Abstract
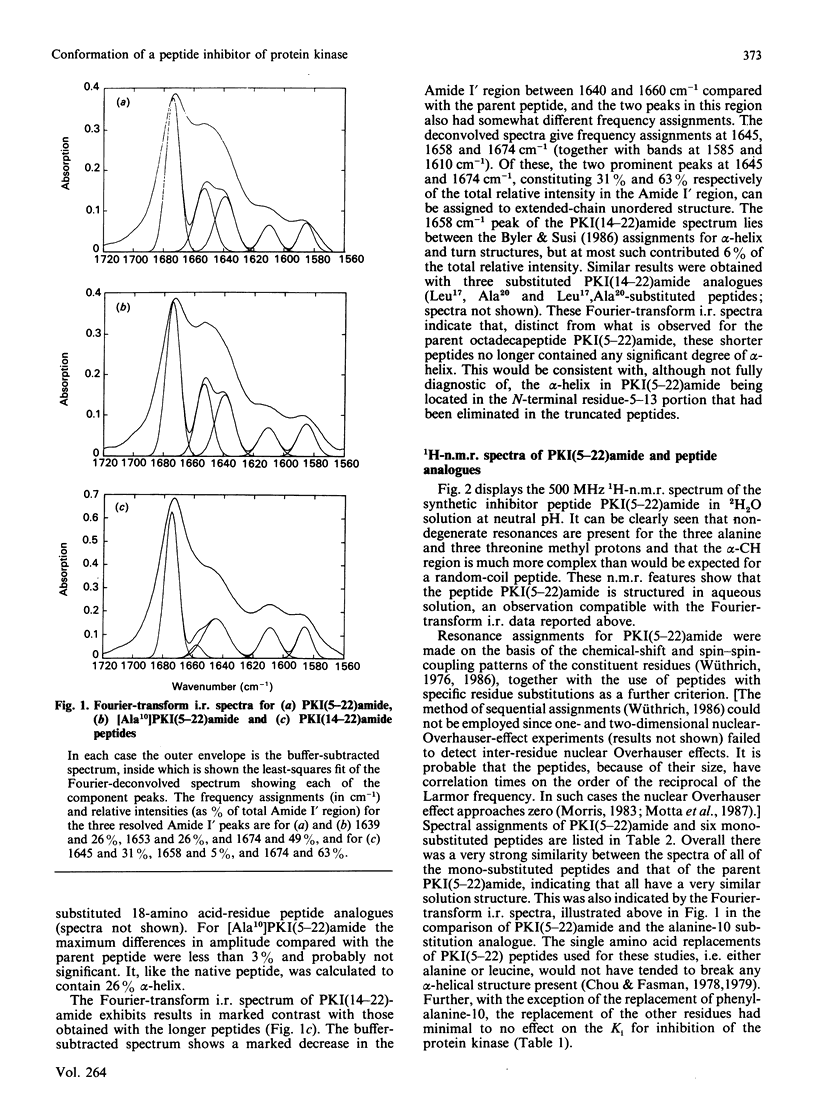
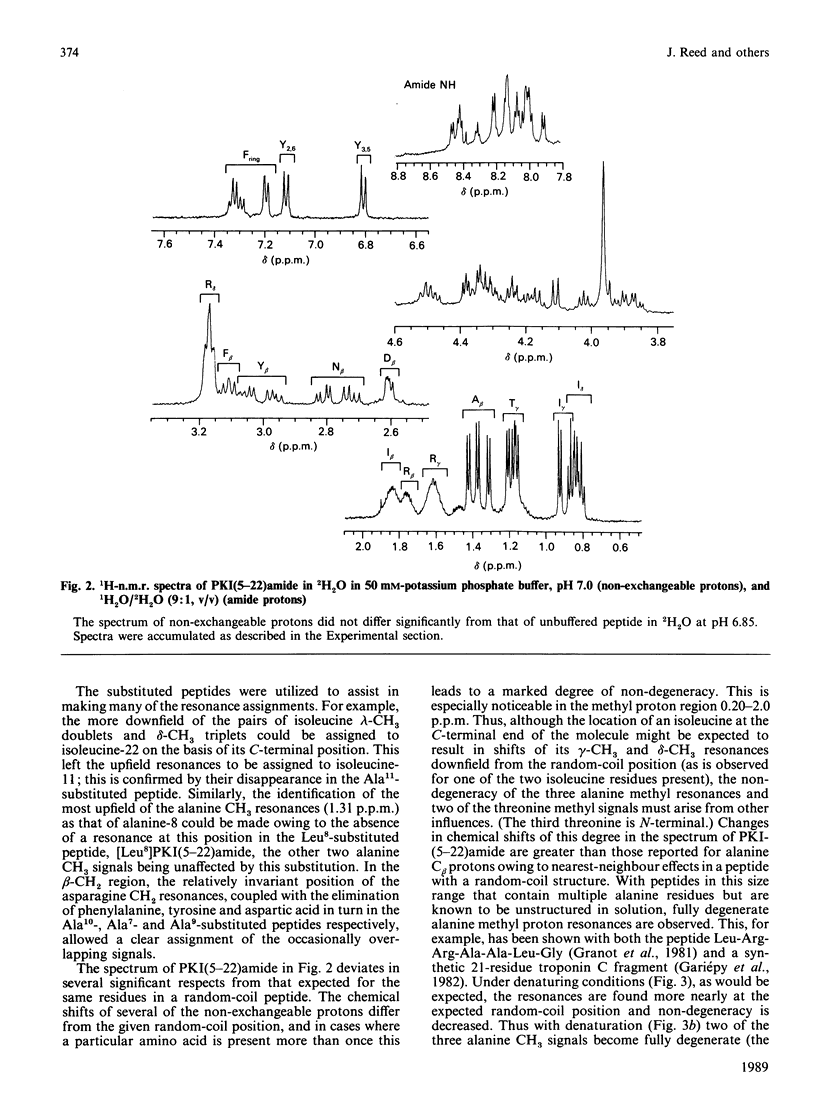
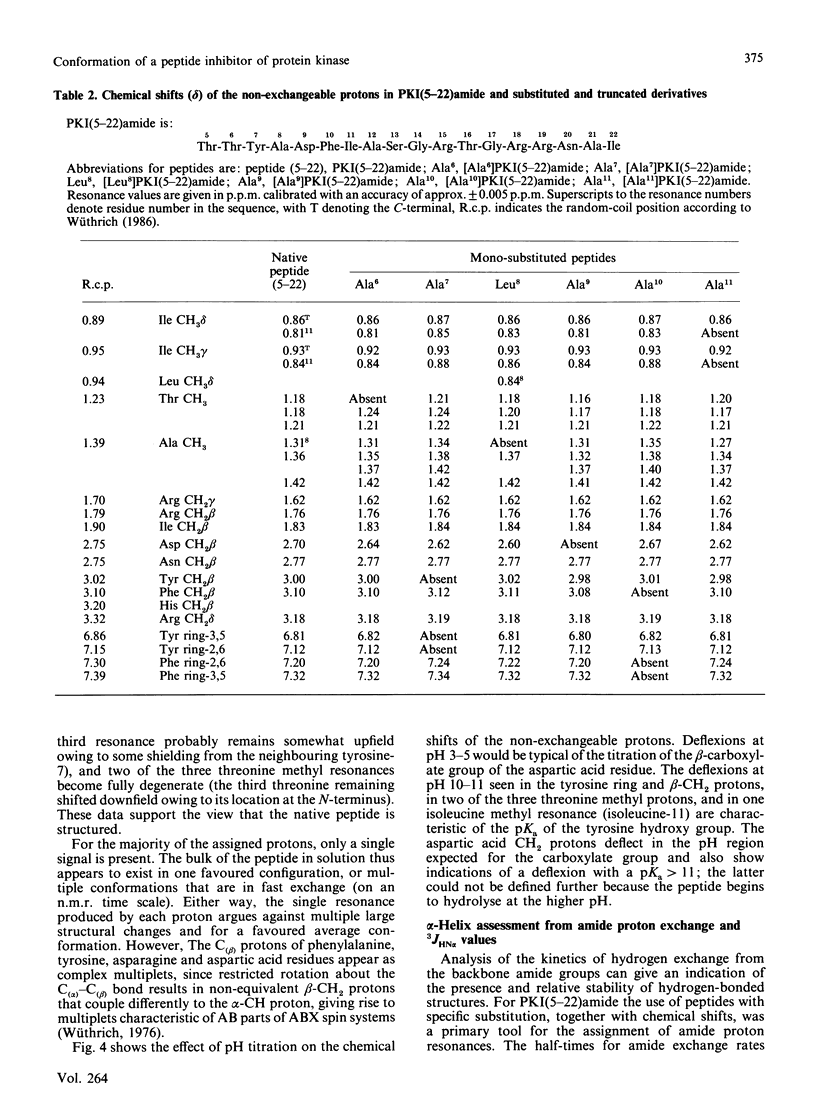
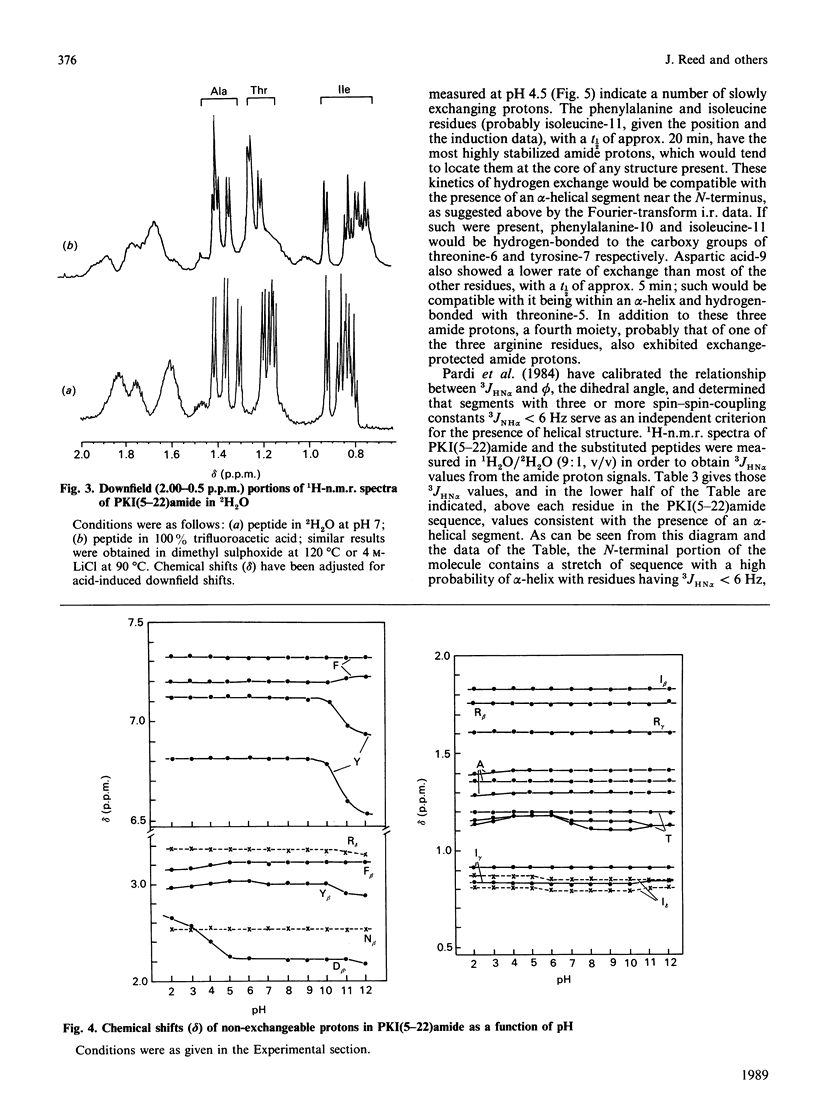
Fourier-transform i.r. spectroscopy, 1H-n.m.r. spectroscopy and X-ray scattering were used to study the conformation and shape of the peptide PKI(5-22)amide, which contains the active site of the inhibitor protein of the cyclic AMP-dependent protein kinase [Cheng, Van Pattern, Smith & Walsh (1985) Biochem. J. 231, 655-661]. The X-ray-scattering solution studies show that the peptide has a compact structure with Rg 0.9 nm (9.0 A) and a linear maximum dimension of 2.5 nm (25A). Compatible with this, Fourier-transform i.r. and n.m.r. determinations indicate that the peptide contains approx. 26% alpha-helix located in the N-terminal one-third of the molecule. This region contains the phenylalanine residue that is one essential recognition determinant for high-affinity binding to the protein kinase catalytic site.
