Abstract
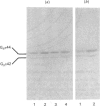
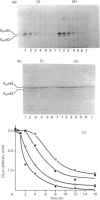
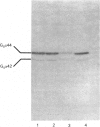
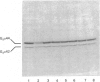
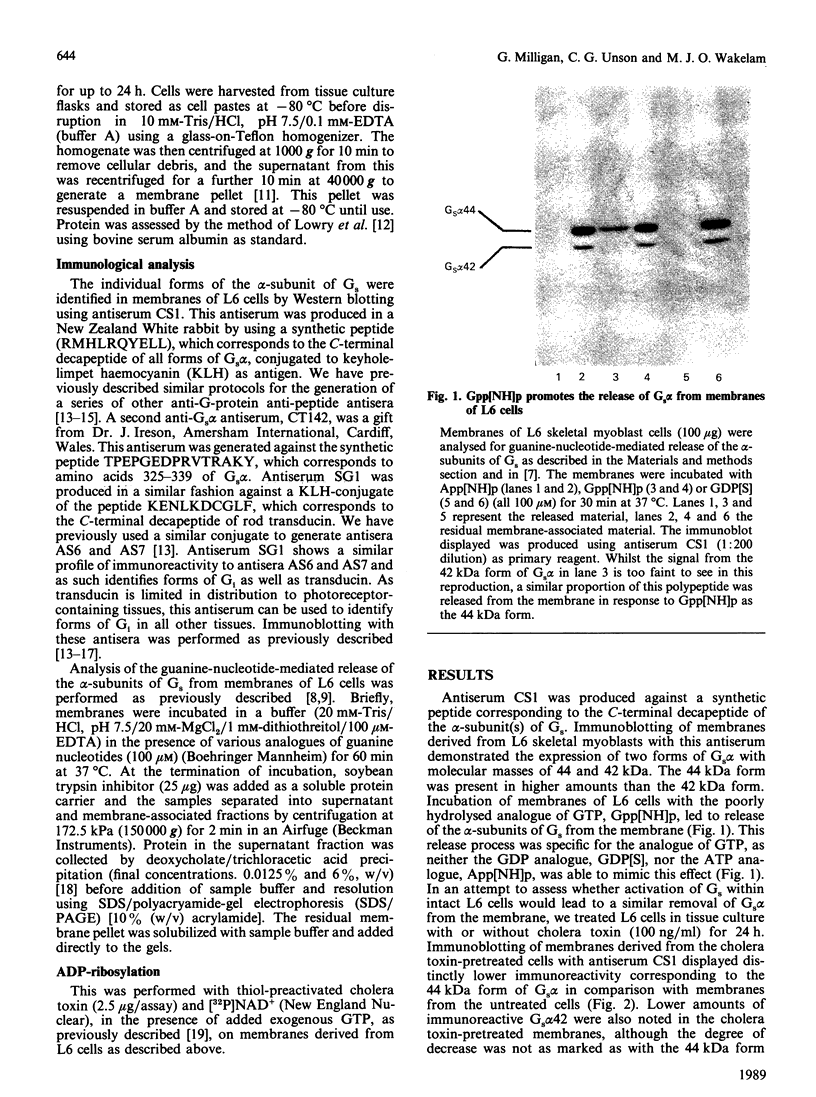
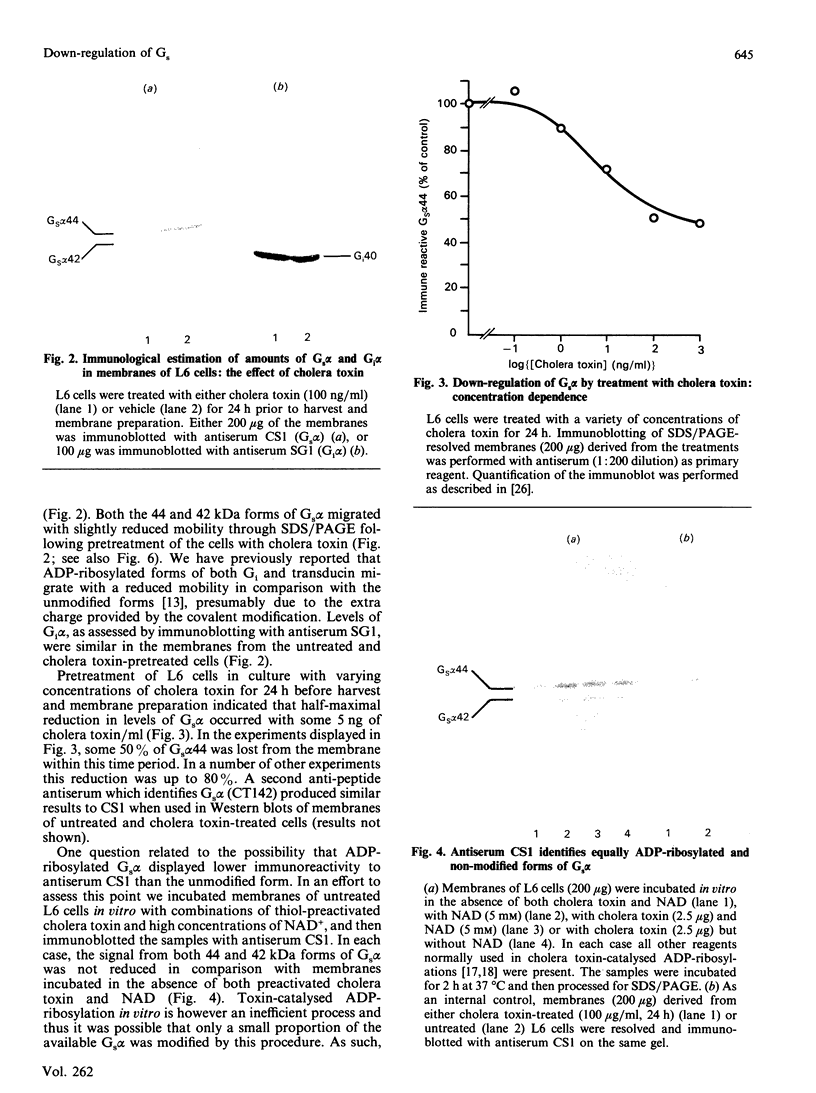
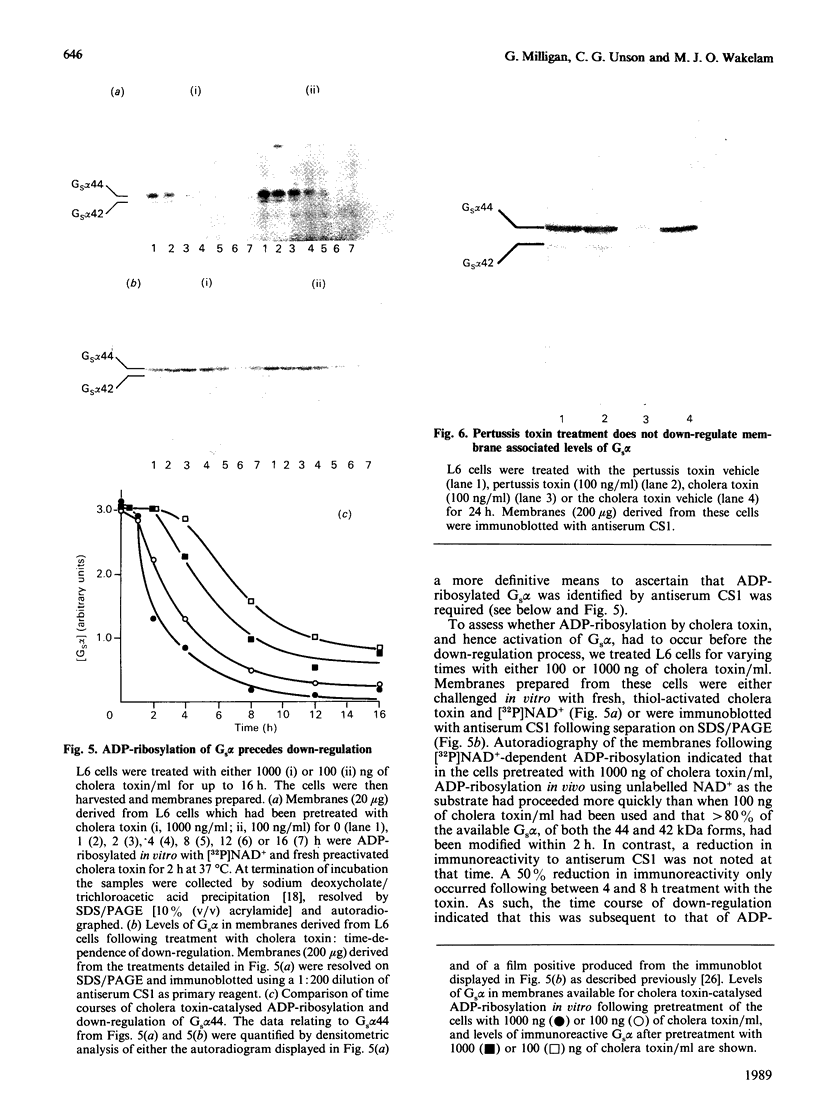
The effect of activation of the alpha-subunit(s) of the stimulatory guanine-nucleotide-binding protein, Gs, on levels of this polypeptide(s) associated with the plasma membrane of L6 skeletal myoblasts was ascertained. Incubation of these cells with cholera toxin led to a time- and concentration-dependent 'down-regulation' of both 44 and 42 kDa forms of Gs alpha as assessed by immunoblotting with an anti-peptide antiserum (CS1) able to identify the extreme C-terminus of Gs. The effect of cholera toxin was specific for Gs; levels of Gi alpha in membranes of cholera toxin-treated cells were not different from untreated cells. Down-regulation of Gs was absolutely dependent upon prior ADP-ribosylation, and hence activation of Gs and was not mimicked by other agents which elevate intracellular levels of cyclic AMP. Pretreatment with pertussis toxin, which catalyses ADP-ribosylation of Gi but not of Gs, did not down-regulate either Gi or Gs, demonstrating that covalent modification by ADP-ribosylation is alone not a signal for removal of G-proteins from the plasma membrane.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bray P., Carter A., Simons C., Guo V., Puckett C., Kamholz J., Spiegel A., Nirenberg M. Human cDNA clones for four species of G alpha s signal transduction protein. Proc Natl Acad Sci U S A. 1986 Dec;83(23):8893–8897. doi: 10.1073/pnas.83.23.8893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buss J. E., Mumby S. M., Casey P. J., Gilman A. G., Sefton B. M. Myristoylated alpha subunits of guanine nucleotide-binding regulatory proteins. Proc Natl Acad Sci U S A. 1987 Nov;84(21):7493–7497. doi: 10.1073/pnas.84.21.7493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gierschik P., Jakobs K. H. Receptor-mediated ADP-ribosylation of a phospholipase C-stimulating G protein. FEBS Lett. 1987 Nov 16;224(1):219–223. doi: 10.1016/0014-5793(87)80451-9. [DOI] [PubMed] [Google Scholar]
