Abstract
Chronic traumatic encephalopathy (CTE) is a neurodegenerative disease caused by repetitive head impacts (RHI) and pathologically defined as neuronal phosphorylated tau aggregates around small blood vessels and concentrated at sulcal depths. Cross-sectional studies suggest that tau inclusions follow a stereotyped pattern that begins in the neocortex in low stage disease, followed by involvement of the medial temporal lobe and subcortical regions with significant neocortical burden in high stage CTE. Here, we define a subset of brain donors with high stage CTE and with a low overall cortical burden of tau inclusions (mean semiquantitative value ≤1) and classify them as cortical-sparing CTE (CSCTE). Of 620 brain donors with pathologically diagnosed CTE, 66 (11%) met criteria for CSCTE. Compared to typical high stage CTE, those with CSCTE had a similar age at death and years of contact sports participation and were less likely to carry apolipoprotein ε4 (p < 0.05). CSCTE had less overall tau pathology severity, but a proportional increase of disease burden in medial temporal lobe and brainstem regions compared to the neocortex (p’s < 0.001). CSCTE also had lower prevalence of comorbid neurodegenerative disease. Clinically, CSCTE participants were less likely to have dementia (p = 0.023) and had less severe cognitive difficulties (as reported by informants using the Functional Activities Questionnaire (FAQ); p < 0.001, meta-cognitional index T score; p = 0.002 and Cognitive Difficulties Scale (CDS); p < 0.001,) but had an earlier onset age of behavioral (p = 0.006) and Parkinsonian motor (p = 0.013) symptoms when compared to typical high stage CTE. Other comorbid tauopathies likely contributed in part to these differences: when cases with concurrent Alzheimer dementia or frontal temporal lobar degeneration with tau pathology were excluded, differences were largely retained, but only remained significant for FAQ (p = 0.042), meta-cognition index T score (p = 0.014) and age of Parkinsonian motor symptom onset (p = 0.046). Overall, CSCTE appears to be a distinct subtype of high stage CTE with relatively greater involvement of subcortical and brainstem regions and less severe cognitive symptoms.
Keywords: Chronic traumatic encephalopathy, Neurodegenerative disease, Repetitive head impacts, Traumatic brain injury
Introduction
Chronic traumatic encephalopathy (CTE) is a neurodegenerative disease at least partially caused by repetitive head impacts (RHI). Clinically, diagnostic criteria for traumatic encephalopathy syndrome (TES) have been proposed and updated [19]. The criteria include cognitive impairment (i.e., in episodic memory and/or executive function) and/or neurobehavioral dysregulation (e.g., impulsivity, “short fuse”) as core clinical features. Supportive features include depression, anxiety, apathy, motor signs and symptoms, and a clinical history of delayed symptomatology after RHI. As is true in other neurodegenerative diseases, pathological subtypes and comorbid pathologies may contribute to the significant clinical heterogeneity in TES.
CTE is defined by the presence of intraneuronal hyperphosphorylated tau (p-tau) inclusions that occur around small blood vessels and at the depths of the cortical sulci [41]. There is a spectrum of pathology in CTE associated with age and duration of exposure to RHI. Thus, CTE is typically progressive with increasing severity associated with level of contact sports play (particularly American football), the duration of play, neuroinflammation, apolipoprotein ε4 (APOE4) allele frequency, and age at death [4, 7, 23, 26–28]. Four pathological stages of CTE have been described [24, 25]. Stage I is characterized by 1 or 2 isolated foci of p-tau neurofibrillary tangles (NFTs) along with dot-like neurites aggregated around small blood vessels at the depths of the sulci in the frontal cortex. Stage II is characterized by three or more of these lesions in multiple cortical lobes. In stage III, p-tau inclusions are present in both the bank and the gyral crest in multiple cortical lobes and involve the medial temporal lobe, brainstem, striatum, thalamus, and mamillary body. Finally, in stage IV, p-tau NFTs are abundant within the cortex with pronounced degeneration of the medial temporal lobe as well as involvement of the diencephalon, brainstem, cervical spinal cord, and dentate nucleus of the cerebellum. In 2021 a simplified Low/High staging system was proposed that involved the determination of neurofibrillary tangles in 10 different regions or patterns [9]. These included involvement of the gyral side adjacent to CTE lesion, gyral crest adjacent to lesion, superficial cortical laminae, CA4 of hippocampus, CA2 of hippocampus, entorhinal cortex, amygdala, thalamus, mammillary body and the dentate nucleus of the cerebellum. Low stage is defined by p-tau pathology within less than five of these and high stage in five or more. In general, Low stage CTE corresponds to McKee stages I–II and High stage to III–IV [25, 39]. Both McKee stages I–IV and Low/High stages are associated with increasing years of RHI, age, and APOE4 status, suggesting that CTE is typically a progressive disease that begins and worsens in severity in the neocortex and later involves medial temporal lobe and subcortical structures.
In both CTE staging systems, however, there is a subset of high stage (III–IV) individuals with relatively less cortical p-tau pathology given the subcortical burden. For instance, Alosco et al. performed hierarchical clustering on regional p-tau pathology scores in CTE and found a unique cluster of high stage (stage III) CTE that had a lower cortical p-tau burden [2]. Neuropathological subtypes that appear to skip the typical progression of disease are common in other neurodegenerative diseases. For instance, the limbic-sparing subtype of Alzheimer disease (AD) appears to skip the usual Braak neurofibrillary tangle progression, and the amygdala predominant subtype of Lewy body disease (LBD) does not follow the typical pattern of Lewy body progression [8, 16, 18]. Here, we examined brain donors with CTE that met criteria for high stage disease yet had only mild cortical p-tau pathology (cortical-sparing CTE, CSCTE). We examined the pathological distribution of p-tau pathology in CSCTE and tested whether CSCTE had different genetic, pathological, and clinical characteristics than typical high stage CTE.
Methods
Participants
Autopsy participants with a history of RHI exposure through contact sports participation and neuropathologically diagnosed with CTE (n = 620) were drawn from the Understanding Neurologic Injury and Traumatic Encephalopathy (UNITE) study. Inclusion criteria for brain donors in the UNITE study have been published previously [27, 29, 43]. A history of RHI (e.g., from contact sports, military service, physical violence) and tissue quality sufficient for neuropathological diagnosis using immunohistochemistry are the main inclusionary criteria. Criteria have also recently been expanded to include donors with a history of moderate to severe traumatic brain injury. Cases were procured either by having the donor’s next-of-kin contact the CTE center, referral by the medical examiner, or recruitment from the Concussion Legacy Foundation. The donor’s next-of-kin consented to the study enrollment, and approval for all study protocols was obtained from the BU Medical Campus and VA Bedford Healthcare System Institutional Review Boards.
Neuropathological assessment
Neuropathological processing included comprehensive screening for neurodegenerative conditions following procedures established for the UNITE brain bank [29, 43]. Briefly, all cases were evaluated based on paraffin-embedded tissue sections taken from standardized regions for histochemical and immunohistochemical staining [29, 43]. CTE was neuropathologically diagnosed using the NIH consensus criteria, which include abnormal perivascular accumulations of hyperphosphorylated tau (p-tau) in neurons, with or without astrocytes, and cell processes concentrated at the sulcal depths [9, 41]. CTE was then staged based on regional p-tau involvement according to the McKee staging system and then dichotomized in Low (stages I and II) and High (stage III and IV) [2, 9]. Secondary analyses were performed using the NINDS consensus staging criteria. Of note, 100% of High CTE and 88% of CSCTE (see definition below) cases remained “high stage” when applying the consensus criteria. Neuronal p-tau was required for diagnosis and to distinguish from aging-related tau astrogliopathy.
