Abstract
Background
Transanal total mesorectal excision (TaTME), a novel approach for treating low rectal cancer, holds promise. However, concerns exist in certain countries about their oncologic safety due to less-than-optimal outcomes on global studies. This research seeks to evaluate the long-term oncologic outcomes focusing on local recurrence rate and overall survival after TaTME surgery in Germany.
Patients and methods
This study analyzed data from patients who underwent elective TaTME surgery between 2014 and 2021 in four certified colorectal cancer centers in Germany. Primary endpoints were 3-year local recurrence rate and local recurrence-free survival (LRFS). Secondary outcomes encompassed overall survival (OS), operative time, completeness of local tumor resection, lymph node resection, and postoperative complications.
Results
A total of 378 patients were analyzed (mean age 61.6 years; 272 males, 72%). After a median follow-up period of 2.5 years, 326 patients with UICC-stages I–III and tumor operability included in survival analyses. Local recurrence was observed in 8 individuals, leading to a 3-year cumulative local recurrence rate of 2.2% and a 3-year LRFS rate of 88.1%. The 3-year OS rate stood at 88.9%. Within 30 days after surgery, anastomotic leakage occurred in 19 cases (5%), whereas a presacral abscess was present in 12 patients (3.2%).
Conclusion
TaTME proves effective in addressing the anatomical and technical challenges of low rectal surgery and is associated with pleasing short- and long-term results. However, its safe integration into surgical routine necessitates sufficient knowledge and a previously completed training program.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00384-024-04704-w.
Keywords: Rectal cancer, Transanal total mesorectal excision, Short-term outcome, Long-term outcome, Oncologic safety
Introduction
Colorectal cancer ranks third in global malignancy prevalence, with a recorded global incidence of 1.9 million cases in 2020 [1]. In localized tumors, surgical resection of the affected bowel segment together with lymphadenectomy represents the most important part of a curative treatment approach. Over the past decades, surgical treatment options have evolved from open procedures to minimally invasive techniques [2, 3]. Among the contemporary minimally invasive techniques for resection of rectal tumors is transanal total mesorectal excision (TaTME), a technique devised to surmount the anatomical complexities of rectal surgery by using a two-stage approach involving laparoscopic abdominal and endoscopic transanal preparation [4–6]. This process affords enhanced visualization of the pelvic planes and consequently improves nerve-orientated mobilization of the distal rectum [7], which is believed to lead to a higher rate of R0 resections and a diminished frequency of conversions to open procedures compared to laparoscopic total mesorectal excision (LapTME) [8].
While trials in centers—predominantly with high expertise—have shown encouraging results in terms of a 3-year local recurrence rate ranging from 2.0 to 6.6%, an observational study by Larsen et al. raised apprehensions regarding the oncologic integrity of the novel technique [9–16]. This study, based on data sourced from the Norwegian Cancer Registry, identified an unusual pattern of local recurrence after TaTME, observed at a rate of 9.5% after a median follow-up of 11 months. In contrast, the local recurrence rate obtained over the same follow-up period for TME was only 3.4%. However, TaTME procedures in this study were performed by a substantial number of hospitals with low institutional caseloads and, consequently, a lack of experience with this challenging surgical technique [14, 17, 18]. Furthermore, neoadjuvant treatment was not offered to 90% of the patients with recurrent rectal cancer, despite their advanced disease stages. With regard to the doubts about the oncological success rate of TaTME, another study was recently published by Swiss surgeons comparing the outcome of the laparoscopic TME technique with the new TaTME technique. Although the authors initially appeared to be skeptical, their study showed no significant difference in the local recurrence rate of the two surgical procedures [19]. Despite the rather small patient population from Ammann et al., this finding is also consistent with a recent review [16].
The aim of the present study was to contribute to the growing evidence base on the TaTME technique by analyzing the short- and long-term oncologic outcomes of patients undergoing TaTME surgery in four certified colorectal cancer centers across Germany.
