Abstract
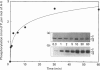
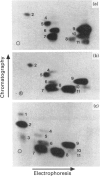
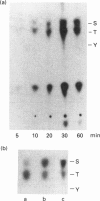
Annexin I is a member of the annexin family of Ca(2+)- and phospholipid-binding proteins. The ability of this protein to aggregate and to mediate the fusion of various types of vesicles has supported the hypothesis that this protein might be involved in intracellular membrane fusion processes such as exocytosis. Although annexin I has been described as a major in vitro substrate of both protein kinase C and the epidermal-growth-factor-receptor protein tyrosine kinase, the functional consequences of these phosphorylation events have not been investigated. In this paper we examine the effect of the phosphorylation of annexin I by protein kinase C on the phospholipid aggregation activity of the protein. The stoichiometry of phosphorylation of the protein was affected by the method of preparation of the phospholipid. Under optimal assay conditions protein kinase C catalysed the incorporation of 2.83 +/- 0.23 mol of phosphate/mol of annexin I (mean +/- S.E.M., n = 21). Studies with the Ca(2+)- and phospholipid-independent form of protein kinase C suggested that the phosphorylation of annexin I was stimulated by phospholipid in the absence of Ca2+, although maximal phosphorylation was achieved in the presence of both phospholipid and Ca2+. Phosphorylation of annexin I resulted in a dramatic decrease in the rate and extent of phospholipid vesicle aggregation, without significantly disrupting the binding of the protein to the phospholipid vesicles. The phosphorylation of annexin I increased the EC50 (Ca2+) of phospholipid vesicle aggregation from 19 +/- 10 microM (mean +/- S.D., n = 7) for the native protein to 290 +/- 95 microM (mean +/- S.D., n = 5) for the phosphorylated protein. These results suggest that protein kinase C may act to inhibit the phospholipid vesicle aggregation activity of annexin I.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ali S. M., Burgoyne R. D. The stimulatory effect of calpactin (annexin II) on calcium-dependent exocytosis in chromaffin cells: requirement for both the N-terminal and core domains of p36 and ATP. Cell Signal. 1990;2(3):265–276. doi: 10.1016/0898-6568(90)90054-e. [DOI] [PubMed] [Google Scholar]
- Ando Y., Imamura S., Hong Y. M., Owada M. K., Kakunaga T., Kannagi R. Enhancement of calcium sensitivity of lipocortin I in phospholipid binding induced by limited proteolysis and phosphorylation at the amino terminus as analyzed by phospholipid affinity column chromatography. J Biol Chem. 1989 Apr 25;264(12):6948–6955. [PubMed] [Google Scholar]
- Antonicelli F., Omri B., Breton M. F., Rothhut B., Russo-Marie F., Pavlovic-Hournac M., Haye B. Identification of four lipocortin proteins and phosphorylation of lipocortin I by protein kinase C in cytosols of porcine thyroid cell cultures. FEBS Lett. 1989 Dec 4;258(2):346–350. doi: 10.1016/0014-5793(89)81690-4. [DOI] [PubMed] [Google Scholar]
- Blackwood R. A., Ernst J. D. Characterization of Ca2(+)-dependent phospholipid binding, vesicle aggregation and membrane fusion by annexins. Biochem J. 1990 Feb 15;266(1):195–200. doi: 10.1042/bj2660195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Burgoyne R. D. Secretory vesicle-associated proteins and their role in exocytosis. Annu Rev Physiol. 1990;52:647–659. doi: 10.1146/annurev.ph.52.030190.003243. [DOI] [PubMed] [Google Scholar]
- Campos-Gonzalez R., Kanemitsu M., Boynton A. L. Tumor promoter-dependent phosphorylation of a Triton X-100 extractable form of lipocortin I in T51B rat liver cells. Exp Cell Res. 1989 Oct;184(2):287–296. doi: 10.1016/0014-4827(89)90328-5. [DOI] [PubMed] [Google Scholar]
- Dedman J. R. Mediation of intracellular calcium: variances on a common theme. Cell Calcium. 1986 Dec;7(5-6):297–307. doi: 10.1016/0143-4160(86)90034-5. [DOI] [PubMed] [Google Scholar]
- Drust D. S., Creutz C. E. Aggregation of chromaffin granules by calpactin at micromolar levels of calcium. Nature. 1988 Jan 7;331(6151):88–91. doi: 10.1038/331088a0. [DOI] [PubMed] [Google Scholar]
