Abstract
BACKGROUND
Recent research has underscored the potentially protective role of dietary antioxidants against chronic conditions, such as cardiovascular diseases and stroke. The composite dietary antioxidant index (CDAI), which reflects the overall intake of key dietary antioxidants, has been identified as a crucial metric for exploring this relationship. Although previous research has shown a negative correlation between CDAI levels and stroke risk in prediabetic individuals, there remains a substantial gap in understanding this association among individuals with dia-betes, who are at an inherently greater risk for cerebrovascular events.
AIM
To investigate the association between CDAI and stroke risk in individuals with diabetes.
METHODS
Using a cross-sectional study design, this investigation analyzed data from the National Health and Nutrition Examination Survey spanning from 2003 to 2018 that included 6735 participants aged over 20 years with diabetes. The CDAI was calculated from 24-h dietary recalls to assess intake of key antioxidants: Vitamins A, C, and E; carotenoids; selenium; and zinc. Multivariate logistic regression and restricted cubic spline analysis were used to rigorously examine the relationship between CDAI and stroke risk.
RESULTS
The participant cohort, with an average age of 59.5 years and a slight male majority, reflected the broader demographic characteristics of individuals with diabetes. The analysis revealed a strong inverse relationship between CDAI levels and stroke risk. Remarkably, those in the highest quintile of CDAI demonstrated a 43% lower prevalence of stroke compared to those in the lowest quintile, even after adjustments for various confounders. This finding not only highlights the negative association between CDAI and stroke risk but also underscores the significant potential of antioxidant-rich diets in reducing stroke prevalence among patients with diabetes.
CONCLUSION
Our findings suggested that CDAI was inversely associated with stroke prevalence among patients with diabetes. These results suggest incorporating antioxidant-rich foods into dietary regimens as a potential strategy for stroke prevention.
Keywords: Stroke, Diabetes, Composite dietary antioxidant index, National Health and Nutrition Examination Survey, Cross-sectional study
Core Tip: Previous research on the composite dietary antioxidant index (CDAI) and its impact on stroke risk among individuals with diabetes is limited. Our study addressed this gap by examining the association between higher CDAI scores and stroke prevalence. Our findings revealed that higher CDAI scores correlated with reduced stroke risk in this population, indicating that a diet rich in diverse antioxidants may play a crucial role in mitigating stroke risk among individuals with diabetes.
INTRODUCTION
Stroke is the second largest cause of death worldwide and a major contributor to long-term disability[1]. Additionally, the chronic condition of diabetes, which is prevalent globally, has been identified as a significant stroke risk factor[2,3]. Importantly, individuals with diabetes face a 1.5 to 2-fold greater risk of stroke than individuals without diabetes, which intensifies with a longer duration of diabetes[4]. Additionally, patients with diabetes tend to experience worse post-stroke outcomes and a heightened risk of recurrent strokes[5-10]. The 2024 ADA Standards of Care underscore the critical role of dietary management in diabetes to mitigate the risk of associated complications, including stroke[11].
Hyperglycemia, a hallmark of diabetes, is known to induce mitochondrial dysfunction and endoplasmic reticulum stress, leading to increased accumulation of reactive oxygen species (ROS). This buildup is crucial in causing cellular damage, hastening the development of diabetes-related complications like stroke[12-15]. Furthermore, hyperglycemia-induced oxidative stress is implicated in the upregulation of proinflammatory factors, triggering cellular apoptosis and impairing nitric oxide release[16]. This interplay between oxidative stress and inflammation forms a vicious cycle, exacerbating the progression of atherosclerosis, a critical process in stroke pathogenesis[17,18]. Emerging research suggests that increasing daily dietary antioxidant intake can increase plasma antioxidant levels, effectively mitigating oxidative stress-related damage[19,20]. Therefore, dietary modifications aimed at reducing oxidative stress present a promising approach for decreasing stroke risk in individuals with diabetes.
The efficacy of individual antioxidants, such as vitamin E and carotenoids, in reducing cardiovascular disease (CVD) risk remains controversial[21-26]. Measurements based on individual antioxidants, including vitamins A, C, E, zinc, selenium, and total carotenoids, may not accurately capture overall antioxidant intake. In contrast to individual antioxidants, the composite dietary antioxidant index (CDAI) provides a more extensive evaluation of total antioxidant consumption, which correlates with specific inflammatory biomarkers, including tumor necrosis factor-α and interleukin-1β[27,28]. Although elevated CDAI scores have been inversely associated with the risks of coronary heart disease, stroke, depression, and cancer[17,29-31], its specific impact on stroke risk among individuals with diabetes is yet to be studied. To the best of our knowledge, this study represents the initial investigation into the correlation between the CDAI and stroke risk within a diabetic population. Our cross-sectional analysis examines how CDAI relates to stroke risk among this group, which may inform future preventive and therapeutic approaches.
MATERIALS AND METHODS
Study population
The National Health and Nutrition Examination Survey (NHANES) supplies essential information related to the health and diet specifics of the United States population. Employing a stratified, multistage probability sampling approach, NHANES ensures a demographically representative sample. Data collection encompasses structured personal interviews in participants’ homes, comprehensive health assessments at mobile examination centers, and laboratory analysis of collected specimens. Every participant consented to the study by signing an informed consent form.
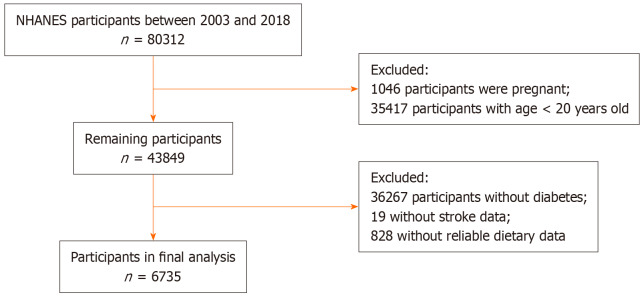
Our study focused on adults aged 20 years or older with hyperglycemia, participating in NHANES from 2003 to 2018. Diagnosis of diabetes mellitus in participants was established in accordance with the Standards of Medical Care in Diabetes. Criteria included a fasting plasma glucose level of at least 126 mg/dL (7.0 mmol/L), a hemoglobin A1c level of 6.5% or higher (48 mmol/mol), a self-reported diagnosis, or prior use of antidiabetic drugs. Exclusion criteria included being under 20 years of age, pregnancy at study onset, lack of dietary information, or absence of stroke condition data. After rigorous screening, 6735 individuals diagnosed with diabetes from the 2003-2018 NHANES cohorts were selected for analysis. Figure 1 illustrates the detailed screening process and the participant breakdown.
