Abstract
Background
Microplastics, widely present in the environment, are implicated in disease pathogenesis through oxidative stress and immune modulation. Prevailing research, primarily based on animal and cell studies, falls short in elucidating microplastics' impact on human cardiovascular health. This cross-sectional study detected blood microplastic concentrations in patients presenting with chest pain using pyrolysis–gas chromatography/mass spectrometry and evaluating inflammatory and immune markers through flow cytometry, to explore the potential effects of microplastic on acute coronary syndrome.
Results
The study included 101 participants, comprising 19 controls and 82 acute coronary syndrome cases. Notably, acute coronary syndrome patients exhibited elevated microplastic concentrations, with those suffering from acute myocardial infarction presenting higher loads compared to those with unstable angina. Furthermore, patients at intermediate to high risk of coronary artery disease displayed significantly higher microplastic accumulations than their low-risk counterparts. A significant relationship was observed between increased microplastic levels and enhanced IL-6 and IL-12p70 contents, alongside elevated B lymphocyte and natural killer cell counts.
Conclusion
These results suggest an association between microplastics and both vascular pathology complexity and immunoinflammatory response in acute coronary syndrome, underscoring the critical need for targeted research to delineate the mechanisms of this association.
Highlights
Blood microplastic levels escalate from angiographic patency, to angina patients, peaking in myocardial infarction patients.
Microplastics in acute coronary syndrome patients are predominantly PE, followed by PVC, PS, and PP.
Microplastics may induce immune cell-associated inflammatory responses in acute coronary syndrome patients.
Graphical abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s12989-024-00596-4.
Keywords: Microplastic, Acute coronary syndrome, Coronary angiography, Pyrolysis–gas chromatography/mass spectrometry, Flow cytometry
Background
Microplastic (MP), defined as plastic fragments with a diameter under 5 mm, primarily arises from the direct release of granulated products or the breakdown of larger plastics 1, 2. As a global emerging pollutant, MPs persist in the environment owing to the slow degradation rate, exhibiting a range of harmful effects including reproductive toxicity, inflammation, carcinogenicity, and mutagenicity 3. While studies on the human health impacts of MPs remains nascent, evidence has revealed the presence of MPs in various organs [4–6], especially within cardiac and arterial tissues as highlighted by our recent findings 7, 8.
MP durability presents challenges to complete clearance by the immune system. Initial evidence points to potential local and systemic damage from MPs due to mechanical injury and toxic additive release 9. Both mechanical endothelial damage and heightened inflammatory responses underscore the potential significance of MPs as an emergent cardiovascular risk factor 10, 11. Given their crucial role in the circulatory system, vascular tissues seem particularly vulnerable to MP exposure. Recent findings associating the presence of MPs with acute arterial thrombosis support this hypothesis 12. Nonetheless, the relationship between MPs and vascular diseases remains under-researched.
Acute coronary syndrome (ACS) is characterized by sudden diminution in cardiac blood supply, leading to symptoms such as chest pain, alterations in 12-lead electrocardiogram, and elevated markers of myocardial injury 13. Globally, ACS diagnoses surpass 7 million annually, with a mortality rate nearing 5% prior to discharge 14. Although causes such as atherosclerotic plaque rupture, coronary spasms, and coronary dissections are identified, comprehensive preventive strategies remain elusive, indicating gaps in our comprehension of ACS etiologies. A recent study has highlighted an association between concentrations of nano- and microplastic (NMP) and the presence of carotid atherosclerotic plaques, as well as an increased incidence of adverse events, including myocardial infarction, stroke, and death from any cause 15. However, the correlation between MP concentration and the pathophysiology of ACS has yet to be elucidated.
In this study, utilizing invasive coronary angiography, pyrolysis gas chromatography mass spectrometry (Py-GC/MS), and flow cytometry, we aim to ascertain MP concentrations in ACS patients, explore associations with lymphocytes and inflammatory cytokines, and investigate the cardiovascular repercussions of MP concentration, in order to further elucidate ACS pathogenesis.
Methods
Ethics statement
This study was carried out in accordance with relevant guidelines and regulations with ethical approval obtained from the Ethics Committee of Beijing Anzhen Hospital (Approval No. KS2023049) and was conducted following the Declaration of Helsinki.
Participants
Patients who presented to Beijing Anzhen Hospital with a complaint of chest pain between July 2023 and September 2023 were evaluated for inclusion in the study. All patients received coronary angiogram. The complexity of coronary artery was evaluated by the SYNTAX [Synergy Between percutaneous coronary intervention (PCI) With Taxus and coronary artery bypass surgery] score, which was calculated using SYNTAX Score calculator (https://syntaxscore.org/calculator/start.htm) after the coronary angiogram procedure. A SYNTAX Score of 0 indicates that no significant stenosis or blockage were observed in the major coronary arteries 16, 17.Exclusion criteria: a history of previous coronary intervention or myocardial infarction (MI) within 6 month; received treatments for chest pain at other hospitals prior to their visit to Beijing Anzhen Hospital; declined participation; other conditions such as aortic dissection and pulmonary embolism.
Quality control
The quality control system was designed before the experiment. (1) contamination control: a vacuum pump filtered all chemical solutions through a 0.1 μm pore size polytetrafluoroethylene membrane filter. During the sample collection process, medical personnel wore cotton coats, polypropylene (PP) surgical masks, and latex gloves for collecting blood. During the laboratory processing phase, samples were placed inside a plastic free fume hood. Before processing the samples, the surface of the fume hood was wiped with alcohol. All blood samples and control filtered pure water samples were stored in glass tubes featuring heparin anticoagulation (equipped with rubber tube caps). Five glass vacutainers with 0.45 μm filtered deionized water were set up as blanks, and 1 g filtered deionized water of vacutainers were pre-treated and analyzed along with the experimental samples. (2) recovery experiment: MP solutions of known concentrations were introduced into one blood sample, post the different pre-treatment protocols, to conduct recovery assessments. Recovery rate was calculated by dividing the measured concentration by the nominal spiked concentration × 100% and are tabulated in the Table S1. (3) Limits of detection and quantification: the limit of detection and quantification were three and ten times the baseline noise, respectively (Table S2).
Sample collection
While patients were admitted to the hospital ward, a venipuncture was performed to collect whole blood samples. These samples were drawn directly into 10 mL heparinized glass vacutainers, ensuring that the containers remained sealed throughout both the sampling process and subsequent storage. The venipuncture was conducted using a sterile stainless-steel surgical-grade needle, which was connected to the glass vacutainer under a vacuum system, ensuring a direct and sterile transfer of blood from the vein to the vacutainer.
