Abstract
Hyperalgesic priming, a form of pain plasticity initiated by initial injury, leads to heightened sensitivity to subsequent noxious stimuli, contributing to chronic pain development in animals. While astrocytes play active roles in modulating synaptic transmission in various pain models, their specific involvement in hyperalgesic priming remains elusive. Here, we show that spinal astrocytes are essential for hyperalgesic priming formation in a mouse model of acid-induced muscle pain. We observed spinal astrocyte activation 4 h after initial acid injection, and inhibition of this activation prevented chronic pain development upon subsequent acid injection. Chemogenetic activation of spinal astrocytes mimicked the first acid-induced hyperalgesic priming. We also demonstrated that spinal phosphorylated extracellular regulated kinase (pERK)-positive neurons were mainly vesicular glutamate transporter-2 positive (Vglut2+) neurons after the first acid injection, and inhibition of spinal pERK prevented astrocyte activation. Furthermore, pharmacological inhibition of astrocytic glutamate transporters glutamate transporter-1 and glutamate–aspartate transporter abolished the hyperalgesic priming. Collectively, our results suggest that pERK activation in Vglut2+ neurons activate astrocytes through astrocytic glutamate transporters. This process eventually establishes hyperalgesic priming through spinal D-serine. We conclude that spinal astrocytes play a crucial role in the transition from acute to chronic pain.
Keywords: chronic muscle pain, hyperalgesic priming, spinal astrocytes, D-serine
Significance Statement.
The transition from acute to chronic pain is a complex process involving various molecular and cellular mechanisms, presenting significant challenges for both patients and healthcare providers. Hyperalgesic priming defines this transition. Using intramuscular acid-induced hyperalgesic priming and chronic muscle pain model, we discover the crucial role of spinal astrocytes in priming formation. Our study indicates that the initial intramuscular acid injection activates spinal phosphorylated extracellular regulated kinase in Vglut2+ neurons, leading to spinal astrocyte activation and the initiation of the priming signal mediated by D-serine. These findings offer insights into potential therapeutic strategies for managing the transition from acute pain to chronic muscle pain.
Introduction
Chronic pain represents a prevalent clinical condition in humans, and it is impacting a significant portion of society (1, 2). Chronic pain is marked by adaptive alterations in both the peripheral and central nervous systems, these alterations are believed to directly or indirectly contribute to the persistence of chronic pain and hold potential as targets for therapeutic intervention (3–7). Although peripheral and central nervous system alterations play a role in transitioning from acute to chronic pain, the exact mechanism remains elusive (8–10). Hyperalgesic priming is a prolonged state in which animals display heightened sensitivity to acute noxious stimuli (11–14). Understanding the mechanism underlying the formation of hyperalgesic priming may provide an opportunity to treat or manage chronic pain (13, 14).
Priming can occur in the peripheral or central nervous systems (11, 15, 16). For example, activation of protein kinase C epsilon (PKCε) in peripheral neurons induces hyperalgesic priming in peripheral terminals of isolectin B4-positive nociceptors in an inflammatory pain model (17). Eliminating spinal NK1-positive neurons resulted in the complete prevention of hyperalgesic priming in IL-6 or carrageenan-induced hyperalgesic priming model (18). Spinal ERK signaling regulates spinal cord neuron plasticity, and it is crucial for the hyperalgesic priming formation in the acid-induced muscle pain (AIMP) model (12). These investigations provided evidence supporting the involvement of plasticity in both peripheral nociceptors (17, 19–22) and the spinal dorsal horn (SDH) (12, 20, 23).
In the central nervous system, astrocytes provide various metabolic support to neurons and contribute to information processing within neuronal circuits by modulating neuronal excitability and synaptic transmission (24–27). Astrocytes can be activated by multiple neurotransmitters and, in turn, secrete glial transmitters such as adenosine 5′-triphosphate (ATP), glutamate, D-serine, and γ-aminobutyric acid (GABA) (28, 29). Inhibition of astrocytes by L-Aminoadipate (LAA) or knocking down the expression of spinal glial fibrillary acidic protein (GFAP) reduces pain sensitivity in nerve-injured animals (30–32). A pinch on the hind paw or intraplantar injection of capsaicin or formalin leads to increased astrocyte activity in the SDH (30–32). Chemogenetic activation of SDH astrocytes induces mechanical hypersensitivity via a D-serine/N-methyl-D-aspartate receptors-dependent pathway (33, 34). These findings suggest that SDH astrocytes are sensitive to noxious stimuli from the periphery and play a critical role in pain modulation in different pain models (30–32).
Given the importance of astrocytes in modulating neuronal activities in various pathophysiological conditions, including chronic pain, it is still unclear whether spinal astrocytes are involved in the hyperalgesic priming and acute pain transition to chronic pain. To answer this, we investigated the role of spinal astrocytes in hyperalgesic priming using the AIMP model. We showed that the first intramuscular injection of acid saline (pH 4.0) activated spinal astrocytes in the SDH. This spinal astrocyte activation depended on the activation of ERK in Vglut2+ neurons. These activated astrocytes induced hyperalgesic priming through D-serine. Interestingly, we found that pharmacological inhibition or knockdown of astrocytic glutamate transporters glutamate transporter-1 (GLT-1) or GLAST prevented the hyperalgesic priming without affecting the acute hyperalgesia after the first acid injection. Our findings demonstrate that spinal astrocytes are involved in the development of hyperalgesic priming.
Results
Activation of spinal astrocytes contributes to the first acid inducedhyperalgesic priming
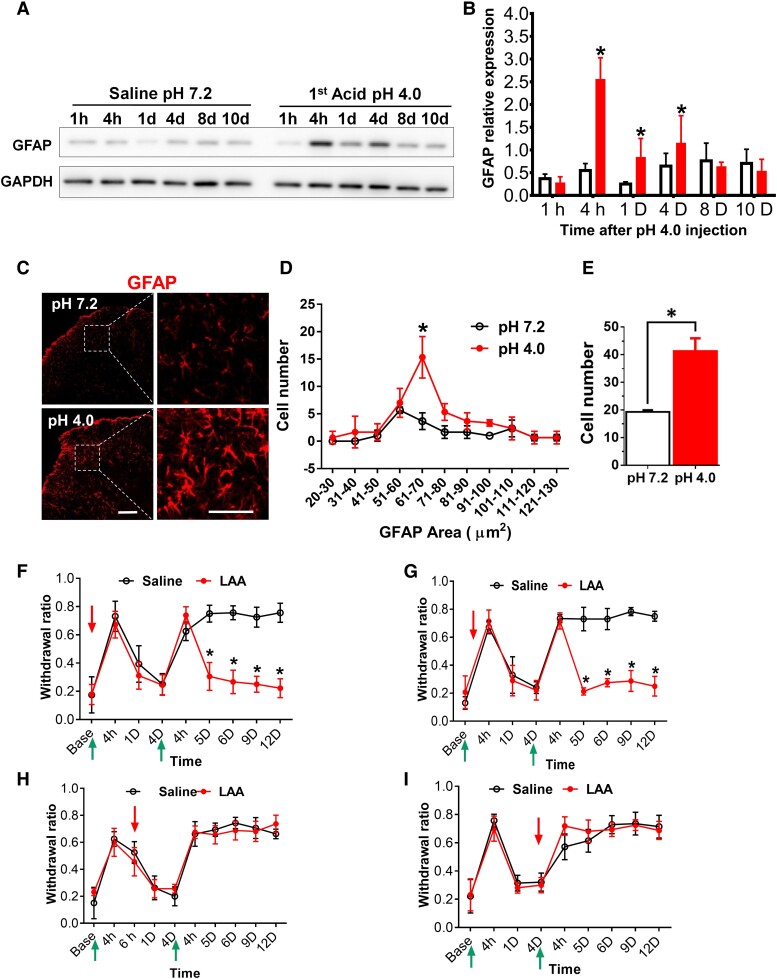
Peripheral inflammation and nerve injuries activate astrocytes in the brainstem and the spinal cord (33). GFAP is a component of astrocyte intermediate filaments. The expression of GFAP increases when astrocytes are activated; thus, increased GFAP is indicated as astrocyte activation (35, 36). To investigate whether spinal astrocytes are involved in hyperalgesic priming, we examined the expression of spinal GFAP after the first acid injection (pH 4.0) in the AIMP model. Compared to the control group (pH 7.2) saline, the protein level of GFAP increased significantly 4 h, 1 day, and 4 days after acid injection (pH 4.0) (Fig. 1A and B). The expression of GFAP decreased at 8 and 10 days in the acid-injected group and became comparable to those in the control group. Immunostaining of GFAP revealed that the activated number and size of ipsilateral SDH astrocytes increased significantly 4 h after acid injection (Fig. 1C–E). Interestingly, the results show that there are no changes in the expression of GFAP mRNA 4 h after the first acid injection (pH 4.0) compared with the saline injection (pH 7.2) (Fig. S1A). Similar increases in the GFAP staining were also observed on the contralateral side (Fig. S1B and C). We immunostained the astrocyte nuclear marker Sox9 4 h after the acid (pH 4.0) or saline (pH 7.2) injection in the SDH. Although Sox9 colocalized with GFAP, the Sox9 signals were equally expressed in both groups (Fig. S1D). These results suggest that acid injection activates spinal astrocytes. This activation of astrocytes peaks at 4 h and lasts until 4 days after acid injection.
Fig. 1.
