Abstract
Background
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) using umbilical cord blood is a valuable therapy option for patients with acute leukemia (AL). Acute graft-versus-host disease (aGVHD) remains the most frequently encountered complication. This study investigated risk factors for aGVHD and assessed whether post-transplant serum ferritin (SF) within 2 weeks is a potential biomarker for aGVHD in pediatric patients with AL undergoing umbilical cord blood transplantation (UCBT).
Material/Methods
We conducted a retrospective cohort study of 71 patients with AL who underwent UCBT at the Children’s Hospital of Soochow University between 2017 and 2022. We evaluated several factors related to aGVHD. Univariate and multivariate analyses were performed using the proportional subdistribution hazard regression model of Fine and Gray. Analyses of overall survival (OS) were performed using the Kaplan-Meier method, and differences were compared using log-rank tests.
Results
Of the 71 patients, 23 (32.4%) experienced grade II–IV aGVHD, of whom 18 (25.4%) developed grade III–IV aGVHD. Patients with grade II–IV and III–IV aGVHD had worse 5-year OS (69.4±10%, p=0.01; and 60.6±11.6, P=0.007, respectively). Conditioning intensity was a risk factor for grade III–IV aGVHD (HR: 0.34, 95% CI: 0.13–0.89, P=0.027). An SF level >1650 ng/mL within 2 weeks post-transplant was associated with an increased risk of severe aGVHD (HR: 3.61, 95% CI: 1.09–11.97, P=0.036).
Conclusions
Post-transplant SF within 2 weeks was a potential biomarker for developing severe aGVHD. Higher levels of post-transplant SF are associated with a higher incidence of grade II–IV aGVHD and grade III–IV aGVHD.
Keywords: Hospitals, Pediatric; Cord Blood Stem Cell Transplantation; Graft vs Host Disease; Ferritins
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is a valuable therapy option for patients with acute leukemia (AL). Umbilical cord blood, as a conveniently available donor source with low immunogenicity and less stringent human leukocyte antigen (HLA)-matching requirements, has emerged as an alternative donor source because of the scarcity of HLA-matched related donors [1,2]. Although the outcomes of umbilical cord blood transplantation (UCBT) have considerably improved, acute graft-versus-host disease (aGVHD) remains the most common and potentially life-threatening complication [3,4]. The risk factors for aGVHD and outcomes of aGVHD in adults using conventional donor sources are well documented. HLA matching, sex disparity between the donor and recipient, conditioning intensity, increased age, multiparous female donors, ineffective aGVHD prophylaxis, graft source, gut bacterial diversity, and total body irradiation have been implicated as risk factors for the onset of aGVHD [5–10]. However, little is known about risk factors for aGVHD after UCBT in children, and it is unclear whether the risk factors and outcomes are similar to those of conventional donor sources in adults.
Patients undergoing allo-HSCT are often transfused with multiple units of blood products, which can result in iron overload. Serum ferritin (SF) is an acute-phase reactant protein which can become elevated in response to iron overload. SF levels have been shown to correlate with long-term outcomes. According to a meta-analysis of 25 trials involving 4545 patients, a high pretransplant SF level was associated with worse overall survival (OS) [11]. Elevated post-transplant SF levels have also been linked to increased non-relapse mortality and lower OS [12–14]. Furthermore, Lin et al [15] demonstrated that increased pretransplant SF levels are linked to a higher incidence of grade II–IV aGVHD and grade III–IV aGVHD in patients with severe aplastic anemia (SAA). However, the relationship between post-transplant SF and aGVHD in children with AL after UCBT is yet to be clarified.
Therefore, we performed a retrospective study at our center to analyze the outcomes of aGVHD and possible risk factors in pediatric patients with AL after UCBT. The purpose was to identify risk factors for aGVHD to improve the quality of life after UCBT in children with AL.
Material and Methods
Patients
This study included children aged 1–18 years diagnosed with AL who underwent UCBT at Children’s Hospital of Soochow University, Suzhou, China, from 2017 to 2022. The patients’ parents and stem cell donors provided written informed consent before beginning the transplantation process. The patient selection criteria were as follows: (i) diagnosis of acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL), or mixed phenotype acute leukemia (MAL); (ii) aged 0–18 years at the time of transplantation; (iii) received a single-unit of cord blood; (iv) no prior allo-HSCT; and (v) neutrophils were successfully engrafted.