Full text
PDF



Selected References
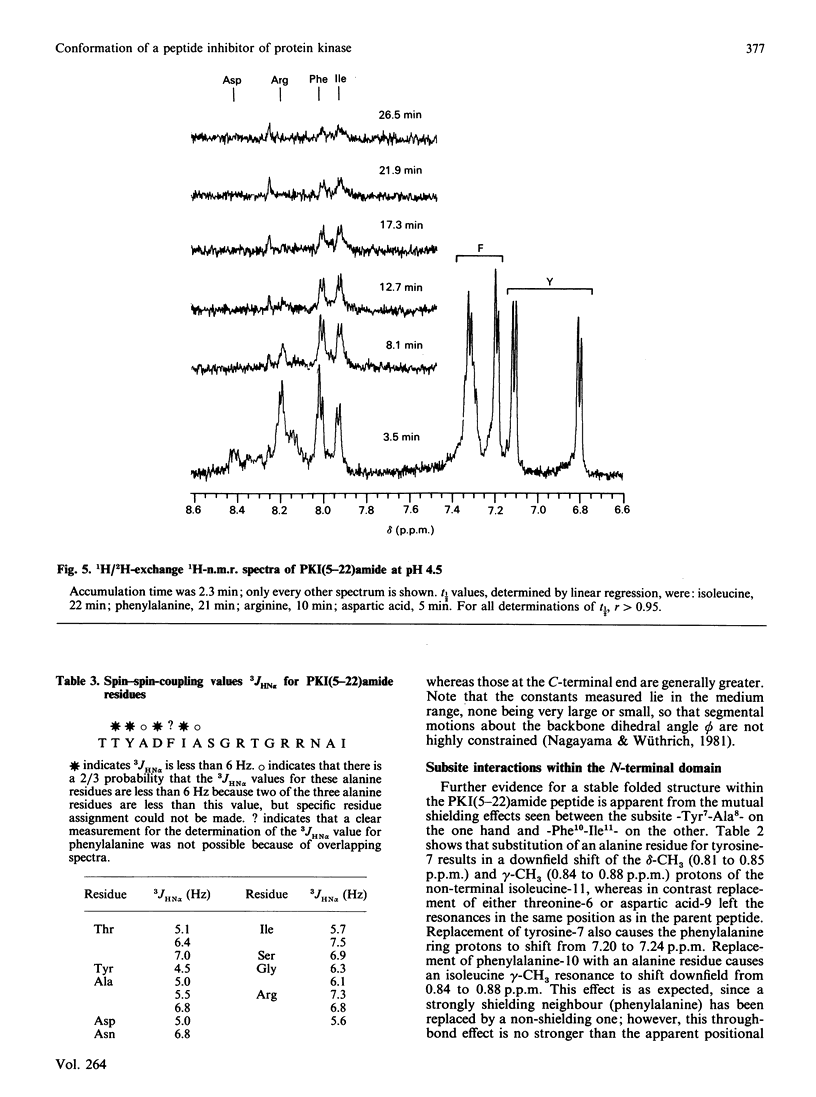
These references are in PubMed. This may not be the complete list of references from this article.
