Abstract
Self-report is typically used to differentiate between asymptomatic neurocognitive impairment (ANI) and mild neurocognitive disorder (MND) in the assessment of HIV-associated neurocognitive disorders (HAND). Yet, it is unclear whether the lack of self-reported functional impairments in individuals with ANI is indicative of a genuine absence of functional impairment, or of inaccurate self-reports. In the present study, we examined the relationship between previously validated self-report (patient’s assessment of own functioning inventory; instrumental activities of daily living inventory) and performance-based (the Texas Functional Living Scale) measures of functional abilities in 112 virologically-controlled HIV-infected, and 40 well-matched, HIV-uninfected participants. Participants with symptomatic cognitive impairment (CI) had significantly lower overall scores and higher rates of impairment on a performance-based measure of everyday functioning as compared to participants with either asymptomatic CI or normal cognitive performance WNL within normal limits]; all p < 0.05), while asymptomatic CI and WNL participants had comparable rates of impairment and performance within the average range on the performance-based measure. The concordance between self-report and performance-based measures of everyday functioning in asymptomatic and symptomatic CI provide support for ANI and MND as clinically distinct diagnostic entities, and support the use of self-reports as appropriate measures of everyday functioning in the diagnosis of HAND.
Keywords: HIV-associated neurocognitive disorders, Everyday functioning, Self-reports, Texas Functional Living Scale, Instrumental activities of daily living
Introduction
In 1991, the American Academy of Neurology (AAN) published criteria for HIV-associated dementia (HAD) and minor cognitive motor disorder (MCMD), acknowledging the cognitive and behavioral sequelae of HIV infection [1]. In addition to demonstrated cognitive impairment on exam, a diagnosis of either HAD or MCMD required evidence of impairment or increased dependence at work or in instrumental activities of daily living (IADLs). Though this diagnostic algorithm was useful in identifying more severe cases of cognitive and behavioral impairment, it became evident that many HIV-infected individuals who demonstrated mild-to-moderate cognitive impairment on formal neuropsychological exams did not report any difficulties in work or in IADLs, thus precluding them from receiving a diagnosis of MCMD. It is largely for this reason that in 2007, the AAN diagnostic algorithm for HIV-associated neurocognitive disorders (HAND) was developed to reflect this subgroup of ‘asymptomatic’ individuals [2]. While the criteria for HAD remained largely the same, the category formerly known as MCMD was divided into asymptomatic neurocognitive impairment (ANI) and mild neurocognitive disorder (MND). Both diagnostic categories (i.e., ANI and MND) require evidence of mild impairment in at least two cognitive domains, though the number and type of tests that should be administered are not specified. Additionally, for a diagnosis of MND, individuals must exhibit difficulties in work and/or IADLs. A diagnosis of ANI is given if difficulties in IADLs are not reported or detected through formal assessment.
IADLs are frequently assessed by self-reports in the diagnosis of HAND [3] not because self-reports have been accepted as the ‘gold standard’ in the field, but rather because they are inexpensive, quick, require little training to administer, and do not require testing materials other than paper forms and writing utensils. While informant reports possess the same advantages, obtaining informant reports in the context of HIV infection has traditionally been difficult because of a variety of factors including social stigma and resulting social isolation [4–6]. Although self-report data is used in the diagnosis of HAND, it is unclear whether such data are accurate approximations of cognition and everyday function.
Some evidence suggests no relationship between self-reported neuropsychological impairments and impairments on formal neuropsychological testing in individuals with HIV in both the pre-highly active retroviral therapy (HAART) [7, 8] and post-HAART eras [9–13], as well as no relationship between self-reported functional impairments and impairments on performance-based measures of everyday functioning. Findings from a recent exploratory study [14], in which HIV-infected participants with ANI and MND manifested the same level of functional impairments on performance-based tasks of IADLs, suggest that functional impairments in ANI do exist, but simply go unreported [14]. However, that study was limited by a small sample size (n = 16 with HAND) and a demographically non-representative sample (100% men over age 60). A prior study using performance-based tests of medication management and financial abilities found similar levels of function in CI HIV-infected participants who self-reported functional impairment and those who did not self-report functional impairments, further suggesting a discordance between actual and self-reported functional impairment [15].
Conversely, Blackstone et al. [16] found that in 76% of participants with CI, there was a concordance between impairments in self-reports and impairments on a performance-based measure of everyday functioning. While these results provide initial evidence suggesting that the reliance on self-reports rather than objective performance-based measures of everyday functioning as part of HAND algorithms results in accurate representations of everyday functioning (and concomitantly, accurate diagnoses), it remains unknown whether these results would be replicable in samples with lower HIV severity (e.g., 64% of the sample in Blackstone et al. [16] had a current CD4 count <200 or AIDS-defining illness), current controlled viremia (in Blackstone et al. [21] approximately 50% of participants had detectable viremia), or when using different tests of IADLs. Additionally, since Blackstone et al. [16] calculated and reported rates of impairment on functional measures relative to cognitively unimpaired participants as a dichotomous impaired/unimpaired variable, the magnitude of the difference in performance-based measures across HAND diagnostic categories when considering everyday functioning on a continuum warrants further study.
It has been proposed that observed discrepancies between performance-based and self-reported evaluations of functioning may be due to impaired self-awareness experienced at increasing levels of cognitive impairment [17–19] such that individuals who are cognitively impaired do not realize the problems they have, and thus do not report them. In the context of HAND specifically, this could result in an underestimation of the prevalence of MND and concomitant overestimation of the prevalence of ANI [14]. Further, these potential inaccuracies in diagnosis may contribute to the current inability to find biomarkers of clinical interventions for cognitive and IADL impairment in HIV.
The aim of the present study was to determine whether the current reliance on self-reports to evaluate functional impairment in the diagnosis of HAND is justified. To do so, we evaluated the relationship between self-reports and performance-based measures of everyday functioning across participants with asymptomatic cognitive impairment (CI), symptomatic CI, and normal cognitive performance (WNL) in a large cohort of virologically suppressed HIV-infected participants and demographically matched HIV-uninfected controls. Based on data from prior research [14, 15], we hypothesized that participants with asymptomatic CI and symptomatic CI would show no differences on performance-based tasks and simultaneously be impaired relative to the performance of WNL participants, while asymptomatic CI participants would self-report functional statuses similar to those of WNL participants.
Materials and Methods
Participants
Participants were evaluated during the initial screening visit of a 5-year, IRB-approved, longitudinal protocol investigating the clinical course of cognitive impairment in individuals with HIV at the National Institutes of Health (NIH) in Bethesda, MD (clinicaltrials.gov, NCT01875588).
The participants were recruited from existing HIV protocols at NIH, HIV clinics, and physician offices in the Washington, DC metropolitan area. To minimize the confounding effect of socioeconomic and demographic factors, both HIV-infected and HIV-uninfected participants were recruited from the same communities, and HIV-infected participants were encouraged to inform friends and relatives about the study. Advertisements recruiting HIV-infected participants for a study on thinking, mood, and memory were placed on buses and bus stops in the Washington, DC area. Similar advertisements at the same locations were used to recruit HIV-uninfected participants.
