Abstract
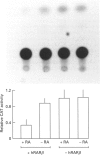
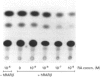
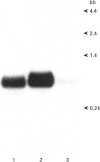
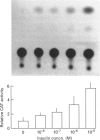
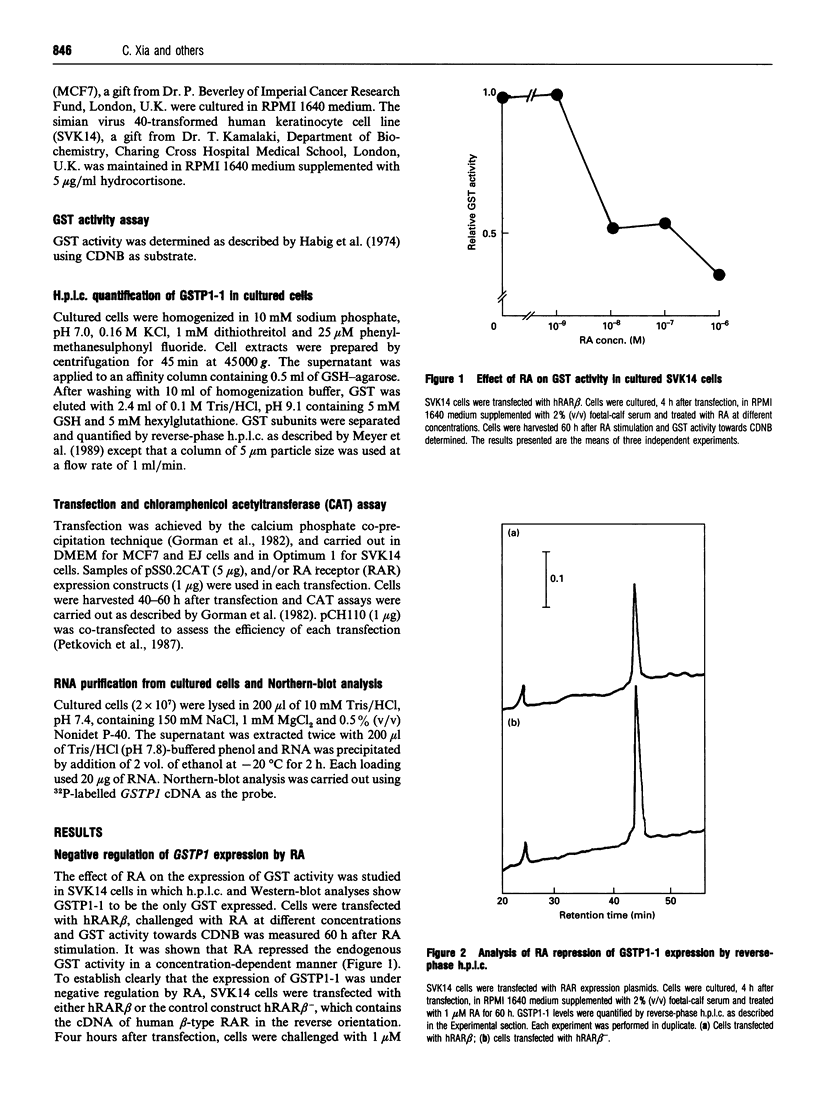
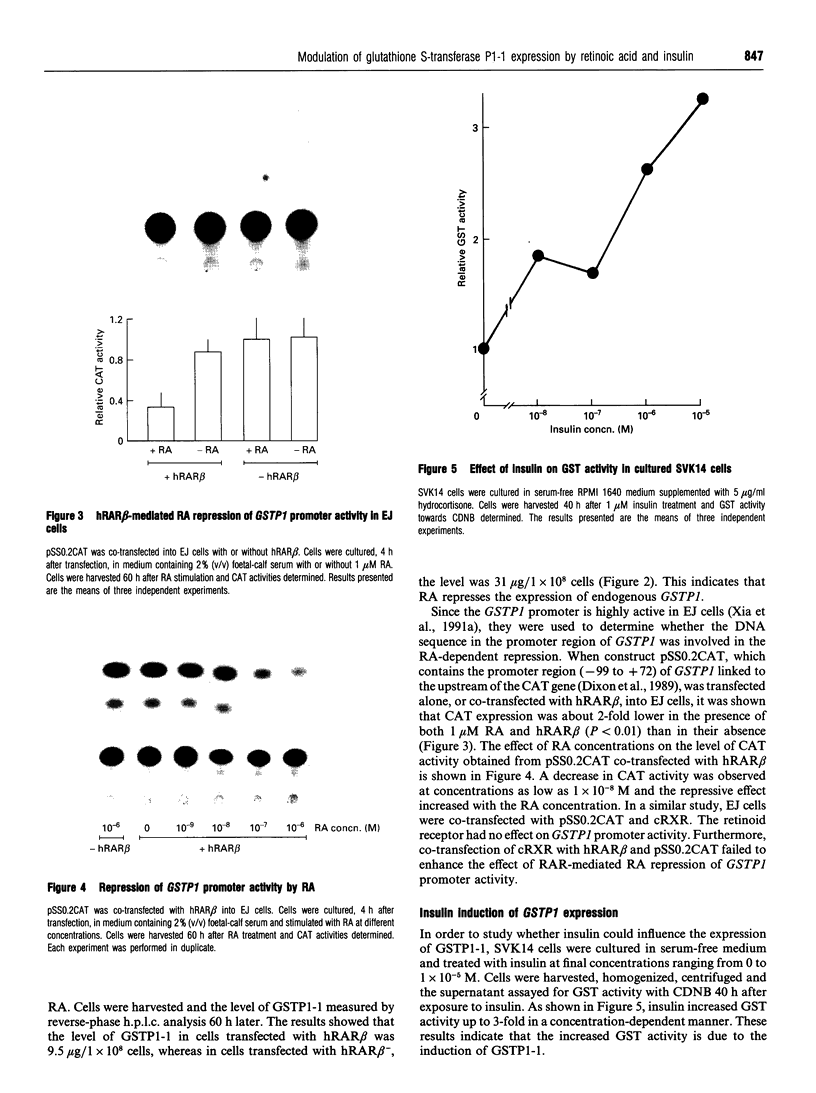
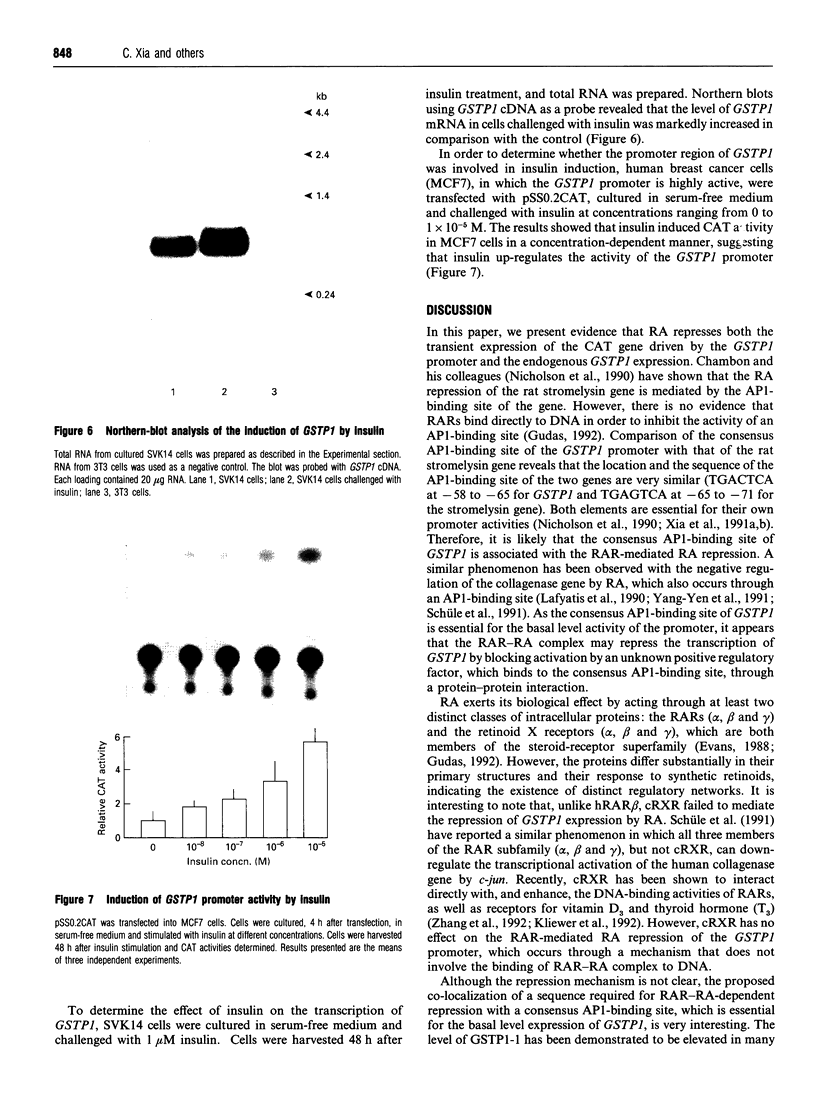
Glutathione S-transferases (GSTs) are a group of enzymes which play an important role in the detoxication of xenobiotics. It is shown that the expression of human glutathione S-transferase P1-1 (GSTP1-1) is suppressed by retinoic acid (RA) as the result of decreased transcription from its gene, GSTP1. Chloramphenicol acetyltransferase (CAT) assays indicate that the effect of RA on the transcription of a GSTP1 promoter-CAT fusion gene is mediated by the region -99 to +72 of GSTP1. A consensus activator protein 1-binding site, located at nucleotide position -59 to -65 of GSTP1, is suggested to be responsible for RA repression. This effect of RA on GSTP1 expression is mediated by the human beta-type RA receptor, hRAR beta, but not the chicken retinoid X receptor, cRXR. The retinoid X receptor does not augment the action of hRAR beta on GSTP1. In addition, it is shown that GSTP1-1 expression is enhanced by insulin as a result of increased transcription of GSTP1. Assay of CAT activity indicates that the effect of insulin on the transcription of GSTP1 is also mediated by the region -99 to +72 of GSTP1. Comparison with sequences of other insulin-responsive genes, suggests that insulin enhancement of GSTP1 expression is effected by an eight-base-pair sequence, 'CCCGCGTC', located at +48 to +55 in intron 1 of the gene. These results are discussed in relation to the increased expression of GSTP1-1 in many tumour cells.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alcalay M., Zangrilli D., Pandolfi P. P., Longo L., Mencarelli A., Giacomucci A., Rocchi M., Biondi A., Rambaldi A., Lo Coco F. Translocation breakpoint of acute promyelocytic leukemia lies within the retinoic acid receptor alpha locus. Proc Natl Acad Sci U S A. 1991 Mar 1;88(5):1977–1981. doi: 10.1073/pnas.88.5.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batist G., Tulpule A., Sinha B. K., Katki A. G., Myers C. E., Cowan K. H. Overexpression of a novel anionic glutathione transferase in multidrug-resistant human breast cancer cells. J Biol Chem. 1986 Nov 25;261(33):15544–15549. [PubMed] [Google Scholar]