Figure 1.
Overview of the study design.
Dietary assessment
Dietary data were collected through structured interviews at the NHANES. Dietary intake data from the subjects was gathered via recall interviews conducted by experienced technicians, with participants asked to specify the food and beverages they consumed in the 24 h prior to the interview. This information facilitated the estimation of energy consumption, various nutrients, and other dietary components. The assessment of dietary antioxidants focused on six key antioxidants: Vitamin A; vitamin C; vitamin E; zinc; selenium; and total carotenoids. Total carotenoids were quantified as the sum of five distinct carotenoids: α-carotene; β-carotene; β-cryptoxanthin; lutein/zeaxanthin; and lycopene. To evaluate the cumulative impact of dietary antioxidants on stroke risk in patients with diabetes, we utilized a modified CDAI[32]. This index was derived from the six mentioned antioxidants and is calculated through the following steps. First, we stratify each antioxidant intake by sex, calculating the mean and standard deviation for males and females separately. Next, we standardize each individual’s antioxidant intake. For each individual, we subtract the sex-specific mean from their intake and then divide by the sex-specific standard deviation. In this way, we obtain the standardized intake of dietary antioxidants. Finally, we aggregate the standardized intakes of the six antioxidants to calculate each individual’s overall standardized intake of dietary antioxidants. The following formula is used for the calculation:
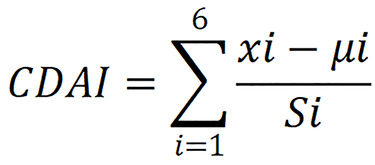
In this formula, xi represents the daily intake of antioxidant i; μi represents the sex-specific mean value of xi for the antioxidants i; Si represents the standard deviation for μi.
Diagnosis of stroke
Stroke diagnosis in this research was determined based on participants’ self-reported medical history of stroke[33]. During the NHANES interview process, participants were directly asked, “Have you ever been informed by a doctor or other health professional that you experienced a stroke?” A positive response to this question was considered indicative of a prior stroke occurrence. Conversely, a negative response was interpreted as an indication that the participant had no history of stroke. This method aligns with standard practices in epidemiological research, where self-reported medical histories are commonly used to identify previous health events.
Covariates
Informed by clinical expertise and prior research, this study carefully considered various potential confounders that might influence the association between the CDAI and stroke risk, integrating them as covariates in our analysis. These included demographic data such as age, sex, and race/ethnicity, educational levels spanning from below high school to college education and above, marital status categories, economic indicators like the family poverty-income ratio (PIR), lifestyle aspects like smoking habits and alcohol consumption, and health-related metrics like body mass index (BMI), daily energy intake, and histories of hypertension and hypercholesterolemia. This thorough selection of covariates ensured a robust analysis by accounting for potential confounders in the association under investigation.
Statistical analysis
This study utilized data from the NHANES, a complex, multistage, cluster research survey conducted by the National Center for Health Statistics in the United States. To ensure representativeness of the United States population, the analysis was weighted according to NHANES-specific sample weights. The study population was divided into two groups for analysis: Those with a history of stroke and those without. Continuous variables with a normal distribution were presented as means ± standard deviation, while those with skewed distributions were expressed as medians with interquartile range and subjected to log transformation to approximate normality prior to analysis. Categorical variables were shown as percentages (%).
To assess differences between groups, independent sample t-tests or non-parametric Mann-Whitney U tests were employed for continuous variables, and χ2 tests were utilized for categorical variables. Multivariate logistic regression analyses were performed to explore the relationship between CDAI and stroke. Three distinct models were employed: An unadjusted model 1; model 2 adjusted for demographic variables such as age, sex, race, education level, marital status, and family PIR; and model 3 that included additional adjustments for lifestyle and comorbidity like alcohol consumption, smoking status, BMI, energy intake, hypertension, and hypercholesterolemia.
Results were presented as odds ratios (ORs) and 95% confidence intervals (CI). The dose-response relationship between CDAI and stroke risk was analyzed using restricted cubic spline functions. Additionally, subgroup analyses were conducted to further validate the stability of the results. All statistical analyses were performed with R software, version 4.3.2 (http://www.R-project.org). A two-tailed P value of less than 0.05 was considered statistically significant.
RESULTS
In this study, 6735 participants from the NHANES dataset were rigorously screened and included in the analysis. The mean age of the subjects was 59.5 ± 13.6 years, and 51.5% of the participants were males. Among these, 611 individuals were identified as having a history of stroke. The baseline characteristics of the study population, encompassing age, sex, race, education, marital status, family PIR, alcohol consumption status (drinker), smoking status, BMI, and histories of hypertension and hypercholesterolemia, are detailed in Table 1. A comparative analysis revealed significant distinctions in several clinical characteristics between the stroke and non-stroke groups. Notably, the stroke group comprised older participants (P < 0.001), and significant racial composition differences were observed (P < 0.001). Additionally, the stroke group demonstrated lower levels of educational and economic status compared to the non-stroke group. Prevalence rates of hypertension and hypercholesterolemia were also found to be significantly higher in the stroke group, underscoring the potential impact of these conditions on stroke occurrence. The non-stroke group exhibited significantly higher levels of vitamin A, vitamin E, zinc, selenium, and carotenoids compared to their counterparts in the stroke group (Table 2). However, no significant difference in vitamin C was observed between the two groups (P = 0.093).
Table 1.