Inhibition of spinal astrocyte activation disrupts hyperalgesic priming and prevents chronic pain. A) Western blot analysis of spinal GFAP protein at 1, 4 h, and 1, 4, 8, and 10 days after intramuscular (i.m.) first acid pH 4.0 injection. B) Quantification of GFAP protein expression levels. GFAP level at each time point was normalized to the level of GAPDH and is presented as fold changes, compared to the control group pH 7.2 saline. All data were presented as mean ± SEM (n = 4). *P < 0.05. C) Representative confocal images of GFAP in the dorsal horn 4 h after the first acid injection (n = 4), (scale bar, 100 µm). D and E) The GFAP-expressing cell number and area at 4 h after acid pH 4.0 injection, data were presented as mean ± SEM (n = 4). *P < 0.05 compared to control group saline pH 7.2. F) Mechanical threshold measurement for mice pretreated with intrathecal (i.t.) administration of 100 nmol LAA 90 min before and G) 2 h after the first acid injection compared with i.t. saline. H) i.t. LAA at 6 h after the first acid, or I) at day 4 (90 min before the second acid injection). Red arrows indicate the intrathecal injection of the drugs, and the green arrows indicate the i.m. injection of the acid pH 4.0, n = 8/group, data are presented as mean ± SEM, *P < 0.05 compared to i.t. saline pH 7.2 group.
To determine whether the acid-induced activation of astrocytes plays a role in hyperalgesic priming in the AIMP model, we intrathecally infused astrocyte inhibitor LAA (37), dose (100 nmol) 1.5 h before the first acid injection in AIMP model. We found that infusion of LAA prevented the development of chronic pain induced by the second acid injection but had no effect on the first acid-induced hyperalgesia (Fig. 1F). Since the peak of increased GFAP was at 4 h after acid injection (Fig. 1A), we also infused LAA 2 h after the first acid injection. The effect of LAA infused 2 h after acid injection was similar to the 1.5 h before acid injection (Fig. 1G). To confirm the effect of LAA on astrocyte activation, we examined the protein level and immunostaining of spinal GFAP 4 h after LAA treatment with one acid injection. LAA treatment successfully decreased the level of GFAP and decreased the number and size of activated astrocytes induced by acid injection (Fig. S2A and B). Next, we asked whether the activation of GFAP after 4 h is involved in the hyperalgesic priming. Infusion of LAA 6 h or 4 days after the first acid injection did not affect the first acid-induced hyperalgesia or the second acid-induced chronic pain (Fig. 1H and I). These results suggest that astrocyte activation at 4 h induced by acid injection is required for the formation of hyperalgesic priming in the AIMP model.
Chemogenetic activation of spinal astrocyte is sufficient to induce the hyperalgesic priming
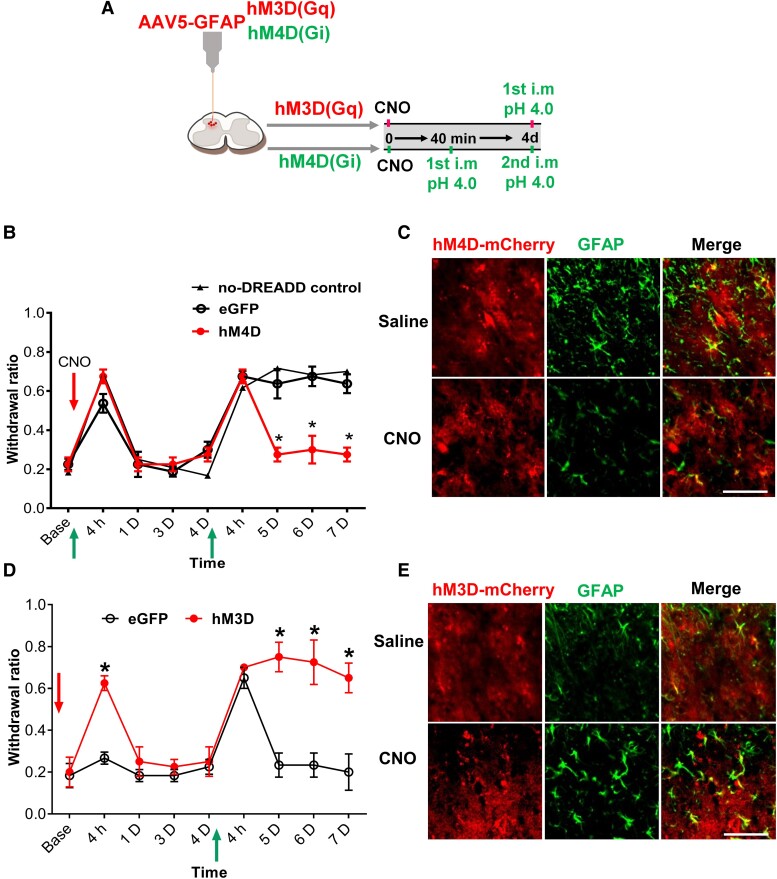
To further confirm the involvement of astrocyte activation in the priming formation, we used a designer receptor exclusively activated by designer drugs (DREADD) chemogenetic approach to inhibit or activate spinal astrocyte activity ipsilaterally by expressing hM4D(Gi) or hM3D(Gq) driven by GFAP promoter, respectively (38, 39). The experimental procedure is outlined in Fig. 2A. hM3D(Gq) or hM4D(Gi) was activated by intraperitoneal (i.p.) injection of clozapine-N-oxide (CNO) (3 mg/kg). Treatment of CNO has no effect on the first acid-induced transient hyperalgesia in the hM4D, eGFP, and no-DREADD expression control groups (Fig. 2B). However, second acid-induced chronic pain was prevented in the hM4D but not the eGFP and no-DREADD expression control groups (Fig. 2B). To confirm the inhibitory effect of hM4D on astrocytes, we examined the expression of GFAP after CNO treatment. Activation of hM4D by i.p. CNO led to a reduction of GFAP expression after the first acid injection compared with the i.p. saline group (Fig. 2C). Similar to those treated with LAA shown in Fig. 1F, these results suggest that ipsilateral chemogenetic inhibition of astrocytes before the first acid injection disrupts priming signal and thus the second acid-induced chronic pain.
Fig. 2.
Chemogenetic activation of spinal astrocytes induces hyperalgesic priming. A) Schematic diagram of the experimental procedures. B) Mechanical withdrawal measurement for hM4D group, the first acid was injected 40 min after CNO and the second acid injection was at day 4 (n = 7). C) A Representative confocal image showing spinal GFAP immunofluorescent staining in hM4D mCherry-expressing astrocytes in mice pretreated with i.p. CNO followed by first acid injection, the samples were collected 4 h after the first acid injection (n = 4). D) Mechanical withdrawal measurement of mice received i.p. CNO followed by the first acid injection at day 4 in hM3D group (n = 7). E) A representative confocal image showing spinal GFAP immunofluorescent staining in hM3D mCherry-expressing astrocytes following i.p. CNO (n = 4). Behavior data are presented as mean ± SEM, *P < 0.05 compared to the eGFP control group. *P < 0.05; red arrows indicate CNO i.p. injection, and the green arrows indicate the intramuscular injection of the acid pH 4.0 (scale bar, 100 µm).
Next, we test whether direct activation of astrocytes could mimic the effect of the first acid injection and induce hyperalgesic priming. Treatment of CNO-induced transient hyperalgesia in the hM3D group but not in the eGFP control group. Treatment of CNO-induced transient hyperalgesia in the hM3D group but not in the eGFP control group (Fig. 2D). Four days later, a single acid injection induced chronic pain in the hM3D group but transient hyperalgesia in the eGFP control group (Fig. 2D). Activation of hM3D by CNO led to an increased expression of GFAP, indicative of astrocyte activation, compared to saline group (Fig. 2E).
To examine the effect of contralateral manipulation of astrocytes on hyperalgesic priming, we expressed hM4D or hM3D on the right side of SDH (Fig. S3). Interestingly, the inhibition of contralateral astrocyte activation by hM4D did not prevent acid-induced hyperalgesic priming, as evidenced by the second acid-induced prolonged mechanical hypersensitivity (Fig. S3A). Similarly, although right-side astrocyte activation by hM3D induced transient hyperalgesia, a following left-side acid injection 4 days later did not induce prolonged mechanical hypersensitivity (Fig. S3B). These findings suggest that localized astrocyte activations can only prime neurons on the same side of the SDH.
To assure the specific expression of DREADD in astrocytes, we immunostained the neuronal (NeuN), astrocytic (GFAP), and microglial (Iba1) markers in the hM3D-mCherry-expressing SDH and quantified their colocalization. Most of the mCherry signals are colocalized with GFAP signals, with very few mCherry signals colocalized with NeuN or Iba1 signals (Fig. S4A and B).
