Abstract
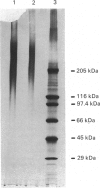
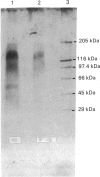
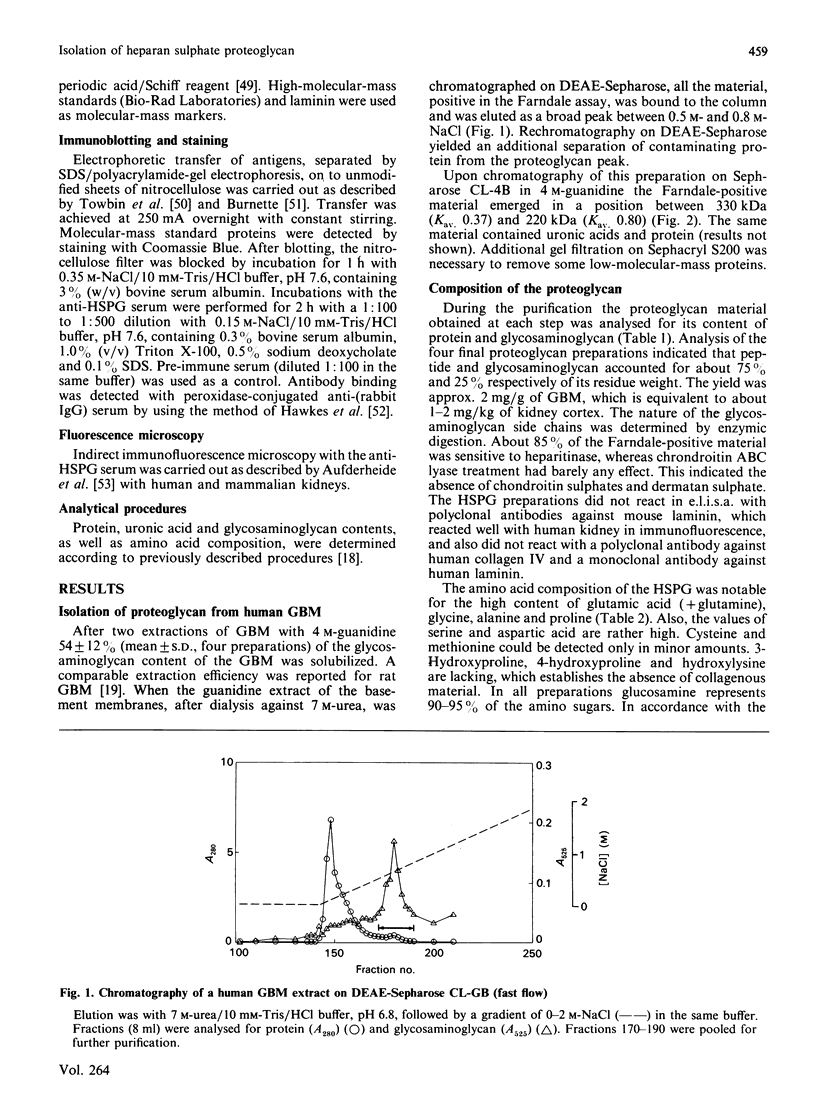
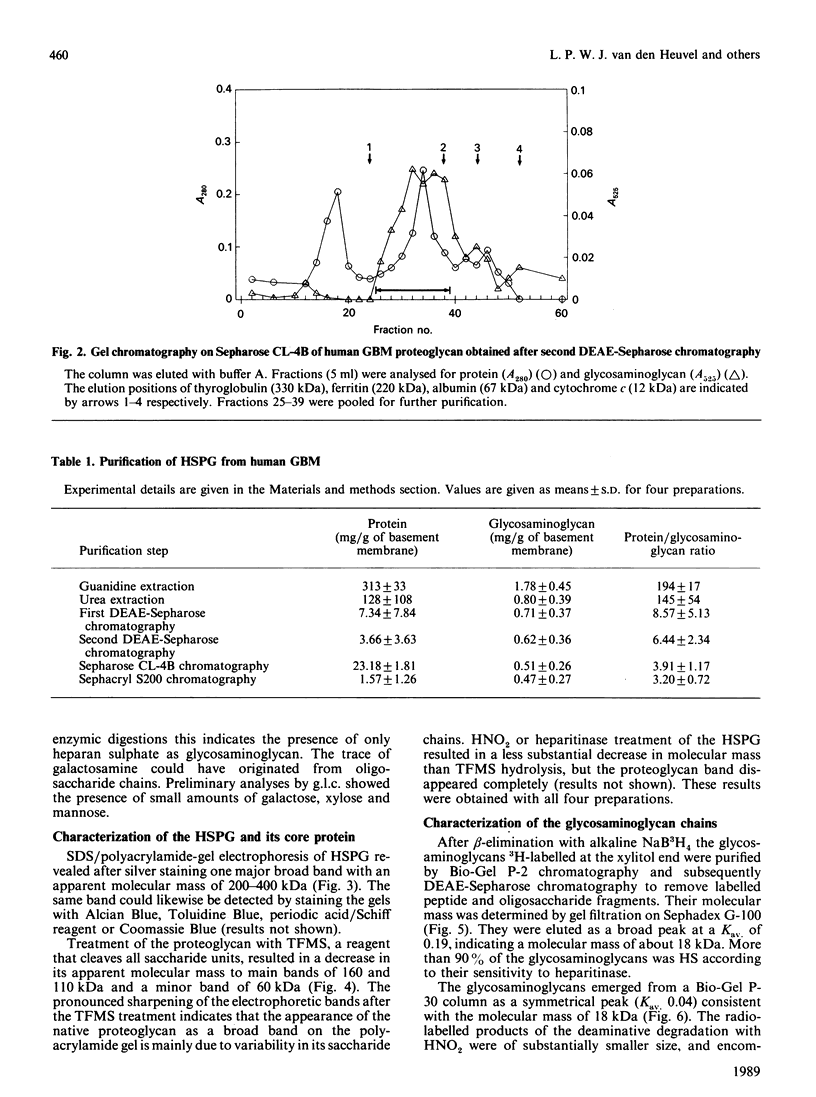
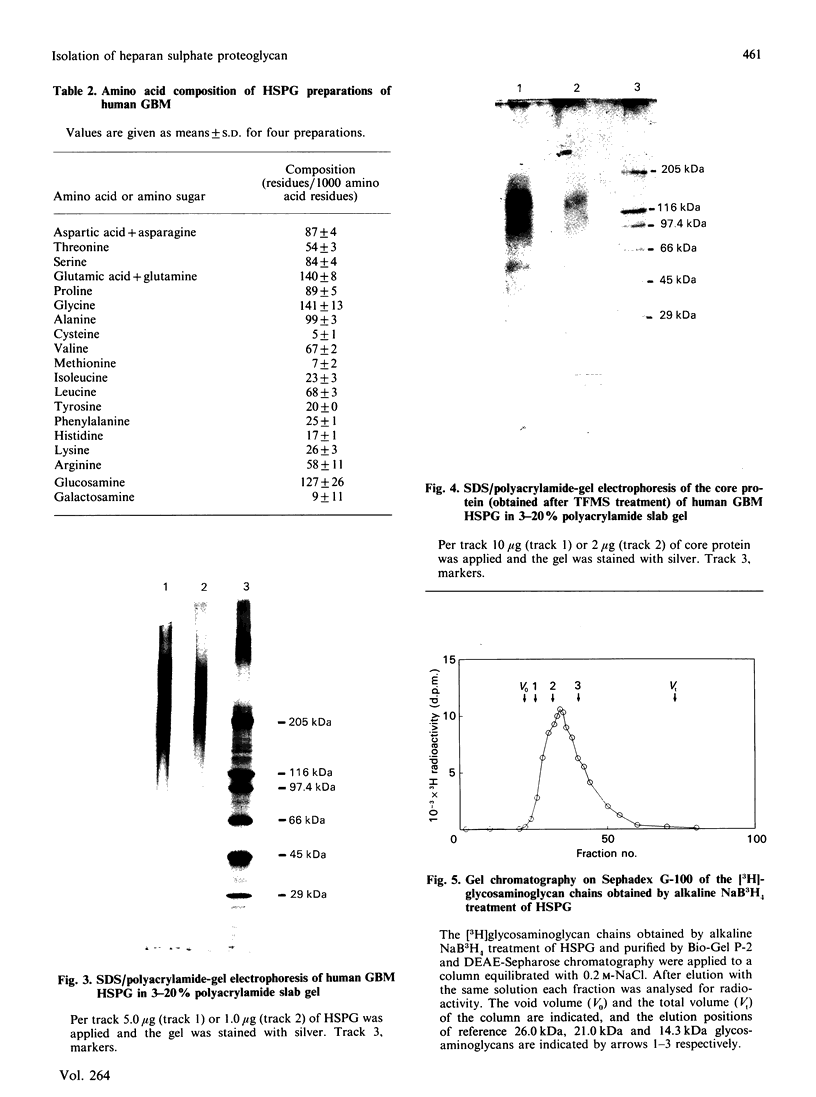
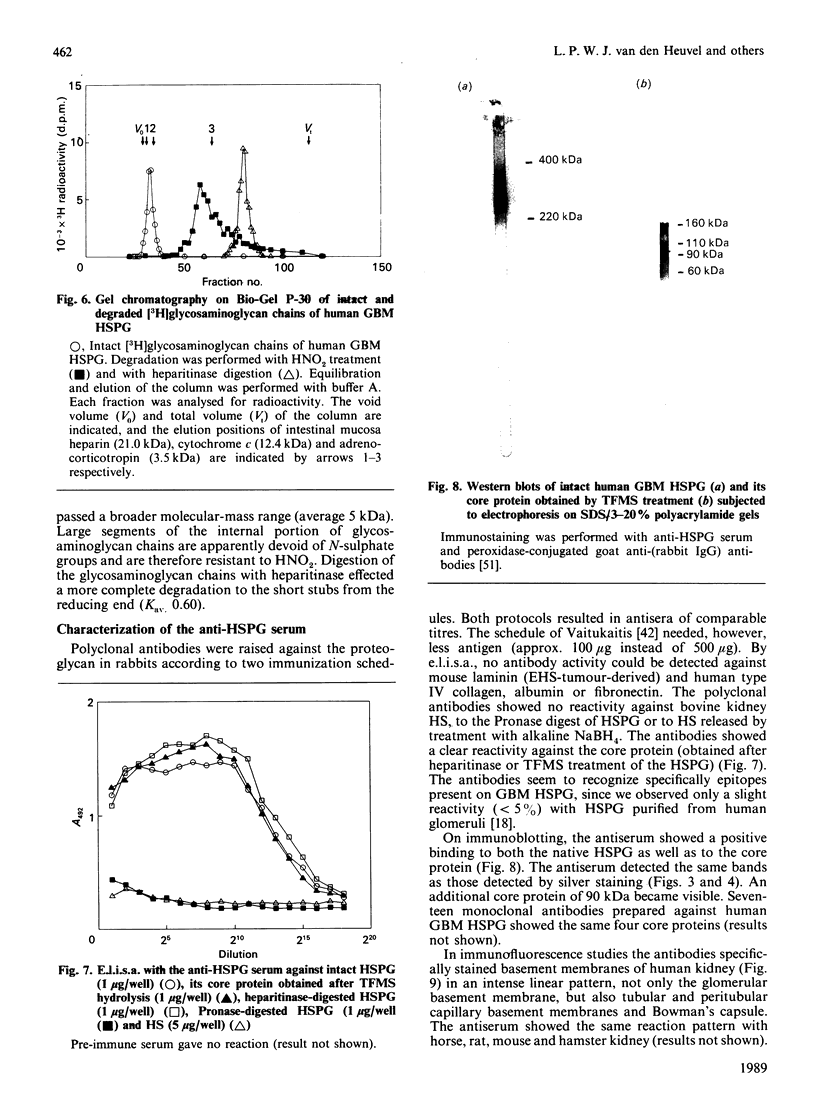
Heparan sulphate proteoglycan was solubilized from human glomerular basement membranes by guanidine extraction and purified by ion-exchange chromatography and gel filtration. The yield of proteoglycan was approx. 2 mg/g of basement membrane. The glycoconjugate had an apparent molecular mass of 200-400 kDa and consisted of about 75% protein and 25% heparan sulphate. The amino acid composition was characterized by a high content of glycine, proline, alanine and glutamic acid. Hydrolysis with trifluoromethanesulphonic acid yielded core proteins of 160 and 110 kDa (and minor bands of 90 and 60 kDa). Alkaline NaBH4 treatment of the proteoglycan released heparan sulphate chains with an average molecular mass of 18 kDa. HNO2 oxidation of these chains yielded oligosaccharides of about 5 kDa, whereas heparitinase digestion resulted in a more complete degradation. The data suggest a clustering of N-sulphate groups in the peripheral regions of the glycosaminoglycan chains. A polyclonal antiserum raised against the intact proteoglycan showed reactivity against the core protein. It stained all basement membranes in an intense linear fashion in immunohistochemical studies on frozen kidney sections from man and various mammalian species.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aufderheide E., Chiquet-Ehrismann R., Ekblom P. Epithelial-mesenchymal interactions in the developing kidney lead to expression of tenascin in the mesenchyme. J Cell Biol. 1987 Jul;105(1):599–608. doi: 10.1083/jcb.105.1.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Edge A. S., Faltynek C. R., Hof L., Reichert L. E., Jr, Weber P. Deglycosylation of glycoproteins by trifluoromethanesulfonic acid. Anal Biochem. 1981 Nov 15;118(1):131–137. doi: 10.1016/0003-2697(81)90168-8. [DOI] [PubMed] [Google Scholar]