Patients and methods
Study setting
This is a retrospective analysis of prospectively collected data of four certified colorectal cancer centers in Germany (Caritas Krankenhaus St. Josef Regensburg, Charité Universitätsmedizin Berlin, the University Medical Center of the Johannes Gutenberg University Mainz, and the Klinikum Leverkusen). Prior to implementing the TaTME technique on patients, participating surgeons underwent a dedicated training program aligned with the consensus on structured training curriculum for transanal total mesorectal excision [20]. In addition, all cases were presented to an interdisciplinary tumor board prior to surgical intervention to discuss the best possible treatment options for each individual patient. Preoperative procedures related to diagnosis, preoperative staging, and additional treatment options such as radiochemotherapy were carried out according to the German S3-treatment guideline [21]. Since the current study is a retrospective analysis of data in which neither animal nor human experiments were performed, no ethics approval was required. All patients signed a declaration of consent which explicitly stated that their data would be used in pseudonymized form for study purposes. Since the four participating hospitals are certified national training centers for colorectal surgery and TaTME in particular, the surgical intervention was comparable between the individual clinics and regular meetings were held to share knowledge. Details of the surgical procedure have been described previously [12, 13].
Data collection and inclusion criteria
Data were collected individually by the clinics and subsequently compiled by the authors and processed for common analysis. Patients and their data were recruited from hospital documents on diagnosis and treatment, completed by data from regional cancer registries on follow-up regarding survival. In total, data from 378 patients with primary rectal adenocarcinoma who underwent elective TaTME surgery between 2014 and 2021 were analyzed. In order to avoid selection bias, all patients receiving TaTME in the defined recruitment period were included, except few patients (N = x) with incomplete data. Patients with inoperability of the primary tumor and patients classified as Union for International Cancer Control (UICC) stage IV were excluded from survival analyses. Thus, 326 patients were included in the survival analysis. Patients with limited follow-up time were right-censored in survival analysis. The dataset contains information on the preoperative, intraoperative, and postoperative period. Preoperatively, data regarding patient demographics, tumor characteristics, and additional therapies were collected. Intraoperative data included the extent of local tumor resection, operative time, and the number of harvested lymph nodes. Postoperative data contained information on survival, recurrence, and postoperative complications according to the Clavien–Dindo classification. In this context, Clavien–Dindo I or II is defined as minor complications that can be treated with pharmaceutical agents and do not lead to further invasive treatment. Patients classified as Clavien–Dindo III require surgical, endoscopic, or radiologic intervention. Clavien–Dindo IV is classified as a life-threatening complication such as single or multiorgan dysfunction requiring critical care management [22, 23].
Statistical analysis and outcome
The primary endpoints were 3-year local recurrence rate and local recurrence-free survival (LRFS). Secondary outcomes included overall survival (OS), operative time, completeness of local tumor resection, lymph node resection, and postoperative complications. Descriptive statistics involved categorization for nominal variables and calculating the median and mean, along with standard deviation (SD) and interquartile range (IQR), for scalar variables. Mean and median follow-up were determined using the reverse Kaplan–Meier method. Surgery date served as the starting point for all Kaplan–Meier survival analyses, with events including local recurrence, metachronous distant metastasis, and death. Recurrences within 3 months after surgery were regarded as early events. If a patient experienced multiple events, only the first was considered an endpoint for the corresponding analyses. For the statistical analyses, IBM SPSS 29 (IBM Corp., SPSS for Windows, Armonk, NY, USA) was used.
Results
Patient and tumor characteristics
Between 2014 and 2021, a total of 378 patients were treated using the TaTME technique. The four participating centers contributed 42, 54, 100, and 182 cases, respectively. Patient demographics and tumor characteristics are summarized in Tables 1 and 2. The majority of patients were male (n = 272, 72%). At the time of surgery, the mean age was 61.6 years (SD 12.2 years), with a minimum of 20 and a maximum of 87.1 years (median age 62.3 years, IQR 54.1–70.9 years). The patients’ body mass index (BMI) averaged 26.6 kg/m2 (range 15.8–45.2 kg/m2). According to the American Society of Anesthesiologists physical status classification system (ASA), 269 patients were classified as ASA 1 or 2 (71.2%), 95 as ASA 3 (25.1%), and 8 as ASA 4 (2.1%). The majority of patients (n = 243, 64.3%) presented initially with tumors classified as UICC stage III, while 32 patients (8.5%) had metastatic disease at diagnosis and were therefore classified as UICC stage IV. Following the German guidelines, 273 patients (72.2%) underwent neoadjuvant therapy and 227 cases (60.1%) received adjuvant therapy postoperatively. After neoadjuvant therapy and/or surgery, 132 patients were classified as UICC stage I (34.9%), 58 as UICC stage II (15.3%), and 87 as UICC stage III (23.0%); the number of patients with UICC stage IV increased to 42 (11.1%). In 59 patients with preoperative neoadjuvant treatment (15.6%), tumor tissue was no longer detectable postoperatively in the specimens removed (ypT0). Over 40% of the patients had tumors < 6 cm (Table 2).
