Abstract
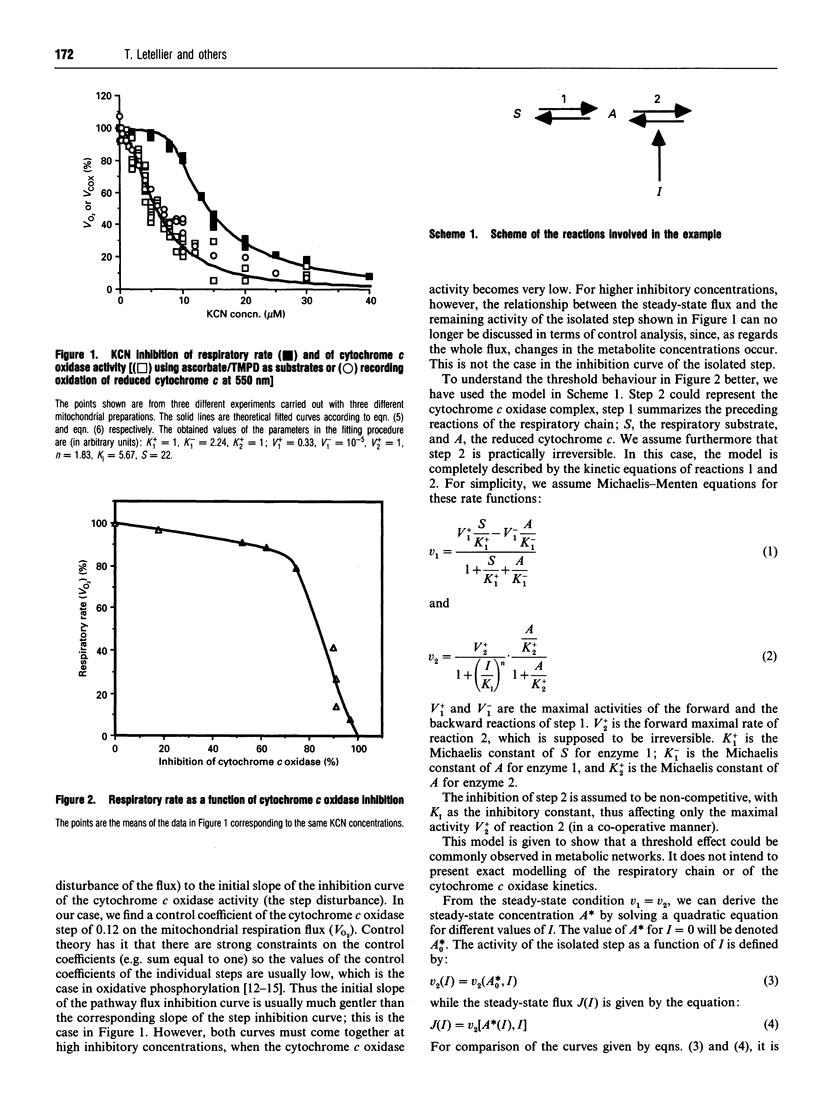
Threshold effects in the expression of metabolic diseases have often been observed in mitochondrial pathologies, i.e. the clinical demonstration of the disease appears only when the activity of a step has been reduced to a rather low level. We show experimentally that an inhibition of cytochrome c oxidase activity by cyanide, simulating a defect in this step, leads to a decrease in mitochondrial respiration which then exhibits a threshold behaviour similar to that observed in mitochondrial diseases. We discuss this behaviour in terms of metabolic control theory and construct a mathematical model simulating this behaviour.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- CHANCE B., WILLIAMS G. R. The respiratory chain and oxidative phosphorylation. Adv Enzymol Relat Subj Biochem. 1956;17:65–134. doi: 10.1002/9780470122624.ch2. [DOI] [PubMed] [Google Scholar]
- Chomyn A., Martinuzzi A., Yoneda M., Daga A., Hurko O., Johns D., Lai S. T., Nonaka I., Angelini C., Attardi G. MELAS mutation in mtDNA binding site for transcription termination factor causes defects in protein synthesis and in respiration but no change in levels of upstream and downstream mature transcripts. Proc Natl Acad Sci U S A. 1992 May 15;89(10):4221–4225. doi: 10.1073/pnas.89.10.4221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint H. J., Porteous D. J., Kacser H. Control of the flux in the arginine pathway of Neurospora crassa. The flux from citrulline to arginine. Biochem J. 1980 Jul 15;190(1):1–15. doi: 10.1042/bj1900001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint H. J., Tateson R. W., Barthelmess I. B., Porteous D. J., Donachie W. D., Kacser H. Control of the flux in the arginine pathway of Neurospora crassa. Modulations of enzyme activity and concentration. Biochem J. 1981 Nov 15;200(2):231–246. doi: 10.1042/bj2000231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groen A. K., Wanders R. J., Westerhoff H. V., van der Meer R., Tager J. M. Quantification of the contribution of various steps to the control of mitochondrial respiration. J Biol Chem. 1982 Mar 25;257(6):2754–2757. [PubMed] [Google Scholar]
- Hayashi J., Ohta S., Kikuchi A., Takemitsu M., Goto Y., Nonaka I. Introduction of disease-related mitochondrial DNA deletions into HeLa cells lacking mitochondrial DNA results in mitochondrial dysfunction. Proc Natl Acad Sci U S A. 1991 Dec 1;88(23):10614–10618. doi: 10.1073/pnas.88.23.10614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinrich R., Rapoport T. A. A linear steady-state treatment of enzymatic chains. General properties, control and effector strength. Eur J Biochem. 1974 Feb 15;42(1):89–95. doi: 10.1111/j.1432-1033.1974.tb03318.x. [DOI] [PubMed] [Google Scholar]
