Abstract
Malignant tumors remain a primary cause of human mortality. Among the various treatment modalities for neoplasms, tumor vaccines have consistently shown efficacy and promising potential. These vaccines offer advantages such as specificity, safety, and tolerability, with mRNA vaccines representing promising platforms. By introducing exogenous mRNAs encoding antigens into somatic cells and subsequently synthesizing antigens through gene expression systems, mRNA vaccines can effectively induce immune responses. Katalin Karikó and Drew Weissman were awarded the 2023 Nobel Prize in Physiology or Medicine for their great contributions to mRNA vaccine research. Compared with traditional tumor vaccines, mRNA vaccines have several advantages, including rapid preparation, reduced contamination, nonintegrability, and high biodegradability. Tumor-targeted therapy is an innovative treatment modality that enables precise targeting of tumor cells, minimizes damage to normal tissues, is safe at high doses, and demonstrates great efficacy. Currently, targeted therapy has become an important treatment option for malignant tumors. The application of mRNA vaccines in tumor-targeted therapy is expanding, with numerous clinical trials underway. We systematically outline the targeted delivery mechanism of mRNA vaccines and the mechanism by which mRNA vaccines induce anti-tumor immune responses, describe the current research and clinical applications of mRNA vaccines in tumor-targeted therapy, and forecast the future development trends of mRNA vaccine application in tumor-targeted therapy.
Keywords: mRNA vaccines, Tumor-targeted therapy, Mechanism, Clinical application, Development trends
Background
Although there have been considerable advancements in cancer treatment, malignant tumors still remain a primary cause of human mortality [1]. Conventional modalities such as surgery, chemotherapy, and radiotherapy remain widely utilized. Additionally, immune checkpoint inhibitors (ICIs) have pioneered novel avenues in tumor-targeted therapy, showing efficacy across diverse malignancies [2]. The growing landscape of tumor-targeted therapy offers hope to cancer patients. This innovative modality enables precise tumor cell targeting, minimizes damage to normal tissues, is tolerable at high doses, and demonstrates significant therapeutic efficacy [3]. mRNA vaccines represent a novel technology at the intersection of molecular biology and immunology and is at the forefront of gene therapy (Table 1). In these vaccines, exogenous antigens encoded by mRNA are introduced into somatic cells, promoting the synthesis of antigenic proteins. This concurrent activation of the body’s principal immune mechanisms, namely, cellular and humoral immunity [4, 5], underscores the pivotal role of mRNA vaccines in tumor-targeted therapy. In recent decades, great strides in experimental techniques have catalysed the widespread utilization of mRNA vaccines across diverse domains, with an emphasis on tumor-targeted therapy. Currently, mRNA vaccines are used for the treatment of various diseases, yielding favourable outcomes [6–10]. (Fig. 1). This review comprehensively discusses the targeted delivery mechanisms of mRNA vaccines and their pivotal role in tumor-targeted therapy. It explores in detail the processes by which mRNA vaccines activate the immune system to recognize and attack tumor cells. Furthermore, this review explores the role of mRNA vaccines in modulating the tumor microenvironment, emphasizing their potential to enhance anti-tumor efficacy by optimizing the working conditions of immune cells. A thorough assessment of the research and application progress of mRNA vaccines in tumor-targeted therapy is presented, including numerous clinical trials that demonstrate their actual effectiveness and potential across multiple cancer types. Finally, this review anticipates future trends in this field, which are expected to further advance the development of mRNA vaccines in cancer treatment. Through this review, we aim to provide readers with a comprehensive and in-depth perspective that aids in understanding the central role of mRNA vaccines in cancer therapy and their promising future prospects.
Table 1.
Breakthroughs of mRNA vaccines in research
| Year | Breakthrough in mRNA vaccine research | References |
|---|---|---|
| 1990 | Concept proposal of mRNA vaccines | [11] |
| 1995 | mRNA tested as cancer vaccine (in mice) | [12] |
| 2000 | Exploration of mRNA as a novel vaccine approach | [13] |
| 2002 | The first clinical trial with ex vivo DCs transfected with mRNA against cancer | [14] |
| 2006–2008 | mRNA modifications to enhance stability and efficacy, leading to improved performance and longevity in vaccine applications and therapeutic interventions | [15–18] |
| 2010 | Significant Progress of mRNA technology in infectious disease vaccines | [19] |
| 2012 | Intranodal delivery of mRNA transfects DCs and elicits anti-tumor immunity | [20] |
| 2013 | Debate on type I IFN in efficacy and safety of mRNA vaccines | [21] |
| 2020–2022 | FDA approval of two mRNA vaccines of COVID-19 | [22, 23] |
| 2022 to present | Research and clinical trials on personalized mRNA cancer vaccines | [24–26] |
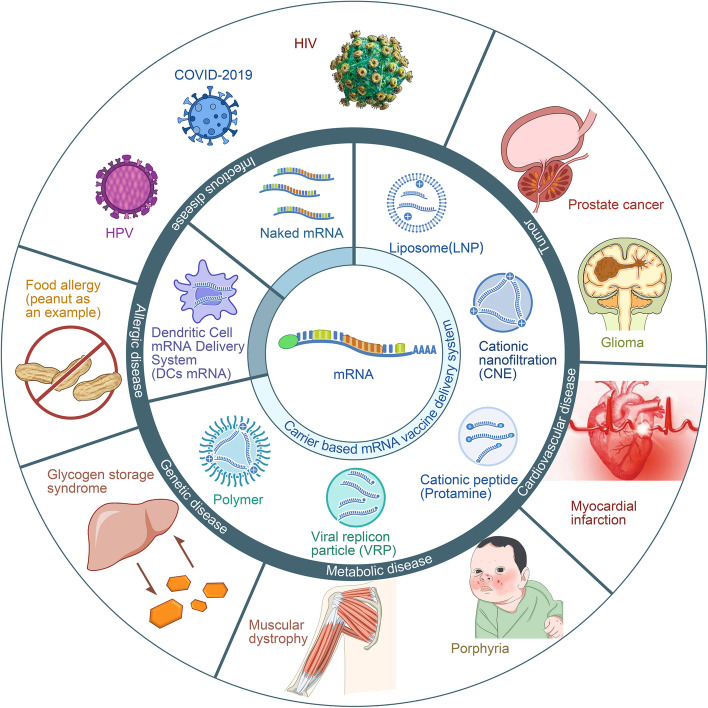
Fig. 1.
Application field of mRNA vaccines. Legend: The mRNA vaccine delivery systems primarily encompass three categories: 1) Carrier-based delivery systems, including lipid nanoparticles (LNPs), cationic nanoemulsions (CNEs), cationic peptides (e.g., protamine), viral replicating particles (VRPs), and polymers. 2) Dendritic cell mRNA delivery systems (DCs mRNA). 3) Naked mRNA. Presently, mRNA vaccines are predominantly employed in the treatment of various diseases, such as: 1) Cardiovascular diseases, including myocardial infarction and heart failure [6]. 2) Metabolic diseases, such as muscular dystrophy [7] and porphyria [8]. 3) Genetic disorders, including glycogen storage disease [9]. 4) Allergic diseases, such as food allergies [27]. 5) Infectious diseases, including human papillomavirus (HPV) [28], Corona Virus Disease 2019 (COVID-19) [29], and human immunodeficiency virus (HIV) [30], among others. 6) Tumors, such as prostate cancer [10] and glioma [31], among others. Naked mRNA vaccines are primarily utilized in the treatment of tumors [32] and infectious diseases [33]. DC-loaded mRNA vaccines are mainly applied in the treatment of tumors [10]
Overview of mRNA vaccines: principle, classification, synthesis and biomarkers
mRNA vaccines are based on the "central dogma" of molecular biology and involve the optimization, chemical modification, and purification of mRNAs with specific antigens [34]. There are two main types of mRNA vaccines: self-amplifying (SAM) and nonreplicating vaccines [35]. SAM vaccines alter a virus's genome to include mRNAs encoding antigens, allowing self-replication without viral protein synthesis and increasing safety and efficiency. Nonreplicating vaccines contain only full-length mRNAs encoding the antigen, with a 5' cap structure and 3' poly(A) tail. Despite their simple structure and direct antigen focus, they have the drawbacks of a short half-life and low in vivo antigen expression [36], necessitating higher doses for effectiveness. The generation of designed DNA templates into an RNA strand is guided by the principle of base complementarity [37]. This process is accomplished through in vitro transcription (IVT), which involves sequence construction, IVT, capping, and tailing, is the primary method for synthesizing mRNA vaccines [38]. DNA templates for IVT must have an open reading frame (ORF), a 5' UTR and 3' UTR, and for self-amplification, a long ORF. The ORF contains start and stop codons [39], allowing splicing for mature mRNA production. The 5' UTR and 3' UTR regulate mRNA stability and translation [40]. The primary challenge facing IVT mRNAs is their immunogenicity. To address this issue, modifications using nucleotides can increase RNA stability and minimize immunogenicity. Among various nucleotide modification methods, chemical alterations, poly(A) tail addition, and sequence optimization are commonly employed [15]. Furthermore, mRNA purification is crucial for eliminating immunogenic properties [41]. Purification techniques mainly include different chromatographic methods (e.g., high-performance liquid chromatography (HPLC) [42], ion exchange [43], size exclusion [44], affinity [45], and Fast protein liquid chromatography (FPLC) [46]), adsorption [47], and membrane technology [41]. The cap structure shields mRNA from exonuclease degradation, ensuring mRNA stability and enhancing translation efficiency [48]. Methylation can be utilized to cap mRNAs in three primary forms: Cap0, Cap1, and Cap2. Traditional enzymatic capping is achieved by enzymatic capping [49], which involves RNA 5'-triphosphatase (RTPase) hydrolysing the 5′ end of RNA, which transfers guanosine monophosphate (GMP) via guanylyltransferase (GTase) to form a cap structure (m7GpppNp), which can be further modified to cap1 (m7GpppN1mp) or cap2 through 2'-O-methyltransferase. Cotranscriptional capping with a Cap analogue (m7GpppG) is also a common method [50] used during mRNA transcription [51]. However, studies have indicated that capping analogues may disrupt mRNA binding, affecting translation efficiency by hindering ribosome recognition and proper 5' end determination [52, 53]. Adding a poly(A) tail is essential for ensuring the stability of mRNA after transcription [54]. There are two main methods used for adding poly(A) tails to mRNAs. The first involves traditional enzymatic polyadenylation, in which the poly(A) tail is added to the 3' end of the mRNA without changing the length of the tail [55]. The other method involves obtaining a poly(A) tail of controllable length by formatting a fixed-length poly(A) sequence on the basis of a DNA template and transcribing it [56]. The ideal length of the poly(A) tail falls within the range of 120 to 150 nucleotides [57–59]. Research on biomarkers for mRNA vaccines remains limited, encompassing two primary areas: 1) Immunophenotyping. Studies have indicated that immunophenotyping can reflect the expression levels of immune checkpoint (ICP) and immunogenic cell death (ICD) regulators, suggesting its potential as a therapeutic biomarker for mRNA vaccines [60]. 2) Tumor antigens. Lin et al. identified six genes that may serve as vaccine targets and stimulate antigen-presenting cell (APC) activation in glioblastoma (GBM), suggesting that these genes are potential biomarkers for mRNA vaccines [61]. Another study demonstrated that patients with malignant mesothelioma (MESO) characterized by high expression of the oncogene fibronectin 1 (FN1) may develop resistance to mRNA vaccination. Consequently, the authors suggest that FN1 could serve as a potential biomarker for mRNA vaccines [62]. However, these studies are primarily bioinformatics analyses, and further exploration through in vitro and in vivo studies is necessary to elucidate the biomarkers associated with mRNA vaccines.
Targeted delivery mechanism of mRNA vaccines
A reliable and secure targeted delivery mechanism is highly important for the progress of mRNA vaccine technology [63]. Currently, several mRNA delivery systems are known, including, Carrier based delivery system, naked mRNA and the dendritic cell-mRNA delivery system (DCs-mRNA) [64](Table 2).
Table 2.
mRNA vaccine delivery systems
| Delivery types | Delivery subtypes | Advanntages | Challenges |
|---|---|---|---|
| Carrier based delivery system | Liposomes and their derivatives, mainly lipid nanoparticles(LNPs) [65] |
Lipid nanoparticles (LNPs) demonstrate a remarkable mRNA encapsulation efficiency, which is pivotal for the protection of mRNA from nuclease degradation and subsequent stable delivery to the target cells Moreover, LNPs possess a distinct advantage in terms of tissue penetration, which facilitates deeper penetration into tissues and organs, thereby enabling more widespread and efficient cellular uptake. The nanoscale dimensions of LNPs contribute to their enhanced intracellular delivery, as they can easily traverse cellular barriers and accumulate within the target cells In addition to their delivery efficiency, LNPs exhibit low cytotoxicity and immunogenicity, which are critical attributes for their application in therapeutic settings Another notable feature of LNPs is their potent adjuvant properties, which are essential for enhancing the immune response when delivering vaccines or immunotherapies [66–68]. |
Lipid nanoparticles (LNPs) are susceptible to degradation, showcasing suboptimal stability under storage conditions, with a propensity for aggregation and fusion phenomena, which can compromise their structural integrity and therapeutic efficacy [69]. |
| Polymers [70] | Certain polymers have demonstrated the ability to significantly enhance the process of endosomal escape, thereby improving the delivery efficiency of therapeutic agents. Additionally, these polymers provide protection for messenger RNAs (mRNAs) against enzymatic degradation, ensuring their stability, and facilitate a safe and effective release of mRNAs into the cytoplasm for subsequent translation [71]. | The low purity and high molecular weight of polymer-based delivery vectors, coupled with their high charge density, can result in significant cytotoxicity [72] | |
| Virus-like replicon particles [73] | Viral replicon particles (VRPs) have the unique capacity to encapsulate self-amplifying RNA (saRNA)-encoded antigens, effectively facilitating their transport to the cytosol. Through in vitro synthesis, viral structural proteins can be produced and utilized for encapsulating saRNAs that encode specific antigens. Extensive researches [74] has illuminated the therapeutic potential of mRNA vaccines administered via VRPs across a diverse array of viral, bacterial diseases, and cancer. This method enhances RNA replication, elicits potent innate immune responses, and promotes the maturation of dendritic cells, contributing to the vaccines' efficacy and immunogenicity. | Viral replicon particles (VRPs) possess a notable disadvantage, as they have been observed to elicit neutralizing antibody responses specifically targeted against the viral surface proteins, as evidenced by studies [75, 76]. | |
| Cationic nanoemulsion (CNE) [77] | CNE can enhance the efficacy of mRNA vaccines by binding to saRNA in a pH-dependent manner, comprising nanoemulsions and cationic lipids. Nanoemulsions can be generated via techniques such as ultrasound, microfluidics, and vigorous stirring [78]. Among the CNE components, the cationic lipid 1.2-diol sn glycerol-3-phosphate choline (DOTAP) stands out for its positive charge, being emulsified with MF59, the identical adjuvant component of the lotion [79]. Additionally, CNE has shown promising therapeutic effects in its ability to deliver saRNA, indicating that lower doses of adjuvant subunits in CNE complexes can elicit substantial immune responses [80]. Numerous studies have been conducted to investigate the stability, toxicity, and biodistribution of CNE, with findings confirming its stability [81]. | However, the conclusions regarding the toxicity of CNE vary across different models. One study demonstrated that the toxicity of nanoemulsions on human foetal lung cells (MRC-5) is dose-dependent [82]. In contrast, another investigation revealed that the rabies animal model exhibited suitable tolerance to CNE-delivered self-amplifying mRNA (SAM) vaccines [83] | |
| Cationic cell-penetrating peptides (CPP) [84, 85] | Cationic peptides, including protamine, a well-established cationic peptide utilized for mRNA transport [86], facilitate the formation of nanosized complexes with mRNAs. These complexes effectively shield the mRNA from enzymatic degradation, maintain immunogenicity across varying temperatures, and preserve the potency of antigen-encoded mRNA vaccines [87]. Protamine's ability to spontaneously condense mRNA via electrostatic interactions serves to protect the enclosed mRNA from degradation by extracellular RNases [88, 89]. Furthermore, the protamine-mRNA complexes demonstrate adjuvant properties, stimulating TLR7/8 to trigger robust innate immune responses [90]. | The specific combination ratio and binding strength between protamine and mRNA are crucial factors that can significantly influence the translation process. These parameters may impose limitations on the efficiency of vaccine protein expression, ultimately affecting the overall effectiveness of the vaccine in eliciting an immune response and providing protection [91]. | |
| Naked mRNA | - | First, the mRNA cannot be integrated into the genome, reducing the risk of genetic mutations. Second, ribosomes can bind directly to the mRNA in the cytoplasm, causing the mRNA to be translated immediately and rapidly initiating an immune response after vaccination. Third, the final position of the mRNA determines the site of protein expression, allowing for precise control of protein expression [92, 93]. | The lack of a carrier during the delivery process can lead to unstable protein translation and expression. However, this can be mitigated by altering the administration method and proper chemical modifications. However, research in this area is relatively limited at present [94]. |
| Dendritic Cell-mRNA Delivery System (DCs-mRNA) | - | Dendritic cells (DCs) serve as the orchestrators of the immune response, exhibiting unparalleled efficiency in their ability to capture and present antigens. This is achieved through a meticulously regulated process involving internalization and proteolytic degradation. Following this intricate mechanism, DCs proceed to present antigens to CD8 + T or CD4 + T cells via major histocompatibility complexes (MHCs), specifically MHC class I (MHCI) or MHC class II (MHCII). By doing so, they initiate an adaptive immune response [95]. The pivotal role of DCs in this context underscores their significance as prime targets for vaccination strategies. | Challenges primarily include the two aspects: Firstly, serum protein aggregation and mRNA degradation upon systemic administration [96], compromising vaccine integrity. Additionally, the second challenge involves the efficient systematic dissemination of mRNA vaccines, ensuring uniform distribution [97] for optimal immune response. |
Carrier based delivery system
Two major types of carrier molecules have been utilized in nucleotide delivery systems: viral carriers and non-viral carriers [98]. However, owing to associated limitations such as potential immunogenicity, tumorigenicity, and low drug loading, the use of viral carriers has been limited. Conversely, nonviral carriers, including liposomes and their derivatives [65], polymers [70], virus-like replicon particles [73], cationic nanoemulsion (CNE) [77], and cationic cell-penetrating peptides (CPP) [84, 85], have garnered significant attention. Among these carriers, liposomes and their derivatives, particularly lipid nanoparticles (LNPs) [99, 100], stand out as widely employed delivery systems. LNPs typically have four key components: ionizable amino lipids, cholesterol, polyethylene glycol lipids, and auxiliary lipids such as double stearyl phosphatidylcholine (DSPC) [101–103]. LNPs, which are approximately 100 nm in diameter, are strikingly similar in both size and composition to various viral entities, mirroring the dimensions of infectious agents such as the SARS-CoV-2 virus (approximately 100 nm), influenza A virus (ranging from 80 to 120 nm), and mature HIV particles (approximately 100 nm in diameter) [104]. Post-administration, LNPs are dynamically transported to cells expressing lipid or scavenger receptors akin to natural apolipoprotein conveyance. LNPs offer notable advantages, including high delivery efficacy [105] and commendable biocompatibility. Polymer materials primarily feature cationic liposome polymers (LPPs) with a positive charge that are proficient in mRNA encapsulation to enable protein expression while mitigating degradation risks. However, these methods have limitations such as polydispersity and macromolecule elimination [106]. Lipid nanoparticles (LNPs) exhibit high mRNA encapsulation efficiency and effective cellular transfection, coupled with robust tissue penetration, low cytotoxicity and immunogenicity, and potent adjuvant properties [66–68]. However, LNPs are prone to degradation and exhibit relatively poor stability during storage, tending to aggregate and fuse [69]. Nevertheless, numerous preclinical and clinical trials have confirmed that LNPs hold promising potential as mRNA vaccine carriers, capable of effectively activating immune responses. Continuous technological advancements have led to LNPs with more complex structures and enhanced physical stability [107], yielding substantial achievements in the innovation of vaccine delivery systems [71]. LPPs encompass diverse materials like polyethyleneimine (PEI) [108], polyamide amine (PAMAM) dendritic polymer [109], dendritic macromolecular polypropylene imine [pol (propylene imine), PPI], polyurethane [poly (aminoester), PAE], and polysaccharides [110]. Polyethylenimine (PEI) has been shown to enhance endosomal escape, protect mRNAs from degradation, and facilitate safe release into the cytoplasm [111]. However, the low purity and high molecular weight of polymer-based delivery vectors, coupled with their high charge density, can result in significant cytotoxicity [72]. Cationic peptides, characterized by cations or amphiphilic amino groups (e.g., arginine) in the main and side chains, facilitate mRNA delivery. Notably, protamine, a renowned cationic peptide for mRNA transport [86], forms nanosized complexes with mRNAs to safeguard against RNA enzyme degradation and stabilize immunogenicity across temperatures while preserving the efficacy of antigen-encoded mRNA vaccines [87]. Protamine can spontaneously condense mRNA through electrostatic interactions, thereby protecting the encapsulated mRNA from degradation by extracellular RNases [88, 89]. Additionally, protamine-mRNA complexes can function as adjuvants, activating TLR7/8 to elicit innate immune responses [90]. The combination ratio and binding strength between protamine and mRNA can have implications for the translation process, potentially limiting vaccine protein expression efficiency and overall vaccine effectiveness [91]. Viral replicon particles (VRPs) have the capacity to encapsulate self-amplifying RNA (saRNA)-encoded antigens and facilitate their transportation to the cytosol. In vitro synthesis of viral structural proteins allows for their encapsulation as saRNAs encoding specific antigens. Numerous studies have highlighted the therapeutic potential of mRNA vaccines delivered via VRPs against a spectrum of viral diseases, bacterial diseases, and cancer [74]. It enhances RNA replication, triggers innate immune responses, and promotes the maturation of dendritic cells. However, it also has the drawback of inducing neutralizing antibody responses against the viral surface [75, 76]. CNE can enhance the efficacy of mRNA vaccines by binding to saRNA in a pH-dependent manner, comprising nanoemulsions and cationic lipids. Nanoemulsions can be generated via techniques such as ultrasound, microfluidics, and vigorous stirring [78]. Notably, among the CNE components, the cationic lipid 1.2-diol sn glycerol-3-phosphate choline (DOTAP) stands out for its positive charge, being emulsified with MF59, the identical adjuvant component of the lotion [79]. Additionally, a preclinical study conducted by Brito et al. on the ability of CNE saRNA delivery in rabbits, mice, and nonhuman primates revealed promising therapeutic effects of CNE and indicated that lower doses of adjuvant subunits in CNE complexes could elicit substantial immune responses [80]. Numerous studies have been conducted to investigate the stability, toxicity, and biodistribution of CNE, with findings confirming its stability [81]. However, the conclusions regarding its toxicity vary across different models. One study demonstrated that the toxicity of nanoemulsions on human foetal lung cells (MRC-5) is dose-dependent [82]. In contrast, another investigation revealed that the rabies animal model exhibited suitable tolerance to CNE-delivered self-amplifying mRNA (SAM) vaccines [83]. Biomimetic carriers represent an innovative drug delivery concept employing endogenous substances, biological structures, and processes. Exosomes, a type of lipid bilayer microvesicle characterized by small size and low immunogenicity, are a particularly auspicious biomimetic carrier. Exosomes can prolong the duration of drugs in circulation by evading mononuclear phagocytic system clearance, thereby increasing drug delivery efficiency [112]. In addition, promising new materials for mRNA vaccine delivery research, such as inorganic nanomaterials and hydrogels, are also being explored. Compared with traditional materials, these new materials have great advantages in terms of improving the efficiency and intensity of vaccine mRNA translation [113].
