Abstract
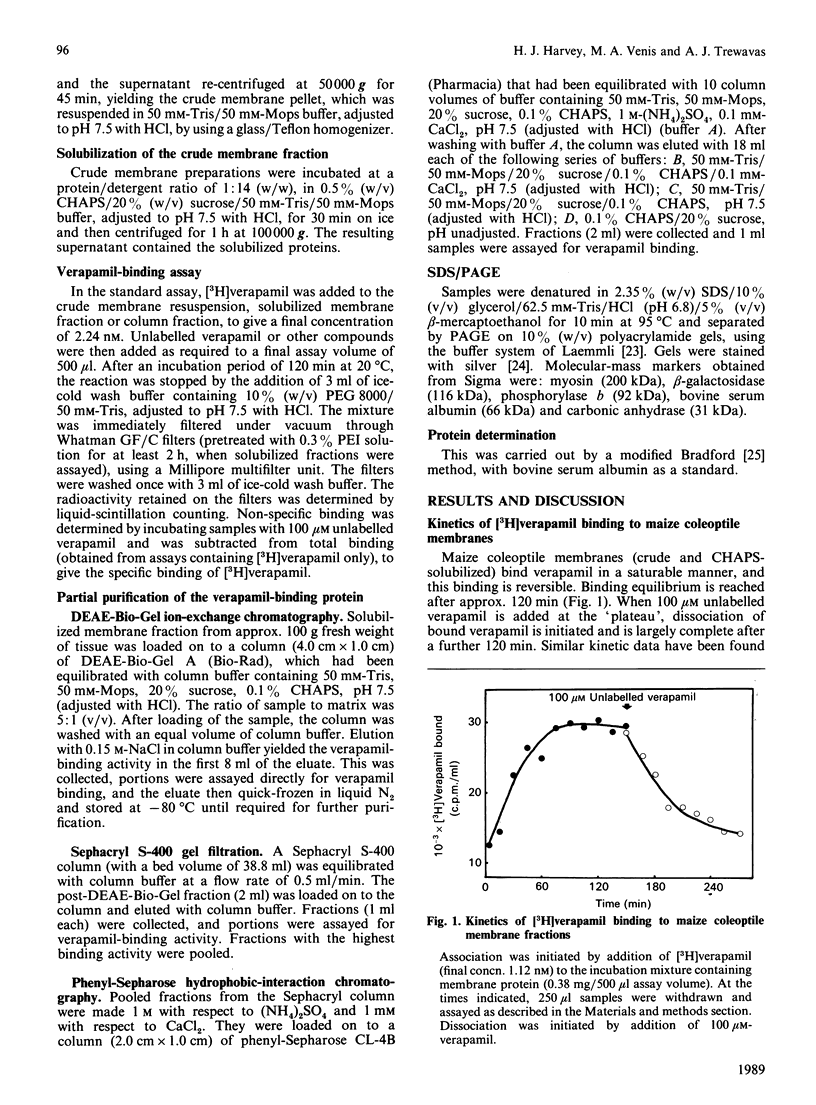
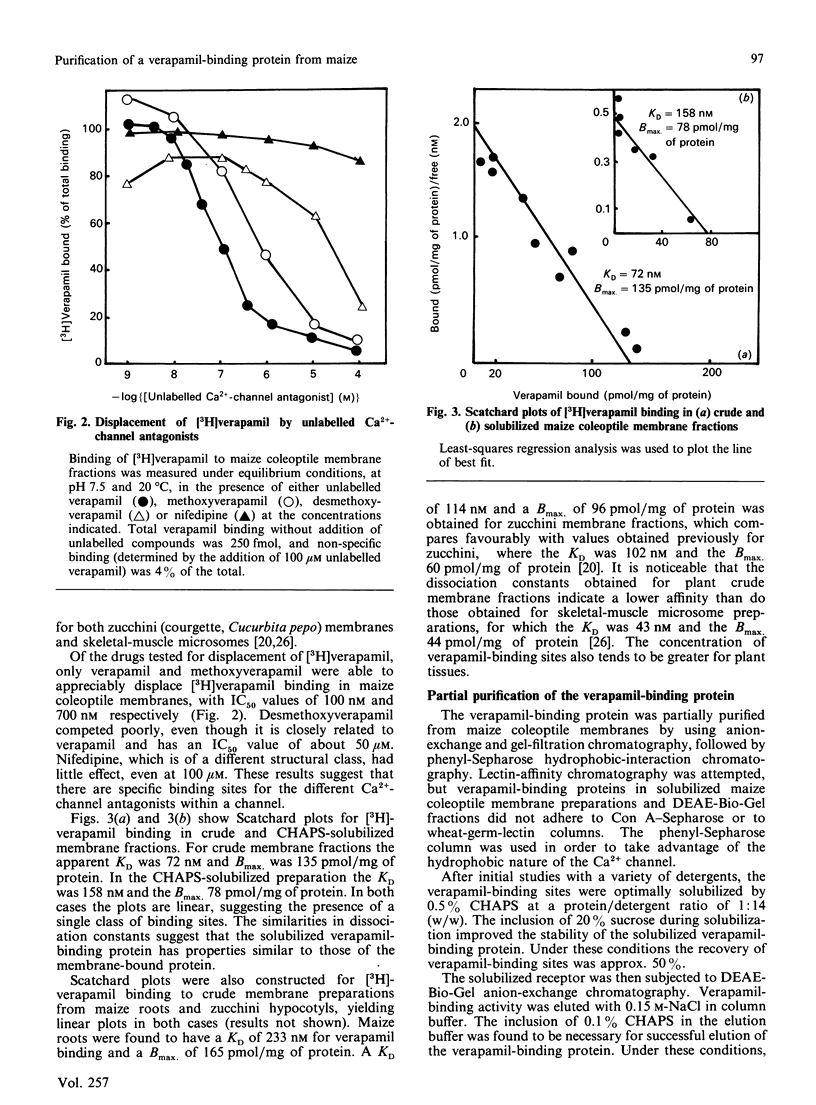
A protein that binds the calcium-channel antagonist verapamil has been partially purified from maize (Zea mays) coleoptile membranes. The protein was solubilized with the detergent CHAPS ([ 3-(3-cholamidopropyl)dimethylammonio]propane-1-sulphonate) and purified by a combination of ion-exchange, gel-filtration and hydrophobic-interaction chromatography. This resulted in a 120-fold purification. SDS/polyacrylamide-gel-electrophoretic analysis of the polypeptides from the final purification step indicated that the verapamil-binding protein may have a major component of Mr 169,000. The dissociation constants for specific binding of [3H]verapamil to crude and CHAPS-solubilized maize coleoptile membrane fractions are 72 nM and 158 nM respectively, with respective binding-site concentrations of 135 pmol/mg of protein and 78 pmol/mg of protein. In both cases the Scatchard plots are linear, indicating a single class of binding sites. [3H]Verapamil binding to crude maize coleoptile membrane fractions could not be displaced by unlabelled desmethoxyverapamil or by nifedipine, but could be displaced by unlabelled methoxyverapamil.