- Edge A. S., Spiro R. G. Selective deglycosylation of the heparan sulfate proteoglycan of bovine glomerular basement membrane and identification of the core protein. J Biol Chem. 1987 May 15;262(14):6893–6898. [PubMed] [Google Scholar]
- Faaber P., Capel P. J., Rijke G. P., Vierwinden G., van de Putte L. B., Koene R. A. Cross-reactivity of anti-DNA antibodies with proteoglycans. Clin Exp Immunol. 1984 Mar;55(3):502–508. [PMC free article] [PubMed] [Google Scholar]
- Farndale R. W., Sayers C. A., Barrett A. J. A direct spectrophotometric microassay for sulfated glycosaminoglycans in cartilage cultures. Connect Tissue Res. 1982;9(4):247–248. doi: 10.3109/03008208209160269. [DOI] [PubMed] [Google Scholar]
- Fujiwara S., Wiedemann H., Timpl R., Lustig A., Engel J. Structure and interactions of heparan sulfate proteoglycans from a mouse tumor basement membrane. Eur J Biochem. 1984 Aug 15;143(1):145–157. doi: 10.1111/j.1432-1033.1984.tb08353.x. [DOI] [PubMed] [Google Scholar]
- Gallagher J. T., Lyon M., Steward W. P. Structure and function of heparan sulphate proteoglycans. Biochem J. 1986 Jun 1;236(2):313–325. doi: 10.1042/bj2360313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassell J. R., Leyshon W. C., Ledbetter S. R., Tyree B., Suzuki S., Kato M., Kimata K., Kleinman H. K. Isolation of two forms of basement membrane proteoglycans. J Biol Chem. 1985 Jul 5;260(13):8098–8105. [PubMed] [Google Scholar]
- Hawkes R., Niday E., Gordon J. A dot-immunobinding assay for monoclonal and other antibodies. Anal Biochem. 1982 Jan 1;119(1):142–147. doi: 10.1016/0003-2697(82)90677-7. [DOI] [PubMed] [Google Scholar]
- Hovingh P., Piepkorn M., Linker A. Biological implications of the structural, antithrombin affinity and anticoagulant activity relationships among vertebrate heparins and heparan sulphates. Biochem J. 1986 Jul 15;237(2):573–581. doi: 10.1042/bj2370573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hök M., Kjellén L., Johansson S. Cell-surface glycosaminoglycans. Annu Rev Biochem. 1984;53:847–869. doi: 10.1146/annurev.bi.53.070184.004215. [DOI] [PubMed] [Google Scholar]
