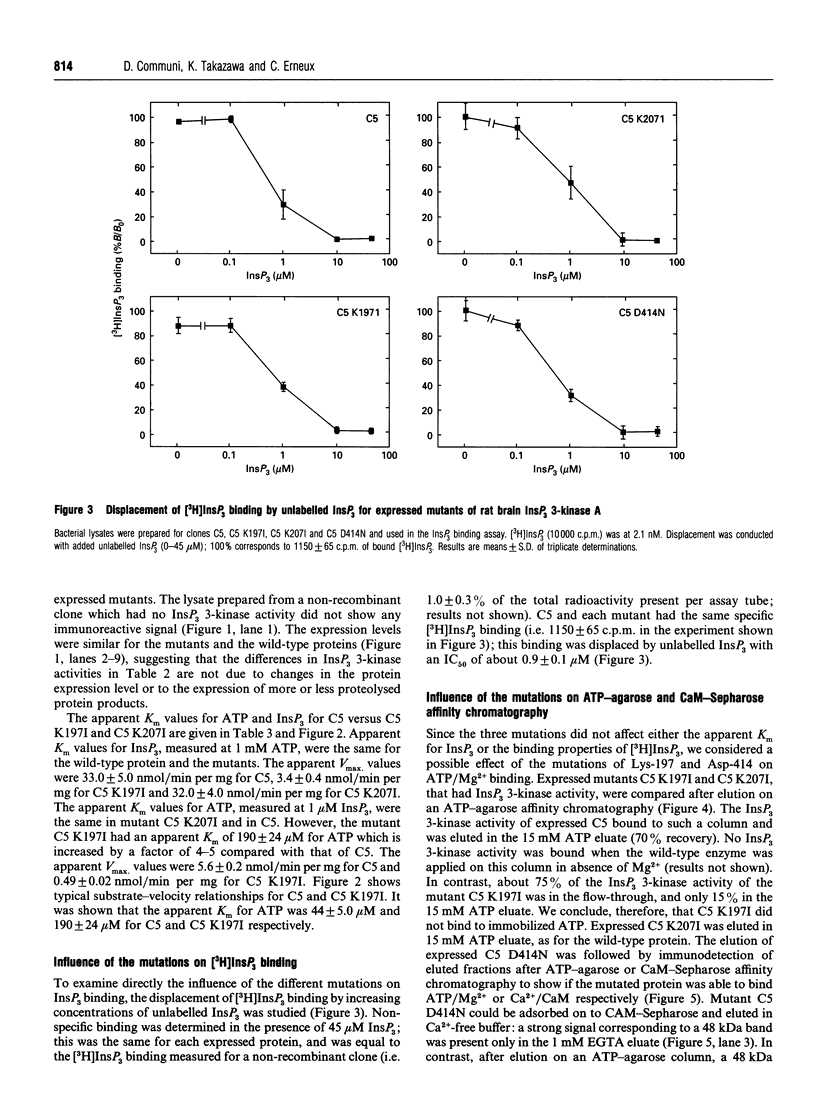
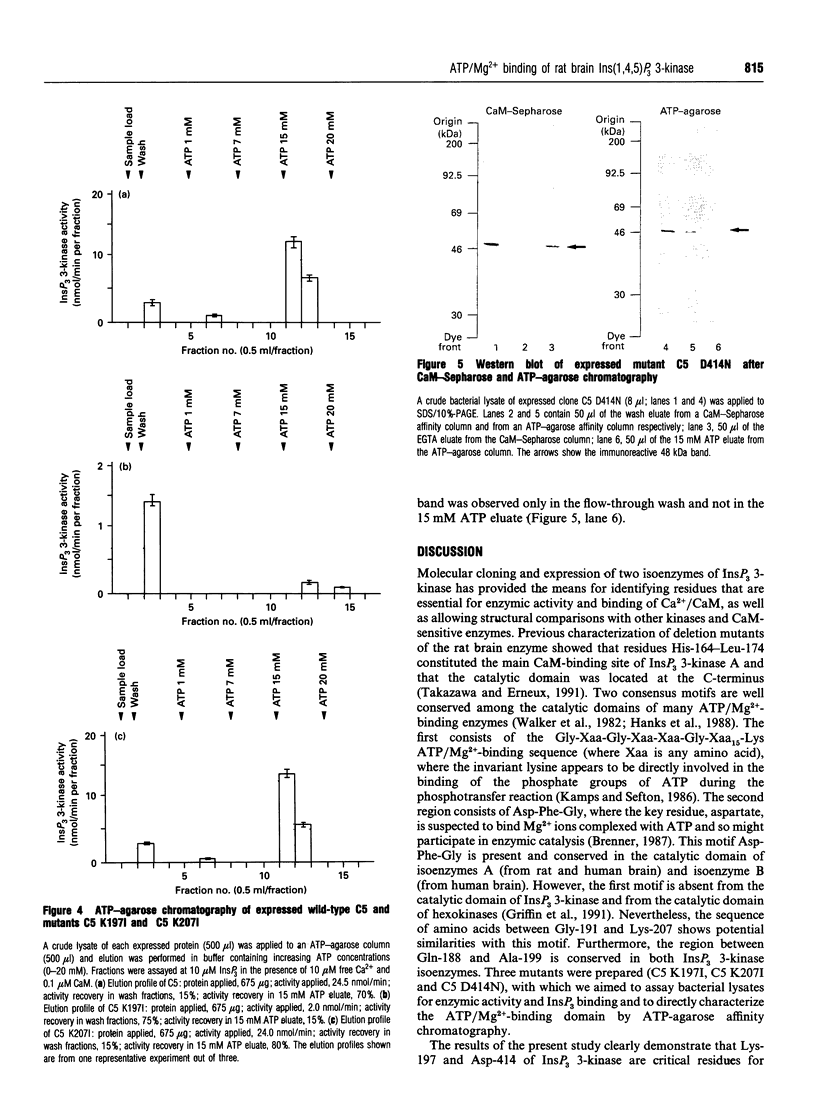
Rat brain inositol 1,4,5-trisphosphate (InsP3) 3-kinase A was expressed in Escherichia coli in order to identify the amino acid residues involved in substrate ATP/Mg2+ binding. Two amino acid regions that are conserved in the catalytic domain of InsP3 3-kinase isoenzymes A and B had characteristics consistent with two ATP/Mg(2+)-binding motives. Site-directed mutagenesis was performed on residues Lys-197, Lys-207 and Asp-414 to generate three mutant enzymes, referred to as C5 K197I, C5 K207I and C5 D414N. Comparison of the wild-type and mutant proteins with regard to enzymic activity revealed that C5 K197I exhibited 10% of control enzyme activity, C5 D414N was totally inactive and C5 K207I was fully active. The reduced levels of enzyme activity for C5 K197I and C5 D414N were correlated with an altered ability of the mutant enzymes to bind ATP/Mg2+, as determined by ATP-agarose affinity chromatography. Neither Ca2+/calmodulin binding nor InsP3 binding appeared to be affected. Mutant C5 K207I showed the same characteristics as the wild-type enzyme. Taken together, these results strongly indicated (i) that amino acid residues Lys-197 and Asp-414 are necessary for InsP3 3-kinase activity and form part of the ATP/Mg(2+)-binding domain, and (ii) that amino acid residues Lys-197, Lys-207 and Asp-414 are not involved in either InsP3 binding or enzyme stimulation by Ca2+/calmodulin.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berridge M. J., Irvine R. F. Inositol phosphates and cell signalling. Nature. 1989 Sep 21;341(6239):197–205. doi: 10.1038/341197a0. [DOI] [PubMed] [Google Scholar]
- Biden T. J., Comte M., Cox J. A., Wollheim C. B. Calcium-calmodulin stimulates inositol 1,4,5-trisphosphate kinase activity from insulin-secreting RINm5F cells. J Biol Chem. 1987 Jul 15;262(20):9437–9440. [PubMed] [Google Scholar]
- Black M. E., Hruby D. E. Identification of the ATP-binding domain of vaccinia virus thymidine kinase. J Biol Chem. 1990 Oct 15;265(29):17584–17592. [PubMed] [Google Scholar]
- Brenner S. Phosphotransferase sequence homology. Nature. 1987 Sep 3;329(6134):21–21. doi: 10.1038/329021a0. [DOI] [PubMed] [Google Scholar]