Naked mRNA
Naked mRNA delivery refers to the direct administration of mRNA. This technology has been successfully used in vivo for immune responses, specifically targeting antigen-presenting cells through intradermal [92, 114] and intranodular injections [115, 116]. There are many advantages associated with this delivery method [92, 93]. First, the mRNA cannot be integrated into the genome, reducing the risk of genetic mutations. Second, ribosomes can bind directly to the mRNA in the cytoplasm, causing the mRNA to be translated immediately and rapidly initiating an immune response after vaccination. Third, the final position of the mRNA determines the site of protein expression, allowing for precise control of protein expression. Despite its advantages, naked mRNA delivery also has some major drawbacks [94]. The lack of a carrier during the delivery process can lead to unstable protein translation and expression. However, this can be mitigated by altering the administration method and proper chemical modifications. However, research in this area is relatively limited at present.
Dendritic Cell-mRNA Delivery System (DCs-mRNA)
DCs are the orchestrators of the immune response, showing unparalleled efficiency in capturing and presenting antigens through a meticulously regulated process of internalization and proteolytic degradation. Subsequently, DCs present antigens to CD8 + T or CD4 + T cells through major histocompatibility complexes (MHCs), specifically MHCI or MHCII, thereby initiating an adaptive immune response [95]. This highlights DCs as prime targets for vaccination. Typically, specific mRNAs encoding antigens are delivered into DCs via electroporation, lipid transfection, nuclear transfection, or in vitro acoustic evaporation. Among these techniques, electroporation is preferred [24]due to its high transfection efficacy and independence from carrier molecules. Challenges primarily include the following two aspects: Firstly, serum protein aggregation and mRNA degradation upon systemic administration. Experts suggest addressing this issue by supplementing DCs with granulocyte–macrophage colony-stimulating factor (GMCSF) and IL-4 [96]. The second challenge lies in the systematic dissemination of mRNA vaccines [97].
Mechanism of mRNA Vaccine-Induced anti- tumor immune response
The mechanisms by which mRNA vaccines induce an anti-tumor immune response involve two primary mechanisms. First, they directly induce tumor-specific T-cell responses, including both innate and adaptive immune responses. During this process, mRNA vaccines deliver tumor-associated antigens or tumor-specific antigens generated by intratumoral mutations to the immune system, activating antigen-presenting cells (APCs) and T cells and thereby initiating a specific antitumor immune response. Second, they achieve induction of an antitumor immune response by modulating the tumor microenvironment.
Key factors in the induction of anti- tumor immune response by mRNA vaccines
The key factors in the mRNA vaccine-induced antitumor immune response include the following aspects. First, the design of the vaccine is crucial [117]. By precisely selecting tumor-associated antigens, a vaccine can ensure high specificity and effectiveness. Second, efficient antigen presentation is a key step in inducing an anti-tumor immune response [118]. mRNA vaccines express tumor-associated antigens, making them targets antigen-presenting cells (APCs) in vivo and thereby enhancing antigen presentation efficiency [119]. Third, a refined immune regulatory mechanism contributes to achieving immune balance [120]. mRNA vaccines can induce the generation of immunosuppressive cells and inflammatory factors to balance the immune response, preventing excessive immune damage [121].
Molecular mechanisms underlying the activation of anti- tumor immune response by mRNA vaccines
The molecular mechanisms underlying mRNA vaccine-induced activation of anti-tumor immune responses involve multiple factors, including antigen presentation, immune cell activation, immune regulation, and antigen stimulation of B cells [122].
Inducing innate immunity
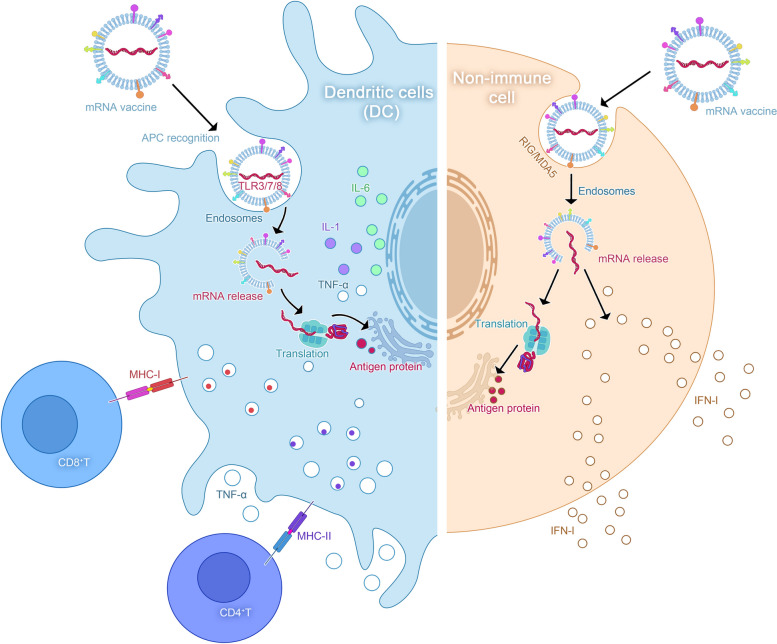
Congenital immune stimulation is driven primarily by the response mechanism of immune cells in defence against pathogens. The Golgi apparatus and endoplasmic reticulum cooperate to translate protein fragments via the MHC [123]. Following DC activation, the MHC can identify corresponding T cells and initiate cytotoxic lymphocyte immunity [124]. MHC complexes and TCRs found on the surface of T cells are the first signals that trigger cellular immune responses. Several components of mRNA vaccines can interact with pattern recognition receptors (PRRs) in endosomes, including TLR3/7/8, which can detect pathogen-associated molecular patterns (PAMPs) in mRNAs. TLR7/8 recognizes single-stranded RNA (ssRNA), whereas TLR3 detects double-stranded RNA (dsRNA). APCs can identify mRNAs and activate TLRs [125]. The activated TLR detects PAMPs and triggers the second signal. The activated second messenger translocates to the nucleus and functions as a potential transcription factor. It can recruit various transactivating factors to promote the expression of proinflammatory cytokines and chemokines such as interleukin-6(IL-6), interleukin-2(IL-2), and tumor necrosis factor-α (TNF-α), thereby activating naive T cells via dual signalling pathways. However, there may be insufficient T lymphocytes to initiate cellular immune responses. Therefore, when these stored cells are exposed to the same antigen again, they are quickly activated. The proper absorption of APCs is a prerequisite for the activation of an immune response, with DCs being primarily responsible. A previous study [126] showed that mRNA vaccines have the ability to stimulate DC cell maturation. In non-immune cells, RIG/MDA5 recognize exogenous mRNA, activating cytokine and chemokine production [127]. Subsequently, they are able to recruit innate immune cells (Fig. 2).
Fig. 2.
mRNA vaccine induces innate immune mechanism. Legend: Upon stimulation of DC cells, the T cells undergo identification, whereby the MHC complex and TCR receptor on their surface serve as the initial signals for cellular immune response. Antigen-presenting cells (APCs) recognize the mRNA, activating TLR and prompting the detection of PAMP, thereby initiating the second signal. The activated second signal translocates to the nucleus as a transcription factor, recruiting various Trans-acting factors to facilitate the expression of proinflammatory cytokines and chemokines. This dual signal pathway effectively activates the initial T cells. In non-immune cells, RIG-I and MDA5 are involved in sensing exogenous mRNA and inducing cytokines/chemokines to recruit innate immune cells
Inducing adaptive immunity
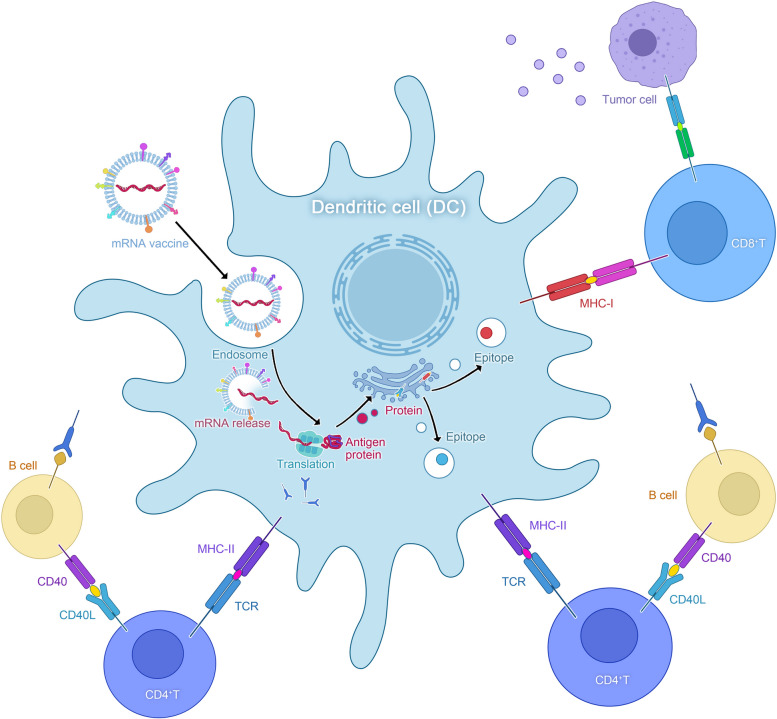
After translation, APCs, such as DCs, take up the protein encoded by mRNA by a variety of mechanisms including micropinocytosis, endocytosis, or phagocytosis [128]. Antigens can be transiently expressed and accumulate in the cytoplasm, allowing for rapid processing into peptides that can be recognized by MHC I. Ribosomal translation generates various antigenic proteins, which are then degraded into fragments in proteasomes and presented as CD8 + T-cell epitopes by MHC-I. Alternatively, antigens can also be transported directly from the cytoplasm to lysosomes, or lysosomal-targeting sequence antigen proteins can be incorporated into mRNA structural design, followed by lysosomal disintegration and presentation as CD4 + T-cell epitopes by MHC-II. In summary, APCs can present exogenous antigens to CD4 + T cells through MHC-II while also cross-presenting exogenous antigens to CD8 + T cells via MHC-I, resulting in the activation of cytotoxic T cells. This stimulation method is termed cross-stimulation. CD4 + T cells can provide support to other immune cells, including B cells and CD8 + T cells, through their helper functions. Ultimately, the cloning amplification of alloantigen-specific T and B cells can result in the elimination of target cells. Furthermore, all nucleated cells possess the ability to process mRNA and present various translated proteins as well as peptides in the MHC-I pathway. Among them, only APCs can present on both MHC-I and MHC-II, triggering immunological responses from CD4 + T or B cells. However, prior to activating adaptive immunity, understanding how cells recognize non-self mRNAs and activate signaling cascades through the interplay of mRNAs, PRRs, and PAMPs is critical. PRRs that can perceive these PAMPs are mainly categorized into two distinct types: extracellular and intracellular [129]. PRRs that recognize RNA contribute to the production of IFN-I. Furthermore, IFN-γ can stimulate the activation of Protein Kinase R (PKR) and eIF2α phosphorylation, leading to cellular and humoral immune responses. As a result, IFN-γ is expected to provide immunological protection to the body. However, mRNA vaccines can overstimulate the immune response, causing excited cells to generate a significant amount of IFN-I, which inhibits mRNA translation and promotes mRNA degradation. Consequently, this downregulates the expression of the target protein, causing a negative reaction on the immune response. Therefore, an effective mRNA vaccine should completely activate innate immunity before initiating adaptive immunity (Fig. 3).
Fig. 3.
mRNA vaccine induces adaptive immune mechanism. Legend. After translation, the proteins encoded by mRNA are taken up by antigen-presenting cells (APCs) via mechanisms such as micropinocytosis, endocytosis, or phagocytosis. These antigens are subsequently processed into peptides and loaded onto the MHC class I pathway. The translation conducted by ribosomes produces immunogenic proteins, which are degraded into fragments within the proteasome and presented to CD8 + T cells via MHC-I. An alternative pathway allows for the direct transport of antigens from the cytoplasm to lysosomes, or the incorporation of a lysosome-targeting sequence within the mRNA structure for lysosomal degradation. The resulting MHC-II peptide complexes are then recognized by the T cell receptor (TCR) on CD4 + T cells
Modulation of the tumor microenvironment by mRNA vaccines
Based on definition provided in a previous report [130], the tumor microenvironment can be described as a local inner environment composed of tumor-infiltrating immune cells, interstitial cells, and active mediators released by these cells along with tumor cells. This microenvironment is critical for tumor growth and progression because it provides important nutrients and energy while also assisting tumor cells in evading immune system responses. Furthermore, the tumor microenvironment has been shown to increase the propensity of tumors to metastasize to other parts of the body. mRNA vaccines, as novel strategies for cancer immunotherapy, also greatly affect the tumor microenvironment [131].
The role of tumor microenvironment during tumor progression
The tumor microenvironment is a complex and dynamic ecosystem within tumor tissue that consists of a diverse array of components, such as tumor cells, immune cells, fibroblasts, extracellular matrix proteins, and an intricate network of cytokines and chemokines. These elements interact in a highly regulated manner, playing crucial roles in tumor growth, invasion, immune evasion, and response to therapy. Understanding the interactions within the tumor microenvironment is essential for developing effective strategies for cancer treatment and improving patient outcomes [132]. For example, cancer-associated fibroblasts (CAFs) and other immune cells have been reported to contribute to this process [133].The tumor microenvironment provides a favorable habitat in which the tumor cells can rapidly proliferate, evade immune surveillance, and metastasize [134]. Tumor cells adapt and evade the immune system by modulating immune suppression signals in response to antitumor immunological pressure [135]. Ultimately, tumor cells create an immunosuppressive microenvironment, which can enhance anti-tumor immunity and promote tumor survival. Thus, an ideal mRNA vaccine may alter the composition of local immune cells while restoring tumor immune surveillance.
mRNA vaccines can alter the distribution of cytokines in the tumor microenvironment
First, mRNA vaccines can alter the levels of cytokines in the tumor microenvironment by expressing tumor-associated antigens, thereby influencing the activation of immune cells and inflammatory responses [136]. The expression of tumor-associated antigens activates immune cells, particularly CD4 + T cells and CD8 + T cells, prompting them to release more cytokines [137]. An increase in these cytokines can disrupt the balance between immunosuppressive cells and inflammatory factors in the TME, making it easier for immune cells to penetrate into tumor tissues and eliminate tumor cells [138]. mRNA vaccines can promote DC maturation through TLR signaling. mRNA vaccines activate the transcription factor NF-κB via the MyD88 and TRIF pathway, thus promoting the generation of cytokines such as interleukins(ILs), tumor necrosis factors(TNFs), and interferon(IFNs), as well as the maturation of cytotoxic T lymphocytes (CTLs), resulting in the elimination of solid tumors during tumor targeted therapy [139]. Furthermore, mRNA vaccines can promote cytokine release by helper T cells, thereby increasing the level of antibodies of the humoral dependent immunity [140]. Furthermore, mRNA vaccines can enhance the recruitment and activation of antigen-presenting cells (APCs) in the TME [141]. APCs, like dendritic cells, macrophages, and B cells, initiate immune responses by presenting tumor antigens to T cells through mRNA vaccines, activating specific anti-tumor immunity [142]. This process helps to establish a bridge between innate and adaptive immune responses in the TME, thereby enhancing anti-tumor immune activity. In addition, mRNA vaccines can also regulate the expression of immune checkpoint molecules in the TME [143]. Immune checkpoints are a type of immune inhibitory molecules that play a key role in regulating immune responses and maintaining self-tolerance [144]. mRNA vaccines can influence the expression of immune checkpoint molecules on immune cells and tumor cells, potentially overcoming the immune evasion mechanisms employed by tumors [28]. By intervening in immune checkpoints, mRNA vaccines can enhance anti-tumor immune responses and improve the efficacy of tumor immune therapy [119].
mRNA vaccines can modulate tumor immune microenvironment (TIME)
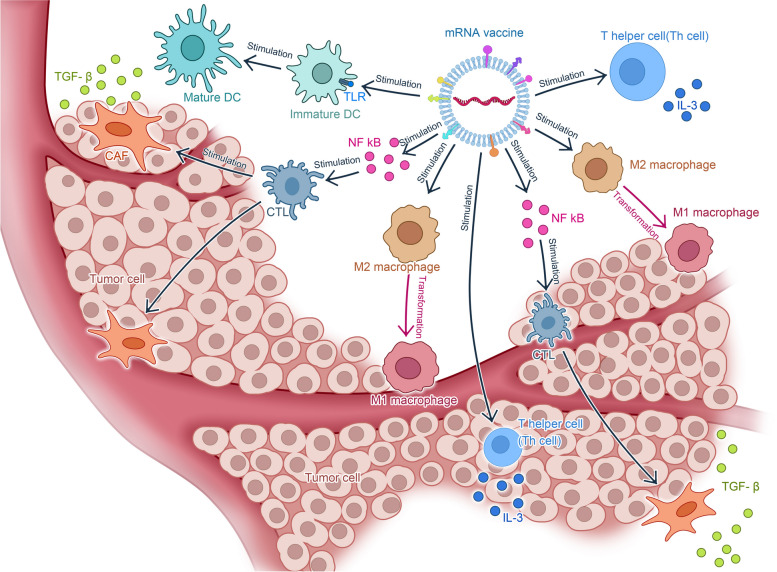
mRNA vaccines have the potential to reshape the tumor immune microenvironment (TIME) via two primary mechanisms: 1) regulating the balance between M1 and M2 macrophages and 2) stimulating cytokine release by different types of T cells (Fig. 4). The interaction of malignant cells and immunological components in the tumor microenvironment (TME) has a great effect on tumor growth and maturation. Tumor cells frequently exploit immunosuppressive mechanisms, such as the production of immunosuppressive proteins, to evade immune surveillance. However, studies have shown that mRNA vaccines can restore tumor immunosurveillance by increasing MHC-I expression [145]. mRNA vaccines can also inhibit tumor growth by regulating the ratio of M1 to M2 macrophages to control tumor progression. Macrophages carry out their functions through two subtypes: M1 and M2. M1 macrophages promote inflammation, whereas M2 macrophages suppress it. In the tumor microenvironment, macrophages mostly exhibit the M2 phenotype. However, studies have shown that mRNA vaccines can increase the ratio of M1 macrophages to M2 macrophages by promoting the transformation of M2 macrophages into M1 macrophages. This is particularly useful for suppressing tumor growth and mitigating tumor immune escape [146].
Fig. 4.
mRNA vaccines reshape tumor immune microenvironment (TIME). Legend.mRNAvaccine possesses the potential to reshape the tumor immune microenvironment via two primary mechanisms. Firstly, it regulates the equilibrium between M1 and M2 macrophages, thus transforming M2 macrophages into M1 macrophages. Secondly, it induces the secretion of cytokines by various T cells (For example T helper cell). Additionally, the vaccine promotes the maturation of dendritic cells (DC) through Toll-like receptor (TLR) receptors, activates the transcription factor NF kB to stimulate the maturation of cytotoxic T lymphocytes (CTL), and prompts T helper cells to secrete cytokines
Research and current applications of mRNA vaccines in tumor targeted therapy
In the realm of tumor-targeted therapy, mRNA vaccines are utilized in two main ways: mRNA tumor vaccines directly target tumor cells, and increasing treatment effectiveness by combining mRNA vaccines with other tumor-targeted therapies, such as immune checkpoint inhibitors. The continual evolution of these strategies has revolutionized tumor-targeted therapy, presenting novel avenues to enhance treatment outcomes among cancer patients and illustrating the promising role of mRNA vaccines in combating cancer.
mRNA tumor vaccines
Currently, mRNA tumor vaccines produced using IVT mainly target four distinct types of molecules: (1) encoding tumor-associated antigens (TAA), (2) encoding tumor-specific antigens (TSA), (3) encoding tumor-Associated Viruses.
mRNA vaccines encoding TAAs
TAAs are expressed in normal cells as well, but at relatively higher levels in tumor cells [147]. These antigens typically arise from abnormal differentiation or dysfunction of tumor cells, such as carcinoembryonic antigen (CEA), PRAME, NY-ESO-1, etc. [148–150]. Although TAAs are expressed to some extent in normal tissues, the significant upregulation of their expression in tumor cells allows the immune system to generate targeted immune responses [151]. The utilization of TAAs as targets for mRNA vaccines has already initiated clinical investigations in various solid tumors and haematologic malignancies. mRNA vaccines have the potential to be designed for TAAs that are selectively expressed in cancer cells. CA125 is a TAA in ovarian epithelial carcinoma, whereas AFP is a TAA in liver cancer. Several clinical trials have used mRNA vaccines targeting similar TAAs for therapy, including NCT00831467, NCT03164772, and NCT01995708 [24]. (Table 3).