Established criteria were used to diagnose the presence of neurodegenerative pathology including fronto-temporal lobar degeneration with p-tau or transactive response DNA-binding protein 43 (TDP-43) inclusions (FTLD-tau; FTLD-TDP), neocortical Lewy body disease (LBD), cerebral amyloid angiopathy (CAA), hippocampal sclerosis (HS), and white matter rarefaction (WMR) [8, 14, 22, 28, 32, 37, 38, 40]. NIA-Reagan criteria were used to diagnose Alzheimer’s disease (AD) [31, 33, 42]. TDP-43 inclusions were assessed within the amygdala, entorhinal cortex, hippocampus, and dorsolateral frontal cortex and marked positive if present in any of those regions. Most TDP-43 inclusions were limbic predominant and indistinguishable from limbic-associated TDP-43 encephalopathy-neuropathologic change (LATE-NC); however, TDP-43 deposition has been shown to occur at a younger age and more commonly involve the frontal cortex and has been associated with RHI in CTE [34]. Compared to AD and primary age-related tauopathy (PART), CTE typically has greater involvement of CA4, as opposed to CA1 and CA2 in AD and PART [14]. Therefore, relatively greater involvement of CA4 was used as working criteria to distinguish hippocampal involvement by CTE versus PART. Although the original criteria for PART state that a diagnosis of PART should not be made in the presence of NFTs from another neurodegenerative disorder, we choose to make this distinction given that it is likely that both CTE and PART can coexist. Brain tissue was also evaluated for the presence of vascular pathology including gross infarcts, microinfarcts, moderate-severe atherosclerosis, and moderate-severe arteriolosclerosis. Pathologies were dichotomized as absent or present (CTE stage III or IV, intermediate or high AD, neocortical LBD, hippocampal sclerosis, FTLD, TDP-43 inclusions, and gross and microinfarcts). For pathologies rated on a none-mild-moderate-severe scale (CAA, white matter rarefaction, atherosclerosis, and arteriolosclerosis), scores were dichotomized as moderate/severe vs none/mild.
Semiquantitative assessment of pathology
Independent semi-quantitative assessments p-tau pathology were performed using semi-quantitative rating scales (0–3 scale; 0 = none, 1 = mild, 2 = moderate, 3 = severe) in 19 regions: dorsolateral frontal cortex, Rolandic cortex, inferior frontal cortex, inferior parietal cortex, superior temporal cortex, basal ganglia temporal pole, calcarine cortex, CA1-hippocampus, CA2-hippocampus, CA3-hippocampus, CA4-hippocampus, entorhinal cortex, amygdala, thalamus, substantia nigra, locus coeruleus, dorsal and median raphe nuclei at the level of the pons, and the dentate nucleus of the cerebellum. These assessments were performed on AT8-immunostaining of 10 μm thick paraffin-embedded sections, and regions were selected because of their involvement in CTE [9, 27, 29]. Previous studies have found that p-tau pathology in CTE is primarily neuronal, so only neuronal tau was included when assessing for p-tau burden in the various regions [11]. Aging-related tau astrogliopathy (ARTAG) was evaluated separately as a co-morbid pathology [20]; although as ARTAG is a recently described entity, data were only available for subset of cases (n = 33). Semi-quantitative assessments reflected overall p-tau burden within the given region and were performed by neuropathologists (ACM, TDS, BRH) for which interrater reliability was previously shown to be very good (0.83–0.96) [2].
Defining CSCTE
The mean cortical burden was defined as the mean semi-quantitative score of the superior frontal, dorsolateral frontal, Rolandic, inferior frontal, superior temporal, inferior parietal, and calcarine cortices. The overall mean and median cortical burden of high stage CTE were 1.77 and 2.00 respectively. However, a previous clustering analysis showed a high stage CTE cluster with a mean and median cortical burden ≅1 [2].Therefore, stages III–IV CTE was divided into typical high stage CTE (High CTE) with a mean cortical burden of >1 and cortical-sparing CTE (CSCTE) with a mean cortical burden of ≤1. This yielded 230 cases of low stage CTE (Low CTE), 324 cases of High CTE and 66 cases of CSCTE (Table 1).
Table 1.
Demographic, repetitive head impact, and genetic characteristics of low stage, high stage, and cortical-sparing CTE
| Low CTE | High CTE | CSCTE | p value | |
|---|---|---|---|---|
|
| ||||
| Total (%) | 230 (37%) | 324 (52%) | 66 (11%) | – |
| Age at death, years [IQR] | 46.0 [30.0–62.0] | 73.5 [64.0–80.0] | 70.0 [63.0–78.0] | <0.001 a,b |
| Range | 17–89 | 27–97 | 44–93 | |
| Unnatural cause of death | 107/118 (48%) | 29/289 (9.1%) | 6/58 (0.9%) | <0.001 a,b,c |
| Suicide | 66/159 (29%) | 18/300 (6.0%) | 5/59 (7.8%) | |
| Accidental drug overdose | 22/203 (10%) | 4/314 (1.3%) | 1/63 (1.6%) | |
| Injury | 19/206 (8.4%) | 7/311 (2.2%) | 0/64 (0%) | |
| Race | 0.242 | |||
| White | 181/45 (80%) | 261/55 (83%) | 46/20 (70%) | |
| Black/African American | 38/188 (17%) | 50/266 (16%) | 20/46 (30%) | |
| Asian | 0/226 (0%) | 1/315 (0.32%) | 0/66 (0%) | |
| Pacific Islander | 2/224 (0.88%) | 1/315 (0.32%) | 0/66 (0%) | |
| American Indian/Alaska Native | 1/223 (0.44%) | 1/315 (0.32%) | 0/66 (0%) | |
| Other | 4/220 (1.8%) | 2/314 (0.63%) | 0/66 (0%) | |
| Education Level | 0.058b | |||
| No High School | 0/226 (0%) | 2/314 (0.63%) | 0/66 (0%) | |
| High School/GED | 13/213 (5.8%) | 14/300 (4.3%) | 0/66 (0%) | |
| College | 171/55 (76%) | 212/104 (67%) | 47/19 (71%) | |
| Beyond College | 42/182 (19%) | 88/228 (28%) | 19/47 (29%) | |
| Primary Sport | 0.002 a | |||
| Football | 178/34 (84%) | 280/31 (90%) | 63/2 (97%) | |
| Hockey | 17/195 (8%) | 11/300 (3.5%) | 1/64 (1.5%) | |
| Boxing | 2/210 (0.9%) | 13/298 (4.2%) | 0/65 (0%) | |
| Soccer | 7/205 (3.3%) | 3/308 (0.96%) | 0/65 (0%) | |
| Wrestling | 8/204 (3.8%) | 4/307 (1.3%) | 1/64 (1.5%) | |
| Military History | 31/197 (14%) | 90/228 (28%) | 14/52 (21%) | <0.001 a |
| Total years playing contact sports, median [IQR] | 14.0 [10.0–18.0] | 16.0 [12.0–20.0] | 15.0 [12.0–18.0] | <0.001 a |
| Age at first contact sports exposure, years [IQR] | 9.0 [7.00–13.0] | 12.0 [10.0–14.0] | 13.0 [9.0–14.0] | <0.001 a,b |
| APOE4, one or more ε4 alleles | 42/97 (30%) | 97/141 (41%) | 10/32 (24%) | 0.029 a,c |
| TMEM106B, homozygous for C:C | 16/67 (18%) | 35/102 (26%) | 6/18 (25%) | 0.446 |
Categorical variables are displayed as yes/no (% positive). For continuous variables, medians with interquartile ranges (IQR) are listed. Kruskal Wallis statistical tests were performed on continuous variables and Chi-squared tests for categorical variables. Post-hoc pairwise comparisons were adjusted by the Bonferroni correction for multiple tests. p < 0.05 in bold
CSCTE, cortical-sparing CTE; GED, General Educational Development test for high school equivalency; APOE4, apolipoprotein E ε4; TMEM106B, transmembrane protein 106B
p < 0.05 between High CTE and Low CTE
p < 0.05 between CSCTE and Low CTE
p < 0.05 between CSCTE and High CTE
The ratios to determine proportional burden were calculated by dividing the semiquantitative score for each region of interest by the cortical mean for that case. The medial temporal lobe consisted of the averaged ratios of the entorhinal cortex, amygdala, temporal pole, and hippocampal fields. The basal ganglia consisted of the striatum and globus pallidus. The brainstem included the locus coeruleus, dorsal/median raphe nuclei, and substantia nigra. The cerebellum was evaluated within the dentate nucleus.
