Abstract
Background/Aim
The aim of this study was to analyze rs2234693 and rs9340799 polymorphisms of the ESR1 gene in the context of breast cancer risk in Polish patients.
Materials and Methods
The study involved a group of 117 patients with breast cancer and 106 controls. The analyses were carried out using the polymerase chain reaction – restriction fragments length polymorphism technique.
Results
The presence of the CC genotype in rs2234693 more than doubled the risk of breast cancer (p=0.04), whereas the presence of the TT genotype in rs2234693 significantly reduced the risk of developing this type of cancer (p=0.0002). The presence of the GG genotype in rs9340799 more than doubled the risk of breast cancer (p=0.04), which was confirmed by the analysis of the recessive model (p=0.04).
Conclusion
The polymorphisms rs2234693 and rs9340799 of the ESR1 gene may be associated with the risk of breast cancer among Polish women.
Keywords: ESR1, polymorphism, rs2234693, rs9340799, breast cancer
Hormone-dependent cancer of the breast is the most common cancer in women (1-3). Hormones are one of the main factors that regulate the function of individual organs of our body and sex hormones, including estrogens, can act as carcinogens (4,5). They can cause chromosome segregation errors and structural chromosome changes. Estrogens can stimulate the uncontrolled proliferation of mutated breast cells, damage DNA and induce cancer (6,7). As a result of binding of the steroid-receptor complex to DNA in the cell nucleus, estrogens have a direct effect on target cells (8). In this way, estrogens regulate gene expression and can cause proto-oncogenes to be converted into oncogenes (9). Cell proliferation is stimulated by estrogens by enhancing DNA synthesis in stromal and glandular tissues (10). Estrogen metabolism leads to the formation of several metabolites that, when converted into semiquinones and quinones, generate reactive oxygen species and directly damage DNA (11).
Literature data indicate an association of endogenous and exogenous circulating estrogen with the etiology of breast cancer. Premenopausal women have an increased risk of this cancer (12). Estrogen receptors (ERs) mediate the effects of estrogen on breast cells. Estrogen binding to its receptor promotes the growth and differentiation of normal breast cells and can lead to breast cancer (13-15). A recent research indicated that ER mediates the suppression of the expression of Capillary morphogenesis gene 2 (CMG2), which is involved in breast cancer progression, in relevant cancer cells. Studies have shown that CMG2 expression is inversely correlated with ER status in breast cancer (16).
A distinction is made between two types of ER: ERα and ERβ, encoded by two different genes, ESR1 and ESR2, respectively. ERα shows a higher level of expression in breast tissue compared to ERβ, which is why it is often associated with the development of breast cancer (17).
The aim of the presented work was to examine whether polymorphisms in ESR1, which is highly polymorphic, affect breast cancer incidence. Of the many polymorphisms in ESR1, the two best studied are rs2234693 (also known as PvuII or 397T>C) and rs9340799 (also known as XbaI or 351A>G). Both polymorphisms are located in intron 1, 1,397 bp and 351 bp, respectively, upstream of exon 2 of the gene. These polymorphisms may be associated with breast cancer and endometrial cancer (18-20). Studies indicate that ESR1 amplification may be an early event in the development of endometrial cancer (21). Amplification of ESR1 is independent of known clinical-pathological factors associated with poor prognosis and expression of certain proteins (PTEN, p53, HER2, MLH1, and ARID1A) (21). However, many studies have found conflicting results as to the association of these two polymorphisms with breast cancer susceptibility (22-25). In order to explain these discrepancies, a meta-analysis of the polymorphism rs2234693 was performed in 2018. In that study, rs2234693 was shown to be associated with reduced susceptibility to breast cancer (26). As for rs9340799, Zhang et al. found no significant association between the polymorphism and breast cancer susceptibility (27). The polymorphisms rs2234693 and rs9340799 in the ESR1 gene were not associated with breast cancer susceptibility in the Mexican population (27). However, the CTAG and CCGG haplogenotypes of rs2234693 and rs9340799 may significantly contribute to breast cancer risk susceptibility of patients who have had miscarriages or had significant tobacco consumption (27,28).
The aim of our research was to determine: 1) the effect of two single nucleotide polymorphisms (SNPs - rs2234693, rs9340799) of the ESR1 gene on the risk of breast cancer in Polish women; 2) the effect of ESR1 gene haplotypes in relation to the risk of breast cancer in women; 3) the association of the rs2234693 and rs9340799 polymorphisms of the ESR1 gene with clinical-pathological features.
Materials and Methods
Patients. We used 117 venous blood samples from women diagnosed with breast cancer and 106 venous blood samples from women who were diagnosed with breast cancer. The samples were obtained from the Department of Surgical Oncology and Breast Diseases, Polish Mother’s Memorial Hospital Research Institute, Lodz, Poland. The patients were operated on between 2015 and 2018.None of the patients had received prior neoadjuvant therapy, chemotherapy, or radiotherapy. The characteristics of the patients are presented in Table I. Approval of the Bioethics Committee at the Polish Mothers Memorial Hospital Research Institute in Lodz was obtained (Nr. 20/2015).
Table I. Characteristics of patients.