- Choi K. Y., Kim H. K., Lee S. Y., Moon K. H., Sim S. S., Kim J. W., Chung H. K., Rhee S. G. Molecular cloning and expression of a complementary DNA for inositol 1,4,5-trisphosphate 3-kinase. Science. 1990 Apr 6;248(4951):64–66. doi: 10.1126/science.2157285. [DOI] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Prediction of the secondary structure of proteins from their amino acid sequence. Adv Enzymol Relat Areas Mol Biol. 1978;47:45–148. doi: 10.1002/9780470122921.ch2. [DOI] [PubMed] [Google Scholar]
- Fry D. C., Kuby S. A., Mildvan A. S. ATP-binding site of adenylate kinase: mechanistic implications of its homology with ras-encoded p21, F1-ATPase, and other nucleotide-binding proteins. Proc Natl Acad Sci U S A. 1986 Feb;83(4):907–911. doi: 10.1073/pnas.83.4.907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin L. D., Gelb B. D., Wheeler D. A., Davison D., Adams V., McCabe E. R. Mammalian hexokinase 1: evolutionary conservation and structure to function analysis. Genomics. 1991 Dec;11(4):1014–1024. doi: 10.1016/0888-7543(91)90027-c. [DOI] [PubMed] [Google Scholar]
- Hanks S. K., Quinn A. M., Hunter T. The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science. 1988 Jul 1;241(4861):42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- Irvine R. F. Is inositol tetrakisphosphate the second messenger that controls Ca2+ entry into cells? Adv Second Messenger Phosphoprotein Res. 1992;26:161–185. [PubMed] [Google Scholar]
- Irvine R. F., Letcher A. J., Heslop J. P., Berridge M. J. The inositol tris/tetrakisphosphate pathway--demonstration of Ins(1,4,5)P3 3-kinase activity in animal tissues. Nature. 1986 Apr 17;320(6063):631–634. doi: 10.1038/320631a0. [DOI] [PubMed] [Google Scholar]
- Kamps M. P., Sefton B. M. Neither arginine nor histidine can carry out the function of lysine-295 in the ATP-binding site of p60src. Mol Cell Biol. 1986 Mar;6(3):751–757. doi: 10.1128/mcb.6.3.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S. Y., Sim S. S., Kim J. W., Moon K. H., Kim J. H., Rhee S. G. Purification and properties of D-myo-inositol 1,4,5-trisphosphate 3-kinase from rat brain. Susceptibility to calpain. J Biol Chem. 1990 Jun 5;265(16):9434–9440. [PubMed] [Google Scholar]