Spinal phosphorylated extracellular regulated kinase inhibition reduces GFAP expression at 4 h after the first acid injection
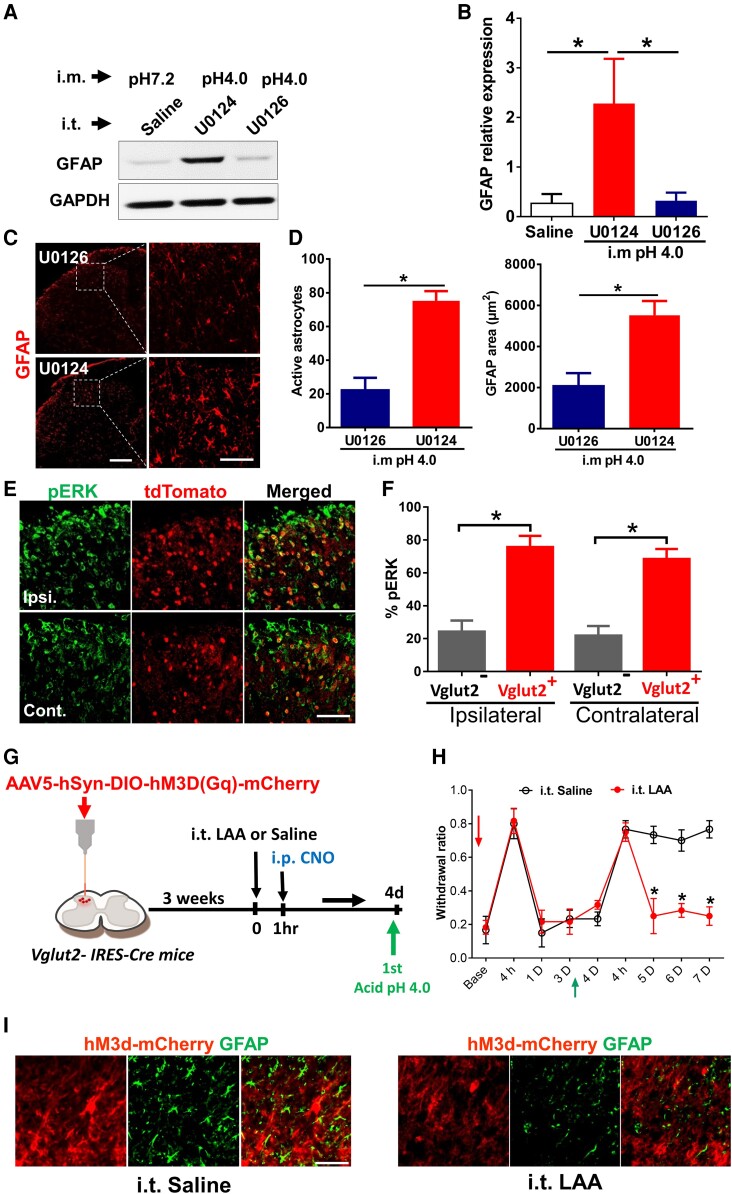
We previously demonstrated that spinal neuronal activities, characterized by increased phosphorylated extracellular regulated kinase (pERK) signals, play an important role in the first acid-induced hyperalgesic priming (12). Since astrocytes can be activated by neuronal activities (24, 26, 40), we asked whether spinal astrocytes are also activated by neurons after the first acid injection. To test this, we infused U0126 intrathecally, a mitogen-activated protein kinase1/2 inhibitor, to inhibit pERK signals. U0126, but not its inactive analog U0124, blocked the increased protein level of GFAP induced by acid injection (Fig. 3A and B). U0126 but not U0124 significantly reduced the size and number of GFAP+ astrocytes (Fig. 3C and D). These results showed that inhibition of pERK signals prevented the activation of astrocytes induced by acid injection, which implies neuronal activation is required for the astrocyte activation during hyperalgesic priming.
Fig. 3.
Activation of spinal pERK-Vglut2+ neurons is required for astrocyte activation during hyperalgesic priming. A) Representative western blots showing the detection of GFAP protein after the first acid injection pretreated with i.t. ERK inhibitor U0126, compared with the control group pretreated with i.t. U0124 and third group is i.m. saline pH 7.2 pretreated with i.t. saline. B) Quantification of GFAP protein expression levels. GFAP level was normalized to the level of GAPDH; all data were presented as mean ± SEM (n = 3). *P < 0.05. C) Representative confocal images of GFAP immunofluorescent staining in the lumbar spinal cord 4 h after the first acid pretreated with i.t. U0126 vs. i.t. U0124 and D) Quantification of GFAP positive cell number (Left panel), and area in square micrometer (right panel). n = 4. E) Representative confocal images of bilateral pERK immunofluorescent staining in a transverse section of the spinal cord of Vglut2-Cre::Ai14 mice, pERK positive signals (green) are located in tdTomato (Red). F) Quantification indicating the percentage of pERK positive neurons in Vglut2 glutamatergic neurons in both ipsilateral (Ipsi.) and contralateral (Cont.) sides to acid injection at 4 h; data were presented as mean ± SEM. *P < 0.05. (n = 4). G) A diagram illustrating the spinal injection of AAV5-hSyn-DIO-hM3D(Gq)-mCherry virus in Vglut2 − IRES-Cre mice, at day 4 after CNO; the first acid was injected into two groups pretreated with i.t. LAA or i.t. saline. H) Mechanical response measurements following administration of CNO in both the i.t. LAA and saline groups. I) Representative confocal images of spinal GFAP after i.p. CNO injection in i.t. LAA group after first acid injection in comparison with i.t. saline group. Red arrow indicates CNO and the green arrow indicates the intramuscular injection of the acid pH 4.0 (scale bar, 100 µm).
Activation of spinal Vglut2+ excitatory neurons induces pain-like behaviors (41–43). Vglut2+ excitatory neurons are expressed mainly at the laminae II/III of the SDH (42). To test whether the pERK+ neurons involved in the hyperalgesic priming are Vglut2+ neurons, we examined the expression of pERK in Vglut2-Cre::Ai14 reporter mice after one acid injection. We found that pERK+ neurons were mainly colocalized with Vglut2+ neurons 2 h after acid injection on both sides of SDH (Fig. 3E and F). To test whether activation of Vglut2+ neurons leads to astrocyte activation, we expressed hM3D specifically in Vglut2+ neurons by intraspinal injection of AAV5-hSyn-DIO-hM3D(Gq)-mCherry in Vglut2 − IRES-Cre mice. The experimental procedure is outlined in Fig. 3G, and the infection numbers of neurons are confirmed in Fig. S5. Chemogenetic activation of Vglut2+ neurons induced transient hyperalgesia and a single acid injection 4 days later induced chronic pain (Fig. 3H, i.t. saline group). Activation of Vglut2+ neurons also increased the size and number of GFAP + astrocytes (Fig. 3I). These results suggest that activation of Vglut2+ neurons mimics the effect of the first acid injection and is sufficient to induce hyperalgesic priming. We then test whether activation of astrocytes following Vglut2+ neurons is required for hyperalgesic priming. LAA treatment before i.p. injection of CNO had no effect on the transient hyperalgesia induced by activating Vglut2+ neurons (Fig. 3H, i.t. LAA group). However, LAA treatment prevented the activated Vglut2+ neurons-induced hyperalgesic priming as indicated by the prevention of single acid-induced chronic pain (Fig. 3H, i.t. LAA group) and reduced GFAP + astrocytes (Fig. 3I). These results suggest that acid injection induces activation of spinal Vglut2+ neurons, subsequently activating astrocytes to initiate hyperalgesic priming.
Inhibition or knockdown of spinal astrocytic glutamate transporters blunts the hyperalgesic priming after the first acid injection
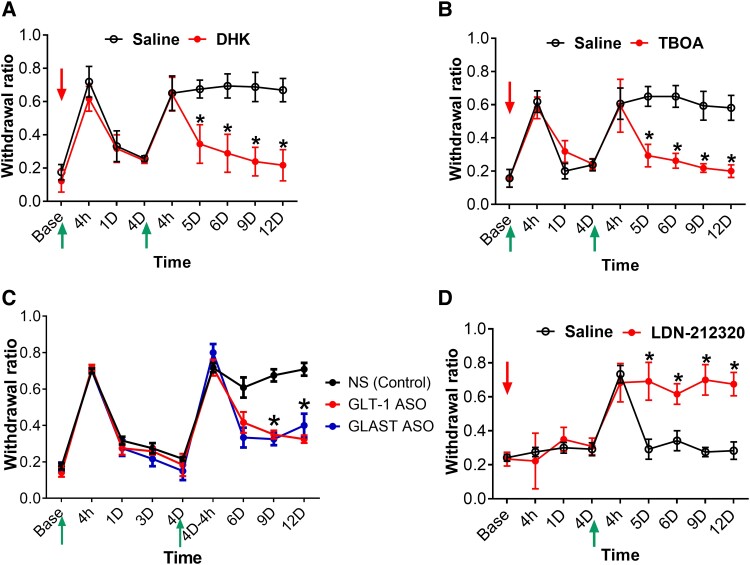
Glutamate is the main neurotransmitter in the spinal Vglut2+ neurons (43, 44), contributing to nociceptive transmission in different pain models (41, 42, 45). We hypothesized that first acid injection triggers glutamate released from Vglut2+ neurons and the released glutamate activates astrocytes. Since astrocytes uptake glutamate and control extracellular glutamate concentration via specific astrocytic glutamate transporters, glutamate transporters GLT-1, and glutamate–aspartate transporter (GLAST) (46–48), we tested whether inhibition of glutamate uptake by GLT-1 and GLAST could prevent acid-induced hyperalgesic priming. Inhibition of GLT-1 or GLAST using 1 μg threo-β-benzyloxyaspartic acid (TBOA) or dihydrokainate (DHK), respectively (49), prevented the second acid-induced chronic pain but did not affect the first acid-induced transient hyperalgesia (Fig. 4A and B). To ensure the inhibition of GLT-1 or GLAST prevents the activation of astrocytes induced by acid injection, we examined the expression of GFAP after TBOA or DHK treatment. TBOA or DHK treatment reduced the size and number of GFAP+ astrocytes induced by acid injection (Fig. S6). In addition to the pharmacological approach, we also used antisense oligonucleotides (ASOs) to down-regulate the expression of GLT-1 and GLAST. Specific ASO targeting GLT-1 and GLAST were i.t. injected 7 days before the first acid injection. Treatments of GLT-1 ASO or GLAST ASO also prevented the development of second acid-induced chronic pain (Fig. 4C). These results suggest that spinal astrocyte glutamate transporters GLT-1 and GLAST are important for the acid-induced hyperalgesic priming. The mRNA knockdown efficiency of GLAST-ASO and GLT-1-ASO was validated by qPCR, and the spinal GLAST and GLT-1 mRNA expression reduced until 1 month after intrathecal infusion of ASO to GLAST or GLT-1 separately (Fig. S7).
Fig. 4.