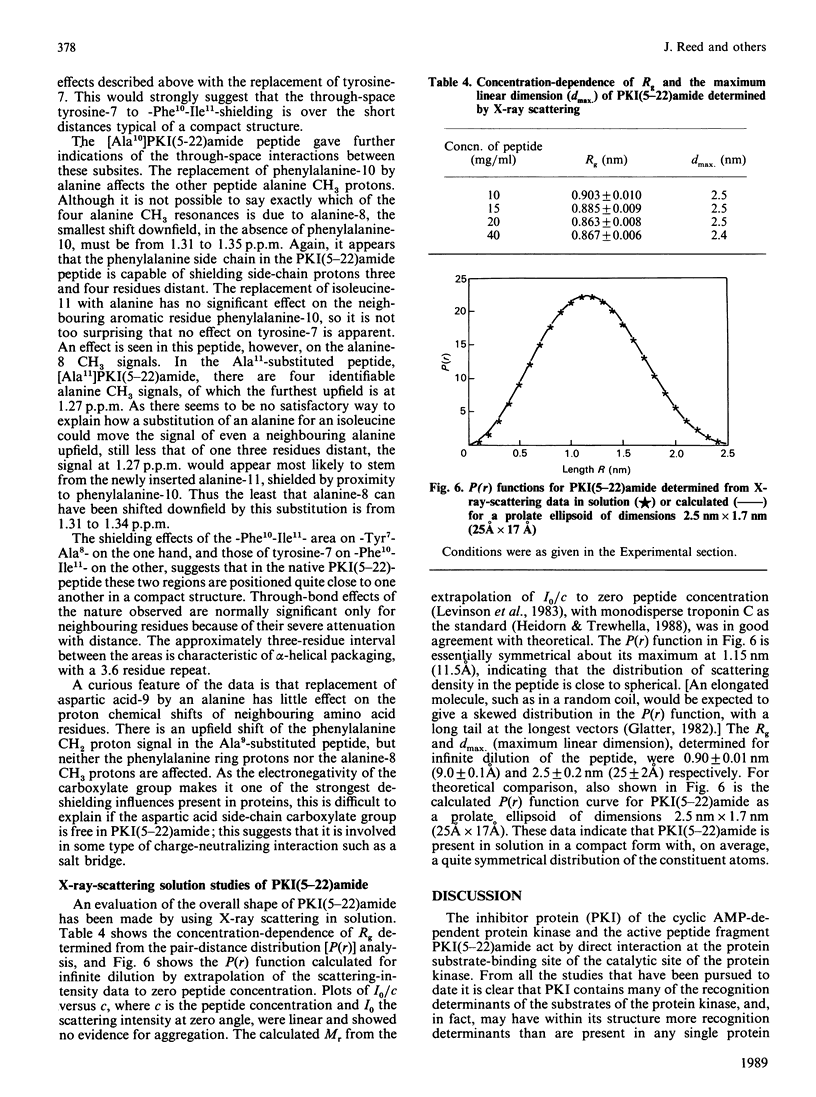
- Ashby C. D., Walsh D. A. Characterization of the interaction of a protein inhibitor with adenosine 3',5'-monophosphate-dependent protein kinases. I. Interaction with the catalytic subunit of the protein kinase. J Biol Chem. 1972 Oct 25;247(20):6637–6642. [PubMed] [Google Scholar]
- Byler D. M., Susi H. Examination of the secondary structure of proteins by deconvolved FTIR spectra. Biopolymers. 1986 Mar;25(3):469–487. doi: 10.1002/bip.360250307. [DOI] [PubMed] [Google Scholar]
- Cheng H. C., Kemp B. E., Pearson R. B., Smith A. J., Misconi L., Van Patten S. M., Walsh D. A. A potent synthetic peptide inhibitor of the cAMP-dependent protein kinase. J Biol Chem. 1986 Jan 25;261(3):989–992. [PubMed] [Google Scholar]
- Cheng H. C., van Patten S. M., Smith A. J., Walsh D. A. An active twenty-amino-acid-residue peptide derived from the inhibitor protein of the cyclic AMP-dependent protein kinase. Biochem J. 1985 Nov 1;231(3):655–661. doi: 10.1042/bj2310655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Empirical predictions of protein conformation. Annu Rev Biochem. 1978;47:251–276. doi: 10.1146/annurev.bi.47.070178.001343. [DOI] [PubMed] [Google Scholar]
- Chou P. Y., Fasman G. D. Prediction of beta-turns. Biophys J. 1979 Jun;26(3):367–383. doi: 10.1016/S0006-3495(79)85259-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demaille J. G., Peters K. A., Fischer E. H. Isolation and properties of the rabbit skeletal muscle protein inhibitor of adenosine 3',5'-monophosphate dependent protein kinases. Biochemistry. 1977 Jul 12;16(14):3080–3086. doi: 10.1021/bi00633a006. [DOI] [PubMed] [Google Scholar]
- Gariépy J., Sykes B. D., Reid R. E., Hodges R. S. Proton nuclear magnetic resonance investigation of synthetic calcium-binding peptides. Biochemistry. 1982 Mar 30;21(7):1506–1512. doi: 10.1021/bi00536a007. [DOI] [PubMed] [Google Scholar]
- Gill G. N., Holdy K. E., Walton G. M., Kanstein C. B. Purification and characterization of 3':5'-cyclic GMP-dependent protein kinase. Proc Natl Acad Sci U S A. 1976 Nov;73(11):3918–3922. doi: 10.1073/pnas.73.11.3918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glass D. B., Cheng H. C., Kemp B. E., Walsh D. A. Differential and common recognition of the catalytic sites of the cGMP-dependent and cAMP-dependent protein kinases by inhibitory peptides derived from the heat-stable inhibitor protein. J Biol Chem. 1986 Sep 15;261(26):12166–12171. [PubMed] [Google Scholar]
- Glass D. B., Cheng H. C., Mende-Mueller L., Reed J., Walsh D. A. Primary structural determinants essential for potent inhibition of cAMP-dependent protein kinase by inhibitory peptides corresponding to the active portion of the heat-stable inhibitor protein. J Biol Chem. 1989 May 25;264(15):8802–8810. [PubMed] [Google Scholar]
- Glass D. B., Lundquist L. J., Katz B. M., Walsh D. A. Protein kinase inhibitor-(6-22)-amide peptide analogs with standard and nonstandard amino acid substitutions for phenylalanine 10. Inhibition of cAMP-dependent protein kinase. J Biol Chem. 1989 Aug 25;264(24):14579–14584. [PubMed] [Google Scholar]