- Menetski J. P., Bear D. G., Kowalczykowski S. C. Stable DNA heteroduplex formation catalyzed by the Escherichia coli RecA protein in the absence of ATP hydrolysis. Proc Natl Acad Sci U S A. 1990 Jan;87(1):21–25. doi: 10.1073/pnas.87.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris A. J., Downes C. P., Harden T. K., Michell R. H. Turkey erythrocytes possess a membrane-associated inositol 1,4,5-trisphosphate 3-kinase that is activated by Ca2+ in the presence of calmodulin. Biochem J. 1987 Dec 1;248(2):489–493. doi: 10.1042/bj2480489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson G. L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem. 1977 Dec;83(2):346–356. doi: 10.1016/0003-2697(77)90043-4. [DOI] [PubMed] [Google Scholar]
- Rose T., Glaser P., Surewicz W. K., Mantsch H. H., Reinstein J., Le Blay K., Gilles A. M., Bârzu O. Structural and functional consequences of amino acid substitutions in the second conserved loop of Escherichia coli adenylate kinase. J Biol Chem. 1991 Dec 15;266(35):23654–23659. [PubMed] [Google Scholar]
- Ryu S. H., Lee S. Y., Lee K. Y., Rhee S. G. Catalytic properties of inositol trisphosphate kinase: activation by Ca2+ and calmodulin. FASEB J. 1987 Nov;1(5):388–393. doi: 10.1096/fasebj.1.5.2824270. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraste M., Sibbald P. R., Wittinghofer A. The P-loop--a common motif in ATP- and GTP-binding proteins. Trends Biochem Sci. 1990 Nov;15(11):430–434. doi: 10.1016/0968-0004(90)90281-f. [DOI] [PubMed] [Google Scholar]
- Sayers J. R., Schmidt W., Eckstein F. 5'-3' exonucleases in phosphorothioate-based oligonucleotide-directed mutagenesis. Nucleic Acids Res. 1988 Feb 11;16(3):791–802. doi: 10.1093/nar/16.3.791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sim S. S., Kim J. W., Rhee S. G. Regulation of D-myo-inositol 1,4,5-trisphosphate 3-kinase by cAMP-dependent protein kinase and protein kinase C. J Biol Chem. 1990 Jun 25;265(18):10367–10372. [PubMed] [Google Scholar]
- Snyder M., Elledge S., Sweetser D., Young R. A., Davis R. W. Lambda gt 11: gene isolation with antibody probes and other applications. Methods Enzymol. 1987;154:107–128. doi: 10.1016/0076-6879(87)54073-3. [DOI] [PubMed] [Google Scholar]
