Abstract
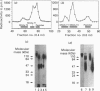
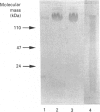
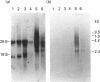
The low-molecular-mass human salivary mucin has at least two isoforms, MG2a and MG2b, that differ primarily in their sialic acid and fucose content. In this study, we characterize further these isoforms, particularly their peptide moieties. Trypsin digests of MG2a and MG2b yielded high- and low-molecular-mass glycopeptides following gel filtration on Sephacryl S-300. The larger glycopeptides from MG2a and MG2b had similar amino acid compositions and identical N-terminal sequences, suggesting common structural features between their peptides. An oligonucleotide probe generated from the amino acid sequence of the smaller glycopeptide from MG2a was employed in Northern-blot analysis. This probe specifically hybridized to two mRNA species from human submandibular and sublingual glands. A cDNA clone selected from a human submandibular gland cDNA expression library with antibody generated against deglycosylated MG2a also hybridized to these two mRNA species. In both cases, the larger mRNA was polydisperse, and the hybridization signal was more intense in the sublingual gland. In addition, the N-terminal amino acid sequence of the larger glycopeptide was found to be part of one of the selected MG2 cDNA clones.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bhargava A. K., Woitach J. T., Davidson E. A., Bhavanandan V. P. Cloning and cDNA sequence of a bovine submaxillary gland mucin-like protein containing two distinct domains. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6798–6802. doi: 10.1073/pnas.87.17.6798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bobek L. A., Aguirre A., Levine M. J. Human salivary cystatin S. Cloning, sequence analysis, hybridization in situ and immunocytochemistry. Biochem J. 1991 Sep 15;278(Pt 3):627–635. doi: 10.1042/bj2780627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Byrd J. C., Lamport D. T., Siddiqui B., Kuan S. F., Erickson R., Itzkowitz S. H., Kim Y. S. Deglycosylation of mucin from LS174T colon cancer cells by hydrogen fluoride treatment. Biochem J. 1989 Jul 15;261(2):617–625. doi: 10.1042/bj2610617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen R. E., Aguirre A., Neiders M. E., Levine M. J., Jones P. C., Reddy M. S., Haar J. G. Immunochemistry and immunogenicity of low molecular weight human salivary mucin. Arch Oral Biol. 1991;36(5):347–356. doi: 10.1016/0003-9969(91)90004-E. [DOI] [PubMed] [Google Scholar]
- Eckhardt A. E., Timpte C. S., Abernethy J. L., Zhao Y., Hill R. L. Porcine submaxillary mucin contains a cystine-rich, carboxyl-terminal domain in addition to a highly repetitive, glycosylated domain. J Biol Chem. 1991 May 25;266(15):9678–9686. [PubMed] [Google Scholar]
- Gendler S. J., Burchell J. M., Duhig T., Lamport D., White R., Parker M., Taylor-Papadimitriou J. Cloning of partial cDNA encoding differentiation and tumor-associated mucin glycoproteins expressed by human mammary epithelium. Proc Natl Acad Sci U S A. 1987 Sep;84(17):6060–6064. doi: 10.1073/pnas.84.17.6060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gendler S. J., Lancaster C. A., Taylor-Papadimitriou J., Duhig T., Peat N., Burchell J., Pemberton L., Lalani E. N., Wilson D. Molecular cloning and expression of human tumor-associated polymorphic epithelial mucin. J Biol Chem. 1990 Sep 5;265(25):15286–15293. [PubMed] [Google Scholar]
- Gerard C., Eddy R. L., Jr, Shows T. B. The core polypeptide of cystic fibrosis tracheal mucin contains a tandem repeat structure. Evidence for a common mucin in airway and gastrointestinal tissue. J Clin Invest. 1990 Dec;86(6):1921–1927. doi: 10.1172/JCI114925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbons R. J., Qureshi J. V. Selective binding of blood group-reactive salivary mucins by Streptococcus mutans and other oral organisms. Infect Immun. 1978 Dec;22(3):665–671. doi: 10.1128/iai.22.3.665-671.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gum J. R., Byrd J. C., Hicks J. W., Toribara N. W., Lamport D. T., Kim Y. S. Molecular cloning of human intestinal mucin cDNAs. Sequence analysis and evidence for genetic polymorphism. J Biol Chem. 1989 Apr 15;264(11):6480–6487. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lan M. S., Batra S. K., Qi W. N., Metzgar R. S., Hollingsworth M. A. Cloning and sequencing of a human pancreatic tumor mucin cDNA. J Biol Chem. 1990 Sep 5;265(25):15294–15299. [PubMed] [Google Scholar]