Supportive Care
Calcineurin inhibitors (cyclosporine or tacrolimus) were used in combination with methotrexate or mycophenolate mofetil for GVHD prophylaxis. All patients were given routine supportive treatment, including antiviral (acyclovir) prophylaxis for herpes simplex virus-positive recipients, preemptive cytomegalovirus prophylaxis, and antifungal prophylaxis.
Definitions
AGVHD was diagnosed and graded according to previously published consensus guidelines [16]. OS considered death from any cause as the event, and surviving patients were censored at the date of last contact. Neutrophil engraftment was defined as an absolute neutrophil count ≥0.5×109/L for ≥3 days. Platelet engraftment was defined as platelet count ≥20×109/L without transfusion support for ≥7 days.
Statistical Analysis
To compare differences between groups, the chi-square test and Mann–Whitney U test were used to assess the statistical significance of differences in categorical and continuous variables, respectively, between groups. Univariate and multivariable analyses were performed using Gray’s test and the Fine and Gray proportional subdistribution hazards regression model. Death before day 180 without severe aGVHD was considered a competing event. Variables with P<0.10 were included in the multivariable model. OS was estimated using the Kaplan-Meier method, and differences were compared using log-rank tests. The statistical analyses were performed using RStudio software (version 4.3.1; RStudio Team, Boston, MA, USA; URL: http://www.rstudio.com/).
Results
Outcomes of Acute Graft-Versus-Host Disease
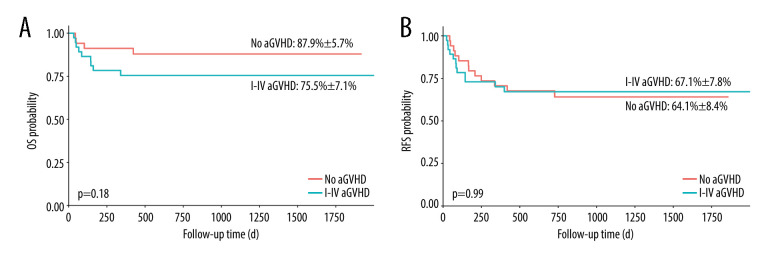
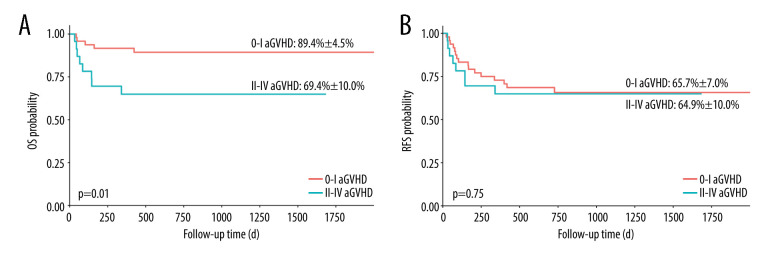
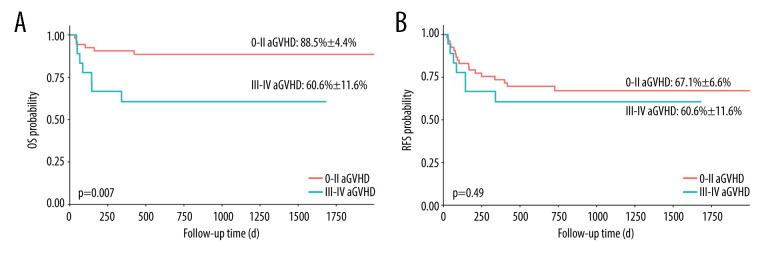
We compared the OS and relapse-free survival (RFS) of the patients between groups of different grades of aGVHD. This study did not find a statistically significant difference in the OS and RFS of patients with grade I–IV aGVHD (Figure 1). However, patients with grade II–IV or grade III–IV aGVHD had worse OS (Figures 2 and 3, 69.4±10%, P=0.01 and 60.6± 11.6%, P=0.007, respectively).
Figure 1.