Characteristics of subjects with or without stroke
| Characteristics | Total, n = 6735 | Non-stroke, n = 6124 | Stroke, n = 611 | P value |
| Age in years | 59.5 ± 13.6 | 58.8 ± 13.6 | 67.4 ± 10.7 | < 0.001 |
| Sex | 0.156 | |||
| Male | 51.5 | 51.8 | 48.7 | |
| Female | 48.5 | 48.2 | 51.3 | |
| Race | < 0.001 | |||
| Non-Hispanic White | 60.7 | 60.3 | 65.0 | |
| Non-Hispanic Black | 15.4 | 15.2 | 17.2 | |
| Mexican American | 9.8 | 10.1 | 6.4 | |
| Other Hispanic | 5.7 | 6.0 | 2.5 | |
| Other race | 8.4 | 8.4 | 8.8 | |
| Education | < 0.001 | |||
| Lower than high school | 10.6 | 10.4 | 13.2 | |
| High school | 39.4 | 38.6 | 48.1 | |
| Higher than high school | 50.0 | 51.0 | 38.6 | |
| Marital status | 0.053 | |||
| Married/living with partner | 62.5 | 63.3 | 54.7 | |
| Widowed/divorced/separated | 27.9 | 26.8 | 40.1 | |
| Never married | 9.6 | 10.0 | 5.2 | |
| Poverty income ratio | < 0.001 | |||
| ≤ 1.30 | 26.2 | 25.5 | 33.1 | |
| 1.3 to ≤ 3.5 | 39.7 | 39.1 | 47.3 | |
| > 3.5 | 34.1 | 35.4 | 19.6 | |
| Smoking status | 0.007 | |||
| Nonsmoker | 48.6 | 49.2 | 42.7 | |
| Former smoker | 34.7 | 34.4 | 37.1 | |
| Current smoker | 16.7 | 16.4 | 20.2 | |
| Alcohol use | 0.002 | |||
| No | 33.3 | 32.7 | 40.1 | |
| Yes | 66.7 | 67.3 | 59.9 | |
| Energy intake, kcal | 1940.82 ± 900.52 | 1967.09 ± 909.80 | 1655.64 ± 734.62 | < 0.001 |
| BMI in kg/m2 | 0.429 | |||
| < 25.0 | 11.77 | 11.77 | 11.86 | |
| 25 to < 30 | 25.68 | 25.88 | 23.36 | |
| ≥ 30 | 62.55 | 62.35 | 64.78 | |
| Hypertension | < 0.001 | |||
| No | 34.3 | 36.0 | 16.4 | |
| Yes | 65.7 | 64.1 | 83.6 | |
| Hypercholesterolemia | 0.002 | |||
| No | 38.6 | 39.1 | 32.5 | |
| Yes | 61.4 | 60.9 | 67.5 | |
| CDAI | 0.28 ± 3.97 | 0.38 ± 4.01 | -0.77 ± 3.29 | < 0.001 |
Data are presented as mean (SD) or n (%). BMI: Body mass index; CDAI: Composite dietary antioxidant index.
Table 2.
Comparison of each component of the composite dietary antioxidant index among the non-stroke group and stroke group
| Variables | Total, n = 6735 | Non-stroke, n = 6124 | Stroke, n = 611 | P value |
| Vitamin A in mcg | 449 (252-736) | 453 (255-743) | 400 (220-676) | 0.026 |
| Vitamin C in mg | 50.6 (21.4-105.2) | 51.3 (22.0-105.4) | 44.2 (18.0-101.3) | 0.093 |
| Vitamin E in mg | 5.99 (3.77-9.23) | 6.10 (3.81-9.38) | 5.13 (3.28-7.46) | < 0.001 |
| Zinc in mg | 8.99 (6.10-13.06) | 9.09 (6.20-13.18) | 7.81 (5.04-11.66) | < 0.001 |
| Selenium in mcg | 94.3 (65.1-131.2) | 95.9 (66.3-133.4) | 79.1 (55.8-111.5) | < 0.001 |
| Carotenoid in mcg | 4740.0 (1675.0-10809.0) | 4851.0 (1759.2-10902.8) | 3655.0 (1124.0-9827.0) | 0.001 |
Data are presented as the median (interquartile range).
As a continuous variable, CDAI exhibited a negative correlation with stroke prevalence, with an OR of 0.91 (95%CI: 0.89-0.94) in unadjusted logistic regression analysis (Table 3). After adjusting for potential confounders, the negative association between CDAI and stroke remained statistically significant (OR = 0.96, 95%CI: 0.92-0.99). Of note, individuals with the highest quintile of CDAI (Q5) displayed a 43% lower stroke risk compared to those in the lowest quintile (Q1) (OR = 0.57, 95%CI: 0.40-0.83). Moreover, a dose-response relationship was observed, indicating a gradual increase in stroke risk as CDAI levels decreased (Figure 2).
Table 3.
Association of composite dietary antioxidant index and stroke
| Factor |
Model 1
|
Model 2
|
Model 3
|
|||
|
OR (95%CI)
|
P value
|
OR (95%CI)
|
P value
|
OR (95%CI)
|
P value
|
|
| CDAI (continuous) | 0.91 (0.89-0.94) | < 0.001 | 0.93 (0.90-0.96) | < 0.001 | 0.96 (0.92-0.99) | 0.019 |
| CDAI-Q1 | Ref. | - | Ref. | - | Ref. | - |
| CDAI-Q2 | 0.70 (0.55-0.88) | 0.003 | 0.70 (0.55-0.89) | 0.004 | 0.74 (0.57-0.96) | 0.024 |
| CDAI-Q3 | 0.59 (0.46-0.75) | < 0.001 | 0.60 (0.46-0.77) | < 0.001 | 0.68 (0.51-0.90) | 0.007 |
| CDAI-Q4 | 0.56 (0.43-0.72) | < 0.001 | 0.62 (0.48-0.80) | < 0.001 | 0.72 (0.53- 0.98) | 0.037 |
| CDAI-Q5 | 0.38 (0.29-0.51) | < 0.001 | 0.46 (0.34-0.61) | < 0.001 | 0.57 (0.40-0.83) | 0.003 |
| P for trend | < 0.0001 | < 0.0001 | 0.007 | |||
Model 1 was unadjusted; Model 2 was adjusted for age, sex, race, education, marital status and poverty income ratio; Model 3 was further adjusted for drinker, smoker, body mass index, energy intake, hypertension and hypercholesterolemia. Subjects with a composite dietary antioxidant index in the lowest quintile group served as the reference group. CDAI: Composite dietary antioxidant index; CI: Confidence interval; OR: Odds ratio; Q: Quintile; Ref.: Reference.
