Abstract
The leaves of Nicotiana glauca (N. glauca; Solanaceae) plant are a known, major human health concern. This study investigated the antioxidant activity and polyphenols composition of aerial parts of N. glauca collected from its wild habitat in Jordan, using Methanol-Conventional (MC) and deep eutectic solvents (DES) extraction methods in addition to nicotine content determination using UHPLC. Our results showed that the MC extract contains fewer total phenols and flavonoid content than the 90% DES extract, (0.1194 ± 0.009 and 0.311 ± 0.020 mg/mL equivalent to gallic acid) and (0.01084 ± 0.005 and 0.928 ± 0.09 mg/mL equivalent to rutin), respectively. Moreover, this study showed that the prepared MC extract contain 635.07 ppm nicotine, while the 90% DES extract contain 1194.91 ppm nicotine. Extracts prepared using the MC and the DES methods exhibited weak antioxidant activities; the highest was a 33% inhibition rate (equivalent to ascorbic acid), obtained by the 90% DES extract,. The performed UHPLC-MS/MS analysis in this study also revealed the presence of variations in the detected compounds between the two extraction methods. Furthermore, this study found that environmentally friendly DES extraction of N. glauca produced higher phenol and flavonoid content than the MC method; this highlights the superior efficiency and environmental benefits of sustainable chemistry methods for extracting valuable phytoconstituents.
Keywords: Nicotiana glauca, chromatography, mass spectrometry, antioxidants, deep eutectic solvents, sustainable extraction
1. Introduction
Tobacco, which is derived from the leaves of the Nicotiana plant, is known for its abuse and is recognized as a major global health concern. However, it has been used in traditional medicine by Native Americans to treat respiratory, parasitic, and mental problems [1]. Later in Europe, the tobacco plant was enlisted in various pharmacopeias, with therapeutic applications in treating catarrh, colds, and fevers. It was also used as a digestion aid, a purgative, and a narcotic. Later in the 20th century, some reports suggested that tobacco might lower the risk of Alzheimer’s disease, Parkinson’s, and Tourette’s syndrome [2].
Several compounds, like alkaloids, steroids, tannins, and flavonoids, were isolated from Nicotiana species. Many of these metabolites are bioactive with reported anti-inflammatory, antitumor, antibacterial, and antioxidant activities [3]. For instance, Nicotiana glauca Graham (Solanaceae) was shown to contain anabasine as the major alkaloid in the methanolic extract of their leaves [4], which is known to possess antiparasitic activity [5]. A study by Ameya et al. (2017) revealed that N. tabacum L. contains pyridine alkaloids with antibacterial activity against biofilm-forming pathogens [6]. These alkaloids were used to treat strep throat caused by Streptococcus pyogenes [7] and showed activity against Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, and Pseudomonas aeruginosa [8].
Moreover, several reports have highlighted the antioxidant activity of tobacco plant extracts, suggesting potential applications for various purposes. According to a recent study the methanol extract of N. glauca contains high levels of phenolic compounds, such as Chlorogenic acid and rutin [9]. These compounds were found to contribute to anti-inflammatory, anti-aging, and anticancer effects [3]. Another study from Saudi Arabia has demonstrated the antimicrobial effects of N. glauca against E. coli and S. aureus. The study concluded that the extracts from the leaves and flowers had the highest amounts of phytochemicals [10].
Conventional methods for extracting natural alkaloids and flavonoids, such as Soxhlet, maceration, percolation, and organic solvent extraction, are well-studied but have significant drawbacks. These techniques are time-consuming, inefficient, and often require large quantities of toxic, flammable, and non-biodegradable solvents, making them non-specific and not cost-effective. To address these issues, innovative solvents like deep eutectic solvents (DES) and natural deep eutectic solvents (NaDES) have been recently utilized [11,12,13,14]. As a subclass of ionic liquids (ILs), NaDES are considered less toxic, lower-cost, greener, and more efficient alternatives to both conventional organic solvents and ILs [15]. NaDES are usually prepared from a hydrogen bond donor and a hydrogen bond acceptor, which, when mixed in certain ratios, form a liquid at room temperature. Overall, the versatility, low toxicity, and environmentally friendly nature of NaDES make them attractive for a broad spectrum of industrial and research applications as solubilizers [16], drug delivery vehicles [17,18,19,20], stability enhancers [21], extraction and purification [22,23]. Additionally, NaDES themselves have been reported to possess antimicrobial [24], antioxidant [25], antibiofilm agents [26], and wound healing activity [17] among other beneficial effects. Choline chloride (a hydrogen bond acceptor) and malonic acid (a hydrogen bond donor) are natural compounds and are among the most commonly used substances for preparing NaDES [27].
In the context of the application of NaDES, this method was utilized for the extraction of polyphenols from Citrus aurantium L. peel [28]. The results showed enhanced recovery efficiency of polyphenols in the obtained extracts. Regarding tobacco plants, Hong et al. (2022) proposed the DES method for the extraction of solanesol from waste tobacco leaves [29]. Recently, cembranoid-type diterpenes compounds, known for their anticancer and antimicrobial effects, were extracted from tobacco flower waste using DES. Findings revealed the importance of green technologies in waste management and the extraction of bioactive natural compounds [30].
Hassan et al. (2014) conducted a phytochemical analysis of the N. glauca growing in Egypt [31]. Findings showed that the content of flavonoids in N. glauca was influenced by its habitat’s different conditions, which also affected the antioxidant activities. Therefore, research should take into consideration the use of medicinal plants relative to their composition of active and/or toxic metabolites collected from different regional areas.
This study aims to investigate the antioxidant activity and polyphenols composition of aerial parts of N. glauca species collected from its wild habitat in Jordan, using Methanol-Conventional (MC) and deep eutectic solvents (DES) extraction methods. This may provide information relevant to phenols content and antioxidant effect of the prepared extracts, revealing novel proposed uses with economic values ensuring the sustainable use of this plant species.
2. Materials and Methods
2.1. Plant Material
Fresh leaves of N. glauca were collected from widely grown plants in North Jordan, during the spring of 2022. The plant material was authenticated by an expert botanist in the Royal Botanical Gardens, Jordan. The voucher sample was deposited in the laboratory of Al-Ahliyya Amman University (Amman, Jordan). N. glauca leaves were dried under shade before the reduction in size using a conventional grinder and kept in a dark dry place at room temperature until used.
2.2. Methanol Conventional Extraction (MC)
An extract of the study plants was prepared using 50 g of dry plant material in 500 mL of methanol using the soaking method for 72 h at room temperature. This process was repeated twice, then the suspension was filtered and concentrated until a fine powder was obtained using Benchtop Manifold Freeze Dryer from Millrock Technology®.
2.3. Deep Eutectic Solvents (DES) Extraction
The DES were prepared from malonic acid and choline chloride in a 1:1 w/w molar ratio by physically mixing the two components gently on a hotplate to around 50–80 °C until a clear, homogeneous liquid was formed. The prepared DES mixture was mixed with deionized water to prepare three different extraction mixtures namely 30%, 70%, and 90% v/v. The cold extraction method was utilized by adding 5 g of the dry powder plant material in 25 mL of the DES extraction media. The plant material was soaked in the solvent for 72 h and then filtered to complete the extraction process.
2.4. Determination of Total Phenolic
The total phenolic content was measured using the Folin-Ciocalteu method as described by Alnsour et al., 2022 [32]. The phenolic content was determined calorimetrically at 765 nm. The total phenolic content (mg/mL) was determined as gallic acid equivalent. A stock solution of the plant extract was prepared at a concentration of 5 mg/mL. Serial dilutions were made, and an aliquot of each sample concentration (80 μL) was added to Folin–Ciocalteu (400 μL) reagent in a test tube, mixed with 7.5% sodium carbonate solution (320 μL). The solution was incubated in a dark place at 45 °C water bath for 30 min. Total phenolic content was expressed as gallic acid equivalent (mg/g), using the standard curve (Equation (1)):
| y = 0.0049x + 0.0426, R2 = 0.9991 | (1) |
y = absorbance at 765 nm and x = concentration of total phenolic content gallic acid equivalent mg/mL.
2.5. Determination of Total Flavonoids
The determination of total flavonoids was performed using a colorimetric method based on the formation of a complex flavonoid–aluminum, measured at a wavelength of 510 nm using a UV-spectrophotometer as described by Ubaydee et al. (2022) [33]. The results were expressed as (mg/mL) equivalents to quercetin. Briefly, a stock solution of the plant extract at a concentration of 5 mg/mL was prepared. Serial dilutions were made, 1 mL of each concentration was added into (0.5 mL) AlCl3, (0.5 mL) NaNO2, (2 mL) NaOH, and (4 mL) distilled water. The mixture was incubated at room temperature for 15 min.
Total Flavonoid content was expressed as rutin equivalent (mg/mL), using the standard curve (Equation (2)):
| y = 0.0009x + 0.613, R2 = 0.994 | (2) |
y = absorbance at 510 nm and x = concentration of total phenolic content rutin equivalent mg/mL.
2.6. In-Vitro Antioxidant Activity
The 2,2-diphenyl-1-picrylhydrazyl (DPPH) scavenging activity was used as described by Al-Bayati et al. (2023) [34]. For the reaction reagent, DPPH was dissolved in methanol at a concentration of (0.04 g/mL). The reaction was performed by dissolving plant extract in methanol at a concentration of (0.01 g/mL). An aliquot of 1 mL of the plant extract solution was mixed with 3 mL of DPPH and completed to a final volume of 10 mL using methanol, then allowed to stand in darkness for 30 min. Absorbance was measured at a wavelength of 517 nm. Ascorbic acid was used as a reference for comparison (Sigma Aldrich, Darmstadt, Germany). A calibration curve of ascorbic acid was used for the calculation of the effective concentration required for scavenging DPPH free radicals (% inhibition Equation (3)).
| % inhibition = [(A control − A sample)/A control] × 100 | (3) |
where: A control = absorbance of the control sample, and A (sample) = absorbance of the sample.
2.7. UHPLC-MS/MS Methodology
Instrumentation and MS Parameters
The UHPLC coupled with Impact II QTOFMS Bruker Daltonik (Bremen, Germany) was used for screening the compounds of interest using the same method previously described by Al-Bayati et al. (2023) [34]. The instrument operation conditions were as follows: Apollo II ion funnel electrospray source, capillary voltage (2500 V), nebulizer gas (2 bar), and nitrogen dry gas at a flow rate of 8 L/min (200 °C). The mass accuracy was <1 ppm; with Full Sensitivity Resolution (50000 FSR) and the TOF repetition rate of 20 kHz.