- Gierschik P., Milligan G., Pines M., Goldsmith P., Codina J., Klee W., Spiegel A. Use of specific antibodies to quantitate the guanine nucleotide-binding protein Go in brain. Proc Natl Acad Sci U S A. 1986 Apr;83(7):2258–2262. doi: 10.1073/pnas.83.7.2258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilman A. G. G proteins: transducers of receptor-generated signals. Annu Rev Biochem. 1987;56:615–649. doi: 10.1146/annurev.bi.56.070187.003151. [DOI] [PubMed] [Google Scholar]
- Goldsmith P., Gierschik P., Milligan G., Unson C. G., Vinitsky R., Malech H. L., Spiegel A. M. Antibodies directed against synthetic peptides distinguish between GTP-binding proteins in neutrophil and brain. J Biol Chem. 1987 Oct 25;262(30):14683–14688. [PubMed] [Google Scholar]
- Kahn R. A., Goddard C., Newkirk M. Chemical and immunological characterization of the 21-kDa ADP-ribosylation factor of adenylate cyclase. J Biol Chem. 1988 Jun 15;263(17):8282–8287. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- McArdle H., Mullaney I., Magee A., Unson C., Milligan G. GTP analogues cause release of the alpha subunit of the GTP binding protein, GO, from the plasma membrane of NG108-15 cells. Biochem Biophys Res Commun. 1988 Apr 15;152(1):243–251. doi: 10.1016/s0006-291x(88)80706-x. [DOI] [PubMed] [Google Scholar]
- McKenzie F. R., Kelly E. C., Unson C. G., Spiegel A. M., Milligan G. Antibodies which recognize the C-terminus of the inhibitory guanine-nucleotide-binding protein (Gi) demonstrate that opioid peptides and foetal-calf serum stimulate the high-affinity GTPase activity of two separate pertussis-toxin substrates. Biochem J. 1988 Feb 1;249(3):653–659. doi: 10.1042/bj2490653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G. Foetal-calf