Abstract
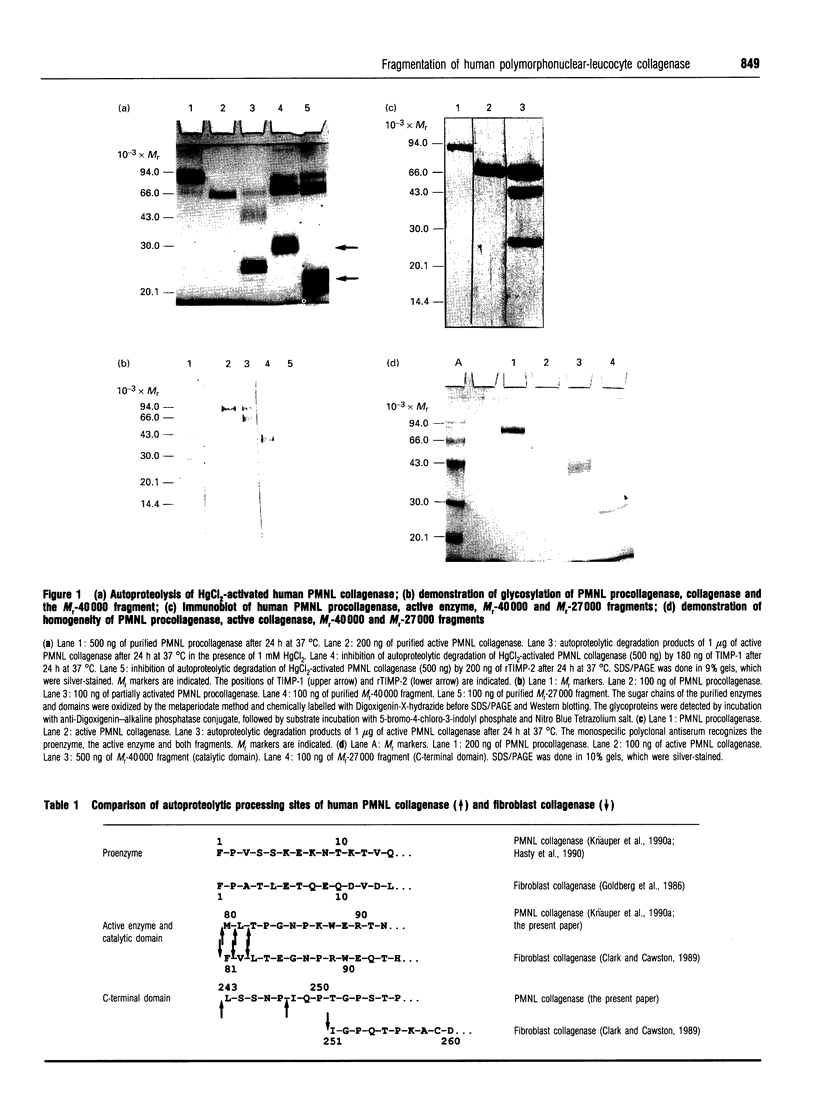
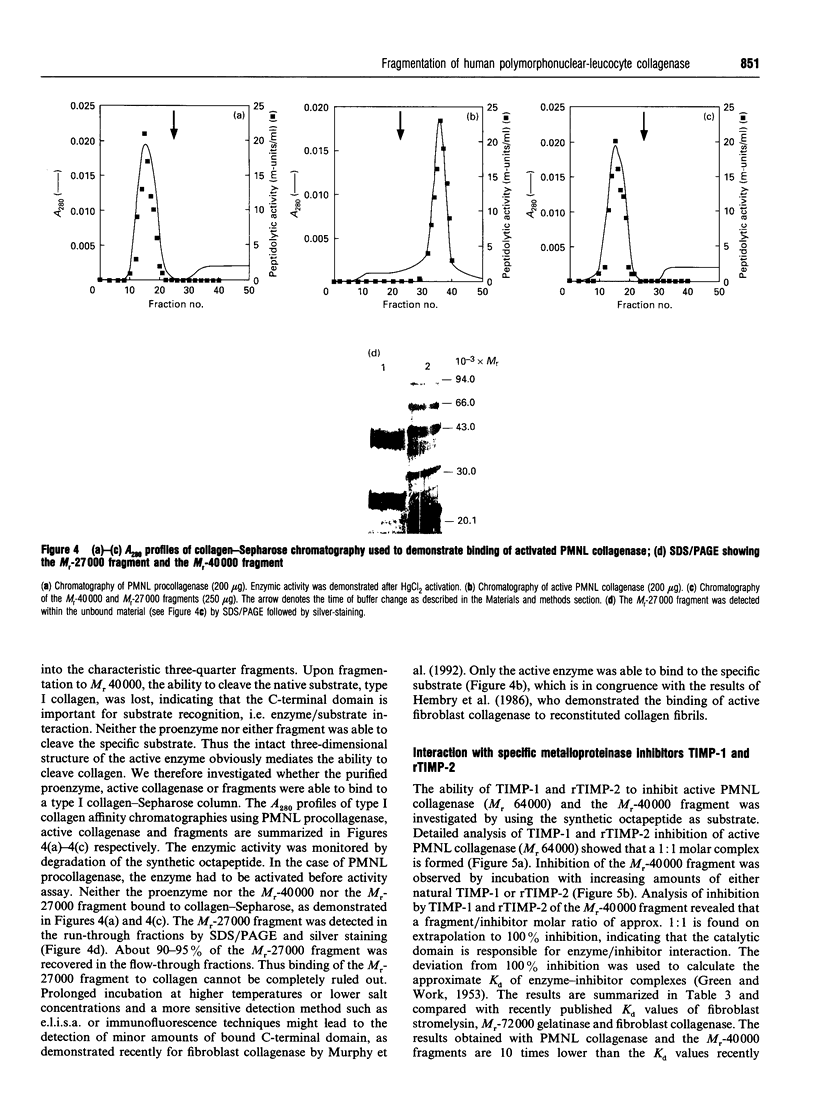
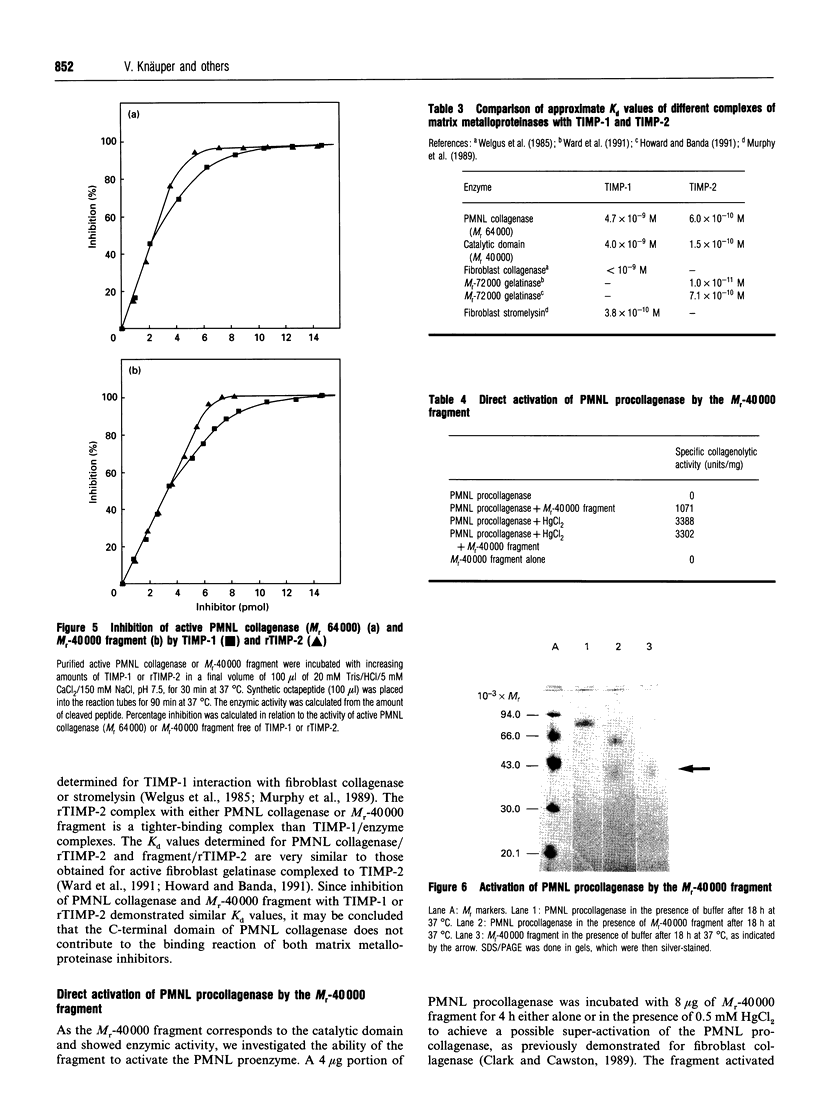
Human polymorphonuclear-leucocyte collagenase (M(r) 64,000) shows autoproteolytic degradation to two major fragments of M(r) 40,000 and M(r) 27,000. N-terminal sequence data and investigation of the substrate specificity of the fragments demonstrate that the M(r)-40,000 fragment corresponds to the catalytic domain, whereas the M(r0-27,000 fragment shows no enzymic activity. The activity profile of the M(r)-40,000 fragment is comparable with the specificity of the intact active collagenase (M(r) 64,000), but the ability to cleave collagen was lost. The enzymic activity of this fragment can be inhibited by either tissue inhibitor of metalloproteinase (TIMP)-1 or recombinant TIMP-2 in a 1:1 molar ratio. The C-terminal part of the enzyme (M(r) 27,000), important for the binding reaction with collagen substrates, is involved in collagenolysis.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allan J. A., Hembry R. M., Angal S., Reynolds J. J., Murphy G. Binding of latent and high Mr active forms of stromelysin to collagen is mediated by the C-terminal domain. J Cell Sci. 1991 Aug;99(Pt 4):789–795. doi: 10.1242/jcs.99.4.789. [DOI] [PubMed] [Google Scholar]
- Bergmann U., Michaelis J., Oberhoff R., Knäuper V., Beckmann R., Tschesche H. Enzyme linked immunosorbent assays (ELISA) for the quantitative determination of human leukocyte collagenase and gelatinase. J Clin Chem Clin Biochem. 1989 Jun;27(6):351–359. doi: 10.1515/cclm.1989.27.6.351. [DOI] [PubMed] [Google Scholar]
- Bläser J., Knäuper V., Osthues A., Reinke H., Tschesche H. Mercurial activation of human polymorphonuclear leucocyte procollagenase. Eur J Biochem. 1991 Dec 18;202(3):1223–1230. doi: 10.1111/j.1432-1033.1991.tb16494.x. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Chin J. R., Murphy G., Werb Z. Stromelysin, a connective tissue-degrading metalloendopeptidase secreted by stimulated rabbit synovial fibroblasts in parallel with collagenase. Biosynthesis, isolation, characterization, and substrates. J Biol Chem. 1985 Oct 5;260(22):12367–12376. [PubMed] [Google Scholar]