TNM: Tumor, nodules and metastases; ER: estrogen receptor; PR: progesterone receptor; HER: human epidermal growth factor receptor 2.
Application of histopathology scanner (3d Histech). Histological examination was performed by experienced pathologists using a digital slide scanner and slide review software (Case Viewer 2.3, 3D Histech, Budapest, Hungary). Histological sections were scanned with a panoramic scanner (3D Histech) and digital images were obtained. It is considered that the scanned digital form of the histological specimen, which can be evaluated using automated programmatic analysis modules, is an independent and objective method of histopathological examination. The study received funding from the Digital Poland Operational Programme MDB-BANK (grant no. POPC.02.03.01-00-0091/19).
DNA isolation from whole blood. DNA was isolated from whole blood using the GenElute Blood Genomic DNA Kit (Sigma-Aldrich, Steinheim am Albuch, Germany) according to the manufacturer’s recommendations. DNA was stored at -20˚C. The purity of the obtained DNA preparations was determined spectrophotometrically measuring the absorbance of each sample twice at wavelengths of 260 nm and 280 nm. The accepted criterion for DNA purity was a A260/A280 value within the range of 1.8-2.0. The concentration of DNA was determined spectrophotometrically based on absorbance values measured at a wavelength of 260 nm considering that 1OD=50 μg DNA/ml.
Analysis of selected polymorphisms of the ESR1 gene. On the basis of data available in the SNP database NCBI (National Center for Biotechnology Information, http://www.ncbi.nlm.nih.gov/snp), two single nucleotide polymorphisms located in the ESR1 gene were selected. The SNPs selected are NC_000006.11:g.152163335T>C (rs2234693) and NC_000006.11:g.152163381A>G (rs9340799). The analysis of the incidence of the above-mentioned polymorphisms in the group of patients with breast cancer as well as in the control group was carried out using PCR reaction combined with restriction fragment analysis, PCR-RFLP (Polymerase Chain Reaction – Restriction Fragments Length Polymorphism). Selected fragments of the ESR1 gene were amplified and then digested using restriction enzymes - rs2234693 (PvuII); rs9340799 (XbaI). The nucleotide sequence of the amplified fragments and the characteristics of the primers used are presented in Table II.
Table II. Nucleotide sequence of the amplified area and sequences of the primers used to carry out the amplification reaction of the ESR1 gene fragment (27).
The PCR reaction was carried out in a final volume of 10 μl. The reaction mixture contained approximately 50 ng of genomic DNA, 1 μl of 5μM solution of each primer, 1 μl of GeneAmp 10× PCR Buffer with MgCl2 (Applied Biosystems, Foster City, CA, USA), 0.2 μl of AmpliTaq Gold® polymerase (5U/μl) (Applied Biosystems), 1 μl of 10 μM dNTPs, and water. Initial denaturation was carried out for 5 min at 95˚C, then the selected fragment of the tested gene was amplified during 40 cycles with the following parameters: 30 s at 95˚C, 1 min at 62˚C and 1 min at 72˚C.
The products of DNA amplification were digested for 16 h using commercially available restriction enzymes - PvuII and XbaI (New England Biolabs, Ipswich, MA, USA) (29). The reaction products were then electrophoresed in 2% agarose gel, stained with ethidium bromide, and visualized under ultraviolet light.
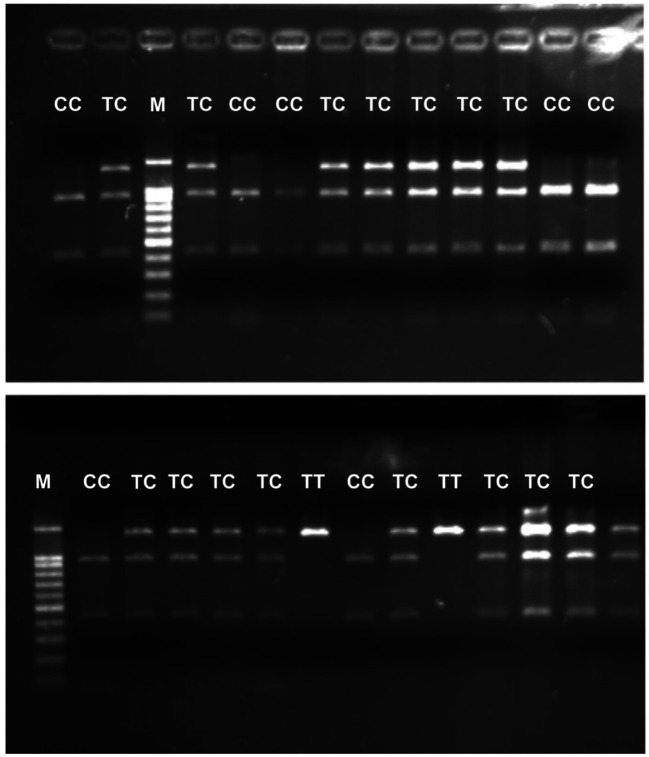