Inhibition or knockdown of astrocytic glutamate transporters (GLT-1 and GLAST) prevents hyperalgesic priming. A and B) Mechanical threshold measurement for the effect of i.t. (DHK and TBOA) prior to first acid pH 4.0 followed by second acid at day 4; (i.t. Dose = 1 µg/5 µL) in comparison with i.t. saline as a control group, n = 7. C) Mechanical threshold measurement after intrathecal injection of 5μg of GLT-1 or GLAST ASO 7 days before the first acid injection, the ASO-treated groups compared with i.t. ASO-NS (nonspecific control) as control n = 6. *P < 0.05 compared to i.t. ASO-NS control group. D) Mechanical threshold measurement shows the effect of i.t. GLT-1 activator LDN-212320 followed by the first acid injection at day 4, n = 6, vs. i.t. saline control group. Data were presented as mean ± SEM *P < 0.05 compared to i.t. control groups. Red arrows indicate the intrathecal injection of the drugs, and the green arrows indicate the intramuscular injection of the acid pH 4.0.
To test whether direct activation of glutamate transporter can mimic the effect of first acid injection in hyperalgesic priming, we intrathecally injected the GLT-1 activator LDN-212320 and injected acid 4 days later. Treatment of LDN-212320 did not induce hyperalgesic sensitivity, but a single acid injection 4 days later induced chronic pain compared to the saline group (Fig. 4D). Intrathecal injection of LDN-212320 increased the expression of spinal GFAP compared to the saline group (Fig. S8). These results suggest that activation of the spinal glutamate transporter activates astrocytes and thus induces hyperalgesic priming.
D-Serine plays an important role in hyperalgesic priming
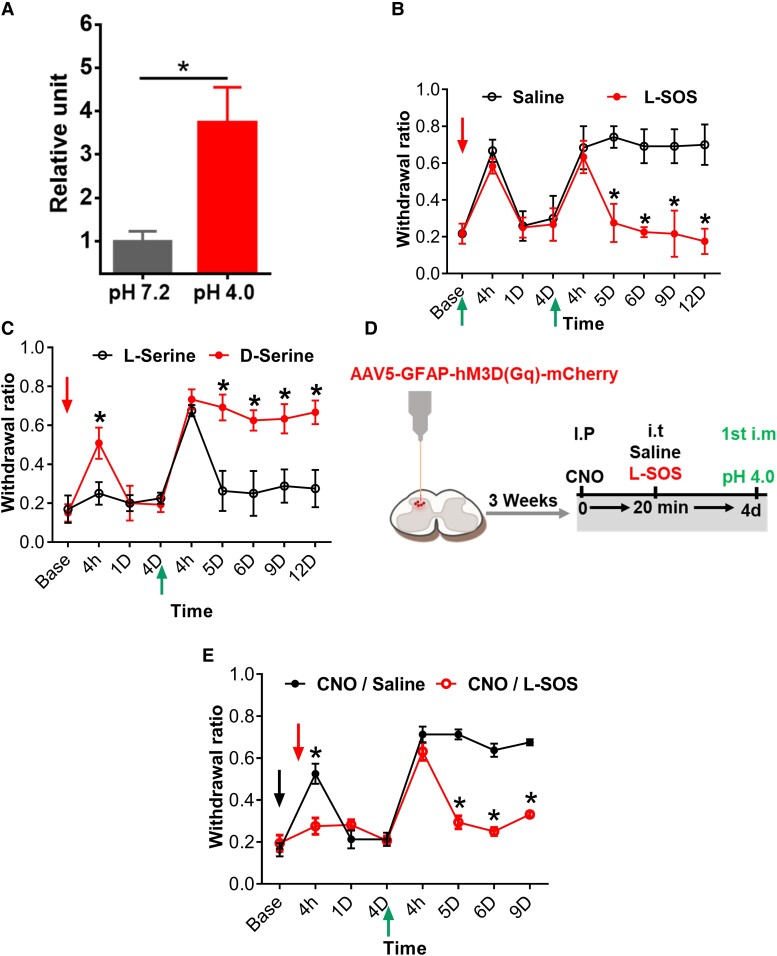
Activation of astrocytes releases gliotransmitters such as ATP, glutamate, D-serine, and GABA in response to various neurotransmitters (50, 51). These gliotransmitters subsequently engage with presynaptic and postsynaptic receptors to regulate synaptic transmission (28, 29). Direct i.t. administration of D-serine induces mechanical hypersensitivity in naive mice, and this hypersensitivity depends on the endogenous levels and activities of NMDA glutamate receptors in the spinal cord (52–54). Thus, we measured and found that the level of spinal cord D-amino acid increased significantly 4 h after acid injection compared to the saline group (Fig. 5A). D-Serine is abundant D-amino acid and is converted from L-serine by serine racemase (55). To test whether D-serine is involved in the hyperalgesia priming, we inhibited serine racemase using L-serine O-sulfate potassium salt (L-SOS) (56). L-SOS (10 nmol) had no effect on the transient hyperalgesia induced by the first acid injection (Fig. 5B); however, it prevented the second acid-induced chronic pain (Fig. 5B). We also tested whether D-serine can mimic the effect of first acid injection in hyperalgesic priming. Intrathecal injection of D-serine (10 μg) but not L-serine induced transient hyperalgesia. However, a single acid injection 4 days later induced chronic pain in the D-serine but not the L-serine group (Fig. 5C).
Fig. 5.
D-serine is the potential gliotransmitter responsible for the hyperalgesic priming. A) Measurement of spinal D-amino acid level at 4 h after the first acid injection in compared with i.m. pH 7.2 saline, n = 3. B) Mechanical threshold measurement after 10 nmol L-SOS (serine racemase inhibitor) before the first acid pH 4.0 injection, and C) after the i.t. injection of endogenous D-serine followed by i.m. first acid injection 4 days after in compared with L-serine as control, n = 7, D) Experimental scheme showing spinal injection of AAV5-GFAP-hM3d(Gq)-mCherry and experiment procedures, the first acid pH 4.0 has been injected at day 4 after CNO injection into two groups pretreated with i.t. L-SOS or i.t. saline. E) Bilateral mechanical test, chemogenetic activation of astrocytes with CNO in i.t. L-SOS group in comparison with the i.t. saline group, n = 7. Data were presented as mean ± SEM *P < 0.05. The red arrows indicate the intrathecal injection, and the black arrow for the CNO injection and the green arrows indicate the acid injection.
To confirm that D-serine is necessary for the establishment of hyperalgesic priming triggered by astrocyte activation, we tested the effect of L-SOS after chemogenetic activation of astrocytes on hyperalgesic priming. The experimental procedure is outlined in Fig. 5D. L-SOS treatment prevented the hyperalgesic priming induced by hM3D-mediated activation of astrocytes as evidenced by the lack of chronic pain induced by a single acid injection 4 days later compared to the saline group (Fig. 5E). These results implied that D-serine is most likely required for hyperalgesic priming after chemogenetic activation of astrocytes.
Discussion
The current study demonstrates that spinal astrocyte activation plays a pivotal role in the hyperalgesic priming in the AIMP model. The primary conclusions of this study are as follows: (i) increased spinal GFAP expression peaked at 4 h after the first acid injection and lasted only until 4 days; (ii) the activation of the spinal astrocytes at 4 h after the first acid injection is required for the establishment of hyperalgesic priming but not essential for the transient hyperalgesia; (iii) the activation of SDH Vglut2+ neurons is required for the activation of astrocytes induced by the first acid injection; (iv) the activation of astrocytes is mediated by astrocytic glutamate transporters (GLT-1 or/and GLAST); (v) direct activation of ipsilateral astrocytes using chemogenetic tools induces transient hyperalgesia and hyperalgesic priming; and (vi) spinal D-serine is the potential transmitter for the hyperalgesic priming following the first acid or astrocyte activation. The graphical summary is presented in Fig. S9.
Activated astrocytes have been observed in response to nerve injuries like spinal nerve ligation (57–60), chronic constriction injury (59–61), and spinal cord injury (62, 63), as well as in various other pain models, including inflammatory pain models (64, 65), chemotherapy-induced pain (66, 67) and arthritis (65, 68). These activated astrocytes regulate synaptic plasticity within the SDH in pain neural circuits, suggesting their involvement in the establishment and persistence of chronic pain (69).
Here, we showed that spinal GFAP transiently increased at 4 h after the first acid injection; additionally, using pharmacological and chemogenetic approaches, we demonstrated that astrocyte activation in the spinal cord involved in the hyperalgesic priming initiation after the first acid injection, while not affect the transient hyperalgesia. However, chemogenetic activation of spinal astrocytes induced a transient hyperalgesia and mimicked the first acid-induced hyperalgesic priming when the following acid is injected ipsilaterally but not contralaterally. These findings suggest that localized astrocyte activation can only prime neurons on the same side of the dorsal horn. The underlying mechanism of this lateralization effect of astrocyte activation on hyperalgesic priming needs further investigation.
The reason why direct chemogenetic activation of spinal astrocytes induced transient hyperalgesia is not clear. One possibility is that direct chemogenetic activation of astrocytes via the Gq-dependent pathway may induce different downstream effects compared to those induced by acid injection (70–72). The findings are consistent with previous research that demonstrated how optogenetic activation of spinal astrocytes expressing ChR2 under the GFAP promoter can induce transient mechanical sensitivity in naïve mice (71, 73). Our chemogenetic and pharmacological findings suggest that early activation of astrocytes is required for the hyperalgesic priming; nonetheless, inhibition of astrocytes after priming formation, 4 h, 1 day, or 4 days after the first acid, did not affect the chronic muscle pain induced by the second acid. These results are consistent with our previous findings that hyperalgesic priming formation required 4 to 6 h to initiate after the first acid injection (12).