Bruker Solo 2.0_C-18 UHPLC column (100 mm × 2.1 mm × 2.0 μm) was used for chromatographic separation at a flow rate of 0.51 mL/min (40 °C). The mobile phase consists of (A: 0.05% formic acid in water), and (B: acetonitrile). Gradient elution was used as follows: 0–27 min linear gradient from 5–80% B; 27–29 min 95% B; 29.1 min 5% B. The total analysis time was 35 min in positive mode and 35 min in negative mode, with an injection volume of 3 µL.
MC and DES samples stock solutions were prepared by dissolving an appropriate amount of the plant extract in dimethyl sulfoxide-DMSO (analytical grade), then diluted with acetonitrile to complete 50 mL, then centrifugation was performed at 4000 rpm was applied for 2 min. All the other reagents, Acetonitrile, methanol, water, and formic acid used were LC-MS grade.
Sample preparation: 100 µL of each sample has been dissolved in 900 µL of MeOH. A 1.0 mL was transferred to an autosampler and injected. Identification of phenols and flavonoid compounds was based on the retention time (Rt), mass spectrum (m/z), and molecular formula, compared to a previously developed integrated library of natural compounds.
2.8. Nicotine Content Determination
The nicotine content was determined according to the process described by Kheawfu et al. in their 2021 study [35]. Briefly, each obtained extract was analyzed by UHPLC coupled with Impact II QTOFMS Bruker Daltonik (Bremen, Germany). Bruker Solo 2.0_C-18 UHPLC column (100 mm × 2.1 mm × 2.0 μm). A linear elution mobile phase composed of Sodium acetate, methanol, and trimethylamine (88:12:0.5 v/v) (pH = 4.2) was used. The mobile phase elution was adjusted to a flow rate of 1 mL/min and measured at UV = 259 nm. The total analysis time was 20 min in positive mode.
Nicotine standard (AccuStandard®, Inc., New Haven, CT, USA) was used for establishing the calibration curve at concentrations ranging from (0.10–2.00 µg/mL) in water and was used for the calculation of nicotine content in plant extracts as ppm values.
Sample preparation for UHPLC-MS/MS analysis: (A) 500 µL from MC or DES extract samples were diluted with 500 µL methanol, then the solution was centrifuged at 4000 rpm for 2.0 min. Next, 1.0 mL was transferred to the autosampler and 3.0 µL was injected into the system (D.f. = 2). (B) Then, 50 µL was taken from sample (A), and diluted with 1950 µL of methanol. Next, 1.0 mL was transferred to the autosampler and 3.0 µL was injected into the system (D.f. = 40). (C) Then, 100 µL was taken from sample (B), and diluted with 1900 µL of methanol. Next, 1.0 mL was transferred to the autosampler and 3.0 µL was injected into the system (D.f. = 20), (D.f.total = 2 × 40 × 20).
3. Results