Table 1.
Patient demographics (BMI, body mass index; ASA, American Society of Anesthesiologists Physical Status System; ns, not specified; TaTME, transanal total mesorectal excision)
| Study cohort | |||
|---|---|---|---|
| TaTME patients | |||
| n | % | ||
| Sex | Male | 272 | 72.0 |
| Female | 106 | 28.0 | |
| Age at diagnosis (years) | < 50 | 62 | 16.4 |
| 50–59 | 107 | 28.3 | |
| 60–69 | 111 | 29.4 | |
| 70–79 | 84 | 22.2 | |
| ≥ 80 | 14 | 3.7 | |
| BMI (kgm−2) | < 20 (underweight) | 18 | 4.8 |
| 20–25 (normal weight) | 140 | 37.0 | |
| 25–30 (overweight) | 128 | 33.9 | |
| ≥ 0 (obese) | 83 | 22.0 | |
| ns | 9 | 2.4 | |
| ASA | I | 23 | 6.1 |
| II | 246 | 65.1 | |
| III | 95 | 25.1 | |
| IV | 8 | 2.1 | |
| ns | 6 | 1.6 | |
| Total | 378 | 100.0 | |
Table 2.
Tumor characteristics (UICC, Union for International Cancer Control staging system; ns, not specified; TaTME, transanal total mesorectal excision)
| Study cohort | |||
|---|---|---|---|
| TaTME patients | |||
| n | % | ||
| Tumor height (third UICC) | < 6 cm | 156 | 41.3 |
| 6–11 cm | 208 | 55.0 | |
| ≥ 12 cm | 2 | 0.5 | |
| ns | 12 | 3.2 | |
| Clinical UICC stage | I | 55 | 14.6 |
| II | 48 | 12.7 | |
| III | 243 | 64.3 | |
| IV | 32 | 8.5 | |
| cT clinical tumor size | T1 | 21 | 5.6 |
| T2 | 68 | 18.0 | |
| T3 | 259 | 68.5 | |
| T4 | 15 | 4.0 | |
| TX/ns | 15 | 4.0 | |
| cN clinical nodal status | cN0 | 108 | 28.6 |
| cN1 | 229 | 60.6 | |
| cN2 | 41 | 10.8 | |
| cM clinical metastases status | cM0 | 346 | 91.5 |
| cM1 | 32 | 8.5 | |
| Postoperative UICC stage | ypT0/Tis | 59 | 15.6 |
| I | 132 | 34.9 | |
| II | 58 | 15.3 | |
| III | 87 | 23.0 | |
| IV | 42 | 11.1 | |
| pT postoperative tumor size | T0 | 64 | 16.9 |
| T1 | 41 | 10.8 | |
| T2 | 122 | 32.3 | |
| T3 | 140 | 37.0 | |
| T4 | 11 | 2.9 | |
| N postoperative nodal status | pN0 | 262 | 69.3 |
| pN1 | 76 | 20.1 | |
| pN2 | 40 | 10.6 | |
| M postoperative metastases status | cM0 | 336 | 88.9 |
| c/pM1 | 42 | 11.1 | |
| Total | 378 | 100.0 | |
Treatment characteristics
The median operative time was 234 min (IQR 186–328 min). Over the surveyed period, a reduction in the median operative time from 258 min in 2014 to 213 min in 2021 was observed. The median postoperative hospital stay was 9.0 days (mean 13.5 days, IQR 7–14 days). Complete resection of rectal cancer was possible in 364 patients (96.3%). In 87.3% of instances, a total of 12 or more lymph nodes were collected.