Activation of spinal astrocytes in the neuropathic pain model depends on the continuous signals from nerve injury (63, 74), resulting in increased excitatory synaptic transmission and central sensitization, ultimately leading to chronic pain (75, 76). Additional research indicates that enhanced neuronal activity results in the activation of astrocytes within the spinal cord and various regions of the brain (26, 40, 77). Astrocytic activation is delayed compared with the early response of microglia and neuronal pathways in the SDH in the neuropathic pain model (78–80). Astrocyte activation is crucial for maintaining the chronic pain states after peripheral nerve injury (62, 80). However, the underlying synaptic pathways from the peripheral fibers to astrocyte activation in the SDH in the neuropathic pain model remain unclear Following peripheral injury, astrocytes in the SDH are activated by pro-inflammatory cytokines, ATP, and signaling molecules released from damaged neurons and immune cells, resulting in elevated expression of reactive astrocyte markers and further release of inflammatory mediators (81, 82).
In the acid-induced chronic muscle pain model, one single acid injection leads to bilateral transient mechanical hypersensitivity, and a second acid injection ipsilaterally or contralaterally 2–8 days after the first acid injection leads to bilateral chronic mechanical but not thermal hypersensitivity (11, 83–86). Additionally, the acid injection produces no obvious inflammation or damage in the muscle (84, 86). Unlike the neuropathic pain model, where a continuous nociceptive signal from the injury site leads to chronic pain, the first acid injection does not produce continuous stimulation from the injection site and only induces transient mechanical hypersensitivity. Our study shows that astrocytes are activated 4 h after acid injection, and this activation requires ERK activity in Vglut2+ neurons and astrocytic glutamate transporters in the SDH (Figs. 3 and S9).
Excitation of Vglut2+ neurons increases neuronal activity and synaptic glutamate release in the SDH region (87). Extrasynaptic glutamate is cleared from the synaptic cleft by astrocytic glutamate transporters GLT-1 and additional glutamate transporter GLAST (46, 49, 88). We showed that inhibition or knockdown of GLAST or GLT-1 using specific glutamate transporters inhibitors (DHK and TBOA) or ASOs prevented the hyperalgesic priming induced by the first acid injection. DHK or TBOA also attenuated the astrocyte activation, as indicated by the decrease in spinal GFAP immunoreactivity after the first acid injection. These results suggest that astrocyte glutamate transporters are most likely responsible for the astrocyte activation induced by first acid injection via Vglut2+ neurons in the dorsal horn region.
In the neuropathic pain model, the expression of GLT-1 and GLAST in the spinal cord initially increased after nerve injury and then gradually decreased (89). Spinal administration of the glutamate transporter activator Riluzole effectively attenuated neuropathic pain in rats (90). Additionally, inhibition of spinal glutamate transporters induces spontaneous nociceptive behaviors and heightened sensitivity to thermal and mechanical pain in naïve mice (49, 91). However, inhibition of spinal GLT-1 or GLAST has been shown to reduce pain in the complete Freund's adjuvant inflammatory pain model and the formalin model (92). Knockdown of spinal GLT-1 resulted in a significant decrease in nociceptive behavior in the formalin model (92, 93). These studies suggest that astrocyte glutamate transporters are important modulators in chronic pain. Manipulation of these transporters could have positive or negative effects on chronic pain, depending on the pain models. In this study, inhibition or knockdown of astrocyte glutamate transporters prevented the astrocyte activation and thus the acid-induced hyperalgesic priming.
D-Serine, glutamate, ATP, and GABA are known gliotransmitters released by activated astrocytes (28, 29). These gliotransmitters act on neurons in seconds and minutes to regulate neuronal synaptic transmission and plasticity (28, 94). Several studies utilized pharmacological and molecular approaches to demonstrate the effect of D-serine on naïve mice. For example, i.t. injection of D-serine induced mechanical allodynia, and this nociception depends on the endogenous levels and activities of NMDA glutamate receptors in the spinal cord (52–54). D-Serine modulates PKC-dependent phosphorylation of the NMDA receptor GluN1 subunit and contributes to nociception facilitation (95). In the context of hyperalgesic priming, our study suggests that spinal D-serine levels may increase 4 h after the first acid injection.
Furthermore, reducing spinal D-serine by racemase inhibitor L-SOS blunted the hyperalgesic priming induced by the first acid injection or by chemogenetic activation of astrocytes. Besides, a direct i.t. injection of exogenous D-serine but not L-serine mimicked the first acid-induced hyperalgesic priming. In the current study, we i.t. injected 10 µg D-serine, which induced transient mechanical hyperalgesia lasting up to 4 h in C57BL/6 female mice. A previous study showed that i.t. injection of 500 nmol D-serine increased paw withdrawal frequency to mechanical stimuli lasting up to 90 min after injection in Institute of Cancer Research (ICR) male mice (54). The contradictory results could be due to different concentrations of D-serine, gender, and strain of mice used in these studies. Our results suggest that D-serine is the gliotransmitter that may be released after the first acid injection and is required for priming formation. D-Serine plays a crucial role in the induction of long-term potentiation (LTP) (96, 97). Our previous study showed that spinal LTP was enhanced by PKC activation during the priming state 4 days after acid injection, even in the absence of mechanical hyperalgesia (12). Previous studies have shown that D-serine facilitates NMDA-induced nociception, partly by modulating PKC-dependent pGluN1 expression (95, 96, 98). In conclusion, the current study discovers a distinct role of spinal astrocytes mediating hyperalgesic priming which is responsible for the transition from acute to chronic pain. Our results support the working model that the first acid injection triggers pERK signaling pathway within the Vglut2-expressing neurons in the dorsal horn, which leads to the activation of spinal astrocytes through glutamate transporters (GLAST and GLT-1). Activated astrocytes are likely to release D-serine, potentially affecting spinal neurons to activate the hyperalgesic priming signaling. Further exploration is needed to fully understand spinal D-serine's prevalence and physiological consequences in initiating hyperalgesic priming and its impact on the spinal neural network.
Materials and methods
Animals
All research was conducted at the Institute of Biomedical Sciences, Academia Sinica (Taipei, Taiwan). The experiments were approved under the protocols AS_ Institutional Animal Care and Use Committee 21-12-1772 and 24-04-2156. All the mice were kept in a specific pathogen-free habitat and ranged in age from 7 to 11 weeks. Female C57BL6 mice from (National Laboratory Animal Center, Taiwan) and Vglut2-IRES-Cre mice (Gift from Dr Jin-Chung Chen, Chang Gung University, Taiwan). Mice were roughly distributed across various experimental groups. The experimenter was not acquainted with the various pharmacological treatments before the investigation.
Acid-induced hyperalgesic priming and chronic muscle pain
Mice were subjected to anesthesia using 2% vaporized isoflurane, and 20 μL of either acidic (pH 4.0) or control saline (pH 7.2) was intramuscularly injected (84). The intramuscular injections were administered into the left gastrocnemius muscle on day 0, followed by a second injection on day 4 on the same side. Subsequently, the animals, after a 30-min acclimatization period in an acrylic chamber, received ten applications of Von Frey 1.0 g filaments (North Coast Medical, Morgan Hill, CA, United States of America), with a 30-s interval between each application, as previously described (99). The time intervals we chose were 4 h, 1, 4, 5, 6, 9, and 12 days after intramuscular acidic saline pH 4.0 or control pH 7.2 injections. Bilateral mechanical scores were assessed by testing the response count to stimulation of ten applications of Von Frey filaments.
Intrathecal administration
The drugs were administered intrathecally using a 30-gauge needle into the dorsal spinal cord (100, 101). To do this, we used a 30G needle connected to a Hamilton micro-syringe, which was carefully inserted between the L5/L6 vertebrae at the cauda equina, with confirmation of successful insertion through the dura by observing a reflexive tail flick. Subsequently, we delivered a 5 μL volume of either control saline or the drug.
We utilized LAA (Sigma) as an astroglial toxin, administered intrathecally at a concentration of 100 nmol per 5 μL of saline (0.9% NaCl). Two inhibitors of glutamate transporter, DL-TBOA and DHK (Tocris Bioscience, Ellisville, MO, United States of America) were used. Also, LDN-212320, a GLT-1 activator, was obtained from MedChem Express, NJ, United States of America. The dosages administered were determined based on previous studies (49). The serine racemase inhibitor L-SOS (Santa Cruz Biotechnology Inc., CA, United States of America), D-serine, and L-serine from Sigma were also used. All treatments were dissolved in 0.9% physiological saline. Additionally, 7 days before the first acid injection, intrathecal injections of ASO targeting mouse EAAT-1 (GLAST), EAAT-2 (GLT-1), and ASO-NS nonspecific control were performed (102, 103). The ASO drugs were dissolved in saline at a concentration of 1 μg/μL in a total volume of 5 μL. The sequences mentioned are detailed in Table 1.
Table 1.
ASOs sequences.
| Target gene | Name of ASO | Sequence of ASO |
|---|---|---|
| EAAT1 (GLAST) | EAAT1-1st | GTTGCTTTTGGTCATATTTTATCT |
| EAAT2 (GLT-1) | EAAT2-866 | CTTCCCACAAATCAAGCAGGCG |
| Not specified | ASO-NS1 | GTT TTC AAA TAC ACC TTC AT |
| Not specified | ASO-NS2 | CCT TCC CTG AAG GTT CCT CC |
Western blot
Mice given intramuscular injections of acidic saline (pH 4.0 vs. pH 7.2) were decapitated. The spinal cord's lumbar segments (L3–L5) were surgically extracted, quickly frozen in liquid nitrogen, and kept at −80°C.