Participant inclusion criteria were age between 18 and 61 years, ability to provide informed consent, ≥7 years of formal education, and the ability to speak, read, and understand English. HIV infection was documented by an OraQuick rapid test or with an HTV-l/HIV-2 Multispot rapid test and Western blot as determined by the NIH Clinical Pathology Laboratory or by Leidos Biomedical Research Monitoring Laboratory. Abbott Real Time HIV assays were used to measure plasma HIV viral load levels. HIV-uninfected participants were required to be HIV-antibody negative. HIV-infected participants were required to have controlled HIV viremia on ART for at least one year at time of testing. Controlled HIV viremia was defined as having a viral load <50 copies/mL. However, participants with a transitory episode of a viral load >50 copies/mL preceded and followed by controlled viremia (i.e., a transient viral load ‘blip’) were also included. Participants were excluded if they were found to have any illness that could confound neurocognitive outcomes (i.e., prior CNS infection), conditions (other than HAND) associated with cognitive impairment, concurrent severe substance abuse, or a concurrent unstable or acute psychiatric illness that would interfere with study participation. Psychiatric and substance abuse-related measures were obtained through the Client Diagnostic Questionnaire, a structured psychiatric interview developed specifically for use in patients with HIV/AIDS [20]. Current levels of depression were assessed using the Beck Depression Inventory (BDI-II), a 21 item self-report questionnaire addressing both physical symptoms (e.g., fatigue, weight loss) and cognitive symptoms (e.g., irritability, hopelessness) of depression [21]. Consent was obtained once participants were made aware of the risks, benefits, and aims of the study.
Cognitive Assessments
All participants were tested by a licensed neuropsychologist or trained psychometrists. Consistent with Antinori et al. criteria [2] stating that at least five domains must be assessed in the evaluation of HAND, we evaluated seven cognitive domains as well as estimated premorbid IQ, as measured by the Wechsler test of adult reading (WTAR; see Table 1 for domains and tests). Participants were also administered a measure of performance validity (Medical Symptom Validity Test [MSVT] [22]). Only participants demonstrating good effort, as evaluated by a senior neuropsychologist [JS], were included in the present analyses.
Table 1.
Neuropsychological tests administered per cognitive domain
| Domain | Test/variable |
|---|---|
|
| |
| Estimated premorbid IQ | WTAR |
| Attention/working memory | PASAT; WAIS-III Letter-Number Sequencing |
| Executive functioning | Trail Making Test Part B; WCST-64 (perseverative responses) |
| Information processing | Trail Making Test Part A; WAIS-III Digit-Symbol Coding; WAIS-III Symbol Search |
| Verbal fluency | COWA FAS; COWA animals |
| Learning | HVLT-R (total recall); BVMT-R (total recall) |
| Psychomotor functioning | Grooved pegboard |
| Memory | HVLT-R (delayed recall); BVMT-R (delayed recall) |
| Everyday functioning | PAOFI (self-report); IADL questionnaire (self-report); TFLS (performance-based) |
WTAR Wechsler test of adult reading, PASAT Paced Auditory Serial Addition Task, WAIS-III Wechsler Adult Intelligence Scale (3rd edition), WCST-64 Wisconsin card sorting test-64 card version, COWA Controlled oral word association test, HVLT-R Hopkins verbal learning test-revised, BVMT-R brief visuospatial memory test-revised, PAOFI patient’s assessment of own functioning, IADL instrumental activities of daily living, TFLS Texas Functional Living Scale
Raw scores were transformed to T-scores using widely available neuropsychological normative software based on large national samples, and were corrected for demographic variables such as age, gender, race, and education level, when available [23–25]. T-scores were then used to determine whether participants met criteria for normal cognitive performance (WNL) or CI, based on a previously outlined algorithm developed to evaluate cognition in HAND [26].
Self-Report of Functional Ability
The Woods et al. [26] criteria for functional impairment in HAND require evaluation of two different self-report forms and current employment status. Participants who exhibit impairment in at least two of these measures (i.e., impairment on one of the self-report measures described below and currently unemployed, impairment on both self-report measures and currently employed, or impairment on both self-report measures and currently unemployed) are considered symptomatic or functionally impaired.
The PAOFI [27] is a 41-item self-report form that evaluates how participants feel they have been performing lately in terms of memory, language and communication, use of hands, sensory-perceptual abilities, and higher level cognitive and intellectual functions. The PAOFI has been used frequently to assess functional abilities in various clinical and research populations [26, 28, 29]. Participants are instructed to indicate how frequently they experience problems within the aforementioned domains using a Likert scale ranging from 1 (almost always) to 6 (almost never).
Responses rated as 3 (fairly often), 2 (very often), and 1 (almost always) are considered indicative of functional difficulty. Participants indicating functional difficulty (i.e., selecting ≤3) on three or more items were considered to be clinically significantly impaired in their everyday functioning. For consistency with previous research [10], participants who had elevated depression, defined as a BDI-II score ࣙ17, were not considered impaired in their everyday functioning unless they endorsed complaints on 10 or more PAOFI items.
The IADL questionnaire [30] is a self-report form to assess functional decline in common tasks such as housekeeping, cooking, keeping track of medication, and managing finances. The form assesses decline by requiring participants to compare their current perceived ability to perform tasks to their highest perceived ability level. Participants reporting functional decline in two or more domains were considered to exhibit clinically significant decline in their everyday functioning.
Performance-Based Measure of Functional Ability
The Texas Functional Living Scale (TFLS) [31] was used as a performance-based measure of functional ability. TFLS performance was not used as part of the HAND diagnostic algorithm. The TFLS requires participants to perform tasks simulating activities performed in daily life, and has previously been found to have high ecological and face validity among cognitively unimpaired participants, as well as among participants with known neurologic diagnoses that deleteriously impact everyday functioning [32–34]. While everyday functioning is somewhat associated with cognitive functioning, TFLS scores are only weakly correlated (r = 0.26) with measures of general intelligence [31]; this suggests that the TFLS is not simply a different neuropsychological measure, but that it indeed measures abilities not captured by traditional neuropsychological tests. Sample TFLS tasks include having to tell time, write a check, count change, look up a phone number, take a proper dose of medication at a certain time, follow a recipe, and address an envelope. Tasks are divided into four scales: time, money and calculation, communication, and memory. Raw scores were converted to T-scores using available normative data based on a national sample [31]. TFLS T-scores are not corrected for any demographic variables, and thus are informative about absolute levels of performance (i.e., how a participant’s performance compares to that of the general population). TFLS overall T-scores were used for the present analyses, and T-scores below 40 were considered impaired.