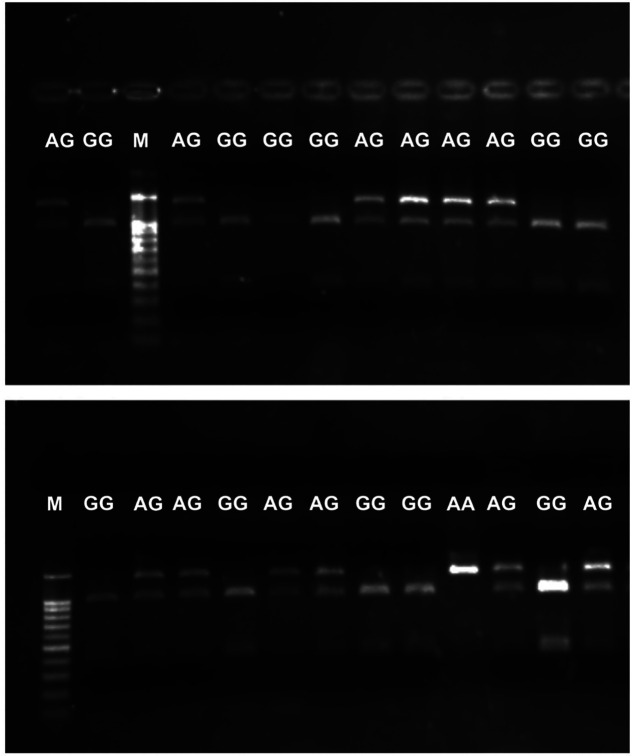
The size of the products for the analysis of the PvuII polymorphism (rs2234693, T/C) and XbaI polymorphism (rs9340799, A/G) is summarized in Table III. The selected PCR products were additionally sequenced using the Sanger method at the Department of Cytobiochemistry (University of Lodz, Poland) to corroborate the genotypes previously estimated using the PCR-RFLP method. An example of the electrophoretic separation of PCR-RFLP reaction products is shown in Figure 1 and Figure 2.
Table III. The size of the products obtained in the analysis of the PvuII polymorphism (rs2234693, T/C) and in the analysis of the XbaI polymorphism (rs9340799, A/G).
Figure 1. An exemplary picture of the electrophoretic separation of the PCR-RFLP reaction products for the Pull polymorphism (rs2234693, T/C) in the ESR1. M: DNA fragment size standard (DNA Marker 100-1000 ladder, Sigma-Aldrich, Burlington, MA, USA); T: normal allele; C: mutant allele.
Figure 2. An exemplary picture of the electrophoretic separation of the PCR-RFLP reaction products for the Pull polymorphism (rs2234693, T/C) in the ESR1. M: DNA fragment size standard (DNA Marker 100-1000 ladder, Sigma-Aldrich, Burlington, MA, USA); T: normal allele; C: mutant allele.
Statistical analysis. Statistical analysis was performed with the use of using STATISTICA 11 (StatSoft, Poznań, Poland), Chaplin 1.2 (genetics.emory.edu) and THESIAS (www.genecanvas.org). The effect of ESR1 gene polymorphisms on breast cancer risk was assessed using odds ratio (OR) and a 95% confidence interval (CI) using a logistic regression model. In this case, OR allows assessment of how many times the risk of disease increases/decreases with an increase/decrease by one unit of the variable treated as a risk factor.
In order to examine whether the Hardy-Weinberg law of equilibrium is observed in the studied populations, the calculation program on the website www.ihg.gfs.de (Institute of Human Genetics, Technical University Munich and Helmholtz Center Munich) was used. To investigate the effect of ERS1 gene haplotypes on cancer risk, Chaplin 1.2 and THESIAS were used.
All statistical tests were performed at a significance level of α=0.05. The assessment of the consistency of the distribution of the values of the studied variables with the normal distribution was carried out using the Shapiro-Wilk test. In order to verify the hypothesis of age significance in the study and control groups, the χ2 analysis was used.
Results
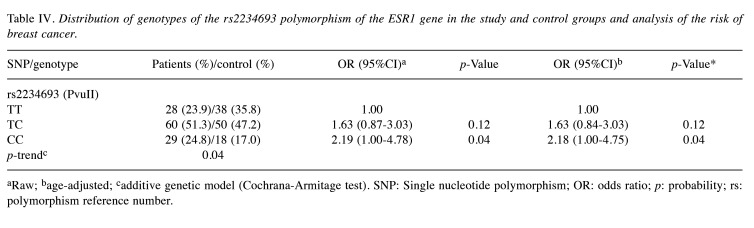
The obtained results indicate that the presence of the CC genotype in rs2234693 more than doubles the risk of breast cancer (OR=2.19; 95%CI=1.00-4.78); p=0.04 (Table IV), while the presence of the TT genotype in rs2234693 significantly reduces the risk of developing this type of cancer (OR=0.31; 95%CI=0.16-0.60); p=0.0002 (Table V). The age of the women had no effect on the prevalence of the individual genotypes or the associated risk of disease. The results of the analysis are also presented in the form of adjusted odds ratios.
Table IV. Distribution of genotypes of the rs2234693 polymorphism of the ESR1 gene in the study and control groups and analysis of the risk of breast cancer.
aRaw; bage-adjusted; cadditive genetic model (Cochrana-Armitage test). SNP: Single nucleotide polymorphism; OR: odds ratio; p: probability; rs: polymorphism reference number.