serum stimulates a pertussis-toxin-sensitive high-affinity GTPase activity in rat glioma C6 BU1 cells. Biochem J. 1987 Jul 15;245(2):501–505. doi: 10.1042/bj2450501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G. Guanine nucleotide regulation of the pertussis and cholera toxin substrates of rat glioma C6 BU1 cells. Biochim Biophys Acta. 1987 Jul 6;929(2):197–202. doi: 10.1016/0167-4889(87)90176-5. [DOI] [PubMed] [Google Scholar]
- Milligan G., Klee W. A. The inhibitory guanine nucleotide-binding protein (Ni) purified from bovine brain is a high affinity GTPase. J Biol Chem. 1985 Feb 25;260(4):2057–2063. [PubMed] [Google Scholar]
- Milligan G., McKenzie F. R. Opioid peptides promote cholera-toxin-catalysed ADP-ribosylation of the inhibitory guanine-nucleotide-binding protein (Gi) in membranes of neuroblastoma x glioma hybrid cells. Biochem J. 1988 Jun 1;252(2):369–373. doi: 10.1042/bj2520369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G., Mullaney I., Unson C. G., Marshall L., Spiegel A. M., McArdle H. GTP analogues promote release of the alpha subunit of the guanine nucleotide binding protein, Gi2, from membranes of rat glioma C6 BU1 cells. Biochem J. 1988 Sep 1;254(2):391–396. doi: 10.1042/bj2540391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G., Spiegel A. M., Unson C. G., Saggerson E. D. Chemically induced hypothyroidism produces elevated amounts of the alpha subunit of the inhibitory guanine nucleotide binding protein (Gi) and the beta subunit common to all G-proteins. Biochem J. 1987 Oct 1;247(1):223–227. doi: 10.1042/bj2470223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G. Techniques used in the identification and analysis of function of pertussis toxin-sensitive guanine nucleotide binding proteins. Biochem J. 1988 Oct 1;255(1):1–13. doi: 10.1042/bj2550001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan G., Unson C. G. Persistent activation of the alpha subunit of Gs promotes its