Postoperative results and survival analysis
In terms of postoperative complications, 41 patients (10.8%) were classified as Clavien–Dindo III and 9 patients (2.4%) as Clavien–Dindo IV. Within 30 days after surgery, anastomotic leakage occurred in 19 cases (5%), whereas a presacral abscess was present in 12 patients (3.2%; Table 3). Among 326 patients included in survival analyses, the mean follow-up was 2.4 years (median 2.5 years). Over the observation period, death occurred in 28 patients, 8 patients experienced local recurrence, and there were 37 cases of recurrent distant metastasis (Table 4). This led to a 3-year cumulative rate of 2.2% for local recurrences and 13.9% for metachronous distant metastases (Figs. 1 and 2). The Kaplan–Meier 3-year OS rate was 88.9% (Fig. 3). The 3-year cumulative LRFS rate was 88.1% (Fig. 4).
Table 3.
Treatment, quality indicators, and postoperative complications (ns, not specified; TaTME, transanal total mesorectal excision)
| Study cohort | |||
|---|---|---|---|
| TaTME patients | |||
| n | % | ||
| Neoadjuvant therapy | Yes | 273 | 72.2 |
| No | 105 | 27.8 | |
| Adjuvant therapy | Yes | 227 | 60.1 |
| No | 151 | 39.9 | |
| Lymph nodes harvested | < 12 | 43 | 11.4 |
| ≥ 12 | 330 | 87.3 | |
| ns | 5 | 1.3 | |
| Residual tumor local | R0 | 364 | 96.3 |
| R1/2 | 13 | 3.4 | |
| RX/ns | 1 | 0.3 | |
| Reoperation (30 days) | Yes | 11 | 2.9 |
| No | 367 | 97.1 | |
| Clavien–Dindo | No complication | 86 | 22.8 |
| Grade I | 22 | 5.8 | |
| Grade II | 37 | 9.8 | |
| Grade III | 41 | 10.8 | |
| Grade IV | 9 | 2.4 | |
| Grade V | 4 | 1.1 | |
| ns | 179 | 47.4 | |
| Anastomotic leak (30 days) | Yes | 19 | 5.0 |
| No | 359 | 95.0 | |
| Presacral abscess (30 days) | Yes | 12 | 3.2 |
| No | 366 | 96.8 | |
| Total | 378 | 100.0 | |
Table 4.
Life status and recurrence
| Study cohort | |||
|---|---|---|---|
| TaTME patients | |||
| n | % | ||
| Life status | Alive | 298 | 91.4 |
| Deceased | 28 | 8.6 | |
| Local recurrence status | No recurrence | 318 | 97.5 |
| Recurrence | 8 | 2.5 | |
| Distant recurrence status | No recurrence | 289 | 88.7 |
| Recurrence | 37 | 11.3 | |
| Recurrence status | No recurrence | 283 | 86.8 |
| Recurrence | 43 | 13.2 | |
| Local recurrence/life status | Alive without recurrence | 293 | 89.9 |
| Deceased or recurrence | 33 | 10.1 | |
| Distant recurrence/life status | Alive without recurrence | 271 | 83.1 |
| Deceased or recurrence | 55 | 16.9 | |
| Recurrence/life status | Alive without recurrence | 266 | 81.6 |
| Deceased or recurrence | 60 | 18.4 | |
| Total | 326 | 100.0 | |
Fig. 1.
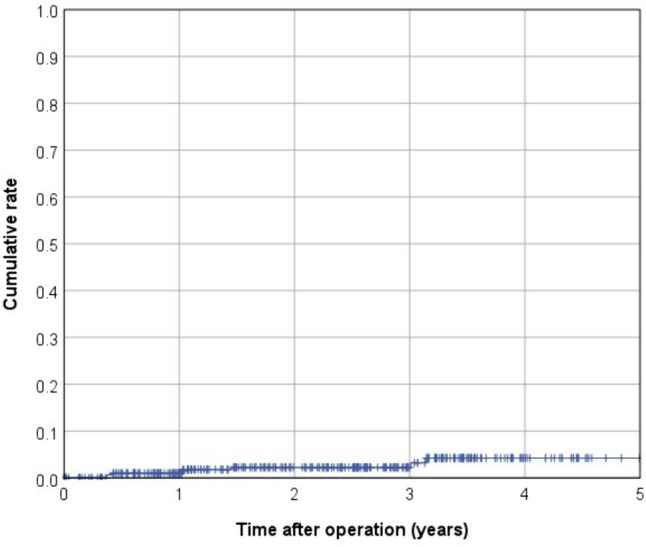
Kaplan–Meier curve for 3-year cumulative local recurrence rate
Fig. 2.