Pre-treatment
Comparative analysis was employed to evaluate the efficacy of divergent pre-treatment modalities on MP quantification, as per modified methodologies delineated in prior investigations 7, 8, 18–20. The analyzed pre-treatment methodologies comprised: (1) acid digestion: samples were subjected to an incubation phase with 68 wt% nitric acid, in a volumetric ratio of 1:3 to the sample, at room temperature for a duration of 48 h, subsequently followed by a thermal treatment at 60 °C for a period of 3 h. (2) alkaline digestion: a 10 wt% sodium hydroxide solution was utilized, in a volumetric ratio of 1:3 to the sample, sustained at 60 °C over an 8-h digestion phase. (3) Solvent extraction: initiated with a chloroform phase to dissolve polymers such as polystyrene (PS), polycarbonate (PC), and polymethyl methacrylate (PMMA). This was succeeded by an extraction with hexafluoroisopropanol targeting polyethylene terephthalate (PET), polyvinyl chloride (PVC), and polyamide (PA) dissolution, and concluded with a xylene-induced dissolution at 150 °C for a decadal minute span to address PP and polyethylene (PE), with each extraction sequence conducted in triplicate. During the digestion or extraction phases, 1 μm polytetrafluoroethylene filtration membranes are repeatedly employed to remove nanoplastic particulates. Upon a comparative evaluation of the pre-treatment methodologies, the solvent extraction technique was adjudicated as the optimal method (Table 1).
Table 1.
MP Detection efficiencies in Py-GC/MS analysis post various pre-treatments
| Polymers | PS | PE | PP | PMMA | PVC | PC | PET | PA6 | PA66 |
|---|---|---|---|---|---|---|---|---|---|
| Acid digestion (μg/g) | 3.8360 | 136.2368 | ND | ND | 161.3833 | ND | ND | ND | ND |
| Alkaline digestion (μg/g) | ND | 171.8356 | 22.6134 | ND | 158.4786 | ND | ND | ND | ND |
| Solvent extraction (μg/g) | 2.9510 | 200.2428 | 29.7244 | ND | 142.4334 | ND | ND | ND | ND |
Sample details: Patient #22, a 55-year-old male with a diagnosis of non-ST elevation acute coronary syndrome (NSTE-ACS), was chosen at random. ND: non-detectable
Sample analysis
Sample analyses were performed at room temperature and in a dust-free environment. After pre-treatment, samples were subjected to pyrolysis at 550 °C (PY-3030D and AS-1020E, Frontier, Fukushima, Japan). The resulting pyrolysis products were run on a GC–MS system fitted with a 30 m × 0.25 mm × 0.25 μm column (Rtx-5MS, Restek, Lisses, France). Helium was used as the carrier gas at a linear velocity of 36.1 cm/s. The oven program consisted of an initial 2 min hold at 40 °C, followed by an increase to 320 °C at a rate of 20 °C/min, which was then maintained for 14 min, resulting in a total run time of 30 min. Mass spectra were obtained using GC/MS (GCMS-QP2020, Shimadzu, Kyoto, Japan). To avoid re-condensation, the interface temperature was set at 320 °C. Mass spectra were recorded from m/z 29 to 600. The LabSolutions software 4.45 was utilized for sample identification. The target polymers were PS, PE, PP, PMMA, PVC, PC, PET, PA6, and PA66. Each polymer was identified based on its specific characteristic components and ions (Table S3 and Figs. S1 and S2). The composition was quantified using the calibration curves of the selected particles presented in Table S4.
Monocyte counts, lymphocyte population, and immunoglobulin detection
Monocyte counts were collected from the medical records of recruited patients. According to the manufacturer’s instructions, the populations of T-, B-, and natural killer (NK)-lymphocytes from peripheral blood without pre-treatment were enumerated using the Multitest™ IMK Kit (BD Biosciences, San Jose, USA) with CD3+ (CD3+/CD4+ and CD3+/CD8+), CD3−/CD19+, and CD3−/CD56+CD16+ assays, respectively, and a BD FACSCanto II flow cytometer with BD FACSCanto™ clinical software version 2.1 (BD Biosciences, San Jose, USA) was used for the analysis. Immunoglobulins G, immunoglobulin A, and immunoglobulin M were detected by an automatic analyzer (Hitachi 7600–120, Tokyo, Japan).
Cytokine assay
In accordance with the manufacturer's instructions, the serum cytokine quantification were determined by FACSCanto II system (BD Biosciences, San Jose, USA), using a multiplex kit for human IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, IL-17, TNF-α, IFN-α, and IFN-γ (Ruisikeer Biotechnology, Shandong, China), and the further analysis using FACSCanto Clinical Software (BD Biosciences, San Jose, USA). High-sensitivity C-reactive protein (hs-CRP) levels are obtained from routine examination procedures performed by the clinical laboratory department in our hospital.
Materials
Details regarding the reagents and consumables are listed in the Table S5.
Statistical methods
Categorical variables were summarized by frequency (n) or percentages (%) and compared by using the chi-square test. Continuous and non-normally distributed variables are presented as the mean and standard deviation, and medians and interquartile ranges, respectively. Two-tailed Student’s t test and Mann–Whitney U test were used for the comparisons of normally distributed data and non-normally distributed data, respectively. The correlations for immunological and inflammatory markers were assessed through linear regression analysis, while Tobit regression—a regression analysis method suitable for handling truncated data, which estimates the linear dependence between variables by modeling the restricted distribution of the data (e.g., SYNTAX scores, which are positive values)—was employed to evaluate coronary complexity. The correlation between MP contents and Syntax Scores was adjusted for age using the residual method, which involves subtracting the predicted values from the actual values. Statistical analyses were performed with Prism software (v.10.0.3, Boston, USA) and STATA software (v.17, Texas, USA).
Results
Positive correlation between blood microplastic levels and coronary artery complexity
While existing research has identified MP pollutants across various human organ systems, the nexus between these particulates and human pathologies, particularly cardiovascular disorders, is still emerging. This study embarked on elucidating the potential association between MPs and ACS by enrolling patients with acute chest pain at admission.
A total of 145 individuals presenting with chest pain were initially enrolled in the study. Of these, 19 had a history of PCI or MI within the past 6 months, 15 withdrew consent, and 10 were diagnosed with non-coronary conditions, such as aortic dissection. Consequently, the study ultimately included 101 participants. Coronary angiography excluded coronary artery disease in 19 of these individuals, while confirming coronary pathology in the remaining 82 individuals. Baseline characteristics of the participants are summarized in Table 2. The average age was 56.85 years, the average body mass index was 26.56 kg/m2, and the majority (79.21%) were male. Cardiac, liver, and kidney function markers were within normal ranges for all participants, and no differences were found in blood lipid levels or routine blood examination results between groups, based on mean or median values. Notably, 79.21% of the participants had a history of medical interventions, including pharmacological injection treatments, history of surgical procedures (encompassing general surgical interventions and cardiac interventional surgeries conducted over 6 months prior), and intravenous imaging examinations.
Table 2.