- Fabiato A., Fabiato F. Calculator programs for computing the composition of the solutions containing multiple metals and ligands used for experiments in skinned muscle cells. J Physiol (Paris) 1979;75(5):463–505. [PubMed] [Google Scholar]
- Fava R. A., Cohen S. Isolation of a calcium-dependent 35-kilodalton substrate for the epidermal growth factor receptor/kinase from A-431 cells. J Biol Chem. 1984 Feb 25;259(4):2636–2645. [PubMed] [Google Scholar]
- Geisow M. J., Ali S. M., Boustead C., Burgoyne R. D., Taylor W. R., Walker J. H. Structures and functions of a supergene family of calcium and phospholipid binding proteins. Prog Clin Biol Res. 1990;349:111–121. [PubMed] [Google Scholar]
- Genge B. R., Wu L. N., Wuthier R. E. Identification of phospholipid-dependent calcium-binding proteins as constituents of matrix vesicles. J Biol Chem. 1989 Jun 25;264(18):10917–10921. [PubMed] [Google Scholar]
- Glenney J. R., Jr Calpactins: calcium-regulated membrane-skeletal proteins. Bioessays. 1987 Oct;7(4):173–175. doi: 10.1002/bies.950070408. [DOI] [PubMed] [Google Scholar]
- Glenney J. R., Jr, Tack B., Powell M. A. Calpactins: two distinct Ca++-regulated phospholipid- and actin-binding proteins isolated from lung and placenta. J Cell Biol. 1987 Mar;104(3):503–511. doi: 10.1083/jcb.104.3.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haigler H. T., Schlaepfer D. D., Burgess W. H. Characterization of lipocortin I and an immunologically unrelated 33-kDa protein as epidermal growth factor receptor/kinase substrates and phospholipase A2 inhibitors. J Biol Chem. 1987 May 15;262(14):6921–6930. [PubMed] [Google Scholar]
- Haigler H. T., Schlaepfer D. D. Expression of lipocortin 1 and endonexin II as a function of cellular growth state. Prog Clin Biol Res. 1990;349:91–98. [PubMed] [Google Scholar]
- Hannun Y. A., Loomis C. R., Bell R. M. Protein kinase C activation in mixed micelles. Mechanistic implications of phospholipid, diacylglycerol, and calcium interdependencies. J Biol Chem. 1986 Jun 5;261(16):7184–7190. [PubMed] [Google Scholar]
- Hirata F. The regulation of lipomodulin, a phospholipase inhibitory protein, in rabbit neutrophils by phosphorylation. J Biol Chem. 1981 Aug 10;256(15):7730–7733. [PubMed] [Google Scholar]
- Johnsson N., Gerke V., Weber K. P36, member of the CA2+/lipid binding proteins (annexins, calpactins, lipocortins) and its complex with P11; molecular aspects. Prog Clin Biol Res. 1990;349:123–133. [PubMed] [Google Scholar]
- Johnstone S. A., Hubaishy I., Waisman D. M. Phosphorylation of annexin II tetramer by protein kinase C inhibits aggregation of lipid vesicles by the protein. J Biol Chem. 1992 Dec 25;267(36):25976–25981. [PubMed] [Google Scholar]
- Karasik A., Pepinsky R. B., Shoelson S. E., Kahn C. R. Lipocortins 1 and 2 as substrates for the insulin receptor kinase in rat liver. J Biol Chem. 1988 Aug 25;263(24):11862–11867. [PubMed] [Google Scholar]
- Khanna N. C., Helwig E. D., Ikebuchi N. W., Fitzpatrick S., Bajwa R., Waisman D. M. Purification and characterization of annexin proteins from bovine lung. Biochemistry. 1990 May 22;29(20):4852–4862. doi: 10.1021/bi00472a015. [DOI] [PubMed] [Google Scholar]
- Khanna N. C., Tokuda M., Waisman D. M. Phosphorylation of lipocortins in vitro by protein kinase C. Biochem Biophys Res Commun. 1986 Dec 15;141(2):547–554. doi: 10.1016/s0006-291x(86)80208-x. [DOI] [PubMed] [Google Scholar]
- Khanna N. C., Tokuda M., Waisman D. M. Purification of three forms of lipocortin from bovine lung. Cell Calcium. 1987 Jun;8(3):217–228. doi: 10.1016/0143-4160(87)90020-0. [DOI] [PubMed] [Google Scholar]
- Kitano T., Go M., Kikkawa U., Nishizuka Y. Assay and purification of protein kinase C. Methods Enzymol. 1986;124:349–352. doi: 10.1016/0076-6879(86)24026-4. [DOI] [PubMed] [Google Scholar]