Data Analysis
Since HIV-uninfected and HIV-infected participants were combined for our primary analyses, we compared demographic variables to ensure the two groups were well-matched. Specifically, we conducted chi-squared tests to determine whether the two groups differed on the basis of race and sex. One-way analyses of variance (ANOVA) were used to test for differences in age at testing, years of formal education, estimated premorbid IQ, and depression (BDI-II) scores. These tests were then run to determine whether there were differences among neuropsychological diagnostic groups, irrespective of HIV diagnosis.
To evaluate differences among neuropsychological diagnostic groups in mean TFLS T-scores, we conducted a series of ANOVAs. If the omnibus ANOVA was significant, post hoc Tukey tests were run to elucidate the specific between-group differences that were significant. TFLS scores were also analyzed by creating a dichotomous variable of TFLS performance (TFLS impaired or unimpaired) and then using a series of Chi square tests to determine whether there was an association with TFLS impairment and neuropsychological diagnostic group. We then repeated these primary analyses restricting our sample to HIV-infected participants.
Finally, we compared scores between the HIV-infected and HIV-uninfected groups using a series of ANOVAs and Chi squared analyses to elucidate the effect of HIV diagnosis on neuropsychological test scores, scores on the performance-based measure, and self-reports of functional impairment.
All tests were two-tailed, and significance level was set to α ≤ 0.05. Effect sizes are reported as eta squared (η2) for omnibus ANOVAs, as Cohen’s d for between-group comparisons, and as φ for Chi square analyses. All analyses were run using SPSS version 22 (IBM, Armonk, NY).
Results
Sample Characteristics
A total of 152 participants (40 HIV-uninfected and 112 HIV-infected) completed neuropsychological testing and demonstrated good effort on the measure of performance validity. Data from six participants who did not pass the performance validity measure were excluded. Participants were predominantly African American (67.8%, n = 103) and male (61.2%, n = 93). Thirty-nine percent (n = 49) of participants were employed full time, 15.8% (n = 24) part time, and 44.7% (n = 68) were unemployed. The mean age at time of testing was 49.05 (SD = 9.06). The mean years of formal education for participants was 13.71 years (SD = 2.81). Twelve percent of participants (n = 18) had elevated depression (i.e., BDI-II scores ≥ 17). The omnibus ANOVA testing differences in BDI-II scores across NP diagnostic groups was trending towards significance (F[2,149] = 2.66, p = 0.07, η2 = 0.04) with post hoc tests revealing the symptomatic CI group had a higher mean BDI-H score than the WNL (p = 0.04), but not the asymptomatic CI group, and no difference between the WNL and asymptomatic CI groups (both p > 0.10). Similarly, the omnibus ANOVA testing differences in premorbid IQ was significant (F[2,149] = 3.57, p = 0.03, η2 = 0.05), with post hoc tests revealing that the symptomatic CI group had a lower mean estimated premorbid IQ than the WNL (p = 0.02), but not the asymptomatic CI group, and no difference between the WNL and asymptomatic CI group (both p > 0.10). No significant differences were found in age (F[2,149] = 1.03, p = 0.36, η2 = 0.01), education (F[2,149] = 1.80, p = 0.17, η2 = 0.02), gender (χ2[1] = 1.10, p = 0.58, φ = 0.09), and race (χ2[1] = 2.47, p = 0.29, φ = 0.13). See Table 2 for additional sample information stratified by neuropsychological diagnosis.
Table 2.
Sample characteristics stratified by neuropsychological diagnosis
| Overall (n = 152) | Cognition WNL (n = 117) | Asymptomatic CI (n = 25) | Symptomatic CI (n = 10) | |
|---|---|---|---|---|
|
| ||||
| Mean age (SD) | 40.05 (9.06) | 49.44 (8.61) | 46.72 (10.18) | 50.30 (11.29) |
| Mean years of education (SD) | 13.71 (2.81) | 13.80 (2.80) | 13.92 (2.96) | 12.10 (2.13) |
| Mean estimated premorbid IQ (SD) | 98.41 (12.28) | 99.51 (11.84) | 97.00 (14.14) | 89.20 (8.28)* |
| Mean BDI-II score (SD) | 8.01 (7.33) | 7.56 (6.92) | 8.20 (8.08) | 13.33 (9.04)* |
| Male | 61.2% | 62.4% | 56.0% | 60.0% |
| African American | 67.8% | 65.0% | 72.0% | 90.0% |
| MSM | 37.5% | 39.3% | 32.0% | 30.0% |
| Employment | ||||
| Full-time | 38.8% | 43.6% | 28.0% | 10.0% |
| Part-time | 15.8% | 15.4% | 20.0% | 10.0% |
| Unemployed | 45.4% | 41.1% | 52.0% | 80.0% |
| History of alcohol abuse | 17.8% | 16.2% | 28.0% | 10.0% |
| History of drug abuse | 31.6% | 31.6% | 32.0% | 30.0% |
| History of depression | 42.1% | 41.0% | 44.0% | 50.0% |
| Currently prescribed psychiatric medication | 30.9% | 29.1% | 36.0% | 40.0% |
| Diabetes | 4.6% | 3.4% | 8.0% | 10.0% |
| Hypertension | 31.6% | 34.2% | 20.0% | 30.0% |
| Detectable Hepatitis C virus (HCV) RNA | 4.6% | 2.6% | 0.0%† | 40.0%* |
Significantly different than WNL at p < 0.05
Significantly different than symptomatic CI at p < 0.05
There were no significant differences between the HIV-uninfected and HIV-infected groups in distributions of race (χ2[1] = 0.2, p = 0.6, φ = 0.04), sex (χ2[1] = 0.02, p = 0.90, φ = 0.01), employment (χ2[1]0.3, p = 0.8, cp = 0.05), estimated premorbid IQ (t[150] = 1.7, p = 0.09, d = 0.3) and years of education (t[150] = 1.5, p = 0.15, d = 0.3). The mean age of HIV-infected participants was approximately 4 years higher than that of HTV-uninfected participants (t[150] = 2.2, p = 0.03, d = 0.5), HIV-infected participants had higher scores on the BDI-II (t[150] = 2.2, p = 0.02, d = 0.5), and a higher proportion of HIV-infected participants were MSM (χ2[1] = 29.1, p < 0.01, φ = 044). See Table 3 for sample information stratified by HIV diagnosis.
Table 3.