Full text
PDF
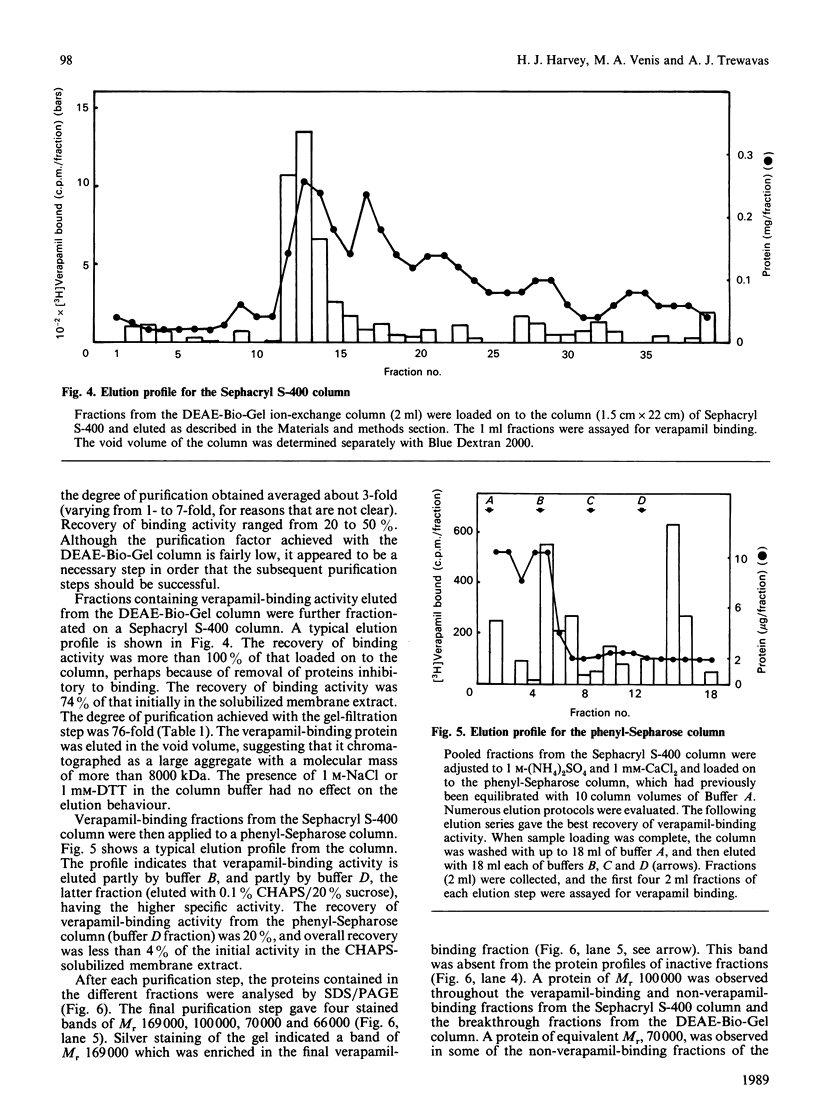


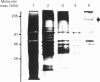
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrejauskas E., Hertel R., Marmé D. 3,4,5-Triiodobenzoic acid affects [3H]verapamil binding to plant and animal membrane fractions and smooth muscle contraction. Biochem Biophys Res Commun. 1986 Aug 14;138(3):1269–1275. doi: 10.1016/s0006-291x(86)80420-x. [DOI] [PubMed] [Google Scholar]
- Andrejauskas E., Hertel R., Marmé D. Specific binding of the calcium antagonist [3H]verapamil to membrane fractions from plants. J Biol Chem. 1985 May 10;260(9):5411–5414. [PubMed] [Google Scholar]
- Baker P. F., Knight D. E. Calcium control of exocytosis and endocytosis in bovine adrenal medullary cells. Philos Trans R Soc Lond B Biol Sci. 1981 Dec 18;296(1080):83–103. doi: 10.1098/rstb.1981.0174. [DOI] [PubMed] [Google Scholar]
- Bearden J. C., Jr Quantitation of submicrogram quantities of protein by an improved protein-dye binding assay. Biochim Biophys Acta. 1978 Apr 26;533(2):525–529. doi: 10.1016/0005-2795(78)90398-7. [DOI] [PubMed] [Google Scholar]
- Borsotto M., Barhanin J., Fosset M., Lazdunski M. The 1,4-dihydropyridine receptor associated with the skeletal muscle voltage-dependent Ca2+ channel. Purification and subunit composition. J Biol Chem. 1985 Nov 15;260(26):14255–14263. [PubMed] [Google Scholar]
- Ferry D. R., Glossmann H. Evidence of multiple receptor sites within the putative calcium channel. Naunyn Schmiedebergs Arch Pharmacol. 1982 Oct;321(1):80–83. doi: 10.1007/BF00586355. [DOI] [PubMed] [Google Scholar]
- Galizzi J. P., Borsotto M., Barhanin J., Fosset M., Lazdunski M. Characterization and photoaffinity labeling of receptor sites for the Ca2+ channel inhibitors d-cis-diltiazem, (+/-)-bepridil, desmethoxyverapamil, and (+)-PN 200-110 in skeletal muscle transverse tubule membranes. J Biol Chem. 1986 Jan 25;261(3):1393–1397. [PubMed] [Google Scholar]
- Galizzi J. P., Fosset M., Lazdunski M. Properties of receptors for the Ca2+-channel blocker verapamil in transverse-tubule membranes of skeletal muscle. Stereospecificity, effect of Ca2+ and other inorganic cations, evidence for two categories of sites and effect of nucleoside triphosphates. Eur J Biochem. 1984 Oct 15;144(2):211–215. doi: 10.1111/j.1432-1033.1984.tb08451.x. [DOI] [PubMed] [Google Scholar]
- Goll A., Ferry D. R., Glossmann H. Target size analysis and molecular properties of Ca2+ channels labelled with [3H]verapamil. Eur J Biochem. 1984 May 15;141(1):177–186. doi: 10.1111/j.1432-1033.1984.tb08172.x. [DOI] [PubMed] [Google Scholar]
