Abstract
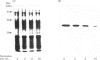
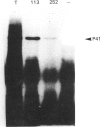
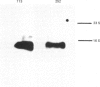
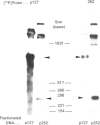
Putative protochlorophyllide reductase cDNA clones (252 and 113) were isolated from an etiolated-oat (Avena sativa) cDNA library. These were used to indirectly characterize a further clone, p127, isolated from a lambda-phage gt11 cDNA library. The latter (1.15 kb in length) was sequenced, and the derived amino acid sequence was shown to be remarkably similar to that derived from chemical analysis of a CNBr-cleavage fragment of the purified reductase, p127 codes for more than 95% of the reductase protein.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apel K., Gollmer I., Batschauer A. The light-dependent control of chloroplast development in barley (Hordeum vulgare L). J Cell Biochem. 1983;23(1-4):181–189. doi: 10.1002/jcb.240230115. [DOI] [PubMed] [Google Scholar]
- Apel K. The protochlorophyllide holochrome of barley (Hordeum vulgare L.). Phytochrome-induced decrease of translatable mRNA coding for the NADPH: protochlorophyllide oxidoreductase. Eur J Biochem. 1981 Nov;120(1):89–93. doi: 10.1111/j.1432-1033.1981.tb05673.x. [DOI] [PubMed] [Google Scholar]
- Batschauer A., Mösinger E., Kreuz K., Dörr I., Apel K. The implication of a plastid-derived factor in the transcriptional control of nuclear genes encoding the light-harvesting chlorophyll a/b protein. Eur J Biochem. 1986 Feb 3;154(3):625–634. doi: 10.1111/j.1432-1033.1986.tb09444.x. [DOI] [PubMed] [Google Scholar]
- Beer N. S., Griffiths W. T. Purification of the enzyme NADPH: protochlorophyllide oxidoreductase. Biochem J. 1981 Apr 1;195(1):83–92. doi: 10.1042/bj1950083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellemare G., Bartlett S. G., Chua N. H. Biosynthesis of chlorophyll a/b-binding polypeptides in wild type and the chlorina f2 mutant of barley. J Biol Chem. 1982 Jul 10;257(13):7762–7767. [PubMed] [Google Scholar]
- Broglie R., Coruzzi G., Fraley R. T., Rogers S. G., Horsch R. B., Niedermeyer J. G., Fink C. L., Chua N. H. Light-regulated expression of a pea ribulose-1,5-bisphosphate carboxylase small subunit gene in transformed plant cells. Science. 1984 May 25;224(4651):838–843. doi: 10.1126/science.6719112. [DOI] [PubMed] [Google Scholar]
- Cashmore A. R. Reiteration frequency of the gene coding for the small subunit of ribulose--1,5--bisphosphate carboxylase. Cell. 1979 Jun;17(2):383–388. doi: 10.1016/0092-8674(79)90164-8. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Collins P. D., Hague D. R. Light-stimulated synthesis of NADP malic enzyme in leaves of maize. J Biol Chem. 1983 Mar 25;258(6):4012–4018. [PubMed] [Google Scholar]
- Cuming A. C., Bennett J. Biosynthesis of the light-harvesting chlorophyll a/b protein. Control of messenger RNA activity by light. Eur J Biochem. 1981 Aug;118(1):71–80. doi: 10.1111/j.1432-1033.1981.tb05487.x. [DOI] [PubMed] [Google Scholar]
- Deng G., Wu R. An improved procedure for utilizing terminal transferase to add homopolymers to the 3' termini of DNA. Nucleic Acids Res. 1981 Aug 25;9(16):4173–4188. doi: 10.1093/nar/9.16.4173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths W. T. Reconstitution of chlorophyllide formation by isolated etioplast membranes. Biochem J. 1978 Sep 15;174(3):681–692. doi: 10.1042/bj1740681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall T. C., Ma Y., Buchbinder B. U., Pyne J. W., Sun S. M., Bliss F. A. Messenger RNA for G1 protein of French bean seeds: Cell-free translation and product characterization. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3196–3200. doi: 10.1073/pnas.75.7.3196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Oliver R. P., Griffiths W. T. Covalent labelling of the NADPH: protochlorophyllide oxidoreductase from etioplast membranes with [3H]N-phenylmaleimide. Biochem J. 1981 Apr 1;195(1):93–101. doi: 10.1042/bj1950093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham H. R., Jackson R. J. An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem. 1976 Aug 1;67(1):247–256. doi: 10.1111/j.1432-1033.1976.tb10656.x. [DOI] [PubMed] [Google Scholar]
- Roman R., Brooker J. D., Seal S. N., Marcus A. Inhibition of the transition of a 40 S ribosome-Met-tRNA-i-Met complex to an 80 S ribosome-Met-tRNA-i-Met- complex by 7-Methylguanosine-5'-phosphate. Nature. 1976 Mar 25;260(5549):359–360. doi: 10.1038/260359a0. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sims T. L., Hague D. R. Light-stimulated increase of translatable mRNA for phosphoenolpyruvate carboxylase in leaves of maize. J Biol Chem. 1981 Aug 25;256(16):8252–8255. [PubMed] [Google Scholar]
- Southard J. H., Van der Laan N. C., Lutz M., Pavlock G. S., Belzer J. P., Belzer F. O. Comparison of the effect of temperature on kidney cortex mitochondria from rabbit, dog, pig, and human: Arrhenius plots of ADP-stimulated respiration. Cryobiology. 1983 Aug;20(4):395–400. doi: 10.1016/0011-2240(83)90029-9. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Young R. A., Davis R. W. Efficient isolation of genes by using antibody probes. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1194–1198. doi: 10.1073/pnas.80.5.1194. [DOI] [PMC free article] [PubMed] [Google Scholar]