Table 3.
Clinical trials of mRNA vaccines encoding TAAs
| Cancer type | NCT number | Drug administration | Phase | TAA type | Status | Delivery system | Result | |
|---|---|---|---|---|---|---|---|---|
| Respiratory system tumors (mainly non-small cell lung cancer) | non-small cell lung cancer | NCT03164772 | BI 1361849 (CV9202) + Durvalumab + / − Tremelimumab | I/II | EGFR | Recruiting | Protamine | Good tolerance, and most patients (84%) have found antigen specific immune responses [24] |
| NCT00923312 | mRNACV9201 | I/II | EGFR | Recruiting | Protamine | Good tolerance and immune response detected after treatment; The median progression and overall survival time were 5 months and 10.8 months, respectively [86] | ||
| NCT01915524 | With local irradiation (with or without pemetrexed and with or without EGFR tyrosine-kinase inhibitor) | I | EGFR | Recruiting | Naked RNA | Detectableantigen-specificimmunity in 21 (84%) patients. One (4%) patient had partial response in combination with chemotherapy treatment, and 12 (46%) patients had stable disease [25] | ||
| Reproductive system tumors | ovarian cancer | NCT04163094 | W_ova1 + carboplatin/paclitaxel | I | OVA-1 | Recruiting | Naked RNA | Not published |
| recurrent epithelialOC | NCT01334047 | DC-006 vaccine (mRNA encoding hTERT, survivin) | I/II | hTERT | Recruiting | DC | Not published | |
| Penile Neoplasms Malignant | NCT03418480 | BNT113 (HPV16 E6 and E7 oncoproteins) | I/II | E6/E7 | Recruiting | Unkown | Not published | |
| ovarian cancer | NCT01456065 | DCs loaded with TERT-mRNA and Survivin-peptide | I | TERT-mRNA and Survivin-peptide | Unknown | DC | Not published | |
| Skin tumor (mainly melanoma) | melanoma | NCT02410 733 | NY-ESO-1, tyrosinase, MAGE-A3, and TPTE | I | NY-ESO-1, MAGE-A3, tyrosinase, TPTE | Active, not publishedt recruiting | Lipid nanoparticles | Immune responses against a minimum of one tumourassociated antigen in 39 (75%) patients. mRNA vaccine with anti-PD-1 therapy: six (35%) patients had partial response and two (12%) had stable disease; mRNA vaccine monot publishedtherapy: three (12%) patients had partial response, and seven (28%) had stable disease [152] |
| NCT04526899 | BNT111 (NY-ESO-1, tyrosinase, MAGE-A3, and TPTE) + cemiplimab | II | NY-ESO-1, MAGE-A3, tyrosinase, TPTE | Recruiting | RNA-LPX | Good tolerance, strong CD4 + and CD8 + T cell immunity after treatment, combined with PD-1 inhibitors, achieving an objective response rate of 35% [153] | ||
| NCT00940004 | Dendritic cells electroporated with mRNA encoding gp100 and tyrosinase | I/II | gp100 | Completed | DC | Not published | ||
| NCT01676779 | mRNA; b.TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100 | II | NY-ESO-1, MAGE-A3, tyrosinase, TPTE | Completed | DC | Good tolerance (symptoms: transient local skin reactions, flu like symptoms, shivering after infusion), and may increase the one-year survival rate (71% in the treatment group, 35% in the control group) [154] | ||
| NCT01302496 | mRNA; b.TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100 | II | NY-ESO-1, MAGE-A3. tyrosinase. TPTE | Completed | DC | 12 out of 15 patients showed T cell stimulation response.Some patients have strong immune responses; Both single therapy and combination therapy can induce multifunctional CD8 + T cell responses, which may provide a benchmark for achieving the immune stimulation levels required for sustained clinical remission [26] | ||
| NCT01676779 | mRNA; TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100 | II | mRNA; TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100 | Completed | DC | Good tolerance (symptoms: transient local skin reactions, flu like symptoms, shivering after infusion), and may increase the one-year survival rate (71% in the treatment group, 35% in the control group) | ||
| NCT01456104 | Langerhans-type dendritic cells (a.k.a. Langerhans cells or LCs) | I | HLA-A | Completed | DC | Unknown | ||
| NCT00978913 | DCs transfected with hTERT, survivin and p53 | I | hTERT, survivin and p53 | Completed | DC | Unknown | ||
| NCT00961844 | Dendritic cells—transfected with hTERT-, survivin- and tumor cell derived mRNA + ex vivo T cell expansion and reinfusion + Temozolomid | I/II | hTERT-, survivin- and tumor cell derived mRNA | Terminated | DC | Unknown | ||
| NCT00929019 | Autologous dendritic cells electroporated with mRNA | I/II | HLA-A2 | Terminated | DC | Not reported | ||
| NCT01302496 | mRNA; b.TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100s | II | MAGE-A3, MAGE-C2, tyrosinase, gp100s | Completed | DC | Among the 15 patients, 12 had T cell stimulation response; Some patients have strong immune responses; Both single therapy and combination therapy can induce multifunctional CD8 + T cell responses, which may provide a benchmark for achieving immune stimulation levels required for sustained clinical remission [24] | ||
| Urinarysystem tumor (mainly prostate cancer) | Prostate cancer | NCT04382898 | BNT112 (PAP, PSA, and three undisclosed antigens) + cemiplimab | I/II | PAP, PSA, and three undisclosed antigens | recruiting | RNA-LPX | Not published |
| NCT01817738 | mRNA vaccine CV9104 | I/II | Terminated | Protamine | Not published | |||
| NCT01446731 | DCs transfected with PSA, PAP, survivin and hTERT mRNA + docetaxel | II | PSA, PAP, survivin and hTERT | Completed | DC | Not published | ||
| NCT02692976 | DC loaded with protamine/mRNA encoding keyhole limpet hemocyanin (KLH) + DC loading with MHC I binding peptides, NY-ESO-1 and MUC1 PepTivator® | II | keyhole limpet hemocyanin (KLH) | Completed | DC | Not published | ||
| NCT01197625 | Dendritic cell vaccine | I/II | PSA | Active, not recruiting | DC | Not published | ||
| NCT01153113 | Human telomerase reverse transcriptase mRNA (hTERT mRNA) transfected dendritic cell | I/II | hTERT | Withdrawn | DC | Not published | ||
| NCT00831467 | CV9103:PSA, PSCA, PSMA, STEAP1 | I/II | PSA, PSCA, PSMA, STEAP1 | Completed | Protamine | Good tolerance and immunogenicity | ||
| NCT00831467 | CV9103 (mRNA encoding 4 PSAs, PSCA, PSMA, and STEAP1) | I/II | PSAs, PSCA, PSMA, and STEAP1 | Completed | protamine-stabilized mRNA | well tolerated,prolonged patient survival | ||
| Blood System Cancer(leukemia mainly) | leukemia | NCT05000801 | Dendritic cell vaccine | I | PSAs, PSCA, PSMA, and STEAP1 | recruiting | DC | Not published |
| NCT01686334 | Dendritic cell vaccine | I/II | PSAs, PSCA, PSMA, and STEAP1 | recruiting | DC | Not published | ||
| NCT03083054 | Autologous dendritic cells electroporated with WT1 mRNA | I/II | WT1 | Not published | DC | Not published | ||
| NCT00834002 | Wilms Tumor Gene (WT1) mRNA-transfected autologous dendritic cell | I | WT1 | Completed | DC | Not published | ||
| NCT02649829 | dendritic cell vaccination plus chemotherapy | I/II | WT1 |
Active, not recruiting |
DC | Not published | ||
| NCT01734304 | DCs electroporated with mRNA encoding WT1, PRAME, and CMVpp65 | I/II | WT1, PRAME, and CMVpp65 | Completed | DC | Not published | ||
| NCT00510133 | GRNVAC1 (mRNA encoding human telomerase reverse transcriptase (hTERT) and a portion of the lysosome-associated membrane protein LAMP-1 (LAMP)) | II | hTERT, LAMP-1 (LAMP) | Completed | DC | Not published | ||
| NCT02528682 | MiHA mRNA-loaded PD-L-silenced DC | I/II | WT1 | Completed | DC | Not published | ||
| NCT01686334 | Autologous WT1 mRNA-electroporated DCs | II | WT1 | Recruiting | DC | Not published | ||
| NCT01995708 | CT7, MAGE-A3, and WT1 mRNA-electroporated Langerhans cells (LCs) | I | CT7, MAGE-A3, and WT1 | Active, recruiting | DC |
safe and therapeutic with a slight adverse event [24] |
||
| NCT03083054 | Autologous dendritic cells electroporated with WT1 mRNA | I/II | WT1 |
Active, not recruiting |
DC | Not published | ||
| NCT00965224 | mRNA encoding for Wilms’ tumor antigen WT1 | II | WT1 | Recruiting | DC | Not published | ||
| NCT00514189 | mRNA in AML cell lysate | I | WT1 | Recruiting | DC | Not published | ||
| NCT02405338 | mRNA encoding WT1 and PRAME | I/II | WT1 |
Active, not recruiting |
DC | Not published | ||
| Digestive System Cancer | Colorectal Cancer with Liver Metastases | NCT05533697 |
mRNA-4359 (mRNA encoding IDO and PD-L1) |
I/II | IDO | Recruiting | Unknown | ongoing |
| NCT00228189 | CEA mRNA-loaded DCs | I/II | CEA | Completed | DC | Not published | ||
| Neurological tumors | Glioblastoma | NCT03688178 | Cytomegalovirus pp65-LAMP + temozolomide, varlilumab, and Td | II | Recruiting | DC | Not published | |
| NCT00639639 | Cytomegalovirus pp65-LAMP + autologous lymphocyte transfer and Td | I | pp65-LAMP | Completed | DC | Not published | ||
| NCT04573140 | Autologous total tumor mRNA and pp65 full length (fl) lysosomal associated membrane protein (LAMP) mRNA loaded DOTAP liposome vaccine administered intravenously (RNA loaded lipid particles, RNA-LPs) | I | LAMP | Recruiting | RNA-LPX | Not published | ||
| NCT02649582 | Dendritic cell vaccine + temozolomide chemotherapy | I/II | WT1 | Recruiting | DC | No | ||
| NCT01291420 | WT1 mRNA-electroporated autologous dendritic cell | I/II | WT1 | Unknown | DC | No | ||
| NCT00961844 | mRNA encoding hTERT, survivin, and tumor mRNA | I/II | hTERT | Recruiting | DCs loaded and ex vivo T cell expansion and reinfusion | Not published | ||
| NCT02366728 | Human CMV pp65-LAMP mRNA-pulsed autologous DCs | II | pp65-flLAMP | Active, not recruiting | DC | Not published | ||
| NCT03548571 | Dendritic cell immunization + Adjuvant temozolomide | II/III | Active,not recruiting | DC | Not reported | |||
| Other Cancers | unresectable/metastatic/recurrent head and neck cancer | NCT04534205 | BNT113 (mRNA encoding E6/E7) | II | E6/E7 | Active, not recruiting | LPX | Not published |
mRNA vaccines encoding TSAs
During the carcinogenesis process, malignant cells develop somatic mutations, resulting in the expression of protein sequences that are not expressed in normal cells. Proteasomes can convert these proteins into peptides. Thereafter, the peptides produced can bind to MHC-I receptors and be recognized as new antigens by T-cell receptors. These novel antigens are distinct to each patient, representing tumor specificity and providing opportunities for tumor-targeted therapies [25, 155]. The specific process procedure involves removing a single tumor and identifying specific novel antigens via next-generation sequencing. The new antigens encoded by mRNAs are subsequently injected into the same patient, eliciting an immune response that can attack the tumor [156]. mRNA can encode several antigens, resulting in the presentation of many epitopes. mRNA can express multiple neoantigens, either as individual molecular forms or concatenated forms of multiple coding sequences. Some tumor types can produce a large number of novel antigens, and expressing multiple epitopes may stimulate T cell responses from a perspective of inducing a broad immune response. The most critical aspect of this therapy is verifying the precise immunogenic non-synonymous somatic mutation found in the patients' tumors and discovering new gene expression epitopes. Interestingly, in a clinical trial (NCT03394937), 20 postoperative (IIc, III, and IV) melanoma patients received an intranasal injection of non-formulated mRNA vaccine (ECI-006) [32]. The results indicated that patients tolerated the low-dose mRNA vaccination (600 μg) well and experienced a significant immunological response. No adverse reactions of level 3 or higher were noted [32]. In a second study cohort (NCT03394937), patients with metastatic melanoma in stable condition following conventional treatment for 3–12 months were given ECI-006 vaccination and standard anti-PD-1 treatment, but the results have yet to be announced [32]. Some clinical trials have reported persistent objective reactions in cancer patients following treatment without unmanageable toxic effects (NCT03323398, NCT03480152, etc.) [24, 26] (Table 4).
Table 4.
Clinical trials of neoantigen mRNA vaccines
| Cancer type | NCT number | Drug administration | Phase | Neoantigen | Status | Delivery system | Result | |
|---|---|---|---|---|---|---|---|---|
| Respiratory system tumors (mainly non-small cell lung cancer) | non-small cell lung cancer | NCT03908671 | Personalized mRNA vaccine encoding neoantigen | I | Individual tumour mutations | Not yet recruiting | LPP(liposubcutaneousplex)nanodelivery | Not published |
| NCT03948763 | mRNA-5671 (KRAS gene driver mutations) + pembrolizumab | I | KRAS gene driver mutations | Recruiting | Lipid nanoparticles | Not published | ||
| Skin tumor (mainly melanoma) | melanoma | NCT03897881 | mRNA-4157 (personalised cancer vaccine encoding 20 different mutated neoepitopes) + With pembrolizumab | II | 20 different mutated neoepitopes | Active,not recruiting | Lipid nanoparticles | Not published |
| NCT03480152 | (NCI)-4650, a mRNA-based, personalized cancer vaccine | I | Immunogenic T-cell epitopes derived from neoantigens | Terminated | Naked mRNA | Mutation-specific CD4 + and CD8 + T-cell responses against predicted neoepitopes in three (75%) of four patients.No objective clinical responses [24, 26] | ||
| NCT02035956 | An individualised tumour mutation signature with ten selected neoepitopes for each patient | I | Ten selected neoepitopes for each patient | Completed | Naked mRNA | Out of 125 selected new epitopes, 60% can stimulate T cell responses; Good tolerance to vaccination | ||
| NCT03480152 | NCI-4650 | I/II | Immunogenic neoantigens | Active, not recruiting | LNP | safe with a slight adverse event | ||
| NCT03468244 | personalized mRNA tumor vaccine | NA | Individual tumour mutations | Recruiting | LPP | Not published | ||
| Urinary system tumor (mainly prostate cancer) | Prostate cancer | NCT03289962 | BNT122 (personalised cancer vaccine encoding individual tumour mutations) | I | Individual tumour mutations | Active, not recruiting | RNA-LPX | Good tolerance when used in combination with atezumab; Inducing the release of pro-inflammatory cytokines and peripheral T cell responses in most patients [25] |
| Blood System Cancer(leukemia mainly) | leukemia | NCT03468244 | Personalized mRNA vaccine encoding neoantigen | I | Individual tumour mutations | Recruiting | LPP (lipo subcutaneous plex) nanodelivery | Not published |
| NCT04486378 | BNT122 (personalised cancer vaccine encoding individual tumour mutations) | II | Individual tumour mutations | Recruiting | RNA-LPX | Not published | ||
| Digestive System Cancer | Colorectal Cancer with Liver Metastases | NCT04161755 | BNT122 (personalised cancer vaccine encoding individual tumour mutations) With oxaliplatin, irinotecan, fluorouracil, leucovorin, and atezolizumab | I | Individual tumour mutations | Active, not recruiting | RNA-LPX | Not published |
| Esophagus Cancer | NCT03480152 | National Cancer Institute (NCI)-4650, a messenger ribonucleic acid (mRNA)-based, Personalized Cancer Vaccine | I/II | Individual tumour mutations | Terminated | Lipid nanoparticles | It has safety and immunogenicity, with a maximum tested dose of 0.39 mg and no serious side effects observed | |
|
Colorectal Cancer |
NCT04534205 |
BNT122 (personalised cancer vaccine encoding individual tumour mutations) |
II | Individual tumour mutations | Recruiting | RNA-LPX | Not published | |
|
Pancreatic cancer |
NCT02316457 |
BNT-114 plus BNT-122 (personalised set of pre-manufactured non-mutated shared tumour-associated antigens plus a personalised cancer vaccine encoding individual tumour mutations) |
I | Individual tumour mutations | Active, not recruiting | DC | Not published | |
| gastric cancer, esophageal cancer, and liver cancer | NCT05192460 | neoantigen tumor vaccine | I | individual tumour mutations | Active, not recruiting | Unknown | Not published | |
| colonic neoplasms and colorectal neoplasms | NCT05456165 | GRT-C901/GRTR902 | II | Deoxyribonucleic acid (DNA) mutations present peptides | Recruiting | Chimpanzee adenovirus | Ongoing | |
| Other Cancer | TNBC | NCT02316457 | IVAC_W_bre1_uID and IVAC_M_uID | I | Tumor-specific mutations | Active, not recruiting | LPX | Ongoing |
mRNA vaccines targeting tumor-associated viruses
Tumor viruses are a class of viruses capable of inducing and promoting carcinogenesis in host cells [157]. Currently, the main viruses associated with human tumors include hepatitis B virus (HBV), hepatitis C virus (HCV), Epstein-Barr virus (EBV), human papillomavirus (HPV), and human T-lymphotropic virus type 1 (HTLV-1) [158]. In recent years, significant progress has been made in mRNA vaccine research targeting HPV. Zhou et al. developed an mRNA-based vaccine against the late oncoproteins E6 and E7 of HPV16, which are abundantly expressed in high-grade squamous intraepithelial lesions (HSIL). In vitro and in vivo studies demonstrated that the translated mRNA was functional and elicited antigen-specific adaptive immune responses. Mice with HPV16 + lesions exhibited tumor growth inhibition, extended lifespan, and the development of protective immune memory following vaccination [159]. Hepatitis B virus (HBV) infection is a major pathogenic factor for liver cancer [160]. Research has shown that mRNA vaccines can activate the innate immune system, inducing the production of potent immunogenicity, high levels of virus-specific antibodies, memory B cells, and T cells, offering prospects for functional cure and prevention of HBV recurrence in chronic patients. However, further in-depth evaluation of mRNA vaccines is needed [161]. HIV infection increases the risk of certain tumors, such as Kaposi's sarcoma and non-Hodgkin's lymphoma [162]. Xie et al. [163] utilized mRNA technology to induce the generation of broadly neutralizing antibody (bnAb) precursors essential for multiple HIV subtypes, providing evidence for the feasibility of germline targeting (GT) and progressive immunization strategies in HIV vaccine development. Through animal models, researchers have successfully elicited specific immune responses to HIV bnAbs, providing new strategies for the development of effective HIV vaccines (Table 5).
Table 5.
Clinical trials of mRNA vaccines targeting tumors associated viruses
| Cancer type | NCT number | Drug administration | Phase | Viruse | Status | Delivery system | Result | |
|---|---|---|---|---|---|---|---|---|
| Reproductive system tumors | cervical carcinoma | NCT06273553 | RG002 Injection (an mRNA Therapeutic Vaccine) | I | HPV16/18 | Not yet recruiting | Lipid nanoparticles | Not published |
| NCT02116920 | mRNA E6/E7 | I | HPV genotypes 16, 18, 31, 33 and 45 | Not yet recruiting | Lipid nanoparticles | Unknown | ||
| NCT05119855 | mRNA-1273 Vaccine | IV | 9-valent human papillomavirus (Types 6, 11, 16, 18, 31, 33, 45, 52, 58) | Completed | Lipid nanoparticles | Not published | ||
| Skin tumor | Kaposi's sarcoma | NCT05217641 | BG505 MD39.3 mRNA, BG505 MD39.3 gp151 mRNA or BG505 MD39.3 gp151 CD4KO mRNA | I | HIV | Active,not recruiting | Lipid nanoparticles | Not published |
| NCT02413645 | TriMix 100, TriMix 300 | I | HIV | Completed | Lipid nanoparticles | The vaccine was secure and well tolerated. There were 31 grade 1/2 and 1 grade 3 adverse events, mostly unrelated to the vaccination. Patients who received the highest dose showed a moderate increase in T-cell responses spanning HTI sequence at week 8. In addition, the proportion of responders receiving any dose of HTI increased from 31% at w0 to 80% postvaccination. The intervention had no impact on caHIV-DNA levels, however, caHIV-RNA expression and usVL were transiently increased at weeks 5 and 6 in the highest dose of iHIVARNA, and these changes were positively correlated with HIV-1-specific-induced immune responses. | ||
| NCT00833781 | mRNA-transfected autologous dendritic cells | I | HIV | Completed | DC | There were no differences in interferon-gamma enzyme-linked immunospot responses to HIV-1 Gag or Nef in the vaccine or placebo group. CD4 proliferative responses to KLH increased 2.4-fold (P = 0.026) and CD8 proliferative responses to KLH increased 2.5-fold (P = 0.053) after vaccination. There were increases in CD4 proliferative responses to HIV-1 Gag (2.5-fold vs. baseline, 3.4-fold vs. placebo, P = 0.054) and HIV-1 Nef (2.3-fold vs. baseline, 6.3-fold vs. placebo, P = 0.009) among vaccine recipients, but these responses were short-lived. | ||
| Lymphatic system tumor | Burkitt's lymphoma | NCT05144748 | EBV mRNA vaccine | I | EBV | Recruiting | Lipid nanoparticles | Unknown |
| Digestive System Cancer | Hepatocellular carcinoma | NCT05738447 | HBV mRNA vaccine | I | HBV | Recruiting | Lipid nanoparticles | Unknown |
Adjuvants for mRNA vaccines
Adjuvants are additional immunostimulatory agents in vaccines that activate the innate immune system and provide the necessary "help" to increase the magnitude and quality of adaptive responses, thereby offering maximal protection against specific pathogens [164]. Different adjuvants can elicit various immune responses, influencing overall vaccine outcomes. Currently, the adjuvants used in mRNA vaccines generally include four categories: 1) the intrinsic adjuvant effects of mRNA vaccines; 2) mRNAs encoding immunostimulatory molecules; 3) mRNAs encoding antibodies; and 4) adjuvants for mRNA vaccines on the basis of delivery carrier components.