- Clark I. M., Cawston T. E. Fragments of human fibroblast collagenase. Purification and characterization. Biochem J. 1989 Oct 1;263(1):201–206. doi: 10.1042/bj2630201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeClerck Y. A., Yean T. D., Lu H. S., Ting J., Langley K. E. Inhibition of autoproteolytic activation of interstitial procollagenase by recombinant metalloproteinase inhibitor MI/TIMP-2. J Biol Chem. 1991 Feb 25;266(6):3893–3899. [PubMed] [Google Scholar]
- Desrochers P. E., Mookhtiar K., Van Wart H. E., Hasty K. A., Weiss S. J. Proteolytic inactivation of alpha 1-proteinase inhibitor and alpha 1-antichymotrypsin by oxidatively activated human neutrophil metalloproteinases. J Biol Chem. 1992 Mar 5;267(7):5005–5012. [PubMed] [Google Scholar]
- Devarajan P., Mookhtiar K., Van Wart H., Berliner N. Structure and expression of the cDNA encoding human neutrophil collagenase. Blood. 1991 Jun 15;77(12):2731–2738. [PubMed] [Google Scholar]
- Evans C. H., Ridella J. D. An evaluation of fluorometric proteinase assays which employ fluorescamine. Anal Biochem. 1984 Nov 1;142(2):411–420. doi: 10.1016/0003-2697(84)90485-8. [DOI] [PubMed] [Google Scholar]
- Fields G. B., Van Wart H. E. Unique features of the tissue collagenase cleavage site in interstitial collagens. Matrix Suppl. 1992;1:68–70. [PubMed] [Google Scholar]
- Fontana A., Fassina G., Vita C., Dalzoppo D., Zamai M., Zambonin M. Correlation between sites of limited proteolysis and segmental mobility in thermolysin. Biochemistry. 1986 Apr 22;25(8):1847–1851. doi: 10.1021/bi00356a001. [DOI] [PubMed] [Google Scholar]
- GREEN N. M., WORK E. Pancreatic trypsin inhibitor. II. Reaction with trypsin. Biochem J. 1953 May;54(2):347–352. doi: 10.1042/bj0540347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg G. I., Strongin A., Collier I. E., Genrich L. T., Marmer B. L. Interaction of 92-kDa type IV collagenase with the tissue inhibitor of metalloproteinases prevents dimerization, complex formation with interstitial collagenase, and activation of the proenzyme with stromelysin. J Biol Chem. 1992 Mar 5;267(7):4583–4591. [PubMed] [Google Scholar]
- Goldberg G. I., Wilhelm S. M., Kronberger A., Bauer E. A., Grant G. A., Eisen A. Z. Human fibroblast collagenase. Complete primary structure and homology to an oncogene transformation-induced rat protein. J Biol Chem. 1986 May 15;261(14):6600–6605. [PubMed] [Google Scholar]
