Abstract
ABSTRACT
Objectives
Rheumatoid arthritis (RA) is associated with an increased risk for osteoporosis and osteoporotic fractures. Since the treatment of RA has improved significantly in recent years, we can expect RA-associated osteoporosis to decrease with good disease control. Therefore, we conducted a retrospective study to investigate whether the frequency of osteoporosis and osteoporotic fractures has changed during 24 years in RA.
Methods
We analysed the data of 1.086 RA patients from the time of the first osteological assessment with bone mineral density (BMD) measurement and collection of osteologically important data during the years 1996 and 2019 at our clinic. According to the treatment period, the patients were divided into cohort 1 (investigation between 1996 and 2004; n=539) and cohort 2 (investigation between 2005 and 2019; n=547). The data of the two cohorts were compared, and predictors of BMD were analysed by linear regression analysis.
Results
Prevalence of osteoporosis (28.3% vs 48.4%; p<0.001) as well as osteoporotic peripheral fractures (11.5% vs 21%; p<0.001) and vertebral fractures (6.6% vs 10.9%; p=0.011) were significantly lower and treatment with biologicals (19.7% vs 5.0%; p<0.001) significantly more common and glucocorticoid use was significantly less common (p=0.005) in cohort 2. In RA patients with a disease duration of more than 2 years, BMD was significantly higher under treatment with biologicals (p<0.001) despite increased cumulative glucocorticoid dosages (p<0.001).
Conclusion
Our study showed a significant decline in osteoporosis and osteoporotic fractures in RA for 24 years. This positive effect is associated with the more frequent use of biologicals in the years between 2005 and 2019.
Keywords: Rheumatoid Arthritis, Osteoporosis, Bone Density, Glucocorticoids
WHAT IS ALREADY KNOWN ON THIS TOPIC
Rheumatoid arthritis (RA) is an important risk factor for osteoporosis and fractures. The treat-to-target strategy and the use of biologicals have significantly improved the treatment of RA. It is unclear whether these advances as well as targeted osteological diagnostics are also associated with a decrease in the prevalence of osteoporosis and fractures in clinical practice.
WHAT THIS STUDY ADDS
For the first time, the study shows a highly significant decrease in osteoporosis and fractures in RA over the course of 24 years. Early osteological assessment allows intervention at a time when the prevalence of osteoporosis and fractures is lower. In the case of long-standing RA, pretreatment with biologicals is associated with a reduced prevalence of osteoporosis, despite higher cumulative doses of glucocorticoids.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
In RA patients, osteological assessment should be carried out as early as possible. In addition, the use of biologicals should be considered as part of the treatment strategy in those patients with a high risk of osteoporosis.
Introduction
Osteoporosis is a chronic disease characterised by the loss of bone mineral density (BMD) and increased fracture risk and is one of the most important comorbidities in rheumatoid arthritis (RA). The incidence of systemic osteoporosis in patients with RA is about twice that of the general population in the same age group, the risk of femur fracture is 1.3 times higher, and RA patients are more than twice as likely to experience vertebral fractures, even in people who do not take glucocorticoids (GCs).1 Osteoporosis-related fractures are considerably associated with higher morbidity and mortality in RA patients, place an enormous medical and personal burden and take a significant economic toll.2 Therefore, management of bone health and reducing the risk of developing osteoporosis and fractures are of critical importance in RA.
Possible factors contributing to the high incidence of osteoporosis in RA include older age,3,11 low body mass index (BMI) or low weight,3,13 therapy with GCs in particular with high cumulative and daily doses,3,69 10 13 high disease activity,4 7 12 high extent of periarticular bone destruction,5 7 10 low functional status (disability),34 8,10 15 long disease duration,38,10 15 female gender7 8 and postmenopausal status.3 6 13 RA-specific risks for fractures encompass older age,316,21 chronic exposure to GC,3 16 18 19 21 22 low functional status (disability),3 17 18 21 22 prior fractures,16,1820 female gender,19 postmenopausal status,22 low BMD17 23 and falls.21
Addressing modifiable risk factors is essential to reduce osteoporosis and osteoporotic fractures in RA. This includes the avoidance of long-term or higher dose GC therapy as well as achieving adequate and sustained control of RA disease activity using non-GC medications such as biologic disease-modifying antirheumatic drugs (bDMARDs) and targeted synthetics (tsDMARDs).24,26 Further key approaches for early detection of osteoporosis in RA patients encompass regular BMD measurement, fracture risk assessment using tools such as the FRAX algorithm, and adequate and consistent osteoporosis treatment with bisphosphonates (BP), denosumab and osteoanabolic substances.
In the last 15 years, many recommendations such as the treat-to-target strategy27 and the regularly updated recommendations of the European League Against Rheumatism27 28 have improved the outcome of RA patients and also addressed bone health. In addition, the therapeutic armamentarium for RA has increased substantially over the last 25 years and it can be expected that the tremendous success in controlling inflammatory arthritis with modern antirheumatic treatment will also decrease RA-associated osteoporosis.
Therefore, our retrospective study evaluated possible changes in the prevalence of osteoporosis and osteoporotic fractures in RA patients from the time of the first osteological assessment with BMD measurement and collection of osteologically important data during the years 1996 and 2019 at our clinic. If changes were identified, we investigated the possible causes.
Methods
Study design and patients
We analysed the medical records data of 1.086 patients with RA who were admitted to our department during the years 1996 and 2019. The primary reason for the visit of patients in our centre was RA management, including discussion for biological use. Of special interest were the following parameters: age, disease duration of RA defined as the time interval between diagnosis of RA and first osteodensitometry, gender and postmenopausal status, BMI, GC therapy including cumulative GC dose, therapy with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and bDMARDs, pretreatment with BP, and prevalence of peripheral (pFx) and vertebral osteoporotic fractures (vFx). In addition, parameters of disease activity such as erythrocyte sedimentation rate (ESR) and C reactive protein (CRP) were determined. Serological status based on the detection of anti-citrullinated protein antibodies (ACPA) and rheumatoid factor (RF) was also included in the analyses. BMD values measured with dual X-ray absorptiometry (DXA) were assessed for total hip (BMD-H), femoral neck (BMD-FN) and lumbar spine BMD (BMD-LS). Based on the lowest T-score, the patients were classified into those with normal BMD (T-score higher than −1.0), osteopenia (T-score between −1 and −2.5) and osteoporosis (T-score lower than −2.5).
GC use was defined as pre-existing therapy that was still ongoing at the time of osteodensitometry. The cumulative glucocorticoid dose was determined as accurately as possible by means of detailed questioning of patients about onset, duration and daily doses and on the basis of the available medical documents.
Use of bDMARD was defined as at least 1 year of existing and continued therapy at the time of osteodensitometry.
With regard to fractures, all patients were interviewed in detail. The location of reported fractures was confirmed by X-rays. In addition, the patients received an X-ray examination of the thoracic and lumbar spine to detect vertebral fractures.
All osteodensitometric examinations were carried out using the DXA technique. The DXA device was changed twice during the study period. Between 1996 and June 2006, the HOLOGIC 4500A device (HOLOGIC, Waltham, USA) was used, from June 2006 to March 2017 the device Lunar Prodigy Advanced (GE Healthcare, Solingen, Germany), and from April 2017 the device Lunar Prodigy (GE Healthcare). By means of cross-calibration and the use of appropriate software by the manufacturer, the comparability of the measurement results was ensured over time.
According to the period of first osteological diagnosis, the patients were divided into those with investigations between 1996 and 2004 (cohort 1; n=539) and those with investigations between 2005 and 2019 (cohort 2; n=547). The division into both groups was made because after 2004, the therapy of RA in our centre had become significantly more effective, especially due to the increasing use of tumour necrosis factor (TNF) inhibitors, and the aims of the therapy had become more ambitious.