- Jalkanen M., Nguyen H., Rapraeger A., Kurn N., Bernfield M. Heparan sulfate proteoglycans from mouse mammary epithelial cells: localization on the cell surface with a monoclonal antibody. J Cell Biol. 1985 Sep;101(3):976–984. doi: 10.1083/jcb.101.3.976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jalkanen M., Rapraeger A., Bernfield M. Mouse mammary epithelial cells produce basement membrane and cell surface heparan sulfate proteoglycans containing distinct core proteins. J Cell Biol. 1988 Mar;106(3):953–962. doi: 10.1083/jcb.106.3.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanwar Y. S., Hascall V. C., Farquhar M. G. Partial characterization of newly synthesized proteoglycans isolated from the glomerular basement membrane. J Cell Biol. 1981 Aug;90(2):527–532. doi: 10.1083/jcb.90.2.527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanwar Y. S., Linker A., Farquhar M. G. Increased permeability of the glomerular basement membrane to ferritin after removal of glycosaminoglycans (heparan sulfate) by enzyme digestion. J Cell Biol. 1980 Aug;86(2):688–693. doi: 10.1083/jcb.86.2.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanwar Y. S., Veis A., Kimura J. H., Jakubowski M. L. Characterization of heparan sulfate-proteoglycan of glomerular basement membranes. Proc Natl Acad Sci U S A. 1984 Feb;81(3):762–766. doi: 10.1073/pnas.81.3.762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato M., Koike Y., Suzuki S., Kimata K. Basement membrane proteoglycan in various tissues: characterization using monoclonal antibodies to the Engelbreth-Holm-Swarm mouse tumor low density heparan sulfate proteoglycan. J Cell Biol. 1988 Jun;106(6):2203–2210. doi: 10.1083/jcb.106.6.2203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein D. J., Brown D. M., Oegema T. R., Brenchley P. E., Anderson J. C., Dickinson M. A., Horigan E. A., Hassell J. R. Glomerular basement membrane proteoglycans are derived from a large precursor. J Cell Biol. 1988 Mar;106(3):963–970. doi: 10.1083/jcb.106.3.963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein D. J., Brown D. M., Oegema T. R., Jr Partial characterization of heparan and dermatan sulfate proteoglycans synthesized by normal rat glomeruli. J Biol Chem. 1986 Dec 15;261(35):16636–16652. [PubMed] [Google Scholar]