The spinal cord tissues were lysed using a modified lysis buffer (20 mM Tris pH 7.5, 1% Triton-X, NaCl 150 mM, Ethylenediamine tetraacetic acid 1 mM, and Ethylene glycol tetraacetic acid 1 mM). Also, the cocktail of protease inhibitors was added during the lysis process (Roche). The crude homogenate was centrifuged for 12 min at 1,000 rpm. The supernatant was collected, and the pellets were discarded. Proteins were then extracted (40 g of protein per sample was calculated using the Bradford protein assay (Bio-Rad), After protein concentration was measured, samples were heated for 5 min at 95°C and loaded onto 10% stacking, 5% separating sodium dodecyl sulfate–polyacrylamide gels for protein separation. Protein was electrophoretically and transferred using nitrocellulose blotting membranes (GE Healthcare). All membranes were blocked for 1 h at room temperature in TBS (150 mM NaCl, 20 mM TrisCl; pH = 7.2) containing 5% skimmed milk. The membranes were then subjected to primary antibodies for an overnight duration at 4°C temperature, washed three times at room temperature, incubated for an hour at room temperature with the corresponding secondary antibody, and rinsed at least three times for 10 min each time. After the final washing, protein-antibodies were identified using an ECL chemiluminescent kit (GE Healthcare). Rabbit monoclonal anti-mouse monoclonal anti-GFAP was the primary antibody (1:500, cell signaling). Secondary antibodies with dilution were produced using goat horseradish peroxidase (HRP)-conjugated anti-mouse secondary H + L IgG (1:5,000, Cell signaling). GAPDH antibody HRP (1:10,000 Proteintech) was used for loading control. The positive pixel area of specific bands was measured using ImageJ software (ImageJ 1.45 s; National Institutes of Health, Bethesda, MD, United States of America) and normalized against the corresponding GAPDH loading control bands.
Immunofluorescent staining
For anesthesia, we utilized 2% isoflurane. Avertin (Tribromoethanol) + 2-methyl-2-butanol with tert-amyl alcohol (0.25 mg/g) (Sigma-Aldrich). Avertin was administered intraperitoneally to mice. After dissection, phosphate-buffer saline 25 mL of 100 mM (PBS, pH 7.2) was transcardially administered, then 20 mL of 4% paraformaldehyde (PFA), the spinal cord's lumbar portion (L3–L5) was placed in 4% PFA for a second night at 4°C. The tissues were then transferred to and kept at 4°C for 2 to 3 days while being treated in 30% sucrose in 100 mM PBS (pH 7.2). After being cryoprotected, the spinal cord tissues were embedded in O.C.T. (Leica-Cryo-embedding media) to be sectioned on a cryostat 20 μm in all sections. Spinal cord sections were preincubated in blocking buffer (5% bovine serum albumin with 0.1% Triton-X-100 in TBS) for 1 h at room temperature before starting the immunostaining process. The sections were then treated with anti-GFAP (GA5) Mouse mAb conjugated Alexa Fluor 555 (1:5,000; cell signaling), anti-pERK polyclonal antibody, and anti-Neun (1:500; Cell signaling). The slides were washed three times with TBS + 0.05 Tween 20 (TBSt) and then incubated with secondary antibodies overnight at 4°C (1:500; Thermo Fisher), Fluoromount (Diagnostic BioSystems, Pleasanton, CA, United States of America) was used to mount the immunolabeled sections on coated glass slides. For examination, we used an LSM700 confocal scanning laser microscopy and analyzed using (ZEN software). For the quantification of GFAP-activated cells based on intensity and area, the MetaMorph Image Analysis Software was performed using an image analyzer.
Chemogenetic manipulation and intraspinal virus injection
We administered GFAP promoter-expressing AAV-5 to spinal astrocytes, which express the inhibitory or excitatory G protein-coupled receptor hM4D(Gi) or hM3D(Gq), respectively (54) and administered the hM4D or hM3D agonist CNO(3 mg/kg, i.p.) to inhibit or activate astrocytes, respectively. We used AAVs carrying DREAD receptors includes AAV5-hM3D(Gq) or hM4D(Gi) (Gift from Bryan Roth, Addgene viral prep # 50478 and # 50479 ; http://n2t.net/addgene:50478 and http://n2t.net/addgene:50479; RRID: Addgene_50478 and Addgene_50479). For control we used AAV5.GFAP.eGFP.WPRE.hGH (Gift from James M. Wilson [Addgene viral prep # 105549-AAV5; http://n2t.net/addgene:105549]; RRID:Addgene_105549) (38, 104). The mice underwent lumber surgery as follows.
Mice lumbar surgery for intraspinal viral injection
The procedure for intraspinal viral injection was modified as described (105). The mice were under continuous isoflurane anesthesia, and breathing rates were monitored during the whole procedure. We injected an AAV5 under GFAP promoter to express the excitatory or inhibitory G protein-coupled receptor in astrocytes (AAV5-GFAP-hM3D(Gq)-mCherry) or (AAV5-GFAP-hM4D(Gi)-mCherry) or AAV5.GFAP.eGFP.WPRE.hGH using pulled glass capillary micropipette. The depth of the micropipette in the dorsal is 300 μm. Topical antibiotic ointment and an iodine disinfectant were applied to the sutured area after the closure of the incision using suture clips. The animal's general health and behavior were under control, and the wound was observed until it had completely healed.
Measurement of the spinal D-amino acid after the first acid injection
The L3–L5 lumbar spinal cord tissues were homogenized in a buffer containing 100 mM Tris–HCl pH 8.8 and 50 mM NaCl, then centrifuged at 12,000×g for 10 min. The supernatants were filtered using an Amicon Ultra Centrifugal Filter (Merk Millipore) to remove molecules larger than 3 kD. D-Amino acid levels in the filtrates were measured using DAAO as previously described (98, 106). Briefly, 10 μL of tissue filtrates or standard D-serine were mixed with HRP (3 U/mL), 0.8 μg/mL Flavin Adenine Dinucleotide, and luminol. After 20 min, the background luminescence was measured, and then 2 U/mL DAAO (Sigma-Aldrich) was added immediately before the next luminescent measurement. The peak luminescence intensity of the tissue filtrate was adjusted by subtracting the background intensity at the time point just before the addition of DAAO. The amount of D-amino acid in the spinal cord was calculated from the fitted regression line of the standard D-serine. One-way ANOVA followed by post hoc Tukey was used to test statistically significant differences.
Statistics
The number of samples (n) mentioned in the figure legend and the data are presented as mean ± SEM. The Prism 6 program from GraphPad software was used. For all data analysis, two-way ANOVA was used with Bonferroni's multiple comparison test to measure statistical differences between groups. The western blot gels were analyzed with Image J program software (NIH, United States) using the gel analysis tool.
Supplementary Material
Acknowledgments
The authors thank the Common Facilities Core Laboratory, Animal Core, the Pathology Core, and the Common Facilities Core Laboratory at the Institute of Biomedical Sciences, Academia Sinica, Taiwan. The authors also thank Dr Jin-Chung Chen, Chang Gung University, Taiwan, for sharing vglut2-Cre mouse line.
Contributor Information
Mohamed Abbas Abdelaziz, Taiwan International Graduate Program in Interdisciplinary Neuroscience, National Cheng Kung University and Academia Sinica, Taipei 11529, Taiwan; Institute of Biomedical Sciences, Academia Sinica, Taipei 11529, Taiwan; Zoology Department, Faculty of Science, Al-Azhar University Assiut Branch, Assiut 71524, Egypt.
Wei-Hsin Chen, Institute of Biomedical Sciences, Academia Sinica, Taipei 11529, Taiwan.
Yu-Wang Chang, Institute of Biomedical Sciences, Academia Sinica, Taipei 11529, Taiwan.
Selomon Assefa Mindaye, Taiwan International Graduate Program in Interdisciplinary Neuroscience, National Cheng Kung University and Academia Sinica, Taipei 11529, Taiwan; Institute of Biomedical Sciences, Academia Sinica, Taipei 11529, Taiwan.
Chien-Chang Chen, Institute of Biomedical Sciences, Academia Sinica, Taipei 11529, Taiwan.
Supplementary Material
Supplementary material is available at PNAS Nexus online.
Funding
This work was supported by Academia Sinica grants (AS-IR-(111-113)-05-A), Ministry of Science and Technology, Taiwan grants (MOST 108-2320-B-001-025-MY3 and MOST 111-2320-B-001-007-MY3) to C.-C.C. and (MOST 111-2811-B-001-009 and MOST 112-2811-B-001-038) to W.-H.C.
Author Contributions
M.A.A., W.-H. C., and C.-C.C. designed the study, interpreted the results, and wrote the manuscript. M.A.A. and Y.W. C. conducted the experiments and analyzed the data. S.A.M. contributed to behavioral experiments.
Data Availability
All relevant data have been included in the submitted materials.