- Grant G. A., Eisen A. Z., Marmer B. L., Roswit W. T., Goldberg G. I. The activation of human skin fibroblast procollagenase. Sequence identification of the major conversion products. J Biol Chem. 1987 Apr 25;262(12):5886–5889. [PubMed] [Google Scholar]
- Hasty K. A., Pourmotabbed T. F., Goldberg G. I., Thompson J. P., Spinella D. G., Stevens R. M., Mainardi C. L. Human neutrophil collagenase. A distinct gene product with homology to other matrix metalloproteinases. J Biol Chem. 1990 Jul 15;265(20):11421–11424. [PubMed] [Google Scholar]
- Hembry R. M., Murphy G., Cawston T. E., Dingle J. T., Reynolds J. J. Characterization of a specific antiserum for mammalian collagenase from several species: immunolocalization of collagenase in rabbit chondrocytes and uterus. J Cell Sci. 1986 Mar;81:105–123. doi: 10.1242/jcs.81.1.105. [DOI] [PubMed] [Google Scholar]
- Howard E. W., Banda M. J. Binding of tissue inhibitor of metalloproteinases 2 to two distinct sites on human 72-kDa gelatinase. Identification of a stabilization site. J Biol Chem. 1991 Sep 25;266(27):17972–17977. [PubMed] [Google Scholar]
- Howard E. W., Bullen E. C., Banda M. J. Regulation of the autoactivation of human 72-kDa progelatinase by tissue inhibitor of metalloproteinases-2. J Biol Chem. 1991 Jul 15;266(20):13064–13069. [PubMed] [Google Scholar]
- Knäuper V., Krämer S., Reinke H., Tschesche H. Characterization and activation of procollagenase from human polymorphonuclear leucocytes. N-terminal sequence determination of the proenzyme and various proteolytically activated forms. Eur J Biochem. 1990 Apr 30;189(2):295–300. doi: 10.1111/j.1432-1033.1990.tb15489.x. [DOI] [PubMed] [Google Scholar]
- Knäuper V., Reinke H., Tschesche H. Inactivation of human plasma alpha 1-proteinase inhibitor by human PMN leucocyte collagenase. FEBS Lett. 1990 Apr 24;263(2):355–357. doi: 10.1016/0014-5793(90)81412-h. [DOI] [PubMed] [Google Scholar]
- Knäuper V., Triebel S., Reinke H., Tschesche H. Inactivation of human plasma C1-inhibitor by human PMN leucocyte matrix metalloproteinases. FEBS Lett. 1991 Sep 23;290(1-2):99–102. doi: 10.1016/0014-5793(91)81235-z. [DOI] [PubMed] [Google Scholar]