In the present study, the efficiency of the synthesized DES to recover phenolic compounds from N. glauca leaf was tested using two representative phytochemical indices such as TPC and TFC. The structure of DES significantly influences their physicochemical properties, impacting their extraction efficiency. In this study, DES were created using CC as a hydrogen bond acceptor (HBA) and MA as a hydrogen bond donor (HBD), with water added at 10%, 30%, and 70% concentrations. All the solvents remained stable without precipitating during preparation, extraction, and analysis. Viscosity and polarity are crucial for DES’s efficiency and can be modified by adding water. Adding water helps lower viscosity and enhances tunability. However, excessive water can weaken interactions within the DES and with extracted components, reducing efficiency. The optimal water addition (25–30%) improves extraction, while higher amounts (40–75%) can diminish it. Previous studies, such as Dai et al.’s 2013 study [36], noted that significant water addition can alter polarity and disrupt hydrogen bonds in DES. Additionally, the biological activities of extracts using DES were compared to those obtained with traditional solvents like methanol.
3.1. Total Phenol Content
The extract that obtained from the MC method was shown to contain less total phenolic compounds (0.1194 ± 0.009 mg/mL equivalent to gallic acid), compared to the DES extracts which showed similar total phenol content for the three prepared extract ratios with almost no significant difference between the three tested DES ratios (30, 70, and 90%) corresponding to an average of 0.312 ± 0.13 mg/mL (equivalent to gallic acid) (Table 1). This finding is in close accordance with the results reported by others who found that carboxylic-based DES have a high ability to extract phenolics from plants [37,38,39]. The high extraction efficiency of DES may be due to the hydrogen bond interaction between the phenolic compounds and the DES’s components.
Table 1.
Total phenols content in MC and DES extracts at three extract ratios.
| Extraction Media | mg/mL ± SD (Equivalent to Gallic Acid) |
|---|---|
| DES 30% | 0.326 ± 0.11 |
| DES 70% | 0.300 ± 0.03 |
| DES 90% | 0.311 ± 0.02 |
| MC | 0.119 ± 0.01 |
3.2. Total Flavonoid Content
Total flavonoid content was also analyzed to compare the extraction efficiency of DES and MC, as well as to evaluate the difference between DES with varying water content (Table 2). The DES with the lowest water content (10%) resulted in the highest total flavonoid content, which was 7–8 times higher than the other two DES and about 84 times higher than MC. The extraction pattern of flavonoids by the tested DES with different water content and with MC was found to be similar to that of the phenolic extraction. It can be concluded that extraction yields not only depend on solvent types but also on the physical and chemical characteristics of the samples. It should be mentioned that flavonoids themselves can also act as hydrogen bond donors, potentially competing with the HBD used in DES preparation. However, in this study, only one class of DES were tested, so this factor had no effect.
