Abstract
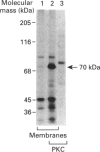
The major platelet integrin, glycoprotein IIb-IIIa, binds soluble fibrinogen only after platelet activation. To investigate the mechanism by which platelets convert glycoprotein IIb-IIIa into a functional fibrinogen receptor, we characterized the opening and closing of fibrinogen-binding sites in isolated platelet membranes and compared the regulatory properties of membrane-bound glycoprotein IIb-IIIa with those of the detergent-solubilized receptor. Basal fibrinogen binding to the membranes possessed many of the properties of fibrinogen binding to activated platelets; however, less than 10% of glycoprotein IIb-IIIa in the membranes was capable of binding fibrinogen. Preincubating the membranes with either an activating glycoprotein IIb-IIIa antibody or alpha-chymotrypsin increased fibrinogen binding. In contrast, agents that require intracellular mediators, such as platelet agonists, guanine-nucleotide-binding-protein activators and purified protein kinase C, did not stimulate fibrinogen binding to the membranes, suggesting that cytosolic factor(s) may be required for activation of the receptor in platelets. Occupancy of glycoprotein IIb-IIIa in the membranes with RGD (Arg-Gly-Asp)-containing peptides reversibly exposed neoantigenic epitopes and fibrinogen-binding sites in the receptor. These conformational changes required membrane fixation to be maintained following peptide removal. Similar results were obtained with purified glycoprotein IIb-IIIa incorporated into phospholipid vesicles, indicating that the resting state of the receptor is favoured in these environments. In contrast, when the conformation of detergent-solubilized glycoprotein IIb-IIIa was altered by exposure to RGD-containing peptides, the receptor remained active even after incorporation into phospholipid vesicles. These results demonstrate that platelet membranes are a useful model in which to study the regulation of glycoprotein IIb-IIIa and suggest that the environment surrounding the receptor may have a profound influence on this process.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrieux A., Hudry-Clergeon G., Ryckewaert J. J., Chapel A., Ginsberg M. H., Plow E. F., Marguerie G. Amino acid sequences in fibrinogen mediating its interaction with its platelet receptor, GPIIbIIIa. J Biol Chem. 1989 Jun 5;264(16):9258–9265. [PubMed] [Google Scholar]
- Bennett J. S., Shattil S. J., Power J. W., Gartner T. K. Interaction of fibrinogen with its platelet receptor. Differential effects of alpha and gamma chain fibrinogen peptides on the glycoprotein IIb-IIIa complex. J Biol Chem. 1988 Sep 15;263(26):12948–12953. [PubMed] [Google Scholar]
- Bennett J. S., Vilaire G. Exposure of platelet fibrinogen receptors by ADP and epinephrine. J Clin Invest. 1979 Nov;64(5):1393–1401. doi: 10.1172/JCI109597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y. P., Djaffar I., Pidard D., Steiner B., Cieutat A. M., Caen J. P., Rosa J. P. Ser-752-->Pro mutation in the cytoplasmic domain of integrin beta 3 subunit and defective activation of platelet integrin alpha IIb beta 3 (glycoprotein IIb-IIIa) in a variant of Glanzmann thrombasthenia. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10169–10173. doi: 10.1073/pnas.89.21.10169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheresh D. A., Berliner S. A., Vicente V., Ruggeri Z. M. Recognition of distinct adhesive sites on fibrinogen by related integrins on platelets and endothelial cells. Cell. 1989 Sep 8;58(5):945–953. doi: 10.1016/0092-8674(89)90946-x. [DOI] [PubMed] [Google Scholar]