- Borrow J., Goddard A. D., Sheer D., Solomon E. Molecular analysis of acute promyelocytic leukemia breakpoint cluster region on chromosome 17. Science. 1990 Sep 28;249(4976):1577–1580. doi: 10.1126/science.2218500. [DOI] [PubMed] [Google Scholar]
- Burgering B. M., Medema R. H., Maassen J. A., van de Wetering M. L., van der Eb A. J., McCormick F., Bos J. L. Insulin stimulation of gene expression mediated by p21ras activation. EMBO J. 1991 May;10(5):1103–1109. doi: 10.1002/j.1460-2075.1991.tb08050.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coles B., Ketterer B. The role of glutathione and glutathione transferases in chemical carcinogenesis. Crit Rev Biochem Mol Biol. 1990;25(1):47–70. doi: 10.3109/10409239009090605. [DOI] [PubMed] [Google Scholar]
- Cowell I. G., Dixon K. H., Pemble S. E., Ketterer B., Taylor J. B. The structure of the human glutathione S-transferase pi gene. Biochem J. 1988 Oct 1;255(1):79–83. doi: 10.1042/bj2550079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deffie A. M., Alam T., Seneviratne C., Beenken S. W., Batra J. K., Shea T. C., Henner W. D., Goldenberg G. J. Multifactorial resistance to adriamycin: relationship of DNA repair, glutathione transferase activity, drug efflux, and P-glycoprotein in cloned cell lines of adriamycin-sensitive and -resistant P388 leukemia. Cancer Res. 1988 Jul 1;48(13):3595–3602. [PubMed] [Google Scholar]
- Dixon K. H., Cowell I. G., Xia C. L., Pemble S. E., Ketterer B., Taylor J. B. Control of expression of the human glutathione S-transferase pi gene differs from its rat orthologue. Biochem Biophys Res Commun. 1989 Sep 15;163(2):815–822. doi: 10.1016/0006-291x(89)92295-x. [DOI] [PubMed] [Google Scholar]
- Evans R. M. The steroid and thyroid hormone receptor superfamily. Science. 1988 May 13;240(4854):889–895. doi: 10.1126/science.3283939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garewal H. S., Meyskens F., Jr Retinoids and carotenoids in the prevention of oral cancer: a critical appraisal. Cancer Epidemiol Biomarkers Prev. 1992 Jan-Feb;1(2):155–159. [PubMed] [Google Scholar]
- Gorman C. M., Moffat L. F., Howard B. H. Recombinant genomes which express chloramphenicol acetyltransferase in mammalian cells. Mol Cell Biol. 1982 Sep;2(9):1044–1051. doi: 10.1128/mcb.2.9.1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gudas L. J. Retinoids, retinoid-responsive genes, cell differentiation, and cancer. Cell Growth Differ. 1992 Sep;3(9):655–662. [PubMed] [Google Scholar]
- Habig W. H., Pabst M. J., Jakoby W. B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem. 1974 Nov 25;249(22):7130–7139. [PubMed] [Google Scholar]