Table 2.
Total flavonoid content in MC and DES extracts at three extract ratios.
| Extraction Media | mg/mL ± SD (Equivalent to Rutin) |
|---|---|
| DES 30% | 0.128 ± 0.03 |
| DES 70% | 0.115 ± 0.14 |
| DES 90% | 0.928 ± 0.09 |
| MC | 0.011 ± 0.01 |
3.3. Antioxidant Activity (DPPH Assay) for N. glauca Leaf Extracts
Maceration is a traditional and one of the most ancient extraction processes applied for the extraction of bioactive substances such as phenolic compounds. Although maceration is a time-consuming method, it has been reported to be adequate and subsequent for the recovery of antioxidants from various plant materials. The Antioxidant capacities of the extracts obtained by DES and MC were measured using DPPH. The most concentrated DES extract (90%) exhibited the highest % Inhibition of free radical activity at 33%, equivalent to ascorbic acid. In contrast, the other DES ratios and MC exhibited weak antioxidant activities and failed to change the color of the DPPH reagent from dark purple to pale yellow, rendering the antioxidant test ineffective for these samples. It can be concluded that the nature of the extraction solvent greatly influences the type of extractives obtained and their antioxidant activity. Many reports in the literature demonstrated that extracts prepared in organic acid-based DES show better antioxidant activity than those prepared in aqueous, methanolic, and ethanolic extracts. Pavic et al., (2019) [40] found that Ruta leaf extracts prepared in NADES of CC and citric acid in a 2:1 molar ratio had the highest phenolic content and the highest DPPH radical scavenging activity. Additionally, Bakirtzi et al., (2016) [41] reported the highest reducing power in sage extracts obtained using NADES of lactic acid and CC in a 3:1 molar ratio.
3.4. Identification of Phenols Using UHPLC-MS/MS Analysis
3.4.1. Methanol Conventional Extraction (MC)
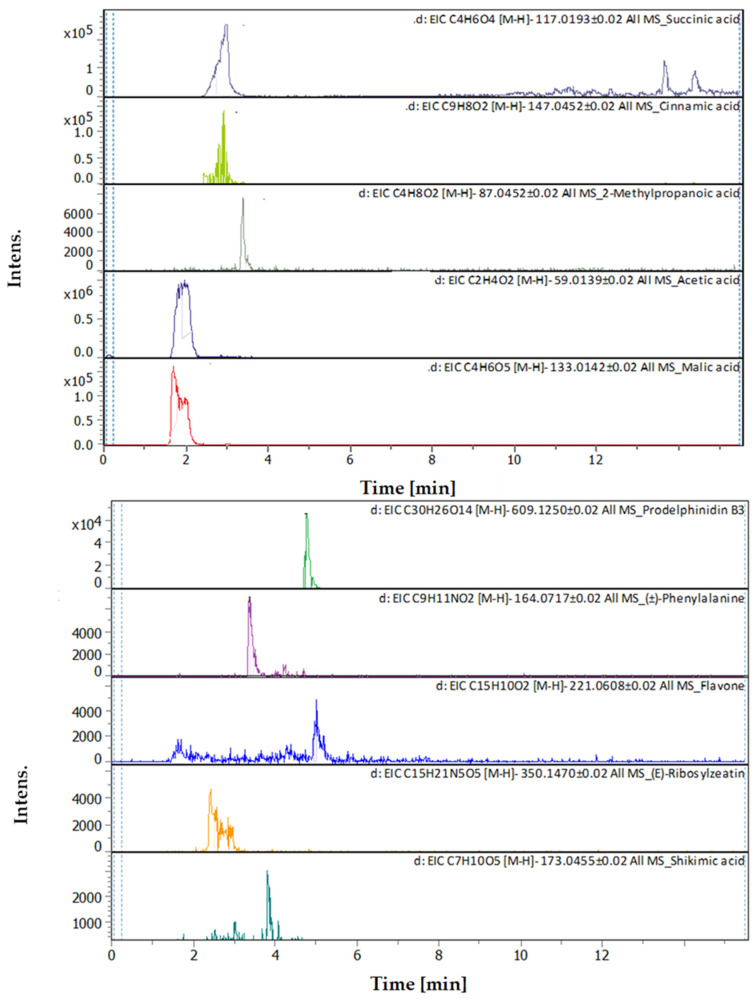
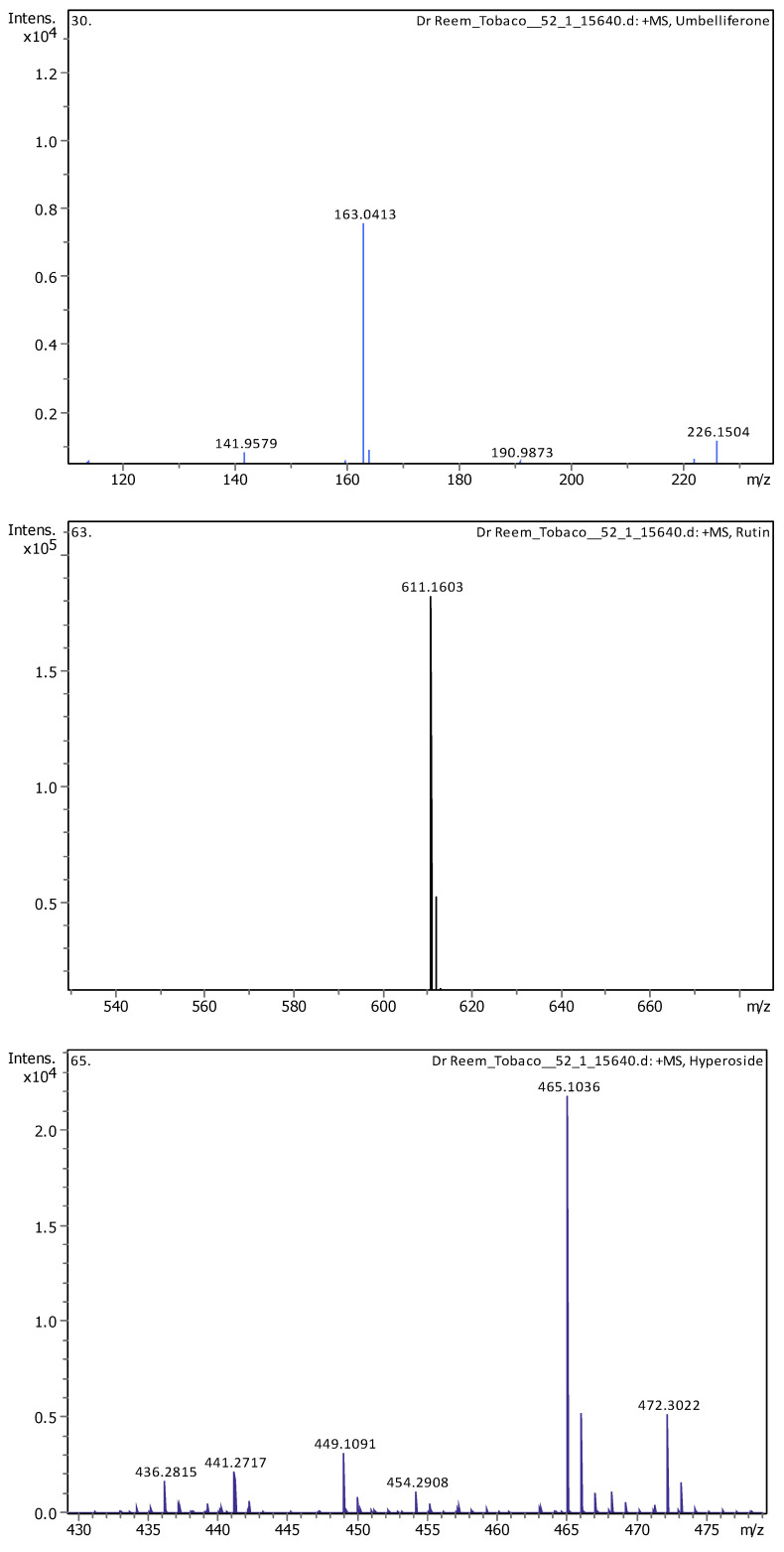
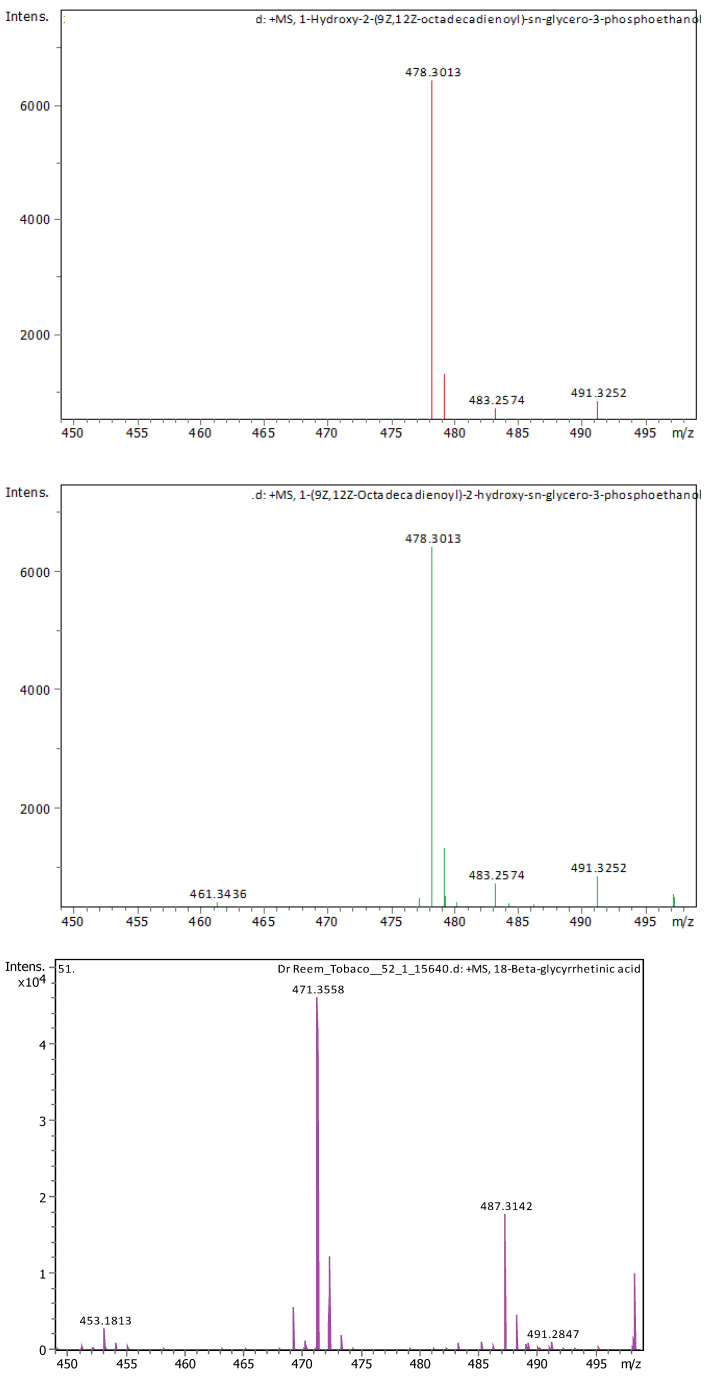
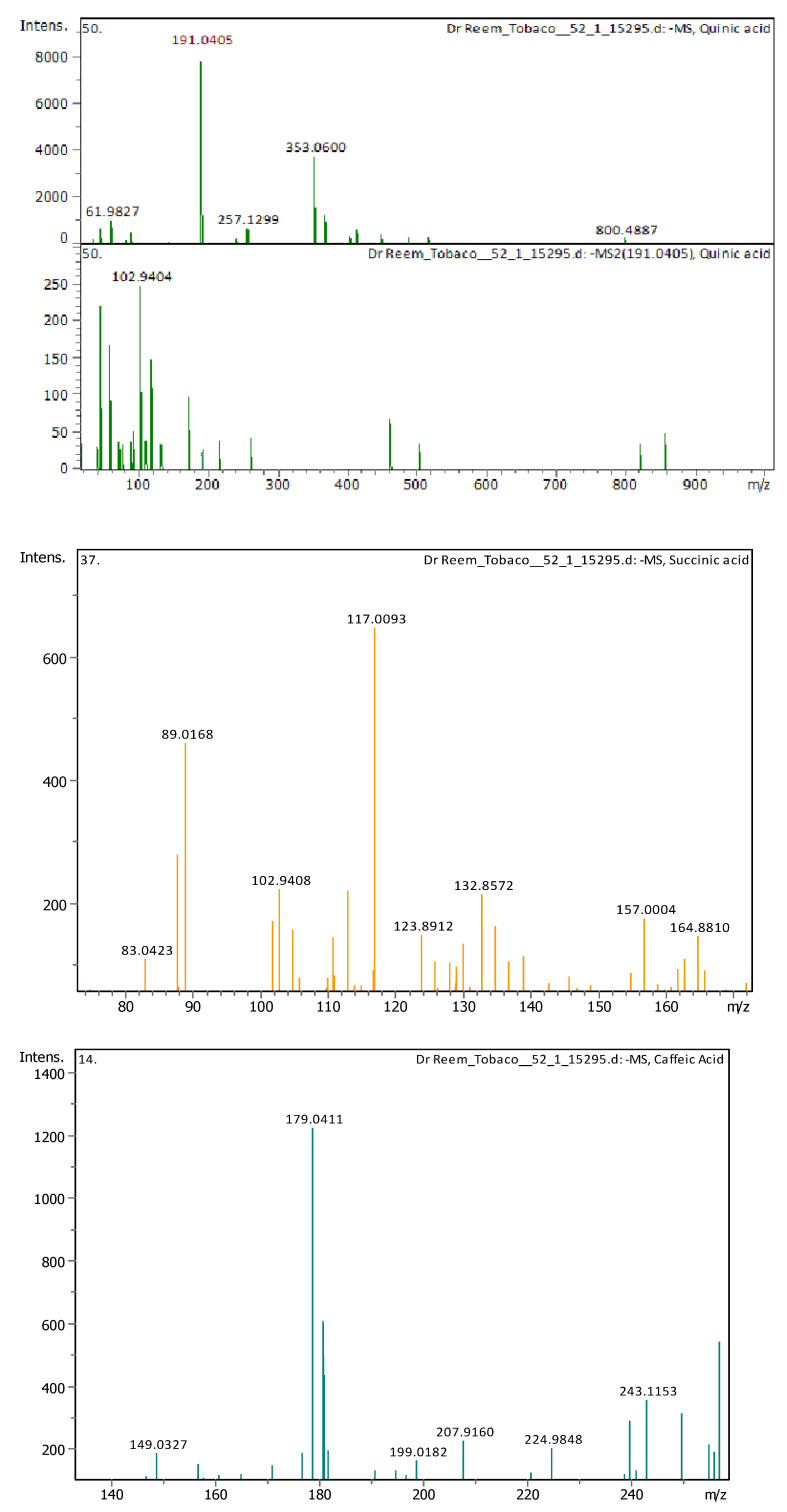
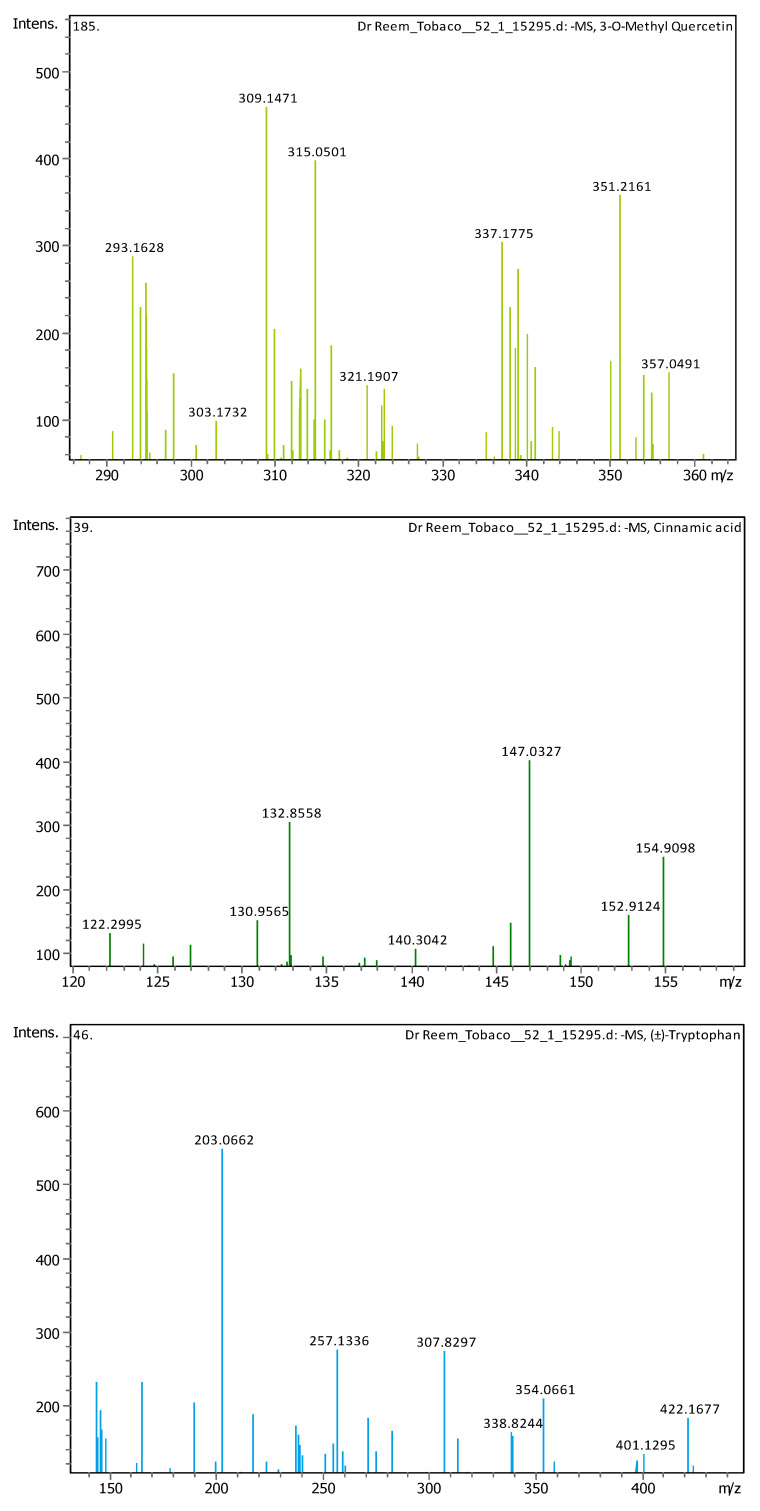
A total of twenty-three different phenolic components have been detected in the MC extract using the LC-MS/MS analysis, and the integrated natural compounds library. The retention time (Rt)mass-to-charge ratio (m/z), and molecular formula for the detected compounds (positive and negative ion modes) are listed in Table 3. A total of fourteen compounds were detected- in the negative mode, and nine were detected in the positive mode.
Table 3.
UHPLC-MS/MS analysis (positive and negative modes) showing all components detected in N. glauca MC extract based on retention time (Rt), Mass-to-charge ratio (m/z), and molecular formula.
| # | Rt [min] | m/z Meas. | M Meas. | Ions | Name | Molecular Formula |
|---|---|---|---|---|---|---|
| 1 | 0.61 | 131.04612 | 132.05339 | [M–H]− | L-Asparagine | C4H8N2O3 |
| 2 | 0.62 | 114.05616 | 115.06344 | [M–H]− | Proline | C5H9NO2 |