Overall survival (OS) and relapse-free survival (RFS) in patients with and without acute graft-versus-host disease (aGVHD). (A) OS in patients with grade I–IV aGVHD vs no (grade 0) aGVHD; (B) RFS in patients with grade I–IV aGVHD vs no (grade 0) aGVHD. aGHVD – acute graft-versus-host disease; OS – overall survival; RFS – relapse-free survival. Software used for the creation of the figure: RStudio (version 4.3.1, Microsoft).
Figure 2.
Overall survival (OS) and relapse-free survival (RFS) in patients with and without acute graft-versus-host disease (aGVHD). (A) OS in patients with grade II–IV aGVHD vs grade 0-I aGVHD; (B) RFS in patients with grade II–IV aGVHD vs grade 0-I aGVHD. aGHVD – acute graft-versus-host disease; OS – overall survival; RFS – relapse-free survival. Software used for the creation of the figure: RStudio (version 4.3.1, Microsoft).
Figure 3.
Overall survival (OS) and relapse-free survival (RFS) in patients with and without acute graft-versus-host disease (aGVHD). (A) OS in patients with grade III–IV aGVHD vs grade 0-II aGVHD; (B) RFS in patients with grade III–IV aGVHD vs grade 0-II aGVHD. aGHVD – acute graft-versus-host disease; OS – overall survival; RFS – relapse-free survival. Software used for the creation of the figure: RStudio (version 4.3.1, Microsoft).
Characteristics of Patients with Grade 0-II and III–IV Acute Graft-Versus-Host Disease
The patients were divided into 2 groups: grade 0-II and grade III–IV aGVHD. The characteristics of patients in each group are shown in Table 1. The grade 0-II aGVHD group included 53 patients (27 boys and 26 girls) with a median age of 3.58 years (range: 0.67–12.57 years). There were 30 patients with AML and 23 patients with ALL. The median doses of mononuclear cells (MNC) and CD34+ cells were 6.20×107/kg (range: 1.30–11.33×107/kg) and 2.73×105/kg (range: 1.70–6.41×105/kg), respectively. The grade II–IV aGVHD group included 18 patients (8 boys and 10 girls) with a median age of 3.98 years (range: 1.42–8.83 years), 8 with AML, 8 with ALL, and 2 with MAL. The median doses of MNC and CD34+ cells were 5.90×107/kg (range: 1.37–11.79×107/kg) and 2.78×105/kg (range: 0.83–9.20×105/kg), respectively.
Table 1.
Baseline characteristics of patients with grade 0-II and grade III–IV acute graft-versus-host disease.
| Variable | Grade 0-II aGVHD (n=53) | Grade III–IV aGVHD (n=18) | P value |
|---|---|---|---|
| Recipient age in years, median (range) | 3.58 (0.67–12.57) | 3.98 (1.42–8.83) | 0.463 |
| Sex | 0.634 | ||
| Male, n (%) | 27 (51%) | 8 (44%) | |
| Female, n (%) | 26 (49%) | 10 (56%) | |
| Disease | 0.053 | ||
| AML, n (%) | 30 (57%) | 8 (44%) | |
| ALL, n (%) | 23 (43%) | 8 (44%) | |
| MAL, n (%) | 0 (0%) | 2 (12%) | |
| HLA matching | 0.076 | ||
| ≥9/10, n (%) | 36 (68%) | 8 (44%) | |
| <9/10, n (%) | 17 (32%) | 10 (56%) | |
| ABO matching | 0.663 | ||
| Matched, n (%) | 12 (23%) | 5 (28%) | |
| Mismatched, n (%) | 41 (77%) | 13 (72%) | |
| Disease status at transplant | 0.405 | ||
| CR1, n (%) | 32 (60%) | 13 (72%) | |
| CR2, n (%) | 11 (21%) | 2 (11%) | |
| PR, n (%) | 7 (13%) | 3 (17%) | |
| NR, n (%) | 3 (6%) | 0 (0%) | |
| Conditioning intensity | 0.037 | ||
| MAC, n (%) | 43 (81%) | 10 (56%) | |
| RIC, n (%) | 10 (19%) | 8 (44%) | |
| ATG | 0.340 | ||
| Yes, n (%) | 11 (21%) | 2 (11%) | |
| No, n (%) | 42 (79%) | 16 (89%) | |
| Conditioning drug dose median (range) | |||
| Bu, mg/kg | 12.31 (8.64–14.20) | 12.80 (8.40–14.10) | 0.450 |
| CTX, mg/kg | 120.00 (75.79–141.00) | 120.00 (72.00–141.18) | 0.317 |
| Neutrophil engraftment time, median (range), days | 15.00 (10.00–29.00) | 15.00 (12.00–23.00) | 0.680 |
