Abstract
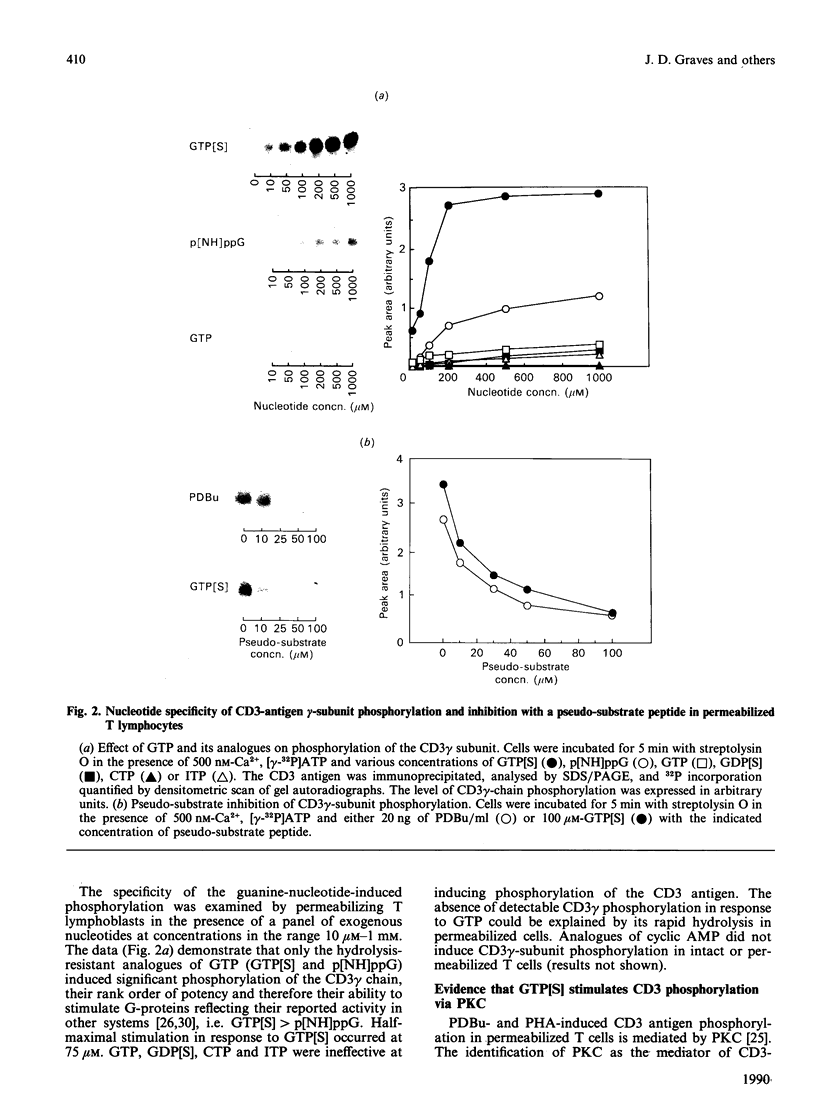
A method of membrane permeabilization of T lymphocytes with the bacterial cytotoxin streptolysin O has allowed the effect of guanine nucleotide analogues on phosphatidylinositol metabolism and protein kinase C (PKC) activation to be investigated. The data demonstrate that, in permeabilized cells, phosphorylation of the gamma subunit of the CD3 antigen can be induced in response to the PKC activator phorbol 12,13-dibutyrate, the polyclonal mitogen phytohaemagglutinin (PHA) and the stimulatory guanine nucleotide analogue guanosine 5'-[gamma-thio]triphosphate (GTP[S]). Application of a pseudo-substrate inhibitor of PKC indicated that CD3gamma-chain phosphorylation induced in response to all three agonists was mediated by PKC. PHA and GTP[S] also stimulated inositol phospholipid turnover and inositol phosphate accumulation. The kinetics and concentration-dependence of PHA-induced inositol phospholipid hydrolysis correlated with PHA-induced CD3gamma phosphorylation, suggesting that PHA may regulate CD3gamma phosphorylation via diacylglycerol produced as a consequence of inositol phospholipid hydrolysis. However, there was an inconsistency in that PHA induced greater (greater than 200%) levels of inositol phospholipid turnover than did GTP[S], but much weaker (less than 50%) levels of CD3-antigen phosphorylation. There was also a discrepancy between GTP[S] effects on phosphatidylinositol turnover and PKC activation, in that the half-maximal GTP[S] concentration for inositol phosphate production and CD3gamma-chain phosphorylation was 0.75 microM and 75 microM respectively. Moreover, 10 microM-GTP[S] induced maximal inositol phosphate production, but only 10% of maximal CD3gamma-chain phosphorylation. The data are consistent with the idea that other signal-transduction pathways, in addition to those involving inositol phosphate production, exist for the regulation of PKC in T lymphocytes.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander D. R., Hexham J. M., Lucas S. C., Graves J. D., Cantrell D. A., Crumpton M. J. A protein kinase C pseudosubstrate peptide inhibits phosphorylation of the CD3 antigen in streptolysin-O-permeabilized human T lymphocytes. Biochem J. 1989 Jun 15;260(3):893–901. doi: 10.1042/bj2600893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrowman M. M., Cockcroft S., Gomperts B. D. Two roles for guanine nucleotides in the stimulus-secretion sequence of neutrophils. Nature. 1986 Feb 6;319(6053):504–507. doi: 10.1038/319504a0. [DOI] [PubMed] [Google Scholar]
- Bell R. M. Protein kinase C activation by diacylglycerol second messengers. Cell. 1986 Jun 6;45(5):631–632. doi: 10.1016/0092-8674(86)90774-9. [DOI] [PubMed] [Google Scholar]
- Berridge M. J., Dawson R. M., Downes C. P., Heslop J. P., Irvine R. F. Changes in the levels of inositol phosphates after agonist-dependent hydrolysis of membrane phosphoinositides. Biochem J. 1983 May 15;212(2):473–482. doi: 10.1042/bj2120473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berridge M. J., Irvine R. F. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature. 1984 Nov 22;312(5992):315–321. doi: 10.1038/312315a0. [DOI] [PubMed] [Google Scholar]
