Abstract
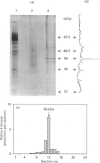
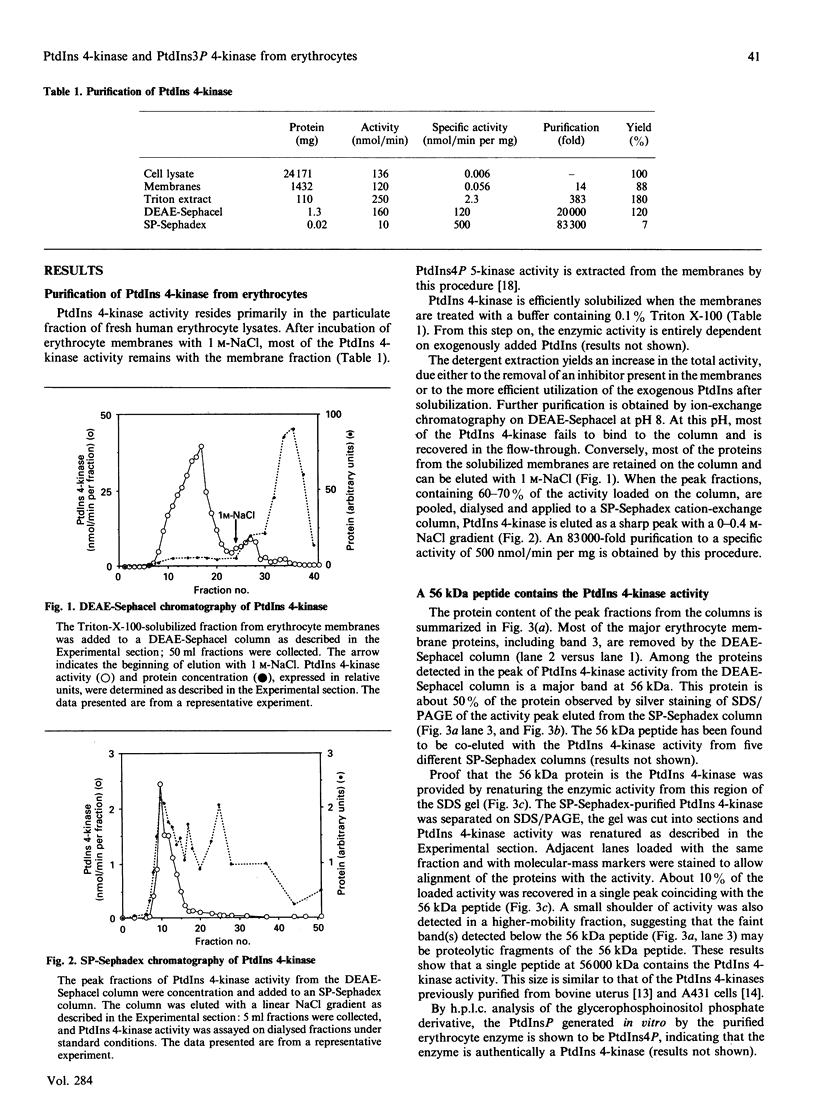
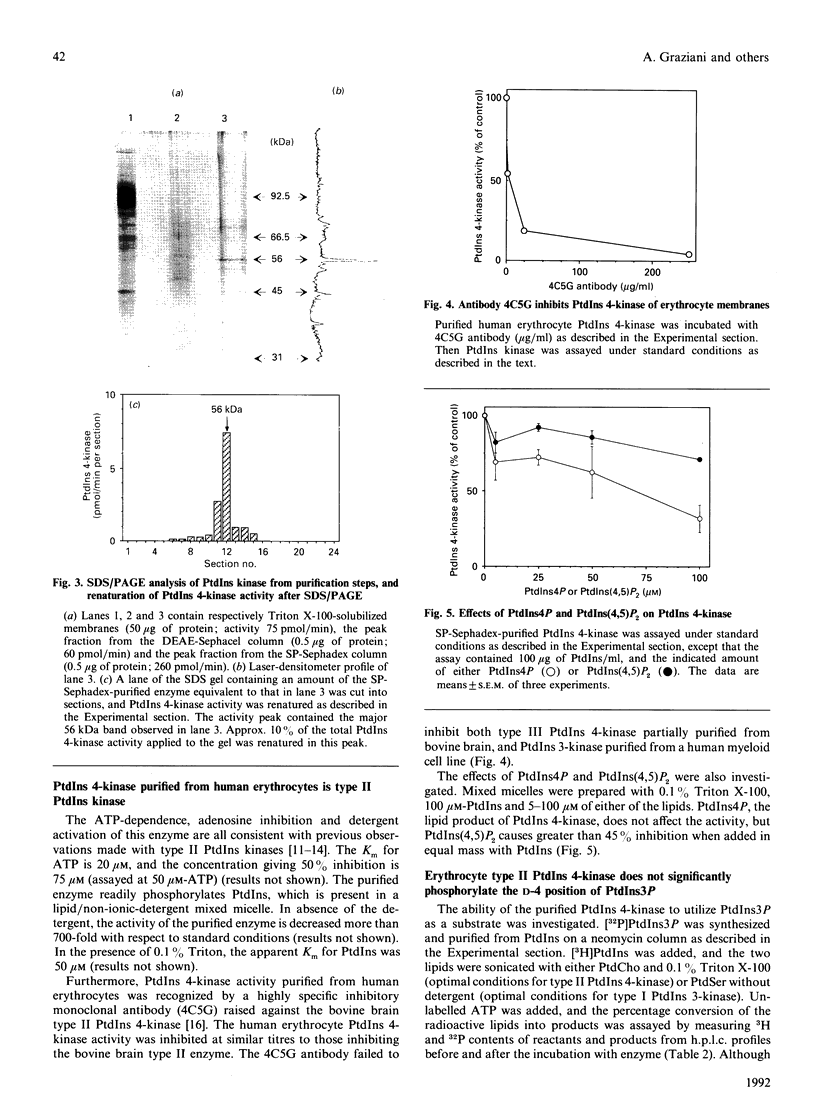
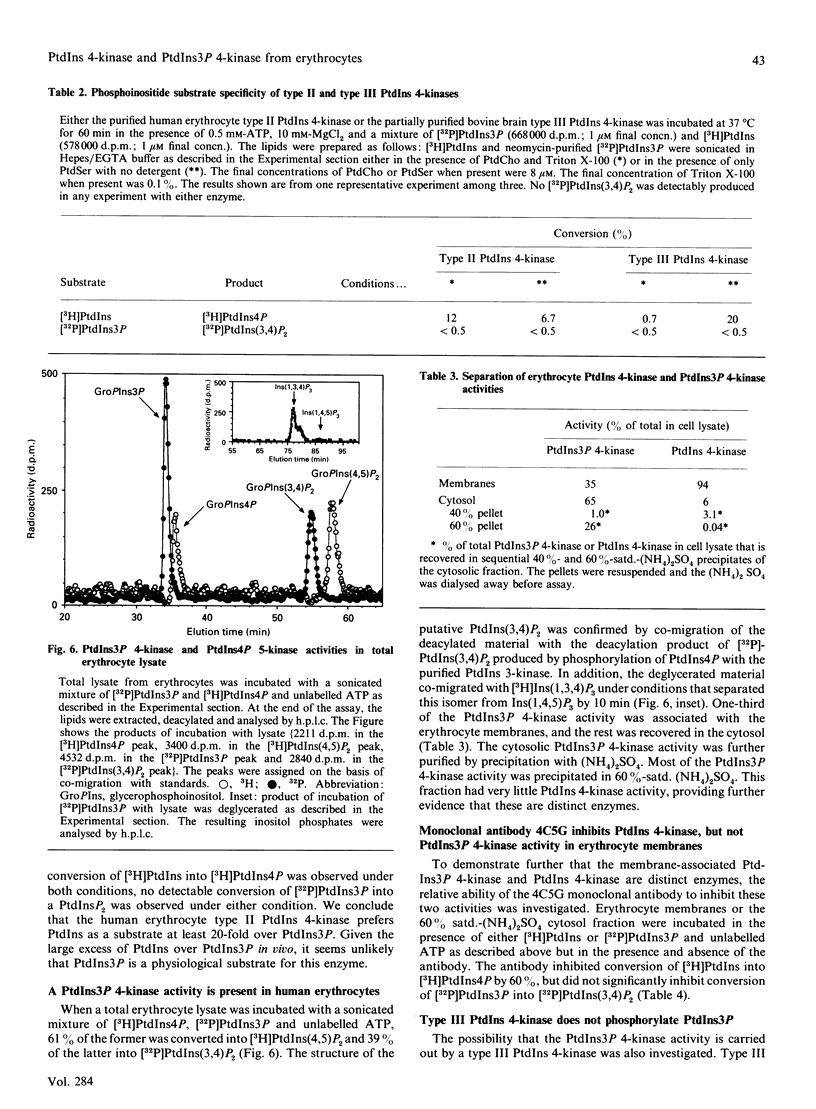
PtdIns 4-kinase has been purified 83,000-fold from human erythrocyte membranes. The major protein detected by SDS/PAGE is of molecular mass 56 kDa, and enzymic activity can be renatured from this band of the gel. The characteristics of this enzyme are similar to other type II PtdIns kinases previously described: PtdIns presented in Triton X-100 micelles is preferred as a substrate over PtdIns vesicles, the enzyme possesses a relatively low Km for ATP (20 microM), and adenosine is an effective inhibitor. A monoclonal antibody raised against bovine brain type II PtdIns 4-kinase is an effective inhibitor of the purified enzyme. PtdIns(4,5)P2 inhibits by approx. 50% when added in equimolar amounts with PtdIns; PtdIns4P has little effect on activity. A PtdIns3P 4-kinase activity has also been detected in erythrocyte lysates. Approximately two-thirds of this activity is in the cytosolic fraction and one-third in the membrane fraction. No PtdIns3P 4-kinase activity could be detected in the purified type II PtdIns 4-kinase preparation, nor could this activity be detected in a bovine brain type III PtdIns 4-kinase preparation. The monoclonal antibody that inhibits the type II PtdIns 4-kinase does not affect the PtdIns3P 4-kinase activity in the membrane fraction. The cytosolic PtdIns3P 4-kinase can be efficiently recovered from a 60%-satd.-(NH4)2SO4 precipitate that is virtually free of PtdIns 4-kinase activity. We conclude that PtdIns3P 4-kinase is a new enzyme distinct from previously characterized PtdIns 4-kinases, and that this enzyme prefers PtdIns3P over PtdIns as a substrate.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Auger K. R., Serunian L. A., Soltoff S. P., Libby P., Cantley L. C. PDGF-dependent tyrosine phosphorylation stimulates production of novel polyphosphoinositides in intact cells. Cell. 1989 Apr 7;57(1):167–175. doi: 10.1016/0092-8674(89)90182-7. [DOI] [PubMed] [Google Scholar]