- Hatayama I., Yamada Y., Tanaka K., Ichihara A., Sato K. Induction of glutathione S-transferase P-form in primary cultured rat hepatocytes by epidermal growth factor and insulin. Jpn J Cancer Res. 1991 Jul;82(7):807–814. doi: 10.1111/j.1349-7006.1991.tb02706.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imagawa M., Osada S., Koyama Y., Suzuki T., Hirom P. C., Diccianni M. B., Morimura S., Muramatsu M. SF-B that binds to a negative element in glutathione transferase P gene is similar or identical to trans-activator LAP/IL6-DBP. Biochem Biophys Res Commun. 1991 Aug 30;179(1):293–300. doi: 10.1016/0006-291x(91)91368-m. [DOI] [PubMed] [Google Scholar]
- Imagawa M., Osada S., Okuda A., Muramatsu M. Silencer binding proteins function on multiple cis-elements in the glutathione transferase P gene. Nucleic Acids Res. 1991 Jan 11;19(1):5–10. doi: 10.1093/nar/19.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakoby W. B. The glutathione S-transferases: a group of multifunctional detoxification proteins. Adv Enzymol Relat Areas Mol Biol. 1978;46:383–414. doi: 10.1002/9780470122914.ch6. [DOI] [PubMed] [Google Scholar]
- Kliewer S. A., Umesono K., Mangelsdorf D. J., Evans R. M. Retinoid X receptor interacts with nuclear receptors in retinoic acid, thyroid hormone and vitamin D3 signalling. Nature. 1992 Jan 30;355(6359):446–449. doi: 10.1038/355446a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knowles B. B., Howe C. C., Aden D. P. Human hepatocellular carcinoma cell lines secrete the major plasma proteins and hepatitis B surface antigen. Science. 1980 Jul 25;209(4455):497–499. doi: 10.1126/science.6248960. [DOI] [PubMed] [Google Scholar]
- Lafyatis R., Kim S. J., Angel P., Roberts A. B., Sporn M. B., Karin M., Wilder R. L. Interleukin-1 stimulates and all-trans-retinoic acid inhibits collagenase gene expression through its 5' activator protein-1-binding site. Mol Endocrinol. 1990 Jul;4(7):973–980. doi: 10.1210/mend-4-7-973. [DOI] [PubMed] [Google Scholar]
- Lippman S. M., Kavanagh J. J., Paredes-Espinoza M., Delgadillo-Madrueño F., Paredes-Casillas P., Hong W. K., Holdener E., Krakoff I. H. 13-cis-retinoic acid plus interferon alpha-2a: highly active systemic therapy for squamous cell carcinoma of the cervix. J Natl Cancer Inst. 1992 Feb 19;84(4):241–245. doi: 10.1093/jnci/84.4.241. [DOI] [PubMed] [Google Scholar]