- Kobayashi S., Oguri K., Kobayashi K., Okayama M. Isolation and characterization of proteoheparan sulfate synthesized in vitro by rat glomeruli. J Biol Chem. 1983 Oct 10;258(19):12051–12057. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Laterra J., Silbert J. E., Culp L. A. Cell surface heparan sulfate mediates some adhesive responses to glycosaminoglycan-binding matrices, including fibronectin. J Cell Biol. 1983 Jan;96(1):112–123. doi: 10.1083/jcb.96.1.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl U., Hök M. Glycosaminoglycans and their binding to biological macromolecules. Annu Rev Biochem. 1978;47:385–417. doi: 10.1146/annurev.bi.47.070178.002125. [DOI] [PubMed] [Google Scholar]
- Louvard D., Reggio H., Warren G. Antibodies to the Golgi complex and the rough endoplasmic reticulum. J Cell Biol. 1982 Jan;92(1):92–107. doi: 10.1083/jcb.92.1.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyon M., Steward W. P., Hampson I. N., Gallagher J. T. Identification of an extended N-acetylated sequence adjacent to the protein-linkage region of fibroblast heparan sulphate. Biochem J. 1987 Mar 1;242(2):493–498. doi: 10.1042/bj2420493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Hernandez A., Miller E. J., Damjanov I., Gay S. Laminin-secreting yolk sac carcinoma of the rat. Biochemical and electron immunohistochemical studies. Lab Invest. 1982 Sep;47(3):247–257. [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- Oohira A., Wight T. N., McPherson J., Bornstein P. Biochemical and ultrastructural studies of proteoheparan sulfates synthesized by PYS-2, a basement membrane-producing cell line. J Cell Biol. 1982 Feb;92(2):357–367. doi: 10.1083/jcb.92.2.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parthasarathy N., Spiro R. G. Isolation and characterization of the heparan sulfate proteoglycan of the bovine glomerular basement membrane. J Biol Chem. 1984 Oct 25;259(20):12749–12755. [PubMed] [Google Scholar]