- Bocckino S. B., Blackmore P. F., Wilson P. B., Exton J. H. Phosphatidate accumulation in hormone-treated hepatocytes via a phospholipase D mechanism. J Biol Chem. 1987 Nov 5;262(31):15309–15315. [PubMed] [Google Scholar]
- Burch R. M., Luini A., Axelrod J. Phospholipase A2 and phospholipase C are activated by distinct GTP-binding proteins in response to alpha 1-adrenergic stimulation in FRTL5 thyroid cells. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7201–7205. doi: 10.1073/pnas.83.19.7201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantrell D. A., Davies A. A., Crumpton M. J. Activators of protein kinase C down-regulate and phosphorylate the T3/T-cell antigen receptor complex of human T lymphocytes. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8158–8162. doi: 10.1073/pnas.82.23.8158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantrell D., Davies A. A., Londei M., Feldman M., Crumpton M. J. Association of phosphorylation of the T3 antigen with immune activation of T lymphocytes. Nature. 1987 Feb 5;325(6104):540–542. doi: 10.1038/325540a0. [DOI] [PubMed] [Google Scholar]
- Cockcroft S., Gomperts B. D. Role of guanine nucleotide binding protein in the activation of polyphosphoinositide phosphodiesterase. Nature. 1985 Apr 11;314(6011):534–536. doi: 10.1038/314534a0. [DOI] [PubMed] [Google Scholar]
- Davies A. A., Hexham J. M., Cantrell D. A., Crumpton M. J. Evidence that a GTP binding protein regulates phosphorylation of the CD3 antigen in human T lymphocytes. Biochem Biophys Res Commun. 1988 Oct 14;156(1):328–334. doi: 10.1016/s0006-291x(88)80844-1. [DOI] [PubMed] [Google Scholar]
- Friedrich B., Gullberg M. The role of protein kinase C in early activation vs. growth of T lymphocytes. Eur J Immunol. 1988 Mar;18(3):489–492. doi: 10.1002/eji.1830180327. [DOI] [PubMed] [Google Scholar]
- Gilman A. G. G proteins: transducers of receptor-generated signals. Annu Rev Biochem. 1987;56:615–649. doi: 10.1146/annurev.bi.56.070187.003151. [DOI] [PubMed] [Google Scholar]
- House C., Kemp B. E. Protein kinase C contains a pseudosubstrate prototope in its regulatory domain. Science. 1987 Dec 18;238(4834):1726–1728. doi: 10.1126/science.3686012. [DOI] [PubMed] [Google Scholar]
- Howell T. W., Gomperts B. D. Rat mast cells permeabilised with streptolysin O secrete histamine in response to Ca2+ at concentrations buffered in the micromolar range. Biochim Biophys Acta. 1987 Feb 18;927(2):177–183. doi: 10.1016/0167-4889(87)90132-7. [DOI] [PubMed] [Google Scholar]
- Irving H. R., Exton J. H. Phosphatidylcholine breakdown in rat liver plasma membranes. Roles of guanine nucleotides and P2-purinergic agonists. J Biol Chem. 1987 Mar 15;262(8):3440–3443. [PubMed] [Google Scholar]
- McPhail L. C., Clayton C. C., Snyderman R. A potential second messenger role for unsaturated fatty acids: activation of Ca2+-dependent protein kinase. Science. 1984 May 11;224(4649):622–625. doi: 10.1126/science.6231726. [DOI] [PubMed] [Google Scholar]