Statistical analysis
The data were compared between both cohorts in general and in different subgroups stratified by gender, menopausal status, GC therapy, BMI, disease duration and age. Furthermore, data between cohort 1 and cohort 2 and data between patients with and without osteoporosis were compared separately in patients with a disease duration of ≤2 years and those with disease duration >2 years. Statistical analyses were performed with the SPSS for Windows Statistical programme. We used the Mann-Whitney U test to compare metric and ordinal parameters of the different patient groups and the χ2 test to compare the nominally scaled parameters. For correlation analysis, we used the Spearman correlation coefficient.
Predictors of BMD-LS, BMD-FN and BMD-H in patients with a disease duration of ≤2 years and of those with a disease duration of >2 years were identified by multiple linear regression analysis using important factors known to be associated with BMD in RA (age, disease duration, BMI, gender, postmenopausal status and cumulative GC dose) as well as therapeutic parameters (pretreatment with bDMARDs and BP), and those of disease activity (ESR and CRP) and seropositivity (detection of ACPA and/or RF) as independent variables.
Multifactorial analysis in these two subgroups according to disease duration were repeated after exclusion of patients with BP pretreatment.
Results
Patient characteristics
The patients had a mean age of 59.8±12.9 years (18–91 years) and a mean disease duration of 9.0±10.2 years (0.1–60 years). 644 patients (59.3%) were postmenopausal women, 181 (16.7%) were premenopausal women and 261 (24%) were men. A total of 805 patients (74.1%) were receiving GC therapy.
Demographic, clinical and therapeutic parameters in the two time periods in the entire group
Demographic data as well as clinical and laboratory findings and medications in the two different cohorts are shown in table 1. Patients in cohort 2 were significantly older (p=0.002), had a higher BMI (p<0.001), a shorter disease duration or a shorter interval between the diagnosis of RA and the first bone density measurement (p<0.001) and a lower mean ESR (p=0.001) compared with cohort 1. In addition, cohort 2 encompassed fewer postmenopausal women (p=0.043) and more men (p=0.001). The percentage of patients with a disease duration of ≤2 years was highly significantly higher in cohort 2 (p<0.001).
Table 1. Frequency of osteoporosis and fractures, demographic data, clinical/laboratory findings, and therapy in cohort 1 (period 1996–2004) and cohort 2 (period 2005–2019) (entire RA group, n=1.086).
| Years 1996–2004cohort 1(n=539) | Years 2005–2019cohort 2(n=547) | P value | |
| Frequency of osteoporosis and fractures | |||
| Osteoporosis (%) | 48.4 | 28.3 | <0001 |
| Peripheral fractures (%) | 21 | 11.5 | <0.001 |
| Vertebral fractures (%) | 10.9 | 6.6 | 0.011 |
| BMD | |||
| BMD-LS (g/cm2) | 0.85±0.15 | 1.03±0.21 | <0.001 |
| BMD-FN (g/cm2) | 0.71±0.13 | 0.83±0.14 | <0.001 |
| BMD-H (g/cm2) | 0.83±0.16 | 0.93±0.17 | <0.001 |
| Demographic and clinical data | |||
| Age (years) | 58.3±13.1 | 61.3±12.6 | 0.002 |
| Postmenopausal women (%) | 62.3 | 56.3 | 0.043 |
| Premenopausal women (%) | 18.0 | 15.0 | ns |
| Men (%) | 19.7 | 28.3 | 0.001 |
| BMI (kg/m2) | 26.6±5.0 | 28.5±5.6 | <0.001 |
| Disease duration | 12.0±10.5 | 6.0±8.9 | <0.001 |
| Patients with disease duration ≤2 years (%) | 16.9 | 53.9 | <0.001 |
| ESR (mm/hour) | 36±27 | 30±25 | 0.001 |
| CRP (mg/L) | 29.9±38.1 | 28.8±36.3 | ns |
| Seropositivity (%) | 71.6 | 68.4 | ns |
| RA therapy and BP use | |||
| GC therapy (%) | 78 | 70 | 0.005 |
| Cumulative GC dose (g) | 15.5±20.4 | 12.8±25.6 | 0.021 |
| csDMARD (total) (%) | 85.2 | 59.7 | <0.001 |
| csDMARD without bDMARD (%) | 80.3 | 41.9 | <0.001 |
| Number of csDMARDS | 1.8 | 1.0 | <0.001 |
| bDMARDs (sequential treatment with csDMARDs and bDMARDs) (%) | 5.0 | 19.7 | <0.001 |
| BP pretreatment (%) | 13 | 14.7 | ns |
bDMARDsbiologic disease-modifying antirheumatic drugsBMIbody mass indexBPbisphosphonatesCRPC reactive proteincsDMARDSconventional synthetic disease-modifying antirheumatic drugsESRerythrocyte sedimentation rateGCglucocorticoidsnsnot significantRArheumatoid arthritis
The percentage of GC-treated patients (p=0.005) and the cumulative GC dose (p=0.021) were lower in cohort 2. The use of csDMARDs without biologicals and the number of csDMARDs per patient were almost twice as high in cohort 1 as in cohort 2 (p<0.001). In contrast, the percentage of patients receiving combined or sequential treatment with csDMARDs and bDMARDs was significantly higher in cohort 2 compared with cohort 1 (19.7% vs 5%; p<0.001). There was no difference in BP-pretreatment between the two cohorts (13% vs 14.7%).
Prevalence of osteoporosis, BMD values and osteoporotic peripheral/vertebral fractures in cohorts 1 and 2
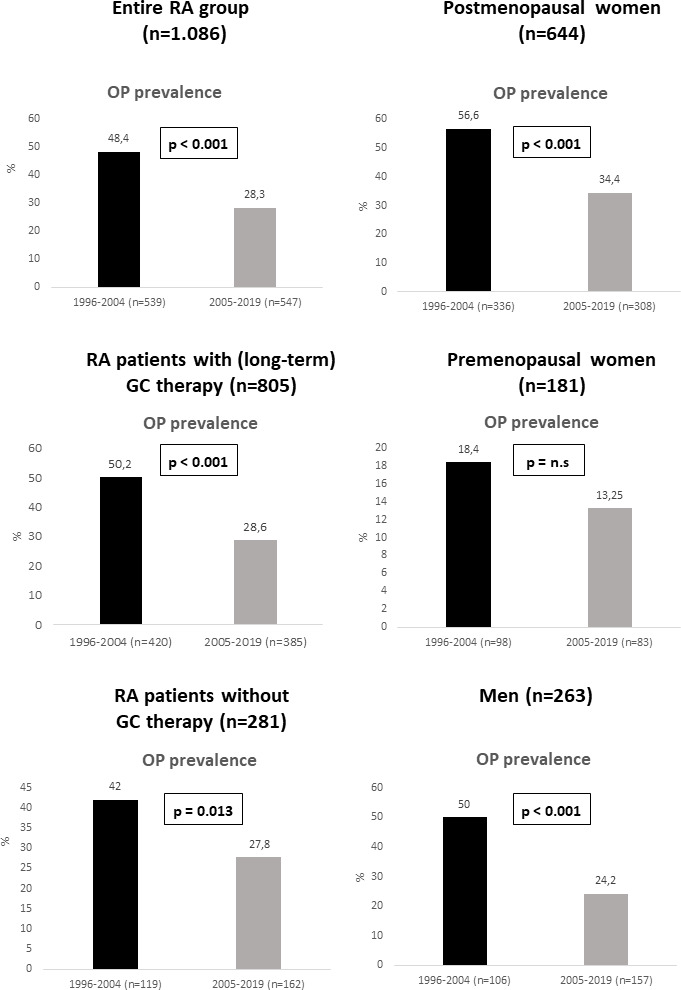
Frequency of osteoporosis in the entire cohort was significantly lower in cohort 2 (28.3%) compared with cohort 1 (48.4%; p<0.001) (figure 1). Accordingly, BMD values at all three skeletal sites were significantly higher in cohort 2 versus cohort 1 (p<0.001) (table 1). Furthermore, patients in cohort 2 had significantly fewer osteoporotic peripheral fractures (pFx) (11.5% vs 21%; p<0.001) and vertebral fractures (vFx) (6.6% vs 10.9%; p=0.011) versus cohort 1 (table 1).