- Lippman S. M., Parkinson D. R., Itri L. M., Weber R. S., Schantz S. P., Ota D. M., Schusterman M. A., Krakoff I. H., Gutterman J. U., Hong W. K. 13-cis-retinoic acid and interferon alpha-2a: effective combination therapy for advanced squamous cell carcinoma of the skin. J Natl Cancer Inst. 1992 Feb 19;84(4):235–241. doi: 10.1093/jnci/84.4.235. [DOI] [PubMed] [Google Scholar]
- Mannervik B., Awasthi Y. C., Board P. G., Hayes J. D., Di Ilio C., Ketterer B., Listowsky I., Morgenstern R., Muramatsu M., Pearson W. R. Nomenclature for human glutathione transferases. Biochem J. 1992 Feb 15;282(Pt 1):305–306. doi: 10.1042/bj2820305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannervik B., Danielson U. H. Glutathione transferases--structure and catalytic activity. CRC Crit Rev Biochem. 1988;23(3):283–337. doi: 10.3109/10409238809088226. [DOI] [PubMed] [Google Scholar]
- Meyer D. J., Coles B., Pemble S. E., Gilmore K. S., Fraser G. M., Ketterer B. Theta, a new class of glutathione transferases purified from rat and man. Biochem J. 1991 Mar 1;274(Pt 2):409–414. doi: 10.1042/bj2740409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer D. J., Lalor E., Coles B., Kispert A., Alin P., Mannervik B., Ketterer B. Single-step purification and h.p.l.c. analysis of glutathione transferase 8-8 in rat tissues. Biochem J. 1989 Jun 15;260(3):785–788. doi: 10.1042/bj2600785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohn K. L., Laz T. M., Melby A. E., Taub R. Immediate-early gene expression differs between regenerating liver, insulin-stimulated H-35 cells, and mitogen-stimulated Balb/c 3T3 cells. Liver-specific induction patterns of gene 33, phosphoenolpyruvate carboxykinase, and the jun, fos, and egr families. J Biol Chem. 1990 Dec 15;265(35):21914–21921. [PubMed] [Google Scholar]
- Morrow C. S., Goldsmith M. E., Cowan K. H. Regulation of human glutathione S-transferase pi gene transcription: influence of 5'-flanking sequences and trans-activating factors which recognize AP-1-binding sites. Gene. 1990 Apr 16;88(2):215–225. doi: 10.1016/0378-1119(90)90034-o. [DOI] [PubMed] [Google Scholar]
- Nasrin N., Ercolani L., Denaro M., Kong X. F., Kang I., Alexander M. An insulin response element in the glyceraldehyde-3-phosphate dehydrogenase gene binds a nuclear protein induced by insulin in cultured cells and by nutritional manipulations in vivo. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5273–5277. doi: 10.1073/pnas.87.14.5273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholson R. C., Mader S., Nagpal S., Leid M., Rochette-Egly C., Chambon P. Negative regulation of the rat stromelysin gene promoter by retinoic acid is mediated by an AP1 binding site. EMBO J. 1990 Dec;9(13):4443–4454. doi: 10.1002/j.1460-2075.1990.tb07895.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. M., Granner D. K. Regulation of gene expression by insulin. Biochem J. 1991 Sep 15;278(Pt 3):609–619. doi: 10.1042/bj2780609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okuda A., Imagawa M., Maeda Y., Sakai M., Muramatsu M. Structural and functional analysis of an enhancer GPEI having a phorbol 12-O-tetradecanoate 13-acetate responsive element-like sequence found in the rat glutathione transferase P gene. J Biol Chem. 1989 Oct 5;264(28):16919–16926. [PubMed] [Google Scholar]
- Okuda A., Imagawa M., Sakai M., Muramatsu M. Functional cooperativity between two TPA responsive elements in undifferentiated F9 embryonic stem cells. EMBO J. 1990 Apr;9(4):1131–1135. doi: 10.1002/j.1460-2075.1990.tb08219.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okuda A., Sakai M., Muramatsu M. The structure of the rat glutathione S-transferase P gene and related pseudogenes. J Biol Chem. 1987 Mar 15;262(8):3858–3863. [PubMed] [Google Scholar]
- Petkovich M., Brand N. J., Krust A., Chambon P. A human retinoic acid receptor which belongs to the family of nuclear receptors. Nature. 1987 Dec 3;330(6147):444–450. doi: 10.1038/330444a0. [DOI] [PubMed] [Google Scholar]
- Sakai M., Muramatsu M., Nishi S. Suppression of glutathione transferase P expression by glucocorticoid. Biochem Biophys Res Commun. 1992 Sep 16;187(2):976–983. doi: 10.1016/0006-291x(92)91293-y. [DOI] [PubMed] [Google Scholar]
- Sakai M., Okuda A., Muramatsu M. Multiple regulatory elements and phorbol 12-O-tetradecanoate 13-acetate responsiveness of the rat placental glutathione transferase gene. Proc Natl Acad Sci U S A. 1988 Dec;85(24):9456–9460. doi: 10.1073/pnas.85.24.9456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato K. Glutathione transferases as markers of preneoplasia and neoplasia. Adv Cancer Res. 1989;52:205–255. doi: 10.1016/s0065-230x(08)60214-6. [DOI] [PubMed] [Google Scholar]
- Satoh K., Kitahara A., Soma Y., Inaba Y., Hatayama I., Sato K. Purification, induction, and distribution of placental glutathione transferase: a new marker enzyme for preneoplastic cells in the rat chemical hepatocarcinogenesis. Proc Natl Acad Sci U S A. 1985 Jun;82(12):3964–3968. doi: 10.1073/pnas.82.12.3964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schüle R., Rangarajan P., Kliewer S., Ransone L. J., Bolado J., Yang N., Verma I. M., Evans R. M. Functional antagonism between oncoprotein c-Jun and the glucocorticoid receptor. Cell. 1990 Sep 21;62(6):1217–1226. doi: 10.1016/0092-8674(90)90397-w. [DOI] [PubMed] [Google Scholar]