| 3 | 0.97 | 180.06594 | 181.07322 | [M–H]− | L-Tyrosine | C9H11NO3 |
| 4 | 1 | 117.01933 | 118.02661 | [M–H]− | Succinic acid | C4H6O4 |
| 5 | 1.28 | 147.04508 | 148.05236 | [M–H]− | Cinnamic acid | C9H8O2 |
| 6 | 1.28 | 164.0718 | 165.07908 | [M–H]− | (±)-Phenylalanine | C9H11NO2 |
| 7 | 2.03 | 203.08226 | 204.08954 | [M–H]− | (±)-Tryptophan | C11H12N2O2 |
| 8 | 2.9 | 191.05607 | 192.06335 | [M–H]− | Quinic acid | C7H12O6 |
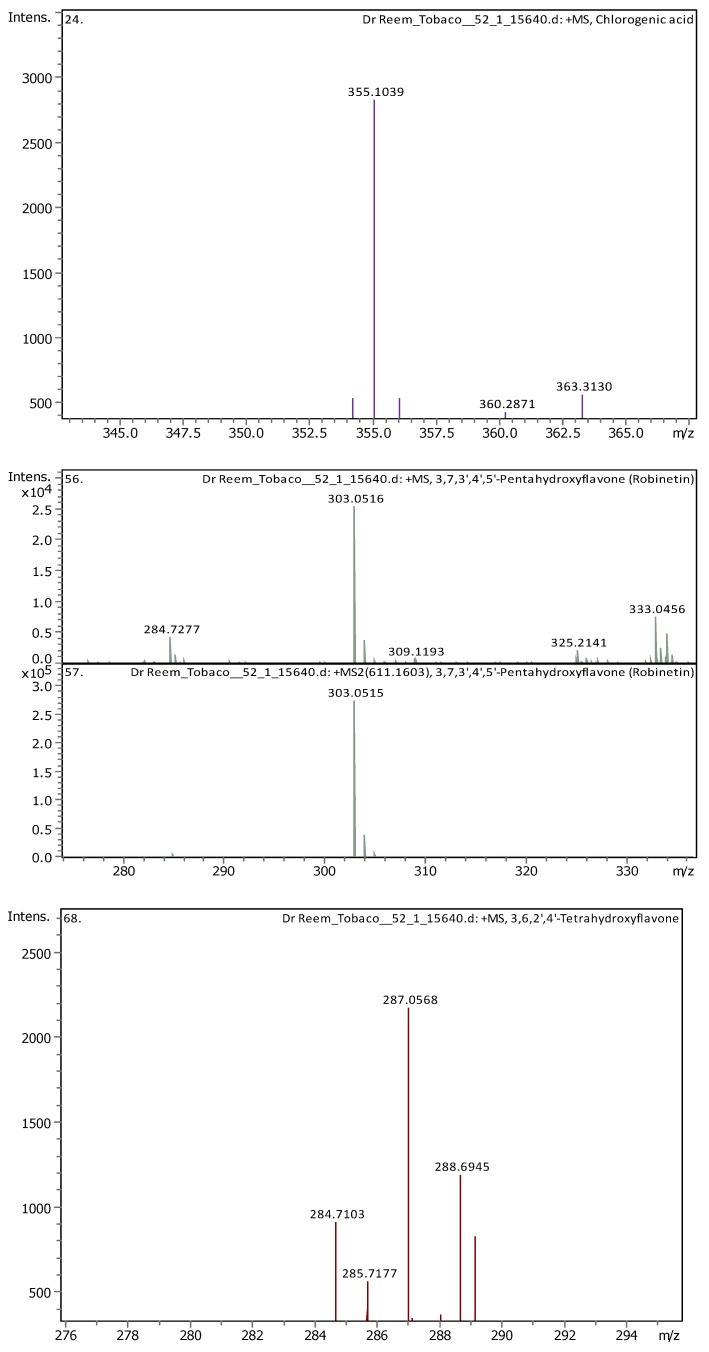
| 9 | 2.96 | 355.10248 | 354.09521 | [M+H]+ | Chlorogenic acid | C16H18O9 |
| 10 | 3.05 | 179.03487 | 180.04214 | [M–H]− | Caffeic Acid | C9H8O4 |
| 11 | 5.07 | 163.03979 | 162.03251 | [M+H]+ | Umbelliferone | C9H6O3 |
| 12 | 5.12 | 203.08228 | 204.08955 | [M–H]− | (±)-Tryptophan | C11H12N2O2 |
| 13 | 5.57 | 609.1455 | 610.15278 | [M–H]− | Quercetin 3-rutinoside | C27H30O16 |
| 14 | 5.61 | 303.05014 | 302.04287 | [M+H]+ | Robinetin | C15H10O7 |
| 15 | 5.61 | 465.10293 | 464.09566 | [M+H]+ | Hyperoside | C21H20O12 |
| 16 | 5.62 | 611.16099 | 610.15353 | [M+H]+, [M+Na]+ | Rutin | C27H30O16 |
| 17 | 6.31 | 179.05592 | 180.0632 | [M–H]− | Starch | C6H12O6 |
| 18 | 6.37 | 287.0557 | 286.04842 | [M+H]+ | 3,6,2′,4′-Tetrahydroxyflavone | C15H10O6 |
| 19 | 9.1 | 315.05061 | 316.05788 | [M–H]− | 3-O-Methyl Quercetin | C16H12O7 |
| 20 | 21.09 | 478.28898 | 477.2817 | [M+H]+ | 1-Hydroxy-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphoethanolamine (NMR) | C23H44NO7P |
| 21 | 22.31 | 478.28916 | 477.28189 | [M+H]+ | 1-(9Z,12Z-Octadecadienoyl)-2-hydroxy-sn-glycero-3-phosphoethanolamine (NMR) | C23H44NO7P |
| 22 | 22.49 | 471.35137 | 470.34409 | [M+H]+ | 18-Beta-glycyrrhetinic acid | C30H46O4 |
| 23 | 28.62 | 221.15517 | 222.16244 | [M–H]− | Histamine | C10H18N6 |
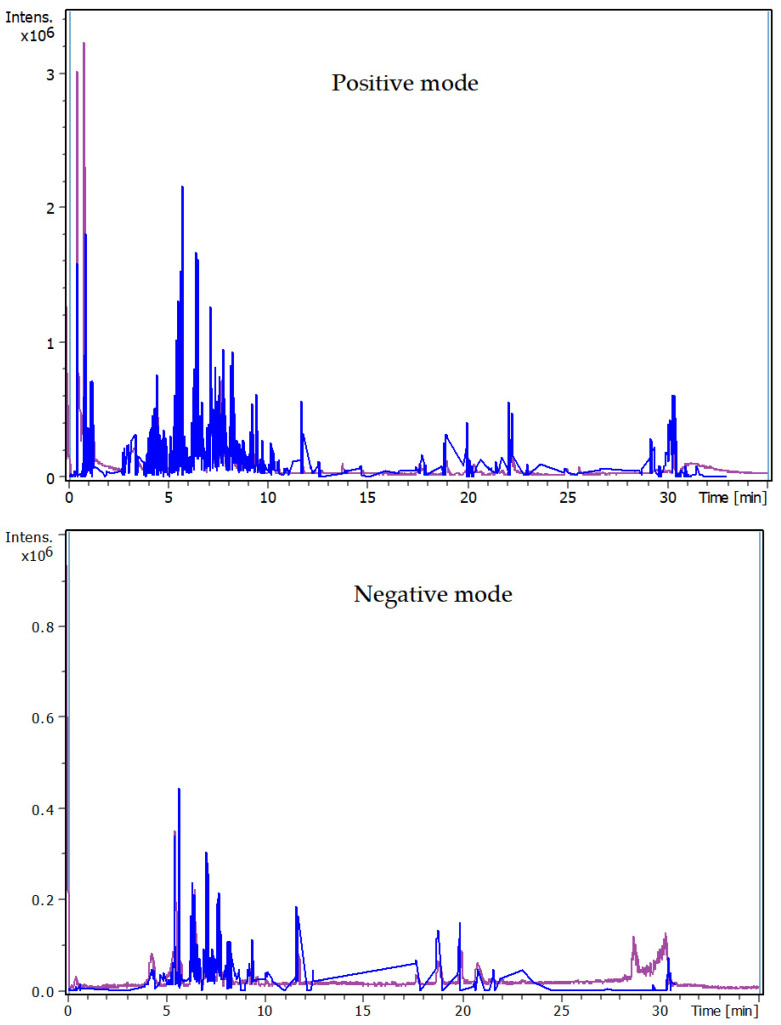
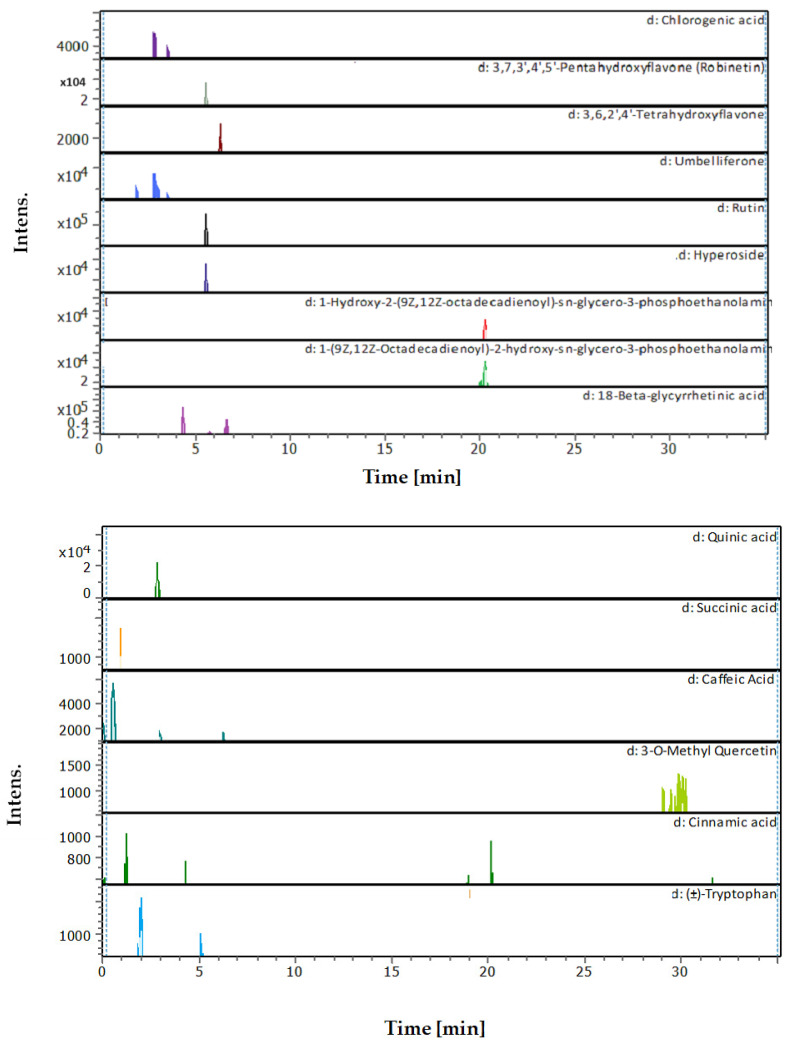
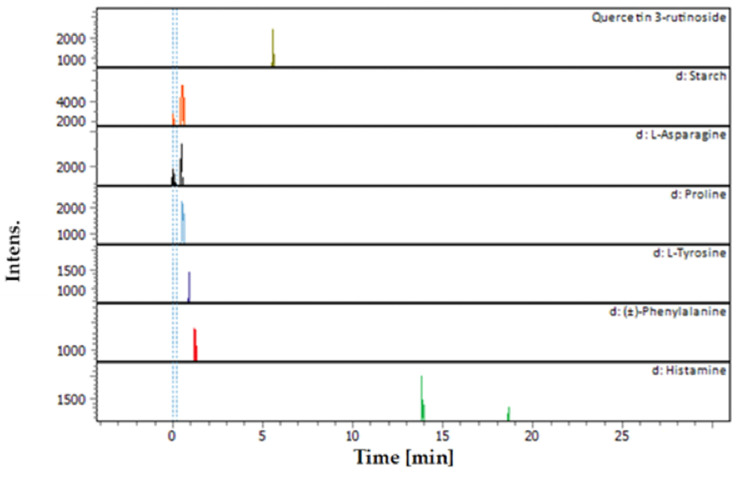
Figure 1 shows the total ion chromatogram for all compounds detected in the MC leaves extracts. The UHPLC-chromatograms which display the peaks and retention time of each compound detected in the extract are shown in Figure 2. The mass spectrum (m/z) and fragments for each compound detected in the MC extract are presented in Appendix A.
Figure 1.
Total ion chromatograms for all compounds detected in N. gluca MC extract.
Figure 2.
The UHPLC-chromatograms show peaks and retention time of each compound detected in N. glauca prepared by MC extraction.
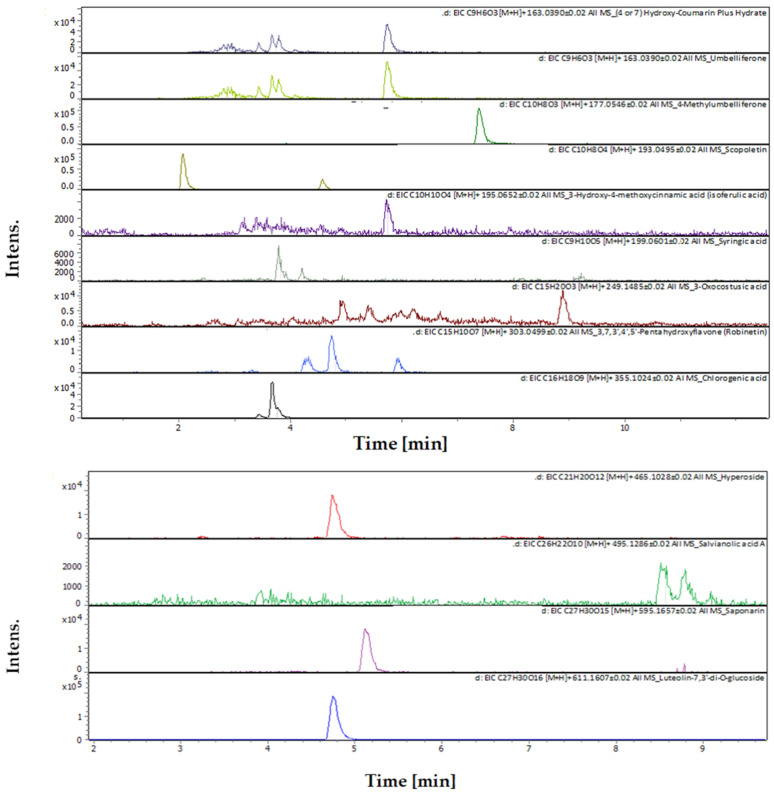
3.4.2. Deep Eutectic Extraction (DES)
A total of twenty-three different phenolic components have been detected in the DES extract using the UHPLC-MS/MS analysis, and the integrated natural compounds library. The Rt, m/z, and molecular formula for the detected compounds (positive and negative ion modes) are listed in Table 4. A total of ten compounds were detected in the negative mode, and thirteen were detected in the positive mode.
Table 4.