Sample characteristics by HIV diagnosis
| HIV-infected (n = 112) | HIV-uninfected (n = 40) | |
|---|---|---|
|
| ||
| Mean age (SD)* | 50.01 (8.50) | 46.35 (10.11) |
| Mean years of education (SD) | 13.54 (2.83) | 14.18 (2.74) |
| Mean WTAR estimated IQ (SD) | 97.67 (12.36) | 100.48 (11.91) |
| Mean BDI-II score (SD)* | 8.78 (7.11) | 5.85 (7.58) |
| Male | 62.5% | 57.5% |
| African American | 66.1% | 72.5% |
| MSM* | 48.2% | 7.5% |
| Employment | ||
| Full-time | 38.4% | 42.5% |
| Part-time | 17.0% | 12.5% |
| Unemployed | 44.6% | 45.0% |
| History of alcohol abuse | 21.4% | 7.5% |
| History of drug abuse | 36.6% | 17.5% |
| History of depression | 47.3% | 27.5% |
| Currently prescribed psychiatric medication | 33.9% | 22.5% |
| Diabetes | 5.4% | 2.5% |
| Hypertension | 35.7% | 20.0% |
| Detectable Hepatitis C virus (HCV) RNA | 5.4% | 2.5% |
Significant difference between HIV-infected and HIV-uninfected groups at p < 0.05
Plasma viral load was undetectable (<40 copies/mL) for 92% HIV-infected participants. As the detectable viral loads observed in 8% of participants were preceded and followed by a viral load <50 copies/mL (i.e. viral load ‘blips’), these participants were retained in analyses as per study protocol. All participants with detectable viral loads had viral loads <300 copies/mL.
Mean reported duration of time since HIV diagnosis was 17.77 years (SD = 8.51), mean reported nadir CD4 count was 200.91 (SD = 175.58), and mean CD4 count at testing was 600.15 (SD = 299.81). At time of testing, 5.5% of HIV-infected participants had a CD4 count <200.
Using previously published diagnostic criteria [2, 26], 77.0% (n = 117) of participants did not meet criteria for CI (WNL), 16.4% (n = 25) met criteria for asymptomatic CI, and 6.6% (n = 10) met criteria for symptomatic CI Similar rates of CI have previously been reported in studies of HAND amongst virologically-controlled participants [35, 36].
Performance-Based Measures
TFLS T-scores were highest in the WNL group (M = 52.23, SD = 10.0), followed by the asymptomatic CI (M = 49.32, SD = 10.87) and the symptomatic CI group (M = 37.90, SD = 8.45; see Fig. 1). The omnibus ANOVA was significant (F[2,149] = 9.67, p < 0.01, η2 = 0.17), indicating a significant main effect of neuropsychological diagnostic group. Post-hoc tests revealed that TFLS T-scores were significantly lower in the symptomatic CI group than in the WNL and asymptomatic CI groups (both p < 0.01; d = 1.57 and 1.12, respectively). There was no significant difference between the asymptomatic CI and WNL groups (p = 0.5, d = 0.36). When restricting analyses to HIV-infected participants only (n = 112), there was again a significant main effect of neuropsychological diagnostic group (F[2,109] = 7.4, p < 0.01, η2 = 0.11) with post hoc analyses revealing significantly lower scores in the symptomatic CI group (M = 38.67, SD = 8.59) than the WNL group (M = 51.33, SD = 10.35; p < 0.01, d = 1.28). The difference between TFLS scores in the asymptomatic CI group (M = 46.07, SD = 9.81) and symptomatic CI group trended towards significance (p = 0.08, d = 0.80).
Fig. 1.
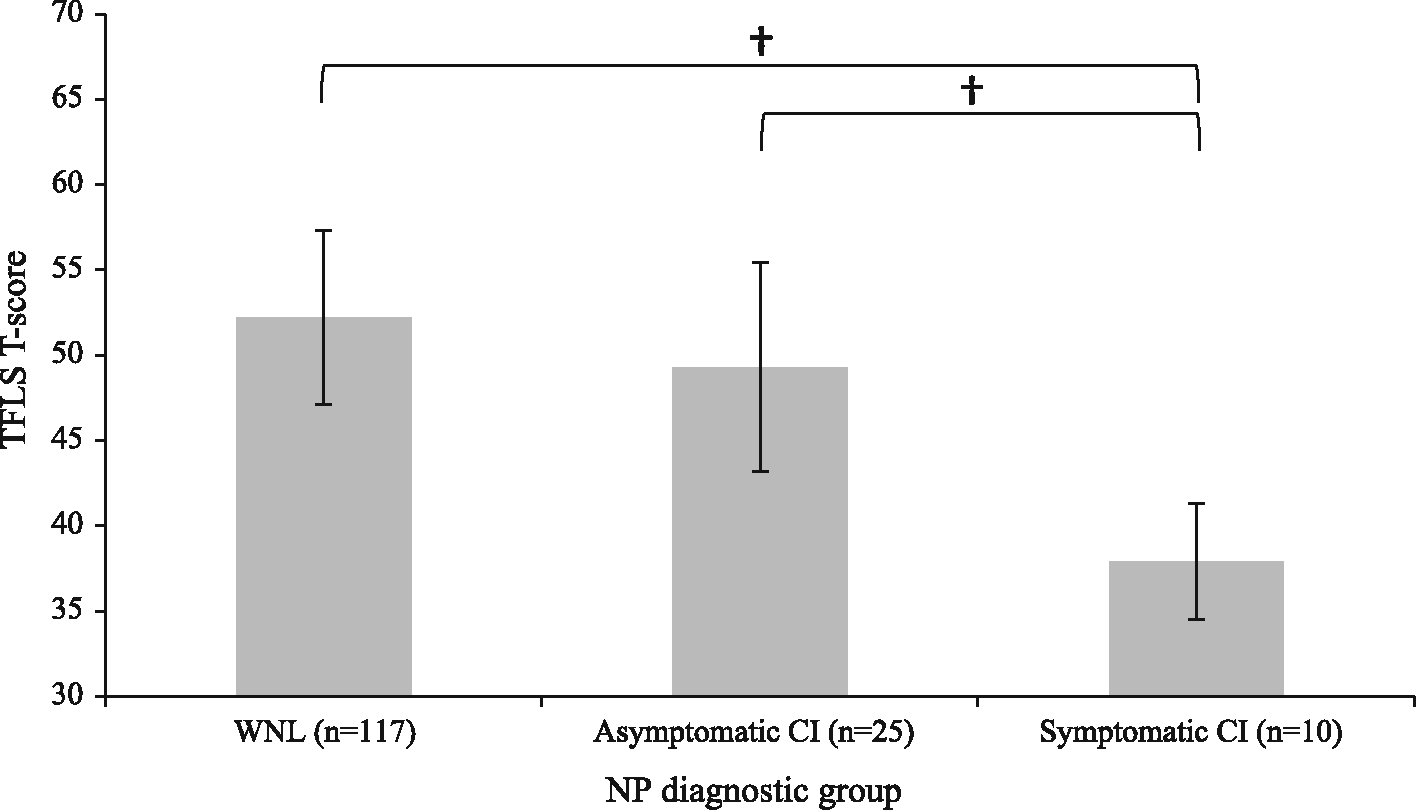
Mean TFLS T-scores by NP diagnostic group. Bars represent standard error, † significant at p < 0.01
As the percent of participants with detectable Hepatitis C (HCV) RNA (determined using chemiluminescence immunoassay detecting HCV antibodies) was significantly higher in the symptomatic CI group than in the asymptomatic CI and WNL groups and previous work has shown an association between HCV and cognition [37, 38], analyses were run to elucidate the potentially confounding effect of HCV on TFLS performance. An independent samples t test revealed that participants with detectable HCV RNA (n = 7) had significantly lower TFLS scores than those with undetectable HCV RNA (t[150] = 3.34, p < 0.01, d = 1.31). Thus, the analyses described above were re-run with HCV entered as a covariate. The omnibus ANCOVA revealed a significant main effect of neuropsychological diagnostic group (F[2,149] = 5.52, p < 0.01, η2 = 0.07), and a trending main effect of detectable HCV RNA (F[1,149] = 3.21, p = 0.08, η2= 0.02). Subsequent post hoc pairwise comparisons revealed that TFLS scores remained significantly lower in the symptomatic CI group than in the asymptomatic CI and WNL groups (both p < 0.05), and that there was no significant difference in TFLS scores in the asymptomatic CI and WNL groups (p = 0.16).