Clinical characteristics of participantsa
| Variable | Participants (n = 101) | Total (n = 101) | ||
|---|---|---|---|---|
| Control groupb (n = 19) | ACS group (n = 82) | P-value | ||
| Diagnosis | < 0.001 | |||
| No significant stenosis or blockage | 19 (100.00) | 0 (0.00) | 19 (18.81) | |
| UA | 0 (0.00) | 46 (56.10) | 46 (45.54) | |
| NSTE-ACS | 0 (0.00) | 32 (39.02) | 32 (31.68) | |
| STEMI | 0 (0.00) | 4 (4.88) | 4 (3.96) | |
| Age (years) | 53.58 ± 11.97 | 57.61 ± 10.40 | 0.1423 | 56.85 ± 10.77 |
| Male sex | 15 (78.95) | 65 (79.26) | 0.9755 | 80 (79.21) |
| BMI (kg/m2) | 26.61 ± 4.08 | 26.54 ± 4.07 | 0.9438 | 26.56 ± 4.07 |
| Hypertension | 7 (36.84) | 45 (54.88) | 0.1595 | 52 (51.49) |
| Diabetes mellitus | 4 (21.05) | 23 (28.05) | 0.5394 | 27 (26.73) |
| Cerebral infarction | 0 (0.00) | 9 (10.98) | 0.1329 | 9 (8.91) |
| Malignancies | 1 (5.26) | 3 (3.66) | 0.7496 | 4 (3.96) |
| Current smoking | 9 (47.37) | 26 (31.71) | 0.1999 | 35 (34.65) |
| SYNTAX Scorec | 0 ± 0.00 | 17.00 (10.00, 25.00) | < 0.001 | 13.00 (7.00, 23.50) |
| LVEF (%) | 60.67 ± 7.47 | 60.97 ± 5.86 | 0.8588 | 60.93 ± 6.10 |
| ALT (U/L) | 23 (15, 47) | 18 (14, 29) | 0.0762 | 19 (14, 30) |
| AST (U/L) | 24 (18, 35) | 20 (16, 30) | 0.3464 | 20 (16, 32) |
| Hemoglobin (g/L) | 150.60 ± 12.80 | 147.60 ± 17.07 | 0.4672 | 148.20 ± 16.35 |
| White blood cell (*109/L) | 6.08 (5.21, 9.52) | 7.31 (5.63, 8.52) | 0.3342 | 7.20 (5.62, 8.54) |
| Platelets (*109/L) | 218.90 ± 63.82 | 228.10 ± 63.80 | 0.5760 | 226.30 ± 63.58 |
| Low-density lipoprotein (mmol/L) | 2.47 ± 1.02 | 2.54 ± 1.10 | 0.7906 | 2.53 ± 1.07 |
| High-density lipoprotein (mmol/L) | 1.05 ± 0.25 | 1.08 ± 0.28 | 0.6005 | 1.08 ± 0.28 |
| Total cholesterol (mmol/L) | 4.17 ± 1.13 | 4.37 ± 1.20 | 0.5007 | 4.33 ± 1.19 |
| Triglycerides (mmol/L) | 1.73 (1.47, 3.08) | 1.69 (1.14, 3.00) | 0.6742 | 1.71 (1.15, 3.00) |
| eGFR (mL/min/1.73m2)d | 93.75 ± 16.85 | 100.60 ± 15.50 | 0.1064 | 95.05 ± 16.75 |
| History of Medical Interventionse | 16 (84.21) | 64 (78.05) | 0.5510 | 80 (79.21) |
| Subcutaneous injection therapy | 2 (10.53) | 8 (9.76) | 0.9191 | 10 (9.90) |
| Cardiac interventional procedures | 5 (26.32) | 32 (39.02) | 0.3002 | 37 (36.63) |
| Surgical operations | 5 (26.32) | 19 (23.17) | 0.7716 | 24 (23.76) |
| Intravenous contrast imaging | 9 (47.37) | 29 (35.37) | 0.3305 | 29 (28.71) |
aData displayed as mean ± standard deviation, median values (interquartile range), and n (%). Results with statistical significance are indicated in bold. UA, unstable angina; NSTE-ACS, non ST-elevation acute coronary syndrome; STEMI, ST-segment elevation myocardial infarction; BMI, body mass index; LVEF, left ventricular ejection fraction; ALT, alanine transaminase; AST, aspartate aminotransferase; eGFR, estimated glomerular filtration rate
bThe control group consisted of patients who presented with symptoms suggestive of acute coronary syndrome but displayed no vascular anomalies upon coronary angiography, signified by a SYNTAX Score of 0
cThe SYNTAX Score gauges the intricacy of coronary artery disease via coronary angiography. It delineates the position, type, and magnitude of coronary abnormalities, with scores spanning from 0 to upper ranges. Elevated SYNTAX Scores represent heightened complexity in coronary artery disease, facilitating the decision-making process between coronary artery bypass graft and PCI
dEstimated glomerular filtration rate was calculated using the CKD Epidemiology Collaboration creatinine equation
eHistory of medical interventions encompasses the patient's previous personal medical intervention history, including pharmacological treatments such as insulin injections, proprotein convertase subtilisin/kexin type 9 inhibitor injections, surgical histories like cardiac interventional surgeries conducted over 6 months prior, orthopedic operations, cesarean sections, as well as imaging examinations including myocardial perfusion imaging and coronary CT scans. A subset of patients possess multiple histories of medical interventions
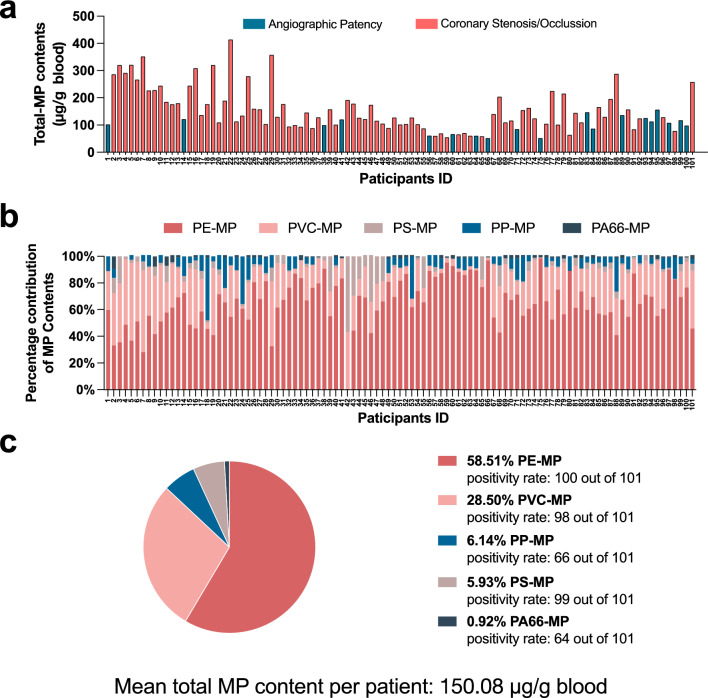
Subsequent to hospital admission, we quantified the levels of MPs in preoperative venous blood samples using Py-GC/MS. As shown in Fig. 1a, MPs were detected in the blood of all 101 participants, irrespective of their final diagnosis of ACS, with concentrations peaking at 413.99 μg per gram of blood. Five types of MPs were identified: PE, PVC, PP, PS, and PA66, as shown in Fig. 1b. Among the MPs identified, PE was the most prevalent type, constituting 58.51% of all MP detections, followed by PVC at 28.50%, PP at 6.14%, PS at 5.93%, and PA66 at 0.92% (Fig. 1c), with an average concentration across all types of 150.08 μg per gram of blood.
Fig. 1.