- Coller B. S., Peerschke E. I., Scudder L. E., Sullivan C. A. A murine monoclonal antibody that completely blocks the binding of fibrinogen to platelets produces a thrombasthenic-like state in normal platelets and binds to glycoproteins IIb and/or IIIa. J Clin Invest. 1983 Jul;72(1):325–338. doi: 10.1172/JCI110973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Souza S. E., Ginsberg M. H., Burke T. A., Lam S. C., Plow E. F. Localization of an Arg-Gly-Asp recognition site within an integrin adhesion receptor. Science. 1988 Oct 7;242(4875):91–93. doi: 10.1126/science.3262922. [DOI] [PubMed] [Google Scholar]
- Du X. P., Plow E. F., Frelinger A. L., 3rd, O'Toole T. E., Loftus J. C., Ginsberg M. H. Ligands "activate" integrin alpha IIb beta 3 (platelet GPIIb-IIIa). Cell. 1991 May 3;65(3):409–416. doi: 10.1016/0092-8674(91)90458-b. [DOI] [PubMed] [Google Scholar]
- Fitzgerald L. A., Leung B., Phillips D. R. A method for purifying the platelet membrane glycoprotein IIb-IIIa complex. Anal Biochem. 1985 Nov 15;151(1):169–177. doi: 10.1016/0003-2697(85)90067-3. [DOI] [PubMed] [Google Scholar]
- Frelinger A. L., 3rd, Cohen I., Plow E. F., Smith M. A., Roberts J., Lam S. C., Ginsberg M. H. Selective inhibition of integrin function by antibodies specific for ligand-occupied receptor conformers. J Biol Chem. 1990 Apr 15;265(11):6346–6352. [PubMed] [Google Scholar]
- Frelinger A. L., 3rd, Du X. P., Plow E. F., Ginsberg M. H. Monoclonal antibodies to ligand-occupied conformers of integrin alpha IIb beta 3 (glycoprotein IIb-IIIa) alter receptor affinity, specificity, and function. J Biol Chem. 1991 Sep 15;266(26):17106–17111. [PubMed] [Google Scholar]
- Frelinger A. L., 3rd, Lam S. C., Plow E. F., Smith M. A., Loftus J. C., Ginsberg M. H. Occupancy of an adhesive glycoprotein receptor modulates expression of an antigenic site involved in cell adhesion. J Biol Chem. 1988 Sep 5;263(25):12397–12402. [PubMed] [Google Scholar]
- Gulino D., Ryckewaert J. J., Andrieux A., Rabiet M. J., Marguerie G. Identification of a monoclonal antibody against platelet GPIIb that interacts with a calcium-binding site and induces aggregation. J Biol Chem. 1990 Jun 5;265(16):9575–9581. [PubMed] [Google Scholar]
- Hillery C. A., Smyth S. S., Parise L. V. Phosphorylation of human platelet glycoprotein IIIa (GPIIIa). Dissociation from fibrinogen receptor activation and phosphorylation of GPIIIa in vitro. J Biol Chem. 1991 Aug 5;266(22):14663–14669. [PubMed] [Google Scholar]
- Hynes R. O. Integrins: a family of cell surface receptors. Cell. 1987 Feb 27;48(4):549–554. doi: 10.1016/0092-8674(87)90233-9. [DOI] [PubMed] [Google Scholar]
- Jackowski S., Rock C. O. Stimulation of phosphatidylinositol 4,5-bisphosphate phospholipase C activity by phosphatidic acid. Arch Biochem Biophys. 1989 Feb 1;268(2):516–524. doi: 10.1016/0003-9861(89)90318-4. [DOI] [PubMed] [Google Scholar]
- KAZAL L. A., AMSEL S., MILLER O. P., TOCANTINS L. M. THE PREPARATION AND SOME PROPERTIES OF FIBRINOGEN PRECIPITATED FROM HUMAN PLASMA BY GLYCINE. Proc Soc Exp Biol Med. 1963 Aug-Sep;113:989–994. doi: 10.3181/00379727-113-28553. [DOI] [PubMed] [Google Scholar]
- Kitano T., Go M., Kikkawa U., Nishizuka Y. Assay and purification of protein kinase C. Methods Enzymol. 1986;124:349–352. doi: 10.1016/0076-6879(86)24026-4. [DOI] [PubMed] [Google Scholar]
- Kornecki E., Niewiarowski S., Morinelli T. A., Kloczewiak M. Effects of chymotrypsin and adenosine diphosphate on the exposure of fibrinogen receptors on normal human and Glanzmann's thrombasthenic platelets. J Biol Chem. 1981 Jun 10;256(11):5696–5701. [PubMed] [Google Scholar]
- Kornecki E., Tuszynski G. P., Niewiarowski S. Inhibition of fibrinogen receptor-mediated platelet aggregation by heterologous anti-human platelet membrane antibody. Significance of an Mr = 66,000 protein derived from glycoprotein IIIa. J Biol Chem. 1983 Aug 10;258(15):9349–9356. [PubMed] [Google Scholar]
- Kouns W. C., Kirchhofer D., Hadváry P., Edenhofer A., Weller T., Pfenninger G., Baumgartner H. R., Jennings L. K., Steiner B. Reversible conformational changes induced in glycoprotein IIb-IIIa by a potent and selective peptidomimetic inhibitor. Blood. 1992 Nov 15;80(10):2539–2547. [PubMed] [Google Scholar]