We then evaluated TFLS performance as a dichotomous (impaired or unimpaired), rather than as a continuous, variable. The highest rate of TFLS impairment was found in the symptomatic CI group (80.0%), followed by the asymptomatic CI group (24.0%), and WNL group (12.8%). There was a significant association between TFLS impairment and neuropsychological diagnostic group. Symptomatic CI participants were significantly more likely to be impaired on the TFLS as compared to asymptomatic CI (χ2[1] = 9.33, p < 0.01, φ = 0.52) and WNL participants (χ2[1] = 28.04, p < 0.01, φ = 0.47), while impairment rates were comparable in the asymptomatic CI and WNL groups (η2[1] = 2.04, p = 0.15, φ = 0.12). Self-reported impairment was significantly associated with TFLS impairment in the total sample (χ2[1] = 8.75, p < 0.01, φ = 0.24). The same results were found when restricting analyses to HIV-infected individuals.
Within the sample of CI participants, there was 77.1% agreement between impairment on performance-based and self-report impairment (54.3% not impaired on both self-report and TFLS, 22.9% impaired on both self-report and TFLS). Six percent of CI participants were impaired on self-report measures only, and 17.1% were impaired on the TFLS only. Within the WNL sample, 71.6% of participants were concordant in their impairment on self-report and TFLS (66.4% not impaired on both measures, 5.2% impaired on both measures). Twenty-one percent of the WNL sample was impaired on self-report measures only, and 7.8% were impaired on the TFLS only.
Relationship Between HIV Diagnosis, Self-reports, and Scores
There was no difference between HIV-infected and HIV-uninfected participants in overall neuropsychological T-scores (t[150] = 0.32, p = 0.75, d = 0.15). The difference between HIV-infected and HIV-uninfected participants in TFLS T-scores was statistically significant (t[150] = 2.37, p = 0.02, d = 0.53), though it is important to note that mean scores for the HIV-infected group (M = 49.61, SD = 10.73) and the HIV-uninfected group (M = 54.18, SD = 9.69) were both within the average range. These results were then replicated when running the analysis with detectable HCV RNA entered as a covariate. There was a trending association between HIV diagnosis and TFLS impairment (10.0% for HIV-uninfected and 22.3% for HIV-infected groups; χ2[1] = 2.90, p = 0.09, φ = 0.14). There was also a significant association between HIV diagnosis and self-reported impairment (χ2[1] = 3.69, p = 0.06, φ = 0.16), with HIV-infected participants being more likely to self-report impairment (30.6%) than HIV-uninfected participants (15.0%).
Post Hoc Analyses
A post hoc 2×2 ANOVA was run with cognitive status (neuropsychologically impaired versus neuropsychologically unimpaired) and self-reported impairment (impaired versus unimpaired) entered as fixed factors to further elucidate the effects of both cognition and self-reported impairment on TFLS scores. There was a trending interaction between neuropsychological impairment and self-re-ported impairment (F[1,149] = 3.26, p = 0.07, χ2 = 0.02). Within the WNL group, mean TFLS scores were in the normal range regardless of whether participants were asymptomatic or symptomatic on self-report (M = 53.24 in WNL asymptomatic participants; M = 49.60 in WNL symptomatic participants). However, within CI participants, scores varied based on whether participants were asymptomatic or symptomatic on self-report (as discussed above; see Fig. 2).
Fig. 2.
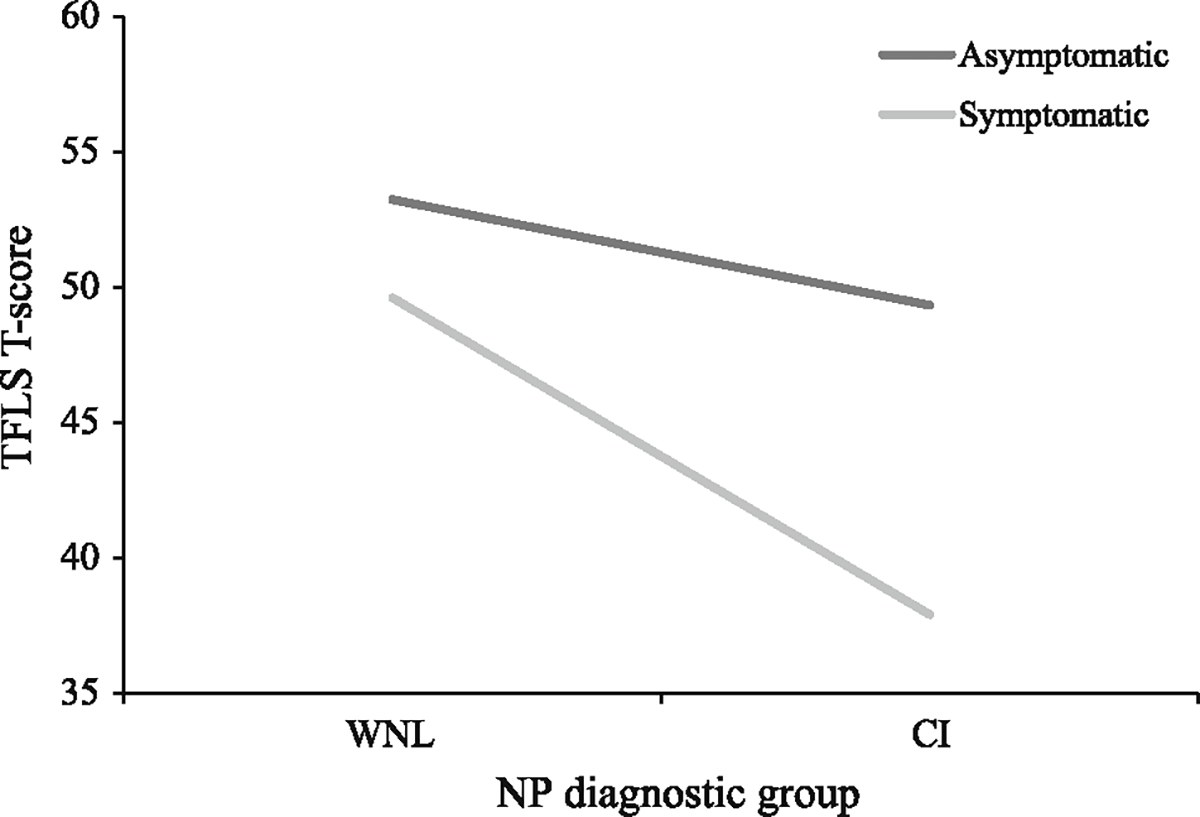
Mean TFLS scores by NP diagnostic group and self-reported impairment in everyday functioning
Discussion
After adding ANI as a diagnostic category, authors of the 2007 revised AAN HAND criteria recognized that ‘further work needs to be conducted on the real-life impact of ANT [2]. Recent work has suggested that rather than being ‘asymptomatic’ as the diagnosis implies, individuals with ANI do in fact experience functional impairment, but do not endorse functional impairments on self-reports [14,15]. In the present study, we expanded upon this work by utilizing a performance-based measure with previously demonstrated high ecological and face validity (the TFLS), and a larger, more demographically representative sample in which all HIV-infected participants were virologically suppressed. Contrary to our hypothesis, our results suggest a concordance between self-report and performance-based measures of everyday functioning in both asymptomatic and symptomatic CI participants, thus providing support for ANI as a distinct diagnostic entity from MND.