Distribution characteristics of microplastics in the blood of 101 participants. a Total microplastic content in the blood of each of the 101 participants. Participants were divided into a group with normal angiography results (n = 19) and a group with abnormal angiography results (n = 82); b Proportion of different types of microplastics in each participant's blood; c Overall characteristics of blood microplastics among the 101 participants
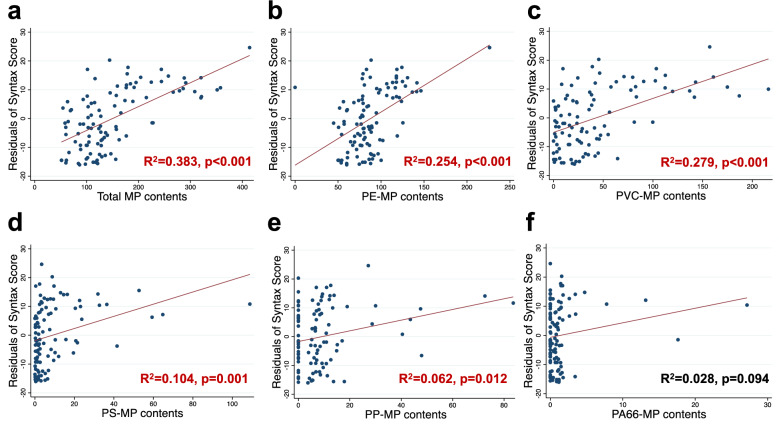
To further explore the association between the preoperative blood MP content and coronary artery pathology, we initially assessed the coronary arteries of all participants using the SYNTAX Score. Subsequent analysis explored the correlation between MP content and SYNTAX Score values. After adjusting for age, our findings revealed that the total MP content, as well as the specific contents of PE, PVC, PS, and PP, were positively correlated with the SYNTAX Score (Figs. 2a–e, Table 3), suggesting that these MPs may promote the progression of coronary artery disease. No positive correlation was observed for PA66-MP content (Fig. 2f, Table 3).
Fig. 2.
Linear regression analyses between syntax score residuals and various MP contents. Each panel depicts the impact of a different type of microplastic on Syntax Score residuals, adjusted for age. Results with statistical significance are indicated in red. a Total microplastic content; b polyethylene microplastic content; c polyvinyl chloride microplastic content; d polypropylene microplastic content; e polystyrene microplastic content; f polyamide 66 microplastic content
Table 3.
Association of MP contents and SYNTAX Score in 101 individuals presenting with chest pain
| MP contents (μg/g blood) | SYNTAX score | |||
|---|---|---|---|---|
| Coefficient (SE) | P-value | Coefficient (SE) | P-value | |
| Total MP | 0.092(0.013) | < 0.001 | ||
| PE-MP | 0.206(0.038) | < 0.001 | ||
| PVC-MP | 0.131(0.023) | < 0.001 | ||
| PS-MP | 0.246(0.074) | 0.001 | ||
| PP-MP | 0.224(0.087) | 0.012 | ||
| PA66-MP | 0.550(0.351) | 0.120 | ||
Results are from Tobit regression models, which used Py-GC/MS data and adjusted for age. Results with statistical significance are indicated in bold. The units of the MPs are μg/g blood. SE, standard error; MP, microplastic; PE, polyethylene; PVC, polyvinyl chloride; PP, polypropylene; PS, polystyrene; PA66, polyamide 66
Elevated microplastic burden in acute coronary syndrome patients
In the initial phase of our study, although a positive correlation between MP content and the SYNTAX Score was identified, the distribution characteristics of MP levels between ACS patients and those with normal angiographic results remained unclear. Consequently, based on the results of coronary angiography, we further stratified the 101 participants into a control group and an ACS patient group.
Baseline demographics and clinical characteristics, as shown in Table 2, revealed no significant discrepancies between the groups concerning age, gender, prevalence of hypertension, diabetes, cerebrovascular incidents, ejection fraction, and liver and renal function indices. The complexity of vasculature, as assessed by the SYNTAX Score, had a median of 17 (interquartile range 10, 25) in the ACS group, compared to a null score in the control group. Furthermore, there were no significant differences between the two groups of patients in terms of medical treatment/examination records prior to admission.
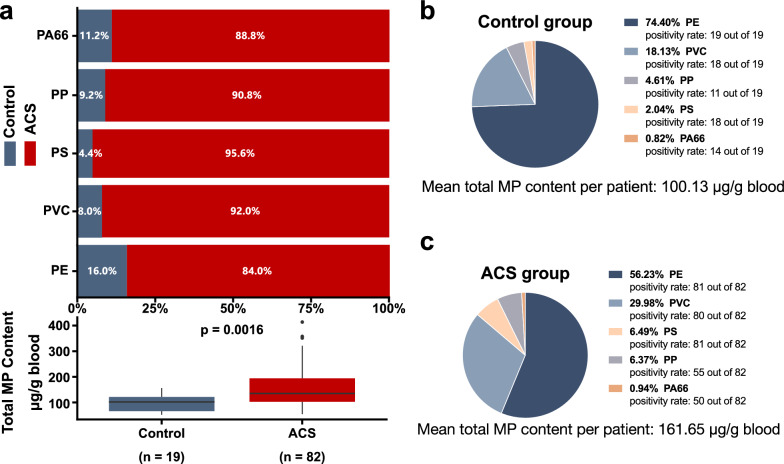
As demonstrated in Fig. 3a, the MP content in the blood of ACS patients was significantly higher than that of the control patients. Moreover, the proportionate contribution of MPs in the blood was notably higher in the ACS group compared to the control group, varying from 84.0 to 95.6%. On average, MPs in the control group measured 100.13 μg per gram of blood (Fig. 3b), increasing to 161.65 μg per gram in the ACS group (Fig. 3c). In the control patients, the highest content was PE, followed by PVC, PP, PS, and PA66. For ACS patients, the order was PE, PVC, PS, PP, and PA66. In both groups, the highest content was PE, accounting for 74.40% in the control group and decreasing to 56.23% in the ACS group. Conversely, the proportion of PVC increased in ACS patients, from 18.13% in the control group to 29.98%.
Fig. 3.