Retrospective clinical assessments
Online surveys, telephone interviews with next of kin, and review of clinical records were used to collect retrospective clinical data along with telephone interviews of the decedent’s next of kin. Both the interviewers and interviewees were blind to neuropathological findings. Semi-structured interviews were conducted by clinicians with expertise in this area. These interviews consisted of adapting standardized clinical scales for retrospective post-mortem administration. The Functional Activities Questionnaire (FAQ) was used to interrogate a decedent’s activities of daily living prior to death. It is a 10-item questionnaire with scores ranging from 0 to 30 and with higher scores representing worse functionality [36]. The Cognitive Difficulties Scale (CDS) is a 39-item instrument that assesses attention, memory, perception, and psychomotor abilities prior to death on a 0 (none) to 4 (very often) scale, and scores are summed to form a global composite (higher scores reflect greater reported cognitive symptoms). The Behavior Rating Inventory of Executive Function-Adult Version (BRIEF-A) Metacognition Index (MI) assessed informant-reported symptoms of executive dysfunction [15]. The BRIEF-A is a 75-item measurement of executive function behaviors. Informants rated frequency of problematic behaviors using a three-point scale (1 = never, 2 = sometimes, 3 = often). The BRIEF-A MI is a subscale that captures activity initiation, problem-solving, working memory, planning, and organization. MI raw scores are converted to age-adjusted T-scores; 65 or higher is clinically significant. A consensus of expert clinicians assigned dementia status via modified DSM-IV-TR criteria. The clinical history (i.e., symptoms, symptom course) and other pertinent history (e.g., medical, family, substance use history) was summarized by a clinician at the consensus conference. The UNITE study has evolved over time: FAQ was used prior to 2014 and after 2014 CDS was used. Additionally, certain other data points were collected, such as demographics, educational attainment, athletic history (type of sports played, level, position, age of first exposure and duration), military history, and traumatic brain injury history were queried during a telephone interview (pre-2014) and/or using an online questionnaire (2014 and on).
Neurobehavioral dysregulation was assessed by the BRIEF-A Behavioral Regulation Index (BRI), the Barratt Impulsiveness Scale-11 (BIS-11), and the Brown-Goodwin Aggression Scale [10, 19]. T-scores from the BRIEF-A BRI were examined and reflect an individual’s ability to control impulses and self-monitor behavior [13]. A T-score of 65 or higher is clinically meaningful. The BIS-11 is a 30-item questionnaire designed to assess impulsiveness in three domains: motor, non-planning, and attention and higher scores reflect more impulsive behaviors (range: 30–120) [35]. The Brown-Goodwin Aggression Scale is a 16-item assessment of lifetime history of aggressive and impulsive behavior (verbal and/or physical), and the adulthood subsection was used. Informants rated the observed frequency of specific aggressive behaviors on a five-point scale (range: 11–44), and a higher score indicates more aggressive behaviors. In addition, based on review of next-of-kin interviews and medical records, expert clinicians judged whether symptoms of parkinsonism, including bradykinesia, resting tremor, rigidity, micrographia, and shuffling gait, were present.
Genotyping
DNA was extracted from fresh frozen brain tissue using the Maxwell RSC Tissue DNA Kit (Promega, Madison, WI) according to the manufacturer’s protocol. Genetic variation in Apolipoprotein E (APOE; rs429358 and rs7412) and transmembrane protein 106B (TMEM106B; rs1990622) were determined using TaqMan single nucleotide polymorphism genotyping assays on the Step One Plus Real-Time PCR System (Applied Biosystems, Waltham, MA). APOE genotype was dichotomized into those with one or two copies of the e4 risk allele versus those with none. TMEM106B genotype was dichotomized as those homozygous for C:C vs. those with a G allele.
Statistical methods
IBM SPSS 27 was used for all statistical analyses. A Kruskall-Wallis H test was used for comparisons of continuous values and a Chi-square for nominal categorical values. Kruskall-Wallis H test with Bonferroni correction was utilized to test differences in ratios between the three pathological groups: low stage CTE (Low CTE), typical high stage CTE (High CTE), and CSCTE for all brain regions. Significance was defined as p < 0.05. The continuous scale variables were age at death, years playing contact sports, FAQ score, age of behavioral symptom onset and age of cognitive symptom onset. The output of these analyses was reported as a median and interquartile range due to failed tests of normality for all variables. The nominal values were dementia history, diagnosis of AD by NIA-Reagan criteria, CTE stage, Lewy body disease (neocortex and limbic or brainstem predominant), TDP43 (neocortex or amygdala and hippocampus), HS, PART, frontotemporal lobar dementia (FTLD), CAA, presence of one or more APOE4 alleles, and TMEM106B risk defined as C:C.
Results
Cortical-sparing CTE was present within 11% of all participants with CTE and 17% of those that met criteria for high stage (III–IV) CTE (Table 1). Participants with Low CTE had a significantly higher rate of unnatural death and died much younger than either CSCTE or High CTE. There were no significant differences in age or in the primary sport played between CSCTE and High CTE (Table 1). American football was by far the most frequent primary sport for all groups. Additionally, there was no difference in years played of contact sports, age of first contact sports exposure or age at death between High CTE and CSCTE. High CTE had an increased frequency of carrying the APOE4 allele when compared to both Low CTE and CSCTE (p = 0.006). No difference in the homozygous frequency of the risk allele for TMEM106B rs1990622 was observed between the three types of CTE.
Given that participants with Low CTE were markedly younger, pathological and clinical findings were compared between CSCTE and High CTE only (Tables 2, 3, and 4). CSCTE had a lower rate of AD by NIA-Reagan criteria compared to High CTE (1.5 vs. 31%, p < 0.001, Table 2). CSCTE also had lower Braak and CERAD scores (p’s < 0.001). CSCTE was significantly less likely to have other comorbid pathologies including all forms of Lewy Body Disease (LBD, p = 0.012), CAA of the leptomeninges (p = 0.014), TDP-43 inclusions (p’s < 0.01), FTLD-TDP (p = 0.026), and cerebral white matter rarefaction (p = 0.044). On the other hand, primary age-related tauopathy was seen at a significantly higher rate in CSCTE (38%) than High CTE (23%; p < 0.001). No difference was seen in frequencies of FTLD-tau, CAA intracortical or MND, and there were no significant differences in frequencies of moderate to severe atherosclerosis or arteriolosclerosis. Although the numbers with available data were small, no difference was found when comparing ARTAG between CSCTE and High CTE (p = 0.300). There was no difference in ARTAG involvement of the amygdala (p = 0.647), neocortex (p = 0.679), subcortical structures (p = 0.519), or brainstem (p = 0.667). Raw median and mean semiquantitative scores were also calculated between the three groups. The overall scores within all regions were lower in CSCTE compared to High CTE, but higher compared to Low CTE (Table 3).
Table 2.