Consistent with the current AAN diagnostic criteria, the underperformance on neuropsychological tests in the asymptomatic CI group did not translate into functional deficits (as measured by TFLS scores), while it did translate into functional deficits for symptomatic CI patients. However, it may be important to note that despite a lack of statistical significance, the impairment rate on the TFLS of asymptomatic CI participants (25.0%) was almost twice as high as that of WNL participants (12.8%), although mean scores were not significantly different between groups. It may be that some participants classified as having asymptomatic CI experience subclinical, but considerable, levels of functional impairment relative to WNL participants. Seventy-seven percent of CI participants were concordant in their ratings of impairment by both self-report and performance-based measures, consistent with the concordance found in Blackstone et al. [16]. That the present study was able to replicate previous findings in a sample with different HIV disease characteristics (e.g., different percentage of participants meeting AIDS criteria, participants with controlled viremia) and by using different tests of everyday functioning suggests that self-reports may in fact be accurately capturing everyday functioning abilities for a large majority of participants. Further, results suggest that there may be little added diagnostic utility in incorporating performance-based measure of everyday functioning in evaluations of HAND as performance-based measures require tremendous investment of time, training, and resources relative to that of self-report measures, making their use less practical in busy clinical settings. While ultimately it is up to individual clinicians to decide whether they believe the benefits of administration of performance-based assessments in addition to self-reports to outweigh the costs, it is important that they be cognizant of the degree of concordance between performance-based and self-report measures corroborated by multiple studies when making such calculations. Additionally, the clinical and diagnostic interpretations of discordant self-report and performance-based classifications remain equivocal; thus, the inclusion of both measures does not necessarily increase diagnostic accuracy, as has been suggested in some prior work [16].
Interestingly, HIV diagnosis was not significantly associated with neuropsychological performance and while there was a trending difference in TFLS scores between HIV-infected and uninfected participants, mean scores for both groups were well within normal limits. Though cognitive impairment observed in HIV-infected individuals has become less severe in the HAART era [39–41], studies continue to report higher rates of mild cognitive and functional impairment in HIV-infected, as compared to HIV-uninfected, individuals [42, 43]. While results from the present study do not support this notion, it is possible that our analyses did not have adequate statistical power to detect relationships that may exist between HIV diagnosis and performance on the measures administered. On the other hand, the socio-demographic characteristics of our sample are representative of the HIV epidemic in the US, and the fact that all participants had a controlled viral load makes these findings particularly relevant to the contemporary clinical practice in the US and other parts of the world, where access to ART is standard and viremia suppression an achievable and realistic goal in most ART-adherent patients.
Differences among neuropsychological diagnostic groups in performance on the TFLS were independent from HCV infection in our adjusted analyses. However, it is intriguing that none of the participants in the asymptomatic CI group had detectable HCV RNA, while 40% of symptomatic CI participants did. On one hand, this finding could be an artifact of the small sample size of participants with CI Alternatively, it may suggest that HCV infection plays an active role in the pathophysiology of both neuropsychological and functional impairment, perhaps by perpetuating systemic inflammatory cascades in HIV-infected patients with controlled HIV viremia. Whereas past studies have linked HCV infection with neuropsychological impairment [37, 38], future adequately powered studies should evaluate the relationship between HCV infection and functional impairment in HAND.
The ability of neuropsychologically impaired individuals to accurately assess one’s own functional abilities may be an important factor in an individual’s prognosis, survival, and quality of life, with prior studies suggesting links between impaired self-awareness and decreased likelihood to seek medical attention and weaker adherence to a medication regimen [44]. Thus, while HIV-infected patients with ANI and MND may exhibit slight neuropsychological deficits, and functional deficits in the case of MND, the potential preservation of self-awareness in both groups may contribute to the health, safety, and quality of life for these individuals.
Our study was not without limitations. The present results and inferences about real-world functional abilities are informative insofar as performance on the TFLS is indicative of real-world functional ability. While prior studies have demonstrated good ecological validity of TFLS [32–34], the most ecologically valid way to measure real-world functional abilities in controlled test settings remains to be elucidated. Comparisons between neuropsychological diagnostic groups may be limited by the accuracy of diagnoses themselves. Individuals with elevated BDI-II scores had to report more complaints to be considered impaired on self-report. However, in the context of HIV infection, the direction of the relationship between self-reported impairments and depression is unknown. Furthermore, some of the same pathophysiological mechanisms that are hypothesized to cause HAND (e.g., inflammation, immune activation) have also been implied in the pathophysiology of depression [45]. Thus, further work is needed to elucidate how depression levels should be optimally incorporated in diagnostic algorithms for HAND. Comparisons between neuropsychological diagnostic groups may also be limited by our relatively small numbers of asymptomatic (n = 25) and symptomatic (n = 10) CI participants in our sample. However, the sample size in the present study is considerably larger than those in prior studies [14, 46, 47], making statistical power a relative strength of the present study. Similarly, analyses comparing sample characteristics of HIV-infected and uninfected participants may have been limited in their ability to detect any true differences that could exist between groups in behavioral risk factors for NP or functional impairment (e.g., history of drug or alcohol abuse), though our recruitment methods mitigate the possibility of extant group differences in such behavioral risk factors. Finally, our study is limited by a lack of informant reports, which were requested of all participants, but received for only four participants with MND and three participants with ANI. The difficulty and low response rate in obtaining informant reports in the present study appears to be the rule rather than an exception as similar observations had been made in prior studies of functional impairment in HIV-infected individuals (see “Introduction” section), therefore questioning the plausibility of obtaining informant reports in a highly stigmatized condition such as HIV/AIDS.