Comparison of detected microplastic in blood across control and acute coronary syndrome group. a Between-group comparison; b Within-group comparisons in the control group; b Within-group comparisons in the acute coronary syndrome group. ACS, acute coronary syndrome; PE, polyethylene; PVC, polyvinyl chloride; PP, polypropylene; PS, polystyrene; PA66, polyamide 66
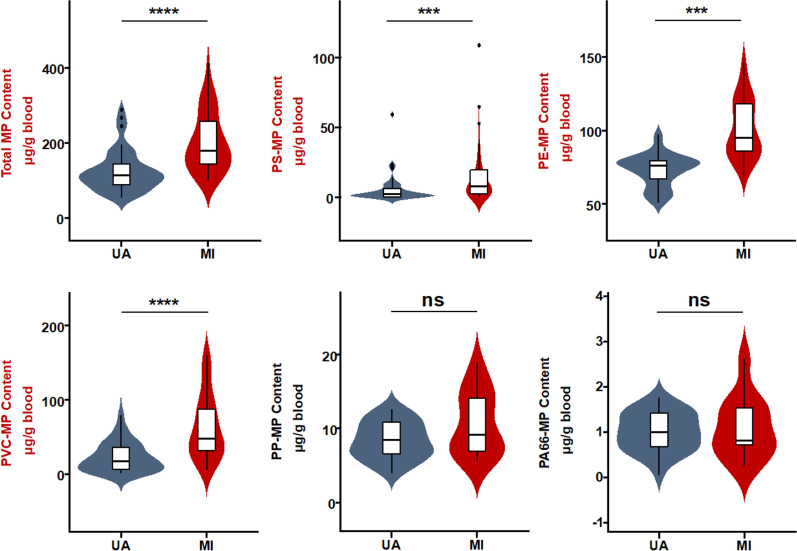
Further analysis among ACS patients was conducted to discern internal variations. Following established guidelines 13, ACS patients were classified into unstable angina (UA, n = 48) and MI (n = 34) groups. Figure 4 shows the total MP concentration in the blood of MI patients upon admission was significantly higher than in UA patients. Specifically, concentrations of PS, PE, and PVC were also significantly elevated in MI patients, while no difference was observed for PP and PA66. Comparisons between male (n = 65) and female (n = 17) ACS patients showed no significant differences in total or individual MP concentrations (Fig. S3a). Moreover, a comparison between early-onset (n = 44, defined as men under 55 years of age and women under 65 years of age) and late-onset (n = 38, male aged 55 or above and women aged 65 or above) coronary heart disease (CAD) patients also revealed no significant differences in MP levels (Fig. S3b) 21.
Fig. 4.
Differential microplastic burdens in blood samples of unstable angina and myocardial infarction patients. Results with statistical significance are indicated in red. The concentrations of total microplastics and specific MP types (PS, PE, PP, PVC, PA66) in µg/g of blood in MI (n = 34) patients and UA (n = 48) patients. ***p < 0.001; ****p < 0.0001. UA, unstable angina; MI, myocardial infarction
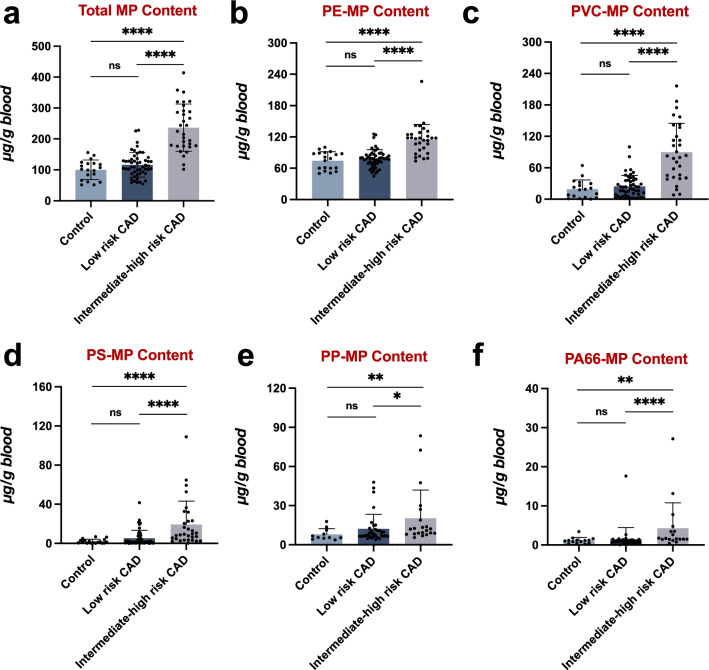
To delve deeper, we further stratified ACS patients based on their SYNTAX Score into two groups: those with a low-risk CAD (SYNTAX Score below 23, n = 51), and those with medium-to-high-risk CAD (SYNTAX Score of 23 or above, n = 31). Our analysis proceeded to compare MP concentrations across three distinct cohorts: control patients, low-risk CAD, and medium-to-high-risk CAD patients. Our findings revealed that, compared to control patients, low-risk ACS patients exhibited an upward trend in MP concentration, albeit not statistically significant. Conversely, the total MP content in medium-to-high-risk ACS patients was markedly higher (Fig. 5a). This observed pattern was consistent across each individual MP type (Fig. 5b–f), suggesting a potential association between increased MP dose and higher prognostic risk in ACS patients.
Fig. 5.
Exploring the relationship between microplastic burden and the complexity of coronary artery lesions. The MP content across different risk categories of CAD and control patients were analyzed: a Total MP content. b PS-MP content. c PE-MP content. d PP-MP content. e PVC-MP content. f PA66-MP content. Results with statistical significance are indicated in red. *p < 0.05, **p < 0.01, ****p < 0.0001. ns, not significant
These results indicate that the severity and type of ACS, rather than the age of onset or gender, are associated with changes in MP distribution.
Increased immunological and inflammatory burden associated with microplastic concentration
Although a correlation between the coronary complexity and elevated MP concentrations in the blood was observed, the extent to which MPs influence factors affecting coronary lesions is not well-established. Considering the significance of the immune-inflammatory state in coronary lesions, we further analyzed the association between MP concentrations and the immune/inflammatory profile of participants.
We examined correlations between the levels of a panel of inflammatory cytokines—specifically IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, IL-17, TNF-α, IFN-α, and IFN-γ—and the levels of high-sensitivity C-reactive protein (hs-CRP) in relation to the concentration of various MPs. Our results revealed that total MP contents were significantly and positively correlated with IL-6 and IL-12p70 levels (Table 4). In the correlation analysis of different MP types with cytokine concentrations, adjusted for age, only specific MP types demonstrated significant correlations. PA66-MP negatively correlated with IL-1β, while PVC-MP positively correlated with IL-6. Both PE-MP and PVC-MP, along with PS-MP, were positively correlated with IL-12p70. Additionally, PA66-MP was negatively correlated with IL-17, PE-MP negatively correlated with IFN-γ, and PS-MP positively correlated with IFN-γ (Table S6). The heterogeneous effects of MPs on inflammatory cytokines may stem from the distinct physicochemical properties of different MP materials. Despite variations in MP types among individuals, the widespread presence of various MPs in human blood indicates that MP accumulation could potentially upregulate specific pro-inflammatory cytokines involved in coronary pathologies.
Table 4.