Figure 1. Prevalence of osteoporosis (OP) in rheumatoid arthritis (RA) patients according to the time period of first osteological diagnosis, including subgroups stratified by glucocorticoid (GC) therapy, gender and menopausal status.
Subgroup analysis of osteoporosis and osteoporotic fractures
Prevalence of osteoporosis
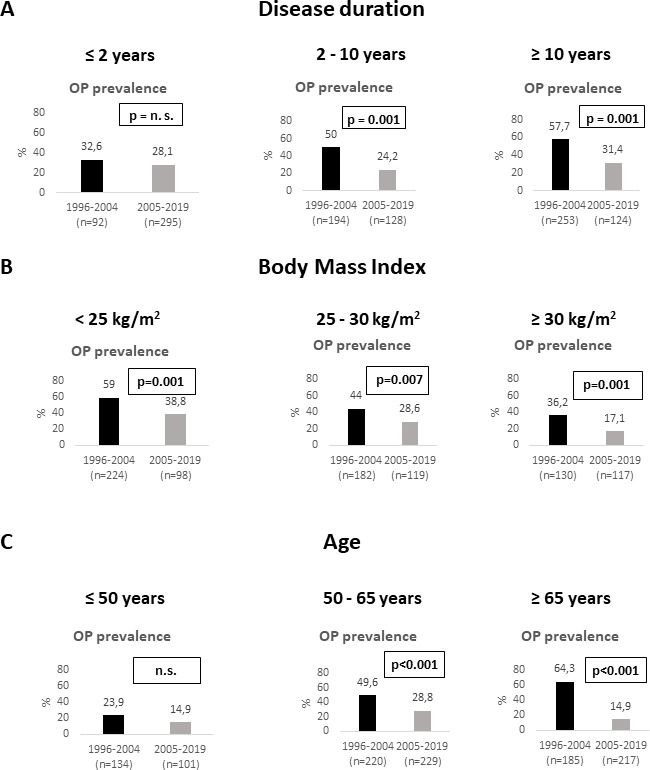
The frequency of osteoporosis in subgroups stratified by gender, menopausal status, GC therapy, disease duration, BMI and age is shown in figures1 2. Patients in cohort 2 were significantly less likely to develop osteoporosis compared with cohort 1 (p<0.001). This was true not only for the entire group, but also for the subgroups of patients treated with (p<0.001) and without GC (p=0.013), for postmenopausal women (p<0.001), and in men (p<0.001) as well as in patients with an RA disease duration of >2–10 years and >10 years (p=0.001). Furthermore, a lower frequency of osteoporosis was documented in cohort 2—patients with a BMI of <25 kg/m2 (p=0.001), 25 to <30 kg/m2 (p=0.007) and ≥30 kg/m2 (p=0.001), in patients aged over 50–65 years (p<0.001) and over 65 years (p<0.001). Furthermore, a lower frequency of osteoporosis in cohort 2 compared with cohort 1 was also observed in seropositive (28.2% vs 51%; p<0.001) and seronegative patients (23.6% vs 39.7%; p=0.003). Only in premenopausal women, patients with disease duration ≤2 years, and those aged ≤50 years no significant differences in the prevalence of osteoporosis between the two cohorts were found (figures1 2).
Figure 2. Prevalence of osteoporosis (OP) in rheumatoid arthritis patients according to period of first osteological diagnosis in subgroups stratified by disease duration (A), body mass index (B) and age (C).
Prevalence of pFx
pFx were significantly lower in the overall cohort 2 as well as in patients under GC therapy (13.2% vs 21.9%; p=0.001), in those without GC therapy (7.4% vs 17.6%; p=0.008), in postmenopausal women (15.9% vs 27.1%; p=0.001), in seropositive patients (12.5% vs 24.4%; p<0.001), in seronegative patients (8% vs 25.5%; p<0.001), in patients with a disease duration >2 to 10 years (11.7% vs 22.2%; p=0.017), in patients with a BMI<25 kg/m2 (5.1% vs 21.4%; p=0.001) and ≥30 kg/m2 (9.4% vs 23.1%; p=0.004) as well as in patients with an age of 50 to 65 years (12.2% vs 21.4%; p=0.009) and those older than 65 years (13.4% vs 27%; p=0.001).
Prevalence of vFx
The frequency of vFx in cohort 2 was lower in GC-treated patients (7% vs 11.9%; p=0.018), in postmenopausal women (7.8% vs 14.2%; p=0.009), in seronegative patients (5.6% vs 14.2%; p=0.011), in patients with a BMI of <25 kg/m2 (1% vs 11.2%; p=0.002), and in those aged over 65 years (9.2% vs 21.1%; p=0.001) compared with cohort 1.
Subgroup analysis in RA patients with disease duration of ≤2 years and those with disease duration of more than 2 years
Prevalence of osteoporosis and fractures in both RA-subgroups according to disease duration
The frequencies of osteoporosis (29.5% vs 43.1%; p<0.001), pFx (7.8% vs 21.1%; p<0.001) and vFx (6.2% vs 10.1%; p=0.028) were significantly lower in patients with a disease duration ≤2 years compared with those with longer disease duration. The frequency of patients with GC (58.4% vs 82.9%; p<0.001) and the cumulative GC dose (1.8±5.4 g vs 20.0±25.4 g; p<0.001) were significantly lower in patients with a disease duration ≤2 years.
Comparison between cohort 1 (1996–2004) and cohort 2 (2005–2019) in both RA subgroups according to disease duration
Frequency of osteoporosis and fractures, demographic and clinical data, RA therapy and BP pretreatment of cohort 1 and cohort 2 in the two RA subgroups according to disease duration are shown in table 2.
Table 2. Frequency of osteoporosis and fractures, demographic and clinical data, RA therapy and BP pretreatment of cohort 1 and cohort 2 in RA subgroups according to disease duration.
| Disease duration ≤2 years(n=386) | Disease duration >2 years(n=700) | |||||
| Years 1996–2004cohort 1(n=92) | Years 2005–2019cohort 2(n=294) | P value* | Years 1996–2004cohort 1(n=398) | Years 2005–2019cohort 2(n=302) | P value* | |
| Frequency of osteoporosis and fractures | ||||||
| Osteoporosis (%) | 33.0 | 28.6 | ns | 51.6 | 28.2 | <0.001 |
| Peripheral fractures (%) | 12.1 | 6.5 | ns | 22.8 | 17.5 | ns |
| Vertebral fractures (%) | 4.5 | 6.8 | ns | 12.3 | 6.3 | 0.013 |
| Demographic data | ||||||
| Age (years) | 58.1±15.2 | 62.4±12.5 | ns | 58.4±12.6 | 60.1±12.6 | ns |
| Disease duration | 0.86±0.63 | 0.28±1.01 | <0.001 | 14.3±10.1 | 12.8±9.3 | ns |
| BMI (kg/m2) | 27.7±5.0 | 28.3±5.8 | ns | 26.4±5.0 | 28.2±5.4 | <0.001 |
| Women/men (%) | 73.6/26.4 | 67.0/33.0 | ns | 81.7/18.3 | 76.6/23.4 | ns |
| Postmenopausal women (%) | 54.9 | 56.5 | ns | 63.8 | 56.4 | ns |
| Disease activity/serology | ||||||
| ESR (mm/hour) | 50±31 | 33±26 | <0.001 | 33±25 | 28±23 | 0.008 |
| CRP (mg/L) | 48.8±35.2 | 23.5±36.8 | <0.001 | 26.0±35.2 | 23.6±35.0 | 0.010 |
| Seropositivity (%) | 61.4 | 64.0 | ns | 73.7 | 74.0 | ns |
| RA therapy and BP use | ||||||
| GC therapy (%) | 50.5 | 50.9 | ns | 83.5 | 81.7 | ns |
| Cumulative GC dose (g) | 2.0±4.5 | 1.7±5.8 | ns | 18.3±20.4 | 23.6±32.1 | <0.001 |
| csDMARD (total) (%) | 40.7 | 34.1 | ns | 94.6 | 91.3 | ns |
| csDMARD without bDMARD (%) | 40.6 | 29.7 | 0.019 | 88.4 | 56.3 | <0.001 |
| bDMARD use (%) | 0 | 4.8 | 0.034 | 6.25 | 36.9 | <0.001 |
| BP pretreatment (%) | 2 | 9.2 | 0.009 | 15.4 | 21.4 | 0.044 |
cComparison between cohort 1 and cohort 2 of the same subgroup according to disease duration.