- Schüle R., Rangarajan P., Yang N., Kliewer S., Ransone L. J., Bolado J., Verma I. M., Evans R. M. Retinoic acid is a negative regulator of AP-1-responsive genes. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6092–6096. doi: 10.1073/pnas.88.14.6092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stumpo D. J., Blackshear P. J. Insulin and growth factor effects on c-fos expression in normal and protein kinase C-deficient 3T3-L1 fibroblasts and adipocytes. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9453–9457. doi: 10.1073/pnas.83.24.9453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchida S., Sato K. Glutathione transferases and cancer. Crit Rev Biochem Mol Biol. 1992;27(4-5):337–384. doi: 10.3109/10409239209082566. [DOI] [PubMed] [Google Scholar]
- Xia C. L., Cowell I. G., Dixon K. H., Pemble S. E., Ketterer B., Taylor J. B. Glutathione transferase pi its minimal promoter and downstream cis-acting element. Biochem Biophys Res Commun. 1991 Apr 15;176(1):233–240. doi: 10.1016/0006-291x(91)90914-s. [DOI] [PubMed] [Google Scholar]
- Yang-Yen H. F., Chambard J. C., Sun Y. L., Smeal T., Schmidt T. J., Drouin J., Karin M. Transcriptional interference between c-Jun and the glucocorticoid receptor: mutual inhibition of DNA binding due to direct protein-protein interaction. Cell. 1990 Sep 21;62(6):1205–1215. doi: 10.1016/0092-8674(90)90396-v. [DOI] [PubMed] [Google Scholar]
- Yang-Yen H. F., Zhang X. K., Graupner G., Tzukerman M., Sakamoto B., Karin M., Pfahl M. Antagonism between retinoic acid receptors and AP-1: implications for tumor promotion and inflammation. New Biol. 1991 Dec;3(12):1206–1219. [PubMed] [Google Scholar]
- Zhang X. K., Hoffmann B., Tran P. B., Graupner G., Pfahl M. Retinoid X receptor is an auxiliary protein for thyroid hormone and retinoic acid receptors. Nature. 1992 Jan 30;355(6359):441–446. doi: 10.1038/355441a0. [DOI] [PubMed] [Google Scholar]
- de Thé H., Chomienne C., Lanotte M., Degos L., Dejean A. The t(15;17) translocation of acute promyelocytic leukaemia fuses the retinoic acid receptor alpha gene to a novel transcribed locus. Nature. 1990 Oct 11;347(6293):558–561. doi: 10.1038/347558a0. [DOI] [PubMed] [Google Scholar]