- Bazenet C. E., Brockman J. L., Lewis D., Chan C., Anderson R. A. Erythroid membrane-bound protein kinase binds to a membrane component and is regulated by phosphatidylinositol 4,5-bisphosphate. J Biol Chem. 1990 May 5;265(13):7369–7376. [PubMed] [Google Scholar]
- Berridge M. J., Heslop J. P., Irvine R. F., Brown K. D. Inositol trisphosphate formation and calcium mobilization in Swiss 3T3 cells in response to platelet-derived growth factor. Biochem J. 1984 Aug 15;222(1):195–201. doi: 10.1042/bj2220195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantley L. C., Auger K. R., Carpenter C., Duckworth B., Graziani A., Kapeller R., Soltoff S. Oncogenes and signal transduction. Cell. 1991 Jan 25;64(2):281–302. doi: 10.1016/0092-8674(91)90639-g. [DOI] [PubMed] [Google Scholar]
- Carpenter C. L., Cantley L. C. Phosphoinositide kinases. Biochemistry. 1990 Dec 25;29(51):11147–11156. doi: 10.1021/bi00503a001. [DOI] [PubMed] [Google Scholar]
- Carpenter C. L., Duckworth B. C., Auger K. R., Cohen B., Schaffhausen B. S., Cantley L. C. Purification and characterization of phosphoinositide 3-kinase from rat liver. J Biol Chem. 1990 Nov 15;265(32):19704–19711. [PubMed] [Google Scholar]
- Endemann G. C., Graziani A., Cantley L. C. A monoclonal antibody distinguishes two types of phosphatidylinositol 4-kinase. Biochem J. 1991 Jan 1;273(Pt 1):63–66. doi: 10.1042/bj2730063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Endemann G., Dunn S. N., Cantley L. C. Bovine brain contains two types of phosphatidylinositol kinase. Biochemistry. 1987 Oct 20;26(21):6845–6852. doi: 10.1021/bi00395a039. [DOI] [PubMed] [Google Scholar]