Frequency of comorbid pathologies in cortical-sparing CTE versus typical high stage CTE
| High CTE | CSCTE | p value | |
|---|---|---|---|
|
| |||
| Diagnosis of AD (NIA-Reagan) | 98/223 (31%) | 1/65 (1.5%) | <0.001 |
| Braak stage | <0.001 | ||
| 0 | 16/307 (5.0%) | 1/65 (1.5%) | |
| I–II | 34/289 (11%) | 19/47 (29%) | |
| III–IV | 168/155 (52%) | 46/20 (70%) | |
| V–VI | 99/224 (31%) | 0/66 (0%) | |
| CERADa | |||
| Diffuse plaques | 169/155 (52%) | 17/49 (25%) | <0.001 |
| Neuritic plaques | 70/254 (22%) | 1/65 (1.5%) | <0.001 |
| Lewy body disease | 0.012 | ||
| Neocortex and limbic | 44/280 (13%) | 5/61 (10%) | |
| Brainstem | 37/287 (11%) | 1/65 (3.4%) | |
| TDP43, neocortex | 97/208 (32%) | 8/53 (13%) | 0.004 |
| TDP43, amygdala and hippocampus | 136/174 (44%) | 14/48 (23%) | 0.002 |
| FTLD | |||
| FTLD-tau (PiD) | 1/323 (0.3%) | 0/66 (0%) | 0.651 |
| FTLD-tau (CBD) | 2/321 (0.6%) | 0/66 (0%) | 0.522 |
| FTLD-tau (PSP) | 8/315 (2.5%) | 0/66 (0%) | 0.197 |
| FTLD-TDP43 | 23/300 (7.1%) | 0/66 (0%) | 0.026 |
| Hippocampal sclerosis | 106/214 (33%) | 14/51 (28%) | 0.087 |
| Primary age-related tauopathy (PART) | 74/249 (23%) | 35/31 (38%) | <0.001 |
| Motor neuron disease | 14/310 (4.3%) | 2/64 (3.0%) | 0.918 |
| CAA intracorticala | 38/278 (12%) | 3/62 (5.3%) | 0.079 |
| CAA leptomeningeala | 78/238 (25%) | 7/58 (11%) | 0.014 |
| Atherosclerosisa | 86/218 (28%) | 13/52 (20%) | 0.171 |
| Arteriolosclerosisa | 192/130 (60%) | 42/24 (64%) | 0.544 |
| White matter rarefactiona | 199/119 (63%) | 30/36 (45%) | 0.044 |
| McKee stage III | 162/162 (50%) | 64/2 (99%) | <0.001 |
Data are displayed as yes/no (% positive). The Kruskal Wallis statistical test was performed on continuous variables and Chi-squared test was used for categorical variables. Post-hoc pairwise comparisons were adjusted by the Bonferroni correction for multiple tests
CAA intracortical, CAA leptomeningeal, atherosclerosis, arteriolosclerosis, and white matter rarefaction. p < 0.05 in bold. CSCTE, cortical-sparing CTE; AD, Alzheimer disease; CAA, cerebral amyloid angiopathy
The numbers with moderate to severe disease are shown for CERAD score
Table 3.
Median and mean raw semiquantitative scores
| Low CTE | High CTE | CSCTE | p value | |
|---|---|---|---|---|
|
| ||||
| Medial Temporal Lobe, median (mean) | 0.5 (0.679) | 3.0 (2.21) | 2.0 (1.64) | 0.001 a,b,c |
| Basal Ganglia, median (mean) | 0.0 (0.162) | 1.0 (0.950) | 0.5 (0.770) | 0.001 a,b,c |
| Thalamus and mamillary bodies, median (mean) | 0.0 (0.337) | 1.5 (1.68) | 0.5 (0.354) | 0.001 a,b,c |
| Brainstem, median (mean) | 1.0 (0.815) | 2.0 (2.16) | 1.0 (1.50) | 0.001 a,b,c |
| Cerebellum, median (mean) | 0.0 (0.045) | 1.0 (0.907) | 0.0 (0.231) | 0.001 a,b,c |
Kruskal Wallis statistical tests were performed on all variables. Post-hoc pair wise comparisons were adjusted by the Bonferroni correction for multiple tests. p < 0.05 in bold
Low CTE, low stage CTE; High CTE, high stage typical CTE; CSCTE, cortical-sparing CTE
p < 0.05 between High CTE and Low CTE
p < 0.05 between CSCTE and Low CTE
p < 0.05 between CSCTE and High CTE
Table 4.
Clinical characteristics of cortical-sparing CTE versus typical high stage CTE
| High CTE | CSCTE | p value | |
|---|---|---|---|
|
| |||
| Dementia history | 230/66 (86%) | 39/22 (64%) | 0.023 |
| Functional activities questionnaire, median [IQR] | 25.00 [15.00–30.00] | 13.5 [5.00–24.00] | <0.001 |
| Meta-cognition index T score, median [IQR] | 90.00 [75.00–103.00] | 75.50 [62.75–90.25] | 0.002 |
| Cognitive difficulty scale score, median [IQR] | 126.50 [72.25–151.00] | 80.50 [54.00–115.75] | <0.001 |
| Age of cognitive symptom onset, years, [IQR] | 61.00 [51.00–70.00] | 62.50 [47.25–70.25] | 0.619 |
| Age of behavioral symptom onset, years, [IQR] | 55.00 [35.00–70.00] | 42.00 [30.00–59.00] | 0.006 |
| Age of Parkinsonian motor symptom onset, years, [IQR] | 71.00 [76.00–63.00] | 65.00 [57.50–73.75] | 0.013 |
| Parkinsonian motor symptoms | 125/144 (26%) | 21/40 (34%) | 0.087 |
| Brown-Goodwin adult sum, median [IQR] | 15.00 [12.00–19.00] | 16.00 [13.00–20.00] | 0.357 |
| Behavioral regulation index (BRI) T-score, median [IQR] | 83.00 [68.00–95.00] | 77.50 [62.75–91.25] | 0.082 |
| Barratt impulsiveness scale-11 Total, median [IQR] | 72.00 [82.00–60.00] | 70.00 [ 57.75–70.25] | 0.890 |
Categorical variables are displayed as yes/no (% positive). For continuous variables, medians with interquartile ranges [IQR] are listed. Kruskal Wallis statistical tests were performed on continuous variables and Chi-squared tests for categorical variables. Higher scores on the functional activities questionnaire, meta-cognition index, cognitive difficulty scale, BRI, and Barratt impulsiveness scale-11 reflect greater functional difficulties, cognitive symptoms, behavioral dysregulation, and impulsivity, respectively
The ratios of the different brain regions to the mean cortical burden varied significantly by subtype. CSCTE cases had a significantly higher proportional burden of disease than High CTE in the medial temporal lobe (Fig. 1a; p < 0.001) and brainstem (Fig. 1d; p < 0.001), but not in the basal ganglia (Fig. 1b), thalamus and mammillary bodies (Fig. 1c) or cerebellum (Fig. 1e). However, Low CTE had significantly lower proportional burden of p-tau pathology in the medial temporal lobe, basal ganglia, and thalamus and mammillary bodies compared to CSCTE and High CTE (p’s < 0.001).
Fig. 1.
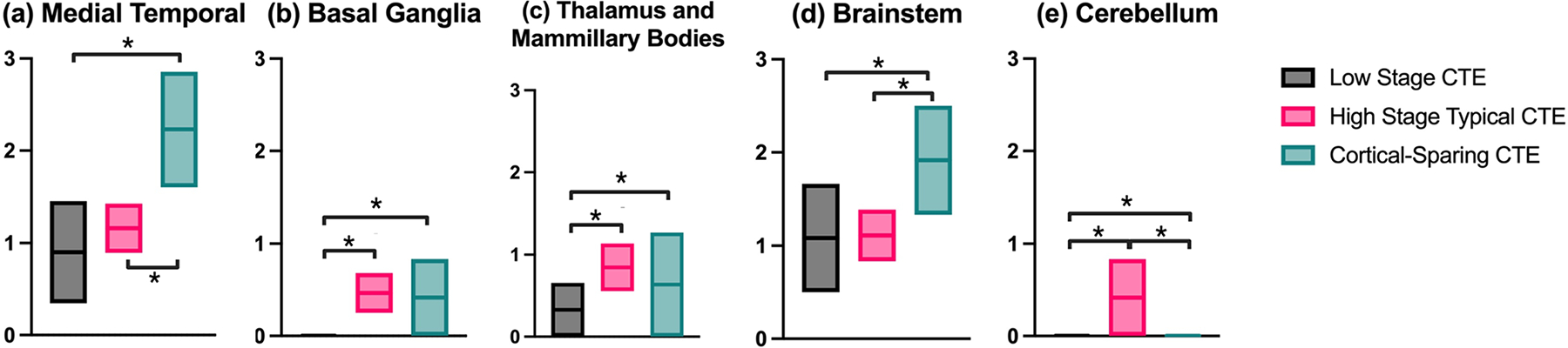
Comparison of proportional p-tau burden summary scores in different regions of the brain versus the neocortex in Low CTE, High CTE, and CSCTE. *p < 0.001
The two regions where CSCTE was found to have a higher proportional burden, the medial temporal lobe and brainstem, were further delineated by substructure (Figs. 2 and 3). CSCTE had significantly more proportional p-tau pathology burden in CA1, CA2 and CA4 (p’s < 0.001) when compared to Low CTE and High CTE. With respect to the brainstem, CSCTE had a significantly higher proportion of disease in the substantia nigra, locus coeruleus and dorsal and median raphe nuclei (p’s < 0.001) when compared to the other forms. Low CTE also had significantly lower proportional disease burden than High CTE (p’s < 0.001) in the substantia nigra and raphe nucleus.