UHPLC-MS/MS analysis (positive and negative modes) showing all components detected in N. glauca DES extract based on retention time (Rt), Mass (m/z), and molecular formula.
| # | Rt [min] | m/z Meas. | M Meas. | Ions | Name | Molecular Formula |
|---|---|---|---|---|---|---|
| 1 | 1.65 | 133.00998 | 134.0174 | [M–H]−, [M–H H2O]− |
Malic acid | C4H6O5 |
| 2 | 1.91 | 59.01128 | 60.01856 | [M–H]− | Acetic acid | C2H4O2 |
| 3 | 2.39 | 350.14397 | 351.15125 | [M–H]− | (E)-Ribosylzeatin | C15H21N5O5 |
| 4 | 2.96 | 117.01473 | 118.022 | [M–H]− | Succinic acid | C4H6O4 |
| 5 | 3.32 | 147.04052 | 148.0478 | [M–H]− | Cinnamic acid | C9H8O2 |
| 6 | 3.35 | 87.04212 | 88.04939 | [M–H]− | 2-Methylpropanoic acid | C4H8O2 |
| 7 | 3.36 | 164.06681 | 165.07409 | [M–H]− | (±)-Phenylalanine | C9H11NO2 |
| 8 | 3.69 | 163.038920 | 162.031640 | [M+H]+ | Umbelliferone | C9H6O3 |
| 9 | 3.7 | 355.102500 | 354.095190 | [M+H]+, [M+K]+, [M+Na]+ |
Chlorogenic acid | C16H18O9 |
| 10 | 3.79 | 173.04114 | 174.04841 | [M–H]− | Shikimic acid | C7H10O5 |
| 11 | 3.8 | 199.057710 | 198.050440 | [M+H]+ | Syringic acid | C9H10O5 |
| 12 | 4.6 | 193.049450 | 192.042170 | [M+H]+ | Scopoletin | C10H8O4 |
| 13 | 4.76 | 465.102820 | 464.095540 | [M+H]+ | Hyperoside | C21H20O12 |
| 14 | 4.77 | 303.050010 | 302.042740 | [M+H]+ | Robietin | C15H10O7 |
| 15 | 4.77 | 611.160240 | 610.152970 | [M+H]+ | Luteolin-7,3′-di-O-glucoside | C27H30O16 |
| 16 | 4.91 | 609.1281 | 610.13537 | [M–H]− | Prodelphinidin B3 | C30H26O14 |
| 17 | 5 | 221.05958 | 222.06686 | [M–H]− | Flavone | C15H10O2 |
| 18 | 5.15 | 595.165470 | 594.158150 | [M+H]+, [M+Na]+ | Saponarin | C27H30O15 |
| 19 | 5.74 | 195.065060 | 194.057780 | [M+H]+ | 3-Hydroxy-4-methoxycinnamic acid (isoferulic acid) | C10H10O4 |
| 20 | 7.39 | 163.039120 | 162.031850 | [M+H]+ | (4 or 7) Hydroxy-Coumarin Plus Hydrate | C9H6O3 |
| 21 | 7.41 | 177.054460 | 176.047190 | [M+H]+ | 4-Methylumbelliferone | C10H8O3 |
| 22 | 8.54 | 495.125630 | 494.118360 | [M+H]+ | Salvianolic acid A | C26H22O10 |
| 23 | 11.6 | 249.148000 | 248.140730 | [M+H]+ | 3-Oxocostusic acid | C15H20O3 |
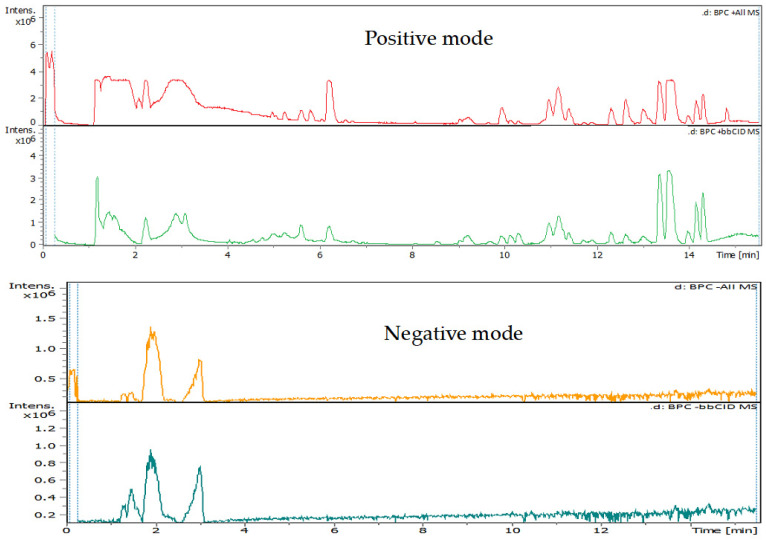
Figure 3 shows the total ion chromatogram for all compounds detected in the DES leaf extracts. The UHPLC-chromatograms showing peaks and retention time of each compound detected in the extract are shown in Figure 4. The Mass spectrum (m/z) and fragments for each compound detected in the MC extract are available in Appendix B.
Figure 3.
Total ion chromatograms for all compounds detected in N. glauca DES extract.
Figure 4.
The UHPLC-chromatograms show peaks and retention time of each compound detected in N. glauca DES extract.
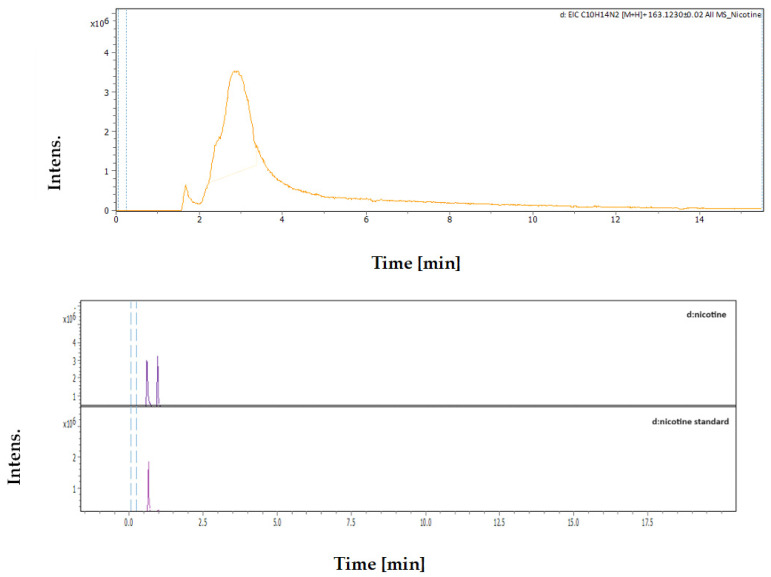
3.4.3. Identification and Quantification of Nicotine
The identification of nicotine in the extract samples was performed using a multiple external standards method using the UHPLC-MS/MS system, based on the retention time, m/z, and molecular formula as shown in Table 5 and Figure 5.
Table 5.
UHPLC-MS/MS analysis for nicotine detected in N. glauco extracts based on retention time.
| Rt [min] | m/z Meas. | M Meas. | Ions | Name | Molecular Formula |
|---|---|---|---|---|---|
| 2.89 | 163.12293 | 324.2313 | [M+H+H]2+ | Nicotine | C10H14N2 |
Figure 5.
The UHPLC-chromatogram and mass spectra show peaks and retention time of nicotine detected in N. glauca extract.
The results indicated that the concentration of nicotine in the MC extract was 635.07 ppm (µg/mL) (0.064%), while the concentration in the DES extract was 1194.91 ppm (µg/mL) (0.119%) (i.e., almost twice). This suggests that the DES extraction method yielded a higher concentration of nicotine compared with the MC method, proving that the DES method is more effective for the extraction of this alkaloid. The levels of nicotine extracted using the two techniques are illustrated in Table 6.
Table 6.
Area under the curve and concentration of nicotine (ppm) in the MC and DES extracts based on multiple external standards method.
| Sample | MC Extract | DES Extract |
|---|---|---|
| Area of Nicotine in Sample | 2,526,713 | 4,192,477 |
| Concentration of Nicotine | 635.07 ppm | 1194.91 ppm |
Alkaloids are the dominant class of constitutive secondary chemicals in N. glauca, but in contrast to most Nicotiana species, the major alkaloid in N. glauca tissues is anabasine [42]. To the best of our knowledge, limited work was performed to study the content of nicotine in the species N. glauca. Comparing our findings with the other extraction methods mentioned below for the species N. tabacum, both extracts (MC and DES) showed a lower content of nicotine than the previously published data. A similar investigation was performed by Tayoub et al. (2015) [43] aimed at measuring nicotine levels in the leaves of seven different varieties of N. tabacum, which were cultivated in Syria. The study reported that nicotine is naturally present in a concentration of 0.3 to 3%. Tantullavetch et al. (2007) [44] reported that nicotine extraction yield from tobacco leaves with acid-base extraction was approximately 4.2%. Kheawfu et al., (2021) [35] studied the effect of the use of different extraction solvents on the yield of nicotine extracted from N. tabacum leaves collected from different positions on the stem. Findings using the acid-base extraction method presented the highest nicotine content (43.28–63.17%) compared to the maceration extraction method using water (1.27–12.07%) and ethanol (10.78–16.99%).
In a study by Banožić et al., (2021) [45] nicotine was extracted using a microwave-assisted extraction method. The extraction took place under different conditions and temperatures. The study’s findings showed that nicotine is the dominant compound with concentrations in the range 1.512–5.480, 1.886–3.709, 2.628–4.840, and 0.867–1.783% for leaves, scrap, dust, and midrib extracts, respectively. Therefore, the concentration of nicotine is highly dependent on tobacco species type, variety, growing, and environmental conditions, in addition to variations in the extraction techniques used.
4. Discussion
Several studies reported high toxicity of the plant N. glauca caused by its alkaloid content, namely nicotine and other derivatives [46,47]. The use of N. glauca as an anti-jaundice plant among herbalists and traditional healers was reported in Jordanian folk medicine [48]. Nevertheless, few reports investigated the phytochemical composition of the wild species grown in Jordan. Researchers detected the presence of different phenolic compounds in tobacco plants such as kaempferol-3-O-rutinoside, quercetin-3-O-rutinoside, in addition to the main components of tobacco polyphenols are chlorogenic acid and rutin [4,49,50]. In this study, the UHPLC-MS/MS analysis revealed the presence of variations in the detected compounds between the two extraction methods used. Mainly, 3,7,3′,4′,5′-Pentahydroxyflavone (Robinetin), Chlorogenic acid, Hyperoside, Rutin, and Umbelliferone, were detected in both extracts. Whereas (4 or 7) Hydroxy-Coumarin Plus Hydrate, 3-Hydroxy-4-methoxycinnamic acid (isoferulic acid), 3-Oxocostusic acid, 4-Methylumbelliferone, Luteolin-7,3′-di-O-glucoside, Salvianolic acid A, Saponarin, Scopoletin, and Syringic acid, were detected in the DES extract only. On the other side, 1-(9Z,12Z-Octadecadienoyl)-2-hydroxy-sn-glycero-3-phosphoethanolamine,18-Beta glycyrrhetinic acid, 1-Hydroxy-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphoethanolamine, and 3,6,2′,4′-Tetra hydroxy flavone, were detected in the MC extract only.