bDMARDsbiologic disease-modifying antirheumatic drugsBMIbody mass indexBPbisphosphonatesCRPC reactive proteincsDMARDSconventional synthetic disease-modifying antirheumatic drugsESRerythrocyte sedimentation rateGCglucocorticoidsnsnot significantRArheumatoid arthritis
The prevalences of osteoporosis and fractures were not different between cohort 1 (1996–2004) and cohort 2 (2005–2019) in RA with a disease duration of ≤2 years, despite a shorter disease duration in cohort 2 (p<0.001). In contrast, in patients with a disease duration of more than 2 years, no differences in disease duration were observed between cohort 1 and cohort 2, but the prevalences of osteoporosis (p<0.001) and of vertebral fractures (p<0.013) were significantly lower in cohort 2 compared with cohort 1. In patients with disease duration >2 years, BMI was significantly higher in cohort 2 (p<0.001).
In both RA subgroups due to disease duration, ESR (p<0.001 and p=0.008, respectively) and CRP (p<0.001 and p=0.010, respectively) were significantly lower in cohort 2 compared with cohort 1.
In patients with a disease duration of ≤2 years, higher rates of BP pretreatment (p=0.009) and of bDMARD use (p=0.034) were observed. However, the rate of pretreatment with bDMARD and BP was very low in cohorts 1 and 2.
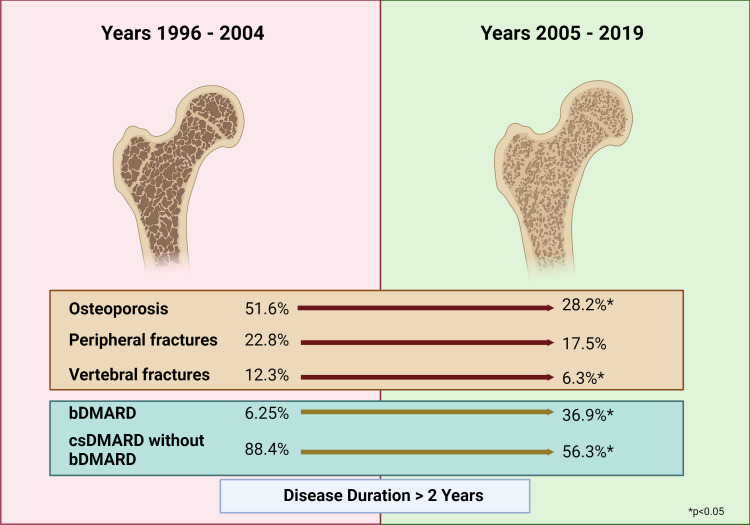
In the RA subgroup with a disease duration of more than 2 years, the percentage of GC users was not different between cohort 1 and cohort 2. However, the cumulative GC dose was higher in cohort 2 (p<0.001). Furthermore, the percentage of patients with bDMARD therapy was highly significantly higher in cohort 2 (36.9 %) in comparison to cohort 1 (6.25 %; p<0.001) and the percentage of patients treated with csDMARDs without bDMARD therapy was significantly lower in cohort 2 (p<0.001) (figure 3). Patients in cohort 2 were treated with BP slightly more frequently (p=0.044).
Figure 3. The reduction of osteoporosis, peripheral fractures and vertebral fractures based on the use of biologic disease-modifying antirheumatic drugs (bDMARDs) (csDMARD, conventional synthetic disease-modifying antirheumatic drugs). Created with BioRender.com.
In patients with more than 2 years of disease, BMD at all sites was significantly higher in the bDMARD pretreatment group compared with those not treated with biologicals (p<0.001) despite a higher cumulative GC dosage (p<0.001) (table 3A). This result was confirmed after exclusion of patients with BP (table 3B).
Table 3. BMD, daily GC dose (GD/day) and cumulative GC dose (cGCD) in patients with and without pretreatment with bDMARDS with disease duration >2 years ((A) all patients; (B) patients without bisphosphonates).
| BMD-LS (g/cm2) | BMD-SH (g/cm2) | BMD-H (g/cm2) | GD/day (mg) | cGCD (g) | |
| (A)Allpatients | |||||
| No pretreatment with bDMARDS(n=578) | 0.89±0.18 | 0.72±0.15 | 0.84±0.17 | 5.2±6.7 | 18.7±25.9 |
| Pretreatment with bDMARDS(n=122) | 1.00±0.21 | 0.80±0.13 | 0.90±0.14 | 5.4±3.5 | 26.8±21.0 |
| P value | <0.001 | <0.001 | <0.001 | ns. | <0.001 |
| (B)Patientswithout bisphosphonates | |||||
| No pretreatment with bDMARDS(n=487) | 0.90±0.19 | 0.74±0.15 | 0.85±0.17 | 5.3±7.2 | 17.1±26.0 |
| Pretreatment with bDMARDS(n=91) | 1.03±0.22 | 0.82±0.13 | 0.92±0.14 | 5.3±3.4 | 25.5±22.9 |
| P value | <0.001 | <0.001 | <0.001 | ns. | <0.001 |
bDMARDs, biologic disease-modifying antirheumatic drugs; BMD, bone mineral density; BMD-FN, BMD femoral neck; BMD-H, BMD total hip; BMD-LS, BMS lumbar spine; cGCD, cumulative glucocorticoid dose; GD/day, glucocorticoid dose per day; ns, not significant
Comparison between patients with and without osteoporosis in both RA subgroups according to disease duration
Differences between patients with and without osteoporosis in RA in the two RA subgroups according to disease duration are shown in table 4. In both subgroups of RA patients, osteoporotic RA patients were older (p<0.001) and had a lower BMI (p<0.001) compared with patients without osteoporosis. In addition, osteoporosis was also more common in postmenopausal women (p<0.001) and pretreatment with BP was more frequent in patients with osteoporosis (p<0.001).
Table 4. Demographic and clinical data, RA therapy, BP pretreatment and prevalence of fractures in patients without and with osteoporosis in RA subgroups according to disease duration.
| Disease duration ≤2 years(n=386) | Disease duration >2 years(n=700) | |||||
| Without osteoporosis (n=272) | With osteoporosis(n=114) | P value* | Without osteoporosis (n=398) | With osteoporosis(n=302) | P value* | |
| Demographic data | ||||||
| Age (years) | 59.6±13.5 | 65.6±12.0 | <0.001 | 56.5±13.2 | 62.3±10.9 | <0.001 |
| Disease duration (years) | 0.38±0.69 | 0.51±1.58 | ns | 12.2±8.6 | 15.7±10.9 | <0.001 |
| BMI (kg/m2) | 28.8±5.5 | 26.3±5.0 | <0.001 | 27.8±5.3 | 25.7±4.9 | <0.001 |
| Women/men (%) | 66.8/33.2 | 72.8/27.2 | ns | 79.6/20.4 | 80.1/19.9 | ns |
| Postmenopausal women (%) | 50.5 | 69.3 | <0.001 | 53.0 | 71.9 | <0.001 |
| Disease activity/serology | ||||||
| ESR (mm/hour) | 36±27 | 40±31 | ns | 28±24 | 34±26 | 0.002 |
| CRP (mg/L) | 34.4±38.6 | 42.1±42.3 | ns | 23.2±34.0 | 34.5±25.8 | <0.001 |
| Seropositivity (%) | 61.8 | 67.6 | ns | 71 | 77.7 | ns |
| RA therapy and BP use | ||||||
| GC therapy (%) | 59 | 57 | ns | 81.4 | 84.6 | ns |
| Cumulative GC dose (g) | 1.7±3.9 | 2.1±7.8 | ns | 17.5±25.4 | 23.1±25.0 | 0.003 |
| Therapy with csDMARDs (%) | 34.7 | 38.1 | ns | 91.9 | 96 | 0.016 |
| csDMARDs without subsequent use of bDMARDs (%) | 31.0 | 35.4 | n.s | 71.1 | 84.4 | <0.001 |
| bDMARD use (%) | 3.4 | 3.5 | ns | 21.4 | 11.9 | <0.001 |
| BP pretreatment (%) | 3.0 | 17.5 | <0.001 | 11.3 | 25.8 | <0.001 |
| Fractures | ||||||
| Peripheral fractures (%) | 6.3 | 11.4 | ns | 17.3 | 25.5 | 0.008 |
| Vertebral fractures (%) | 5.9 | 7.0 | ns | 5.0 | 16.9 | <0.001 |
cComparison between patients with and without osteoporosis of the same subgroup according to disease duration.