Table V. Distribution of genotypes of the rs2234693 polymorphism of the ESR1 gene in the study and control groups – dominant and recessive model.
aRaw; bage-adjusted; cdominant genetic model; drecessive genetic model. SNP: Single nucleotide polymorphism; OR: odds ratio; p: probability; rs: polymorphism reference number.
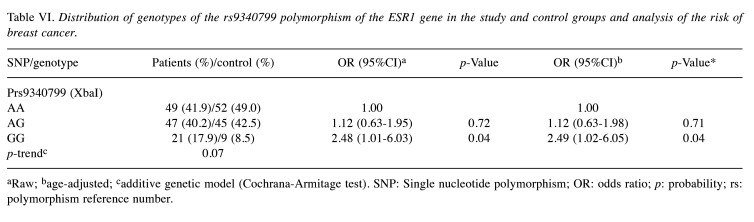
In the case of the second polymorphism, rs9340799, it was found, analogously to the previous studies, that the presence of the GG genotype more than doubled the risk of breast cancer (OR=2.48; 95%CI=1.01-6.03) p=0.04) (Table VI), which was confirmed by the analysis of the recessive model (OR=2.36; 95%CI=1.02-5.41; p=0.049) (Table VII).
Table VI. Distribution of genotypes of the rs9340799 polymorphism of the ESR1 gene in the study and control groups and analysis of the risk of breast cancer.
aRaw; bage-adjusted; cadditive genetic model (Cochrana-Armitage test). SNP: Single nucleotide polymorphism; OR: odds ratio; p: probability; rs: polymorphism reference number.
Table VII. Distribution of genotypes of the rs9340799 polymorphism of the ESR1 gene in the study and control groups – dominant and recessive model.
aRaw; bage-adjusted; cdominant genetic model; drecessive genetic model. SNP: Single nucleotide polymorphism; OR: odds ratio; p: probability; rs: polymorphism reference number.
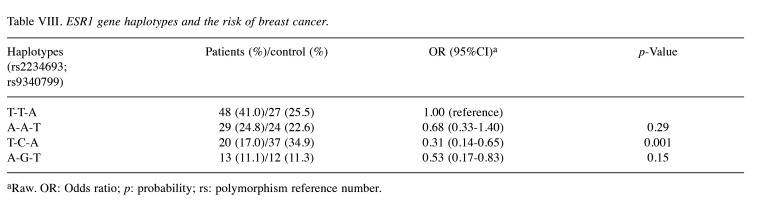
Haplotypes analysis. The analysis of ESR1 gene haplotypes in relation to the rs2234693 and rs9340799 polymorphisms showed that among the examined patients, only the occurrence of the T-C-A haplogenotypes was associated with a significant reduction in the risk of cancer in women (OR=0.31; 95%CI=0.14-0.65; p=0.001). Detailed results of the analyses are summarized in Table VIII.
Table VIII. ESR1 gene haplotypes and the risk of breast cancer.
aRaw. OR: Odds ratio; p: probability; rs: polymorphism reference number.
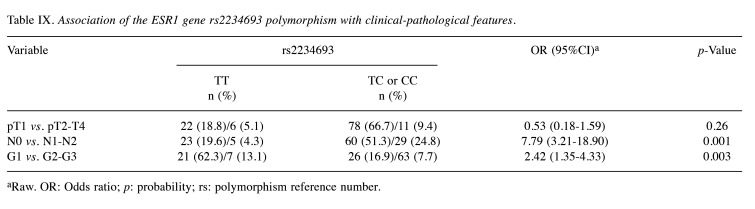
Analysis of the association of the rs2234693 and rs9340799 polymorphisms of the ESR1 gene with clinical-pathological features. It was found that the presence of the TC or CC genotype (presence of the C allele) in rs2234693 was significantly associated with a more than seven-fold increase in the tendency of breast cancers to metastasize (OR=7.79; 95%CI=3.21-18.90; p=0.001) and with a higher degree of anaplasia (OR=2.42; 95%CI=1.35-4.33; p=0.003) (Table IX).
Table IX. Association of the ESR1 gene rs2234693 polymorphism with clinical-pathological features.
aRaw. OR: Odds ratio; p: probability; rs: polymorphism reference number.
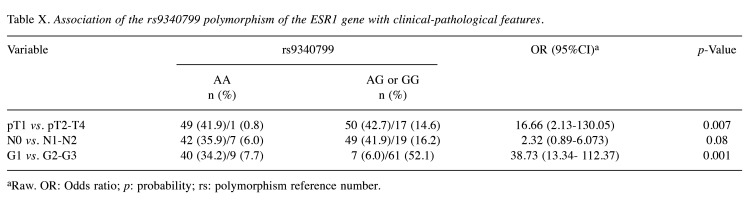
In the case of rs9340799 polymorphism, the presence of AG or GG genotype (presence of the G allele) was associated with a larger tumor size (OR=16.66; 95%CI=2.13-130.05; p=0.007) and a higher degree of anaplasia (OR=38.73; 95%CI=13.34-112.37; p=0.001) (Table X).