- Parthasarathy N., Tanzer M. L. Isolation and characterization of a low molecular weight chondroitin sulfate proteoglycan from rabbit skeletal muscle. Biochemistry. 1987 Jun 2;26(11):3149–3156. doi: 10.1021/bi00385a031. [DOI] [PubMed] [Google Scholar]
- Paulsson M., Dziadek M., Suchanek C., Huttner W. B., Timpl R. Nature of sulphated macromolecules in mouse Reichert's membrane. Evidence for tyrosine O-sulphate in basement-membrane proteins. Biochem J. 1985 Nov 1;231(3):571–579. doi: 10.1042/bj2310571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rennard S. I., Berg R., Martin G. R., Foidart J. M., Robey P. G. Enzyme-linked immunoassay (ELISA) for connective tissue components. Anal Biochem. 1980 May 1;104(1):205–214. doi: 10.1016/0003-2697(80)90300-0. [DOI] [PubMed] [Google Scholar]
- Reubsaet F. A., Langeveld J. P., Veerkamp J. H. Glycosaminoglycan content of glomerular and tubular basement membranes of various mammalian species. Biochim Biophys Acta. 1985 Jan 28;838(1):144–150. doi: 10.1016/0304-4165(85)90260-0. [DOI] [PubMed] [Google Scholar]
- Reubsaet F. A., Veerkamp J. H., Monnens L. A. Sulfated glycosaminoglycan content of glomerular and tubular basement membranes of individuals of different ages. Nephron. 1985;41(4):344–347. doi: 10.1159/000183612. [DOI] [PubMed] [Google Scholar]
- Seno N., Anno K., Kondo K., Nagase S., Saito S. Improved method for electrophoretic separation and rapid quantitation of isomeric chondroitin sulfates on cellulose acetate strips. Anal Biochem. 1970 Sep;37(1):197–202. doi: 10.1016/0003-2697(70)90280-0. [DOI] [PubMed] [Google Scholar]
- Shimomura H., Spiro R. G. Studies on macromolecular components of human glomerular basement membrane and alterations in diabetes. Decreased levels of heparan sulfate proteoglycan and laminin. Diabetes. 1987 Mar;36(3):374–381. doi: 10.2337/diab.36.3.374. [DOI] [PubMed] [Google Scholar]