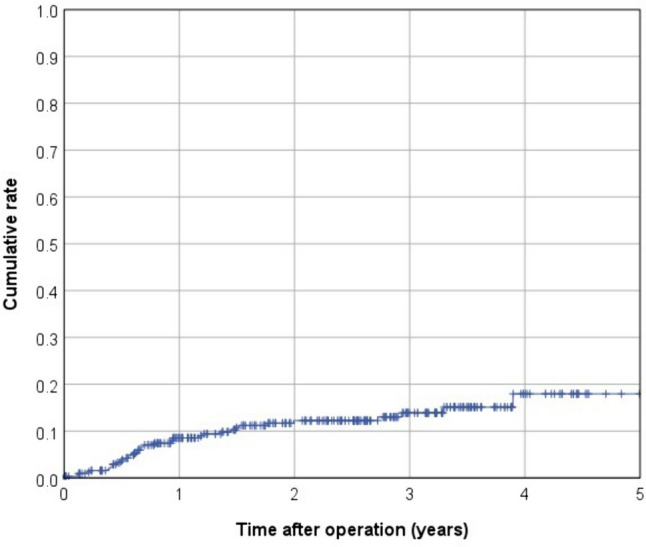
Kaplan–Meier curve for 3-year cumulative distant metastasis rate
Fig. 3.
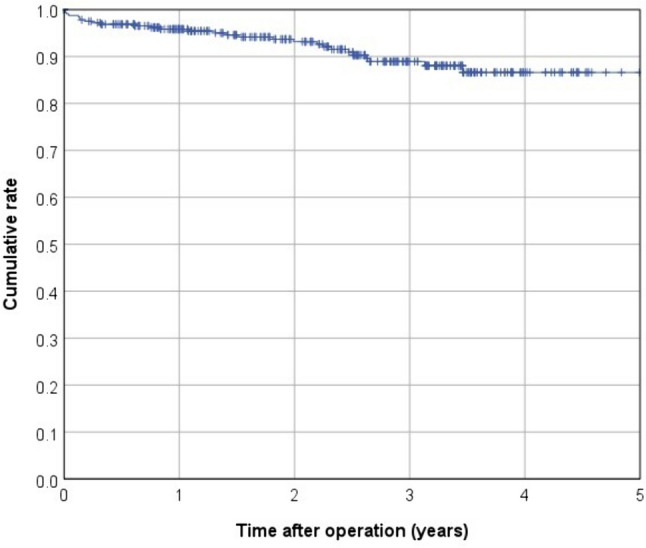
Kaplan–Meier curve for overall survival (OS)
Fig. 4.
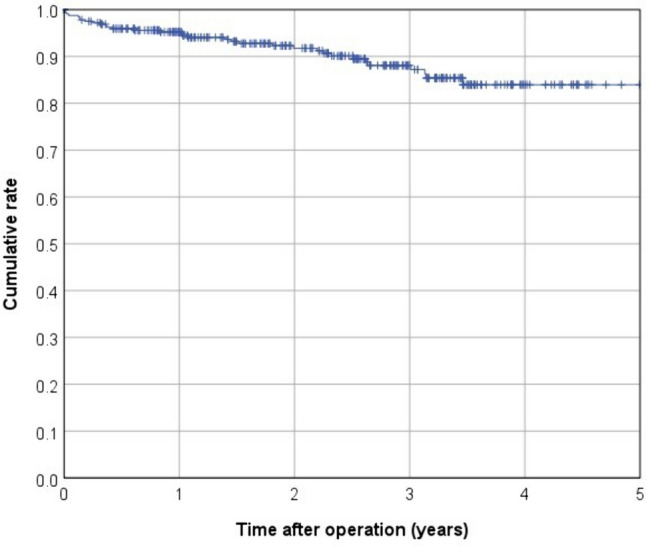
Kaplan–Meier curve for local recurrence-free survival (LRFS)
Discussion
This study presents the short- and long-term effects of applying TaTME in four certified colorectal centers in Germany. The beneficial outcome is particularly evident in the low rate of local recurrences, with a 3-year cumulative rate of 2.2% and a 3-year cumulative OS rate of 88.9%. These findings align closely with other international research regarding TaTME surgery. A study by Hol et al. published in 2019 showed a 3-year local recurrence rate of 2% and 3-year OS of 83.6% [10]. Fortunately, the current results show greater success with regard to postoperative complications: the reoperation rate in Hol et al. was 22.6%, whereas, in the present study, reoperation occurred in only 2.9% of cases, and the results are also better in terms of anastomotic leakage (5.0% vs. Hol et al. 6.3%) and presacral abscesses (3.2% vs. Hol et al. 8.8%). Similarly, a multicenter study conducted in Canada showed a local recurrence rate of 3.6% after a median follow-up of 27 months [24]. With regard to postoperative complications, the distribution of patients according to the Clavien–Dindo classification in the current study exhibits a slight advantage compared to the findings of the Canadian colleagues (Clavien–Dindo III 10.8% vs. Caycedo-Marulanda et al. 14.5%) and the results of anastomotic leakage are also more encouraging (5.0% vs. Caycedo-Marulanda et al. 7.6%). Furthermore, the present patient collective shows significantly less favorable tumor and patient characteristics: the majority of cases was classified clinically as UICC stage III (64.3%), whereas in the Canadian study only 42.1% presented with UICC stage III and the majority of cases were UICC stage I (21.7%) and II (27.3%) [24]. In addition, there are already studies comparing the oncological outcomes between TaTME and LapTME. In 2020, Lacy et al. published a multicenter comparative analysis showing the superiority of TaTME regarding the 3-year cumulative local recurrence rate of 3.6% compared to LapTME (9.6%). This study also demonstrated a superiority of TaTME over LapTME in terms of overall survival (87.2% vs. 82.2%) [9].