- Kouns W. C., Wall C. D., White M. M., Fox C. F., Jennings L. K. A conformation-dependent epitope of human platelet glycoprotein IIIa. J Biol Chem. 1990 Nov 25;265(33):20594–20601. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lam S. C., Plow E. F., Smith M. A., Andrieux A., Ryckwaert J. J., Marguerie G., Ginsberg M. H. Evidence that arginyl-glycyl-aspartate peptides and fibrinogen gamma chain peptides share a common binding site on platelets. J Biol Chem. 1987 Jan 25;262(3):947–950. [PubMed] [Google Scholar]
- Mooney J. J., Horne W. C., Handin R. I., Schildkraut J. J., Alexander R. W. Sodium inhibits both adenylate cyclase and high-affinity 3H-labeled p-aminoclonidine binding to alpha 2-adrenergic receptors in purified human platelet membranes. Mol Pharmacol. 1982 May;21(3):600–608. [PubMed] [Google Scholar]
- Newman P. J., Allen R. W., Kahn R. A., Kunicki T. J. Quantitation of membrane glycoprotein IIIa on intact human platelets using the monoclonal antibody, AP-3. Blood. 1985 Jan;65(1):227–232. [PubMed] [Google Scholar]
- Niewiarowski S., Budzynski A. Z., Morinelli T. A., Brudzynski T. M., Stewart G. J. Exposure of fibrinogen receptor on human platelets by proteolytic enzymes. J Biol Chem. 1981 Jan 25;256(2):917–925. [PubMed] [Google Scholar]
- Niewiarowski S., Norton K. J., Eckardt A., Lukasiewicz H., Holt J. C., Kornecki E. Structural and functional characterization of major platelet membrane components derived by limited proteolysis of glycoprotein IIIa. Biochim Biophys Acta. 1989 Jul 24;983(1):91–99. doi: 10.1016/0005-2736(89)90384-2. [DOI] [PubMed] [Google Scholar]
- O'Toole T. E., Loftus J. C., Du X. P., Glass A. A., Ruggeri Z. M., Shattil S. J., Plow E. F., Ginsberg M. H. Affinity modulation of the alpha IIb beta 3 integrin (platelet GPIIb-IIIa) is an intrinsic property of the receptor. Cell Regul. 1990 Nov;1(12):883–893. doi: 10.1091/mbc.1.12.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Toole T. E., Mandelman D., Forsyth J., Shattil S. J., Plow E. F., Ginsberg M. H. Modulation of the affinity of integrin alpha IIb beta 3 (GPIIb-IIIa) by the cytoplasmic domain of alpha IIb. Science. 1991 Nov 8;254(5033):845–847. doi: 10.1126/science.1948065. [DOI] [PubMed] [Google Scholar]
- Parise L. V., Helgerson S. L., Steiner B., Nannizzi L., Phillips D. R. Synthetic peptides derived from fibrinogen and fibronectin change the conformation of purified platelet glycoprotein IIb-IIIa. J Biol Chem. 1987 Sep 15;262(26):12597–12602. [PubMed] [Google Scholar]
- Parise L. V., Phillips D. R. Reconstitution of the purified platelet fibrinogen receptor. Fibrinogen binding properties of the glycoprotein IIb-IIIa complex. J Biol Chem. 1985 Sep 5;260(19):10698–10707. [PubMed] [Google Scholar]
- Peerschke E. I., Coller B. S. A murine monoclonal antibody that blocks fibrinogen binding to normal platelets also inhibits fibrinogen interactions with chymotrypsin-treated platelets. Blood. 1984 Jul;64(1):59–63. [PubMed] [Google Scholar]
- Phillips D. R., Baughan A. K. Fibrinogen binding to human platelet plasma membranes. Identification of two steps requiring divalent cations. J Biol Chem. 1983 Sep 10;258(17):10240–10246. [PubMed] [Google Scholar]
- Pidard D., Frelinger A. L., Bouillot C., Nurden A. T. Activation of the fibrinogen receptor on human platelets exposed to alpha chymotrypsin. Relationship with a major proteolytic cleavage at the carboxyterminus of the membrane glycoprotein IIb heavy chain. Eur J Biochem. 1991 Sep 1;200(2):437–447. doi: 10.1111/j.1432-1033.1991.tb16202.x. [DOI] [PubMed] [Google Scholar]
- Plow E. F., Ginsberg M. H. Cellular adhesion: GPIIb-IIIa as a prototypic adhesion receptor. Prog Hemost Thromb. 1989;9:117–156. [PubMed] [Google Scholar]