References
- 1. Breivik H, Collett B, Ventafridda V, Cohen R, Gallacher D. 2006. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur J Pain. 10:287–333. [DOI] [PubMed] [Google Scholar]
- 2. Dueñas M, Ojeda B, Salazar A, Mico JA, Failde I. 2016. A review of chronic pain impact on patients, their social environment and the health care system. J Pain Res. 9:457–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gregory NS, Sluka KA. 2014. Anatomical and physiological factors contributing to chronic muscle pain. Curr Top Behav Neurosci. 20:327–348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Varrassi G, et al. 2010. Pharmacological treatment of chronic pain—the need for CHANGE. Curr Med Res Opin. 26:1231–1245. [DOI] [PubMed] [Google Scholar]
- 5. Zheng Q, Dong X, Green DP, Dong X. 2022. Peripheral mechanisms of chronic pain. Medical Review. 2:251–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ji R-R, Xu Z-Z, Gao Y-J. 2014. Emerging targets in neuroinflammation-driven chronic pain. Nat Rev Drug Discov. 13:533–548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Vincent TL. 2020. Peripheral pain mechanisms in osteoarthritis. Pain. 161:S138–S146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ji R-R, Chamessian A, Zhang Y-Q. 2016. Pain regulation by non-neuronal cells and inflammation. Science. 354:572–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Basbaum AI, Bautista DM, Scherrer G, Julius D. 2009. Cellular and molecular mechanisms of pain. Cell. 139:267–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Glare P, Aubrey KR, Myles PS. 2019. Transition from acute to chronic pain after surgery. Lancet. 393:1537–1546. [DOI] [PubMed] [Google Scholar]
- 11. Jasper LL, MacNeil BJ. 2012. Diverse sensory inputs permit priming in the acidic saline model of hyperalgesia. Eur J Pain. 16:966–973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Chen WH, Chang YT, Chen YC, Cheng SJ, Chen CC. 2018. Spinal protein kinase C/extracellular signal-regulated kinase signal pathway mediates hyperalgesia priming. Pain. 159:907–918. [DOI] [PubMed] [Google Scholar]
- 13. Wood JN, Sun W-H, Su Y-S, Chen C-C. 2020. The transition from acute to chronic pain. In: Wood JN, editors. The Oxford handbook of the neurobiology of pain. Oxford University Press. p. 678–701. doi:10.1093/oxfordhb/9780190860509.013.28. chap. 23. [Google Scholar]
- 14. Price TJ, et al. 2018. Transition to chronic pain: opportunities for novel therapeutics. Nat Rev Neurosci. 19:383–384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kandasamy R, Price TJ. 2015. The pharmacology of nociceptor priming. Handb Exp Pharmacol. 227:15–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kim JV, et al. 2016. Neuroligin 2 regulates spinal GABAergic plasticity in hyperalgesic priming, a model of the transition from acute to chronic pain. Pain. 157:1314–1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Joseph EK, Levine JD. 2010. Hyperalgesic priming is restricted to isolectin B4-positive nociceptors. Neuroscience. 169:431–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kim JY, et al. 2015. Spinal dopaminergic projections control the transition to pathological pain plasticity via a D1/D5-mediated mechanism. J Neurosci. 35:6307–6317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Asiedu MN, et al. 2011. Spinal protein kinase M ζ underlies the maintenance mechanism of persistent nociceptive sensitization. J Neurosci. 31:6646–6653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Melemedjian OK, et al. 2014. Local translation and retrograde axonal transport of CREB regulates IL-6-induced nociceptive plasticity. Mol Pain. 10:45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ferrari LF, Bogen O, Levine JD. 2013. Role of nociceptor αCaMKII in transition from acute to chronic pain (hyperalgesic priming) in male and female rats. J Neurosci. 33:11002–11011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ferrari LF, Bogen O, Levine JD. 2013. Role of nociceptor alphaCaMKII in transition from acute to chronic pain (hyperalgesic priming) in male and female rats. J Neurosci. 33:11002–11011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Corder G, et al. 2013. Constitutive μ-opioid receptor activity leads to long-term endogenous analgesia and dependence. Science. 341:1394–1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Benarroch EE. 2005. Neuron-astrocyte interactions: partnership for normal function and disease in the central nervous system. Mayo Clin Proc. 80:1326–1338. [DOI] [PubMed] [Google Scholar]
- 25. Liddelow SA, Barres BA. 2017. Reactive astrocytes: production, function, and therapeutic potential. Immunity. 46:957–967. [DOI] [PubMed] [Google Scholar]
- 26. Araque A, Carmignoto G, Haydon PG. 2001. Dynamic signaling between astrocytes and neurons. Annu Rev Physiol. 63:795–813. [DOI] [PubMed] [Google Scholar]
- 27. Araque A, Sanzgiri RP, Parpura V, Haydon PG. 1999. Astrocyte-induced modulation of synaptic transmission. Can J Physiol Pharmacol. 77:699–706. [PubMed] [Google Scholar]
- 28. Covelo A, Araque A. 2018. Neuronal activity determines distinct gliotransmitter release from a single astrocyte. Elife. 7:e32237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Araque A, et al. 2014. Gliotransmitters travel in time and space. Neuron. 81:728–739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Farrar MJ, et al. 2012. Chronic in vivo imaging in the mouse spinal cord using an implanted chamber. Nat Methods. 9:297–302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. White FA, Jung H, Miller RJ. 2007. Chemokines and the pathophysiology of neuropathic pain. Proc Natl Acad Sci U S A. 104:20151–20158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ohmichi M, et al. 2014. Activated spinal astrocytes are involved in the maintenance of chronic widespread mechanical hyperalgesia after cast immobilization. Mol Pain. 10:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kohro Y, et al. 2020. Spinal astrocytes in superficial laminae gate brainstem descending control of mechanosensory hypersensitivity. Nat Neurosci. 23:1376–1387. [DOI] [PubMed] [Google Scholar]
- 34. Neame S, et al. 2019. The NMDA receptor activation by d-serine and glycine is controlled by an astrocytic Phgdh-dependent serine shuttle. Proc Natl Acad Sci U S A. 116:20736–20742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Stephenson JL, Byers MR. 1995. GFAP immunoreactivity in trigeminal ganglion satellite cells after tooth injury in rats. Exp Neurol. 131:11–22. [DOI] [PubMed] [Google Scholar]
- 36. Eddleston M, Mucke L. 1993. Molecular profile of reactive astrocytes—implications for their role in neurologic disease. Neuroscience. 54:15–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Khurgel M, Koo AC, Ivy GO. 1996. Selective ablation of astrocytes by intracerebral injections of α-aminoadipate. Glia. 16:351–358. [DOI] [PubMed] [Google Scholar]
- 38. Kol A, et al. 2020. Astrocytes contribute to remote memory formation by modulating hippocampal–cortical communication during learning. Nat Neurosci. 23:1229–1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Miyamoto K, Ishikura Ki, Kume K, Ohsawa M. 2019. Astrocyte-neuron lactate shuttle sensitizes nociceptive transmission in the spinal cord. Glia. 67:27–36. [DOI] [PubMed] [Google Scholar]
- 40. Wang W, et al. 2009. Crosstalk between spinal astrocytes and neurons in nerve injury-induced neuropathic pain. PLoS One. 4:e6973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wang L, et al. 2018. Regulating nociceptive transmission by VGluT2-expressing spinal dorsal horn neurons. J Neurochem. 147:526–540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Scherrer G, et al. 2010. VGLUT2 expression in primary afferent neurons is essential for normal acute pain and injury-induced heat hypersensitivity. Proc Natl Acad Sci U S A. 107:22296–22301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Liu Y, et al. 2010. VGLUT2-dependent glutamate release from nociceptors is required to sense pain and suppress itch. Neuron. 68:543–556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Brumovsky P, Watanabe M, Hökfelt T. 2007. Expression of the vesicular glutamate transporters-1 and-2 in adult mouse dorsal root ganglia and spinal cord and their regulation by nerve injury. Neuroscience. 147:469–490. [DOI] [PubMed] [Google Scholar]
- 45. Lagerström MC, et al. 2011. A sensory subpopulation depends on vesicular glutamate transporter 2 for mechanical pain, and together with substance P, inflammatory pain. Proc Natl Acad Sci U S A. 108:5789–5794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Salvatore MF, Davis RW, Arnold JC, Chotibut T. 2012. Transient striatal GLT-1 blockade increases EAAC1 expression, glutamate reuptake, and decreases tyrosine hydroxylase phosphorylation at ser(19). Exp Neurol. 234:428–436. [DOI] [PubMed] [Google Scholar]
- 47. Barbour B, Brew H, Attwell D. 1988. Electrogenic glutamate uptake in glial cells is activated by intracellular potassium. Nature. 335:433–435. [DOI] [PubMed] [Google Scholar]
- 48. Chiang C-Y, et al. 2007. Astroglial glutamate–glutamine shuttle is involved in central sensitization of nociceptive neurons in rat medullary dorsal horn. J Neurosci. 27:9068–9076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Liaw W-J, et al. 2005. Spinal glutamate uptake is critical for maintaining normal sensory transmission in rat spinal cord. Pain. 115:60–70. [DOI] [PubMed] [Google Scholar]
- 50. Lee S, et al. 2010. Channel-mediated tonic GABA release from glia. Science. 330:790–796. [DOI] [PubMed] [Google Scholar]
- 51. Sahlender DA, Savtchouk I, Volterra A. 2014. What do we know about gliotransmitter release from astrocytes? Philos Trans R Soc Lond B Biol Sci. 369:20130592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Mothet J-P, et al. 2000. D-serine is an endogenous ligand for the glycine site of the N-methyl-D-aspartate receptor. Proc Natl Acad Sci U S A. 97:4926–4931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Lefèvre Y, et al. 2015. Neuropathic pain depends upon D-serine co-activation of spinal NMDA receptors in rats. Neurosci Lett. 603:42–47. [DOI] [PubMed] [Google Scholar]