- Takazawa K., Erneux C. Identification of residues essential for catalysis and binding of calmodulin in rat brain inositol 1,4,5-trisphosphate 3-kinase. Biochem J. 1991 Nov 15;280(Pt 1):125–129. doi: 10.1042/bj2800125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takazawa K., Passareiro H., Dumont J. E., Erneux C. Ca2+/calmodulin-sensitive inositol 1,4,5-trisphosphate 3-kinase in rat and bovine brain tissues. Biochem Biophys Res Commun. 1988 Jun 16;153(2):632–641. doi: 10.1016/s0006-291x(88)81142-2. [DOI] [PubMed] [Google Scholar]
- Takazawa K., Passareiro H., Dumont J. E., Erneux C. Purification of bovine brain inositol 1,4,5-trisphosphate 3-kinase. Identification of the enzyme by sodium dodecyl sulphate/polyacrylamide-gel electrophoresis. Biochem J. 1989 Jul 15;261(2):483–488. doi: 10.1042/bj2610483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takazawa K., Perret J., Dumont J. E., Erneux C. Molecular cloning and expression of a human brain inositol 1,4,5-trisphosphate 3-kinase. Biochem Biophys Res Commun. 1991 Jan 31;174(2):529–535. doi: 10.1016/0006-291x(91)91449-m. [DOI] [PubMed] [Google Scholar]
- Takazawa K., Perret J., Dumont J. E., Erneux C. Molecular cloning and expression of a new putative inositol 1,4,5-trisphosphate 3-kinase isoenzyme. Biochem J. 1991 Sep 15;278(Pt 3):883–886. doi: 10.1042/bj2780883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takazawa K., Vandekerckhove J., Dumont J. E., Erneux C. Cloning and expression in Escherichia coli of a rat brain cDNA encoding a Ca2+/calmodulin-sensitive inositol 1,4,5-trisphosphate 3-kinase. Biochem J. 1990 Nov 15;272(1):107–112. doi: 10.1042/bj2720107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker J. E., Saraste M., Runswick M. J., Gay N. J. Distantly related sequences in the alpha- and beta-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1982;1(8):945–951. doi: 10.1002/j.1460-2075.1982.tb01276.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi K., Hirata M., Kuriyama H. Purification and characterization of inositol 1,4,5-trisphosphate 3-kinase from pig aortic smooth muscle. Biochem J. 1988 Apr 1;251(1):129–134. doi: 10.1042/bj2510129. [DOI] [PMC free article] [PubMed] [Google Scholar]