| Platelet engraftment time, median (range), days | 31.00 (13.00–43.00) | 27.50 (11.00–56.00) | 0.277 |
| Total infused cell count, median (range) | |||
| MNC, ×107/kg | 6.20 (1.30–11.33) | 5.90 (1.37–11.79) | 0.895 |
| CD34+, ×105/kg | 2.73 (1.70–6.41) | 2.78 (0.83–9.20) | 0.648 |
| SF level within 2 weeks post-transplant | 0.022 | ||
| >1650 ng/mL, n (%) | 28 (53%) | 15 (83%) | |
| <1650 ng/mL, n (%) | 25 (47%) | 3 (17%) | |
| SF level within 2 weeks pretransplant | 0.251 | ||
| >1650 ng/mL, n (%) | 6 (11%) | 4 (22%) | |
| <1650 ng/mL, n (%) | 47 (89%) | 14 (78%) |
aGVHD – acute graft-versus-host disease; ALL – acute lymphocytic leukemia; AML – acute myeloid leukemia; ATG – anti-thymocyte globulin; Bu – busulfan; CI – confidence interval; CR – complete response; CTX – cyclophosphamide; HR – hazard ratio; MAL – mixed phenotype acute leukemia; MNC – mononuclear cells; NR – no response; PR – partial response; SF – serum ferritin.
Risk Factors for Grade III–IV Acute Graft-Versus-Host Disease
We assessed several other variables for a potential association with an increased risk of III–IV aGVHD using univariate analysis, including age, sex, disease, HLA matching, ABO matching, disease status at transplant, conditioning intensity, anti-thymocyte globulin (ATG), conditioning drug dose, neutrophil engraftment time, platelet engraftment time, MNC, CD34+, post-transplant SF within 2 weeks (all before aGVHD occurred), and pretransplant SF (Table 2). HLA matching (hazard ratio [HR]: 0.46, 95% CI: 0.18–1.14, P=0.093), conditioning intensity (HR: 0.32, 95% CI: 0.13–0.81, P=0.017), and SF level within 2 weeks post-transplant (HR: 3.98, 95% CI: 1.16–13.60, P=0.028) were associated with an elevated risk of developing III–IV aGVHD. Furthermore, conditioning intensity (HR: 0.34, 95% CI: 0.13–0.89, P=0.027) and SF level within 2 weeks post-transplant (HR: 3.61, 95% CI: 1.09–11.97, P=0.036) were independent risk factors for severe aGVHD in the multivariable analysis.
Table 2.
Risk factors for the occurrence of III–IV acute graft-versus-host disease in pediatric patients undergoing allogeneic hematopoietic stem-cell transplantation.
| Risk factors | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P value | HR | 95% CI | P value | |
| Age | 0.99 | 0.89–1.12 | 0.920 | |||
| Sex female vs male | 1.27 | 0.51–3.17 | 0.600 | |||
| Disease (AL type) | 1.94 | 0.80–4.73 | 0.140 | |||
| HLA matching | 0.46 | 0.18–1.14 | 0.093* | 0.45 | 0.18–1.17 | 0.100 |
| ABO matching | 1.27 | 0.46–3.51 | 0.640 | |||
| Disease status at transplant | ||||||
| CR1 | 1.65 | 0.61–4.48 | 0.330 | |||
| CR2 | 0.51 | 0.12–2.17 | 0.360 | |||
| PR | 1.19 | 0.37–3.84 | 0.770 | |||
| NR | Reference | |||||
| Conditioning intensity | 0.32 | 0.13–0.81 | 0.017* | 0.34 | 0.13–0.89 | 0.027 |
| ATG | 0.50 | 0.12–2.04 | 0.330 | |||
| Conditioning drug dose | ||||||
| Bu | 0.92 | 0.69–1.23 | 0.560 | |||
| CTX | 0.98 | 0.95–1.02 | 0.340 | |||
| Neutrophil engraftment time | 1.00 | 0.90–1.12 | 0.950 | |||
| Platelet engraftment time | 1.01 | 0.97–1.05 | 0.610 | |||
| Total infused cell count | ||||||
| MNC | 1.03 | 0.85–1.24 | 0.800 | |||
| CD34+ | 1.01 | 0.81–1.27 | 0.900 | |||
| SF level within 2 weeks post-transplant | 3.98 | 1.16–13.60 | 0.028* | 3.61 | 1.09–11.97 | 0.036 |
| SF level within 2 weeks pretransplant | 1.97 | 0.66–5.89 | 0.220 | |||
AL – acute leukemia; ATG – anti-thymocyte globulin; Bu – busulfan; CI – confidence interval; CR – complete response; CTX – cyclophosphamide; HR – hazard ratio; MNC – mononuclear cells; NR – no response; PR – partial response; SF – serum ferritin.