- Mustelin T. GTP dependence of the transduction of mitogenic signals through the T3 complex in T lymphocytes indicates the involvement of a G-protein. FEBS Lett. 1987 Mar 9;213(1):199–203. doi: 10.1016/0014-5793(87)81491-6. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. The molecular heterogeneity of protein kinase C and its implications for cellular regulation. Nature. 1988 Aug 25;334(6184):661–665. doi: 10.1038/334661a0. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. The role of protein kinase C in cell surface signal transduction and tumour promotion. Nature. 1984 Apr 19;308(5961):693–698. doi: 10.1038/308693a0. [DOI] [PubMed] [Google Scholar]
- O'Shea J. J., Urdahl K. B., Luong H. T., Chused T. M., Samelson L. E., Klausner R. D. Aluminum fluoride induces phosphatidylinositol turnover, elevation of cytoplasmic free calcium, and phosphorylation of the T cell antigen receptor in murine T cells. J Immunol. 1987 Nov 15;139(10):3463–3469. [PubMed] [Google Scholar]
- Patel M. D., Samelson L. E., Klausner R. D. Multiple kinases and signal transduction. Phosphorylation of the T cell antigen receptor complex. J Biol Chem. 1987 Apr 25;262(12):5831–5838. [PubMed] [Google Scholar]
- Rosoff P. M., Savage N., Dinarello C. A. Interleukin-1 stimulates diacylglycerol production in T lymphocytes by a novel mechanism. Cell. 1988 Jul 1;54(1):73–81. doi: 10.1016/0092-8674(88)90181-x. [DOI] [PubMed] [Google Scholar]
- Schrezenmeier H., Ahnert-Hilger G., Fleischer B. A T cell receptor-associated GTP-binding protein triggers T cell receptor-mediated granule exocytosis in cytotoxic T lymphocytes. J Immunol. 1988 Dec 1;141(11):3785–3790. [PubMed] [Google Scholar]
- Shearman M. S., Berry N., Oda T., Ase K., Kikkawa U., Nishizuka Y. Isolation of protein kinase C subspecies from a preparation of human T lymphocytes. FEBS Lett. 1988 Jul 18;234(2):387–391. doi: 10.1016/0014-5793(88)80122-4. [DOI] [PubMed] [Google Scholar]
- Truneh A., Albert F., Golstein P., Schmitt-Verhulst A. M. Early steps of lymphocyte activation bypassed by synergy between calcium ionophores and phorbol ester. Nature. 1985 Jan 24;313(6000):318–320. doi: 10.1038/313318a0. [DOI] [PubMed] [Google Scholar]
- Whetton A. D., Monk P. N., Consalvey S. D., Huang S. J., Dexter T. M., Downes C. P. Interleukin 3 stimulates proliferation via protein kinase C activation without increasing inositol lipid turnover. Proc Natl Acad Sci U S A. 1988 May;85(10):3284–3288. doi: 10.1073/pnas.85.10.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka G., Eckstein F., Stryer L. Interaction of retinal transducin with guanosine triphosphate analogues: specificity of the gamma-phosphate binding region. Biochemistry. 1986 Oct 7;25(20):6149–6153. doi: 10.1021/bi00368a048. [DOI] [PubMed] [Google Scholar]
- Zilberman Y., Howe L. R., Moore J. P., Hesketh T. R., Metcalfe J. C. Calcium regulates inositol 1,3,4,5-tetrakisphosphate production in lysed thymocytes and in intact cells stimulated with concanavalin A. EMBO J. 1987 Apr;6(4):957–962. doi: 10.1002/j.1460-2075.1987.tb04845.x. [DOI] [PMC free article] [PubMed] [Google Scholar]