Table X. Association of the rs9340799 polymorphism of the ESR1 gene with clinical-pathological features.
aRaw. OR: Odds ratio; p: probability; rs: polymorphism reference number.
Discussion
Genetic polymorphisms in the estrogen receptor-α (ESR1) gene are involved in alterations in receptor expression and function, evolving as important determinants of breast cancer susceptibility. The associations of the single nucleotide polymorphisms (SNPs) rs2234693 and rs9340799 in the ESR1 gene with breast cancer have been studied. However, the data and conclusions are inconsistent and controversial. The rs2234693 polymorphism is found in the first base of intron 397 bp upstream exon 2, in which cytosine is replaced by thymine, and this change is identified by the PvuII endonuclease (30,31).
It is known that localization of a polymorphism in intron regions may lead to modification of splicing in mRNA transcripts. The consequence of these changes is a change in the function of genes (31). There are differences in the incidence of this polymorphisms in different populations. It has been shown that the T or C allele may be a protective or risk factor for breast cancer (32-34,27).
The C allele is considered to be an ancestral allele that is present at a lower frequency in the general population. Studies in the Mexican population have shown a different frequency of the C allele compared to other populations. The first studies were conducted in postmenopausal women (age range=46-80 years) and the C allele was found to be present at a frequency of 30.9% (33), while in another study conducted in women under 45 years of age with C allele was present in 25.8% (34).
Recent studies in the Mexican population regarding these polymorphisms have shown that they are not associated with susceptibility to breast cancer. However, the CTAG and CCGG haplogenotypes of the rs2234693 and rs9340799 polymorphisms may significantly contribute to breast cancer risk susceptibility in women who have had a miscarriage and those who have consumed tobacco (28).
Numerous meta-analyses indicate that women who carry the T allele have a low risk of developing breast cancer. In Caucasian populations, the risk of breast cancer is higher in carriers of the TT genotype, whereas in Asians the CC genotype (32,35).
Kornats et al. found no association between ESR1 gene variants and the risk of developing mastopathy in infertile women, although heterozygous ESR1 gene variants increased the “protective” effect of FSHR (estrogen-stimulating hormone receptor) gene variants and reduced the risk of mastopathy (36).
The ESR1 and ESR2 gene variants have been studied in triple-negative breast cancer (TNBC), where unique genetic variants ESR1 and ESR2 have been shown to be associated with the risk of TNBC. This suggests a possible diagnostic and prognostic role of these polymorphisms in TNBC (37).
A total of 25 case-control studies (meta-analysis) were analyzed to investigate the association between the ESR1 gene polymorphisms rs2234693, rs9340799, and rs1801132 and breast cancer risk in different populations. Three different comparative models were used to assess the association – the dominant model, the recessive model, and the homozygote comparative model. The results showed that regarding the rs2234693 polymorphism, individuals with the TT+TC or TT genotype were at a higher risk of developing breast cancer than those with the CC genotype. However, in the case of the rs9340799 and rs1801132 polymorphisms, none of the 3 models found significant associations.
Analysis of polymorphisms was also performed according to ethnicity (white or Asian) and source of control (hospital or population). An assessment of ethnic subgroups showed that the TT rs2234693 genotype, compared to the CC genotype, conferred a higher risk of breast cancer only in Asians, but not in white populations. In the source-stratified subgroup analysis, individuals with the TT+TC genotype were found to be at higher risk of breast cancer than those with the CC genotype in the hospital subgroup (27).
Polymorphisms in genes involved in estrogen synthesis (UGT2B17 - UGT2B17*2, CYP19A1 rs10046/rs4646 and ESR1 rs2077647/rs2234693/rs9340799) have been correlated with breast cancer risk, prognosis, and response to treatment (38). The studies involved women with ER-positive postmenopausal breast cancer. The UGT2B17*2 polymorphism was associated with higher levels of 17-hydroxyexemestane and a better prognosis compared to homozygous UGT2B17. The rs10046 A and rs4646 C alleles in CYP19A1 were associated with higher estrogen levels. The presence of the rs10046 G and rs4646 A alleles was associated with low estrogen levels and a better prognosis compared to homozygous wild type (median follow-up of 7 years). Thus, postmenopausal hormone-responsive breast cancer is influenced by UGT2B17 and CYP19A1. Carriers of UGT2B17*2 and CYP19A1 variants with low estrogen levels may have a better prognosis. Polymorphisms may be important in optimizing hormone therapy.
Three polymorphisms inESR1, r s2881766, rs9383951, rs9340799 and one in ESR2, rs3020449, were studied in 459 patients and 549 healthy controls (38). The rs2881766 polymorphism was associated with a reduced risk of breast cancer Whereas the rs3020449 was associated with an increased risk of this cancer. The other two polymorphisms were not associated with susceptibility to breast cancer. In addition, rs2881766 was correlated with lymph node metastases and ER expression, and rs3020449 was associated with tumor size, histological grade, and ER expression. The authors of the study emphasize that these findings require further validation in a large population (39).
A recent meta-analysis of 23 studies involving 34,721 patients, found no significant association between the rs9340799 ESR1 polymorphism and breast cancer susceptibility (40). Subgroup analyses by ethnicity, menopausal status, and study quality also found no statistically significant association (40).