- Hosey M. M., Barhanin J., Schmid A., Vandaele S., Ptasienski J., O'Callahan C., Cooper C., Lazdunski M. Photoaffinity labelling and phosphorylation of a 165 kilodalton peptide associated with dihydropyridine and phenylalkylamine-sensitive calcium channels. Biochem Biophys Res Commun. 1987 Sep 30;147(3):1137–1145. doi: 10.1016/s0006-291x(87)80188-2. [DOI] [PubMed] [Google Scholar]
- Keith C. H., Ratan R., Maxfield F. R., Bajer A., Shelanski M. L. Local cytoplasmic calcium gradients in living mitotic cells. 1985 Aug 29-Sep 4Nature. 316(6031):848–850. doi: 10.1038/316848a0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- Ochs D. Protein contaminants of sodium dodecyl sulfate-polyacrylamide gels. Anal Biochem. 1983 Dec;135(2):470–474. doi: 10.1016/0003-2697(83)90714-5. [DOI] [PubMed] [Google Scholar]
- Reuter H. Calcium channel modulation by neurotransmitters, enzymes and drugs. Nature. 1983 Feb 17;301(5901):569–574. doi: 10.1038/301569a0. [DOI] [PubMed] [Google Scholar]
- Saunders M. J., Hepler P. K. Calcium antagonists and calmodulin inhibitors block cytokinin-induced bud formation in Funaria. Dev Biol. 1983 Sep;99(1):41–49. doi: 10.1016/0012-1606(83)90252-x. [DOI] [PubMed] [Google Scholar]
- Saunders M. J., Hepler P. K. Calcium ionophore a23187 stimulates cytokinin-like mitosis in funaria. Science. 1982 Sep 3;217(4563):943–945. doi: 10.1126/science.217.4563.943. [DOI] [PubMed] [Google Scholar]
- Schmid A., Barhanin J., Coppola T., Borsotto M., Lazdunski M. Immunochemical analysis of subunit structures of 1,4-dihydropyridine receptors associated with voltage-dependent Ca2+ channels in skeletal, cardiac, and smooth muscles. Biochemistry. 1986 Jun 17;25(12):3492–3495. doi: 10.1021/bi00360a002. [DOI] [PubMed] [Google Scholar]
- Triggle D. J., Janis R. A. Calcium channel ligands. Annu Rev Pharmacol Toxicol. 1987;27:347–369. doi: 10.1146/annurev.pa.27.040187.002023. [DOI] [PubMed] [Google Scholar]
- Tsien R. W. Calcium channels in excitable cell membranes. Annu Rev Physiol. 1983;45:341–358. doi: 10.1146/annurev.ph.45.030183.002013. [DOI] [PubMed] [Google Scholar]
- Vandaele S., Fosset M., Galizzi J. P., Lazdunski M. Monoclonal antibodies that coimmunoprecipitate the 1,4-dihydropyridine and phenylalkylamine receptors and reveal the Ca2+ channel structure. Biochemistry. 1987 Jan 13;26(1):5–9. doi: 10.1021/bi00375a002. [DOI] [PubMed] [Google Scholar]
- Williamson R. E., Ashley C. C. Free Ca2+ and cytoplasmic streaming in the alga Chara. Nature. 1982 Apr 15;296(5858):647–650. doi: 10.1038/296647a0. [DOI] [PubMed] [Google Scholar]