- Koklitis P. A., Murphy G., Sutton C., Angal S. Purification of recombinant human prostromelysin. Studies on heat activation to give high-Mr and low-Mr active forms, and a comparison of recombinant with natural stromelysin activities. Biochem J. 1991 May 15;276(Pt 1):217–221. doi: 10.1042/bj2760217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mallya S. K., Mookhtiar K. A., Gao Y., Brew K., Dioszegi M., Birkedal-Hansen H., Van Wart H. E. Characterization of 58-kilodalton human neutrophil collagenase: comparison with human fibroblast collagenase. Biochemistry. 1990 Nov 27;29(47):10628–10634. doi: 10.1021/bi00499a008. [DOI] [PubMed] [Google Scholar]
- Marcy A. I., Eiberger L. L., Harrison R., Chan H. K., Hutchinson N. I., Hagmann W. K., Cameron P. M., Boulton D. A., Hermes J. D. Human fibroblast stromelysin catalytic domain: expression, purification, and characterization of a C-terminally truncated form. Biochemistry. 1991 Jul 2;30(26):6476–6483. doi: 10.1021/bi00240a018. [DOI] [PubMed] [Google Scholar]
- Michaelis J., Vissers M. C., Winterbourn C. C. Human neutrophil collagenase cleaves alpha 1-antitrypsin. Biochem J. 1990 Sep 15;270(3):809–814. doi: 10.1042/bj2700809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mookhtiar K. A., Van Wart H. E. Purification to homogeneity of latent and active 58-kilodalton forms of human neutrophil collagenase. Biochemistry. 1990 Nov 27;29(47):10620–10627. doi: 10.1021/bi00499a007. [DOI] [PubMed] [Google Scholar]
- Moore W. M., Spilburg C. A. Purification of human collagenases with a hydroxamic acid affinity column. Biochemistry. 1986 Sep 9;25(18):5189–5195. doi: 10.1021/bi00366a031. [DOI] [PubMed] [Google Scholar]
- Murphy G., Allan J. A., Willenbrock F., Cockett M. I., O'Connell J. P., Docherty A. J. The role of the C-terminal domain in collagenase and stromelysin specificity. J Biol Chem. 1992 May 15;267(14):9612–9618. [PubMed] [Google Scholar]
- Murphy G., Koklitis P., Carne A. F. Dissociation of tissue inhibitor of metalloproteinases (TIMP) from enzyme complexes yields fully active inhibitor. Biochem J. 1989 Aug 1;261(3):1031–1034. doi: 10.1042/bj2611031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy G., Reynolds J. J., Bretz U., Baggiolini M. Collagenase is a component of the specific granules of human neutrophil leucocytes. Biochem J. 1977 Jan 15;162(1):195–197. doi: 10.1042/bj1620195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogata Y., Enghild J. J., Nagase H. Matrix metalloproteinase 3 (stromelysin) activates the precursor for the human matrix metalloproteinase 9. J Biol Chem. 1992 Feb 25;267(6):3581–3584. [PubMed] [Google Scholar]