bDMARDsbiologic disease-modifying antirheumatic drugsBMIbody mass indexBPbisphosphonatesCRPC reactive proteincsDMARDSconventional synthetic disease-modifying antirheumatic drugsESRerythrocyte sedimentation rateGCglucocorticoidsnsnot significantRArheumatoid arthritis
In the subgroup with disease duration <2 years, no differences in paraclinical parameters of inflammatory activity and seropositivity and in RA treatment with regard to the use of GC, csDMARD and bDMARD and for the cumulative GC dose between patients with and without osteoporosis have been found.
In patients with a disease duration of more than 2 years, ESR (p=0.002) and CRP (p<0.001) were significantly higher in osteoporotic patients. Furthermore, patients with osteoporosis showed a higher cumulative GC dose (p=0.003), a highly significantly higher percentage of treatment with csDMARD without subsequent use of bDMARDs (p<0.001) and a highly significantly lower percentage of bDMARD use (p<0.001). Disease duration was significantly longer in osteoporotic patients in this subgroup (p<0.001).
While the fracture prevalence in RA with a disease duration of <2 years did not differ between patients with and without osteoporosis, osteoporotic patients in the subgroup with a disease duration of more than 2 years had significantly more peripheral (p=0.008) and vertebral fractures (p<0.001) compared with patients without osteoporosis.
Predictors of BMD-LS, BMD-FN and BMD-H in both RA subgroups according to disease duration
In the subgroup with a disease duration of ≤2 years, BMD was primarily associated with demographic parameters, showing a positive association at all sites with BMI (p=0.002 to p<0.001) (table 5). BMD-LS was negatively associated with postmenopausal status (p<0.001); older age (both p<0.001) and female gender (p=0.011 and p=0.001) were negatively associated with BMD-FN and BMD-H. In addition, BMD-LS showed a weak positive association with bDMARD use (p=0.034) and a negative association with seropositivity (p=0.031).
Table 5. Predictors of BMD-LS, BMD-FN and BMD-H (independent variables used: age, female gender, postmenopausal status, BMI, disease duration, cumulative GC dose, pretreatment with bDMARD, pretreatment with BP, ESR, CRP, seropositivity) in RA subgroups according to disease duration ((A) all patients; (B) patients without bisphosphonates); multiple linear regression analysis.
| (A) | |||||||
| RA with disease duration ≤2 years (n=386) | RA with disease duration >2 years (n=700) | ||||||
| Site | BMD-LS | BMD-FN | BMD-H | Site | BMD-LS | BMD-FN | BMD-H |
| Age | ns | <0.001 | <0.001 | Age | 0.034 | <0.001 | <0.001 |
| Female | ns | 0.011 | 0.001 | Female | 0.018 | ns | ns |
| PMP | <0.001 | ns | ns | PMP | <0.001 | <0.001 | <0.001 |
| BMI | 0.002 | <0.001 | <0.001 | BMI | <0.001 | <0.001 | <0.001 |
| DD | ns | ns | ns | DD | ns | ns | ns |
| cGCD | ns | ns | ns | cGCD | 0.006 | <0.001 | <0.001 |
| bDMARD | 0.034 | ns | ns | bDMARD | <0.001 | 0.009 | 0.008 |
| BP | ns | ns | ns | BP | 0.027 | ns | 0.016 |
| ESR | ns | ns | ns | ESR | 0.024 | ns | 0.004 |
| CRP | ns | ns | ns | CRP | ns | ns | ns |
| Seropositivity | 0.031 | ns | ns | Seropositivity | ns | ns | ns |
| R2 | 0.166 | 0.224 | 0.279 | R2 | 0.210 | 0.309 | 0.401 |
| P value | <0.001 | <0.001 | <0.001 | P value | <0.001 | <0.001 | <0.001 |
| (B) | |||||||
| RA with disease duration ≤2 years (n=357) | RA with disease duration >2 years (n=577) | ||||||
| Site | BMD-LS | BMD-FN | BMD-H | Site | BMD-LS | BMD-FN | BMD-H |
| Age | 0.033 | <0.001 | <0.001 | Age | 0.060 | <0.001 | <0.001 |
| Female | <0.001 | 0.004 | <0.001 | Female | 0.027 | ns | ns |
| PMP | ns | ns | ns | PMP | 0.001 | <0.001 | <0.001 |
| BMI | 0.011 | <0.001 | <0.001 | BMI | <0.001 | <0.001 | <0.001 |
| DD | 0.018 | ns | ns | DD | ns | 0.028 | ns |
| cGCD | ns | ns | ns | cGCD | 0.021 | 0.020 | <0.001 |
| bDMARD | 0.003 | ns | ns | bDMARD | 0.001 | 0.008 | 0.008 |
| ESR | ns | ns | ns | ESR | 0.007 | ns | 0.012 |
| CRP | ns | ns | ns | CRP | ns | 0.011 | ns |
| Seropositivity | 0.006 | ns | ns | Seropositivity | ns | ns | ns |
| R2 | 0.210 | 0.235 | 0.278 | R2 | 0.202 | 0.364 | 0.428 |
| P value | <0.001 | <0.001 | <0.001 | P value | <0.001 | <0.001 | <0.001 |
Positive associations with BMD are highlighted in bold font.
bDMARDs, biologic disease-modifying antirheumatic drugs; BMD, bone mineral density; BMD-FN, BMD femoral neck; BMD-H, BMD total hip; BMD-LS, BMD lumbar spine; BMI, body mass index; BP, bisphosphonates; cGCD, cumulative glucocorticoid dose; CRP, C reactive protein; DD, disease duration; ESR, erythrocyte sedimentation rate; Female, female gender; ns, not significantPMP, postmenopausal statusRArheumatoid arthritis
Unlike in the cohort with a known RA diagnosis of ≤2 years, in patients with a disease duration of more than 2 years, BMD was not only associated with demographic parameters but was also with RA therapy. Thus, older age (p=0.034 to p<0.001), postmenopausal status (p<0.001) and high cumulative GC dosages (p=0.006 to p<0.001) were the most important negative predictors of BMD at all sites. A higher BMI (p<0.001) and bDMARD therapy (p=0.009 to p<0.001) turned out to be the main positive predictors of BMD at all sites. In addition, BMD-LS and BMD-H were negatively associated with ESR (p=0.024 and p=0.004, respectively). BP pretreatment was negatively associated with BMD-LS (p=0.027) and BMD-H (p=0.016) and female gender was negatively associated with BMD-LS (p=0.018).
The main results of this analysis could also be confirmed after exclusion of patients with BP pretreatment.
Relationship of parameters of inflammation to bDMARD use and to BMD
In the entire group, ESR (28±25 mm/hour vs 34±26 mm/hour; p=0.003) was lower in bDMARD treated patients compared with those without bDMARD. CRP (28.9±41.7 mg/L vs 29.2±36.6 mg/L) and DAS28 (4.6 vs 4.5) were not different between patients with and without bDMARDs. ESR was also lower in bDMARD-treated patients in the subgroup with a disease duration of more than 2 years (p=0.009). CRP and DAS28 were not different according to bDMARD use in the two patient subgroups according to disease duration. In the entire cohort as well as in patients with a disease duration of more than 2 years, patients with osteoporosis showed significantly higher values for ESR (p=0.005 and p=0.002, respectively) and CRP (p=0.014 and p<0.001, respectively) compared with those without osteoporosis, and BMD at all sites showed a negative correlation with ESR (p=0.007 to p<0.001) and CRP (p=0.19 to p<0.001). DAS28 was not related to BMD.