- Holzhütter H. G., Colosimo A. SIMFIT: a microcomputer software-toolkit for modelistic studies in biochemistry. Comput Appl Biosci. 1990 Jan;6(1):23–28. doi: 10.1093/bioinformatics/6.1.23. [DOI] [PubMed] [Google Scholar]
- Kacser H., Burns J. A. The control of flux. Symp Soc Exp Biol. 1973;27:65–104. [PubMed] [Google Scholar]
- Kacser H., Burns J. A. The molecular basis of dominance. Genetics. 1981 Mar-Apr;97(3-4):639–666. doi: 10.1093/genetics/97.3-4.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letellier T., Malgat M., Mazat J. P. Control of oxidative phosphorylation in rat muscle mitochondria: implications for mitochondrial myopathies. Biochim Biophys Acta. 1993 Feb 8;1141(1):58–64. doi: 10.1016/0005-2728(93)90189-m. [DOI] [PubMed] [Google Scholar]
- Morgan-Hughes J. A., Hayes D. J., Clark J. B., Landon D. N., Swash M., Stark R. J., Rudge P. Mitochondrial encephalomyopathies: biochemical studies in two cases revealing defects in the respiratory chain. Brain. 1982 Sep;105(Pt 3):553–582. doi: 10.1093/brain/105.3.553. [DOI] [PubMed] [Google Scholar]
- Reder C. Metabolic control theory: a structural approach. J Theor Biol. 1988 Nov 21;135(2):175–201. doi: 10.1016/s0022-5193(88)80073-0. [DOI] [PubMed] [Google Scholar]
- Shoffner J. M., Lott M. T., Lezza A. M., Seibel P., Ballinger S. W., Wallace D. C. Myoclonic epilepsy and ragged-red fiber disease (MERRF) is associated with a mitochondrial DNA tRNA(Lys) mutation. Cell. 1990 Jun 15;61(6):931–937. doi: 10.1016/0092-8674(90)90059-n. [DOI] [PubMed] [Google Scholar]
- Tager J. M., Wanders R. J., Groen A. K., Kunz W., Bohnensack R., Küster U., Letko G., Böhme G., Duszynski J., Wojtczak L. Control of mitochondrial respiration. FEBS Lett. 1983 Jan 10;151(1):1–9. doi: 10.1016/0014-5793(83)80330-5. [DOI] [PubMed] [Google Scholar]
- Taylor R. W., Birch-Machin M. A., Bartlett K., Lowerson S. A., Turnbull D. M. The control of mitochondrial oxidations by complex III in rat muscle and liver mitochondria. Implications for our understanding of mitochondrial cytopathies in man. J Biol Chem. 1994 Feb 4;269(5):3523–3528. [PubMed] [Google Scholar]
- Taylor R. W., Birch-Machin M. A., Bartlett K., Turnbull D. M. Succinate-cytochrome c reductase: assessment of its value in the investigation of defects of the respiratory chain. Biochim Biophys Acta. 1993 Jun 19;1181(3):261–265. doi: 10.1016/0925-4439(93)90030-5. [DOI] [PubMed] [Google Scholar]