The intrinsic adjuvant effect of mRNA vaccines
Exogenous RNA molecules can induce immune responses in mammalian cells. Unmodified exogenous nucleotide mRNAs used to express antigens in mRNA vaccines exhibit intrinsic adjuvant activity by triggering innate immune signalling pathways. Notably, double-stranded RNA (dsRNA) can activate TLR3, while single-stranded RNA is capable of activating mouse TLR7, and RNA oligonucleotides containing thio-phosphorylated nucleotide linkages serve as ligands for human TLR8 [165]. Polyuridine (U) and short dsRNA with 5' triphosphate blunt ends can enhance immune responses through the TLR3 and retinoic acid-inducible gene (RIG)-I signaling pathways without compromising antigen expression, thus functioning as adjuvants for mRNA vaccines [166, 167]. The activation of TLRs and RIG-I signaling can induce the production of proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), IL-12, IL-1β, and interferon-alpha/beta (IFNα/β) (Fig. 1), which enhances the protective immunity required by mRNA vaccines while potentially leading to excessive inflammation [168]. Pioneering work by Kariko et al. demonstrated that unmodified RNA molecules activate TLR or RIG-I signaling pathways, triggering antiviral-like immune responses that may impair RNA translation and promote RNA degradation [15]. Nucleoside-modified mRNA can circumvent this immune activation, such as pseudouridine, which has been widely applied in mRNA vaccines [145, 165]. Recent studies indicate that the modified mRNA in the Pfizer-BioNTech BNT162b2 mRNA vaccine may be recognized by melanoma differentiation-associated protein 5 (MDA-5), triggering IFNα production and contributing to the magnitude of antigen-specific T cell and antibody responses [169].
mRNA vaccines encoding immune modulators
Immune modulators typically include cytokines, co-stimulatory molecules, and PRR agonists [170]. Immune modulators such as interferons, interleukins, lymphokines, and tumor necrosis factors play different roles in the immune system. Some trigger inflammation, whereas others support cell growth and differentiation, whereas others enhance lymphocyte functions [171]. It is crucial to restore the anti-tumor immune response by inhibiting immune suppression through the modulation of immune modulators [172]. The use of cytokines in cancer therapy has emerged as a viable treatment option in clinical settings for patients battling cancer [173]. One of the challenges associated with current immunomodulatory treatments is the occurrence of dose-related toxicity resulting from the short half-life of the administered agents, necessitating frequent dosing and systemic distribution, as exemplified by IL-12 therapy [174]. Therefore, intratumoral (IT) and intradermal (ID) injections are often used to induce local immune responses. The transient protein expression and prominent advantages of local delivery make mRNA vaccines complementary to immune modulators, making immune modulators important targets for mRNA vaccines. IL-12, an essential cytokine, can activate CTL and NK cells. In 2018, IL-12 mRNA-LNPs were shown to be effective in hepatocellular carcinoma (HCC) treatment [175]. Due to the unique functions of each cytokine, the efficacy of single cytokine therapy in tumor treatment is limited. Therefore, a combination of multiple cytokines with different functions is often used to improve therapeutic outcomes. Research indicates that mRNA vaccines encoding IL-12 and IL-27 can induce NK and CD8 + T cells within the melanoma tumor microenvironment (TME), demonstrating optimal efficacy [176]. Another study found that a mixture of IL-12, GM-CSF, IL-15, and IFN-α mRNA increased the number of CD4 + T cells and CD8 + T cells in the TME, and adding anti-PD-1 antibody improved mouse survival rates [136]. In 2019, Haabeth et al. [177] pioneered a novel approach to initiate anti-cancer immunity by combining cytokines with co-stimulatory molecules using mRNA. They used a specialized mRNA delivery system to locally express cytokines (CD70, IL-12, and IFN-γ) and co-stimulatory molecules (OX40L, CD80, and CD86) in two tumor models (B-cell lymphoma and colorectal cancer.). Their findings showed that mice given mRNA vaccines containing both cytokines and co-stimulatory molecules achieved complete elimination of tumors, unlike those given other mRNA vaccines that only had partial effects. Combining OX40L with CD80 or CD86, or OX40L with IL-12, notably improved survival rates and delayed tumor growth. These preclinical results indicate that specific cytokines and co-stimulatory molecules could effectively enhance T cell responses against cancer. Currently, most clinical trials on mRNA vaccines encoding immune modulators are in phase I/II to evaluate tolerability. One of the pioneers in this field is eTheRNA, which has developed an adjuvant based on TriMix mRNA consisting of three naked mRNA molecules. Both naked TriMix mRNA evaluated in multiple clinical trials and TriMix mRNA loaded onto DCs ex vivo have shown good tolerability and immunogenicity. Moderna, a leading biotech firm, has created two mRNA therapies enclosed in LNP frameworks to trigger immune responses within tumors. These therapies are undergoing phase I clinical trials to assess the safety and tolerance of repeated administration. One of the products, mRNA-2416, contains mRNA encoding OX40L. It is being tested alone or combined with the intravenous PD-L1 inhibitor durvalumab for treating lymphoma and metastatic ovarian cancer (NCT03323398) [154]. Another candidate, mRNA-2752, comprises OX40L/IL-23/IL-36γmRNA for the treatment of lymphoma (NCT03739931) [178]. Here, OX40L generates secondary signals, enhancing T-cell effector functions and promoting T-cell proliferation and survival. Moderna and AstraZeneca have teamed up to work on the development of MED I1191, which is an IL-12 mRNA product designed for intratumoral administration as part of cancer treatment. Preliminary results from the initial clinical trial revealed that sequential or combination therapy of MED I1191 with durvalumab in patients with advanced solid tumors and skin or subcutaneous lesions is safe and feasible. No treatment-related adverse events leading to treatment discontinuation from MEDI1191 or durvalumab were reported. The combination of MEDI1191 and durvalumab has demonstrated preliminary clinical efficacy; 29.0% of patients achieve either a partial response (PR) or stable disease (SD) for a minimum duration of 12 weeks (NCT03946800) [179] (Table 6).
Table 6.
Clinical trials of mRNA vaccines encoding immunomodulator
| Cancer type | NCT number | Drug administration | Phase | Immunomodulator | Status | Delivery system | Result | |
|---|---|---|---|---|---|---|---|---|
| Respiratory system tumors (mainly non-small cell lung cancer) | non-small cell lung cancer | NCT02688686 | Suppressor of cytokine signaling (SOCS) 1, MUC1 and Survivin mRNA-loaded DC + cytokine-induced killer | I/II | suppressor of cytokine signaling (SOCS) 1 | Unknown | DC | No |
| Skin tumors | Melanoma | NCT01066390 | TriMix-DC | I | TLR4, CD40L and CD70 | Completed | DC | 15 patients had good tolerance, 2 patients had complete remission, and 2 patients had partial remission; It has immunogenicity and long-lasting anti-tumor activity for disease control. Antigen specific CD8 + T cells were detected in 4 out of 5 patients [24] |
| NCT00204607 | mRNA + GM-CSF | I/II | GM-CSF | Completed | Naked RNA | Not reported | ||
| NCT00204516 | mRNA coding for melanoma associated antigens + GM-CSF | I/II | GM-CSF | Completed | Naked mRNA | Not published | ||
| NCT01278940 | mRNA-transfected DCs + IL-2 | I/II | IL-2 | Completed | DC | Not reported | ||
| NCT01530698 | autologous dendritic cell vaccine by mRNA Electroporation | I/II | TLR7/8, IL-6 | Completed | DC | Not reported | ||
| NCT04335890 | Vaccination with IKKb matured Dendritic Cells | I | IL-1ß, IL-6 and PGE2 | Active,not recruiting | DC | Not reported | ||
| NCT03394937 | CD40L, CD70, TLR4; tumour-associated antigens: tyrosinase, gp100, MAGE-A3, MAGE-C2, and PRAME | I | CD40L, CD70, TLR4; | Terminated | DC | Good tolerance, low dose (600 μ g) 4/10 and 3/9 of patients with high (1800ug) levels detected vaccine induced immune responses, with immunogenicity in some patients; No adverse reactions of level 3 or above have occurred [32] | ||
| NCT01676779 | mRNA; b.TAAs: MAGE-A3, MAGE-C2, tyrosinase, gp100 | II | Completed | DC | Good tolerance (symptoms: transient local skin reactions, flu like symptoms, shivering after infusion), and may increase the one-year survival rate (71% in the treatment group, 35% in the control group) | |||
| NCT03291002 | CV8102: TLR7/8, RIG-1 | I | TLR7/8, RIG-1 | Active,not recruiting | Protamine | Both individual and combined administration showed good therapeutic effects, and local induced immune responses were observed to transform into systemic immune responses | ||
| Solid Tumor | NCT03946800 | MEDI1191 (mRNA encoding IL-12) | I | IL-12 | Recruiting | LNP | Preliminary results from the initial clinical trial revealed that sequential or combination therapy of MED I1191 with durvalumab in patients with advanced solid tumors and skin or subcutaneous lesions is safe and feasible. No treatment-related adverse events leading to treatment discontinuation from MEDI1191 or durvalumab were reported. The combination of MEDI1191 and durvalumab has demonstrated preliminary clinical efficacy; 29.0% of patients achieve either a partial response (PR) or stable disease (SD) for a minimum duration of 12 weeks [179] | |
| NCT04455620 |
BNT151 (mRNA encoding IL-2) |
I/II | IL-2 | Recruiting | LPX | Ongoing | ||
| NCT04710043 |
BNT152 (mRNA encoding IL-7) plus BNT153 (mRNA encoding IL-2) |
I | IL-7/IL-2 | Recruiting | LPX | Ongoing | ||
| NCT05392699 | ABOD2011 (mRNA encoding IL-12) | I | IL-12 | Recruiting | Naked-mRNA | Ongoing | ||
| Neurological tumors(mainly glioblastoma) | glioblastoma | NCT03396575 | TTRNA-DC vaccines with GM-CSF | I | GM-CSF | Recruiting | DC | No |
| NCT02465268 | HCMV pp65-shLAMP or pp65-flLAMP + temozolomide, GM-CSF, and Td | II | GM-CSF | Recruiting | DC | Not published | ||
| NCT04963413 | Autologous DCs derived from PBMC loaded with RNA encoding the human CMV matrix protein pp65-LAMP plus GM-CSF | I | GM-CSF | Active,not recruiting | DC | Not published | ||
| NCT00626483 | CMV pp65-LAMP mRNA-loaded DC + GM-CSF | I | CMV pp65-LAMP | Completed | DC | Not published | ||
| NCT03927222 | Human CMV pp65-LAMP mRNA-pulsed autologous DCs + temozolomide + tetanusdiphtheria toxoid + GM-CSF | II | CMV pp65-LAMP | Recruiting | DC | Not published | ||
| Urinarysystem tumor (mainly prostatecancer) | Prostate cancer | NCT02452307 | Peptide vaccine + montanide ISA-51 + / − GM-CSF + / − imiquimod + / − mRNA/protamin | I/II | GM-CSF | Unknown | Protamine | No |
| Blood System Cancer(leukemia mainly) | leukemia | NCT00514189 | Autologous dendritic cells | I | GM-CSF | Terminated | DC | No |
| NCT02693236 | Adenovirus-transfected autologous DCs + CIK cells | I/II | cytokine-induced killer (CIK) cell | Unknown | DC | No | ||
| Digestive System Cancer | Colorectal Cancer with Liver Metastases | NCT04157127 | Pancreatic adenocarcinoma mRNA and lysate With standard therapy | I | Th-1 | Recruiting | DC | No |
| NCT03323398 | mRNA-2416:OX40L | I/II | OX40L | Active, not recruiting | Lipid nanoparticles | Good safety and tolerability, with no occurrence of > Level 3 adverse reactions; 14/39 patients were in stable condition, and 4/6 patients with ovarian cancer were in stable condition. The patients receiving treatment showed that OX40L protein and T cell infiltration in the tumor microenvironment increased, PD-L1 transcription was up-regulated, and the expression of proinflammatory genes was activated [154]. | ||
|
Colon Cancer Gastrointestinal Cancer |
NCT03739931 | mRNA-2752:OX40L, IL-23, IL-36Y | I | OX40L, IL-23, IL-36Y | Recruiting | Lipid nanoparticles | Good tolerance; Tumor shrinkage is related to drug use. 0.5 mg RNA combined with Duvalimab, 81% of bladder cancer focus regression was observed; Treatment has a sustained immune regulatory effect, with elevated levels of IFN-y, TNF-a, and PD-L1 detected in tumors and plasma [178]. | |
| Other Cancer | Ductal carcinoma in situ | NCT02872025 |
mRNA-2752 (mRNA encoding OX40L, IL-23, and IL-36γ) |
I | OX40L, IL-23, and IL-36γ | Recruiting | LNP | well tolerated with slight dose-limiting toxicities |
mRNA vaccines encoding antibodies
Since the development of monoclonal antibodies (mAb) using hybridoma technology in 1975, antibodies have risen to prominence as a rapidly expanding category of pharmaceuticals that specifically target cancer cells [180]. These antibodies have anti-tumor effects through mechanisms such as antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), complement-dependent cytotoxicity (CDC), and blockade of immunosuppressive signals. Conventional antibodies consist of Fab and Fc fragments, with Fab binding to tumor antigens and the Fc region interacting with FcγR on NK cells and macrophages to facilitate cancer cell lysis. In addition to traditional antibodies, single-chain variable fragments (scFvs), single-domain antibodies (sdAbs), and bispecific antibodies (bsAbs) have demonstrated potential in immunotherapy. BsAbs can form T-cell–bsAb–tumor cell complexes, mediating immune cell-mediated killing [181]. Researchers at CureVac studied mRNA vaccines targeting antibodies. After 9 years, they were able to develop mRNA vaccines targeting antibodies that effectively reduced tumor growth in a mouse lymphoma model, supporting the use of mAb-targeting mRNA vaccines in cancer immunotherapy [182]. Leiba-Kasper and colleagues conducted a study to explore the intricate relationship between the absorption, distribution, metabolism, and excretion of the mRNA-encoded anti-HER2 antibody trastuzumab, elucidating its impact on the body and its ability to combat cancer. Through their research, they confirmed the potent anticancer properties of this novel therapeutic approach, shedding light on the mechanisms underlying its efficacy in targeting HER2-positive tumors. The findings from this investigation serve to validate the promising therapeutic potential of mRNA-encoded antibodies in the fight against cancer, opening new avenues for optimized treatment strategies and improved patient outcomes [183]. In addition to monoclonal antibodies, a range of mRNA-encoded bispecific antibodies (bsAbs) have been developed. CCL2 and CCL5 play critical roles in tumor-associated macrophage (TAM) accumulation and HCC immunosuppression. The Wang group developed the bispecific antibody BisCCL2/5i, which targets CCL2 and CCL5, promoting TAM polarization towards the anti-tumor M1 phenotype and reversing immune suppression in the tumor microenvironment (TME). BisCCL2/5i sensitizes HCC to PD-L1 blockade and prolongs survival in a murine model of liver malignancy [184].Bi-specific T cell engagers (BiTEs) are a class of bispecific antibodies lacking an Fc region, consisting of two scFv domains—one recognizing CD3 and the other binding to the cancer cell target antigen—facilitating T cell-mediated tumor killing [185]. The Staid team has developed the RiboMab platform, which includes BiTE mRNA targeting three tumor-associated antigens (TAA)—CD3 × CLDN6, CLDN18.2 × CD3, and EpCAM × CD3. The mRNA encoding CD3 × CLDN6 BiTE exhibits a longer half-life in serum compared to the protein counterpart, leading to complete tumor regression in a mouse model without eliciting systemic immune reactions [186]. CD3 × CLDN6 mRNA (BNT142) is currently undergoing Phase I/II clinical trials (NCT05262530) [179]. While research on mRNA vaccine-encoded antibodies remains limited, monoclonal antibodies (mAbs) and bispecific antibodies (bsAbs) have already shown efficacy. By encoding anticancer antigens, blocking immune checkpoint molecules, and mediating T-cell anti-tumor responses through mRNA vaccine-encoded antibodies, the potential of mRNA antibody immunotherapy is vast. (Table 7).
Table 7.
Clinical trials of mRNA vaccines encoding Ab
| Cancer type | NCT number | Drug administration | Phase | Status | Delivery system | Result | |
|---|---|---|---|---|---|---|---|
| Solid Tumor | NCT05262530 | BNT142 (mRNA encoding antibodies targetingCD3 × CLDN6) | I | Recruiting | LNP | Ongoing | |
| Reproductive system tumors (mainly ovarian cancer) | ovarian cancer | NCT04683939 | BNT141 (mRNA encodinganti-Claudin18.2 monoclonal antibody) | I/II | Recruiting | LNP | Ongoing |
Adjuvants for mRNA vaccines based on delivery carrier components
Cationic lipids may play a critical role in the adjuvant activity of lipid nanoparticles (LNPs). LNPs based on the ionizable cationic lipid DLinDMA exhibit immunostimulatory properties and serve as adjuvants for nucleoside-modified mRNA vaccines, effectively eliciting follicular helper T (TFH) cell responses and germinal centre B-cell responses that produce neutralizing antibodies [187]. The cationic lipid-like substance C1 facilitates the delivery of mRNA into cells, promoting the release of inflammatory cytokines such as IL-1β, IL-6, and IL-12P70 and upregulating the expression of costimulatory molecules via the TLR4 signalling pathway [141]. Lipid C12-TLRa, containing a TLR7/8 agonist, enhances mRNA vaccine delivery and TLR responses, collectively inducing high levels of neutralizing antibodies [188]. Another ionizable lipid-like substance, A2-Iso5–2DC18 (A2), activates STING signaling and releases cytokines such as CXCL10, thereby enhancing the immune response [68]. The non-nucleotide STING agonist-derived amino lipid SAL12, formulated into LNPs, induces the production of IFNβ, triggering potent neutralizing antibodies against SARS-Cov-2 [189]. Additionally, the direct incorporation of all-trans retinoic acid (ATRA) during LNP self-assembly results in ATRA-LNPs that effectively activate dendritic cells, eliciting robust systemic T-cell responses and increasing the infiltration of antigen-specific cytotoxic T cells in colorectal tumors [190]. However, the intrinsic immunostimulatory properties of lipid materials are not always beneficial for vaccines. A study reported that lipid components (DOTMA and DOPE) in mRNA vaccines promote mitochondrial ROS production in monocytes, activating the NLRP3 inflammasome and releasing IL-1β, leading to inflammatory side effects [191]. These findings underscore the importance of selecting lipid components with appropriate immunostimulatory effects for the rational design and development of future mRNA vaccines.
Combined application of mRNA vaccines with other tumor targeted therapies
Currently, many patients have developed resistance to tumor-targeted monotherapies, substantially impacting the effectiveness of tumor-targeted therapy. Therefore, the combined application of mRNA vaccines with other tumor-targeted therapies holds tremendous potential in enhancing treatment outcomes. By harnessing the synergistic effects of different therapeutic approaches, this combination strategy offers a promising avenue for overcoming resistance and improving the overall efficacy of tumor-targeted therapy.