Fig. 2.
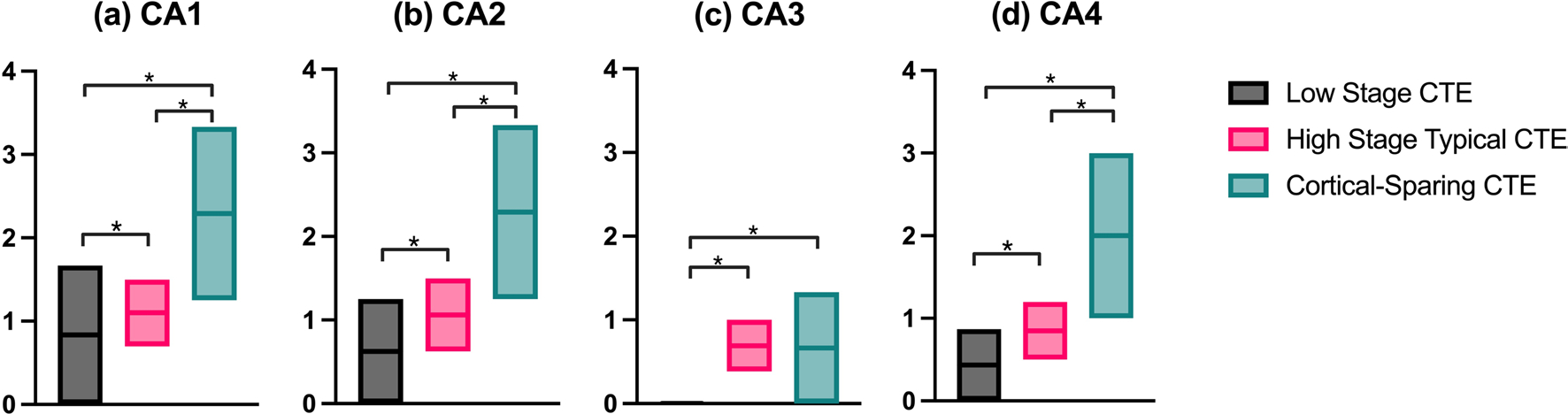
Comparison of proportional p-tau burden in each hippocampal subregion versus the neocortex in Low CTE, High CTE, and CSCTE. *p < 0.001
Fig. 3.
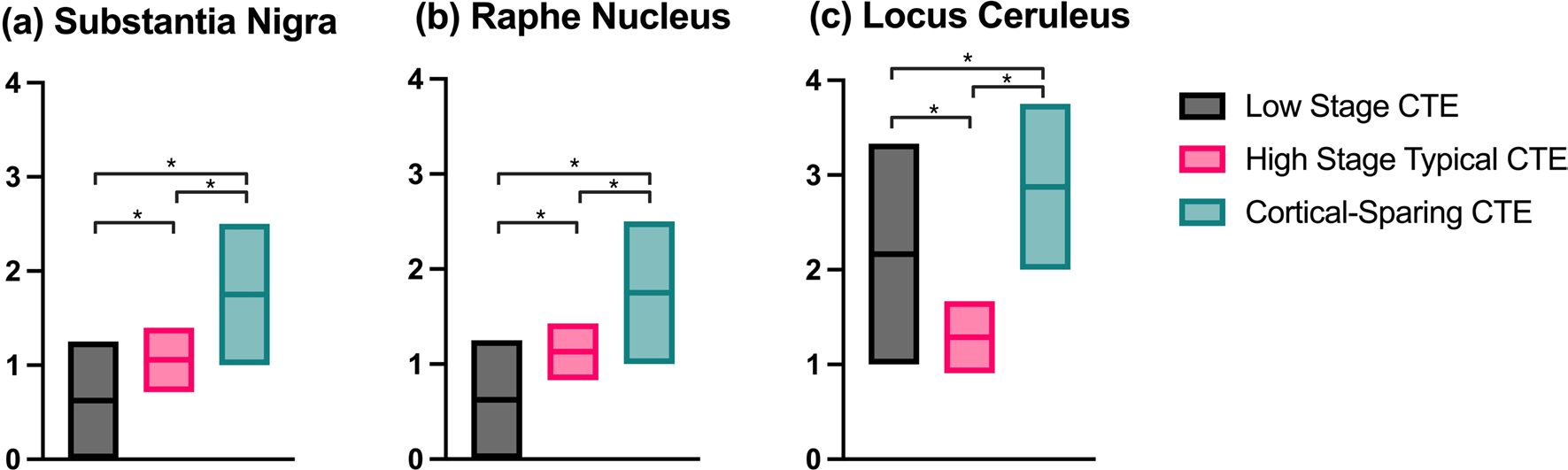
Comparison of proportional p-tau burden in different brainstem nuclei versus the neocortex in Low CTE, High CTE, and CSCTE. *p < 0.001
Histopathological differences between CSCTE and High stage CTE are shown in Fig. 4. For example, in High CTE abundant NFTs can be seen within the dorsolateral frontal cortex (DLF) (Fig. 4a) but are relatively sparse within CSCTE (Fig. 4b). In contrast, NFTs are present within the hippocampus and can be abundant in CSCTE, particularly in the CA4 subregion (Fig. 4c and d). In addition, there is often a moderate burden of tau pathology within the substantia nigra in CSCTE (Fig. 4f) that is comparable to that in typical high stage CTE (Fig. 4e).
Fig. 4.