Discussion
The main result of our study is the significant reduction of osteoporosis and osteoporotic pFx and vFx in RA patients in the period between 2005 and 2019 (cohort 2), compared with the years 1996–2004 (cohort 1). Potential causes for these findings are discussed in the following.
Cohort 1 and cohort 2 differ in some demographic and clinical parameters as well as in therapy across the group. Patients in cohort 2 have a shorter duration of disease, a higher BMI, a lower proportion of postmenopausal women, a lower proportion of patients with GC and a lower cumulative GC dose compared with cohort 1 (table 1). These differences could contribute to the higher frequency of osteoporosis in this group, because a low BMI,3,13 long RA disease duration,38,10 15 postmenopausal status3 6 13 and GC therapy, especially in high cumulative and daily dosages,3,69 10 13 are known risk factors for osteoporosis in RA patients.
However, the significance of each risk factor must be considered in the context of the various subgroup analyses. In view of the importance of disease duration and cumulative GC dosages for the lower prevalence of osteoporosis and fractures in cohort 2 compared with cohort 1, it is essential to differentiate the findings in the two subgroups according to disease duration (table 2).
In RA patients with a disease duration of ≤2 years, we observed no significant differences in the prevalence of osteoporosis and fractures between cohorts 1 and 2, despite the shorter duration of the disease in cohort 2. In contrast, in patients with a disease duration of more than 2 years, the prevalence of osteoporosis and vertebral fractures is significantly lower in cohort 2, although there were no differences in disease duration between these two cohorts. On the other hand, patients with a disease duration of ≤2 years show a significantly lower prevalence of osteoporosis and fractures compared with patients with a longer disease duration. These results suggest that two factors are very important for the lower prevalence of osteoporosis and fractures in cohort 2: the significantly higher proportion of patients with a disease duration of up to 2 years with correspondingly lower osteoporosis and fracture prevalence and the lower prevalence of osteoporosis and fractures in patients with longer disease duration, no matter how long patients have been suffering from RA. Furthermore, multivariate analysis showed no clear association between long disease duration and BMD (table 5). These findings contradict the notion that a shorter duration of RA could be a significant cause of the reduced incidence of osteoporosis in the years 2005–2019 in patients with a disease duration of more than 2 years.
No association was found between BMD or osteoporosis and GC therapy or cumulative GC dose in patients diagnosed with RA for ≤2 years (tables2 5). However, higher cumulative GC doses were significantly negatively associated with BMD at all sites (table 5) and were associated with osteoporosis in patients with disease duration >2 years (table 4). Due to the high proportion of patients with a disease duration of ≤2 years with a relatively low proportion of patients with GC and a low cumulative GC dose, the percentage of patients with GC and cumulative GC doses are lower in cohort 2 compared with cohort 1 in the entire group (tables1 2). Nevertheless, in patients with a disease duration of more than 2 years the percentage of GC use was not different between cohort 1 and cohort 2 and the cumulative GC dose was significantly higher in cohort 2 (table 2). Therefore, the lower prevalence of osteoporosis in cohort 2 in patients with a disease duration of more than 2 years cannot be explained by a more sparing use of GC.
Concerning predictors of BMD, we conducted a multivariate analysis and identified a strong positive correlation between a high BMI and BMD at all sites in the two subgroups stratified by disease duration (table 5). Postmenopausal status was a main negative predictor of BMD at all sites in patients with a disease duration of >2 years. On the other hand, osteoporosis was significantly lower in cohort 2 compared with cohort 1 in all subgroups stratified by BMI and in postmenopausal women and men with RA (figures1 2). Therefore, a higher BMI and the lower percentage of postmenopausal women do not provide a sufficient explanation for the lower prevalence of osteoporosis found in cohort 2.
Although advanced age is one of the key risk factors for osteoporosis3,11 and osteoporotic fractures316,21 in RA, patients of cohort 2 had a lower prevalence of osteoporosis and associated fractures compared with cohort 1 despite an older age. Furthermore, the frequency of osteoporosis and pFx and vFx was significantly reduced in cohort 2 in patients older than 50 years and especially in those over 65 years, but not in those ≤50 years.
In summary, the high proportion of patients with a disease duration of ≤2 years with a correspondingly lower proportion of patients with osteoporosis and fractures as well as the lower rate of GC use and lower cumulative GC dosages in cohort 2 certainly contributed to the lower prevalence of osteoporosis and fractures in the overall group. However, differences in disease duration, demographic factors, GC use and cumulative GC dose do sufficiently explain the differences in the prevalence of osteoporosis and vertebral fractures between cohort 1 and cohort 2 in the subgroup with disease duration of more than 2 years.
With regard to therapy, we observed the highest significance between the two time periods for the use of biologicals (~5% between 1996 and 2004; ~20% between 2005 and 2019; p<0.001). These findings could be confirmed in patients with a disease duration of more than 2 years. In addition, BMD at all sites was significantly higher in patients on bDMARD therapy compared with those without biologicals, despite significantly higher cumulative GC doses (table 3). Osteoporosis also occurred less frequently in the bDMARD-treated group of patients with a disease duration of more than 2 years (table 4). Furthermore, bDMARD therapy was an important positive predictor of BMD at all sites in patients with a disease duration of more than 2 years (table 5). These results suggest that more frequent use of bDMARDs in cohort 2 is a very important factor for the reduced prevalence of osteoporosis compared with cohort 1 in patients with a disease duration of more than 2 years.
These results suggest a protective effect of biologicals regarding systemic bone loss in RA. Such a positive effect with stabilisation or increase of bone density in RA has been shown in a number of studies for TNF-alpha inhibitors (TNFi),25 26 29 30 the anti-interleukin (IL)-6R antibody tocilizumab,31,33 inhibition of costimulation between antigen-presenting cell and T-cell with abatacept34 and B-cell depletion with rituximab.35 In some studies, bDMARDs were compared with csDMARDs regarding bone density loss in RA.14 29 30 36 Two studies observed a stabilisation or a BMD-increase in the lumbar spine as well as the hip and femoral neck under therapy with infliximab, while BMD decreased in the control group (prednisone and MTX/MTX alone).29 30 In addition, data from the UK Biobank revealed an association of low BMD under GC- and csDMARD-therapy, but not with bDMARDs.14 Furthermore, long-term therapy with biologicals or JAK-inhibitors has shown protective effects on bone loss for patients with RA: after 3 years, BMD remained stable at the femoral neck, hip and lumbar vertebra, whereas BMD decreased significantly in csDMARD-treated participants at corresponding sites.36 These data are in accordance with our findings and suggest protective effects of bDMARDs against generalised osteoporosis in patients with RA and no or a negative impact under csDMARDs.
It remains unclear whether this beneficial effect of bDMARDs compared with csDMARDs is due to a greater suppression of disease activity and GC sparing and/or a more specific influence on pathological bone turnover in RA. As high disease activity is a risk factor for bone loss in RA,4 7 12 reducing systemic disease activity may be a cause of the bone protective effect of bDMARDs. The finding of a lower ESR in bDMARD-treated patients and the negative correlation of the parameters of systemic inflammation (ESR and CRP) with BMD in our study may suggest that the protective effect of bDMARDs on the bone is mediated by suppression of inflammation. On the other hand, infliximab, tocilizumab and rituximab showed a protective effect on joint-distant bones which was independent of the effect on disease activity or DAS28.30 31 35 Furthermore, a better effect on BMD-SH was observed with abatacept compared with other biologicals despite a lower reduction in DAS28.34 This suggests that the protective effect of bDMARDs on systemic bone is, at least in part, independent of suppression of inflammatory activity and is mediated by important regulators of bone metabolism.