Acute Graft-Versus-Host Disease After Umbilical Cord Blood Transplantation
Seventy-one patients were included in the analysis, of whom 37 were in the aGVHD group (14 with grade I aGVHD, 5 with grade II aGVHD, 3 with grade III aGVHD, and 15 with grade IV aGVHD). The sites involved included the skin (31 patients), intestines (22 patients), and liver (10 patients). The patients were divided into 2 groups according to their SF level (Table 3).
Table 3.
Characteristics of pediatric patients with acute graft-versus-host disease according to their serum ferritin level.
| Serum ferritin level | |||
|---|---|---|---|
| Total | >1650 ng/mL | <1650 ng/mL | |
| aGVHD classification | |||
| Grade I, n | 14 | 8 | 6 |
| Grade II, n | 5 | 4 | 1 |
| Grade III, n | 3 | 2 | 1 |
| Grade IV, n | 15 | 13 | 2 |
| aGVHD target organ | |||
| Skin aGVHD, n | 31 | 22 | 9 |
| Intestinal aGVHD, n | 22 | 19 | 3 |
| Hepatic aGVHD, n | 10 | 9 | 1 |
aGVHD – acute graft-versus-host disease.
Incidence of Acute Graft-Versus-Host Disease
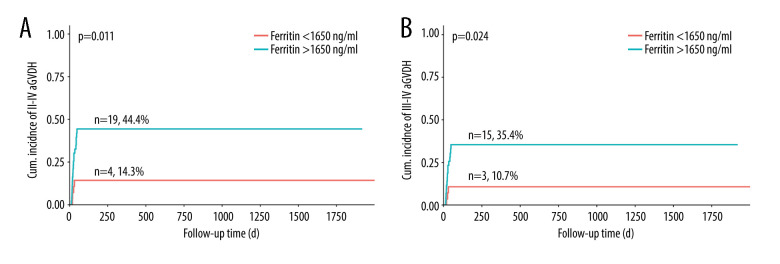
The incidence of grade II–IV was 44.4% in patients with an SF level >1650 ng/mL, as opposed to 14.3% in patients with an SF level <1650 ng/mL, representing a statistically significant difference (P=0.011). Similarly, for grade III–IV aGVHD, the incidence was 35.4% in patients with an SF level >1650 ng/mL and 10.7% in patients with an SF level <1650 ng/mL (P=0.024) (Figure 4).
Figure 4.
(A, B) Cumulative incidence of grades II–IV and III–IV aGVHD in patients with a serum ferritin level >1650 ng/mL and <1650 ng/mL. Software used for the creation of the figure: RStudio (version 4.3.1, Microsoft).
Outcomes According to Serum Ferritin Levels
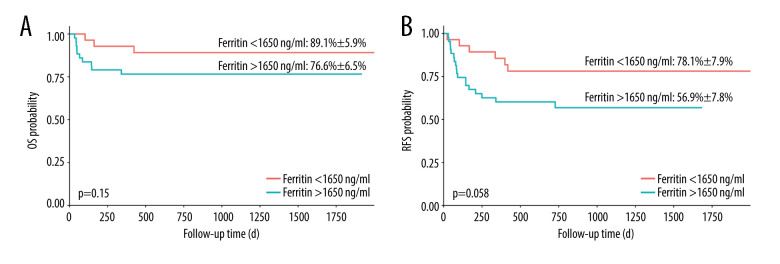
Finally, we compared the OS and RFS of patients in the SF level <1650 ng/mL and >1650 ng/mL groups. OS and RFS were not associated with the SF level (Figure 5, P=0.15 and P=0.058, respectively).