Over 40% of the present patients had tumors located in the lower third of the rectum. Anatomical limitations, especially in the narrow, obese, and male pelvis, thus with a limited view to the distal resection level, can be sufficiently addressed with the transanal approach without the demand for a linear stapling device [25]. Regarding the average duration of surgery, an interesting trend emerges from 2014 to 2021: the overall data from all hospitals indicate a reduction from 270 to 231 min; however, a specific hospital stood out in 2021, with an average operative time of 178 min. This suggests that certain facilities have found ways to operate particularly efficiently. The longer average surgery duration in other hospitals might be attributed to potential staff shortages, making it challenging for them to simultaneously conduct this specific type of surgery. These disparities highlight the importance of resource management and could serve as points of focus for healthcare improvements.
Furthermore, this study has several limitations. First, it is a retrospective cohort analysis of data from four colorectal cancer centers in Germany. Patients diagnosed with rectal cancer were screened for eligibility and then included in further analysis. Since the design was not that of a randomized controlled trial, there was no comparison group and patient selection bias also cannot be excluded. In addition, adequate training has been shown to be an important factor in surgical success and oncologic safety in the past. For instance, the results of the Norwegian colleagues, with a local recurrence rate of 9.5% after performing TaTME, show inadequate oncologic results. This led to the Norwegian Colorectal Cancer Group temporarily halting the use of TaTME for rectal cancer in December 2018. As mentioned in the introduction, these were not specialized colorectal cancer centers and the surgeons were not sufficiently trained in advance with regard to the special circumstances of the TaTME technique [14, 17]. Therefore, our results are not fully reproducible insofar as inexperienced surgeons and low-volume centers regarding TaTME are concerned [26]. Thus, in 2017, Francis et al. called for a structured training curriculum to safely integrate TaTME into daily surgical practice. This program was divided into four steps: self-study, cadaver workshop, proctorship, and independent practice. After the surgeons had familiarized themselves theoretically with the TaTME technique, a subsequent cadaver workshop followed in order to train TaTME on the model. This was followed by the so-called proctorship, i.e., performance of the first TaTME operations under the guidance of an experienced TaTME surgeon. The fourth and final step was independent application in clinical practice [20]. It can be expected that with sufficient preparation and training, pleasing results can be achieved. To inform future steps towards wider implementation of TaTME training and clinical application, more evidence from randomized controlled trials like COLORIII is eagerly awaited.