Immune cells play a major role in the multifactorial pathogenesis of osteoporosis in RA, expressing RANKL (receptor activator of nuclear factor kappa B ligand) and thus activating osteoclasts.25 26 In addition, pro-inflammatory cytokines such as TNF-alpha, IL-1, IL-6 and IL-17 are known to promote osteoclasts differentiation and activation. Furthermore, patients with RA show increased levels of serum Dickkopf-1 (Dkk-1), a soluble inhibitor of the Wnt signalling pathway, leading to the inhibition of anabolic bone processes. Several studies have compared bone turnover markers in RA patients treated with biologicals or placebo and have consistently shown a pattern of increased bone formation and decreased bone resorption for cytokine antagonists such as TNFi,25 26 tocilizumab32 33 37 38 and for rituximab.35 Furthermore, a decrease in RANKL and Dkk-1 was observed under therapy with TNFi and tocilizumab.25 26 These data suggest that bDMARDs may be able to restore a more physiological bone remodelling process in RA.
Notably, we observed in our study that BMD was significantly higher in patients treated with bDMARDs than in those not treated with bDMARDs, despite a significantly higher cumulative dose of GC. This finding suggests that the effects of GC on bone may be partially antagonised by bDMARD therapy. Interestingly, there is accumulating evidence that at least some of the negative effects of GC on the skeleton may be due to increased osteoclastogenesis as a result of the upregulation of RANKL and the inhibition of the Wnt signalling pathway,39 which can in principle be countered by biologicals.26
Finally, the results of our study also revealed a discrepancy between the percentage of patients with RA taking GC who should have received adequate osteoporosis treatment with BP and the percentage of those who did. This gap has also been reported in other studies.40
Our study has some limitations. Due to the design as a retrospective cross-sectional study and the fact that many patients presented to our clinic for the first time, no information about long-term disease activity could be obtained. DAS28 was only collected in cohort 2. The influence of disease activity on the results can therefore only be assessed to a limited extent. Exact data on the extent of periarticular bone destruction at the time of osteodensitometry and on the patients’ functional limitations could not be systematically incorporated into the analysis. Therefore, an overall more severe course of the disease in cohort 1 cannot be definitively ruled out. However, because the course of the disease is largely determined by the therapy, the results of the study suggest that better management of RA, including a treat-to-target strategy and the more frequent use of biologicals, is important for the better bone health of our RA patients with longer disease duration in the period 2005–2019.
Conclusion
The results of our study have shown a significant reduction of osteoporosis and osteoporotic fractures in RA patients treated between the years 2005 and 2019, compared with those between 1996 and 2004. The decrease in the prevalence of osteoporosis is associated with a significantly higher proportion of patients having an osteological assessment within the first 2 years of RA, lower cumulative GC dosages and more frequent use of biologicals in patients with longer disease duration (figure 3). Furthermore, our data suggest a partial antagonisation of negative GC effects on bone by bDMARDs.
Acknowledgements
This study also reflects the dramatic changes in therapeutic options in rheumatology in the first author's clinical career spanning more than 40 years at the University of Jena, Germany.
Footnotes
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Data availability free text: The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Patient consent for publication: Consent obtained directly from patient(s).
Ethics approval: This study involves human participants and the study design was approved by the Ethical Committee of the University Clinic of Jena, Germany (registration number 2020-1731-data). Participants gave informed consent to participate in the study before taking part.
Provenance and peer review: Not commissioned; externally peer reviewed.
Contributor Information
Peter Oelzner, Email: peter.oelzner@gmail.com.
Paul-Heinrich Mueller, Email: info@schwarzatalambulatorium.de.
Tobias Hoffmann, Email: tobias.hoffmann@med.uni-jena.de.
Antje Schwabe, Email: info@antje-schwabe.de.
Gabriele Lehmann, Email: gabriele.lehmann@med.uni-jena.de.
Thorsten Eidner, Email: thorsten.eidner@med.uni-jena.de.
Gunter Wolf, Email: gunter.wolf@med.uni-jena.de.
Alexander Pfeil, Email: alexander.pfeil@med.uni-jena.de.
Data availability statement
Data are available upon reasonable request.
References
- 1.Tanaka Y. Managing Osteoporosis and Joint Damage in Patients with Rheumatoid Arthritis: An Overview. J Clin Med. 2021;10:1241. doi: 10.3390/jcm10061241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wysham KD, Baker JF, Shoback DM. Osteoporosis and fractures in rheumatoid arthritis. Curr Opin Rheumatol. 2021;33:270–6. doi: 10.1097/BOR.0000000000000789. [DOI] [PubMed] [Google Scholar]
- 3.Sinigaglia L, Nervetti A, Mela Q, et al. A multicenter cross sectional study on bone mineral density in rheumatoid arthritis. Italian Study Group on Bone Mass in Rheumatoid Arthritis. J Rheumatol. 2000;27:2582–9. [PubMed] [Google Scholar]
- 4.Haugeberg G, Ørstavik RE, Uhlig T, et al. Clinical decision rules in rheumatoid arthritis: do they identify patients at high risk for osteoporosis? Testing clinical criteria in a population based cohort of patients with rheumatoid arthritis recruited from the Oslo Rheumatoid Arthritis Register. Ann Rheum Dis. 2002;61:1085–9. doi: 10.1136/ard.61.12.1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lodder MC, Haugeberg G, Lems WF, et al. Radiographic damage associated with low bone mineral density and vertebral deformities in rheumatoid arthritis: the Oslo-Truro-Amsterdam (OSTRA) collaborative study. Arthritis Rheum. 2003;49:209–15. doi: 10.1002/art.10996. [DOI] [PubMed] [Google Scholar]
- 6.Oelzner P, Schwabe A, Lehmann G, et al. Significance of risk factors for osteoporosis is dependent on gender and menopause in rheumatoid arthritis. Rheumatol Int. 2008;28:1143–50. doi: 10.1007/s00296-008-0576-x. [DOI] [PubMed] [Google Scholar]
- 7.Hauser B, Riches PL, Wilson JF, et al. Prevalence and clinical prediction of osteoporosis in a contemporary cohort of patients with rheumatoid arthritis. Rheumatology (Oxford) 2014;53:1759–66. doi: 10.1093/rheumatology/keu162. [DOI] [PubMed] [Google Scholar]
- 8.Takahashi K, Setoguchi T, Tawaratsumida H, et al. Risk of low bone mineral density in patients with rheumatoid arthritis treated with biologics. BMC Musculoskelet Disord. 2015;16:269. doi: 10.1186/s12891-015-0732-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee J-H, Sung Y-K, Choi C-B, et al. The frequency of and risk factors for osteoporosis in Korean patients with rheumatoid arthritis. BMC Musculoskelet Disord. 2016;17:98. doi: 10.1186/s12891-016-0952-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tong J-J, Xu S-Q, Zong H-X, et al. Prevalence and risk factors associated with vertebral osteoporotic fractures in patients with rheumatoid arthritis. Clin Rheumatol. 2020;39:357–64. doi: 10.1007/s10067-019-04787-9. [DOI] [PubMed] [Google Scholar]