Our work on the total content of phenolic phytocomponents showed the MC extract contained fewer total phenols and flavonoid content compared to the DES extract. These findings were expected, as previous studies highlighted the advantages of using NaDES as a green solvent in the extraction of phytochemicals, including enhanced extraction yield, and additional environmental benefits [28,29]. The results of the antioxidant DPPH test revealed that both extraction methods have weak antioxidant activities, except for the concentrated (90%) DES extract which showed a moderated antioxidant activity compared to ascorbic acid. The (90%) DES extract was also found to contain the highest total flavonoid content among the other extract samples. These findings are in correlation with phenols and flavonoid content, which are the most contributing natural compounds for antioxidant activity. Our findings agree with a previous study by Trifa, et al. (2020) [51], investigating N. glauca extract collected from central Algeria, which showed good antioxidant activity in the ethyl acetate and n-butanol fractions, which was related to the content of polyphenols. Similarly, Sumengen et al. (2023) [52] conducted a study to investigate the phytochemical composition of N. glauca methanol leaf extract collected from Northern Cyprus. Findings showed that N. glauca methanolic extract had the highest antioxidant activity determined using the DPPH test and were correlated to their content of phenols and flavonoids. The latter compared his findings with others’ previous work, showing that variations in the antioxidant effects are expected, due to variations in the phytochemical composition. These variations may occur due to several factors, such as growing conditions, environmental variations, stage of plant development, season of collection, and geographical origin, in addition to methods of extraction and solvent used.
It was previously found that the acid-base extraction method contains the highest nicotine content, as this method aids in solubilizing the alkaloids by converting them to the salt form, which enhances their solubility in polar solvents [35]. It was concluded that methods of extraction and solvent used, plant part, and many other variables would largely be affecting nicotine content, in addition to other phytocomponents obtained in the plant extract.
The findings of the presented study showed that the MC extract contains 635.07 ppm of nicotine compared to 1194.91 ppm of nicotine found according to the DES extraction method. In agreement with other published work, Kheawfu et al. (2021) [35] found that extraction with water for 24 h gave the highest amount of nicotine. Whereas Puripattanavong et al. (2013) [53] suggested that using methanol and ethanol gave the highest yield percentage of nicotine extraction from N. tabacum leaves compared with other solvents and extraction media. A similar work by Massadeh et al. (2022) [4] screening the phytochemical constituent in the leaves of N. glauca collected from the north region in Jordan [4]. Using the UPLC-MS and GC-MS analysis, anabasine was detected as the major alkaloid, while nicotine was not detected in their studied extract.
5. Conclusions
In conclusion, N. glauca contains a substantial content of valuable phenols and flavonoids, with a low amount of nicotine compared to other Nicotiana species. Moreover, results demonstrated that the extract that resulted from the MC method has the lowest content of the detected phytochemicals compared to the extracts that resulted from the DES method. Similarly, the antioxidant activity of the prepared extracts showed no effects of all extracts except for the concentrated extract from the DES extraction (90%), which showed the highest activity (33%) relative to ascorbic acid.
These results emphasize the critical importance of adopting green chemistry techniques, not only for their environmental benefits but also for their superior efficiency in producing higher yields of valuable phytoconstituents. In addition, optimization of the extraction procedure, as well as plant selection and preparation was shown to largely influence the content of secondary phytocompounds. Despite the work performed and published on the content of alkaloids in the study plant species, further investigation of the proportions of other alkaloids found in N. glauca is still required.
Appendix A
Appendix B
Author Contributions
Conceptualization, R.I. and F.A.-A.; methodology, R.I., L.A. and T.R.A.-S.; validation, S.H.A. and K.W.O.; formal analysis, R.I.; data curation, F.A.-A.; writing—original draft preparation, R.I., F.A.-A. and S.H.A.; writing—review and editing, S.H.A. and K.W.O. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Berlowitz I., Torres E.G., Walt H., Wolf U., Maake C., Martin-Soelch C. “Tobacco Is the Chief Medicinal Plant in My Work”: Therapeutic Uses of Tobacco in Peruvian Amazonian Medicine Exemplified by the Work of a Maestro Tabaquero. Front. Pharmacol. 2020;11:594591. doi: 10.3389/fphar.2020.594591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Charlton A. Medicinal Uses of Tobacco in History. J. R. Soc. Med. 2004;97:292–296. doi: 10.1177/014107680409700614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zou X., Bk A., Abu-Izneid T., Aziz A., Devnath P., Rauf A., Mitra S., Emran T.B., Mujawah A.A.H., Lorenzo J.M., et al. Current Advances of Functional Phytochemicals in Nicotiana Plant and Related Potential Value of Tobacco Processing Waste: A Review. Biomed. Pharmacother. 2021;143:112191. doi: 10.1016/j.biopha.2021.112191. [DOI] [PubMed] [Google Scholar]
- 4.Massadeh R., El-Elimat T., Al-Gharaibeh M., Tawaha K., Alali F. UPLC-HRESI-MS and GC-MS Analysis of the Leaves of Nicotiana glauca. Acta Pharm. 2022;72:97–108. doi: 10.2478/acph-2022-0001. [DOI] [PubMed] [Google Scholar]
- 5.Silva F.D.S., Albuquerque U.P., Costa Júnior L.M., Lima A.D.S., Nascimento A.L.B.D., Monteiro J.M. An Ethnopharmacological Assessment of the Use of Plants against Parasitic Diseases in Humans and Animals. J. Ethnopharmacol. 2014;155:1332–1341. doi: 10.1016/j.jep.2014.07.036. [DOI] [PubMed] [Google Scholar]
- 6.Ameya G., Manilal A., Merdekios B. In Vitro Antibacterial Activity and Phytochemical Analysis of Nicotiana tabacum, L. Extracted in Different Organic Solvents. Open Microbiol. J. 2017;11:352–359. doi: 10.2174/1874285801711010352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anumudu C., Mi N., Cc O., Io N., Ihenetu F. Antimicrobial Activities of Extracts of Tobacco Leaf (Nicotiana tabacum) and Its Grounded Snuff (Utaba) on Candida albicans and Streptococcus pyogenes. PLoS Negl. Trop. Dis. 2019;7:000300. doi: 10.4172/2329-891X.1000300. [DOI] [Google Scholar]
- 8.Fernanda S.A., Amru B.A., Rahmani H.A., Gozan M., Irsyad N.S., Bahar M., Puspita O.S., Zulfa F., Pramono A. Antibacterial Potential of Nicotiana tabacum L. Var Virginia Pyrolysis Extract Against Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, and Pseudomonas aeruginosa. IOP Conf. Ser. Earth Environ. Sci. 2021;755:012013. doi: 10.1088/1755-1315/755/1/012013. [DOI] [Google Scholar]
- 9.Gutiérrez D., Bah M., Garduño M., Mendoza S., Serrano V. Anti-Inflammatory and Antioxidant Activities of Methanol Extracts and Alkaloid Fractions of Four Mexican Medicinal Plants of Solanaceae. Afr. J. Tradit. Complement. Altern. Med. AJTCAM/Afr. Netw. Ethnomed. 2014;11:259–267. doi: 10.4314/ajtcam.v11i3.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ali Alghamdi A. Phytoconstituents Screening and Antimicrobial Activity of the Invasive Species Nicotiana glauca Collected from Al-Baha Region of Saudi Arabia. Saudi J. Biol. Sci. 2021;28:1544–1547. doi: 10.1016/j.sjbs.2020.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Al-Akayleh F., Ali H.H.M., Ghareeb M.M., Al-Remawi M. Therapeutic deep eutectic system of capric acid and menthol: Characterization and pharmaceutical application. J. Drug Deliv. Sci. Technol. 2019;53:101159. doi: 10.1016/j.jddst.2019.101159. [DOI] [Google Scholar]
- 12.Al-Mawla L., Al-Akayleh F., Daadoue S., Mahyoob W., Al-Tameemi B., Al-Remawi M., Adwan S., Agha A.S.A.A. Development, characterization, and ex vivo permeation assessment of diclofenac diethylamine deep eutectic systems across human skin. J. Pharm. Innov. 2023;18:2196–2209. doi: 10.1007/s12247-023-09784-9. [DOI] [Google Scholar]
- 13.Alkhawaja B., Al-Akayleh F., Nasereddin J., Kamran M., Woodman T., Al-Rubaye Z., Qinna N., Al-Remawi M., Olaimat A.R. Structural insights into novel therapeutic deep eutectic systems with capric acid using 1D, 2D NMR and DSC techniques with superior gut permeability. RSC Adv. 2024;14:14793–14806. doi: 10.1039/D4RA01469C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakaweh A., Al-Akayleh F., Al-Remawi M., Abdallah Q., Agha A.S. Deep Eutectic System-Based Liquisolid Nanoparticles as Drug Delivery System of Curcumin for In-Vitro Colon Cancer Cells. J. Pharm. Innov. 2024;19:18. doi: 10.1007/s12247-024-09826-w. [DOI] [Google Scholar]
- 15.Al-Akayleh F., Al-Remawi M., Agha A., Abu-Nameh E. Applications and Risk Assessments of Ionic Liquids in Chemical and Pharmaceutical Domains: An Updated Overview. Jordan J. Chem. (JJC) 2023;18:53–76. [Google Scholar]
- 16.Alkhawaja B., Al-Akayleh F., Nasereddin J., Malek S.A., Alkhawaja N., Kamran M., Al-Rubaye Z., Smairat M., Al-Remawi M., Aburayyan W.S. Levofloxacin–Fatty Acid Systems: Dual Enhancement Through Deep Eutectic Formation and Solubilization for Pharmaceutical Potential and Antibacterial Activity. AAPS PharmSciTech. 2023;24:244. doi: 10.1208/s12249-023-02701-w. [DOI] [PubMed] [Google Scholar]