Association of total MP content with inflammatory cytokines and high-sensitivity c-reactive protein in 101 individuals with chest pain
| IL-1β (pg/mL) | IL-2 (pg/mL) | IL-4 (pg/mL) | IL-5 (pg/mL) | IL-6 (pg/mL) | IL-8 (pg/mL) | IL-10 (pg/mL) | |
|---|---|---|---|---|---|---|---|
| Total MP content | − 0.002 | − 0.000 | − 0.000 | − 0.000 | 0.042* | − 0.077 | − 0.004 |
| (μg/g blood) | (0.002) | (0.001) | (0.002) | (0.001) | (0.020) | (0.052) | (0.004) |
| Constant | 2.014* | 1.523 | 1.462* | 2.462 | 4.470 | 69.024 | 1.677 |
| (0.923) | (0.973) | (0.608) | (1.655) | (6.289) | (55.857) | (1.559) | |
| R2 | 0.017 | 0.002 | 0.000 | 0.027 | 0.087 | 0.025 | 0.027 |
| IL-12p70 (pg/mL) | IL-17 (pg/mL) | TNF-α (pg/mL) | IFN-α (pg/mL) | IFN-γ (pg/mL) | hs-CRP (mg/L) | |
|---|---|---|---|---|---|---|
| Total MP Content | 0.004** | − 0.008 | − 0.004 | 0.001 | − 0.000 | 0.011 |
| (μg/g blood) | (0.001) | (0.007) | (0.003) | (0.001) | (0.000) | (0.006) |
| Constant | 1.191 | 8.257 | 2.907 | 1.269 | 0.861*** | 2.588 |
| (0.612) | (4.215) | (1.609) | (0.821) | (0.268) | (2.708) | |
| R2 | 0.086 | 0.031 | 0.019 | 0.015 | 0.020 | 0.032 |
Results are from linear regression models and adjusted for age. Robust standard errors in parentheses. Results with statistical significance are indicated in bold. The units of the MPs are μg/g blood. **p < 0.01; *p < 0.05. TNF-α, tumor necrosis factor-α; IFN-α, interferon-α; IFN-γ, interferon-γ; hs-CRP, high-sensitivity C-reactive protein
The complex interactions between monocytes, lymphocytes, and cytokines are well-documented, yet research exploring the association between MP levels and these cell types remains limited. To ascertain the relationship between MP dose and immune cells, we assessed the monocyte counts and lymphocyte subpopulation profiles in the blood of all participants and conducted linear regression analyses with MP concentrations (Table 5). Our findings indicate that, after adjusting for age, there is a significant positive correlation between total MP content and the quantities of B cells (CD3−/CD19+ cells) and NK cells (CD3−/CD56+CD16+ cells), with no significant correlation observed with monocytes and T cells. Moreover, only specific types of MPs showed significant associations with certain lymphocyte subpopulations: PS-MP was negatively correlated with T suppressor cells (CD3+/CD8+ cells), while PE-MP, PVC-MP, and PP-MP were positively correlated with both B cells and NK cells (Table S7). To clarify the impact of MP on B cell function, we further analyzed the relationship between MP contents and the levels of three types of immunoglobulins, finding no significant correlation (Table S7).
Table 5.
Association between total MP content and monocytes, lymphocytes, and cytokines profiles in 101 individuals with chest pain
| Monocytes (*109/L) | CD3+/CD4+ (per μL) | CD3+/CD8+ (per μL) | CD3−/CD19+ (per μL) | CD3−/CD56+CD16+ (per μL) | IgA (g/L) | IgG (g/L) | IgM (g/L) | |
|---|---|---|---|---|---|---|---|---|
| Total MP Content | − 0.000 | 0.351 | 0.271 | 0.605** | 0.557** | − 0.001 | 0.001 | 0.001 |
| (μg/g blood) | (0.000) | (0.376) | (0.270) | (0.180) | (0.201) | (0.001) | (0.003) | (0.001) |
| Constant | 0.615** | 777.175** | 544.198** | 296.411** | 104.022 | 2.513** | 8.481** | 1.092** |
| (0.094) | (188.255) | (114.970) | (77.281) | (62.917) | (0.706) | (1.836) | (0.264) | |
| R2 | 0.053 | 0.021 | 0.025 | 0.204 | 0.128 | 0.003 | 0.067 | 0.069 |
Results are from linear regression models and adjusted for age. Robust standard errors in parentheses. Results with statistical significance are indicated in bold. The units of the MPs are μg/g blood. **p < 0.01; *p < 0.05. IgG, immunoglobulin G; IgA, immunoglobulin A; IgM, immunoglobulin M
Discussion
Since the initial recognition of MP contamination in 2004, there has been extensive research into its environmental and biotic impacts. Recently, evidence of MP pollution has been identified in human excreta, bodily fluids, and even organ tissues. However, notwithstanding these advancements, research directly examining the interplay between MP concentration and disease pathophysiology in humans remains scarce. Grounded in existing literature, we propose a novel and concerning hypothesis: MPs may influence inflammatory and immune responses, potentially correlating with more intricate vascular pathologies and exacerbated clinical manifestations in ACS patients.
In our study, MP was ubiquitously detected in all participants, with ACS patients demonstrating a pronounced elevation in MP levels. Due to the absence of pre-onset data, we cannot conclusively determine whether these augmented MP concentrations are attributable to an acute escalation concurrent with the manifestation of chest pain or are indicative of a chronic, elevated baseline. Irrespective of the underlying temporal dynamics, each hypothesis posits a substantial association with the pathophysiological trajectory of ACS. The former scenario posits MPs as a potential co-contributor to ACS onset, a notion particularly salient in the context of MI patients. The latter hypothesis implicates chronic physicochemical insults from the sustained toxic effects of MPs, a phenomenon pertinent to both MI and UA patient cohorts.
From a broader perspective, ACS encompasses a spectrum of clinical syndromes precipitated by rapid constriction of coronary arteries, which includes various clinical ischemic symptoms due to myocardial ischemia induced by coronary artery disease and is classified into three types: UA, Non-ST-Elevation ACS (NSTE-ACS), and ST-Elevation Myocardial Infarction (STEMI) 13. The latter two are predominantly characterized by rupture or erosion of atherosclerotic plaques, leading to either complete or incomplete occlusive thrombus formation. Irrespective of the specific ACS subtype, chronic atherosclerotic plaque development is a fundamental pathophysiological element. These plaques have a complex composition, including lipids, cellular elements, calcium ions, and fibrin. Notably, immune cells within the cellular component play a crucial role in plaque formation and characteristics. For instance, depleting NK cells in mice with specific antibodies alleviates atherosclerotic plaque formation 22, and certain NK cell subtypes are associated with plaque instability 23. Moreover, removal of mature B lymphocytes with specific monoclonal antibodies significantly reduces the area of atherosclerotic plaques in mice 24. In our results, elevated MP concentrations were correlated with increased numbers of NK and B cells, suggesting that MP might promote atherosclerosis progression through immune cell activation. Conversely, the role of T lymphocytes in atherosclerotic plaque development is paradoxical. T helper cells are known to promote atherosclerosis, whereas cytotoxic T cells can contribute to plaque instability but also protect against atherosclerosis by reducing levels of atherogenic IgG 25. Our study found no significant correlation between T cell counts and total MP content, leaving the effect of MPs on T cells in ACS patients and their role in atherosclerosis progression unclear. Monocyte-macrophages play an integral role in the formation of lipid streaks and the subsequent rupture of atherosclerotic plaques 26. Prior research, however, has not demonstrated a significant relationship between PE-MP concentration and macrophage viability 27. Consistent with these findings, our study also shows a lack of correlation between MP concentration and monocyte counts. Still, further exploration is needed to ascertain the potential impact of MPs on the inflammatory cytokine secretion capabilities of monocyte-macrophages.
Although there is no direct evidence at the human level that the MP dose in blood is associated with MI, a recent study underscores a significant relationship between the presence of NMP within carotid atherosclerotic plaques and subsequent adverse cardio-cerebrovascular events. Researchers quantitatively assessed the NMP content within the carotid plaques of 257 individuals using Py-GC/MS. Subsequent to a median follow-up duration of 33.7 ± 6.9 months, it was discerned that individuals with plaques testing positive for NMPs incurred a 4.53-fold increase in the risk of MI, cerebrovascular incidents, and all-cause mortality compared to their counterparts whose plaques were devoid of such MPs 15.