- 11.Hu Z, Zhang L, Lin Z, et al. Prevalence and risk factors for bone loss in rheumatoid arthritis patients from South China: modeled by three methods. BMC Musculoskelet Disord. 2021;22:534. doi: 10.1186/s12891-021-04403-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kweon S-M, Sohn DH, Park J-H, et al. Male patients with rheumatoid arthritis have an increased risk of osteoporosis: Frequency and risk factors. Medicine (Balt) 2018;97:e11122. doi: 10.1097/MD.0000000000011122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Theander L, Willim M, Nilsson JÅ, et al. Changes in bone mineral density over 10 years in patients with early rheumatoid arthritis. RMD Open. 2020;6:e001142. doi: 10.1136/rmdopen-2019-001142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Clynes MA, Jameson K, Prieto-Alhambra D, et al. Impact of Rheumatoid Arthritis and Its Management on Falls, Fracture and Bone Mineral Density in UK Biobank. Front Endocrinol (Lausanne) 2019;10:817. doi: 10.3389/fendo.2019.00817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Abdulkhaliq A, Cheikh M, Almuntashri F, et al. A Comparison of Demographics, Disease Activity, Disability, and Treatment Among Rheumatoid Arthritis Patients with and without Osteoporosis. Open Access Rheumatol. 2021;13:275–83. doi: 10.2147/OARRR.S318810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ørstavik RE, Haugeberg G, Uhlig T, et al. Vertebral deformities in 229 female patients with rheumatoid arthritis: associations with clinical variables and bone mineral density. Arthritis Rheum. 2003;49:355–60. doi: 10.1002/art.11118. [DOI] [PubMed] [Google Scholar]
- 17.Orstavik RE, Haugeberg G, Uhlig T, et al. Incidence of vertebral deformities in 255 female rheumatoid arthritis patients measured by morphometric X-ray absorptiometry. Osteoporos Int. 2005;16:35–42. doi: 10.1007/s00198-004-1631-4. [DOI] [PubMed] [Google Scholar]
- 18.Kim D, Cho S-K, Choi C-B, et al. Incidence and risk factors of fractures in patients with rheumatoid arthritis: an Asian prospective cohort study. Rheumatol Int. 2016;36:1205–14. doi: 10.1007/s00296-016-3453-z. [DOI] [PubMed] [Google Scholar]
- 19.Choi ST, Kwon S-R, Jung J-Y, et al. Prevalence and Fracture Risk of Osteoporosis in Patients with Rheumatoid Arthritis: A Multicenter Comparative Study of the FRAX and WHO Criteria. JCM. 2018;7:507. doi: 10.3390/jcm7120507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pierini FS, Brom M, Scolnik M, et al. Osteoporotic fractures in rheumatoid arthritis patients in Argentina: a matched retrospective cohort study. Adv Rheumatol . 2021;61:21. doi: 10.1186/s42358-021-00179-3. [DOI] [PubMed] [Google Scholar]
- 21.Guañabens N, Olmos JM, Hernández JL, et al. Vertebral fractures are increased in rheumatoid arthritis despite recent therapeutic advances: a case-control study. Osteoporos Int. 2021;32:1333–42. doi: 10.1007/s00198-021-05824-7. [DOI] [PubMed] [Google Scholar]
- 22.Coulson KA, Reed G, Gilliam BE, et al. Factors influencing fracture risk, T score, and management of osteoporosis in patients with rheumatoid arthritis in the Consortium of Rheumatology Researchers of North America (CORRONA) registry. J Clin Rheumatol. 2009;15:155–60. doi: 10.1097/RHU.0b013e3181a5679d. [DOI] [PubMed] [Google Scholar]
- 23.Ørstavik RE, Haugeberg G, Mowinckel P, et al. Vertebral deformities in rheumatoid arthritis: a comparison with population-based controls. Arch Intern Med. 2004;164:420–5. doi: 10.1001/archinte.164.4.420. [DOI] [PubMed] [Google Scholar]
- 24.Haugeberg G, Helgetveit KB, Førre Ø, et al. Generalized bone loss in early rheumatoid arthritis patients followed for ten years in the biologic treatment era. BMC Musculoskelet Disord. 2014;15:289. doi: 10.1186/1471-2474-15-289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zerbini CAF, Clark P, Mendez-Sanchez L, et al. Biologic therapies and bone loss in rheumatoid arthritis. Osteoporos Int. 2017;28:429–46. doi: 10.1007/s00198-016-3769-2. [DOI] [PubMed] [Google Scholar]
- 26.Orsolini G, Fassio A, Rossini M, et al. Effects of biological and targeted synthetic DMARDs on bone loss in rheumatoid arthritis. Pharmacol Res. 2019;147:104354. doi: 10.1016/j.phrs.2019.104354. [DOI] [PubMed] [Google Scholar]
- 27.Smolen JS, Aletaha D, Bijlsma JWJ, et al. Treating rheumatoid arthritis to target: recommendations of an international task force. Ann Rheum Dis. 2010;69:631–7. doi: 10.1136/ard.2009.123919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smolen JS, Landewé RBM, Bijlsma JWJ, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann Rheum Dis. 2020;79:685–99. doi: 10.1136/annrheumdis-2019-216655. [DOI] [PubMed] [Google Scholar]
- 29.Seriolo B, Paolino S, Sulli A, et al. Bone metabolism changes during anti-TNF-alpha therapy in patients with active rheumatoid arthritis. Ann N Y Acad Sci. 2006;1069:420–7. doi: 10.1196/annals.1351.040. [DOI] [PubMed] [Google Scholar]
- 30.Marotte H, Pallot-Prades B, Grange L, et al. A 1-year case-control study in patients with rheumatoid arthritis indicates prevention of loss of bone mineral density in both responders and nonresponders to infliximab. Arthritis Res Ther. 2007;9:R61. doi: 10.1186/ar2219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kume K, Amano K, Yamada S, et al. The effect of tocilizumab on bone mineral density in patients with methotrexate-resistant active rheumatoid arthritis. Rheumatology (Oxford) 2014;53:900–3. doi: 10.1093/rheumatology/ket468. [DOI] [PubMed] [Google Scholar]
- 32.Briot K, Rouanet S, Schaeverbeke T, et al. The effect of tocilizumab on bone mineral density, serum levels of Dickkopf-1 and bone remodeling markers in patients with rheumatoid arthritis. Joint Bone Spine. 2015;82:109–15. doi: 10.1016/j.jbspin.2014.10.015. [DOI] [PubMed] [Google Scholar]
- 33.Chen Y-M, Chen H-H, Huang W-N, et al. Tocilizumab potentially prevents bone loss in patients with anticitrullinated protein antibody-positive rheumatoid arthritis. PLoS One. 2017;12:e0188454. doi: 10.1371/journal.pone.0188454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tada M, Inui K, Sugioka Y, et al. Abatacept might increase bone mineral density at femoral neck for patients with rheumatoid arthritis in clinical practice: AIRTIGHT study. Rheumatol Int. 2018;38:777–84. doi: 10.1007/s00296-017-3922-z. [DOI] [PubMed] [Google Scholar]
- 35.Wheater G, Elshahaly M, Naraghi K, et al. Changes in bone density and bone turnover in patients with rheumatoid arthritis treated with rituximab, results from an exploratory, prospective study. PLoS One. 2018;13:e0201527. doi: 10.1371/journal.pone.0201527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen J-F, Hsu C-Y, Yu S-F, et al. The impact of long-term biologics/target therapy on bone mineral density in rheumatoid arthritis: a propensity score-matched analysis. Rheumatology (Oxford) 2020;59:2471–80. doi: 10.1093/rheumatology/kez655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Garnero P, Thompson E, Woodworth T, et al. Rapid and sustained improvement in bone and cartilage turnover markers with the anti-interleukin-6 receptor inhibitor tocilizumab plus methotrexate in rheumatoid arthritis patients with an inadequate response to methotrexate: results from a substudy of the multicenter double-blind, placebo-controlled trial of tocilizumab in inadequate responders to methotrexate alone. Arthritis Rheum. 2010;62:33–43. doi: 10.1002/art.25053. [DOI] [PubMed] [Google Scholar]
- 38.Karsdal MA, Schett G, Emery P, et al. IL-6 receptor inhibition positively modulates bone balance in rheumatoid arthritis patients with an inadequate response to anti-tumor necrosis factor therapy: biochemical marker analysis of bone metabolism in the tocilizumab RADIATE study ( NCT00106522) Semin Arthritis Rheum. 2012;42:131–9. doi: 10.1016/j.semarthrit.2012.01.004. [DOI] [PubMed] [Google Scholar]
- 39.Chen M, Fu W, Xu H, et al. Pathogenic mechanisms of glucocorticoid-induced osteoporosis. Cytok Grow Factor Rev. 2023;70:54–66. doi: 10.1016/j.cytogfr.2023.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Malochet-Guinamand S, Lambert C, Gossec L, et al. Evaluation of the Implementation of Guidelines on the Treatment of Osteoporosis in Patients with Rheumatoid Arthritis. J Rheumatol. 2020;47:6–14. doi: 10.3899/jrheum.180889. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are available upon reasonable request.