- 17.Alkhawaja B., Al-Akayleh F., Al-Khateeb A., Nasereddin J., Ghanim B.Y., Bolhuis A., Jaber N., Al-Remawi M., Qinna N.A. Deep eutectic liquids as a topical vehicle for tadalafil: Characterisation and potential wound healing and antimicrobial activity. Molecules. 2023;28:2402. doi: 10.3390/molecules28052402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Luhaibi D.K., Ali H.H.M., Al-Ani I., Shalan N., Al-Akayleh F., Al-Remawi M., Nasereddin J., Qinna N.A., Al-Adham I., Khanfar M. The formulation and evaluation of deep eutectic vehicles for the topical delivery of azelaic acid for acne treatment. Molecules. 2023;28:6927. doi: 10.3390/molecules28196927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Daadoue S., Al-Remawi M., Al-Mawla L., Idkaidek N., Khalid R.M., Al-Akayleh F. Deep eutectic liquid as transdermal delivery vehicle of Risperidone. J. Mol. Liq. 2022;345:117347. doi: 10.1016/j.molliq.2021.117347. [DOI] [Google Scholar]
- 20.Al-Akayleh F., Adwan S., Khanfar M., Idkaidek N., Al-Remawi M. A novel eutectic-based transdermal delivery system for risperidone. AAPS PharmSciTech. 2021;22:4. doi: 10.1208/s12249-020-01844-4. [DOI] [PubMed] [Google Scholar]
- 21.Alkhawaja B., Al-Akayleh F., Al-Rubaye Z., Bustami M., Smairat M.A., Agha A.S., Nasereddin J., Qinna N., Michael A., Watts A.G. Dissecting the stability of Atezolizumab with renewable amino acid-based ionic liquids: Colloidal stability and anticancer activity under thermal stress. Int. J. Biol. Macromol. 2024;270:132208. doi: 10.1016/j.ijbiomac.2024.132208. [DOI] [PubMed] [Google Scholar]
- 22.Airouyuwa J.O., Sivapragasam N., Redha A.A., Maqsood S. Sustainable green extraction of anthocyanins and carotenoids using natural deep eutectic solvents (NaDES): A review of recent developments. Food Chem. 2024;448:139061. doi: 10.1016/j.foodchem.2024.139061. [DOI] [PubMed] [Google Scholar]
- 23.Wang Z., Wang D., Fang J., Song Z., Geng J., Zhao J., Fang Y., Wang C., Li M. Green and efficient extraction of flavonoids from Perilla frutescens (L.) Britt. leaves based on natural deep eutectic solvents: Process optimization, component identification, and biological activity. Food Chem. 2024;452:139508. doi: 10.1016/j.foodchem.2024.139508. [DOI] [PubMed] [Google Scholar]
- 24.Al-Akayleh F., Khalid R.M., Hawash D., Al-Kaissi E., Al-Adham I.S.I., Al-Muhtaseb N., Jaber N., Al-Remawi M., Collier P.J. Antimicrobial potential of natural deep eutectic solvents. Lett. Appl. Microbiol. 2022;75:607–615. doi: 10.1111/lam.13699. [DOI] [PubMed] [Google Scholar]
- 25.Yu J., Xu S., Goksen G., Yi C., Shao P. Chitosan films plasticized with choline-based deep eutectic solvents: UV shielding, antioxidant, and antibacterial properties. Food Hydrocoll. 2023;135:108196. doi: 10.1016/j.foodhyd.2022.108196. [DOI] [Google Scholar]
- 26.Nystedt H.L., Grønlien K.G., Rolfsnes R.R., Winther-Larsen H.C., Økstad O.A.L., Tønnesen H.H. Neutral natural deep eutectic solvents as anti-biofilm agents. Biofilm. 2023;5:100114. doi: 10.1016/j.bioflm.2023.100114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsvetov N., Pasichnik E., Korovkina A., Gosteva A. Extraction of bioactive components from Chamaenerion angustifolium (L.) Scop. with choline chloride and organic acids natural deep eutectic solvents. Molecules. 2022;27:4216. doi: 10.3390/molecules27134216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Edrisi S., Bakhshi H. Separation of Polyphenolic Compounds from Citrus aurantium L. peel by deep eutectic solvents and their recovery using a new DES-based aqueous two-phase system. J. Mol. Liq. 2024;402:124790. doi: 10.1016/j.molliq.2024.124790. [DOI] [Google Scholar]
- 29.Hong J., Deng M., Zhao L. Natural Deep Eutectic Solvent Combined with Ultrasonic Enhancement: A Green Extraction Strategy for Solanesol in Tobacco Leaves. Ind. Crops Prod. 2022;187:115355. doi: 10.1016/j.indcrop.2022.115355. [DOI] [Google Scholar]
- 30.Yu T., Yang L., Shang X., Bian S. Recovery of Cembratrien-Diols from Waste Tobacco (Nicotiana tabacum L.) Flowers by Microwave-Assisted Deep Eutectic Solvent Extraction: Optimization, Separation, and In Vitro Bioactivity. Molecules. 2024;29:1563. doi: 10.3390/molecules29071563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nasr H. Ecological and phytochemical studies on Nicotiana glauca from Egypt. Egypt. J. Exp. Biol. 2014;10:87–95. [Google Scholar]
- 32.Alnsour L., Issa R., Awwad S., Albals D., Al-Momani I. Quantification of Total Phenols and Antioxidants in Coffee Samples of Different Origins and Evaluation of the Effect of Degree of Roasting on Their Levels. Molecules. 2022;27:1591. doi: 10.3390/molecules27051591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ubaydee A.H.N., Issa R., Hajleh M.N.A., Ghanim B.Y., Al-Akayleh F., Qinna N.A. The Effect of Medicago sativa Extract and Light on Skin Hypopigmentation Disorders in C57/BL6 Mice. J. Cosmet. Dermatol. 2022;21:6270–6280. doi: 10.1111/jocd.15233. [DOI] [PubMed] [Google Scholar]
- 34.Al-Bayati M., Issa R., Abu-Samak M., Alnsour L., Awwad S. Phytochemical Analysis and Evaluation of Anti-Hyperlipidaemic Effect for Ethanolic Leaf Extract of Equisetum ramosissimum L.: In Vivo Study Rats’ Models. Pharmacia. 2023;70:557–568. doi: 10.3897/pharmacia.70.e101623. [DOI] [Google Scholar]
- 35.Kheawfu K., Kaewpinta A., Chanmahasathien W., Rachtanapun P., Jantrawut P. Extraction of Nicotine from Tobacco Leaves and Development of Fast Dissolving Nicotine Extract Film. Membranes. 2021;11:403. doi: 10.3390/membranes11060403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dai Y., Van Spronsen J., Witkamp G.J., Verpoorte R., Choi Y.H. Ionic liquids and deep eutectic solvents in natural products research: Mixtures of solids as extraction solvents. J. Nat. Prod. 2013;76:2162–2173. doi: 10.1021/np400051w. [DOI] [PubMed] [Google Scholar]
- 37.Chanioti S., Tzia C. Extraction of phenolic compounds from olive pomace by using natural deep eutectic solvents and innovative extraction techniques. Innov. Food Sci. Emerg. Technol. 2018;48:228–239. doi: 10.1016/j.ifset.2018.07.001. [DOI] [Google Scholar]
- 38.Zannou O., Koca I., Aldawoud T.M., Galanakis C.M. Recovery and stabilization of anthocyanins and phenolic antioxidants of roselle (Hibiscus sabdariffa L.) with hydrophilic deep eutectic solvents. Molecules. 2020;25:3715. doi: 10.3390/molecules25163715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Alsaud N., Shahbaz K., Farid M. Application of deep eutectic solvents in the extraction of polyphenolic antioxidants from New Zealand Manuka leaves (Leptospermum scoparium): Optimization and antioxidant activity. J. Mol. Liq. 2021;337:116385. doi: 10.1016/j.molliq.2021.116385. [DOI] [Google Scholar]
- 40.Pavić V., Flačer D., Jakovljević M., Molnar M., Jokić S. Assessment of total phenolic content, in vitro antioxidant and antibacterial activity of Ruta graveolens L. extracts obtained by choline chloride based natural deep eutectic solvents. Plants. 2019;8:69. doi: 10.3390/plants8030069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bakirtzi C., Triantafyllidou K., Makris D.P. Novel lactic acid-based natural deep eutectic solvents: Efficiency in the ultrasound-assisted extraction of antioxidant polyphenols from common native Greek medicinal plants. J. Appl. Res. Med. Aromat. Plants. 2016;3:120–127. doi: 10.1016/j.jarmap.2016.03.003. [DOI] [Google Scholar]
- 42.Alsenidi M., Moustafa M. Variation in Chemicals and Antimicrobial Activities of Nicotiana glauca Graham. Bangladesh J. Bot. 2023;52:403–410. doi: 10.3329/bjb.v52i2.67058. [DOI] [Google Scholar]
- 43.Tayoub G., Sulaiman H., Alorfi M. Determination of nicotine levels in the leaves of some Nicotiana tabacum varieties cultivated in Syria. Herba Pol. 2015;61:23–30. doi: 10.1515/hepo-2015-0028. [DOI] [Google Scholar]
- 44.Tantullavetch Y., Chutrtong W., Nimkulrat S. The Development and Production of Nicotine Gum for Use in Smoking Cessation Therapy (the Second Period) Srinakharinwirot University; Bangkok, Thailand: 2007. [Google Scholar]
- 45.Banožić M., Banjari I., Flanjak I., Paštar M., Vladić J., Jokić S. Optimization of MAE for the Separation of Nicotine and Phenolics from Tobacco Waste by Using the Response Surface Methodology Approach. Molecules. 2021;26:4363. doi: 10.3390/molecules26144363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Furer V., Hersch M., Silvetzki N., Breuer G.S., Zevin S. Nicotiana glauca (Tree Tobacco) Intoxication—Two Cases in One Family. J. Med. Toxicol. 2011;7:47–51. doi: 10.1007/s13181-010-0102-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Schep L.J., Slaughter R.J., Beasley D.M.G. Nicotinic Plant Poisoning. Clin. Toxicol. 2009;47:771–781. doi: 10.1080/15563650903252186. [DOI] [PubMed] [Google Scholar]
- 48.Janakat S., Al-Merie H. Evaluation of Hepatoprotective Effect of Pistacia lentiscus, Phillyrea latifolia and Nicotiana glauca. J. Ethnopharmacol. 2002;83:135–138. doi: 10.1016/S0378-8741(02)00241-6. [DOI] [PubMed] [Google Scholar]
- 49.Rodgman A., Perfetti T. The Chemical Components of Tobacco and Tobacco Smoke. 2nd ed. CRC Press; Boca Raton, FL, USA: 2013. [Google Scholar]
- 50.Wang H., Zhao M., Yang B., Jiang Y., Rao G. Identification of Polyphenols in Tobacco Leaf and Their Antioxidant and Antimicrobial Activities. Food Chem. 2008;107:1399–1406. doi: 10.1016/j.foodchem.2007.09.068. [DOI] [Google Scholar]
- 51.Trifa W., Akkal S., Lefahal M., Benmekhebi L., Khennouf S. Preliminary Screening of Extracts for Determination of Antioxidant Activity by Different Methods. Curr. Issues Pharm. Med. Sci. 2020;33:32–37. doi: 10.2478/cipms-2020-0007. [DOI] [Google Scholar]
- 52.Özdenefe M.S., Takcı A.M., Kayış F.B. Antibacterial, Antioxidant, Antidiabetic Potentials and Chemical Composition of Nicotiana glauca Graham Leaf Extract. J. Anatol. Environ. Anim. Sci. 2023;8:700–706. doi: 10.35229/jaes.1325678. [DOI] [Google Scholar]
- 53.Puripattanavong J., Songkram C., Lomlim L., Amnuaikit T. Development of Concentrated Emulsion Containing Nicotiana tabacum Extract for Use as Pesticide. J. Appl. Pharm. Sci. 2013;3:16–21. doi: 10.7324/JAPS.2013.31104. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are contained within the article.