Figure 5.
(A, B) Overall survival and relapse-free survival of patients with serum ferritin levels <1650 ng/mL and >1650 ng/mL. OS – overall survival; RFS – relapse-free survival. Software used for the creation of the figure: RStudio (version 4.3.1, Microsoft).
Discussion
This study assessed the prognosis of children with aGVHD after UCBT and identified risk factors for the development of grades III–IV aGVHD. In recent years, an increasing number of children with AL have undergone UCBT. Despite significant developments in UCBT, aGVDH remains a crucial factor restricting quality of life, especially in patients with severe aGVHD. Patients with grade II–IV and III–IV aGVHD had worse 5-year OS. Infection is the cause of death in most patients with aGVHD, which may have been exacerbated by more aggressive immunosuppressive strategies. Consequently, it is critical to understand the risk factors for aGVHD.
Several risk factors for aGVHD in patients undergoing UCBT, such as myeloablative conditioning, omission of ATG, and double umbilical cord blood, have been described previously [10,17–19]. In this study, we similarly found that conditioning intensity was an independent risk factor for III–IV aGVHD. We also identified the post-transplant SF level as a risk factor for severe aGVDH in the univariate and multivariable analysis.
Previous studies have found that the maximum post-transplant SF levels were significantly higher in patients during aGVHD after allo-HSCT [20]. However, it remains unclear whether post-transplant SF is associated with the occurrence of aGVHD after UCBT in children with AL. We found the post-transplant SF level within 2 weeks was a potential biomarker for predicting development of severe aGVHD. The SF level was also associated with the incidence of II–IV and III–IV aGVHD.
There are multiple potential causes of hyperferritinemia in patients undergoing UCBT. Brissot et al [21] reported that hyperferritinemia can be caused by inflammation, metabolic syndrome, hepatitis, and alcoholism. Iron overload can also induce hyperferritinemia after allo-HSCT, which may be related to transfusions, dyserythropoiesis, or associated genetic factors, and has been reported as a risk factor for aGVHD [22].
Ferritin can have an impact on effects of antigen presenting cells [23]. The activation of antigen-presenting cells is the main mechanism of aGVDH pathogenesis. Furthermore, ferritin enhances the susceptibility to aGVHD by organ toxicity due to reactive oxygen species [24]. Several in vivo studies have demonstrated that mitigating iron overload could potentially reduce the incidence of aGVHD [25,26].
In a retrospective analysis, Lin et al [15] found that pretransplant SF levels were a risk factor for aGVHD in patients with SAA after allo-HSCT. However, we found that the pretransplant SF level was not an independent predictor of aGVHD after UCBT in children with AL. This may be due to the absence of the need for repeated transfusions in patients with AL compared with patients with SAA before UCBT, which may result in no obvious elevation of pretransplant SF. Several studies have shown that post-transplant SF can be used to predict survival outcomes [12–14,27]. Nevertheless, the 5-year OS and RFS did not differ significantly according to the SF level in our analysis. This may be attributable to the limited sample size.
This study has some limitations, including the retrospective design and limited number of patients included in the analysis. Prospective multicenter trials are needed to confirm the risk factors for aGVHD after UCBT in children with AL.
Conclusions
In conclusion, the post-transplant SF level within 2 weeks was a potential biomarker for developing severe aGVHD. Higher levels of post-transplant SF are associated with a higher incidence of grade II–IV aGVHD and grade III–IV aGVHD. However, these results need to be confirmed in prospective studies.
Footnotes
Conflict of interest: None declared
Publisher’s note: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher
Declaration of Figures’ Authenticity: All figures submitted have been created by the authors, who confirm that the images are original with no duplication and have not been previously published in whole or in part.