In conclusion, results from the present study suggest that individuals meeting research criteria for asymptomatic CI may truly be asymptomatic, thus supporting the validity of current AAN diagnostic categories of HAND. The concordance between self-report measures and performance-based measures of everyday functioning suggests that the addition of performance-based measures in evaluations of HAND would not significantly affect patients’ diagnoses, and that the current reliance on self-reports to examine everyday functioning is justified. Though additional information on everyday functioning gleamed through administration of performance-based measures certainly has the potential to be clinically informative, brief and inexpensive performance-based measures with high ecological validity have yet to be applied to HAND research and diagnosis. Until such measures are utilized, the 77% concordance between performance-based and self-report measures may yield the clinical benefits of relying on self-report measures alone greater than the clinical costs of incorporating both performance-based and self-report measures. Future studies should further explore factors contributing to discrepancies between functional ability and neuropsychological performance in the context of HIV infection. It is unknown why within a group of individuals exhibiting a similar magnitude of neuropsychological impairments, some experience functional difficulties while others do not. Future studies should assess differences in other IADLs (e.g., driving abilities) among HAND diagnostic groups to further elucidate the functional realms that may be deleteriously affected in HAND. Finally, longitudinal studies using both performance-based and self-report measures could further elucidate intra-individual relationships between self-awareness and neuropsychological performance over time.
Acknowledgements
This study was funded by National Institutes of Health (NIH) intramural research grants awarded to Avindra Nath (clinicaltrials.gov study identifier: NCT01875588).
Footnotes
Conflict of interest None of the authors have any conflicts of interest to disclose.
Ethical Approval All procedures performed in studies involving human participants were in accordance with the ethical standards of the institution (NIH) and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent It was obtained from all participants included in the study; all consent forms can be made available upon request.
References
- 1.Janssen RS, Cornblath DR, Epstein LG, Foa RP. Nomenclature and research case definitions for neurologic manifestations of human immunodeficiency virus-type 1 (HIV-1) infection. Neurology. 1991;41(6):778–85. [DOI] [PubMed] [Google Scholar]
- 2.Antinori A, Arendt G, Becker JT, Brew BJ, Byrd DA, Cherner M, et al. Updated research nosology for HIV-associated neurocognitive disorders. Neurology. 2007;69:1789–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Heaton RK, Clifford DB, Franklin DR, Woods SP, Ake C, Vaida F, et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy. Neurology. 2010;75:2087–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grov C, Golub SA, Parsons JT, Brennan M, Karpiak SE. Loneliness and HIV-related stigma explain depression among older HIV-positive adults. AIDS Care. 2010;22(5):630–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rao D, Pryor JB, Gaddist BW, Mayer R. Stigma, secrecy, and discrimination: ethnic/ratioal differences in the concerns of people living with HIV/AIDS. AIDS Behav. 2008;12(2):265–71. [DOI] [PubMed] [Google Scholar]
- 6.Emlet CA. An examination of the social networks and social isolation in older and younger adults living with HIV/AIDS. Health Soc Work. 2006;31(4):299–308. [DOI] [PubMed] [Google Scholar]
- 7.Van Gorp WG, Satz P, Hinkin CH, Seines O, Miller EN, McArthur J, et al. Metacognition in HIV-1 seropositive asymptomatic individuals: self-ratings versus objective neuropsychological performance. J Clin Exp Neuropsychol. 1991;13(5):812–9. [DOI] [PubMed] [Google Scholar]
- 8.Wilkins JW, Robertson KR, Snyder CR, Robertson WK, Van der Horst C, Hall CD. Implications of self-reported cognitive and motor dysfunction in HIV-positive patients. Am J Psychiatry. 1991;148:641–3. [DOI] [PubMed] [Google Scholar]
- 9.Hinkin CH, Van Gorp WG, Satz P, Marcotte T, Durvasula RS, Wood S, et al. Actual versus self-reported cognitive dysfunction in HIV-1 infection: memory-metamemory dissociations. J Clin Exp Neuropsychol. 1996;18(3):431–43. [DOI] [PubMed] [Google Scholar]
- 10.Thames AD, Becker BW, Marcotte T, Hines L, Foley JM, Ramezani A, et al. Depression, cognition, and self-appraisal of functional abilities in HIV: an examination of subjective appraisal versus objective performance. Clin Neuropsychol. 2011;25(2):224–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Juengst S, Skidmore E, Pramuka M, McCue M, Becker JT. Factors contributing to impaired self-awareness of cognitive functioning in an HIV positive and at-risk population. Disabil Rehabil. 2012;34(1): 19–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Moore LH, Van Gorp WG, Hinkin CH, Swales T, Satz P. Subjective complaints versus actual cognitive deficits in predominantly symptomatic HIV-1 seropositive individuals. J Neuropsychiatry Clin Neurosci. 1997;9:37–44. [DOI] [PubMed] [Google Scholar]
- 13.Schifitto G, Kieburtz K, McDermott MP, McArthur J, Marder K, Sacktor N, et al. Clinical trials in HIV-associated cognitive impairment: cognitive and functional outcomes. Neurology. 2001;56(3):415–8. [DOI] [PubMed] [Google Scholar]
- 14.Chiao S, Rosen HJ, Nicolas K, Wendelken LA, Alcantar O, Rankin KP, et al. Deficits in self-awareness impact the diagnosis of Asymptomatic Neurocognitive Impairment in HIV. AIDS Res Hum Retrovir. 2013;29(6):949–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gandhi NS, Skolasky RL, Peters KB, Moxley RT, Creighton J, Roosa HV, et al. A comparison of performance-based measures of function in HIV-associated neurocognitive disorders. J Neurovirol. 2011;17:159–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Blackstone K, Moore DJ, Heaton RK, Franklin DR, Woods SP, Clifford DB, et al. Diagnosing symptomatic HIV-associated neurocognitive disorders: self-report versus performance-based assessment of everyday functioning. J Int Neuropsychol Soc. 2012;18(1):79–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Feher EP, Mahurin RK, Inbody SB, Crook TH, Pirozzolo FJ. Anosognosia in Alzheimer’s disease. Neuropsychiatry Neuropsychol Behav Neurol. 1991;4:136–46. [Google Scholar]