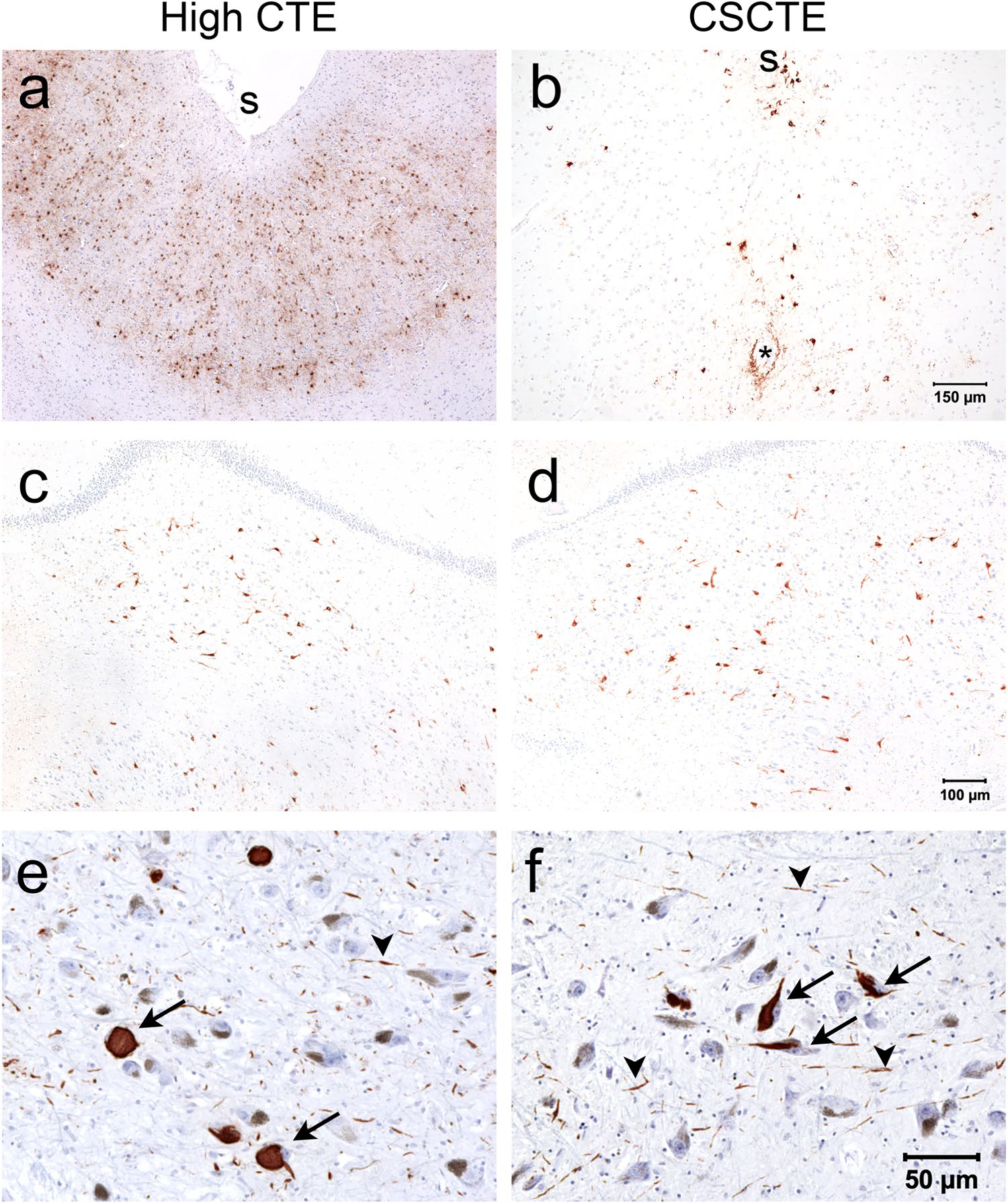
Histopathology of p-tau (AT8 antibody) in High CTE and CSCTE. a and b The dorsolateral frontal cortex has a low burden of p-tau in CSCTE (b) compared to High CTE (a); “asterisk” indicating vascular structure; “s” indicating depth of sulcus. c and d The hippocampus shows abundant neurofibrillary tangles within CA4 in both CSCTE (d) and High CTE (c). e and f The substantia nigra also shows involvement with p-tau neurofibrillary tangles and threads in both CSCTE (f) and High CTE (e); arrows indicate neurofibrillary tangles and arrowheads indicate p-tau threads (reddish-brown vs. pale brown of neuromelanin)
Clinically, CSCTE participants were less likely to be demented (p = 0.023) and had lower (i.e., better) FAQ, meta-cognition index, and CDS scores than High CTE (p’s ≤ 0.002, Table 4). Participants with CSCTE had similar scores for behavioral dysregulation (behavioral regulation index), impulsivity (Barratt impulsiveness scale-11), and Brown-Goodwin compared to High CTE. However, CSCTE presented earlier with behavioral (42 vs. 55 years old, p = 0.006) and Parkinsonian motor symptoms (65 vs. 71 years old, p = 0.013). There was a trend towards more frequent Parkinsonian motor symptoms in CSCTE compared to High CTE (p = 0.087). No significant difference was seen in the age of cognitive symptom onset.
To rule out contributions from co-morbid AD or FTLD, sensitivity analyses were conducted excluding those with intermediate to high AD neuropathological change by NIA-Reagan or FTLD-tau. Those with CSCTE still had significantly less severe FAQ scores, less severe impairment on the meta-cognition index, and younger age of Parkinsonism motor symptom onset than those with High CTE. The frequency of APOE4 remained lower in CSCTE compared to High CTE, but the p-value increased to p = 0.095. The difference in dementia frequency was no longer significant (Supplemental Table 1, online resource). The raw semi-quantitative values for p-tau burden in these cases remained significant, with High CTE having higher burden in all subregions (Supplemental Table 2, online resource). The differences in the proportional p-tau burden also remained significant (p’s < 0.001) for the medial temporal lobe, brainstem, and cerebellum as shown in Fig. 1 (Supplemental Table 3, online resource). In addition, the majority of CSCTE participants were McKee stage III (99%) versus only 50% stage III in typical high stage CTE (Table 2, p < 0.001). When secondary analyses were performed on only stage III brains, all comparisons remained significant.
Discussion
Here we define a subset of high stage CTE with a low overall cortical burden of p-tau pathology (mean semiquantitative score ≤1) and classify these as cortical sparing CTE (CSCTE). Approximately 11% overall and 17% of participants with high stage CTE met criteria for CSCTE. Compared to typical high stage disease, CSCTE participants had similar age at death, years of play, primary sport, and age when first exposed to contact sports but were less likely to carry APOE4. Compared to both low and high stage CTE, CSCTE had a proportional increase of disease burden in medial temporal lobe and brainstem regions compared to the neocortex. Clinically, CSCTE participants had fewer cognitive symptoms but had similar levels of behavioral dysregulation and an earlier onset age of behavioral and Parkinsonism symptoms when compared to typical high stage CTE.
Although CSCTE meets criteria for high stage CTE with either the McKee or NINDS staging systems, there are numerous pathological differences between CSCTE and High CTE. Relative to the p-tau pathology burden in the cortex, CSCTE had greater relative involvement of both medial temporal and brainstem regions. In addition, CSCTE was less likely to have many types of comorbid pathology, including AD, TDP-43, hippocampal sclerosis, LBD, and leptomeningeal CAA. The presence of AD-related pathology exacerbates the cortical p-tau pathology in CTE [40], and thus its absence may lead to decreased cortical p-tau burden in CSCTE compared to high stage CTE. The presence of comorbid AD or FTLD-tau may partially underlie the greater cognitive impairment in High CTE; however, when these were excluded in a sensitivity analysis, CSCTE still showed significantly less impairment in cognitive scores than High CTE. CSCTE was also more likely to be stage III, which may be due to down grading as a result of the low cortical burden. CSCTE had a higher rate of primary age-related tauopathy (PART) despite a similar mean age to High CTE. Since similar regions can be affected, distinguishing PART from CTE can be difficult. Recent studies suggest that CA4 is a region susceptible to CTE, but less involved in PART [14, 17, 44]. The proportional increase of neurofibrillary tangle burden in CA4 in CSCTE versus both Low and High CTE therefore suggest that CSCTE is not simply Low CTE with coexistent PART. Future studies should examine the role of PART in CTE and possible associations with RHI. Furthermore, because of the greater p-tau pathology burden in the medial temporal lobe compared to the cortex, CSCTE may appear similar to PART or early AD on p-tau PET imaging. Therefore, the presence of CSCTE may have implications for clinical studies focused on biomarkers, including tau imaging.
Recent studies suggest that RHI and CTE are associated with numerous neurodegenerative pathologies [1, 6, 28, 34, 38, 40]. In fact, mixed pathologies are common in a brain bank of former contact sport athletes and pathologies such as AD, LBD, hippocampal sclerosis, CAA, and CTE all contribute to cognitive impairment.
In addition to the medial temporal lobe, CSCTE also had a higher proportional burden of disease in the brain-stem compared to High CTE. This included greater relative involvement of the substantia nigra, dorsal/median raphe nuclei, and the locus coeruleus. The upper brainstem may be particularly vulnerable to RHI and TBI due to rotational forces on the cerebrum that pull on this region. CSCTE may therefore be related to a particular type of injury that targets the brainstem over damage to the cortex. Examination of sport positions, sport types, and the forces involved may help elucidate risk factors for developing CSCTE versus High CTE.