Conclusion
Transanal total mesorectal excision is a challenging surgical procedure whose benefits in terms of local recurrence and oncologic safety are realized after sufficient training programs. The results of the current study are promising in terms of the international comparison and demonstrate successful application of the TaTME technique in Germany.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank the participating clinics for their cooperation.
Author contribution
Elena Grundler (Caritas Krankenhaus St. Josef Regensburg, Germany) wrote the main manuscript text and Michael Gerken (University of Regensburg, Germany) and Vinzenz Völkel (University of Regensburg, Germany) prepared the tables and figures. Matthias Biebl and Luca Dittrich were the contacts for patient data from Charité Universitätsmedizin Berlin, Germany. Andreas Rink was the contact person for the patient data from Universitätsmedizin Mainz and Klinikum Leverkusen, Germany. Elena Grundler and Michael Gerken collected the data in pseudo-anonymized form and prepared it for analysis. All authors reviewed the manuscript.
Funding
Open Access funding enabled and organized by Prof. Alois Fürst.
Data availability
This is a retrospective analysis of prospectively collected data of four certified colorectal cancer centers in Germany (Caritas Krankenhaus St. Josef Regensburg, Charité Universitätsmedizin Berlin, the University Medical Center of the Johannes Gutenberg University Mainz, and the Klinikum Leverkusen). The data was collected and analysed by the authors.
Declarations
Informed consent
Based on a retrospective analysis and a fully anonymized set of clinical data, signing an informed consent form was not required.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Vinzenz Völkel and Alois Fürst have contributed equally to this work.
References
- 1.Xi Y, Xu P (2021) Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol 14(10):101174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Draeger T, Völkel V, Gerken M, Klinkhammer-Schalke M, Fürst A (2018) Long-term oncologic outcomes after laparoscopic versus open rectal cancer resection: a high-quality population-based analysis in a Southern German district. Surg Endosc 32(10):4096–4104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bonjer HJ, Deijen CL, Abis GA, Cuesta MA, van der Pas MHGM, de Lange-de Klerk ESM et al (2015) A randomized trial of laparoscopic versus open surgery for rectal cancer. N Engl J Med 372(14):1324–32 [DOI] [PubMed] [Google Scholar]
- 4.Sylla P, Rattner DW, Delgado S, Lacy AM (2010) NOTES transanal rectal cancer resection using transanal endoscopic microsurgery and laparoscopic assistance. Surg Endosc 24(5):1205–1210 [DOI] [PubMed] [Google Scholar]
- 5.Rink AD, Aigner F, Biebl M, Fürst A, Kneist W (2019) Transanale totale mesorektale Exzision – eine sinnvolle Operationstechnik zur individualisierten Behandlung von Patienten mit Rektumkarzinom. coloproctology 41(4):272–9 [Google Scholar]
- 6.Kneist W, Aigner F (2015) Total mesorectal excision via transanal minimally invasive surgery. An alternative technique. coloproctology 37:253–61 [Google Scholar]
- 7.Kneist W, Rink AD, Kauff DW, Konerding MA, Lang H (2015) Topography of the extrinsic internal anal sphincter nerve supply during laparoscopic-assisted TAMIS TME: five key zones of risk from the surgeons’ view. Int J Colorectal Dis 30(1):71–78 [DOI] [PubMed] [Google Scholar]
- 8.Hajibandeh S, Hajibandeh S, Eltair M, George AT, Thumbe V, Torrance AW et al (2020) Meta-analysis of transanal total mesorectal excision versus laparoscopic total mesorectal excision in management of rectal cancer. Int J Colorectal Dis 35(4):575–593 [DOI] [PubMed] [Google Scholar]