- Granot J., Mildvan A. S., Bramson H. N., Thomas N., Kaiser E. T. Nuclear magnetic resonance studies of the conformation and kinetics of the peptide-substrate at the active site of bovine heart protein kinase. Biochemistry. 1981 Feb 3;20(3):602–610. doi: 10.1021/bi00506a024. [DOI] [PubMed] [Google Scholar]
- Heidorn D. B., Trewhella J. Comparison of the crystal and solution structures of calmodulin and troponin C. Biochemistry. 1988 Feb 9;27(3):909–915. doi: 10.1021/bi00403a011. [DOI] [PubMed] [Google Scholar]
- Levinson B. L., Pickover C. A., Richards F. M. Dimerization by colicin E3 in the absence of immunity protein. J Biol Chem. 1983 Sep 25;258(18):10967–10972. [PubMed] [Google Scholar]
- Nagayama K., Wüthrich K. Structural interpretation of vicinal proton-proton coupling constants 3JH alpha H beta in the basic pancreatic trypsin inhibitor measured by two-dimensional J-resolved NMR spectroscopy. Eur J Biochem. 1981 Apr;115(3):653–657. doi: 10.1111/j.1432-1033.1981.tb06252.x. [DOI] [PubMed] [Google Scholar]
- Pardi A., Billeter M., Wüthrich K. Calibration of the angular dependence of the amide proton-C alpha proton coupling constants, 3JHN alpha, in a globular protein. Use of 3JHN alpha for identification of helical secondary structure. J Mol Biol. 1984 Dec 15;180(3):741–751. doi: 10.1016/0022-2836(84)90035-4. [DOI] [PubMed] [Google Scholar]
- Reed J., Kinzel V., Cheng H. C., Walsh D. A. Circular dichroic investigations of secondary structure in synthetic peptide inhibitors of cAMP-dependent protein kinase: a model for inhibitory potential. Biochemistry. 1987 Dec 1;26(24):7641–7647. doi: 10.1021/bi00398a017. [DOI] [PubMed] [Google Scholar]
- Scott J. D., Fischer E. H., Takio K., Demaille J. G., Krebs E. G. Amino acid sequence of the heat-stable inhibitor of the cAMP-dependent protein kinase from rabbit skeletal muscle. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5732–5736. doi: 10.1073/pnas.82.17.5732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott J. D., Glaccum M. B., Fischer E. H., Krebs E. G. Primary-structure requirements for inhibition by the heat-stable inhibitor of the cAMP-dependent protein kinase. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1613–1616. doi: 10.1073/pnas.83.6.1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trewhella J., Liddle W. K., Heidorn D. B., Strynadka N. Calmodulin and troponin C structures studied by Fourier transform infrared spectroscopy: effects of Ca2+ and Mg2+ binding. Biochemistry. 1989 Feb 7;28(3):1294–1301. doi: 10.1021/bi00429a052. [DOI] [PubMed] [Google Scholar]
- Walsh D. A., Ashby C. D., Gonzalez C., Calkins D., Fischer E. H. Krebs EG: Purification and characterization of a protein inhibitor of adenosine 3',5'-monophosphate-dependent protein kinases. J Biol Chem. 1971 Apr 10;246(7):1977–1985. [PubMed] [Google Scholar]