In our study, women who were carriers of the CC rs2234693 and GG rs9340799 genotypes had a higher risk of developing breast cancer. The TT rs2234693 genotype had a protective role. The T-C-A haplogenotype of the rs2234693 and rs9340799 polymorphisms in the ESR1 gene in relation to was associated with a lower risk of breast cancer. Our results differ from the previously cited literature data and shed new light on this topic in the Polish population. We would like to emphasize that this is the first study conducted in Poland. Of course, we are aware of the limitations of our study. First of all, the study population was small (117 patients, 106 controls). The cited research articles are few and the results of the research are contradictory. Further research is needed to confirm or reject the importance of ESR1 polymorphisms in breast cancer.
Conclusion
The CC and GG genotypes of rs2234693 and rs9340799, respectively, of the ESR1 gene increase the risk of breast cancer in women: 1) The TT genotype of the rs2234693 polymorphism in the ESR1 gene lowers the risk of breast cancer in women; 2) The occurrence of the T-C-A haplotype regarding the rs2234693 and rs9340799 polymorphisms of the ESR1 gene is associated with a lower risk of breast cancer in women; 3) In the case of ESR1 gene polymorphisms rs2234693 and rs9340799, the presence of the mutant alleles C and G, respectively, is associated with a higher degree of anaplasia in female breast cancers.
Funding
This work was supported by the Institute of Polish Mother’s Memorial Hospital, Lodz, Poland from the Statutory Development. The study was funded by the Operational Program Digital Poland MDB-MEDICAL DATA BANK (Grant no. POPC.02.03.01-00-0091/19).
Conflicts of Interest
The Authors declare no conflicts of interest in relation to this study.
Authors’ Contributions
Research concept: B.S., AZN.; MB.; EF. and H.R.; Execution of the experiments: EF; patient collection: AZN.; analysis of data: B.S., H.L., D.S.; contribution to the writing of manuscript: B.S., S.L. All Authors have read and agreed to the published version of the manuscript.
Acknowledgements
The Authors would like to thank Filip Jabłoński MD for collecting the blood samples for the study.
References
- 1.Emons G. Hormone-dependent cancers: molecular mechanisms and therapeutical implications. Cells. 2022;12(1):110. doi: 10.3390/cells12010110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boszkiewicz K, Sawicka E, Piwowar A. The impact of xenoestrogens on effectiveness of treatment for hormone-dependent breast cancer – current state of knowledge and perspectives for research. Ann Agric Environ Med. 2020;27(4):526–534. doi: 10.26444/aaem/124165. [DOI] [PubMed] [Google Scholar]
- 3.Chlebowski RT, Manson JE. Menopausal hormone therapy and breast cancer. Cancer J. 2022;28(3):169–175. doi: 10.1097/PPO.0000000000000601. [DOI] [PubMed] [Google Scholar]
- 4.Sapkota Y. Germline DNA variations in breast cancer predisposition and prognosis: a systematic review of the literature. Cytogenet Genome Res. 2014;144(2):77–91. doi: 10.1159/000369045. [DOI] [PubMed] [Google Scholar]
- 5.Smith RA, Andrews KS, Brooks D, Fedewa SA, Manassaram-Baptiste D, Saslow D, Wender RC. Cancer screening in the United States, 2019: A review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin. 2019;69(3):184–210. doi: 10.3322/caac.21557. [DOI] [PubMed] [Google Scholar]
- 6.Jefcoate CR, Jefcoate CR, Liehr JG, Santen RJ, Sutter TR, Yager JD, Yue W, Santner SJ, Tekmal R, Demers L, Pauley R, Naftolin F, Mor G, Berstein L. Tissue-specific synthesis and oxidative metabolism of estrogens. J Natl Cancer Inst Monogr. 2000;27:95–112. doi: 10.1093/oxfordjournals.jncimonographs.a024248. [DOI] [PubMed] [Google Scholar]
- 7.Zhu BT, Conney AH. Functional role of estrogen metabolism in target cells: review and perspectives. Carcinogenesis. 1998;19(1):1–27. doi: 10.1093/carcin/19.1.1. [DOI] [PubMed] [Google Scholar]
- 8.Spelsberg TC, Rories C, Rejman JJ, Goldberger A, Fink K, Lau CK, Colvard DS, Wiseman G. Steroid action on gene expression: possible roles of regulatory genes and nuclear acceptor sites. Biol Reprod. 1989;40(1):54–69. doi: 10.1095/biolreprod40.1.54. [DOI] [PubMed] [Google Scholar]
- 9.Hua H, Zhang H, Kong Q, Jiang Y. Mechanisms for estrogen receptor expression in human cancer. Exp Hematol Oncol. 2018;7:24. doi: 10.1186/s40164-018-0116-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Whitehead MI, Fraser D. The effects of estrogens and progestogens on the endometrium. Modern approach to treatment. Obstet Gynecol Clin North Am. 1987;14:299–320. [PubMed] [Google Scholar]