Combined application of mRNA Vaccines and Adoptive Cell Therapy (ACT)
ACT involves extracting immune-active cells from cancer patients, culturing and evaluating their function outside the body, and finally reintroducing them back into patients to target and destroy tumors directly [192]. Adoptive immune cell therapy mainly includes several categories such as TCR-T and CAR-T [193]. Currently, the most commonly used/most effective applications are CAR-T therapy and TCR-T therapy [194, 195].TCR-T cell therapy entails the isolation of T cells from the patient's body, genetic engineering to express a specific T-cell receptor (TCR), and targeting tumor-associated antigens for recognition and elimination [196]. mRNA vaccines have the ability to induce a broad immune response, encompassing humoral and cellular immunity, while TCR-T therapy allows for direct targeting and destruction of tumor cells [197]. If these two therapies are utilized in conjunction, it has the potential to enhance the anti-tumor capabilities of the innate immune system and directly target [198] specific tumor antigens, thereby demonstrating synergistic efficacy. Furthermore, as a relatively safe and repeatable administration mode, mRNA vaccines could enhance the tolerability of TCR-T cell therapy [199]. However, the joint application of mRNA vaccines and TCR-T cell therapy is still in its early exploratory phase [200].CAR-T is a novel immunotherapy approach that employs genetic engineering technology to modify T cells, allowing them to exert anti-tumor effects [201]. Specifically, CAR-T cells constitute a cutting-edge immunocellular therapy that uses genetic engineering to insert customized chimeric antigen receptors (CARs) into T cells. CARs are generated by combining exogenous antigen recognition domains with T-cell receptor domains. This fusion enables CAR-T cells to accurately target and destroy specific cancer cells. Clinical trials have demonstrated promising outcomes with CAR-T-cell therapy, providing new treatment options for cancer patients. These CAR structures consist of single-chain antibody extracellular domains, extracellular hinge domains, transmembrane domains, and intracellular domains, which facilitate their recognition and binding of specific antigens. CAR-T-cell therapy involves transfecting CAR-T cells with mRNAs encoding target proteins to produce the mRNA‒target-CAR-T complex, which is subsequently administered to the body. This therapeutic strategy has been investigated in cancer patients, and promising results have been reported. For instance, a study by Tchou et al. [202] demonstrated that T cells transfected with CAR mRNA targeting c-Met exhibited good tolerance within the breast tumor tissues and were capable of triggering an inflammatory response. This finding suggested that the combination of CAR-T-cell therapy and mRNA vaccines could be a viable treatment approach, enabling more comprehensive and precise targeting of tumors for greater tumor killing efficacy. In another study, Beatty et al. [203]evaluated T cells transfected with mRNA encoding mesothelin-directed CAR as a potential treatment for pancreatic tumors. Interestingly, in a phase 1 study, these cells did not cause CRS or trigger neurological symptoms. Previous studies have indicated that IVT mRNAs encoding TAMs can be directly delivered into tumors via CAR-T nanoparticle technology to induce local regulation of tumor-associated dendritic cells (TADCs) [204].
mRNA vaccines combined with Immune Checkpoint Inhibitor (ICI)
Immune checkpoints play crucial protective roles in regulating the human immune system, acting as brakes to prevent excessive T-cell activation and other undesirable effects. However, tumor cells frequently exploit this regulatory mechanism by overexpressing immune checkpoint molecules, which effectively dampen immune system responses, evade immunosurveillance, and promote tumor development. The most widely researched and applied ICIs include CTLA4, PD-1, and PD-L1. ICI therapy works by blocking immune checkpoint activity and activating T cells to attack tumors, leading to anti-tumor effects. Furthermore, ICIs can maintain induced immunological responses while inhibiting the induction of T-cell depletion indicators, making them useful partners for mRNA vaccines [205]. Ugur Sahin et al. [206] demonstrated that combining the melanoma mRNA vaccine FixVac with PD-1 inhibitors can result in a synergistic effect. Surprisingly, drug sensitivity can even be restored in patients who have previously developed resistance to ICI treatment using this combination treatment. This trial involved 89 advanced melanoma patients (phase IV) who were treated with at least one vaccine targeting a TAA and who had received one or more ICI therapies. All patients received 8 FixVac vaccinations. Interestingly, among them, 47 out of 89 patients (52.81%) displayed positive responses, with 42 exhibiting the best objective response and 5 exhibiting partial reactions. In addition, 3 patients achieved partial remission, 7 patients remained stable, and 1 patient achieved complete remission of the metastatic lesion among the 25 patients who received FixVac monotherapy. Moreover, among the 17 patients treated with FixVac and PD-1 inhibitors, 6 experienced partial reactions and target lesion regression at all doses. During the two-year follow-up, the majority of patients who achieved partial remission or remained stable had longer disease control. Furthermore, in another important clinical trial, researchers used the mRNA-4157/V940 vaccine in combination with pembrolizumab. The results revealed a significant decrease in the risk of disease relapse among patients who were administered combination therapy compared with those who were solely treated with PD-1 inhibitors [207]. In another study, Lina Liu et al. [208] reported that MUC1-based mRNA vaccination can successfully activate CTL responses against triple-negative breast cancer (TNBC). Furthermore, combining an mRNA vaccine with an anti-CTLA-4 monoclonal antibody can markedly enhance the T-cell immune response, and the effect was substantially superior to that of treatment with an mRNA vaccine alone or anti-CTLA-4 monoclonal antibody therapy alone. Although research on these technologies is still relatively limited, their prospects are highly promising. Furthermore, small interfering RNA (siRNA) has shown great potential in ICIs [209], which can be encapsulated in the same vector as mRNAs to prevent repeated delivery, have shown great potential in the treatment of ICIs. Although there are several limitations associated with its small molecular weight and low encapsulation efficiency, the encapsulation concentration of this therapy is still within acceptable limits.
Combination application of mRNA vaccines and oncogene therapy
Oncogene therapy often involves introducing wild-type copies of tumor suppressor genes or exploiting tumor-specific phenotypic changes to selectively target cancer cells. Tumor suppressor genes (TSGs) are essential for maintaining genomic integrity and regulating cell growth, differentiation, and apoptosis [210]. The loss of TSG function is commonly associated with the occurrence, progression, and treatment resistance of cancer [211]. Furthermore, numerous cancer driver genes, mostly TSGs, have been identified through human cancer exon sequencing studies [212]. The majority of TSGs experience functional loss, leading to overactivation of cancer phenotypes through the aforementioned pathways. In such scenarios, a potential therapeutic approach involves suppressing downstream pathways via supplementation with TSGs. However, difficulties in delivery, genomic integration, and mutation risks pose significant obstacles to gene therapy when functional copies are restored via DNA transfection. mRNA vaccines have been demonstrated to effectively address these issues. In a study from 2018, a PTEN-mRNA vaccine was encapsulated in polyethylene glycol (PEG)-coated polymer‒lipid hybrid nanoparticles (LNPs), successfully introducing PTEN-deficient prostate cancer cells. The therapeutic efficacy of inhibiting the PI3K/Akt signalling pathway and promoting cancer cell apoptosis has been validated [213]. In a study conducted in 2021, PTEN mRNA-NPs were shown to restore the protein expression and autophagy of PTEN-deficient cancer cells, demonstrating therapeutic effects against melanoma and PD-1-resistant prostate cancer [214]. While the utilization of TSG-mRNA vaccines remains largely uncharted territory, their efficacy has been demonstrated in various mouse cancer models, underscoring their considerable practical promise.
Future development trends of mRNA vaccines in tumor targeted therapy
mRNA vaccines, as an emerging immunotherapeutic modality, exhibit a diversified and promising outlook in their future development trends [215]. Here, we discussed the importance and impact of personalized vaccine design, multifunctionality, combination therapy strategies, mucosal immunity, and nanotechnology on the application of mRNA vaccines in targeted tumor therapy. Personalized mRNA tumor vaccines design stands out as a research hotspot [143]. By elucidating the genetic and immune characteristics of patient tumor cells, highly personalized mRNA tumor vaccines can be tailored for individual patients. These customized vaccines can more precisely trigger patient-specific immune responses, thereby enhancing treatment efficacy. In the future, interdisciplinary studies encompassing genomics, immunomics, and bioinformatics will provide a more precise theoretical foundation for personalized vaccine design, laying solid groundwork for clinical applications. The development of personalized vaccines will also benefit from the continuous advancement of high-throughput sequencing technologies, which will aid in the rapid and accurate detection of genomic information in individual tumors. Furthermore, the application of artificial intelligence technology will provide more support. For example, optimizing mRNA sequences via artificial intelligence technology has greatly reduced the immunogenicity of mRNA vaccines [216]. By integrating research findings from different interdisciplinary fields, the design of personalized vaccines will continue to be optimized, providing more effective treatment strategies for a vast number of cancer patients. In addition to directly activating the immune system to attack tumors, the future development trend of mRNA vaccines also includes achieving vaccine multifunctionality. These findings indicate that mRNA vaccines can also regulate the immune microenvironment, inhibit tumor growth and spread, and perform other functions. By incorporating various active components, such as immune modulators and cytokines, mRNA-based tumor vaccines will gradually achieve comprehensive intervention against tumors, suggesting new possibilities for cancer treatment [153]. Future research will focus on deciphering the interaction mechanisms of different components in vaccines to achieve more precise and efficient therapeutic effects. Simultaneously, through techniques such as gene editing, the active components in vaccines may undergo more precise regulation, further enhancing the multifunctional effects of the vaccines. Research on multifunctional vaccines will provide broader insights for the development of personalized treatment strategies. In the context of mRNA-based tumor vaccines, the integration of multiple therapeutic modalities is foreseen to emerge as a pivotal and compelling avenue for improvement [217]. The combination of mRNA vaccines with other anti-tumor treatment modalities holds the promise of further enhancing the therapeutic efficacy of vaccines and achieving synergistic effects via multiple treatment mechanisms. In the future, interdisciplinary research teams will conduct more basic research and clinical trials to explore the mechanisms and application prospects of combined therapeutic strategies in tumor treatment. Interdisciplinary collaboration has become a key approach in the study of combined therapeutic strategies, involving experts from multiple disciplines, including immunology, cell biology, pharmacology, and others. Research on combined therapeutic strategies not only requires a deep understanding of the mechanisms of different treatment modalities but also aims to explore how to rationally combine these modalities to achieve optimal effects in the treatment of tumors at different stages. Mucosal immunology, as a new direction in mRNA tumor vaccine research, has garnered significant attention [218]. By guiding immune responses in local mucosal tissues and designing specific mucosal antigens and adjuvants, prevention and treatment of tumors can be achieved. The introduction of mucosal immunization strategies will lead to novel ideas and possibilities in the field of tumor prevention and control, providing broader insights for the research and application of future tumor vaccines. In future research on mucosal immunology, a deeper exploration of the characteristics of mucosal immune tissues and their relevance to tumor-targeted therapy will be carried out. Additionally, customized mucosal immune vaccines are crucial for enhancing the delivery efficiency and immune effects of vaccines in mucosal immune tissues. Furthermore, interdisciplinary collaborations will open new avenues for the application of mucosal immunization in tumor vaccines. The application of nanotechnology in mRNA tumor vaccines has also attracted considerable attention. Nanocarriers can safely and effectively deliver mRNA vaccines into the body, increasing their bioavailability and immunogenicity [219]. Concurrently, the targeted drug delivery and reduced side effects of nanotechnology significantly increase the therapeutic efficacy and safety of tumor vaccines. The combination of nanotechnology and mRNA-based tumor vaccines will lead to the development of novel therapeutic strategies and possibilities for cancer treatment. Future developments in nanotechnology will focus on improving the stability and targeting of carriers, further reducing their metabolism and excretion rates in the body, thereby prolonging the vaccine's efficacy and impact. Additionally, nanotechnology can provide more possibilities for the modification and functionalization of vaccines to meet the diverse treatment needs of different tumor types and individual patients. The application of nanotechnology in mRNA tumour vaccines will introduce more precise and efficient therapeutic approaches to the field of cancer treatment. Despite the significant potential of mRNA vaccines in targeted cancer therapy, several limitations persist: 1) Poor stability: the chemical structure and biological properties of mRNAs render them inherently unstable and susceptible to degradation, which affects their biological activity and immunogenicity [220]. Although chemical modifications can increase stability, their efficacy is limited, with studies indicating that N1-methylpseudouridine-modified mRNAs undergo ribosomal frameshifting during translation [221]. 2) Low in vivo delivery efficiency: The safe and effective delivery of mRNA to target cells is crucial for its functionality [222]. Current delivery systems, such as liposomes and lipid nanoparticles, have improved in stability and intracellular delivery [67], yet the efficiency of delivery remains suboptimal [152]. 3) Complex tumor immune evasion mechanisms: Tumor cells employ multiple strategies to evade immune surveillance [223], presenting a challenge in the field of mRNA vaccine-based targeted cancer therapy [24]. 4) High interindividual variability: Differences in genetic background, immune status, and disease conditions among individuals influence the immunogenicity and therapeutic efficacy of mRNA vaccines [224]. Based on the aforementioned limitations, we advocate for the following enhancements to mRNA vaccines: Firstly, the essence of mRNA vaccines lies in harnessing the endogenous cellular machinery for antigen protein synthesis to trigger immune responses. The optimization of mRNA structure and sequence can significantly enhance its stability and transcriptional efficiency [216, 225]. Researchers have improved mRNA expression levels and duration within cells by refining the 5' cap structure, 3' poly(A) tail, codon usage, and nucleotide modifications [154, 215, 226]. Secondly, beyond optimizing the mRNA itself, the adoption of novel delivery systems is pivotal for enhancing the immunogenic efficacy of mRNA vaccines. Consequently, the development of safer and more effective new carriers is imperative. Emerging lipid nanomaterials, such as biodegradable fatty acid nanoparticles, have demonstrated superior targeting and immunogenicity in animal models [227]. Similarly, as previously noted, biomimetic carriers can enhance the efficiency and intensity of vaccine mRNA translation compared to conventional materials [113].Finally, for mRNA to be translated into antigen proteins, it must successfully traverse into the cytoplasm of target cells, a process fraught with challenges such as lysosomal degradation post-endocytosis or nuclease degradation [228].Therefore, enhancing mRNA stability and penetrance is crucial [229]. For example, physical methods like electroporation can temporarily disrupt the cell membrane, thereby facilitating increased mRNA transport efficiency [230]. We also discuss key points to consider in the clinical translation of mRNA vaccine technology. First, there is a critical need to strengthen preclinical research to thoroughly investigate the biological characteristics of the vaccine, including its stability, immunogenicity, and routes of administration, to ensure its safety and efficacy. Second, conducting large-scale randomized controlled trials is essential for validating the differences between mRNA vaccines and traditional vaccines, particularly in terms of administration routes, dosage design, and immunogenicity. Furthermore, addressing immune responses across different populations can provide scientific evidence for the precise use of vaccines, ensuring their preventive efficacy and safety. Third, the assessment of long-term efficacy and safety is vital. Continuous monitoring of the duration of immunity and potential adverse reactions postvaccination, especially rare severe events, is necessary to obtain long-term immunogenicity, protective efficacy, and safety data through systematic follow-up analysis, thus providing a reliable basis for clinical applications. Finally, optimizing the design of clinical trial protocols is essential to increase the scientific rigor and reliability of trials. The administration routes and treatment regimens should be flexibly designed according to the characteristics of the vaccine, and trial standards and observational indicators should be optimized on the basis of the immunological characteristics of different populations, with endpoint indicators determined in conjunction with epidemiological considerations.
Conclusion
mRNA vaccines represent a promising solution to overcome the limitations encountered in conventional cancer immunotherapy, offering enhanced and durable treatment alternatives. We posit that the integration of mRNA vaccine technology into tumor-targeted therapy will yield expanded applications, serving as an effective tool in the battle against cancer. The versatility of mRNA vaccines, coupled with their ability to elicit immune responses targeting specific tumor antigens, holds great potential for personalized cancer treatment strategies. As research continues to advance in this area, it is conceivable that mRNA vaccines will play a pivotal role in tumor-targeted therapy.
Acknowledgements
Not applicable.
Abbreviations
- mRNA
Messenger ribonucleic acid
- ICI
Immune checkpoint inhibitors
- SAM
Self-amplifying
- ORF
Open reading frame
- NSP
Non-structural protein
- UTR
Untranslated region
- IVT
In vitro transcription
- TLR
Toll like receptors
- dsRNA
Double-stranded RNA
- HPLC
High performance liquid chromatography
- FPLC
Fast protein liquid chromatography
- ARCA
Anti-Reverse Cap Analog
- DC
Dendritic cells
- CNE
Cationic nanoemulsion
- CPP
Cationic cell penetrating peptides
- LNP
Lipid nanoparticle
- LP
Liposomes
- DSPC
Double stearyl phosphatidylcholine
- PEI
Polyethylene imine
- PAMAM
Polyamide amine
- PPI
Pol (propylene imine)
- PAE
Poly (amino ester)
- PEG
Polyethylene glycol
- PLGA
Poly(lactic-co-glycolic acid)
- PTEN
Phosphatase and tensin homologue
- PLA
Polylatic acid
- PBAE
Poly β-aminoesters
- PGLA
POLY(D,L-LACTIDE-CO-GLYCOLIDE)
- VRP
Virus like replicon particle
- DOTAP
1.2-Diol sn glycerol-3-phosphate choline
- VEEV
Venezuelan equine encephalitis virus
- MHC
Main Histocompatibility complex
- GMCSF
Granulocyte macrophage colony-stimulating factor
- LPX
Lipid complex
- TAA
Tumor associated antigens
- APC
Antigen presenting cell
- HLA
Human leukocyte antigen
- BMDC
Bone marrow-derived dendritic cell
- OVA
Ovalbumin
- PRR
Pattern recognition receptor
- PAMP
Pathogen associated molecular pattern
- ssRNA
Single strand RNA
- TCR
T cell receptor
- RIG-I
Cytosolic retinoic acid inducible gene I
- RLR
Cytosolic retinoic acid inducible gene I (RIG-I) like receptor
- MDA 5
Melanoma differentiation associated gene 5
- IFN
Interferon
- CA
Carbohydrate antigen
- AFP
Alpha fetoprotein
- CAR-T
Chimeric Antigen Receptor T-Cell Immunotherapy
- TADC
Tumor associated dendritic cell
- CTLA
Cytotoxic T-lymphocyte-associated protein
- PD-1
Programmed cell death protein 1
- SiRNA
Small interfering RNA
- TME
Tumor microenvironment
- MDSC
Myeloid suppressor cell
- CAF
Cancer associated fibroblast
- NK cell
Natural killer cell
- TIPE2
Tumor necrosis factor- α Induced protein 8-like 2
- TIME
Tumor immune microenvironment
- TNBC
Triple negative breast cancer
- CTLs
Cytotoxic T lymphocyte
- TNF
Tumor necrosis factor
- IL
Interleukin
- TFH
Follicular helper T
- BiTEs
Bi-specific T cell engagers
- scFvs
Single-chain variable fragments
- sdAbs
Single-domain antibodies
- bsAbs
Bispecific antibodies
- ADCP
Antibody-dependent cellular phagocytosis
- CDC
Complement-dependent cytotoxicity
- mAb
Monoclonal antibodies
- bnAb
Broadly neutralizing antibody
- HSIL
High-grade squamous intraepithelial lesions
- HBV
Hepatitis B virus
- HCV
Hepatitis C virus
- EBV
Epstein-Barr virus
- HPV
Human papillomavirus
- HTLV-1
Human T-lymphotropic virus type 1
- GMCSF
Granulocyte–macrophage colony-stimulating factor
- EGFR
Epidermal growth factor receptor
- hTERT
Human telomerase reverse transcriptase
- NY-ESO-1
New York Esophageal Squamous Cell Carcinoma-1
- MAGE
Melanoma-associated antigen
- LC
Langerhans cell
- HLA
Human leucocyte antigen
- PAP
Prostatic Acid Phosphatase
- PSA
Prostate specific antigen
- KLH
Keyhole Limpet Hemocyanin
- PSCA
Prostate Stem Cell Antigen
- PSMA
Prostate Specific Membrane Antigen
- STEAP
Six-segment transmembrane epithelial antigen of prostate
- WT
Williams Tumor
- PRAME
Preferentially expressed antigen in melanoma
- CMV
Cytomegalovirus
- LAMP1
Lysosomal Associated Membrane Protein 1
- IDO
Indoleamine 2,3-dioxygenase
- KRAS
V-Ki-ras2 Kirsten ratsarcoma viral oncogene homolog
- SOCS
Suppressor of cytokine signaling
- CD40L
CD40 Ligand
- PGE2
Prostaglandin E2
- CIK
Cytokine-induced killer cells
- OX40L
TNF Receptor Superfamily Member 4 Ligand
- CLDN6
Claudin-6
Authors’ contributions
YG and LY conducted the research study. YG drafted the manuscript. LZ provided assistance during the revision and drafting process. XP and HL contributed to the conceptual framework, supervised the study, and revised the manuscript. All authors carefully reviewed the final manuscript and approved it for publication.
Funding
This study was supported by grants from the Basic Research Project Of Educational Department of Liaoning Province(LJKMZ20221210),Shenyang Science and Technology Plan (21–104-0–04/22–321-31–02),Shenyang Science and Technology Talent Project (RC220032), National Natural Science Foundation of China (82203680), Natural Scientific Foundation of Liaoning Province(2021-MS-176), National Natural Science Foundation of China (81472302), Natural Scientific Foundation of Liaoning Province(2022-BS-137).
Availability of data and materials
Not applicable.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yu Gao, Liang Yang and Zhenning Li contributed equally to this work.
Contributor Information
Xueqiang Peng, Email: xqpeng@cmu.edu.cn.
Hangyu Li, Email: sj_li_hangyu@sina.com.