Figure 2.
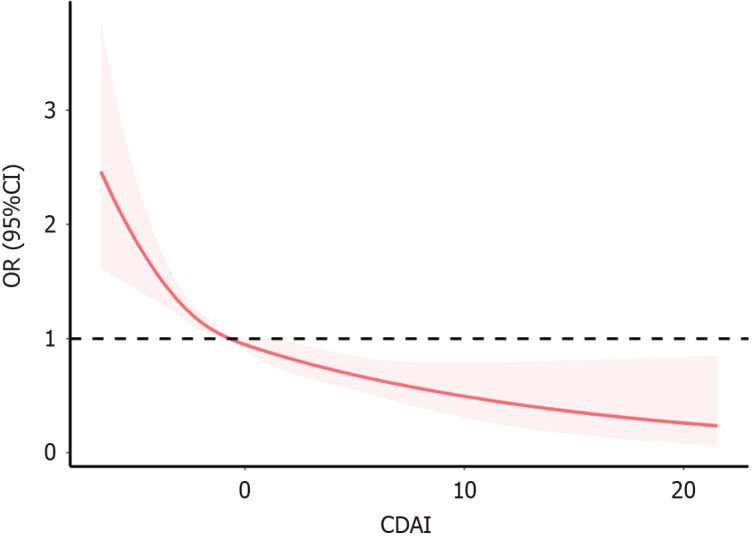
Restricted cubic spline plot of the association between composite dietary antioxidant index and stroke. The shaded part represented the 95% confidence interval (CI). CDAI: Composite dietary antioxidant index; OR: Odds ratio.
Additionally, a comprehensive subgroup analysis was conducted to further elucidate the association between CDAI and stroke risk across various demographic and clinical subgroups. These subgroups were delineated based on age, sex, BMI, race, smoking status, alcohol consumption, hypertension, and hypercholesterolemia (Table 4). However, the subgroup analysis did not reveal any significant statistical interactions, suggesting a consistent relationship between CDAI and stroke risk across these diverse groups.
Table 4.
Stratified analyses of the associations between the composite dietary antioxidant index and stroke
| Characteristics | Quintile 1 | Quintile 2 | Quintile 3 | Quintile 4 | Quintile 5 | P for interaction |
| Sex | 0.135 | |||||
| Male | Ref. | 0.88 (0.62-1.24) | 0.58 (0.39-0.88) | 0.63 (0.40-0.99) | 0.58 (0.34-0.98) | |
| Female | Ref. | 0.60 (0.40-0.88) | 0.77 (0.52-1.14) | 0.82 (0.53-1.26) | 0.56 (0.33-0.95) | |
| Age in years | 0.088 | |||||
| ≤ 60 | Ref. | 0.44 (0.25-0.78) | 0.46 (0.25-0.85) | 0.74 (0.41-1.34) | 0.41 (0.19-0.88) | |
| > 60 | Ref. | 0.88 (0.65-1.17) | 0.80 (0.58-1.11) | 0.74 (0.52-1.07) | 0.68 (0.45-1.03) | |
| BMI | 0.300 | |||||
| < 30 | Ref. | 0.65 (0.45-0.95) | 0.48 (0.31-0.74) | 0.57 (0.36-0.91) | 0.45 (0.26-0.79) | |
| ≥ 30 | Ref. | 0.81 (0.58-1.14) | 0.85 (0.60-1.21) | 0.86 (0.58-1.26) | 0.67 (0.42-1.07) | |
| Race | 0.424 | |||||
| White | Ref. | 0.75 (0.50-1.13) | 0.86 (0.55-1.33) | 0.94 (0.58-1.51) | 0.74 (0.42-1.31) | |
| Others | Ref. | 0.73 (0.52-1.02) | 0.56 (0.38-0.81) | 0.59 (0.39-0.88) | 0.48 (0.29-0.78) | |
| Smoker | 0.611 | |||||
| Yes | Ref. | 0.68 (0.49-0.95) | 0.54 (0.37-0.79) | 0.67 (0.45-1.00) | 0.48 (0.29-0.78) | |
| No | Ref. | 0.77 (0.51-1.16) | 0.83 (0.54-1.27) | 0.74 (0.46-1.19) | 0.67 (0.39-1.17) | |
| Drinker | 0.225 | |||||
| Yes | Ref. | 0.61 (0.42-0.88) | 0.44 (0.29-0.68) | 0.52 (0.33-0.81) | 0.40 (0.24-0.68) | |
| No | Ref. | 0.94 (0.59-1.48) | 0.89 (0.54-1.47) | 1.04 (0.61-1.78) | 0.84 (0.44-1.59) | |
| Hypertension | 0.843 | |||||
| Yes | Ref. | 0.75 (0.57-1.00) | 0.66 (0.48-0.90) | 0.71 (0.51-1.00) | 0.55 (0.36-0.82) | |
| No | Ref. | 0.69 (0.36-1.30) | 0.87 (0.44-1.71) | 0.84 (0.40-1.75) | 0.79 (0.32-1.91) | |
| Hypercholesterolemia | 0.375 | |||||
| Yes | Ref. | 0.68 (0.49-0.95) | 0.66 (0.46-0.95) | 0.57 (0.38-0.85) | 0.47 (0.29-0.76) | |
| No | Ref. | 0.87 (0.56-1.34) | 0.61 (0.37-1.00) | 1.01 (0.61-1.70) | 0.72 (0.38-1.34) |
The odds ratio and 95% confidence interval were obtained from multivariable logistic regression models after adjusting for age, sex, race, education, marital status, poverty income ratio, drinker, smoker, body mass index, energy intake, hypertension and hypercholesterolemia. BMI: Body mass index; Ref.: Reference.
DISCUSSION
In our study, higher CDAI scores were associated with a decreased prevalence of stroke specifically in individuals with diabetes, a novel focus on this high-risk group. The observed dose-response relationship indicated that the risk of stroke inversely correlated with CDAI levels, gradually increasing as CDAI decreased. Importantly, this association remained significant even after accounting for conventional risk factors such as BMI, hypertension, and hypercholesterolemia. These findings suggested that a higher intake of antioxidants, as quantified by the CDAI, may confer a protective effect against stroke in individuals with diabetes, highlighting the unique impact of antioxidants on this population’s heightened oxidative stress, a recognized risk factor for stroke.