- Levine M. J., Herzberg M. C., Levine M. S., Ellison S. A., Stinson M. W., Li H. C., van Dyke T. Specificity of salivary-bacterial interactions: role of terminal sialic acid residues in the interaction of salivary glycoproteins with Streptococcus sanguis and Streptococcus mutans. Infect Immun. 1978 Jan;19(1):107–115. doi: 10.1128/iai.19.1.107-115.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine M. J., Reddy M. S., Tabak L. A., Loomis R. E., Bergey E. J., Jones P. C., Cohen R. E., Stinson M. W., Al-Hashimi I. Structural aspects of salivary glycoproteins. J Dent Res. 1987 Feb;66(2):436–441. doi: 10.1177/00220345870660020901. [DOI] [PubMed] [Google Scholar]
- Ligtenberg M. J., Vos H. L., Gennissen A. M., Hilkens J. Episialin, a carcinoma-associated mucin, is generated by a polymorphic gene encoding splice variants with alternative amino termini. J Biol Chem. 1990 Apr 5;265(10):5573–5578. [PubMed] [Google Scholar]
- Loomis R. E., Prakobphol A., Levine M. J., Reddy M. S., Jones P. C. Biochemical and biophysical comparison of two mucins from human submandibular-sublingual saliva. Arch Biochem Biophys. 1987 Nov 1;258(2):452–464. doi: 10.1016/0003-9861(87)90366-3. [DOI] [PubMed] [Google Scholar]
- Murray P. A., Levine M. J., Reddy M. S., Tabak L. A., Bergey E. J. Preparation of a sialic acid-binding protein from Streptococcus mitis KS32AR. Infect Immun. 1986 Aug;53(2):359–365. doi: 10.1128/iai.53.2.359-365.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray P. A., Levine M. J., Tabak L. A., Reddy M. S. Specificity of salivary-bacterial interactions: II. Evidence for a lectin on Streptococcus sanguis with specificity for a NeuAc alpha 2, 3Ga1 beta 1, 3Ga1NAc sequence. Biochem Biophys Res Commun. 1982 May 31;106(2):390–396. doi: 10.1016/0006-291x(82)91122-6. [DOI] [PubMed] [Google Scholar]
- Prakobphol A., Levine M. J., Tabak L. A., Reddy M. S. Purification of a low-molecular-weight, mucin-type glycoprotein from human submandibular-sublingual saliva. Carbohydr Res. 1982 Oct 1;108(1):111–122. doi: 10.1016/s0008-6215(00)81896-0. [DOI] [PubMed] [Google Scholar]
- Ramasubbu N., Reddy M. S., Bergey E. J., Haraszthy G. G., Soni S. D., Levine M. J. Large-scale purification and characterization of the major phosphoproteins and mucins of human submandibular-sublingual saliva. Biochem J. 1991 Dec 1;280(Pt 2):341–352. doi: 10.1042/bj2800341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy M. S., Levine M. J., Prakobphol A. Oligosaccharide structures of the low-molecular-weight salivary mucin from a normal individual and one with cystic fibrosis. J Dent Res. 1985 Jan;64(1):33–36. doi: 10.1177/00220345850640010601. [DOI] [PubMed] [Google Scholar]
- Stinson M. W., Levine M. J., Cavese J. M., Prakobphol A., Murray P. A., Tabak L. A., Reddy M. S. Adherence of Streptococcus sanguis to salivary mucin bound to glass. J Dent Res. 1982 Dec;61(12):1390–1393. doi: 10.1177/00220345820610120101. [DOI] [PubMed] [Google Scholar]
- Tabak L. A., Levine M. J., Jain N. K., Bryan A. R., Cohen R. E., Monte L. D., Zawacki S., Nancollas G. H., Slomiany A., Slomiany B. L. Adsorption of human salivary mucins to hydroxyapatite. Arch Oral Biol. 1985;30(5):423–427. doi: 10.1016/0003-9969(85)90070-6. [DOI] [PubMed] [Google Scholar]
- Timpte C. S., Eckhardt A. E., Abernethy J. L., Hill R. L. Porcine submaxillary gland apomucin contains tandemly repeated, identical sequences of 81 residues. J Biol Chem. 1988 Jan 15;263(2):1081–1088. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]