removal from the plasma membrane. Biochem J. 1989 Jun 15;260(3):837–841. doi: 10.1042/bj2600837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullaney I., Magee A. I., Unson C. G., Milligan G. Differential regulation of amounts of the guanine-nucleotide-binding proteins Gi and Go in neuroblastoma x glioma hybrid cells in response to dibutyryl cyclic AMP. Biochem J. 1988 Dec 1;256(2):649–656. doi: 10.1042/bj2560649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. M., Houslay M. D., Milligan G., Siddle K. The insulin receptor tyrosyl kinase phosphorylates holomeric forms of the guanine nucleotide regulatory proteins Gi and Go. FEBS Lett. 1987 Feb 23;212(2):281–288. doi: 10.1016/0014-5793(87)81361-3. [DOI] [PubMed] [Google Scholar]
- Robishaw J. D., Smigel M. D., Gilman A. G. Molecular basis for two forms of the G protein that stimulates adenylate cyclase. J Biol Chem. 1986 Jul 25;261(21):9587–9590. [PubMed] [Google Scholar]
- Rotrosen D., Gallin J. I., Spiegel A. M., Malech H. L. Subcellular localization of Gi alpha in human neutrophils. J Biol Chem. 1988 Aug 5;263(22):10958–10964. [PubMed] [Google Scholar]
- Spiegel A. M. Signal transduction by guanine nucleotide binding proteins. Mol Cell Endocrinol. 1987 Jan;49(1):1–16. doi: 10.1016/0303-7207(87)90058-x. [DOI] [PubMed] [Google Scholar]
- Wakelam M. J., Patterson S., Hanley M. R. L6 skeletal muscle cells have functional V1-vasopressin receptors coupled to stimulated inositol phospholipid metabolism. FEBS Lett. 1987 Jan 5;210(2):181–184. doi: 10.1016/0014-5793(87)81333-9. [DOI] [PubMed] [Google Scholar]
- Yatani A., Imoto Y., Codina J., Hamilton S. L., Brown A. M., Birnbaumer L. The stimulatory G protein of adenylyl cyclase, Gs, also stimulates dihydropyridine-sensitive Ca2+ channels. Evidence for direct regulation independent of phosphorylation by cAMP-dependent protein kinase or stimulation by a dihydropyridine agonist. J Biol Chem. 1988 Jul 15;263(20):9887–9895. [PubMed] [Google Scholar]