- Plow E. F., Marguerie G. Inhibition of fibrinogen binding to human platelets by the tetrapeptide glycyl-L-prolyl-L-arginyl-L-proline. Proc Natl Acad Sci U S A. 1982 Jun;79(12):3711–3715. doi: 10.1073/pnas.79.12.3711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plow E. F., Pierschbacher M. D., Ruoslahti E., Marguerie G. A., Ginsberg M. H. The effect of Arg-Gly-Asp-containing peptides on fibrinogen and von Willebrand factor binding to platelets. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8057–8061. doi: 10.1073/pnas.82.23.8057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plow E. F., Pierschbacher M. D., Ruoslahti E., Marguerie G. A., Ginsberg M. H. The effect of Arg-Gly-Asp-containing peptides on fibrinogen and von Willebrand factor binding to platelets. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8057–8061. doi: 10.1073/pnas.82.23.8057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plow E. F., Srouji A. H., Meyer D., Marguerie G., Ginsberg M. H. Evidence that three adhesive proteins interact with a common recognition site on activated platelets. J Biol Chem. 1984 May 10;259(9):5388–5391. [PubMed] [Google Scholar]
- Pytela R., Pierschbacher M. D., Ginsberg M. H., Plow E. F., Ruoslahti E. Platelet membrane glycoprotein IIb/IIIa: member of a family of Arg-Gly-Asp--specific adhesion receptors. Science. 1986 Mar 28;231(4745):1559–1562. doi: 10.1126/science.2420006. [DOI] [PubMed] [Google Scholar]
- Ruggeri Z. M., Houghten R. A., Russell S. R., Zimmerman T. S. Inhibition of platelet function with synthetic peptides designed to be high-affinity antagonists of fibrinogen binding to platelets. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5708–5712. doi: 10.1073/pnas.83.15.5708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. New perspectives in cell adhesion: RGD and integrins. Science. 1987 Oct 23;238(4826):491–497. doi: 10.1126/science.2821619. [DOI] [PubMed] [Google Scholar]
- Shadle P. J., Ginsberg M. H., Plow E. F., Barondes S. H. Platelet-collagen adhesion: inhibition by a monoclonal antibody that binds glycoprotein IIb. J Cell Biol. 1984 Dec;99(6):2056–2060. doi: 10.1083/jcb.99.6.2056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shattil S. J., Brass L. F. Induction of the fibrinogen receptor on human platelets by intracellular mediators. J Biol Chem. 1987 Jan 25;262(3):992–1000. [PubMed] [Google Scholar]
- Shattil S. J., Cunningham M., Wiedmer T., Zhao J., Sims P. J., Brass L. F. Regulation of glycoprotein IIb-IIIa receptor function studied with platelets permeabilized by the pore-forming complement proteins C5b-9. J Biol Chem. 1992 Sep 15;267(26):18424–18431. [PubMed] [Google Scholar]
- Shattil S. J., Hoxie J. A., Cunningham M., Brass L. F. Changes in the platelet membrane glycoprotein IIb.IIIa complex during platelet activation. J Biol Chem. 1985 Sep 15;260(20):11107–11114. [PubMed] [Google Scholar]
- Steiner B., Cousot D., Trzeciak A., Gillessen D., Hadváry P. Ca2+-dependent binding of a synthetic Arg-Gly-Asp (RGD) peptide to a single site on the purified platelet glycoprotein IIb-IIIa complex. J Biol Chem. 1989 Aug 5;264(22):13102–13108. [PubMed] [Google Scholar]
- Taylor D. G., Crawford N. Enzymatic and chemical analyses of pig platelet membrane subfractions isolated by zonal centrifugation. Biochim Biophys Acta. 1976 Jun 4;436(1):77–94. doi: 10.1016/0005-2736(76)90221-2. [DOI] [PubMed] [Google Scholar]
- WARREN L. The thiobarbituric acid assay of sialic acids. J Biol Chem. 1959 Aug;234(8):1971–1975. [PubMed] [Google Scholar]
- van Willigen G., Akkerman J. W. Protein kinase C and cyclic AMP regulate reversible exposure of binding sites for fibrinogen on the glycoprotein IIB-IIIA complex of human platelets. Biochem J. 1991 Jan 1;273(Pt 1):115–120. doi: 10.1042/bj2730115. [DOI] [PMC free article] [PubMed] [Google Scholar]