References
- 1.Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17–48. 10.3322/caac.21763 [DOI] [PubMed] [Google Scholar]
- 2.Terai M, Sato T. Individualised neoantigen cancer vaccine therapy. Lancet. 2024;403(10427):590–1. 10.1016/S0140-6736(23)02463-7 [DOI] [PubMed] [Google Scholar]
- 3.Pérez-Herrero E, Fernández-Medarde A. Advanced targeted therapies in cancer: drug nanocarriers, the future of chemotherapy. Eur J Pharm Biopharm. 2015;93:52–79. 10.1016/j.ejpb.2015.03.018 [DOI] [PubMed] [Google Scholar]
- 4.Verheul MK, Vos M, de Rond L, De Zeeuw-Brouwer ML, Nijhof KH, Smit D, Oomen D, Molenaar P, Bogaard M, van Bergen R, et al. Contribution of SARS-CoV-2 infection preceding COVID-19 mRNA vaccination to generation of cellular and humoral immune responses in children. Front Immunol. 2023;14:1327875. 10.3389/fimmu.2023.1327875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhao GX, Bu GL, Liu GF, Kong XW, Sun C, Li ZQ, Dai DL, Sun HX, Kang YF, Feng GK, et al. mRNA-based vaccines targeting the T-cell Epitope-rich domain of epstein Barr virus latent proteins elicit robust anti-tumor immunity in mice. Adv Sci (Weinh). 2023;10(35):e2302116. 10.1002/advs.202302116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Magadum A, Kaur K, Zangi L. mRNA-Based Protein Replacement Therapy for the Heart. Mol Ther. 2019;27(4):785–93. 10.1016/j.ymthe.2018.11.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Saito T, Saito T, Hashimoto H, Ogata K, Kobayashi M, Takada H, Kuru S, Kimura T, Nakamura A, Matsumura T. Safety and immunogenicity of mRNA COVID-19 vaccine in inpatients with muscular dystrophy. Muscle Nerve. 2023;67(2):117–23. 10.1002/mus.27761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jiang L, Berraondo P, Jericó D, Guey LT, Sampedro A, Frassetto A, Benenato KE, Burke K, Santamaría E, Alegre M, et al. Systemic messenger RNA as an etiological treatment for acute intermittent porphyria. Nat Med. 2018;24(12):1899–909. 10.1038/s41591-018-0199-z [DOI] [PubMed] [Google Scholar]
- 9.Cao J, Choi M, Guadagnin E, Soty M, Silva M, Verzieux V, Weisser E, Markel A, Zhuo J, Liang S, et al. mRNA therapy restores euglycemia and prevents liver tumors in murine model of glycogen storage disease. Nat Commun. 2021;12(1):3090. 10.1038/s41467-021-23318-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zheng X, Xu H, Yi X, Zhang T, Wei Q, Li H, Ai J. Tumor-antigens and immune landscapes identification for prostate adenocarcinoma mRNA vaccine. Mol Cancer. 2021;20(1):160. 10.1186/s12943-021-01452-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wolff JA, Malone RW. Direct gene transfer into mouse muscle in vivo. Science. 1990;247(4949 Pt 1):1465–8. 10.1126/science.1690918 [DOI] [PubMed] [Google Scholar]
- 12.Conry RM, LoBuglio AF. Characterization of a messenger RNA polynucleotide vaccine vector. Cancer Res. 1995;55(7):1397–400. [PubMed] [Google Scholar]
- 13.Weissman D, Ni H, Scales D. HIV gag mRNA transfection of dendritic cells (DC) delivers encoded antigen to MHC class I and II molecules, causes DC maturation, and induces a potent human in vitro primary immune response. J Immunol. 2000;165(8):4710–7. 10.4049/jimmunol.165.8.4710 [DOI] [PubMed] [Google Scholar]
- 14.Nair SK, Morse M, Boczkowski D. Induction of tumor-specific cytotoxic T lymphocytes in cancer patients by autologous tumor RNA-transfected dendritic cells. Ann Surg. 2002;235(4):540–9. 10.1097/00000658-200204000-00013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Karikó K, Muramatsu H, Welsh FA, Ludwig J, Kato H, Akira S, Weissman D. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol Ther. 2008;16(11):1833–40. 10.1038/mt.2008.200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao X, Yu YT. Targeted pre-mRNA modification for gene silencing and regulation. Nat Methods. 2008;5(1):95–100. 10.1038/nmeth1142 [DOI] [PubMed] [Google Scholar]
- 17.Hanashiro K, Ohta S, Sunagawa M. Modification of Cepsilon mRNA expression by EBV-encoded latent membrane protein 1. Mediators Inflamm. 2006;2006(6):68069. 10.1155/MI/2006/68069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reiter M, Walf VM, Christians A. Modification of mRNA expression after treatment with anabolic agents and the usefulness for gene expression-biomarkers. Anal Chim Acta. 2007;586(1–2):73–81. 10.1016/j.aca.2006.10.049 [DOI] [PubMed] [Google Scholar]
- 19.Lorenzi JC, Trombone AP, Rocha CD. Intranasal vaccination with messenger RNA as a new approach in gene therapy: use against tuberculosis. BMC Biotechnol. 2010;10:77. 10.1186/1472-6750-10-77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Van Lint S, Goyvaerts C, Maenhout S. Preclinical evaluation of TriMix and antigen mRNA-based antitumor therapy. Cancer Res. 2012;72(7):1661–71. 10.1158/0008-5472.CAN-11-2957 [DOI] [PubMed] [Google Scholar]
- 21.Pollard C, Rejman J, De Haes W. Type I IFN counteracts the induction of antigen-specific immune responses by lipid-based delivery of mRNA vaccines. Mol Ther. 2013;21(1):251–9. 10.1038/mt.2012.202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Skowronski DM, De Serres G. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2021;384(16):1576–7. 10.1056/NEJMc2036242 [DOI] [PubMed] [Google Scholar]
- 23.Ali K, Berman G, Zhou H, Deng W. Evaluation of mRNA-1273 SARS-CoV-2 Vaccine in adolescents. N Engl J Med. 2021;385(24):2241–51. 10.1056/NEJMoa2109522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.He Q, Gao H, Tan D, Zhang H, Wang JZ. mRNA cancer vaccines: Advances, trends and challenges. Acta Pharm Sin B. 2022;12(7):2969–89. 10.1016/j.apsb.2022.03.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blass E, Ott PA. Advances in the development of personalized neoantigen-based therapeutic cancer vaccines. Nat Rev Clin Oncol. 2021;18(4):215–29. 10.1038/s41571-020-00460-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cafri G, Gartner JJ, Zaks T, Hopson K, Levin N, Paria BC, Parkhurst MR, Yossef R, Lowery FJ, Jafferji MS, et al. mRNA vaccine-induced neoantigen-specific T cell immunity in patients with gastrointestinal cancer. J Clin Invest. 2020;130(11):5976–88. 10.1172/JCI134915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu X, Wang X, Liao YP, Luo L, Xia T, Nel AE. Use of a Liver-Targeting Immune-Tolerogenic mRNA Lipid Nanoparticle Platform to Treat Peanut-Induced Anaphylaxis by Single- and Multiple-Epitope Nucleotide Sequence Delivery. ACS Nano. 2023;17(5):4942–57. 10.1021/acsnano.2c12420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Qiu K, Duan X, Mao M, Song Y, Rao Y, Cheng D, Feng L, Shao X, Jiang C, Huang H, et al. mRNA-LNP vaccination-based immunotherapy augments CD8(+) T cell responses against HPV-positive oropharyngeal cancer. NPJ Vaccines. 2023;8(1):144. 10.1038/s41541-023-00733-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fang E, Liu X, Li M, Zhang Z, Song L, Zhu B, Wu X, Liu J, Zhao D, Li Y. Advances in COVID-19 mRNA vaccine development. Signal Transduct Target Ther. 2022;7(1):94. 10.1038/s41392-022-00950-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mu Z, Haynes BF, Cain DW. HIV mRNA Vaccines-progress and future paths. Vaccines (Basel). 2021;9(2):134. 10.3390/vaccines9020134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mendez-Gomez HR, DeVries A, Castillo P, von Roemeling C, Qdaisat S, Stover BD, Xie C, Weidert F, Zhao C, Moor R, et al. RNA aggregates harness the danger response for potent cancer immunotherapy. Cell. 2024;187(10):2521–2535.e2521. 10.1016/j.cell.2024.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lorentzen CL, Haanen JB, Met Ö, Svane IM. Clinical advances and ongoing trials on mRNA vaccines for cancer treatment. Lancet Oncol. 2022;23(10):e450–8. 10.1016/S1470-2045(22)00372-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abbasi S, Matsui-Masai M, Yasui F, Hayashi A, Tockary TA, Mochida Y, Akinaga S, Kohara M, Kataoka K, Uchida S. Carrier-free mRNA vaccine induces robust immunity against SARS-CoV-2 in mice and non-human primates without systemic reactogenicity. Mol Ther. 2024;32(5):1266–83. 10.1016/j.ymthe.2024.03.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wadhwa A, Aljabbari A, Lokras A, Foged C, Thakur A. Opportunities and challenges in the delivery of mRNA-based vaccines. Pharmaceutics. 2020;12(2):102. 10.3390/pharmaceutics12020102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu Y, Li Y, Hu Q. Advances in saRNA vaccine research against emerging/re-emerging viruses. Vaccines (Basel). 2023;11(7):1142. 10.3390/vaccines11071142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.De A, Ko YT. Why mRNA-ionizable LNPs formulations are so short-lived: causes and way-out. Expert Opin Drug Deliv. 2023;20(2):175–87. 10.1080/17425247.2023.2162876 [DOI] [PubMed] [Google Scholar]
- 37.Dousis A, Ravichandran K, Hobert EM, Moore MJ, Rabideau AE. An engineered T7 RNA polymerase that produces mRNA free of immunostimulatory byproducts. Nat Biotechnol. 2023;41(4):560–8. 10.1038/s41587-022-01525-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Perenkov AD, Sergeeva AD, Vedunova MV, Krysko DV. In vitro transcribed RNA-based platform vaccines: past, present, and future. Vaccines (Basel). 2023;11(10):1600. 10.3390/vaccines11101600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schlesinger D, Elsässer SJ. Revisiting sORFs: overcoming challenges to identify and characterize functional microproteins. Febs j. 2022;289(1):53–74. 10.1111/febs.15769 [DOI] [PubMed] [Google Scholar]
- 40.Imani S, Tagit O, Pichon C. Neoantigen vaccine nanoformulations based on Chemically synthesized minimal mRNA (CmRNA): small molecules, big impact. NPJ Vaccines. 2024;9(1):14. 10.1038/s41541-024-00807-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Javidanbardan A, Messerian KO, Zydney AL. Membrane technology for the purification of RNA and DNA therapeutics. Trends Biotechnol. 2024;42(6):714–27. 10.1016/j.tibtech.2023.11.016 [DOI] [PubMed] [Google Scholar]
- 42.Piao X, Yadav V, Wang E, Chang W, Tau L, Lindenmuth BE, Wang SX. Double-stranded RNA reduction by chaotropic agents during in vitro transcription of messenger RNA. Mol Ther Nucleic Acids. 2022;29:618–24. 10.1016/j.omtn.2022.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Abdul Kareem ZG, Yasser Al-Zamily OM, Al-Khafaji NSK. Purification and characterization of α-galactosidase isolated from Klebsiella pneumoniae in the human oral cavity. Int J Biol Macromol. 2024;261(Pt 1):129550. 10.1016/j.ijbiomac.2024.129550 [DOI] [PubMed] [Google Scholar]
- 44.Lukavsky PJ, Puglisi JD. Large-scale preparation and purification of polyacrylamide-free RNA oligonucleotides. RNA. 2004;10(5):889–93. 10.1261/rna.5264804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goyon A, Tang S, Fekete S, Nguyen D, Hofmann K, Wang S, Shatz-Binder W, Fernandez KI, Hecht ES, Lauber M, et al. Separation of plasmid DNA topological forms, messenger RNA, and lipid nanoparticle aggregates using an ultrawide pore size exclusion chromatography column. Anal Chem. 2023;95(40):15017–24. 10.1021/acs.analchem.3c02944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yuan X, Wu Z, Guo J, Luo D, Li T, Cao Q, Ren X, Fang H, Xu D, Cao Y. Natural Wood-Derived Macroporous Cellulose for Highly Efficient and Ultrafast Elimination of Double-Stranded RNA from In Vitro-Transcribed mRNA. Adv Mater. 2024;36(22):e2303321. [DOI] [PubMed]
- 47.Cho E, Namgung J, Lee JS, Jang J, Jun BH, Kim DE. Mesoporous silica particle as an RNA Adsorbent for facile purification of in vitro-transcribed RNA. Int J Mol Sci. 2023;24(15):12408. 10.3390/ijms241512408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ojha M, Vogt J, Das NK, Redmond E, Singh K, Banna HA, Sadat T, Koirala D. Structure of saguaro cactus virus 3’ translational enhancer mimics 5’ cap for eIF4E binding. Proc Natl Acad Sci U S A. 2024;121(4):e2313677121. 10.1073/pnas.2313677121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang H, Rizvi SRA, Dong D, Lou J, Wang Q, Sopipong W, Su Y, Najar F, Agarwal PK, Kozielski F, et al. Emerging variants of SARS-CoV-2 NSP10 highlight strong functional conservation of its binding to two non-structural proteins, NSP14 and NSP16. Elife. 2023;12:RP87884. 10.7554/eLife.87884.3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schuberth-Wagner C, Ludwig J, Bruder AK, Herzner AM, Zillinger T, Goldeck M, Schmidt T, Schmid-Burgk JL, Kerber R, Wolter S, et al. A Conserved Histidine in the RNA Sensor RIG-I Controls Immune Tolerance to N1–2’O-Methylated Self RNA. Immunity. 2015;43(1):41–51. 10.1016/j.immuni.2015.06.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ziegenhals T, Frieling R, Wolf P, Göbel K, Koch S, Lohmann M, Baiersdörfer M, Fesser S, Sahin U, Kuhn AN. Formation of dsRNA by-products during in vitro transcription can be reduced by using low steady-state levels of UTP. Front Mol Biosci. 2023;10:1291045. 10.3389/fmolb.2023.1291045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Inagaki M, Abe N, Li Z, Nakashima Y, Acharyya S, Ogawa K, Kawaguchi D, Hiraoka H, Banno A, Meng Z, et al. Cap analogs with a hydrophobic photocleavable tag enable facile purification of fully capped mRNA with various cap structures. Nat Commun. 2023;14(1):2657. 10.1038/s41467-023-38244-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Janowski M, Andrzejewska A. The legacy of mRNA engineering: A lineup of pioneers for the Nobel Prize. Mol Ther Nucleic Acids. 2022;29:272–84. 10.1016/j.omtn.2022.07.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xiang K, Bartel DP. The molecular basis of coupling between poly(A)-tail length and translational efficiency. Elife. 2021;10:e66493. 10.7554/eLife.66493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Passmore LA, Coller J. Roles of mRNA poly(A) tails in regulation of eukaryotic gene expression. Nat Rev Mol Cell Biol. 2022;23(2):93–106. 10.1038/s41580-021-00417-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Liu MA, Zhou T, Sheets RL, Meyer H, Knezevic I. WHO informal consultation on regulatory considerations for evaluation of the quality, safety and efficacy of RNA-based prophylactic vaccines for infectious diseases, 20–22 April 2021. Emerg Microbes Infect. 2022;11(1):384–91. 10.1080/22221751.2022.2026742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mockey M, Gonçalves C, Dupuy FP, Lemoine FM, Pichon C, Midoux P. mRNA transfection of dendritic cells: synergistic effect of ARCA mRNA capping with Poly(A) chains in cis and in trans for a high protein expression level. Biochem Biophys Res Commun. 2006;340(4):1062–8. 10.1016/j.bbrc.2005.12.105 [DOI] [PubMed] [Google Scholar]
- 58.Pardi N, Parkhouse K, Kirkpatrick E, McMahon M, Zost SJ, Mui BL, Tam YK, Karikó K, Barbosa CJ, Madden TD, et al. Nucleoside-modified mRNA immunization elicits influenza virus hemagglutinin stalk-specific antibodies. Nat Commun. 2018;9(1):3361. 10.1038/s41467-018-05482-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sahin U, Karikó K, Türeci Ö. mRNA-based therapeutics–developing a new class of drugs. Nat Rev Drug Discov. 2014;13(10):759–80. 10.1038/nrd4278 [DOI] [PubMed] [Google Scholar]
- 60.Huang X, Zhang G, Tang T, Liang T. Identification of tumor antigens and immune subtypes of pancreatic adenocarcinoma for mRNA vaccine development. Mol Cancer. 2021;20(1):44. 10.1186/s12943-021-01310-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lin H, Wang K, Xiong Y, Zhou L, Yang Y, Chen S, Xu P, Zhou Y, Mao R, Lv G, et al. Identification of tumor antigens and immune subtypes of glioblastoma for mRNA vaccine development. Front Immunol. 2022;13:773264. 10.3389/fimmu.2022.773264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang S, Yang Y, Li L, Ma P, Jiang Y, Ge M, Yu Y, Huang H, Fang Y, Jiang N, et al. Identification of Tumor antigens and immune subtypes of malignant mesothelioma for mRNA vaccine development. Vaccines (Basel). 2022;10(8):1168. 10.3390/vaccines10081168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kozak M, Hu J. DNA vaccines: their formulations, engineering and delivery. Vaccines (Basel). 2024;12(1):71. 10.3390/vaccines12010071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chen W, Zhu Y, He J, Sun X. Path towards mRNA delivery for cancer immunotherapy from bench to bedside. Theranostics. 2024;14(1):96–115. 10.7150/thno.89247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shin MD, Shukla S, Chung YH, Beiss V, Chan SK, Ortega-Rivera OA, Wirth DM, Chen A, Sack M, Pokorski JK, et al. COVID-19 vaccine development and a potential nanomaterial path forward. Nat Nanotechnol. 2020;15(8):646–55. 10.1038/s41565-020-0737-y [DOI] [PubMed] [Google Scholar]
- 66.Riley RS, Kashyap MV, Billingsley MM, White B, Alameh MG, Bose SK, Zoltick PW, Li H, Zhang R, Cheng AY, et al. Ionizable lipid nanoparticles for in utero mRNA delivery. Sci Adv. 2021;7(3):eaba1028. 10.1126/sciadv.aba1028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zong Y, Lin Y, Wei T, Cheng Q. Lipid Nanoparticle (LNP) enables mRNA delivery for cancer therapy. Adv Mater. 2023;35(51):e2303261. 10.1002/adma.202303261 [DOI] [PubMed] [Google Scholar]
- 68.Miao L, Li L, Huang Y, Delcassian D, Chahal J, Han J, Shi Y, Sadtler K, Gao W, Lin J, et al. Delivery of mRNA vaccines with heterocyclic lipids increases anti-tumor efficacy by STING-mediated immune cell activation. Nat Biotechnol. 2019;37(10):1174–85. 10.1038/s41587-019-0247-3 [DOI] [PubMed] [Google Scholar]
- 69.Ball RL, Bajaj P, Whitehead KA. Achieving long-term stability of lipid nanoparticles: examining the effect of pH, temperature, and lyophilization. Int J Nanomedicine. 2017;12:305–15. 10.2147/IJN.S123062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kumar R, Santa Chalarca CF, Bockman MR, Bruggen CV, Grimme CJ, Dalal RJ, Hanson MG, Hexum JK, Reineke TM. Polymeric Delivery of Therapeutic Nucleic Acids. Chem Rev. 2021;121(18):11527–652. 10.1021/acs.chemrev.0c00997 [DOI] [PubMed] [Google Scholar]
- 71.Tenchov R, Bird R, Curtze AE, Zhou Q. Lipid nanoparticles─from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano. 2021;15(11):16982–7015. 10.1021/acsnano.1c04996 [DOI] [PubMed] [Google Scholar]
- 72.Khan OF, Kowalski PS, Doloff JC, Tsosie JK, Bakthavatchalu V, Winn CB, Haupt J, Jamiel M, Langer R, Anderson DG. Endothelial siRNA delivery in nonhuman primates using ionizable low-molecular weight polymeric nanoparticles. Sci Adv. 2018;4(6):eaar8409. 10.1126/sciadv.aar8409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ali A, Ganguillet S, Turgay Y, Keys TG, Causa E, Fradique R, Lutz-Bueno V, Chesnov S, Tan-Lin CW, Lentsch V, et al. Surface cross-linking by macromolecular tethers enhances virus-like particles’ resilience to mucosal stress factors. ACS Nano. 2024;18(4):3382–96. 10.1021/acsnano.3c10339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Comes JDG, Pijlman GP, Hick TAH. Rise of the RNA machines - self-amplification in mRNA vaccine design. Trends Biotechnol. 2023;41(11):1417–29. 10.1016/j.tibtech.2023.05.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Biddlecome A, Habte HH, McGrath KM, Sambanthamoorthy S, Wurm M, Sykora MM, Knobler CM, Lorenz IC, Lasaro M, Elbers K, et al. Delivery of self-amplifying RNA vaccines in in vitro reconstituted virus-like particles. PLoS ONE. 2019;14(6):e0215031. 10.1371/journal.pone.0215031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fuchs JD, Frank I, Elizaga ML, Allen M, Frahm N, Kochar N, Li S, Edupuganti S, Kalams SA, Tomaras GD, et al. First-in-human evaluation of the safety and immunogenicity of a recombinant vesicular stomatitis virus human immunodeficiency virus-1 gag Vaccine (HVTN 090). Open Forum Infect Dis. 2015;2(3):ofv082. 10.1093/ofid/ofv082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Shi Y, Huang J, Liu Y, Liu J, Guo X, Li J, Gong L, Zhou X, Cheng G, Qiu Y, et al. Structural and biochemical characteristics of mRNA nanoparticles determine anti-SARS-CoV-2 humoral and cellular immune responses. Sci Adv. 2022;8(47):eabo1827. 10.1126/sciadv.abo1827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bose S, Dahat Y, Kumar D, Haldar S, Das SK. A membrane targeted multifunctional cationic nanoparticle conjugated fusogenic nanoemulsion (CFusoN): induced membrane depolarization and lipid solubilization to accelerate the killing of Staphylococcus aureus. Mater Horiz. 2024;11(3):661–79. 10.1039/D3MH01102J [DOI] [PubMed] [Google Scholar]