Previous studies have not specifically focused on patients with diabetes when examining the relationship between dietary antioxidants and stroke. Our research is the first to highlight the significant role of CDAI in reducing stroke risk among individuals with diabetes, and through restricted cubic splines, we observed that as CDAI increased, the risk of stroke gradually decreased. Our study’s finding contributes to a growing understanding of the impact of diet on chronic inflammation and oxidative stress, key factors in metabolic diseases and cardiovascular health[34,35]. The CDAI, which is designed to reflect the anti-inflammatory potential of dietary components, underscores the role of antioxidants in neutralizing oxidative stress and is implicated in the pathogenesis of atherosclerosis and vascular diseases[36]. Diets rich in antioxidants, targeting ROS, may not only protect against CVD but also are inversely associated with all-cause and CVD-related mortality in adults with diabetes, providing unique insights into the dietary management of this high-risk group[17,37].
This aligns with research indicating that dietary antioxidants can act as neuroprotective agents, safeguarding brain tissues and potentially ameliorating conditions leading to stroke[18-20]. A deficiency in dietary antioxidants is postulated to elevate stroke risk, likely through mechanisms linked to oxidative stress. Excessive ROS, in conjunction with lipid peroxidation, neuroinflammatory responses, and blood-brain barrier disruption, contribute to brain tissue injury[2-4]. Moreover, post-ischemic ROS activity can stimulate nuclear transcription factors and trigger the release of proinflammatory factors, leading to a localized neuroinflammatory response[25].
Oxidative stress also plays a role in the destruction of tissue surrounding hematomas following cerebral hemorrhage[26], and the capacity of the central nervous system for maintaining redox homeostasis is vital for post-stroke brain tissue recovery[5]. The efficacy of antioxidants involves electron donation to free radicals, thus diminishing cellular damage and curbing inflammatory responses[14]. The protective potential of antioxidants extends to the prevention and management of atherosclerosis, a key stroke risk factor, by reducing ROS generation and preventing oxidative damage to lipoproteins[21,22].
The components of CDAI, especially vitamins A, C, and E, are crucial in this context. These non-enzymatic antioxidants help neutralize oxidative stress, a key mechanism in stroke pathogenesis. Vitamin E, for example, effectively scavenges free radicals and protects cell structures, including lipoproteins, from oxidative damage[38,39]. In adult stroke-prone spontaneously hypertensive rats, vitamins C and E mitigate oxidative stress, enhance vascular function and structure, and inhibit hypertension progression potentially through the modulation of enzyme systems responsible for free radical generation[40].
In a 22-year prospective population-based study, it was found that higher dietary intakes of antioxidant vitamins A, C, and E significantly reduced the risk of adverse cardiovascular outcomes among Chinese individuals[41]. Carotenoids offer neuroprotection through the inhibition of neuroinflammation, microglial activation, and the excitotoxic pathway, as well as by modulating autophagy, attenuating oxidative damage, and activating defensive antioxidant enzymes[42]. This protective effect was shown in a 13-year cohort study in which higher plasma levels of carotenoids, indicative of fruit and vegetable intake, were associated with a reduced risk of ischemic stroke[43]. In addition to vitamins, micronutrients in the CDAI, such as zinc and selenium, also contribute significantly to mitigating oxidative stress and inflammation. Zinc acts by suppressing oxidative stress through inhibition of NADPH oxidase[44,45], while selenium, along with selenoproteins, prevents cellular damage due to lipid peroxidation[46].
The CDAI is a widely utilized metric in nutritional research to explore the correlation between dietary antioxidant intake and diverse diseases, particularly those associated with oxidative stress. Previous investigations have indicated a negative correlation between the CDAI and diabetes prevalence[47]. Additionally, research has shown that higher CDAI levels might mitigate the risk of diabetic kidney disease and mortality among individuals with diabetes[48]. Moreover, higher CDAI levels exhibit a significant association with reduced CVD mortality in individuals with diabetes, with those in the highest quartile experiencing a 53% decrease in risk compared to those in the lowest quartile[37]. Collectively, these findings suggest that higher CDAI levels confer protective benefits against multiple health risks, particularly cardiovascular risks, for individuals with diabetes.
Aligning with our findings in individuals with diabetes, the study in the general population has observed an inverse relationship between CDAI and stroke[29]. In the general population, the highest tertile of CDAI was associated with a 23% reduction in stroke prevalence compared to the lowest tertile (OR = 0.77; 95%CI: 0.64-0.92). Remarkably, in our cohort of individuals with diabetes, those in the highest quintile of CDAI exhibited a 43% lower prevalence of stroke compared to those in the lowest quintile, even after adjusting for various confounders. However, it is noteworthy that our study population had a higher prevalence of hypertension, indicating a higher baseline risk for stroke.
However, there exists a degree of heterogeneity in the findings across various studies. While our research supports the notion that increased antioxidant intake is correlated with reduced stroke risk, some studies have reported no significant associations or conflicting results[39,49]. This discrepancy could stem from variations in study designs, population demographics, and the specific types of antioxidants analyzed. For instance, our study’s focus on a composite index rather than individual antioxidants might explain some of these differences. Moreover, the relationship between dietary antioxidants and stroke risk is intricate, as demonstrated by studies showing varied impacts among different sex and age groups. For example, a Swedish cohort study observed a link between dietary antioxidants and ischemic stroke risk in females but not in males. In light of these findings, our study underscored the potential benefit of a holistic dietary approach, emphasizing a high antioxidant intake in managing stroke risk among individuals with diabetes. Nevertheless, the specific mechanisms underlying the CDAI-stroke relationship warrant further investigation to elucidate the complex interplay between diet, oxidative stress, and vascular health in patients with diabetes.