- Okada Y., Nagase H., Harris E. D., Jr A metalloproteinase from human rheumatoid synovial fibroblasts that digests connective tissue matrix components. Purification and characterization. J Biol Chem. 1986 Oct 25;261(30):14245–14255. [PubMed] [Google Scholar]
- Osthues A., Knäuper V., Oberhoff R., Reinke H., Tschesche H. Isolation and characterization of tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2) from human rheumatoid synovial fluid. FEBS Lett. 1992 Jan 13;296(1):16–20. doi: 10.1016/0014-5793(92)80393-u. [DOI] [PubMed] [Google Scholar]
- Park A. J., Matrisian L. M., Kells A. F., Pearson R., Yuan Z. Y., Navre M. Mutational analysis of the transin (rat stromelysin) autoinhibitor region demonstrates a role for residues surrounding the "cysteine switch". J Biol Chem. 1991 Jan 25;266(3):1584–1590. [PubMed] [Google Scholar]
- Quantin B., Murphy G., Breathnach R. Pump-1 cDNA codes for a protein with characteristics similar to those of classical collagenase family members. Biochemistry. 1989 Jun 27;28(13):5327–5334. doi: 10.1021/bi00439a004. [DOI] [PubMed] [Google Scholar]
- Sanchez-Lopez R., Nicholson R., Gesnel M. C., Matrisian L. M., Breathnach R. Structure-function relationships in the collagenase family member transin. J Biol Chem. 1988 Aug 25;263(24):11892–11899. [PubMed] [Google Scholar]
- Schettler A., Thorn H., Jockusch B. M., Tschesche H. Release of proteinases from stimulated polymorphonuclear leukocytes. Evidence for subclasses of the main granule types and their association with cytoskeletal components. Eur J Biochem. 1991 Apr 10;197(1):197–202. doi: 10.1111/j.1432-1033.1991.tb15899.x. [DOI] [PubMed] [Google Scholar]
- Springman E. B., Angleton E. L., Birkedal-Hansen H., Van Wart H. E. Multiple modes of activation of latent human fibroblast collagenase: evidence for the role of a Cys73 active-site zinc complex in latency and a "cysteine switch" mechanism for activation. Proc Natl Acad Sci U S A. 1990 Jan;87(1):364–368. doi: 10.1073/pnas.87.1.364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stetler-Stevenson W. G., Krutzsch H. C., Wacher M. P., Margulies I. M., Liotta L. A. The activation of human type IV collagenase proenzyme. Sequence identification of the major conversion product following organomercurial activation. J Biol Chem. 1989 Jan 25;264(3):1353–1356. [PubMed] [Google Scholar]