- Ferrell J. E., Jr, Huestis W. H. Phosphoinositide metabolism and the morphology of human erythrocytes. J Cell Biol. 1984 Jun;98(6):1992–1998. doi: 10.1083/jcb.98.6.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janmey P. A., Stossel T. P. Gelsolin-polyphosphoinositide interaction. Full expression of gelsolin-inhibiting function by polyphosphoinositides in vesicular form and inactivation by dilution, aggregation, or masking of the inositol head group. J Biol Chem. 1989 Mar 25;264(9):4825–4831. [PubMed] [Google Scholar]
- Kaplan D. R., Whitman M., Schaffhausen B., Pallas D. C., White M., Cantley L., Roberts T. M. Common elements in growth factor stimulation and oncogenic transformation: 85 kd phosphoprotein and phosphatidylinositol kinase activity. Cell. 1987 Sep 25;50(7):1021–1029. doi: 10.1016/0092-8674(87)90168-1. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Ling L. E., Schulz J. T., Cantley L. C. Characterization and purification of membrane-associated phosphatidylinositol-4-phosphate kinase from human red blood cells. J Biol Chem. 1989 Mar 25;264(9):5080–5088. [PubMed] [Google Scholar]
- Majerus P. W., Ross T. S., Cunningham T. W., Caldwell K. K., Jefferson A. B., Bansal V. S. Recent insights in phosphatidylinositol signaling. Cell. 1990 Nov 2;63(3):459–465. doi: 10.1016/0092-8674(90)90442-h. [DOI] [PubMed] [Google Scholar]
- Porter F. D., Li Y. S., Deuel T. F. Purification and characterization of a phosphatidylinositol 4-kinase from bovine uteri. J Biol Chem. 1988 Jun 25;263(18):8989–8995. [PubMed] [Google Scholar]
- Schacht J. Purification of polyphosphoinositides by chromatography on immobilized neomycin. J Lipid Res. 1978 Nov;19(8):1063–1067. [PubMed] [Google Scholar]
- Stephens L., Hawkins P. T., Downes C. P. Metabolic and structural evidence for the existence of a third species of polyphosphoinositide in cells: D-phosphatidyl-myo-inositol 3-phosphate. Biochem J. 1989 Apr 1;259(1):267–276. doi: 10.1042/bj2590267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker D. H., Dougherty N., Pike L. J. Purification and characterization of a phosphatidylinositol kinase from A431 cells. Biochemistry. 1988 Aug 23;27(17):6504–6511. doi: 10.1021/bi00417a046. [DOI] [PubMed] [Google Scholar]