Our study’s strength lies in its use of a comprehensive, multicomponent index to assess dietary antioxidant intake and its large, representative sample. Despite the strengths of our study, it is important to acknowledge certain limitations. First, the cross-sectional study was unable to establish a causal relationship between the CDAI and stroke. Future research should aim to elucidate this relationship through long-term follow-up and cohort studies. Second, the assessment of dietary intake, which forms the basis for calculating CDAI, is not without potential errors and inaccuracies. Dietary data collected through self-report can be subject to recall bias and may not accurately capture day-to-day variations in individual dietary patterns. Third, stroke assessment relied on questionnaire responses, lacking a more detailed classification of stroke types. Fourth, although numerous relevant covariates were included in the analysis, it is possible that not all confounding factors were accounted for. Unidentified confounders might skew the results. Future studies should aim for more comprehensive control of these factors to validate and refine our findings. Moreover, our study’s findings are most applicable to the American population, given that dietary habits can vary significantly across different racial and ethnic groups. This geographic and cultural specificity limits the generalizability of our conclusions. Therefore, further research in diverse populations is needed to broaden our understanding of the association between CDAI and stroke risk globally.
CONCLUSION
In summary, this cross-sectional study found that adults diagnosed with diabetes in the United States with high levels of the CDAI, which measures the overall antioxidant quality of the diet, tend to have a lower risk of stroke. Furthermore, our smooth curve fitting analysis revealed a negative relationship between CDAI and stroke. Based on these findings, it is recommended that patients with diabetes maintain an appropriate intake of dietary antioxidants to increase their CDAI, thereby reducing stroke-related risk factors. In the future, rigorous prospective studies are imperative to validate our findings and provide deeper insights.
ACKNOWLEDGEMENTS
We are grateful to the participants and the researchers involved in the NHANES.
Footnotes
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Endocrinology and metabolism
Country of origin: China
Peer-review report’s classification
Scientific Quality: Grade B, Grade C, Grade C, Grade C, Grade C
Novelty: Grade B, Grade B, Grade C
Creativity or Innovation: Grade B, Grade B, Grade B
Scientific Significance: Grade B, Grade B, Grade C
P-Reviewer: Horowitz M; Li SY; Nagase T; Qureshi W; Soriano-Ursúa MA S-Editor: Qu XL L-Editor: Filipodia P-Editor: Zheng XM
Contributor Information
Hong-Qiang Zhang, Department of Cardiology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Jie Shi, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Tong Yue, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Jia-Hao Weng, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Xu-Lin Wang, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Hao Wang, Graduate School, Bengbu Medical College, Bengbu 233030, Anhui Province, China.
Xiao-Yu Su, Graduate School, Bengbu Medical College, Bengbu 233030, Anhui Province, China.
Xue-Ying Zheng, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Si-Hui Luo, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Yu Ding, Department of Endocrinology, Centre for Leading Medicine and Advanced Technologies of IHM, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China.
Chao-Fan Wang, Department of Endocrinology and Metabolism, The Third Affiliated Hospital of Sun Yat-sen University, Guangdong Provincial Key Laboratory of Diabetology, Guangzhou 510630, Guangdong Province, China. wangchf25@mail.sysu.edu.cn.
References
- 1.GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021;20:795–820. doi: 10.1016/S1474-4422(21)00252-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, Benetos A, Biffi A, Boavida JM, Capodanno D, Cosyns B, Crawford C, Davos CH, Desormais I, Di Angelantonio E, Franco OH, Halvorsen S, Hobbs FDR, Hollander M, Jankowska EA, Michal M, Sacco S, Sattar N, Tokgozoglu L, Tonstad S, Tsioufis KP, van Dis I, van Gelder IC, Wanner C, Williams B ESC Scientific Document Group. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur J Prev Cardiol. 2022;29:5–115. doi: 10.1093/eurjpc/zwab154. [DOI] [PubMed] [Google Scholar]
- 3.Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119. doi: 10.1016/j.diabres.2021.109119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Emerging Risk Factors Collaboration, Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, Di Angelantonio E, Ingelsson E, Lawlor DA, Selvin E, Stampfer M, Stehouwer CD, Lewington S, Pennells L, Thompson A, Sattar N, White IR, Ray KK, Danesh J. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375:2215–2222. doi: 10.1016/S0140-6736(10)60484-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sun Y, Toh MP. Impact of diabetes mellitus (DM) on the health-care utilization and clinical outcomes of patients with stroke in Singapore. Value Health. 2009;12 Suppl 3:S101–S105. doi: 10.1111/j.1524-4733.2009.00639.x. [DOI] [PubMed] [Google Scholar]
- 6.Eriksson M, Carlberg B, Eliasson M. The disparity in long-term survival after a first stroke in patients with and without diabetes persists: the Northern Sweden MONICA study. Cerebrovasc Dis. 2012;34:153–160. doi: 10.1159/000339763. [DOI] [PubMed] [Google Scholar]
- 7.Zhang L, Li X, Wolfe CDA, O'Connell MDL, Wang Y. Diabetes As an Independent Risk Factor for Stroke Recurrence in Ischemic Stroke Patients: An Updated Meta-Analysis. Neuroepidemiology. 2021;55:427–435. doi: 10.1159/000519327. [DOI] [PubMed] [Google Scholar]