- 11.Maiti S, Nazmeen A. Impaired redox regulation of estrogen metabolizing proteins is important determinant of human breast cancers. Cancer Cell Int. 2019;19:111. doi: 10.1186/s12935-019-0826-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Key TJ, Appleby P, Barnes I, Reeves G, Endogenous Hormones and Breast Cancer Collaborative Group Endogenous sex hormones and breast cancer in postmenopausal women: Reanalysis of nine prospective studies. J Natl Cancer Inst. 2002;94:606–616. doi: 10.1093/jnci/94.8.606. [DOI] [PubMed] [Google Scholar]
- 13.Makowski M, Połać I, Pertyński T. Oestrogens and breast cancer. Menopause Rev. 2007;3:150–154. [Google Scholar]
- 14.Soares Júnior JM, Mota BS, Nobrega GB, Filassi JR, Sorpreso ICE, Baracat EC. Breast cancer survivals and hormone therapy: estrogen and melatonin. Rev Assoc Med Bras (1992) 2023;69(10):e6910EDI. doi: 10.1590/1806-9282.6910EDI. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sund M, Garmo H, Andersson A, Margolin S, Ahlgren J, Valachis A. Estrogen therapy after breast cancer diagnosis and breast cancer mortality risk. Breast Cancer Res Treat. 2023;198(2):361–368. doi: 10.1007/s10549-023-06871-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fang Z, Killick C, Halfpenny C, Frewer N, Frewer KA, Ruge F, Jiang WG, Ye L. Sex hormone-regulated CMG2 is involved in breast and prostate cancer progression. Cancer Genomics Proteomics. 2022;19(6):703–710. doi: 10.21873/cgp.20353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Balfe P, McCann A, McGoldrick A, McAllister K, Kennedy M, Dervan P, Kerin MJ. Estrogen receptor α and β profiling in human breast cancer. Eur J Surg Oncol. 2004;30(5):469–474. doi: 10.1016/j.ejso.2004.02.010. [DOI] [PubMed] [Google Scholar]
- 18.Slattery ML, Sweeney C, Herrick J, Wolff R, Baumgartner K, Giuliano A, Byers T. ESR1, AR, body size, and breast cancer risk in Hispanic and non-Hispanic white women living in the Southwestern United States. Breast Cancer Res Treat. 2007;105(3):327–335. doi: 10.1007/s10549-006-9453-z. [DOI] [PubMed] [Google Scholar]
- 19.Dunning AM, Healey CS, Pharoah PD, Teare MD, Ponder BA. A systematic review of genetic polymorphisms and breast cancer risk-PubMed. Cancer Epidemiol Biomark Prev. 1999;8:843–854. [PubMed] [Google Scholar]
- 20.Weiderpass E, Persson I, Melhus H, Wedrén S, Kindmark A, Baron JA. Estrogen receptor gene polymorphisms and endometrial cancer risk. Carcinogenesis. 2000;21(4):623–627. doi: 10.1093/carcin/21.4.623. [DOI] [PubMed] [Google Scholar]
- 21.Rahman MT, Nakayama K, Rahman M, Ishikawa M, Katagiri H, Katagiri A, Ishibashi T, Sato E, Iida K, Ishikawa N, Nakayama N, Miyazaki K. ESR1 gene amplification in endometrial carcinomas: a clinicopathological analysis. Anticancer Res. 2013;33:3775–3781. [PubMed] [Google Scholar]
- 22.González-Zuloeta Ladd AM, Vásquez AA, Rivadeneira F, Siemes C, Hofman A, Stricker BH, Pols HA, Uitterlinden AG, van Duijn CM. Estrogen receptor alpha polymorphisms and postmenopausal breast cancer risk. Breast Cancer Res Treat. 2008;107(3):415–419. doi: 10.1007/s10549-007-9562-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Javed S, Ali M, Sadia S, Aslam MA, Masood AI, Shaikh RS, Sayyed AH. Combined effect of menopause age and genotype on occurrence of breast cancer risk in Pakistani population. Maturitas. 2011;69(4):377–382. doi: 10.1016/j.maturitas.2011.05.008. [DOI] [PubMed] [Google Scholar]
- 24.Onland-Moret NC, Van Gils CH, Roest M, Grobbee DE, Peeters PHM. The estrogen receptor α gene and breast cancer risk (The Netherlands) Cancer Causes Control. 2005;16(10):1195–1202. doi: 10.1007/s10552-005-0307-5. [DOI] [PubMed] [Google Scholar]
- 25.Wedrén S, Lovmar L, Humphreys K, Magnusson C, Melhus H, Syvänen AC, Kindmark A, Landegren U, Fermér ML, Stiger F, Persson I, Baron J, Weiderpass E. Oestrogen receptor alpha gene haplotype and postmenopausal breast cancer risk: a case control study. Breast Cancer Res. 2004;6(4):R437–R449. doi: 10.1186/bcr811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang ZL, Zhang CZ, Li Y, Zhao ZH, Yang SE. Association between ERα gene Pvu II polymorphism and breast cancer susceptibility: A meta-analysis. Medicine (Baltimore) 2018;97(17):e0317. doi: 10.1097/MD.0000000000010317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang Y, Zhang M, Yuan X, Zhang Z, Zhang P, Chao H, Jiang L, Jiang J. Association between ESR1 PvuII, XbaI, and P325P polymorphisms and breast cancer susceptibility: a meta-analysis. Med Sci Monit. 2015;21:2986–2996. doi: 10.12659/MSM.894010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Carrillo-Moreno DI, Eduardo Figuera L, Zuniga González GM, Puebla Perez AM, Jesus Moran Mendoza A, Gallegos Arreola MP. Association of rs2234693 and rs9340799 polymorphisms of ESR1 gene in breast cancer of Mexican population. J BUON. 2019;24:1927–1933. [PubMed] [Google Scholar]