- Stow J. L., Glasgow E. F., Handley C. J., Hascall V. C. Biosynthesis of proteoglycans by isolated rabbit glomeruli. Arch Biochem Biophys. 1983 Sep;225(2):950–957. doi: 10.1016/0003-9861(83)90110-8. [DOI] [PubMed] [Google Scholar]
- Stow J. L., Kjéllen L., Unger E., Hök M., Farquhar M. G. Heparan sulfate proteoglycans are concentrated on the sinusoidal plasmalemmal domain and in intracellular organelles of hepatocytes. J Cell Biol. 1985 Mar;100(3):975–980. doi: 10.1083/jcb.100.3.975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stow J. L., Sawada H., Farquhar M. G. Basement membrane heparan sulfate proteoglycans are concentrated in the laminae rarae and in podocytes of the rat renal glomerulus. Proc Natl Acad Sci U S A. 1985 May;82(10):3296–3300. doi: 10.1073/pnas.82.10.3296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Templeton D. M., Khatchatourian M. Synthesis of heparan sulfate proteoglycans by the isolated glomerulus. Biochem Cell Biol. 1988 Oct;66(10):1078–1085. doi: 10.1139/o88-124. [DOI] [PubMed] [Google Scholar]
- Timpl R., Dziadek M. Structure, development, and molecular pathology of basement membranes. Int Rev Exp Pathol. 1986;29:1–112. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaitukaitis J. L. Production of antisera with small doses of immunogen: multiple intradermal injections. Methods Enzymol. 1981;73(Pt B):46–52. doi: 10.1016/0076-6879(81)73055-6. [DOI] [PubMed] [Google Scholar]
- Wasteson A. A method for the determination of the molecular weight and molecular-weight distribution of chondroitin sulphate. J Chromatogr. 1971 Jul 8;59(1):87–97. doi: 10.1016/s0021-9673(01)80009-1. [DOI] [PubMed] [Google Scholar]
- Woods A., Couchman J. R., Hök M. Heparan sulfate proteoglycans of rat embryo fibroblasts. A hydrophobic form may link cytoskeleton and matrix components. J Biol Chem. 1985 Sep 5;260(19):10872–10879. [PubMed] [Google Scholar]
- van den Heuvel L. P., Veerkamp J. H., Monnens L. A., Schröder C. H. Heparan sulfate proteoglycan from human and equine glomeruli and tubules. Int J Biochem. 1988;20(12):1391–1400. doi: 10.1016/s0020-711x(98)90007-6. [DOI] [PubMed] [Google Scholar]