- 8.Lo JW, Crawford JD, Samaras K, Desmond DW, Köhler S, Staals J, Verhey FRJ, Bae HJ, Lee KJ, Kim BJ, Bordet R, Cordonnier C, Dondaine T, Mendyk AM, Lee BC, Yu KH, Lim JS, Kandiah N, Chander RJ, Yatawara C, Lipnicki DM, Sachdev PS STROKOG Collaboration*. Association of Prediabetes and Type 2 Diabetes With Cognitive Function After Stroke: A STROKOG Collaboration Study. Stroke. 2020;51:1640–1646. doi: 10.1161/STROKEAHA.119.028428. [DOI] [PubMed] [Google Scholar]
- 9.Echouffo-Tcheugui JB, Xu H, Matsouaka RA, Xian Y, Schwamm LH, Smith EE, Bhatt DL, Hernandez AF, Heidenreich PA, Fonarow GC. Diabetes and long-term outcomes of ischaemic stroke: findings from Get With The Guidelines-Stroke. Eur Heart J. 2018;39:2376–2386. doi: 10.1093/eurheartj/ehy036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Li HW, Yang MC, Chung KP. Predictors for readmission of acute ischemic stroke in Taiwan. J Formos Med Assoc. 2011;110:627–633. doi: 10.1016/j.jfma.2011.08.004. [DOI] [PubMed] [Google Scholar]
- 11.American Diabetes Association Professional Practice Committee. 3. Prevention or Delay of Diabetes and Associated Comorbidities: Standards of Care in Diabetes-2024. Diabetes Care. 2024;47:S43–S51. doi: 10.2337/dc24-S003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813–820. doi: 10.1038/414813a. [DOI] [PubMed] [Google Scholar]
- 13.Son SM. Reactive oxygen and nitrogen species in pathogenesis of vascular complications of diabetes. Diabetes Metab J. 2012;36:190–198. doi: 10.4093/dmj.2012.36.3.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maritim AC, Sanders RA, Watkins JB 3rd. Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol. 2003;17:24–38. doi: 10.1002/jbt.10058. [DOI] [PubMed] [Google Scholar]
- 15.Zhu G, Wang X, Chen L, Lenahan C, Fu Z, Fang Y, Yu W. Crosstalk Between the Oxidative Stress and Glia Cells After Stroke: From Mechanism to Therapies. Front Immunol. 2022;13:852416. doi: 10.3389/fimmu.2022.852416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fiorentino TV, Prioletta A, Zuo P, Folli F. Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr Pharm Des. 2013;19:5695–5703. doi: 10.2174/1381612811319320005. [DOI] [PubMed] [Google Scholar]
- 17.Ma R, Zhou X, Zhang G, Wu H, Lu Y, Liu F, Chang Y, Ding Y. Association between composite dietary antioxidant index and coronary heart disease among US adults: a cross-sectional analysis. BMC Public Health. 2023;23:2426. doi: 10.1186/s12889-023-17373-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Clemens DL, Duryee MJ, Hall JH, Thiele GM, Mikuls TR, Klassen LW, Zimmerman MC, Anderson DR. Relevance of the antioxidant properties of methotrexate and doxycycline to their treatment of cardiovascular disease. Pharmacol Ther. 2020;205:107413. doi: 10.1016/j.pharmthera.2019.107413. [DOI] [PubMed] [Google Scholar]
- 19.Daneshzad E, Keshavarz SA, Qorbani M, Larijani B, Azadbakht L. Dietary total antioxidant capacity and its association with sleep, stress, anxiety, and depression score: A cross-sectional study among diabetic women. Clin Nutr ESPEN. 2020;37:187–194. doi: 10.1016/j.clnesp.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 20.Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002;360:23–33. [Google Scholar]
- 21.O'Connor EA, Evans CV, Ivlev I, Rushkin MC, Thomas RG, Martin A, Lin JS. Vitamin and Mineral Supplements for the Primary Prevention of Cardiovascular Disease and Cancer: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2022;327:2334–2347. doi: 10.1001/jama.2021.15650. [DOI] [PubMed] [Google Scholar]
- 22.US Preventive Services Task Force, Mangione CM, Barry MJ, Nicholson WK, Cabana M, Chelmow D, Coker TR, Davis EM, Donahue KE, Doubeni CA, Jaén CR, Kubik M, Li L, Ogedegbe G, Pbert L, Ruiz JM, Stevermer J, Wong JB. Vitamin, Mineral, and Multivitamin Supplementation to Prevent Cardiovascular Disease and Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2022;327:2326–2333. doi: 10.1001/jama.2022.8970. [DOI] [PubMed] [Google Scholar]
- 23.Firuzi O, Miri R, Tavakkoli M, Saso L. Antioxidant therapy: current status and future prospects. Curr Med Chem. 2011;18:3871–3888. doi: 10.2174/092986711803414368. [DOI] [PubMed] [Google Scholar]
- 24.Siti HN, Kamisah Y, Kamsiah J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review) Vascul Pharmacol. 2015;71:40–56. doi: 10.1016/j.vph.2015.03.005. [DOI] [PubMed] [Google Scholar]
- 25.Liguori I, Russo G, Curcio F, Bulli G, Aran L, Della-Morte D, Gargiulo G, Testa G, Cacciatore F, Bonaduce D, Abete P. Oxidative stress, aging, and diseases. Clin Interv Aging. 2018;13:757–772. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Aune D, Keum N, Giovannucci E, Fadnes LT, Boffetta P, Greenwood DC, Tonstad S, Vatten LJ, Riboli E, Norat T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: a systematic review and dose-response meta-analysis of prospective studies. Am J Clin Nutr. 2018;108:1069–1091. doi: 10.1093/ajcn/nqy097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Luu HN, Wen W, Li H, Dai Q, Yang G, Cai Q, Xiang YB, Gao YT, Zheng W, Shu XO. Are dietary antioxidant intake indices correlated to oxidative stress and inflammatory marker levels? Antioxid Redox Signal. 2015;22:951–959. doi: 10.1089/ars.2014.6212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wright ME, Mayne ST, Stolzenberg-Solomon RZ, Li Z, Pietinen P, Taylor PR, Virtamo J, Albanes D. Development of a comprehensive dietary antioxidant index and application to lung cancer risk in a cohort of male smokers. Am J Epidemiol. 2004;160:68–76. doi: 10.1093/aje/kwh173. [DOI] [PubMed] [Google Scholar]
- 29.Wang T, Liu H, Wei X. Association between the Composite Dietary Antioxidant Index and Stroke: A cross-sectional Study. Biol Trace Elem Res. 2023 doi: 10.1007/s12011-023-04011-5. [DOI] [PubMed] [Google Scholar]