- 29.Mohammadi F, Pourahmadi M, Mosalanejad M, Jamali H, Ghobadifar MA, Erfanian S. Association of estrogen receptor α genes PvuII and XbaI polymorphisms with type 2 diabetes mellitus in the inpatient population of a hospital in southern Iran. Diabetes Metab J. 2013;37(4):270–277. doi: 10.4093/dmj.2013.37.4.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sundermann EE, Maki PM, Bishop JR. A review of estrogen receptor alpha gene (ESR1) polymorphisms, mood, and cognition. Menopause. 2010;17(4):874–886. doi: 10.1097/gme.0b013e3181df4a19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Su X, Xu X, Li G, Lin B, Cao J, Teng L. ER-α36: a novel biomarker and potential therapeutic target in breast cancer. Onco Targets Ther. 2014;7:1525–1533. doi: 10.2147/OTT.S65345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhou X, Gu Y, Wang DN, Ni S, Yan J. Eight functional polymorphisms in the estrogen receptor 1 gene and endometrial cancer risk: a meta-analysis. PLoS One. 2013;8(4):e60851. doi: 10.1371/journal.pone.0060851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rojano-Mejía D, Coral-Vázquez RM, Coronel A, Cortes-Espinosa L, del Carmen Aguirre-García M, Valencia-Villalvazo EY, Canto P. Relation of the estrogen receptor and vitamin D receptor polymorphisms with bone mineral density in postmenopausal Mexican-Mestizo women. Gene. 2014;537:10–14. doi: 10.1016/j.gene.2013.12.054. [DOI] [PubMed] [Google Scholar]
- 34.Cahua-Pablo JÁ, Cruz M, Méndez-Palacios A, Antúnez-Ortiz DL, Vences-Velázquez A, del Carmen Alarcón-Romero L, Parra EJ, Tello-Flores VA, Leyva-Vázquez MA, Valladares-Salgado A, Pérez-Macedonio CP, Flores-Alfaro E. Polymorphisms in the LPL and CETP genes and haplotype in the ESR1 gene are associated with metabolic syndrome in women from Southwestern Mexico. Int J Mol Sci. 2015;16:21539–21554. doi: 10.3390/ijms160921539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li LW, Xu L. Menopausal status modifies breast cancer risk associated with ESR1 PvuII and XbaI polymorphisms in Asian women: a HuGE review and meta-analysis. Asian Pacific J Cancer Prev. 2012;13(10):5105–5111. doi: 10.7314/apjcp.2012.13.10.5105. [DOI] [PubMed] [Google Scholar]
- 36.Kornatska AG, Rossokha ZI, Fishchuk LY, Dubenko OD, Medvedieva NL, Flaksemberg МА, Chubei GV, Popova OF, Gorovenko NG. ESR1 gene variants affect FSHR-depended risk of fibrocystic mastopathy in infertile women. Exp Oncol. 2021;43:266–269. doi: 10.32471/exp-oncology2312-8852.vol-43-no-3.16468. [DOI] [PubMed] [Google Scholar]
- 37.Sghaier I, Zidi S, El-Ghali RM, Daldoul A, Aimagambetova G, Almawi WY. Unique ESR1 and ESR2 estrogen receptor gene variants associated with altered risk of triple-negative breast cancer: A case-control study. Gene. 2023;851:146969. doi: 10.1016/j.gene.2022.146969. [DOI] [PubMed] [Google Scholar]
- 38.Johansson H, Aristarco V, Gandini S, Gjerde J, Macis D, Guerrieri-Gonzaga A, Serrano D, Lazzeroni M, Rajasekaran A, Williard CV, Mellgren G, DeCensi A, Bonanni B. Prognostic impact of genetic variants of CYP19A1 and UGT2B17 in a randomized trial for endocrine-responsive postmenopausal breast cancer. Pharmacogenomics J. 2020;20(1):19–26. doi: 10.1038/s41397-019-0087-z. [DOI] [PubMed] [Google Scholar]
- 39.Dai Z, Tian T, Wang M, Yang T, Li H, Lin S, Hao Q, Xu P, Deng Y, Zhou L, Li N, Diao Y. Genetic polymorphisms of estrogen receptor genes are associated with breast cancer susceptibility in Chinese women. Cancer Cell Int. 2019;19:11. doi: 10.1186/s12935-019-0727-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tan SC, Low TY, Mohamad Hanif EA, Sharzehan MAK, Kord-Varkaneh H, Islam MA. The rs9340799 polymorphism of the estrogen receptor alpha (ESR1) gene and its association with breast cancer susceptibility. Sci Rep. 2021;11(1):18619. doi: 10.1038/s41598-021-97935-8. [DOI] [PMC free article] [PubMed] [Google Scholar]