Comparing the high stage forms of disease, CSCTE was far more likely to be McKee stage III with nearly all these donors (99%) falling into this category. Stage IV CTE typically involves the dentate nucleus of the cerebellum, as seen in Fig. 1e [25]. In addition, Stage IV CTE is expected have widespread cortical tau pathology and thus the low cortical burden of tau pathology in CSCTE likely precludes upstaging most of these cases. Nevertheless, when CSCTE was compared only to Stage III High CTE, the reported differences remained significant.
Genetic factors, including the presence of APOE4 and variation in TMEM106B, have been shown to contribute to CTE disease severity [7, 12]. There was no difference in the frequency of the recessive risk allele for TMEM106B in CSCTE. However, CSCTE was significantly less likely to have APOE4 than High CTE. APOE4 might therefore be a risk factor for the more severe High CTE perhaps partially through the development of AD-type pathologies such as neuritic beta-amyloid plaques and CAA [21].
Clinically, CSCTE differed significantly from High CTE. Overall, the rate of dementia was reduced, and scores of cognitive symptoms showed less impairment in CSCTE. Semiquantitative levels of cortical p-tau pathology have been shown to be the greatest predictors of cognitive impairment in CTE [5, 30]. Therefore, this milder phenotype matches well with the milder cortical tau pathology burden in CSCTE. On the other hand, measures of behavioral dysregulation and Parkinsonism symptomatology were similar between CSCTE and High CTE but started significantly earlier in donors with CSCTE. Factors other than p-tau pathology appear to correlate better with mood and behavioral dysfunction, including loss of the myelin proteins myelin-associated glycoprotein (MAG) and proteolipid protein 1 (PLP) [3]. Future studies should examine in CSCTE the role of white matter and axonal damage, inflammation, and other non-tau pathologies as well as the involvement of regions of the brain involved in emotional and behavioral regulation such as the entorhinal cortex and amygdala.
There is also a possibility that CSCTE is a milder form of severe disease that falls in between Low and High CTE given that the raw median and mean scores of the medial temporal lobe, subcortical, and brainstem regions were higher than those of Low CTE, but lower than High CTE. These may be individuals who were in the process of progressing from Low to High stage CTE. However, given the myriad similarities between CSCTE and High CTE (age at death, years of play, age of first contact sports exposure, and age of cognitive symptom onset) but distinct clinical differences (age of behavioral and Parkinsonian motor symptom onset, better cognition scores, less comorbidities), it leads us to favor that this is a distinct, albeit less severe, form of high stage CTE.
There are several limitations to this study. Participants were limited to those with brain donation which may bias towards those with symptoms. The majority of both High CTE and CSCTE played American football and the study was therefore not powered to detect a difference between sport type in either subtype. All participants were male, and additional studies will need to examine the role of sex as more females with a history of RHI and CTE become available. The lower brainstem and spinal cord were not included in this study although both regions may be affected in CTE. Future studies should further examine regional differences in CSCTE. Clinical scales were completed by informants of brain donors and therefore estimates might be inaccurate due to biases from recall and subjectivity. Prospective studies with clinical and cognitive characterization during life among people who agree to brain donation are needed for validation.
Conclusion
CSCTE is a relatively frequent and distinct pathological form of CTE with involvement of the medial temporal lobe and brainstem, but low burden of pathology in the neocortex and with different risk, genetic, and clinical associations than High CTE. CSCTE may have implications for clinical studies focused on biomarkers, including tau imaging.
Supplementary Material
Funding
This work was supported by the United States Department of Veterans Affairs, Veterans Health Administration, Veterans Affairs Biorepository (BX002466); BLRD Merit Award (I01BX005933 and I01BX005161); Alzheimer’s Association (NIRG-305779, NIRG-362697); National Institute of Aging (P30AG072978, R01AG075876); National Institute of Neurological Disorders and Stroke (U54NS115266, U01NS086659, K23NS102399, RF1NS122854); and the Concussion Legacy Foundation. This work was also supported by unrestricted gifts from the Andlinger Foundation and WWE. This material is the result of work supported with resources and the use of facilities at the Bedford VA Healthcare System. We gratefully acknowledge the help of all members of the Boston University CTE Center and the Concussion Legacy Foundation (with special thanks to Lisa McHale), as well as the individuals and families whose participation and contributions made this work possible.
Footnotes
Supplementary Information The online version contains supplementary material available at https://doi.org/10.1007/s00401-024-02690-5.
Data availability
Data available on request to the Boston University Alzheimer’s Disease Center.
References
- 1.Adams JW, Alvarez VE, Mez J, Huber BR, Tripodis Y, Xia W et al. (2018) Lewy body pathology and chronic traumatic encephalopathy associated with contact sports. J Neuropathol Exp Neurol 77:757–768. 10.1093/jnen/nly065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alosco ML, Cherry JD, Huber BR, Tripodis Y, Baucom Z et al. (2020) Characterizing tau deposition in chronic traumatic encephalopathy (CTE): utility of the McKee CTE staging scheme. Acta Neuropathol (Berl) 140:495–512. 10.1007/s00401-020-02197-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alosco ML, Ly M, Mosaheb S, Saltiel N, Uretsky M, Tripodis Y et al. (2023) Decreased myelin proteins in brain donors exposed to football-related repetitive head impacts. Brain Commun 5:fcad019. 10.1093/braincomms/fcad019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alosco ML, Mez J, Tripodis Y, Kiernan PT, Abdolmohammadi B, Murphy L et al. (2018) Age of first exposure to tackle football and chronic traumatic encephalopathy. Ann Neurol 83:886–901. 10.1002/ana.25245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Alosco ML, Mian AZ, Buch K, Farris CW, Uretsky M, Tripodis Y et al. (2021) Structural MRI profiles and tau correlates of atrophy in autopsy-confirmed CTE. Alzheimers Res Ther 13:193. 10.1186/s13195-021-00928-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alosco ML, Stein TD, Tripodis Y, Chua AS, Kowall NW, Huber BR et al. (2019) Association of White Matter Rarefaction, Arteriolosclerosis, and Tau With Dementia in Chronic Traumatic Encephalopathy. JAMA Neurol 76:1298–1308. 10.1001/jamaneurol.2019.2244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Atherton K, Han X, Chung J, Cherry JD, Baucom Z, Saltiel N et al. (2022) Association of APOE genotypes and chronic traumatic encephalopathy. JAMA Neurol 79:787. 10.1001/jamaneurol.2022.1634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Attems J, Toledo JB, Walker L, Gelpi E, Gentleman S, Halliday G et al. (2021) Neuropathological consensus criteria for the evaluation of Lewy pathology in post-mortem brains: a multi-centre study. Acta Neuropathol (Berl) 141:159–172. 10.1007/s00401-020-02255-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bieniek KF, Cairns NJ, Crary JF, Dickson DW, Folkerth RD, Keene CD et al. (2021) The Second NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. J Neuropathol Exp Neurol. Publish Ahead of Print. 10.1093/jnen/nlab001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brown GL, Goodwin FK, Ballenger JC, Goyer PF, Major LF (1979) Aggression in humans correlates with cerebrospinal fluid amine metabolites. Psychiatry Res 1:131–139. 10.1016/0165-1781(79)90053-2 [DOI] [PubMed] [Google Scholar]
- 11.Butler MLMD, Dixon E, Stein TD, Alvarez VE, Huber B, Buckland ME et al. (2022) Tau pathology in chronic traumatic encephalopathy is primarily neuronal. J Neuropathol Exp Neurol 81:773–780. 10.1093/jnen/nlac065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cherry JD, Mez J, Crary JF, Tripodis Y, Alvarez VE, Mahar I et al. (2018) Variation in TMEM106B in chronic traumatic encephalopathy. Acta Neuropathol Commun 6:115. 10.1186/s40478-018-0619-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Crook T (1983) Assessment in geriatric psychopharmacology. Mark Powley Associates, New Canaan, CT [Google Scholar]