- Whitman M., Cantley L. Phosphoinositide metabolism and the control of cell proliferation. Biochim Biophys Acta. 1989 Feb;948(3):327–344. doi: 10.1016/0304-419x(89)90005-x. [DOI] [PubMed] [Google Scholar]
- Whitman M., Downes C. P., Keeler M., Keller T., Cantley L. Type I phosphatidylinositol kinase makes a novel inositol phospholipid, phosphatidylinositol-3-phosphate. Nature. 1988 Apr 14;332(6165):644–646. doi: 10.1038/332644a0. [DOI] [PubMed] [Google Scholar]
- Whitman M., Kaplan D. R., Schaffhausen B., Cantley L., Roberts T. M. Association of phosphatidylinositol kinase activity with polyoma middle-T competent for transformation. Nature. 1985 May 16;315(6016):239–242. doi: 10.1038/315239a0. [DOI] [PubMed] [Google Scholar]
- Whitman M., Kaplan D., Roberts T., Cantley L. Evidence for two distinct phosphatidylinositol kinases in fibroblasts. Implications for cellular regulation. Biochem J. 1987 Oct 1;247(1):165–174. doi: 10.1042/bj2470165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray W., Boulikas T., Wray V. P., Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. doi: 10.1016/0003-2697(81)90179-2. [DOI] [PubMed] [Google Scholar]
- Yamakawa A., Takenawa T. Purification and characterization of membrane-bound phosphatidylinositol kinase from rat brain. J Biol Chem. 1988 Nov 25;263(33):17555–17560. [PubMed] [Google Scholar]
- Yamamoto K., Graziani A., Carpenter C., Cantley L. C., Lapetina E. G. A novel pathway for the formation of phosphatidylinositol 3,4-bisphosphate. Phosphorylation of phosphatidylinositol 3-monophosphate by phosphatidylinositol-3-monophosphate 4-kinase. J Biol Chem. 1990 Dec 25;265(36):22086–22089. [PubMed] [Google Scholar]