Financial support: This work was supported by the National Key R&D Program of China (2022YFC2502700), National Natural Science Foundation of China (NSFC) (81970163, 82170218, 82200177, and 82300244), the Jiangsu Project (BE2021654 and BK20210097), the Suzhou project (SZS201615, GSWS2020039, SKY2022183, and SZS2023014), and the Translational Research Grant of NCRCH (No. 2020WSC07)
References
- 1.Gupta A. Cord-blood transplantation in patients with minimal residual disease. N Engl J Med. 2016;375(22):2203–4. doi: 10.1056/NEJMc1612872. [DOI] [PubMed] [Google Scholar]
- 2.Kindwall-Keller TL, Ballen KK. Umbilical cord blood: The promise and the uncertainty. Stem Cells Transl Med. 2020;9(10):1153–62. doi: 10.1002/sctm.19-0288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sugita J, Atsuta Y, Nakamae H, et al. Comparable survival outcomes with haploidentical stem cell transplantation and cord blood transplantation. Bone Marrow Transplant. 2022;57(11):1681–88. doi: 10.1038/s41409-022-01770-y. [DOI] [PubMed] [Google Scholar]
- 4.Kanda J, Umeda K, Kato K, et al. Effect of graft-versus-host disease on outcomes after pediatric single cord blood transplantation. Bone Marrow Transplant. 2020;55(7):1430–37. doi: 10.1038/s41409-020-0853-1. [DOI] [PubMed] [Google Scholar]
- 5.Jagasia M, Arora M, Flowers ME, et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood. 2012;119(1):296–307. doi: 10.1182/blood-2011-06-364265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tie R, Zhang T, Yang B, et al. Clinical implications of HLA locus mismatching in unrelated donor hematopoietic cell transplantation: A meta-analysis. Oncotarget. 2017;8(16):27645–60. doi: 10.18632/oncotarget.15291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang Y, Chang YJ, Xu LP, et al. Who is the best donor for a related HLA haplotype-mismatched transplant? Blood. 2014;124(6):843–50. doi: 10.1182/blood-2014-03-563130. [DOI] [PubMed] [Google Scholar]
- 8.Wang Y, Wu DP, Liu QF, et al. Donor and recipient age, gender and ABO incompatibility regardless of donor source: Validated criteria for donor selection for haematopoietic transplants. Leukemia. 2018;32(2):492–98. doi: 10.1038/leu.2017.199. [DOI] [PubMed] [Google Scholar]
- 9.Lai YR, Chen YH, Hu DM, et al. Multicenter phase II study of a combination of cyclosporine a, methotrexate and mycophenolate mofetil for GVHD prophylaxis: results of the Chinese Bone Marrow Transplant Cooperative Group (CBMTCG) J Hematol Oncol. 2014;7:59. doi: 10.1186/s13045-014-0059-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.MacMillan ML, Weisdorf DJ, Brunstein CG, et al. Acute graft-versus-host disease after unrelated donor umbilical cord blood transplantation: analysis of risk factors. Blood. 2009;113(11):2410–15. doi: 10.1182/blood-2008-07-163238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yan Z, Chen X, Wang H, et al. Effect of pre-transplantation serum ferritin on outcomes in patients undergoing allogeneic hematopoietic stem cell transplantation: A meta-analysis. Medicine (Baltimore) 2018;97(27):e10310. doi: 10.1097/MD.0000000000010310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Meyer SC, O’Meara A, Buser AS, et al. Prognostic impact of posttransplantation iron overload after allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2013;19(3):440–44. doi: 10.1016/j.bbmt.2012.10.012. [DOI] [PubMed] [Google Scholar]
- 13.Wermke M, Schmidt A, Middeke JM, et al. MRI-based liver iron content predicts for nonrelapse mortality in MDS and AML patients undergoing allogeneic stem cell transplantation. Clin Cancer Res. 2012;18(23):6460–68. doi: 10.1158/1078-0432.CCR-12-1683. [DOI] [PubMed] [Google Scholar]