- 14.Farrell K, Iida MA, Cherry JD, Casella A, Stein TD, Bieniek KF et al. (2022) Differential vulnerability of hippocampal subfields in primary age-related tauopathy and chronic traumatic encephalopathy. J Neuropathol Exp Neurol 81:781–789. 10.1093/jnen/nlac066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gioia GA, Isquith PK, Guy SC, Kenworthy L (2000) TEST REVIEW behavior rating inventory of executive function. Child Neuropsychol 6:235–238. 10.1076/chin.6.3.235.3152 [DOI] [PubMed] [Google Scholar]
- 16.Hyman BT, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Carrillo MC et al. (2012) National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimers Dement 8:1–13. 10.1016/j.jalz.2011.10.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Iida MA, Farrell K, Walker JM, Richardson TE, Marx GA, Bryce CH et al. (2021) Predictors of cognitive impairment in primary age-related tauopathy: an autopsy study. Acta Neuropathol Commun 9:134. 10.1186/s40478-021-01233-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Janocko NJ, Brodersen KA, Soto-Ortolaza AI, Ross OA, Liesinger AM, Duara R et al. (2012) Neuropathologically defined subtypes of Alzheimer’s disease differ significantly from neurofibrillary tangle-predominant dementia. Acta Neuropathol (Berl) 124:681–692. 10.1007/s00401-012-1044-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Katz DI, Bernick C, Dodick DW, Mez J, Mariani ML, Adler CH et al. (2021) National institute of neurological disorders and stroke consensus diagnostic criteria for traumatic encephalopathy syndrome. Neurology 96:848–863. 10.1212/WNL.0000000000011850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kovacs GG, Ferrer I, Grinberg LT, Alafuzoff I, Attems J, Budka H et al. (2016) Aging-related tau astrogliopathy (ARTAG): harmonized evaluation strategy. Acta Neuropathol (Berl) 131:87–102. 10.1007/s00401-015-1509-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu C-C, Kanekiyo T, Xu H, Bu G (2013) Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol 9:106–118. 10.1038/nrneurol.2012.263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mackenzie IRA, Neumann M, Bigio EH, Cairns NJ, Alafuzoff I, Kril J et al. (2010) Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathol (Berl) 119:1–4. 10.1007/s00401-009-0612-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, Budson AE et al. (2009) Chronic traumatic encephalopathy in athletes: progressive tauopathy after repetitive head injury. J Neuropathol Exp Neurol 68:709–735. 10.1097/NEN.0b013e3181a9d503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.McKee AC, Stein TD, Kiernan PT, Alvarez VE (2015) The neuropathology of chronic traumatic encephalopathy. Brain Pathol Zurich Switz 25:350–364. 10.1111/bpa.12248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McKee AC, Stein TD, Nowinski CJ, Stern RA, Daneshvar DH, Alvarez VE et al. (2013) The spectrum of disease in chronic traumatic encephalopathy. Brain 136:43–64. 10.1093/brain/aws307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mendez MF (1995) The neuropsychiatric aspects of boxing. Int J Psychiatry Med 25:249–262. 10.2190/CUMK-THT1-X98M-WB4C [DOI] [PubMed] [Google Scholar]
- 27.Mez J, Daneshvar DH, Abdolmohammadi B, Chua AS, Alosco ML, Kiernan PT et al. (2020) Duration of American football play and chronic traumatic encephalopathy. Ann Neurol 87:116–131. 10.1002/ana.25611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mez J, Daneshvar DH, Kiernan PT, Abdolmohammadi B, Alvarez VE, Huber BR et al. (2017) Clinicopathological evaluation of chronic traumatic encephalopathy in players of American football. JAMA 318:360. 10.1001/jama.2017.8334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mez J, Solomon TM, Daneshvar DH, Murphy L, Kiernan PT, Montenigro PH et al. (2015) Assessing clinicopathological correlation in chronic traumatic encephalopathy: rationale and methods for the UNITE study. Alzheimers Res Ther 7:62. 10.1186/s13195-015-0148-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alosco ML, White M, Bell C, Faheem F, Tripodis Y, Yhang E et al. (2024) Cognitive, functional, and neuropsychiatric correlates of regional tau pathology in autopsy-confirmed chronic traumatic encephalopathy. Mol Neurodegener 19(1):10. 10.1186/s13024-023-00697-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW et al. (2012) National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol (Berl) 123:1–11. 10.1007/s00401-011-0910-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nelson PT, Dickson DW, Trojanowski JQ, Jack CR, Boyle PA, Arfanakis K et al. (2019) Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain J Neurol 142:1503–1527. 10.1093/brain/awz099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Newell KL, Hyman BT, Growdon JH, Hedley-Whyte ET (1999) Application of the National Institute on Aging (NIA)-Reagan Institute criteria for the neuropathological diagnosis of Alzheimer disease. J Neuropathol Exp Neurol 58:1147–1155. 10.1097/00005072-199911000-00004 [DOI] [PubMed] [Google Scholar]
- 34.Nicks R, Clement NF, Alvarez VE, Tripodis Y, Baucom ZH, Huber BR et al. (2023) Repetitive head impacts and chronic traumatic encephalopathy are associated with TDP-43 inclusions and hippocampal sclerosis. Acta Neuropathol (Berl) 145:395–408. 10.1007/s00401-023-02539-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Patton JH, Stanford MS, Barratt ES (1995) Factor structure of the barratt impulsiveness scale. J Clin Psychol 51:768–774. [DOI] [PubMed] [Google Scholar]
- 36.Pfeffer RI, Kurosaki TT, Harrah CH, Chance JM, Filos S (1982) Measurement of functional activities in older adults in the community. J Gerontol 37:323–329. 10.1093/geronj/37.3.323 [DOI] [PubMed] [Google Scholar]
- 37.Roemer SF, Grinberg LT, Crary JF, Seeley WW, McKee AC, Kovacs GG et al. (2022) Rainwater Charitable Foundation criteria for the neuropathologic diagnosis of progressive supranuclear palsy. Acta Neuropathol (Berl) 144:603–614. 10.1007/s00401-022-02479-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Standring OJ, Friedberg J, Tripodis Y, Chua AS, Cherry JD, Alvarez VE et al. (2019) Contact sport participation and chronic traumatic encephalopathy are associated with altered severity and distribution of cerebral amyloid angiopathy. Acta Neuropathol (Berl) 138:401–413. 10.1007/s00401-019-02031-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Stathas S, Alvarez VE, Xia W, Nicks R, Meng G, Daley S et al. (2022) Tau phosphorylation sites serine202 and serine396 are differently altered in chronic traumatic encephalopathy and Alzheimer’s disease. Alzheimers Dement J Alzheimers Assoc 18:1511–1522. 10.1002/alz.12502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Stein TD, Montenigro PH, Alvarez VE, Xia W, Crary JF, Tripodis Y et al. (2015) Beta-amyloid deposition in chronic traumatic encephalopathy. Acta Neuropathol (Berl) 130:21–34. 10.1007/s00401-015-1435-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.the TBI/CTE group, McKee AC, Cairns NJ, Dickson DW, Folkerth RD, Dirk Keene C et al. (2016) The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol (Berl) 131:75–86. 10.1007/s00401-015-1515-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Toledo JB, Gopal P, Raible K, Irwin DJ, Brettschneider J, Sedor S et al. (2016) Pathological α-synuclein distribution in subjects with coincident Alzheimer’s and Lewy body pathology. Acta Neuropathol (Berl) 131:393–409. 10.1007/s00401-015-1526-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vonsattel JPG, Del Amaya MP, Keller CE (2008) Twenty-first century brain banking. Processing brains for research: the Columbia University methods. Acta Neuropathol (Berl) 115:509–532. 10.1007/s00401-007-0311-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Walker JM, Fudym Y, Farrell K, Iida MA, Bieniek KF, Seshadri S et al. (2021) Asymmetry of hippocampal tau pathology in primary age-related tauopathy and Alzheimer disease. J Neuropathol Exp Neurol 80:436–445. 10.1093/jnen/nlab032 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data available on request to the Boston University Alzheimer’s Disease Center.