- 9.de Lacy FB, Roodbeen SX, Ríos J, van Laarhoven J, Otero-Piñeiro A, Bravo R et al (2020) Three-year outcome after transanal versus laparoscopic total mesorectal excision in locally advanced rectal cancer: a multicenter comparative analysis. BMC Cancer 20(1):677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hol JC, van Oostendorp SE, Tuynman JB, Sietses C (2019) Long-term oncological results after transanal total mesorectal excision for rectal carcinoma. Tech Coloproctol 23(9):903–911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.van Oostendorp SE, Belgers HJE, Hol JC, Doornebosch PG, Belt EJT, Oosterling SJ et al (2021) The learning curve of transanal total mesorectal excision for rectal cancer is associated with local recurrence: results from a multicentre external audit. Colorectal Dis 23(8):2020–2029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dittrich L, Biebl M, Schmuck R, Gül S, Weiss S, Haase O, Knoop M, Alkatout I, Pratschke J, Aigner F (2020) Initial experience with the safe implementation of transanal total mesorectal excision (TaTME) as a standardized procedure for low rectal cancer. J Clin Med 10(1):72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Völkel V, Schatz S, Draeger T, Gerken M, Klinkhammer-Schalke M, Fürst A (2022) Transanal total mesorectal excision: short- and long-term results of the first hundred cases of a certified colorectal cancer center in Germany. Surg Endosc 36(2):1172–1180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Larsen SG, Pfeffer F, Kørner H (2019) Norwegian moratorium on transanal total mesorectal excision. Br J Surg 106(9):1120–1121 [DOI] [PubMed] [Google Scholar]
- 15.Roodbeen SX, Spinelli A, Bemelman WA, Di Candido F, Cardepont M, Denost Q et al (2021) Local recurrence after transanal total mesorectal excision for rectal cancer: a multicenter cohort study. Ann Surg 274(2):359–366 [DOI] [PubMed] [Google Scholar]
- 16.Neary E, Ibrahim T, Verschoor CP, Zhang L, Patel SV, Chadi SA et al (2024) A systematic review and meta-analysis of oncological outcomes with transanal total mesorectal excision for rectal cancer. Colorectal Dis 26(5):837–850 [DOI] [PubMed] [Google Scholar]
- 17.Atallah S, Sylla P, Wexner SD (2019) Norway versus The Netherlands: will taTME stand the test of time? Tech Coloproctol 23(9):803–806 [DOI] [PubMed] [Google Scholar]
- 18.Deijen CL, Tsai A, Koedam TWA, VeltcampHelbach M, Sietses C, Lacy AM et al (2016) Clinical outcomes and case volume effect of transanal total mesorectal excision for rectal cancer: a systematic review. Tech Coloproctol 20(12):811–824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ammann Y, Warschkow R, Schmied B, de Lorenzi D, Reißfelder C, Bischofberger S et al (2024) Is survival after transanal total mesorectal excision (taTME) worse than that after traditional total mesorectal excision? A retrospective propensity score-adjusted cohort study. Int J Colorectal Dis 39(1):28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Francis N, Penna M, Mackenzie H, Carter F, Hompes R (2017) Consensus on structured training curriculum for transanal total mesorectal excision (TaTME). Surg Endosc 31(7):2711–2719 [DOI] [PubMed] [Google Scholar]
- 21.Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF): S3-Leitlinie Kolorektales Karzinom, Langversion 2.1, 2019, AWMF-Registernummer: 021/007OL [cited 2024 Aug 10]. Available from https://www.awmf.org/uploads/tx_szleitlinien/021-007OLl_S3_Kolorektales-Karzinom-KRK_2019-01.pdf
- 22.Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Clavien PA, Sanabria JR, Strasberg SM (1992) Proposed classification of complications of surgery with examples of utility in cholecystectomy. Surgery 111(5):518–526 [PubMed] [Google Scholar]
- 24.Caycedo-Marulanda A, Lee L, Chadi SA, Verschoor CP, Crosina J, Ashamalla S et al (2021) Association of transanal total mesorectal excision with local recurrence of rectal cancer. JAMA Netw Open 4(2):e2036330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kneist W, Aigner F (2022) Bedeutung des operativen Zugangs für das Outcome beim Karzinom des unteren Rektumdrittels. coloproctology 44(4):244–50 [Google Scholar]
- 26.Deijen CL, Tsai A, Koedam TWA, VeltcampHelbach M, Sietses C, Lacy AM et al (2016) Clinical outcomes and case volume effect of transanal total mesorectal excision for rectal cancer: a systematic review. Tech Coloproctol 20(12):811–824 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
This is a retrospective analysis of prospectively collected data of four certified colorectal cancer centers in Germany (Caritas Krankenhaus St. Josef Regensburg, Charité Universitätsmedizin Berlin, the University Medical Center of the Johannes Gutenberg University Mainz, and the Klinikum Leverkusen). The data was collected and analysed by the authors.