- 14.Pan T, Ji Y, Liu H, et al. Impact of iron overload and iron chelation with deferasirox on outcomes of patients with severe aplastic anemia after allogeneic hematopoietic stem cell transplantation. Transplant Cell Ther. 2023;29(8):507e1–e8. doi: 10.1016/j.jtct.2023.04.016. [DOI] [PubMed] [Google Scholar]
- 15.Lin F, Zuo Y, Zhang Y, et al. The impact of pretransplant serum ferritin on haploidentical hematopoietic stem cell transplant for acquired severe aplastic anemia in children and adolescents. Pediatr Blood Cancer. 2022;69(9):e29845. doi: 10.1002/pbc.29845. [DOI] [PubMed] [Google Scholar]
- 16.Schoemans HM, Lee SJ, Ferrara JL, et al. EBMT-NIH-CIBMTR Task Force position statement on standardized terminology & guidance for graft-versus-host disease assessment. Bone Marrow Transplant. 2018;53(11):1401–15. doi: 10.1038/s41409-018-0204-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lazaryan A, Weisdorf DJ, DeFor T, et al. Risk factors for acute and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation with umbilical cord blood and matched sibling donors. Biol Blood Marrow Transplant. 2016;22(1):134–40. doi: 10.1016/j.bbmt.2015.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chen YB, Wang T, Hemmer MT, et al. GvHD after umbilical cord blood transplantation for acute leukemia: An analysis of risk factors and effect on outcomes. Bone Marrow Transplant. 2017;52(3):400–8. doi: 10.1038/bmt.2016.265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.ElGohary G, El Fakih R, de Latour R, et al. Haploidentical hematopoietic stem cell transplantation in aplastic anemia: A systematic review and meta-analysis of clinical outcome on behalf of the severe aplastic anemia working party of the European group for blood and marrow transplantation (SAAWP of EBMT) Bone Marrow Transplant. 2020;55(10):1906–17. doi: 10.1038/s41409-020-0897-2. [DOI] [PubMed] [Google Scholar]
- 20.Nogai A, Shi Y, Pérez-Hernandez D, et al. Organ siderosis and hemophagocytosis during acute graft-versus-host disease. Haematologica. 2016;101(8):e344–46. doi: 10.3324/haematol.2016.144519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Brissot E, Savani BN, Mohty M. Management of high ferritin in long-term survivors after hematopoietic stem cell transplantation. Semin Hematol. 2012;49(1):35–42. doi: 10.1053/j.seminhematol.2011.10.003. [DOI] [PubMed] [Google Scholar]
- 22.Lee SE, Yahng SA, Cho BS, et al. Impact of pretransplant red cell transfusion on outcome after allogeneic stem cell transplantation in adult patients with severe aplastic anemia. Bone Marrow Transplant. 2016;51(10):1323–29. doi: 10.1038/bmt.2016.140. [DOI] [PubMed] [Google Scholar]
- 23.Qu Z, Guo Y, Li M, et al. Recombinant ferritin nanoparticles can induce dendritic cell maturation through TLR4/NF-κB pathway. Biotechnol Lett. 2020;42(12):2489–500. doi: 10.1007/s10529-020-02944-8. [DOI] [PubMed] [Google Scholar]
- 24.Kim CH, Leitch HA. Iron overload-induced oxidative stress in myelodysplastic syndromes and its cellular sequelae. Crit Rev Oncol Hematol. 2021;163:103367. doi: 10.1016/j.critrevonc.2021.103367. [DOI] [PubMed] [Google Scholar]
- 25.Bair S, Spaulding E, Parkkinen J, et al. Transplantation of allogeneic T cells alters iron homeostasis in NOD/SCID mice. Blood. 2009;113(8):1841–44. doi: 10.1182/blood-2008-09-178517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gerbitz A, Ewing P, Wilke A, et al. Induction of heme oxygenase-1 before conditioning results in improved survival and reduced graft-versus-host disease after experimental allogeneic bone marrow transplantation. Biol Blood Marrow Transplant. 2004;10(7):461–72. doi: 10.1016/j.bbmt.2004.04.001. [DOI] [PubMed] [Google Scholar]
- 27.Fingrut W, Law A, Lam W, et al. Post-transplant ferritin level predicts outcomes after allogeneic hematopoietic stem cell transplant, independent from pre-transplant ferritin level. Ann Hematol. 2021;100(3):789–98. doi: 10.1007/s00277-020-04363-1. [DOI] [PubMed] [Google Scholar]