- 54. Choi S-R, et al. 2019. Astrocyte D-serine modulates the activation of neuronal NOS leading to the development of mechanical allodynia in peripheral neuropathy. Mol Pain. 15:1744806919843046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Wolosker H, Blackshaw S, Snyder SH. 1999. Serine racemase: a glial enzyme synthesizing D-serine to regulate glutamate-N-methyl-D-aspartate neurotransmission. Proc Natl Acad Sci U S A. 96:13409–13414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Panizzutti R, De Miranda J, Ribeiro CS, Engelender S, Wolosker H. 2001. A new strategy to decrease N-methyl-D-aspartate (NMDA) receptor coactivation: inhibition of D-serine synthesis by converting serine racemase into an eliminase. Proc Natl Acad Sci U S A. 98:5294–5299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Chen G, Luo X, Qadri MY, Berta T, Ji R-R. 2018. Sex-dependent glial signaling in pathological pain: distinct roles of spinal microglia and astrocytes. Neurosci Bull. 34:98–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Colburn R, Rickman A, DeLeo J. 1999. The effect of site and type of nerve injury on spinal glial activation and neuropathic pain behavior. Exp Neurol. 157:289–304. [DOI] [PubMed] [Google Scholar]
- 59. Hald A, Nedergaard S, Hansen RR, Ding M, Heegaard AM. 2009. Differential activation of spinal cord glial cells in murine models of neuropathic and cancer pain. Eur J Pain. 13:138–145. [DOI] [PubMed] [Google Scholar]
- 60. Nakagawa T, Kaneko S. 2010. Spinal astrocytes as therapeutic targets for pathological pain. J Pharmacol Sci. 114:347–353. [DOI] [PubMed] [Google Scholar]
- 61. Ono T, et al. 2020. Mechanical pain of the lower extremity after compression of the upper spinal cord involves signal transducer and activator of transcription 3-dependent reactive astrocytes and interleukin-6. Brain Behav Immun. 89:389–399. [DOI] [PubMed] [Google Scholar]
- 62. Okada S, Hara M, Kobayakawa K, Matsumoto Y, Nakashima Y. 2018. Astrocyte reactivity and astrogliosis after spinal cord injury. Neurosci Res. 126:39–43. [DOI] [PubMed] [Google Scholar]
- 63. Miranpuri GS, et al. 2021. Role of microglia and astrocytes in spinal cord injury induced neuropathic pain. Ann Neurosci. 28:219–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Wei X, et al. 2020. Platelet-rich plasma improves chronic inflammatory pain by inhibiting PKM2-mediated aerobic glycolysis in astrocytes. Ann Transl Med. 8:1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Old EA, Clark AK, Malcangio M. 2015. The role of glia in the spinal cord in neuropathic and inflammatory pain. Pain Control. 227:145–170. [DOI] [PubMed] [Google Scholar]
- 66. Sheng H-Y, Zhang Y-Q. 2020. Emerging molecular targets for the management of cancer pain. Neurosci Bull. 36:1225–1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Luo H, et al. 2019. Interleukin-17 regulates neuron-glial communications, synaptic transmission, and neuropathic pain after chemotherapy. Cell Rep. 29:2384–2397.e2385. [DOI] [PubMed] [Google Scholar]
- 68. Sagar DR, et al. 2011. The contribution of spinal glial cells to chronic pain behaviour in the monosodium iodoacetate model of osteoarthritic pain. Mol Pain. 7:88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Xu Q, et al. 2021. Astrocytes contribute to pain gating in the spinal cord. Sci Adv. 7:eabi6287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Xie AX, Petravicz J, McCarthy KD. 2015. Molecular approaches for manipulating astrocytic signaling in vivo. Front Cell Neurosci. 9:144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Van Den Herrewegen Y, et al. 2021. Side-by-side comparison of the effects of Gq- and Gi-DREADD-mediated astrocyte modulation on intracellular calcium dynamics and synaptic plasticity in the hippocampal CA1. Mol Brain. 14:144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Iwai Y, et al. 2021. Transient astrocytic Gq signaling underlies remote memory enhancement. Front Neural Circuits. 15:658343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Roth BL. 2016. DREADDs for neuroscientists. Neuron. 89:683–694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Tang J, Bair M, Descalzi G. 2021. Reactive astrocytes: critical players in the development of chronic pain. Front Psychiatry. 12:682056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Tsuda M, et al. 2011. JAK-STAT3 pathway regulates spinal astrocyte proliferation and neuropathic pain maintenance in rats. Brain. 134:1127–1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Xu M, Bruchas MR, Ippolito DL, Gendron L, Chavkin C. 2007. Sciatic nerve ligation-induced proliferation of spinal cord astrocytes is mediated by κ opioid activation of p38 mitogen-activated protein kinase. J Neurosci. 27:2570–2581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Farhy-Tselnicker I, Allen NJ. 2018. Astrocytes, neurons, synapses: a tripartite view on cortical circuit development. Neural Dev. 13:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Colburn R, et al. 1997. Dissociation of microglial activation and neuropathic pain behaviors following peripheral nerve injury in the rat. J Neuroimmunol. 79:163–175. [DOI] [PubMed] [Google Scholar]
- 79. Tanga F, Raghavendra V, DeLeo J. 2004. Quantitative real-time RT-PCR assessment of spinal microglial and astrocytic activation markers in a rat model of neuropathic pain. Neurochem Int. 45:397–407. [DOI] [PubMed] [Google Scholar]
- 80. Ueda H, et al. 2018. Involvement of lysophosphatidic acid-induced astrocyte activation underlying the maintenance of partial sciatic nerve injury-induced neuropathic pain. Pain. 159:2170–2178. [DOI] [PubMed] [Google Scholar]
- 81. Dong J, Xu C, Xia R, Zhang Z. 2021. Upregulating miR-130a-5p relieves astrocyte over activation-induced neuropathic pain through targeting CXC motif chemokine receptor 12/CXC motif chemokine receptor 4 axis. Neuroreport. 32:135–143. [DOI] [PubMed] [Google Scholar]
- 82. Smith PA. 2023. Neuropathic pain; what we know and what we should do about it. Front Pain Res. 4:1220034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Tillu DV, Gebhart GF, Sluka KA. 2008. Descending facilitatory pathways from the RVM initiate and maintain bilateral hyperalgesia after muscle insult. Pain. 136:331–339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Sluka KA, Kalra A, Moore SA. 2001. Unilateral intramuscular injections of acidic saline produce a bilateral, long-lasting hyperalgesia. Muscle Nerve. 24:37–46. [DOI] [PubMed] [Google Scholar]
- 85. Sluka KA, et al. 2003. Chronic hyperalgesia induced by repeated acid injections in muscle is abolished by the loss of ASIC3, but not ASIC1. Pain. 106:229–239. [DOI] [PubMed] [Google Scholar]
- 86. Lesnak J, Sluka KA. 2019. Chronic non-inflammatory muscle pain: central and peripheral mediators. Curr Opin Physiol. 11:67–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Zhuang ZY, Gerner P, Woolf CJ, Ji RR. 2005. ERK is sequentially activated in neurons, microglia, and astrocytes by spinal nerve ligation and contributes to mechanical allodynia in this neuropathic pain model. Pain. 114:149–159. [DOI] [PubMed] [Google Scholar]
- 88. Anderson CM, Swanson RA. 2000. Astrocyte glutamate transport: review of properties, regulation, and physiological functions. Glia. 32:1–14. [PubMed] [Google Scholar]
- 89. Sung B, Lim G, Mao J. 2003. Altered expression and uptake activity of spinal glutamate transporters after nerve injury contribute to the pathogenesis of neuropathic pain in rats. J Neurosci. 23:2899–2910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Ji R-R, Donnelly CR, Nedergaard M. 2019. Astrocytes in chronic pain and itch. Nat Rev Neurosci. 20:667–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Weng H-R, Chen J, Cata J. 2006. Inhibition of glutamate uptake in the spinal cord induces hyperalgesia and increased responses of spinal dorsal horn neurons to peripheral afferent stimulation. Neuroscience. 138:1351–1360. [DOI] [PubMed] [Google Scholar]
- 92. Yaster M, et al. 2011. Effect of inhibition of spinal cord glutamate transporters on inflammatory pain induced by formalin and complete Freund’s adjuvant. Anesthesiology. 114:412–423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Niederberger E, Schmidtko A, Rothstein J, Geisslinger G, Tegeder I. 2003. Modulation of spinal nociceptive processing through the glutamate transporter GLT-1. Neuroscience. 116:81–87. [DOI] [PubMed] [Google Scholar]
- 94. Durkee CA, Araque A. 2019. Diversity and specificity of astrocyte-neuron communication. Neuroscience. 396:73–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Choi SR, et al. 2017. Spinal D-serine increases PKC-dependent GluN1 phosphorylation contributing to the sigma-1 receptor-induced development of mechanical allodynia in a mouse model of neuropathic pain. J Pain. 18:415–427. [DOI] [PubMed] [Google Scholar]
- 96. Henneberger C, Papouin T, Oliet SH, Rusakov DA. 2010. Long-term potentiation depends on release of D-serine from astrocytes. Nature. 463:232–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Yang Y, et al. 2003. Contribution of astrocytes to hippocampal long-term potentiation through release of D-serine. Proc Natl Acad Sci U S A. 100:15194–15199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Mothet J-P, et al. 2005. Glutamate receptor activation triggers a calcium-dependent and SNARE protein-dependent release of the gliotransmitter D-serine. Proc Natl Acad Sci U S A. 102:5606–5611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Chen W-K, et al. 2010. Cav3. 2 T-type Ca2+channel-dependent activation of ERK in paraventricular thalamus modulates acid-induced chronic muscle pain. J Neurosci. 30:10360–10368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Maiarù M, et al. 2018. Selective neuronal silencing using synthetic botulinum molecules alleviates chronic pain in mice. Sci Transl Med. 10:eaar7384. [DOI] [PubMed] [Google Scholar]
- 101. Miyakawa A, et al. 2005. Action of neuropeptide Y on nociceptive transmission in substantia gelatinosa of the adult rat spinal dorsal horn. Neuroscience. 134:595–604. [DOI] [PubMed] [Google Scholar]
- 102. Mohan A, et al. 2018. Antisense oligonucleotides selectively suppress target RNA in nociceptive neurons of the pain system and can ameliorate mechanical pain. Pain. 159:139–149. [DOI] [PubMed] [Google Scholar]
- 103. Rao VLR, et al. 2001. Antisense knockdown of the glial glutamate transporter GLT-1, but not the neuronal glutamate transporter EAAC1, exacerbates transient focal cerebral ischemia-induced neuronal damage in rat brain. J Neurosci. 21:1876–1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Yang L, Qi Y, Yang Y. 2015. Astrocytes control food intake by inhibiting AGRP neuron activity via adenosine A1 receptors. Cell Rep. 11:798–807. [DOI] [PubMed] [Google Scholar]
- 105. Kohro Y, et al. 2015. A new minimally-invasive method for microinjection into the mouse spinal dorsal horn. Sci Rep. 5:14306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Lu C-H, et al. 2023. In silico and in vitro screening of serine racemase agonist and in vivo efficacy on Alzheimer's disease Drosophila melanogaster. Pharmaceuticals. 16:280. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All relevant data have been included in the submitted materials.