- 79.Duan X, Zhang Y, Guo M, Fan N, Chen K, Qin S, Xiao W, Zheng Q, Huang H, Wei X, et al. Sodium alginate coating simultaneously increases the biosafety and immunotherapeutic activity of the cationic mRNA nanovaccine. Acta Pharm Sin B. 2023;13(3):942–54. 10.1016/j.apsb.2022.08.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pandey P, Kim SH, Subedi L, Mujahid K, Kim Y, Cho YC, Shim JH, Kim KT, Cho SS, Choi JU, et al. Oral lymphatic delivery of alpha-galactosylceramide and ovalbumin evokes anti-cancer immunization. J Control Release. 2023;356:507–24. 10.1016/j.jconrel.2023.03.010 [DOI] [PubMed] [Google Scholar]
- 81.Teixeira HF, Bruxel F, Fraga M, Schuh RS, Zorzi GK, Matte U, Fattal E. Cationic nanoemulsions as nucleic acids delivery systems. Int J Pharm. 2017;534(1–2):356–67. 10.1016/j.ijpharm.2017.10.030 [DOI] [PubMed] [Google Scholar]
- 82.Silva AL, Marcelino HR, Verissimo LM, Araujo IB, Agnez-Lima LF. do Egito ES: stearylamine-containing cationic nanoemulsion as a promising carrier for gene delivery. J Nanosci Nanotechnol. 2016;16(2):1339–45. 10.1166/jnn.2016.11671 [DOI] [PubMed] [Google Scholar]
- 83.Stokes A, Pion J, Binazon O, Laffont B, Bigras M, Dubois G, Blouin K, Young JK, Ringenberg MA, Ben Abdeljelil N, et al. Nonclinical safety assessment of repeated administration and biodistribution of a novel rabies self-amplifying mRNA vaccine in rats. Regul Toxicol Pharmacol. 2020;113:104648. 10.1016/j.yrtph.2020.104648 [DOI] [PubMed] [Google Scholar]
- 84.Huang J, Wang K, Wu S, Zhang J, Chen X, Lei S, Wu J, Men K, Duan X. Tumor Cell Lysate-Based Multifunctional Nanoparticles Facilitate Enhanced mRNA Delivery and Immune Stimulation for Melanoma Gene Therapy. Mol Pharm. 2024;21(1):267–82. 10.1021/acs.molpharmaceut.3c00826 [DOI] [PubMed] [Google Scholar]
- 85.Porosk L, Härk HH, Arukuusk P, Haljasorg U, Peterson P, Kurrikoff K. The development of cell-penetrating peptides for efficient and selective in vivo expression of mRNA in spleen tissue. Pharmaceutics. 2023;15(3):952. 10.3390/pharmaceutics15030952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chaudhary N, Weissman D, Whitehead KA. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov. 2021;20(11):817–38. 10.1038/s41573-021-00283-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mohamad Razif MI, Nizar N, Zainal Abidin NH, Muhammad Ali SN, Wan Zarimi WNN, Khotib J, Susanti D, Mohd Jailani MT, Taher M. Emergence of mRNA vaccines in the management of cancer. Expert Rev Vaccines. 2023;22(1):629–42. 10.1080/14760584.2023.2232450 [DOI] [PubMed] [Google Scholar]
- 88.Qiu Y, Man RCH, Liao Q, Kung KLK, Chow MYT, Lam JKW. Effective mRNA pulmonary delivery by dry powder formulation of PEGylated synthetic KL4 peptide. J Control Release. 2019;314:102–15. 10.1016/j.jconrel.2019.10.026 [DOI] [PubMed] [Google Scholar]
- 89.Khan MM, Filipczak N, Torchilin VP. Cell penetrating peptides: A versatile vector for co-delivery of drug and genes in cancer. J Control Release. 2021;330:1220–8. 10.1016/j.jconrel.2020.11.028 [DOI] [PubMed] [Google Scholar]
- 90.Fotin-Mleczek M, Duchardt KM, Lorenz C, Pfeiffer R, Ojkić-Zrna S, Probst J, Kallen KJ. Messenger RNA-based vaccines with dual activity induce balanced TLR-7 dependent adaptive immune responses and provide antitumor activity. J Immunother. 2011;34(1):1–15. 10.1097/CJI.0b013e3181f7dbe8 [DOI] [PubMed] [Google Scholar]
- 91.Chen Z, Meng C, Mai J, Liu Y, Li H, Shen H. An mRNA vaccine elicits STING-dependent antitumor immune responses. Acta Pharm Sin B. 2023;13(3):1274–86. 10.1016/j.apsb.2022.11.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Hasan M, Khatun A, Kogure K. Intradermal delivery of naked mRNA vaccines via Iontophoresis. Pharmaceutics. 2023;15(12):2678. 10.3390/pharmaceutics15122678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Stewart MP, Langer R, Jensen KF. Intracellular Delivery by Membrane Disruption: Mechanisms, Strategies, and Concepts. Chem Rev. 2018;118(16):7409–531. 10.1021/acs.chemrev.7b00678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wang Y, Zhang Z, Luo J, Han X, Wei Y, Wei X. mRNA vaccine: a potential therapeutic strategy. Mol Cancer. 2021;20(1):33. 10.1186/s12943-021-01311-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Shrestha A, Zhu Y, Ali A. High-fat diet influences dendritic cells and T-Cell infiltration in apical periodontitis in mice. J Endod. 2024;50(4):506–513.e502. 10.1016/j.joen.2024.01.015 [DOI] [PubMed] [Google Scholar]
- 96.Hiasa M, Abe M, Nakano A, Oda A, Amou H, Kido S, Takeuchi K, Kagawa K, Yata K, Hashimoto T, et al. GM-CSF and IL-4 induce dendritic cell differentiation and disrupt osteoclastogenesis through M-CSF receptor shedding by up-regulation of TNF-alpha converting enzyme (TACE). Blood. 2009;114(20):4517–26. 10.1182/blood-2009-04-215020 [DOI] [PubMed] [Google Scholar]
- 97.Kranz LM, Diken M, Haas H, Kreiter S, Loquai C, Reuter KC, Meng M, Fritz D, Vascotto F, Hefesha H, et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534(7607):396–401. 10.1038/nature18300 [DOI] [PubMed] [Google Scholar]
- 98.Ho W, Gao M, Li F, Li Z, Zhang XQ, Xu X. Next-Generation Vaccines: Nanoparticle-Mediated DNA and mRNA Delivery. Adv Healthc Mater. 2021;10(8):e2001812. 10.1002/adhm.202001812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.John R, Monpara J, Swaminathan S, Kalhapure R. Chemistry and art of developing lipid nanoparticles for biologics delivery: focus on development and scale-up. Pharmaceutics. 2024;16(1):131. 10.3390/pharmaceutics16010131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Xue Y, Zhang Y, Zhong Y, Du S, Hou X, Li W, Li H, Wang S, Wang C, Yan J, et al. LNP-RNA-engineered adipose stem cells for accelerated diabetic wound healing. Nat Commun. 2024;15(1):739. 10.1038/s41467-024-45094-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Dehghani-Ghahnaviyeh S, Smith M, Xia Y, Dousis A, Grossfield A, Sur S. Ionizable amino lipids distribution and effects on DSPC/cholesterol membranes: implications for lipid nanoparticle structure. J Phys Chem B. 2023;127(31):6928–39. 10.1021/acs.jpcb.3c01296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Patel SK, Billingsley MM, Mukalel AJ, Thatte AS, Hamilton AG, Gong N, El-Mayta R, Safford HC, Merolle M, Mitchell MJ. Bile acid-containing lipid nanoparticles enhance extrahepatic mRNA delivery. Theranostics. 2024;14(1):1–16. 10.7150/thno.89913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Thelen JL, Leite W, Urban VS, O’Neill HM, Grishaev AV, Curtis JE, Krueger S, Castellanos MM. Morphological characterization of self-amplifying mRNA lipid nanoparticles. ACS Nano. 2024;18(2):1464–76. 10.1021/acsnano.3c08014 [DOI] [PubMed] [Google Scholar]
- 104.Hassett KJ, Higgins J, Woods A, Levy B, Xia Y, Hsiao CJ, Acosta E, Almarsson Ö, Moore MJ, Brito LA. Impact of lipid nanoparticle size on mRNA vaccine immunogenicity. J Control Release. 2021;335:237–46. 10.1016/j.jconrel.2021.05.021 [DOI] [PubMed] [Google Scholar]
- 105.Chen SP, Blakney AK. Immune response to the components of lipid nanoparticles for ribonucleic acid therapeutics. Curr Opin Biotechnol. 2024;85:103049. 10.1016/j.copbio.2023.103049 [DOI] [PubMed] [Google Scholar]
- 106.Wang H, Yuan Y, Qin L, Yue M, Xue J, Cui Z, Zhan X, Gai J, Zhang X, Guan J, et al. Tunable rigidity of PLGA shell-lipid core nanoparticles for enhanced pulmonary siRNA delivery in 2D and 3D lung cancer cell models. J Control Release. 2024;366:746–60. 10.1016/j.jconrel.2024.01.029 [DOI] [PubMed] [Google Scholar]
- 107.Packer M, Gyawali D, Yerabolu R, Schariter J, White P. A novel mechanism for the loss of mRNA activity in lipid nanoparticle delivery systems. Nat Commun. 2021;12(1):6777. 10.1038/s41467-021-26926-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Voicu G, Rebleanu D, Mocanu CA, Tanko G, Droc I, Uritu CM, Pinteala M, Manduteanu I, Simionescu M, Calin M. VCAM-1 Targeted lipopolyplexes as vehicles for efficient delivery of shRNA-Runx2 to osteoblast-differentiated valvular interstitial cells; implications in calcific valve disease treatment. Int J Mol Sci. 2022;23(7):3824. 10.3390/ijms23073824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Joubert F, Munson MJ, Sabirsh A, England RM, Hemmerling M, Alexander C, Ashford MB. Precise and systematic end group chemistry modifications on PAMAM and poly(l-lysine) dendrimers to improve cytosolic delivery of mRNA. J Control Release. 2023;356:580–94. 10.1016/j.jconrel.2023.03.011 [DOI] [PubMed] [Google Scholar]
- 110.Li C, Cui X, Li Y, Guo D, He S. Identification of ferroptosis and drug resistance related hub genes to predict the prognosis in hepatocellular carcinoma. Sci Rep. 2023;13(1):8681. 10.1038/s41598-023-35796-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Liu X, Huang P, Yang R, Deng H. mRNA cancer vaccines: construction and boosting strategies. ACS Nano. 2023;17(20):19550–80. 10.1021/acsnano.3c05635 [DOI] [PubMed] [Google Scholar]
- 112.Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29(4):341–5. 10.1038/nbt.1807 [DOI] [PubMed] [Google Scholar]
- 113.Rayamajhi S, Wilson S, Aryal S, DeLong R. Biocompatible FePO(4) Nanoparticles: Drug Delivery, RNA Stabilization, and Functional Activity. Nanoscale Res Lett. 2021;16(1):169. 10.1186/s11671-021-03626-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Wang YS, Kumari M, Chen GH, Hong MH, Yuan JP, Tsai JL, Wu HC. mRNA-based vaccines and therapeutics: an in-depth survey of current and upcoming clinical applications. J Biomed Sci. 2023;30(1):84. 10.1186/s12929-023-00977-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bialkowski L, van Weijnen A, Van der Jeught K, Renmans D, Daszkiewicz L, Heirman C, Stangé G, Breckpot K, Aerts JL, Thielemans K. Intralymphatic mRNA vaccine induces CD8 T-cell responses that inhibit the growth of mucosally located tumours. Sci Rep. 2016;6:22509. 10.1038/srep22509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Sahin U, Derhovanessian E, Miller M, Kloke BP, Simon P, Löwer M, Bukur V, Tadmor AD, Luxemburger U, Schrörs B, et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature. 2017;547(7662):222–6. 10.1038/nature23003 [DOI] [PubMed] [Google Scholar]
- 117.Qin S, Tang X, Chen Y, Chen K, Fan N, Xiao W, Zheng Q, Li G, Teng Y, Wu M, et al. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Signal Transduct Target Ther. 2022;7(1):166. 10.1038/s41392-022-01007-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Giampazolias E, Schulz O, Lim KHJ, Rogers NC, Chakravarty P, Srinivasan N, Gordon O, Cardoso A, Buck MD, Poirier EZ, et al. Secreted gelsolin inhibits DNGR-1-dependent cross-presentation and cancer immunity. Cell. 2021;184(15):4016–4031.e4022. 10.1016/j.cell.2021.05.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chen J, Ye Z, Huang C, Qiu M, Song D, Li Y, Xu Q. Lipid nanoparticle-mediated lymph node-targeting delivery of mRNA cancer vaccine elicits robust CD8(+) T cell response. Proc Natl Acad Sci U S A. 2022;119(34):e2207841119. 10.1073/pnas.2207841119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.van Weverwijk A, de Visser KE. Mechanisms driving the immunoregulatory function of cancer cells. Nat Rev Cancer. 2023;23(4):193–215. 10.1038/s41568-022-00544-4 [DOI] [PubMed] [Google Scholar]
- 121.Barmada A, Klein J, Ramaswamy A, Brodsky NN, Jaycox JR, Sheikha H, Jones KM, Habet V, Campbell M, Sumida TS, et al. Cytokinopathy with aberrant cytotoxic lymphocytes and profibrotic myeloid response in SARS-CoV-2 mRNA vaccine-associated myocarditis. Sci Immunol. 2023;8(83):eadh3455. 10.1126/sciimmunol.adh3455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Heine A, Juranek S, Brossart P. Clinical and immunological effects of mRNA vaccines in malignant diseases. Mol Cancer. 2021;20(1):52. 10.1186/s12943-021-01339-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hsu VW, Yuan LC, Nuchtern JG, Lippincott-Schwartz J, Hammerling GJ, Klausner RD. A recycling pathway between the endoplasmic reticulum and the Golgi apparatus for retention of unassembled MHC class I molecules. Nature. 1991;352(6334):441–4. 10.1038/352441a0 [DOI] [PubMed] [Google Scholar]
- 124.Thwe PM, Amiel E. The role of nitric oxide in metabolic regulation of Dendritic cell immune function. Cancer Lett. 2018;412:236–42. 10.1016/j.canlet.2017.10.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jia S, Kim J, Esser-Kahn AP, Deak P. High-throughput screening identification of novel immunomodulatory combinations for the generation of tolerogenic dendritic cells. Front Med (Lausanne). 2023;10:1298424. 10.3389/fmed.2023.1298424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Li D, Zhang Y, Li S, Zheng B. A novel Toxoplasma gondii TGGT1_316290 mRNA-LNP vaccine elicits protective immune response against toxoplasmosis in mice. Front Microbiol. 2023;14:1145114. 10.3389/fmicb.2023.1145114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Aparici-Herraiz I, Sánchez-Sánchez G, Batlle C, Rehues P, López-Serrat M, Valverde-Estrella L, Lloberas J, Celada A. IRF1 is required for MDA5 (IFIH1) Induction by IFN-α, LPS, and poly(I:C) in murine macrophages. J Innate Immun. 2023;15(1):297–316. 10.1159/000527008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Xu G, Peng H, Yao R, Yang Y, Li B. TFEB and TFE3 cooperate in regulating inorganic arsenic-induced autophagy-lysosome impairment and immuno-dysfunction in primary dendritic cells. Cell Biol Toxicol. 2024;40(1):4. 10.1007/s10565-024-09841-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Wolf AJ, Reyes CN, Liang W, Becker C, Shimada K, Wheeler ML, Cho HC, Popescu NI, Coggeshall KM, Arditi M, et al. Hexokinase Is an innate immune receptor for the detection of bacterial peptidoglycan. Cell. 2016;166(3):624–36. 10.1016/j.cell.2016.05.076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Saadh MJ, Rasulova I, Khalil M, Farahim F, Sârbu I, Ciongradi CI, Omar TM, Alhili A, Jawad MJ, Hani T, et al. Natural killer cell-mediated immune surveillance in cancer: role of tumor microenvironment. Pathol Res Pract. 2024;254:155120. 10.1016/j.prp.2024.155120 [DOI] [PubMed] [Google Scholar]
- 131.Zhao D, Liu X, Shan Y, Li J, Cui W, Wang J, Jiang J, Xie Q, Zhang C, Duan C. Recognition of immune-related tumor antigens and immune subtypes for mRNA vaccine development in lung adenocarcinoma. Comput Struct Biotechnol J. 2022;20:5001–13. 10.1016/j.csbj.2022.08.066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sadeghi Rad H, Monkman J, Warkiani ME, Ladwa R, O’Byrne K, Rezaei N, Kulasinghe A. Understanding the tumor microenvironment for effective immunotherapy. Med Res Rev. 2021;41(3):1474–98. 10.1002/med.21765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Xiao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther. 2021;221:107753. 10.1016/j.pharmthera.2020.107753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.López de Andrés J, Griñán-Lisón C, Jiménez G, Marchal JA. Cancer stem cell secretome in the tumor microenvironment: a key point for an effective personalized cancer treatment. J Hematol Oncol. 2020;13(1):136. 10.1186/s13045-020-00966-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zhu KL, Su F, Yang JR, Xiao RW, Wu RY, Cao MY, Ling XL, Zhang T. TP53 to mediate immune escape in tumor microenvironment: an overview of the research progress. Mol Biol Rep. 2024;51(1):205. 10.1007/s11033-023-09097-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Hotz C, Wagenaar TR, Gieseke F, Bangari DS, Callahan M, Cao H, Diekmann J, Diken M, Grunwitz C, Hebert A, et al. Local delivery of mRNA-encoded cytokines promotes antitumor immunity and tumor eradication across multiple preclinical tumor models. Sci Transl Med. 2021;13(610):eabc7804. 10.1126/scitranslmed.abc7804 [DOI] [PubMed] [Google Scholar]
- 137.Tombácz I, Laczkó D, Shahnawaz H, Muramatsu H, Natesan A, Yadegari A, Papp TE, Alameh MG, Shuvaev V, Mui BL, et al. Highly efficient CD4+ T cell targeting and genetic recombination using engineered CD4+ cell-homing mRNA-LNPs. Mol Ther. 2021;29(11):3293–304. 10.1016/j.ymthe.2021.06.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Peña-Romero AC, Orenes-Piñero E. Dual effect of immune cells within tumour microenvironment: pro- and anti-tumour effects and their triggers. Cancers (Basel). 2022;14(7):1681. 10.3390/cancers14071681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kiousi E, Lyraraki V, Mardiki GL, Stachika N, Damianou AK, Malainou CP, Syrigos N, Gomatou G, Kotteas E. Progress and challenges of messenger RNA vaccines in the therapeutics of NSCLC. Cancers (Basel). 2023;15(23):5589. 10.3390/cancers15235589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mudd PA, Minervina AA, Pogorelyy MV, Turner JS, Kim W, Kalaidina E, Petersen J, Schmitz AJ, Lei T, Haile A, et al. SARS-CoV-2 mRNA vaccination elicits a robust and persistent T follicular helper cell response in humans. Cell. 2022;185(4):603–613.e615. 10.1016/j.cell.2021.12.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zhang H, You X, Wang X, Cui L, Wang Z, Xu F, Li M, Yang Z, Liu J, Huang P, et al. Delivery of mRNA vaccine with a lipid-like material potentiates antitumor efficacy through Toll-like receptor 4 signaling. Proc Natl Acad Sci U S A. 2021;118(6):e2005191118. 10.1073/pnas.2005191118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Luo W, Adamska JZ, Li C, Verma R, Liu Q, Hagan T, Wimmers F, Gupta S, Feng Y, Jiang W, et al. SREBP signaling is essential for effective B cell responses. Nat Immunol. 2023;24(2):337–48. 10.1038/s41590-022-01376-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Rojas LA, Sethna Z, Soares KC, Olcese C, Pang N, Patterson E, Lihm J, Ceglia N, Guasp P, Chu A, et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature. 2023;618(7963):144–50. 10.1038/s41586-023-06063-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16(5):275–87. 10.1038/nrc.2016.36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Barbier AJ, Jiang AY, Zhang P, Wooster R, Anderson DG. The clinical progress of mRNA vaccines and immunotherapies. Nat Biotechnol. 2022;40(6):840–54. 10.1038/s41587-022-01294-2 [DOI] [PubMed] [Google Scholar]
- 146.Ma S, Li X, Mai Y, Guo J, Zuo W, Yang J. Immunotherapeutic treatment of lung cancer and bone metastasis with a mPLA/mRNA tumor vaccine. Acta Biomater. 2023;169:489–99. 10.1016/j.actbio.2023.07.059 [DOI] [PubMed] [Google Scholar]
- 147.Takata K, Chong LC, Ennishi D, Aoki T, Li MY, Thakur A, Healy S, Viganò E, Dao T, Kwon D, et al. Tumor-associated antigen PRAME exhibits dualistic functions that are targetable in diffuse large B cell lymphoma. J Clin Invest. 2022;132(10):e145343. 10.1172/JCI145343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Bhagat A, Lyerly HK, Morse MA, Hartman ZC. CEA vaccines hum vaccin immunother. 2023;19(3):2291857. 10.1080/21645515.2023.2291857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lezcano C, Jungbluth AA, Busam KJ. PRAME Immunohistochemistry as an Ancillary Test for the Assessment of Melanocytic Lesions. Surg Pathol Clin. 2021;14(2):165–75. 10.1016/j.path.2021.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Smith SM, Iwenofu OH. NY-ESO-1: a promising cancer testis antigen for sarcoma immunotherapy and diagnosis. Chin Clin Oncol. 2018;7(4):44. 10.21037/cco.2018.08.11 [DOI] [PubMed] [Google Scholar]
- 151.Fortner RT, Damms-Machado A, Kaaks R. Systematic review: Tumor-associated antigen autoantibodies and ovarian cancer early detection. Gynecol Oncol. 2017;147(2):465–80. 10.1016/j.ygyno.2017.07.138 [DOI] [PubMed] [Google Scholar]
- 152.Xu S, Yang K, Li R, Zhang L. mRNA Vaccine era-mechanisms, drug platform and clinical prospection. Int J Mol Sci. 2020;21(18):6582. 10.3390/ijms21186582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Liu C, Shi Q, Huang X, Koo S, Kong N, Tao W. mRNA-based cancer therapeutics. Nat Rev Cancer. 2023;23(8):526–43. 10.1038/s41568-023-00586-2 [DOI] [PubMed] [Google Scholar]