- Klee C. B. Ca2+-dependent phospholipid- (and membrane-) binding proteins. Biochemistry. 1988 Sep 6;27(18):6645–6653. doi: 10.1021/bi00418a001. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Meers P., Mealy T., Pavlotsky N., Tauber A. I. Annexin I-mediated vesicular aggregation: mechanism and role in human neutrophils. Biochemistry. 1992 Jul 21;31(28):6372–6382. doi: 10.1021/bi00143a003. [DOI] [PubMed] [Google Scholar]
- Michener M. L., Dawson W. B., Creutz C. E. Phosphorylation of a chromaffin granule-binding protein in stimulated chromaffin cells. J Biol Chem. 1986 May 15;261(14):6548–6555. [PubMed] [Google Scholar]
- Moss S. E., Crumpton M. J. The lipocortins and the EF hand proteins: Ca2(+)-binding sites and evolution. Trends Biochem Sci. 1990 Jan;15(1):11–12. doi: 10.1016/0968-0004(90)90118-u. [DOI] [PubMed] [Google Scholar]
- Pepinsky R. B., Sinclair L. K. Epidermal growth factor-dependent phosphorylation of lipocortin. Nature. 1986 May 1;321(6065):81–84. doi: 10.1038/321081a0. [DOI] [PubMed] [Google Scholar]
- Reeves J. P., Dowben R. M. Formation and properties of thin-walled phospholipid vesicles. J Cell Physiol. 1969 Feb;73(1):49–60. doi: 10.1002/jcp.1040730108. [DOI] [PubMed] [Google Scholar]
- Russo-Marie F. La lipocortine, protéine de contrôle de l'activation membranaire dans la réaction inflammatoire. Pathol Biol (Paris) 1986 Apr;34(4):227–233. [PubMed] [Google Scholar]
- Sarafian T., Pradel L. A., Henry J. P., Aunis D., Bader M. F. The participation of annexin II (calpactin I) in calcium-evoked exocytosis requires protein kinase C. J Cell Biol. 1991 Sep;114(6):1135–1147. doi: 10.1083/jcb.114.6.1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer D. D., Haigler H. T. Characterization of Ca2+-dependent phospholipid binding and phosphorylation of lipocortin I. J Biol Chem. 1987 May 15;262(14):6931–6937. [PubMed] [Google Scholar]
- Schlaepfer D. D., Haigler H. T. In vitro protein kinase C phosphorylation sites of placental lipocortin. Biochemistry. 1988 Jun 14;27(12):4253–4258. doi: 10.1021/bi00412a008. [DOI] [PubMed] [Google Scholar]
- Sharma R. K., Wang J. H. Calmodulin and Ca2+-dependent phosphorylation and dephosphorylation of 63-kDa subunit-containing bovine brain calmodulin-stimulated cyclic nucleotide phosphodiesterase isozyme. J Biol Chem. 1986 Jan 25;261(3):1322–1328. [PubMed] [Google Scholar]
- Turgeon J. L., Cooper R. H., Waring D. W. Membrane-specific association of annexin I and annexin II in anterior pituitary cells. Endocrinology. 1991 Jan;128(1):96–102. doi: 10.1210/endo-128-1-96. [DOI] [PubMed] [Google Scholar]
- Varticovski L., Chahwala S. B., Whitman M., Cantley L., Schindler D., Chow E. P., Sinclair L. K., Pepinsky R. B. Location of sites in human lipocortin I that are phosphorylated by protein tyrosine kinases and protein kinases A and C. Biochemistry. 1988 May 17;27(10):3682–3690. doi: 10.1021/bi00410a024. [DOI] [PubMed] [Google Scholar]
- Wallner B. P., Mattaliano R. J., Hession C., Cate R. L., Tizard R., Sinclair L. K., Foeller C., Chow E. P., Browing J. L., Ramachandran K. L. Cloning and expression of human lipocortin, a phospholipase A2 inhibitor with potential anti-inflammatory activity. Nature. 1986 Mar 6;320(6057):77–81. doi: 10.1038/320077a0. [DOI] [PubMed] [Google Scholar]
- Wang W., Creutz C. E. Regulation of the chromaffin granule aggregating activity of annexin I by phosphorylation. Biochemistry. 1992 Oct 20;31(41):9934–9939. doi: 10.1021/bi00156a011. [DOI] [PubMed] [Google Scholar]
- William F., Haigler H. T., Kraft A. S. Lack of phosphorylation of lipocortin I in A431 epidermoid carcinoma cells treated with phorbol esters. Biochem Biophys Res Commun. 1989 Apr 28;160(2):474–479. doi: 10.1016/0006-291x(89)92457-1. [DOI] [PubMed] [Google Scholar]
- Zaks W. J., Creutz C. E. Ca(2+)-dependent annexin self-association on membrane surfaces. Biochemistry. 1991 Oct 8;30(40):9607–9615. doi: 10.1021/bi00104a007. [DOI] [PubMed] [Google Scholar]
- Zokas L., Glenney J. R., Jr The calpactin light chain is tightly linked to the cytoskeletal form of calpactin I: studies using monoclonal antibodies to calpactin subunits. J Cell Biol. 1987 Nov;105(5):2111–2121. doi: 10.1083/jcb.105.5.2111. [DOI] [PMC free article] [PubMed] [Google Scholar]