Regarding the potential atherogenic effects of MPs, MPs could contribute to the progression of atherosclerosis in multiple ways. Recent studies have found that in ApoE−/− mice, exposure to PS-NMPs via the digestive tract significantly increases the area of atherosclerotic plaques. This change may be related to alterations in cytokine and immune cell pathways, as well as changes in liver lipid metabolism 28, 29. The imbalance between pro-inflammatory and anti-inflammatory mechanisms is a decisive driver of atherosclerosis 30. Researchers have found that co-culturing human peripheral blood monocytes with PMMA and PS particles results in an upregulation of pro-inflammatory cytokines such as IL-6 in the supernatant 31. Correspondingly, our results also indicated a positive correlation between IL-6 expression and MP contents. Furthermore, MPs have been implicated in endothelial cell damage; endothelial dysfunction is not only a key initiating factor of atherosclerosis but also a crucial condition for thrombus formation 32. Moreover, our team recently found that the concentration of MPs in arteries containing atherosclerotic plaques was significantly higher than that in aortas which did not contain atherosclerotic plaques 7. These findings suggest that MPs may promote atherosclerosis by altering internal inflammation levels or inducing endothelial dysfunction.
Another noteworthy issue is that the concentration of MPs detected in our blood samples far exceeds the total lifetime exposure of just over 10 mg predicted by Mohamed's probabilistic lifetime exposure model 33. Since the estimates by Mohamed et al. are primarily based on exposure through food and air, without accounting for medical-related MP exposures, their model might underestimate human MP exposure levels. Indeed, recent studies have reported PE-MP concentrations in human carotid plaque tissues reaching 21.7 mg per gram of plaque, necessitating further in-depth discussions on MP exposure and hazards based on human-centric studies 15.
In this study, all samples unexpectedly tested negative for PET-MP, which contrasts with previous research reports, including our prior publications based on patients receiving cardiac surgery 8. Considering that the subjects of this study were ACS patients, coupled with the established detection capabilities for PET via Py-GC/MS delineated in our methodological framework, it is presently unclear whether the absence of PET in ACS patient blood is due to a genuinely lower prevalence or concentrations beneath the threshold of detection. This ambiguity highlights the need for additional research.
It is important to acknowledge certain limitations of our study. (1) Given that medical procedures may be another route for human MP exposure 34, our study specifically analyzed MP content in blood samples collected at admission, to minimize potential contamination of MP. This approach limited our analysis to exploring the correlation between blood MP levels and ACS at different time point. Future research should track the dynamics of MP content in the blood of healthy populations, to more definitively ascertain the chronic impacts of MPs on vascular lesion development. (2) The invasive nature of coronary angiography constrains the inclusion of healthy participants in this study, which not only restricted the sample size of the control group (n = 19) but also failed to clarify the potential risks of MPs to healthy populations. Future research needs to further expand the sample size of relatively healthy populations to verify the conclusions of this study, and could also consider exploring the risks of MP exposure in healthy populations using non-invasive indicators such as cardiac MRI. (3) Our study only focused on the analysis of lymphocyte subgroups and monocyte counts, and did not investigate the effects of MPs on human macrophage phenotypes. Future research could delve deeper into the mechanisms of MPs-related cardiovascular diseases from perspectives such as macrophage pro-inflammatory phenotypes or trained immunity.
Conclusions
In summary, by employing coronary angiography, Py-GC/MS, and flow cytometry, we report for the first time a correlation between MPs and the complexity of vascular pathologies as well as the immunoinflammatory status in ACS patients. Navigating the challenge of complete plastic avoidance, particularly in the context of medical diagnostics and treatment in the modern 'Plastic Age', poses a monumental global challenge. Further research is imperative to unravel the pathogenic mechanisms and guide strategies to ameliorate the cardiovascular risks associated with MPs.
Supplementary Information
Acknowledgements
The authors wish to thank G. H. Qu, Y. Y. Ding, S. X. Lin, L. Q. Chen, and Y. Z. Zhu for their strong supports. Graphical abstract was created with BioRender.com (under license number YO26SUB3NP).
Abbreviations
- MP
Microplastic
- ACS
Acute coronary syndrome
- Py-GC/MS
Pyrolysis gas chromatography mass spectrometry
- PCI
Percutaneous coronary intervention
- MI
Myocardial infarction
- PP
Polypropylene
- PS
Polystyrene
- PC
Polycarbonate
- PMMA
Polymethyl methacrylate
- PET
Polyethylene terephthalate
- PVC
Polyvinyl chloride
- PA
Polyamide
- PE
Polyethylene
- hs-CRP
High-sensitivity c-reactive protein
- UA
Unstable angina
- NSTE-ACS
Non ST-elevation acute coronary syndrome
- STEMI
ST-segment elevation myocardial infarction
- LVEF
Left ventricular ejection fraction
- ALT
Alanine transaminase
- AST
Aspartate aminotransferase
- eGFR
Estimated glomerular filtration rate
- TNF
Tumor necrosis factor
- IFN
Interferon
- Ig
Immunoglobulin
- NMP
Nano- and microplastic
- NK cell
Natural killer cell
Author contributions
Yunxiao Yang: conceptualization, methodology, investigation, formal analysis, visualization, writing—original draft. Feng Zhang: data curation, formal analysis, writing—review and editing. Zhili Jiang: methodology, investigation, formal analysis, writing—review and editing. Zhiyong Du: formal analysis, methodology. Sheng Liu: data curation, visualization, investigation. Ming Zhang: investigation. Yanyan Jin: methodology, investigation. Yanwen Qin: investigation, writing—review and editing. Xiubin Yang: methodology, supervision. Chenggang Wang: writing—review and editing, supervision, project administration. Hai Gao: writing—review and editing, supervision, project administration, Funding acquisition.
Funding
The work was supported by the National Natural Science Foundation of China (No. 82100295) and Capital’s Funds for Health Improvement and Research (CFH 2022–1-2062).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
This study was carried out in accordance with relevant guidelines and regulations with ethical approval obtained from the Ethics Committee of Beijing Anzhen Hospital (Approval No. KS2023049).
Consent for publication
Written consent for publication was obtained from the patient.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yunxiao Yang, Feng Zhang, and Zhili Jiang contributed equally to this work
Contributor Information
Chenggang Wang, Email: chamwang@163.com.
Hai Gao, Email: gaohai1221@mail.ccmu.edu.cn.
References
- 1.Thevenon F, Carroll C, Sousa J. Plastic debris in the ocean: the characterization of marine plastics and their environmental impacts, situation analysis report. Gland, Switzerland IUCN. 2014;52:19–26. [Google Scholar]
- 2.Kershaw P. Sources, fate and effects of microplastics in the marine environment: a global assessment: International Maritime Organization. 2015. Report No.: 1020–4873.