- 154.Miao L, Zhang Y, Huang L. mRNA vaccine for cancer immunotherapy. Mol Cancer. 2021;20(1):41. 10.1186/s12943-021-01335-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Wells DK, van Buuren MM, Dang KK, Hubbard-Lucey VM, Sheehan KCF, Campbell KM, Lamb A, Ward JP, Sidney J, Blazquez AB, et al. Key parameters of tumor epitope immunogenicity revealed through a consortium approach improve neoantigen prediction. Cell. 2020;183(3):818–834.e813. 10.1016/j.cell.2020.09.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Esprit A, de Mey W, Bahadur Shahi R, Thielemans K, Franceschini L, Breckpot K. Neo-antigen mRNA vaccines. Vaccines (Basel). 2020;8(4):776. 10.3390/vaccines8040776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Akram N, Imran M, Noreen M, Ahmed F, Atif M, Fatima Z, Bilal Waqar A. Oncogenic Role of Tumor Viruses in Humans. Viral Immunol. 2017;30(1):20–7. 10.1089/vim.2016.0109 [DOI] [PubMed] [Google Scholar]
- 158.Hassan STS. Tumor viruses and their associated cancers: remain on the track with the latest advances. Viruses. 2022;14(2):262. 10.3390/v14020262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhou K, Yuzhakov O, Behloul N, Wang D, Bhagat L, Chu D, Zhang X, Cheng X, Fan L, Huang X, et al. HPV16 E6/E7 -based mRNA vaccine is therapeutic in mice bearing aggressive HPV-positive lesions. Front Immunol. 2023;14:1213285. 10.3389/fimmu.2023.1213285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Jiang Y, Han Q, Zhao H, Zhang J. The Mechanisms of HBV-Induced Hepatocellular Carcinoma. J Hepatocell Carcinoma. 2021;8:435–50. 10.2147/JHC.S307962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Zhao H, Shao X, Yu Y, Huang L, Amor NP, Guo K, Weng C, Zhao W, Yang A, Hu J, et al. A therapeutic hepatitis B mRNA vaccine with strong immunogenicity and persistent virological suppression. NPJ Vaccines. 2024;9(1):22. 10.1038/s41541-024-00813-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Haynes BF, Wiehe K, Borrow P, Saunders KO, Korber B, Wagh K, McMichael AJ, Kelsoe G, Hahn BH, Alt F, et al. Strategies for HIV-1 vaccines that induce broadly neutralizing antibodies. Nat Rev Immunol. 2023;23(3):142–58. 10.1038/s41577-022-00753-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Xie Z, Lin YC, Steichen JM, Ozorowski G, Kratochvil S, Ray R, Torres JL, Liguori A, Kalyuzhniy O, Wang X, et al. mRNA-LNP HIV-1 trimer boosters elicit precursors to broad neutralizing antibodies. Science. 2024;384(6697):eadk0582. 10.1126/science.adk0582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Coffman RL, Sher A, Seder RA. Vaccine adjuvants: putting innate immunity to work. Immunity. 2010;33(4):492–503. 10.1016/j.immuni.2010.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Karikó K, Buckstein M, Ni H, Weissman D. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity. 2005;23(2):165–75. 10.1016/j.immuni.2005.06.008 [DOI] [PubMed] [Google Scholar]
- 166.Uchida S, Yoshinaga N, Yanagihara K, Yuba E, Kataoka K, Itaka K. Designing immunostimulatory double stranded messenger RNA with maintained translational activity through hybridization with poly A sequences for effective vaccination. Biomaterials. 2018;150:162–70. 10.1016/j.biomaterials.2017.09.033 [DOI] [PubMed] [Google Scholar]
- 167.Tockary TA, Abbasi S, Matsui-Masai M, Hayashi A, Yoshinaga N, Boonstra E, Wang Z, Fukushima S, Kataoka K, Uchida S. Comb-structured mRNA vaccine tethered with short double-stranded RNA adjuvants maximizes cellular immunity for cancer treatment. Proc Natl Acad Sci U S A. 2023;120(29):e2214320120. 10.1073/pnas.2214320120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Pulendran B, S Arunachalam P, O’Hagan DT. Emerging concepts in the science of vaccine adjuvants. Nat Rev Drug Discov. 2021;20(6):454–75. 10.1038/s41573-021-00163-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Li C, Lee A, Grigoryan L, Arunachalam PS, Scott MKD, Trisal M, Wimmers F, Sanyal M, Weidenbacher PA, Feng Y, et al. Mechanisms of innate and adaptive immunity to the Pfizer-BioNTech BNT162b2 vaccine. Nat Immunol. 2022;23(4):543–55. 10.1038/s41590-022-01163-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Galon J, Bruni D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat Rev Drug Discov. 2019;18(3):197–218. 10.1038/s41573-018-0007-y [DOI] [PubMed] [Google Scholar]
- 171.Yim HJ, Jean T, Ong PY. Recent advances in immunomodulators for atopic dermatitis. Curr Opin Pediatr. 2023;35(6):671–9. 10.1097/MOP.0000000000001279 [DOI] [PubMed] [Google Scholar]
- 172.Demaria O, Cornen S, Daëron M, Morel Y, Medzhitov R, Vivier E. Harnessing innate immunity in cancer therapy. Nature. 2019;574(7776):45–56. 10.1038/s41586-019-1593-5 [DOI] [PubMed] [Google Scholar]
- 173.Aricò E, Castiello L, Capone I, Gabriele L, Belardelli F. Type I interferons and cancer: an evolving story demanding novel clinical applications. Cancers (Basel). 2019;11(12):1943. 10.3390/cancers11121943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Propper DJ, Balkwill FR. Harnessing cytokines and chemokines for cancer therapy. Nat Rev Clin Oncol. 2022;19(4):237–53. 10.1038/s41571-021-00588-9 [DOI] [PubMed] [Google Scholar]
- 175.Lai I, Swaminathan S, Baylot V, Mosley A, Dhanasekaran R, Gabay M, Felsher DW. Lipid nanoparticles that deliver IL-12 messenger RNA suppress tumorigenesis in MYC oncogene-driven hepatocellular carcinoma. J Immunother Cancer. 2018;6(1):125. 10.1186/s40425-018-0431-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Liu JQ, Zhang C, Zhang X, Yan J, Zeng C, Talebian F, Lynch K, Zhao W, Hou X, Du S, et al. Intratumoral delivery of IL-12 and IL-27 mRNA using lipid nanoparticles for cancer immunotherapy. J Control Release. 2022;345:306–13. 10.1016/j.jconrel.2022.03.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Haabeth OAW, Blake TR, McKinlay CJ, Tveita AA, Sallets A, Waymouth RM, Wender PA, Levy R. Local Delivery of Ox40l, Cd80, and Cd86 mRNA Kindles Global Anticancer Immunity. Cancer Res. 2019;79(7):1624–34. 10.1158/0008-5472.CAN-18-2867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Mai D, June CH, Sheppard NC. In vivo gene immunotherapy for cancer. Sci Transl Med. 2022;14(670):eabo3603. 10.1126/scitranslmed.abo3603 [DOI] [PubMed] [Google Scholar]
- 179.Sun H, Zhang Y, Wang G, Yang W, Xu Y. mRNA-based therapeutics in cancer treatment. Pharmaceutics. 2023;15(2):622. 10.3390/pharmaceutics15020622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Strebhardt K, Ullrich A. Paul Ehrlich’s magic bullet concept: 100 years of progress. Nat Rev Cancer. 2008;8(6):473–80. 10.1038/nrc2394 [DOI] [PubMed] [Google Scholar]
- 181.Labrijn AF, Janmaat ML, Reichert JM, Parren P. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov. 2019;18(8):585–608. 10.1038/s41573-019-0028-1 [DOI] [PubMed] [Google Scholar]
- 182.Thran M, Mukherjee J, Pönisch M, Fiedler K, Thess A, Mui BL, Hope MJ, Tam YK, Horscroft N, Heidenreich R, et al. mRNA mediates passive vaccination against infectious agents, toxins, and tumors. EMBO Mol Med. 2017;9(10):1434–47. 10.15252/emmm.201707678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Rybakova Y, Kowalski PS, Huang Y, Gonzalez JT, Heartlein MW, DeRosa F, Delcassian D, Anderson DG. mRNA Delivery for Therapeutic Anti-HER2 Antibody Expression In Vivo. Mol Ther. 2019;27(8):1415–23. 10.1016/j.ymthe.2019.05.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Wang Y, Tiruthani K, Li S, Hu M, Zhong G, Tang Y, Roy S, Zhang L, Tan J, Liao C, et al. mRNA delivery of a bispecific single-domain antibody to polarize tumor-associated macrophages and synergize immunotherapy against liver malignancies. Adv Mater. 2021;33(23):e2007603. 10.1002/adma.202007603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Klinger M, Benjamin J, Kischel R, Stienen S, Zugmaier G. Harnessing T cells to fight cancer with BiTE® antibody constructs–past developments and future directions. Immunol Rev. 2016;270(1):193–208. 10.1111/imr.12393 [DOI] [PubMed] [Google Scholar]
- 186.Stadler CR, Bähr-Mahmud H, Celik L, Hebich B, Roth AS, Roth RP, Karikó K, Türeci Ö, Sahin U. Elimination of large tumors in mice by mRNA-encoded bispecific antibodies. Nat Med. 2017;23(7):815–7. 10.1038/nm.4356 [DOI] [PubMed] [Google Scholar]
- 187.Alameh MG, Tombácz I, Bettini E, Lederer K, Sittplangkoon C, Wilmore JR, Gaudette BT, Soliman OY, Pine M, Hicks P, et al. Lipid nanoparticles enhance the efficacy of mRNA and protein subunit vaccines by inducing robust T follicular helper cell and humoral responses. Immunity. 2021;54(12):2877–2892.e2877. 10.1016/j.immuni.2021.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Han X, Alameh MG, Butowska K, Knox JJ, Lundgreen K, Ghattas M, Gong N, Xue L, Xu Y, Lavertu M, et al. Adjuvant lipidoid-substituted lipid nanoparticles augment the immunogenicity of SARS-CoV-2 mRNA vaccines. Nat Nanotechnol. 2023;18(9):1105–14. 10.1038/s41565-023-01404-4 [DOI] [PubMed] [Google Scholar]
- 189.Zhang Y, Yan J, Hou X, Wang C, Kang DD, Xue Y, Du S, Deng B, McComb DW, Liu SL, et al. STING agonist-derived LNP-mRNA vaccine enhances protective immunity against SARS-CoV-2. Nano Lett. 2023;23(7):2593–600. 10.1021/acs.nanolett.2c04883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Li W, Li Y, Li J, Meng J, Jiang Z, Yang C, Wen Y, Liu S, Cheng X, Mi S, et al. All-Trans-retinoic acid-adjuvanted mRNA vaccine induces mucosal anti-tumor immune responses for treating colorectal cancer. Adv Sci (Weinh). 2024;11(22):e2309770. 10.1002/advs.202309770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Tahtinen S, Tong AJ, Himmels P, Oh J, Paler-Martinez A, Kim L, Wichner S, Oei Y, McCarron MJ, Freund EC, et al. IL-1 and IL-1ra are key regulators of the inflammatory response to RNA vaccines. Nat Immunol. 2022;23(4):532–42. 10.1038/s41590-022-01160-y [DOI] [PubMed] [Google Scholar]
- 192.Inozume T. Adoptive cell transfer therapy for melanoma. Exp Dermatol. 2023;32(3):250–5. 10.1111/exd.14707 [DOI] [PubMed] [Google Scholar]
- 193.Guedan S, Ruella M, June CH. Emerging Cellular Therapies for Cancer. Annu Rev Immunol. 2019;37:145–71. 10.1146/annurev-immunol-042718-041407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021;11(4):69. 10.1038/s41408-021-00459-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Yang D, Duan Z, Yuan P, Ding C, Dai X, Chen G, Wu D. How does TCR-T cell therapy exhibit a superior anti-tumor efficacy. Biochem Biophys Res Commun. 2023;687:149209. 10.1016/j.bbrc.2023.149209 [DOI] [PubMed] [Google Scholar]
- 196.Baulu E, Gardet C, Chuvin N, Depil S. TCR-engineered T cell therapy in solid tumors: State of the art and perspectives. Sci Adv. 2023;9(7):eadf3700. 10.1126/sciadv.adf3700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Pang Z, Lu MM, Zhang Y, Gao Y, Bai JJ, Gu JY, Xie L, Wu WZ. Neoantigen-targeted TCR-engineered T cell immunotherapy: current advances and challenges. Biomark Res. 2023;11(1):104. 10.1186/s40364-023-00534-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Wei L, Dong C, Zhu W, Wang BZ. mRNA vaccine nanoplatforms and innate immunity. Viruses. 2024;16(1):120. 10.3390/v16010120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Yang F, Zheng X, Koh S, Lu J, Cheng J, Li P, Du C, Chen Y, Chen X, Yang L, et al. Messenger RNA electroporated hepatitis B virus (HBV) antigen-specific T cell receptor (TCR) redirected T cell therapy is well-tolerated in patients with recurrent HBV-related hepatocellular carcinoma post-liver transplantation: results from a phase I trial. Hepatol Int. 2023;17(4):850–9. 10.1007/s12072-023-10524-x [DOI] [PubMed] [Google Scholar]
- 200.Shen L, Yang J, Zuo C, Xu J, Ma L, He Q, Zhou X, Ding X, Wei L, Jiang S, et al. Circular mRNA-based TCR-T offers a safe and effective therapeutic strategy for treatment of cytomegalovirus infection. Mol Ther. 2024;32(1):168–84. 10.1016/j.ymthe.2023.11.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Nakamura N, Jo T, Arai Y, Kitawaki T, Nishikori M, Mizumoto C, Kanda J, Yamashita K, Nagao M, Takaori-Kondo A. Clinical impact of cytokine release syndrome on prolonged hematotoxicity after chimeric antigen receptor T cell therapy: KyoTox A-score, a novel prediction model. Transplant Cell Ther. 2024;30(4):404–14. 10.1016/j.jtct.2024.01.073 [DOI] [PubMed] [Google Scholar]
- 202.Tchou J, Zhao Y, Levine BL, Zhang PJ, Davis MM, Melenhorst JJ, Kulikovskaya I, Brennan AL, Liu X, Lacey SF, et al. Safety and efficacy of intratumoral injections of Chimeric Antigen Receptor (CAR) T Cells in metastatic breast cancer. Cancer Immunol Res. 2017;5(12):1152–61. 10.1158/2326-6066.CIR-17-0189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Beatty GL, O’Hara MH, Lacey SF, Torigian DA, Nazimuddin F, Chen F, Kulikovskaya IM, Soulen MC, McGarvey M, Nelson AM, et al. Activity of mesothelin-specific chimeric antigen receptor T cells against pancreatic carcinoma metastases in a phase 1 trial. Gastroenterology. 2018;155(1):29–32. 10.1053/j.gastro.2018.03.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Zhang F, Parayath NN, Ene CI, Stephan SB, Koehne AL, Coon ME, Holland EC, Stephan MT. Genetic programming of macrophages to perform anti-tumor functions using targeted mRNA nanocarriers. Nat Commun. 2019;10(1):3974. 10.1038/s41467-019-11911-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Han R, Wang Y, Lu L. Sensitizing the efficiency of ICIs by neoantigen mRNA vaccines for HCC treatment. Pharmaceutics. 2023;16(1):59. 10.3390/pharmaceutics16010059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Sahin U, Oehm P, Derhovanessian E, Jabulowsky RA, Vormehr M, Gold M, Maurus D, Schwarck-Kokarakis D, Kuhn AN, Omokoko T, et al. An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature. 2020;585(7823):107–12. 10.1038/s41586-020-2537-9 [DOI] [PubMed] [Google Scholar]
- 207.Poh A. mRNA Vaccine Slows Melanoma Recurrence. Cancer Discov. 2023;13(6):1278. [DOI] [PubMed]
- 208.Liu L, Wang Y, Miao L, Liu Q, Musetti S, Li J, Huang L. Combination Immunotherapy of MUC1 mRNA Nano-vaccine and CTLA-4 Blockade Effectively Inhibits Growth of Triple Negative Breast Cancer. Mol Ther. 2018;26(1):45–55. 10.1016/j.ymthe.2017.10.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Wei T, Li Y, Li B, Xie Q, Huang Y, Wu Z, Chen H, Meng Y, Liang L, Wang M, et al. Plasmid co-expressing siRNA-PD-1 and Endostatin carried by attenuated Salmonella enhanced the anti-melanoma effect via inhibiting the expression of PD-1 and VEGF on tumor-bearing mice. Int Immunopharmacol. 2024;127:111362. 10.1016/j.intimp.2023.111362 [DOI] [PubMed] [Google Scholar]
- 210.Wan PK, Ryan AJ, Seymour LW. Beyond cancer cells: Targeting the tumor microenvironment with gene therapy and armed oncolytic virus. Mol Ther. 2021;29(5):1668–82. 10.1016/j.ymthe.2021.04.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Gao L, Wu ZX, Assaraf YG, Chen ZS, Wang L. Overcoming anti-cancer drug resistance via restoration of tumor suppressor gene function. Drug Resist Updat. 2021;57:100770. 10.1016/j.drup.2021.100770 [DOI] [PubMed] [Google Scholar]
- 212.Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW. Cancer genome landscapes. Science. 2013;339(6127):1546–58. 10.1126/science.1235122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Islam MA, Xu Y, Tao W, Ubellacker JM, Lim M, Aum D, Lee GY, Zhou K, Zope H, Yu M, et al. Restoration of tumour-growth suppression in vivo via systemic nanoparticle-mediated delivery of PTEN mRNA. Nat Biomed Eng. 2018;2(11):850–64. 10.1038/s41551-018-0284-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Lin YX, Wang Y, Ding J, Jiang A, Wang J, Yu M, Blake S, Liu S, Bieberich CJ, Farokhzad OC, et al. Reactivation of the tumor suppressor PTEN by mRNA nanoparticles enhances antitumor immunity in preclinical models. Sci Transl Med. 2021;13(599):eaba9772. 10.1126/scitranslmed.aba9772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Rohner E, Yang R, Foo KS, Goedel A, Chien KR. Unlocking the promise of mRNA therapeutics. Nat Biotechnol. 2022;40(11):1586–600. 10.1038/s41587-022-01491-z [DOI] [PubMed] [Google Scholar]
- 216.Zhang H, Zhang L, Lin A, Xu C, Li Z, Liu K, Liu B, Ma X, Zhao F, Jiang H, et al. Algorithm for optimized mRNA design improves stability and immunogenicity. Nature. 2023;621(7978):396–403. 10.1038/s41586-023-06127-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Chehrazi-Raffle A, Budde LE, Pal SK. Boosting CAR T cells with anti-tumor mRNA vaccines. Nat Med. 2023;29(11):2711–2. 10.1038/s41591-023-02623-x [DOI] [PubMed] [Google Scholar]
- 218.Tang J, Zeng C, Cox TM, Li C, Son YM, Cheon IS, Wu Y, Behl S, Taylor JJ, Chakaraborty R, et al. Respiratory mucosal immunity against SARS-CoV-2 after mRNA vaccination. Sci Immunol. 2022;7(76):eadd4853. 10.1126/sciimmunol.add4853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Gote V, Bolla PK, Kommineni N, Butreddy A, Nukala PK, Palakurthi SS, Khan W. A comprehensive review of mRNA vaccines. Int J Mol Sci. 2023;24(3):2700. 10.3390/ijms24032700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Uddin MN, Roni MA. Challenges of Storage and stability of mRNA-based COVID-19 vaccines. Vaccines (Basel). 2021;9(9):1033. 10.3390/vaccines9091033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Mulroney TE, Pöyry T, Yam-Puc JC, Rust M, Harvey RF, Kalmar L, Horner E, Booth L, Ferreira AP, Stoneley M, et al. N(1)-methylpseudouridylation of mRNA causes +1 ribosomal frameshifting. Nature. 2024;625(7993):189–94. 10.1038/s41586-023-06800-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Ramachandran S, Satapathy SR, Dutta T. Delivery Strategies for mRNA Vaccines. Pharmaceut Med. 2022;36(1):11–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Meyer MA, Baer JM, Knolhoff BL, Nywening TM, Panni RZ, Su X, Weilbaecher KN, Hawkins WG, Ma C, Fields RC, et al. Breast and pancreatic cancer interrupt IRF8-dependent dendritic cell development to overcome immune surveillance. Nat Commun. 2018;9(1):1250. 10.1038/s41467-018-03600-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Lederer K, Bettini E, Parvathaneni K, Painter MM, Agarwal D, Lundgreen KA, Weirick M, Muralidharan K, Castaño D, Goel RR, et al. Germinal center responses to SARS-CoV-2 mRNA vaccines in healthy and immunocompromised individuals. Cell. 2022;185(6):1008–1024.e1015. 10.1016/j.cell.2022.01.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Kim SC, Sekhon SS, Shin WR, Ahn G, Cho BK, Ahn JY, Kim YH. Modifications of mRNA vaccine structural elements for improving mRNA stability and translation efficiency. Mol Cell Toxicol. 2022;18(1):1–8. 10.1007/s13273-021-00171-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Corbett KS, Edwards DK, Leist SR, Abiona OM, Boyoglu-Barnum S, Gillespie RA, Himansu S, Schäfer A, Ziwawo CT, DiPiazza AT, et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature. 2020;586(7830):567–71. 10.1038/s41586-020-2622-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Han X, Xu J, Xu Y, Alameh MG, Xue L, Gong N, El-Mayta R, Palanki R, Warzecha CC, Zhao G, et al. In situ combinatorial synthesis of degradable branched lipidoids for systemic delivery of mRNA therapeutics and gene editors. Nat Commun. 2024;15(1):1762. 10.1038/s41467-024-45537-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Huang X, Kong N, Zhang X, Cao Y, Langer R, Tao W. The landscape of mRNA nanomedicine. Nat Med. 2022;28(11):2273–87. 10.1038/s41591-022-02061-1 [DOI] [PubMed] [Google Scholar]
- 229.Li X, Qi J, Wang J, Hu W, Zhou W, Wang Y, Li T. Nanoparticle technology for mRNA: Delivery strategy, clinical application and developmental landscape. Theranostics. 2024;14(2):738–60. 10.7150/thno.84291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Miller CM, Haimov M. A simple rapid technique for vena cava clip replacement. Surg Gynecol Obstet. 1986;162(1):54–5. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.
No datasets were generated or analysed during the current study.