- 30.Yu YC, Paragomi P, Wang R, Jin A, Schoen RE, Sheng LT, Pan A, Koh WP, Yuan JM, Luu HN. Composite dietary antioxidant index and the risk of colorectal cancer: Findings from the Singapore Chinese Health Study. Int J Cancer. 2022;150:1599–1608. doi: 10.1002/ijc.33925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhao L, Zhang X, Guo S, Han K, Sun Y, Li X, Yan Z, WeiPeng Relationship between composite dietary antioxidant index and depression among overweight and obese adults. J Affect Disord. 2023;341:358–365. doi: 10.1016/j.jad.2023.08.140. [DOI] [PubMed] [Google Scholar]
- 32.Maugeri A, Hruskova J, Jakubik J, Kunzova S, Sochor O, Barchitta M, Agodi A, Bauerova H, Medina-Inojosa JR, Vinciguerra M. Dietary antioxidant intake decreases carotid intima media thickness in women but not in men: A cross-sectional assessment in the Kardiovize study. Free Radic Biol Med. 2019;131:274–281. doi: 10.1016/j.freeradbiomed.2018.12.018. [DOI] [PubMed] [Google Scholar]
- 33.Xu Q, Qian X, Sun F, Liu H, Dou Z, Zhang J. Independent and joint associations of dietary antioxidant intake with risk of post-stroke depression and all-cause mortality. J Affect Disord. 2023;322:84–90. doi: 10.1016/j.jad.2022.11.013. [DOI] [PubMed] [Google Scholar]
- 34.Maiuolo J, Gliozzi M, Carresi C, Musolino V, Oppedisano F, Scarano F, Nucera S, Scicchitano M, Bosco F, Macri R, Ruga S, Cardamone A, Coppoletta A, Mollace A, Cognetti F, Mollace V. Nutraceuticals and Cancer: Potential for Natural Polyphenols. Nutrients. 2021;13 doi: 10.3390/nu13113834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ignarro LJ, Balestrieri ML, Napoli C. Nutrition, physical activity, and cardiovascular disease: an update. Cardiovasc Res. 2007;73:326–340. doi: 10.1016/j.cardiores.2006.06.030. [DOI] [PubMed] [Google Scholar]
- 36.Wanschel ACBA, Guizoni DM, Lorza-Gil E, Salerno AG, Paiva AA, Dorighello GG, Davel AP, Balkan W, Hare JM, Oliveira HCF. The Presence of Cholesteryl Ester Transfer Protein (CETP) in Endothelial Cells Generates Vascular Oxidative Stress and Endothelial Dysfunction. Biomolecules. 2021;11 doi: 10.3390/biom11010069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yang C, Yang Q, Peng X, Li X, Rao G. Associations of composite dietary antioxidant index with cardiovascular disease mortality among patients with type 2 diabetes. Diabetol Metab Syndr. 2023;15:131. doi: 10.1186/s13098-023-01109-7. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 38.Camps-Vilaro A, Subirana I, Ramos R, Cainzos-Achirica M, Tizon-Marcos H, Fito M, Degano IR, Marrugat J. Five-Year Changes in Inflammatory, Metabolic, and Oxidative Biomarkers and 10-Year Cardiovascular Disease Incidence: The REGICOR Cohort Study. Int J Mol Sci. 2023;24 doi: 10.3390/ijms24097934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.An P, Wan S, Luo Y, Luo J, Zhang X, Zhou S, Xu T, He J, Mechanick JI, Wu WC, Ren F, Liu S. Micronutrient Supplementation to Reduce Cardiovascular Risk. J Am Coll Cardiol. 2022;80:2269–2285. doi: 10.1016/j.jacc.2022.09.048. [DOI] [PubMed] [Google Scholar]
- 40.Chen X, Touyz RM, Park JB, Schiffrin EL. Antioxidant effects of vitamins C and E are associated with altered activation of vascular NADPH oxidase and superoxide dismutase in stroke-prone SHR. Hypertension. 2001;38:606–611. doi: 10.1161/hy09t1.094005. [DOI] [PubMed] [Google Scholar]
- 41.Lee CH, Chan RSM, Wan HYL, Woo YC, Cheung CYY, Fong CHY, Cheung BMY, Lam TH, Janus E, Woo J, Lam KSL. Dietary Intake of Anti-Oxidant Vitamins A, C, and E Is Inversely Associated with Adverse Cardiovascular Outcomes in Chinese-A 22-Years Population-Based Prospective Study. Nutrients. 2018;10 doi: 10.3390/nu10111664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Manochkumar J, Doss CGP, El-Seedi HR, Efferth T, Ramamoorthy S. The neuroprotective potential of carotenoids in vitro and in vivo. Phytomedicine. 2021;91:153676. doi: 10.1016/j.phymed.2021.153676. [DOI] [PubMed] [Google Scholar]
- 43.Hak AE, Ma J, Powell CB, Campos H, Gaziano JM, Willett WC, Stampfer MJ. Prospective study of plasma carotenoids and tocopherols in relation to risk of ischemic stroke. Stroke. 2004;35:1584–1588. doi: 10.1161/01.STR.0000132197.67350.bd. [DOI] [PubMed] [Google Scholar]
- 44.Prasad AS. Zinc: an antioxidant and anti-inflammatory agent: role of zinc in degenerative disorders of aging. J Trace Elem Med Biol. 2014;28:364–371. doi: 10.1016/j.jtemb.2014.07.019. [DOI] [PubMed] [Google Scholar]
- 45.Sun R, Wang J, Feng J, Cao B. Zinc in Cognitive Impairment and Aging. Biomolecules. 2022;12 doi: 10.3390/biom12071000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Roman M, Jitaru P, Barbante C. Selenium biochemistry and its role for human health. Metallomics. 2014;6:25–54. doi: 10.1039/c3mt00185g. [DOI] [PubMed] [Google Scholar]
- 47.Chen X, Lu H, Chen Y, Sang H, Tang Y, Zhao Y. Composite dietary antioxidant index was negatively associated with the prevalence of diabetes independent of cardiovascular diseases. Diabetol Metab Syndr. 2023;15:183. doi: 10.1186/s13098-023-01150-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang J, Chen Y, Zou L, Jin L, Yang B, Shu Y, Gong R. Dose-response relationship between dietary antioxidant intake and diabetic kidney disease in the US adults with diabetes. Acta Diabetol. 2023;60:1365–1375. doi: 10.1007/s00592-023-02125-9. [DOI] [PubMed] [Google Scholar]
- 49.Ascherio A, Rimm EB, Hernán MA, Giovannucci E, Kawachi I, Stampfer MJ, Willett WC. Relation of consumption of vitamin E, vitamin C, and carotenoids to risk for stroke among men in the United States. Ann Intern Med. 1999;130:963–970. doi: 10.7326/0003-4819-130-12-199906150-00003. [DOI] [PubMed] [Google Scholar]