- 18.Gil R, Arroyo-Anllo EM, Ingrand P, Gil M, Neau JP, Omon C, et al. Self-consciousness and Alzheimer’s disease. Acta Neurol Scand. 2001;104:296–300. [DOI] [PubMed] [Google Scholar]
- 19.McDaniel KD, Edland SD, Heyman A. Relationship between level of insight and severity of dementia in Alzheimer’s disease. Alzheimer Dis Assoc Disord. 1995;9:101–4. [DOI] [PubMed] [Google Scholar]
- 20.Aidala A, Havens J, Mellins CA, Dodds S, Whetten K, Martin DJ, et al. Development and validation of the Client Diagnostic Questionnaire (CDQ): a mental health screening tool for use in HIV/AIDS service settings. Psychol Health Med. 2004;9(3):362–80. [Google Scholar]
- 21.Beck AT. Beck depression inventory manual. San Antonio: Psychological Corporation; 1992. [Google Scholar]
- 22.Green P. Green’s medical symptom validity test (MSVT) for Microsoft Windows (User Manual). Edmonton: Green’s Publishing; 2004. [Google Scholar]
- 23.Schretlen DJ, Testa SM, Pearlson GD. Calibrated neuropsychological normative system (professional manual). Lutz: Psychological Assessment Resources; 2010. [Google Scholar]
- 24.Heaton RK, Miller SW, Taylor MJ, Grant I. Revised comprehensive norms for an Expanded Halstead–Reitan battery: demographically adjusted neuropsychological norms for African American and Caucasian adults (professional manual). Lutz: Psychological Assessment Resources; 2004. [Google Scholar]
- 25.Heaton RK, Taylor MJ, Manly J, Tulsky DS. Demographic effects and use of demographically corrected norms with the WAIS-III and WMS-in. In: Tulsky DS, Saklofske DH, Chelune GJ, et al. , editors. Clinical interpretation of the WAIS-in and WMS-IIL Burlington: Academic Press; 2003. p. 181–210. [Google Scholar]
- 26.Woods SP, Rippeth JD, Frol AB, Levy JK, Ryan E, Soukup VM, et al. Interrater reliability of clinical ratings and neurocognitive diagnoses in HIV. J Clin Exp Neuropsychol. 2004;26(6):759–78. [DOI] [PubMed] [Google Scholar]
- 27.Chelune GJ, Heaton RK, Lehman RA. Neuropsychological and personality correlates of patients’ complaints of disability. In: Goldstein G, Tarter RE, editors. Advances in clinical neuropsychology. USA: Springer; 1986. p. 95–126. [Google Scholar]
- 28.Richardson-Vejlgaard R, Dawes S, Heaton RK, Bell MD. Validity of cognitive complaints in substance-abusing patients and non-clinical controls: the Patient’s Assessment of Own Functioning Inventory (PAOF1). Psychiatry Res. 2009;169(1):70–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bell MJ, Terhorst L, Bender CM. Psychometric analysis of the Patient Assessment of Own Functioning Inventory in women with breast cancer. J Nurs Meas. 2013;21(2):320–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Heaton RK, Marcotte T, Mindt MR, Sadek J, Moore DJ, Bentley H, et al. The impact of HTV-associated neuropsychological impairment on everyday functioning. J Int Neuropsychol Soc. 2004; 10(3):317–31. [DOI] [PubMed] [Google Scholar]
- 31.Cullum CM, Weiner M, Saine K. Texas Functional Living Scale: examiner’s manual. San Antonio: Pearson; 2009. [Google Scholar]
- 32.Cullum CM, Saine K, Lynette C, Martin-Cook K, Gray K, Weiner M. Performance-based instrument to assess functional capacity in dementia: the Texas Functional Living Scale. Neuropsychiatry Neuropsychol Behav Neurol. 2001;14(2):103–8. [PubMed] [Google Scholar]
- 33.Drozdick LW, Cullum CM. Expanding the ecological validity of WAIS-IV and WMS-IV with the Texas Functional Living Scale. Assessment. 2010;18(2):141–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Weiner M, Davis B, Martin-Cook K, Hynan LS, Saine K, Cullum CM. A direct functional measure to help ascertain optimal level of resident care. Am J Alzheimers Dis Other Dement. 2007;22:355–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sacktor N, Skolasky RL, Seaberg E, Munro C, Becker JT, Martin E, et al. Prevalence of HIV-associated neurocognitive disorders in the Multicenter Aids Cohort study. Neurology. 2016;86(4):334–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cysique LA, Brew BJ. Prevalence of non-confounded HIV-associated neurocognitive impairment in the context of plasma HIV RNA suppression. J Neurovirol. 2011;17(2):176–83. [DOI] [PubMed] [Google Scholar]
- 37.Perry W, Hilsabeck RC, Hassanein TL Cognitive dysfunction in chronic Hepatitis C: a review. Dig Dis Sci. 2008;53(2):307–21. [DOI] [PubMed] [Google Scholar]
- 38.Hilsabeck RC, Perry W, Hassanein TI. Neuropsychological impairment in patients with chronic Hepatitis C. Hepatology. 2002;35(2):440–6. [DOI] [PubMed] [Google Scholar]
- 39.Cysique LA, Brew BJ. Neuropsychological functioning and antiretroviral treatment in HIV/AIDS: a review. Neuropsychol Rev. 2009;19:169–85. [DOI] [PubMed] [Google Scholar]
- 40.Sacktor N, McDermott MP, Marder K, Schifitto G, Seines O, McArthur J, et al. HIV-associated cognitive impairment before and after the advent of combination therapy. J Neurovirol. 2002;8(2): 136–42. [DOI] [PubMed] [Google Scholar]
- 41.Sacktor N, Lyles RH, Skolasky RL, Kleeberger C, Seines O, Miller E. HIV associated neurologic disease incidence changes: multicenter AIDS cohort study, 1990–1998. Neurology. 2001;56(2):257–60. [DOI] [PubMed] [Google Scholar]
- 42.Becker JT, Lopez OL, Dew MA, Aizenstein HJ. Prevalence of cognitive disorders differs as a function of age in HIV virus infection. AIDS. 2004;18:11–8. [PubMed] [Google Scholar]
- 43.Carey CI, Woods SP, Gonzalez R, Conover E, Marcotte T, Grant I, et al. Predictive validity of global deficit scores in detecting neuropsychological impairment in HIV infection. J Clin Exp Neuropsychol. 2004;26(3):307–19. [DOI] [PubMed] [Google Scholar]
- 44.Nurmi ME, Jehkonen M. Recognition and rehabilitation of impaired awareness of illness. Duodecim. 2015;131(3):228–34. [PubMed] [Google Scholar]
- 45.Raison C, Capuron L, Miller A. Cytokines sing the blues: inflammation and the pathogenesis of depression. Trends Immunol. 2006;27(1):24–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thames AD, Kim MS, Becker BW, Foley JM, Hines LJ, Singer EJ, et al. Medication and finance management among HIV-infected adults: the impact of age and cognition. J Clin Exp Neuropsychol. 2011;33(2):200–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mindt MR, Chemer M, Marcotte TD, Moore DJ, Bentley H, Esquivel MM, et al. The functional impact of HIV-associated neuropsychological impairment in Spanish-speaking adults: a pilot study. J Clin Exp Neuropsychol. 2003;25(1):122–32. [DOI] [PMC free article] [PubMed] [Google Scholar]