- Suzuki K., Enghild J. J., Morodomi T., Salvesen G., Nagase H. Mechanisms of activation of tissue procollagenase by matrix metalloproteinase 3 (stromelysin). Biochemistry. 1990 Nov 6;29(44):10261–10270. doi: 10.1021/bi00496a016. [DOI] [PubMed] [Google Scholar]
- Van Wart H. E., Birkedal-Hansen H. The cysteine switch: a principle of regulation of metalloproteinase activity with potential applicability to the entire matrix metalloproteinase gene family. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5578–5582. doi: 10.1073/pnas.87.14.5578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward R. V., Hembry R. M., Reynolds J. J., Murphy G. The purification of tissue inhibitor of metalloproteinases-2 from its 72 kDa progelatinase complex. Demonstration of the biochemical similarities of tissue inhibitor of metalloproteinases-2 and tissue inhibitor of metalloproteinases-1. Biochem J. 1991 Aug 15;278(Pt 1):179–187. doi: 10.1042/bj2780179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welgus H. G., Jeffrey J. J., Eisen A. Z. Human skin fibroblast collagenase. Assessment of activation energy and deuterium isotope effect with collagenous substrates. J Biol Chem. 1981 Sep 25;256(18):9516–9521. [PubMed] [Google Scholar]
- Welgus H. G., Jeffrey J. J., Eisen A. Z., Roswit W. T., Stricklin G. P. Human skin fibroblast collagenase: interaction with substrate and inhibitor. Coll Relat Res. 1985 Mar;5(2):167–179. doi: 10.1016/s0174-173x(85)80038-8. [DOI] [PubMed] [Google Scholar]
- Windsor L. J., Birkedal-Hansen H., Birkedal-Hansen B., Engler J. A. An internal cysteine plays a role in the maintenance of the latency of human fibroblast collagenase. Biochemistry. 1991 Jan 22;30(3):641–647. doi: 10.1021/bi00217a008. [DOI] [PubMed] [Google Scholar]
- Woessner J. F., Jr, Taplin C. J. Purification and properties of a small latent matrix metalloproteinase of the rat uterus. J Biol Chem. 1988 Nov 15;263(32):16918–16925. [PubMed] [Google Scholar]
- de Souza S. J., Brentani R. Collagen binding site in collagenase can be determined using the concept of sense-antisense peptide interactions. J Biol Chem. 1992 Jul 5;267(19):13763–13767. [PubMed] [Google Scholar]