- 3.Suman KH, Haque MN, Uddin MJ, Begum MS, Sikder MH. Toxicity and biomarkers of micro-plastic in aquatic environment: a review. Biomarkers. 2021;26(1):13–25. [DOI] [PubMed] [Google Scholar]
- 4.Schwabl P, Köppel S, Königshofer P, et al. Detection of various microplastics in human stool: a prospective case series. Ann Intern Med. 2019;171(7):453–7. [DOI] [PubMed] [Google Scholar]
- 5.Zhang J, Wang L, Trasande L, Kannan K. Occurrence of polyethylene terephthalate and polycarbonate microplastics in infant and adult feces. Environ Sci Technol Lett. 2021;8(11):989–94. [Google Scholar]
- 6.Ragusa A, Svelato A, Santacroce C, et al. Plasticenta: first evidence of microplastics in human placenta. Environ Int. 2021;146: 106274. [DOI] [PubMed] [Google Scholar]
- 7.Liu S, Wang C, Yang Y, et al. Microplastics in three types of human arteries detected by pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS). J Hazard Mater. 2024;469:133855. [DOI] [PubMed] [Google Scholar]
- 8.Yang Y, Xie E, Du Z, et al. Detection of various microplastics in patients undergoing cardiac surgery. Environ Sci Technol. 2023;57(30):10911–8. [DOI] [PubMed] [Google Scholar]
- 9.Osman AI, Hosny M, Eltaweil AS, et al. Microplastic sources, formation, toxicity and remediation: a review. Environ Chem Lett. 2023;21(4):2129–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ishii T, Warabi E, Mann GE. Mechanisms underlying unidirectional laminar shear stress-mediated Nrf2 activation in endothelial cells: amplification of low shear stress signaling by primary cilia. Redox Biol. 2021;46: 102103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ruscica M, Corsini A, Ferri N, Banach M, Sirtori CR. Clinical approach to the inflammatory etiology of cardiovascular diseases. Pharmacol Res. 2020;159: 104916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Di W, Feng Y, Wang R, et al. Pigment microparticles and microplastics found in human thrombi based on Raman spectral evidence. J Adv Res. 2023;49:141–50. 10.1016/j.jare.2022.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44(38):3720–826. [DOI] [PubMed] [Google Scholar]
- 14.Bhatt DL, Lopes RD, Harrington RA. Diagnosis and treatment of acute coronary syndromes: a review. JAMA. 2022;327(7):662–75. [DOI] [PubMed] [Google Scholar]
- 15.Marfella R, Prattichizzo F, Sardu C, et al. Microplastics and nanoplastics in atheromas and cardiovascular events. N Engl J Med. 2024;390(10):900–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Serruys PW, Onuma Y, Garg S, et al. Assessment of the SYNTAX score in the syntax study. EuroIntervention. 2009;5(1):50–6. [DOI] [PubMed] [Google Scholar]
- 17.Lee PH, Lee JY, Lee CW, et al. Comparison of a simple angiographic approach with a synergy between percutaneous coronary intervention with taxus and cardiac surgery score-based approach for left main coronary artery stenting: a pooled analysis of serial PRECOMBAT (premier of randomized comparison of bypass surgery versus angioplasty using sirolimus-eluting stent in patients with left main coronary artery disease) studies. Circ Cardiovasc Interv. 2018;11(1):e005374. [DOI] [PubMed] [Google Scholar]
- 18.Ceccarini A, Corti A, Erba F, et al. The hidden microplastics: new insights and figures from the thorough separation and characterization of microplastics and of their degradation byproducts in coastal sediments. Environ Sci Technol. 2018;52(10):5634–43. [DOI] [PubMed] [Google Scholar]
- 19.Ke D, Zheng J, Liu X, et al. Occurrence of microplastics and disturbance of gut microbiota: a pilot study of preschool children in Xiamen. China EBioMedicine. 2023;97: 104828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pfeiffer F, Fischer EK. Various digestion protocols within microplastic sample processing—evaluating the resistance of different synthetic polymers and the efficiency of biogenic organic matter destruction. Front Environ Sci. 2020;8: 572424. [Google Scholar]
- 21.Mahtta D, Gupta A, Ramsey DJ, et al. Autoimmune rheumatic diseases and premature atherosclerotic cardiovascular disease: an analysis from the VITAL registry. Am J Med. 2020;133(12):1424-1432.e1421. [DOI] [PubMed] [Google Scholar]
- 22.Selathurai A, Deswaerte V, Kanellakis P, et al. Natural killer (NK) cells augment atherosclerosis by cytotoxic-dependent mechanisms. Cardiovasc Res. 2014;102(1):128–37. [DOI] [PubMed] [Google Scholar]
- 23.Martínez-Rodríguez JE, Munné-Collado J, Rasal R, et al. Expansion of the NKG2C+ natural killer-cell subset is associated with high-risk carotid atherosclerotic plaques in seropositive patients for human cytomegalovirus. Arterioscler Thromb Vasc Biol. 2013;33(11):2653–9. [DOI] [PubMed] [Google Scholar]
- 24.Ait-Oufella H, Herbin O, Bouaziz JD, et al. B cell depletion reduces the development of atherosclerosis in mice. J Exp Med. 2010;207(8):1579–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saigusa R, Winkels H, Ley K. T cell subsets and functions in atherosclerosis. Nat Rev Cardiol. 2020;17(7):387–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Woollard KJ, Geissmann F. Monocytes in atherosclerosis: subsets and functions. Nat Rev Cardiol. 2010;7(2):77–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gautam R, Jo J, Acharya M, et al. Evaluation of potential toxicity of polyethylene microplastics on human derived cell lines. Sci Total Environ. 2022;838(Pt 2): 156089. [DOI] [PubMed] [Google Scholar]
- 28.Zhao J, Gomes D, Yuan F, et al. Oral polystyrene consumption potentiates atherosclerotic lesion formation in ApoE(-/-) mice. Circ Res. 2024;134(9):1228–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wen J, Sun H, Yang B, Song E, Song Y. Long-term polystyrene nanoplastic exposure disrupt hepatic lipid metabolism and cause atherosclerosis in ApoE(-/-) mice. J Hazard Mater. 2024;466: 133583. [DOI] [PubMed] [Google Scholar]
- 30.Bäck M, Yurdagul A Jr, Tabas I, Öörni K, Kovanen PT. Inflammation and its resolution in atherosclerosis: mediators and therapeutic opportunities. Nat Rev Cardiol. 2019;16(7):389–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wolff CM, Singer D, Schmidt A, Bekeschus S. Immune and inflammatory responses of human macrophages, dendritic cells, and T-cells in presence of micro- and nanoplastic of different types and sizes. J Hazard Mater. 2023;459: 132194. [DOI] [PubMed] [Google Scholar]
- 32.Zhu X, Wang C, Duan X, et al. Micro- and nanoplastics: a new cardiovascular risk factor? Environ Int. 2023;171: 107662. [DOI] [PubMed] [Google Scholar]
- 33.Mohamed Nor NH, Kooi M, Diepens NJ, Koelmans AA. Lifetime accumulation of microplastic in children and adults. Environ Sci Technol. 2021;55(8):5084–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu S, Yang Y, Du Z, et al. Percutaneous coronary intervention leads to microplastics entering the blood: interventional devices are a major source. J Hazard Mater. 2024;476: 135054. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.