Abstract
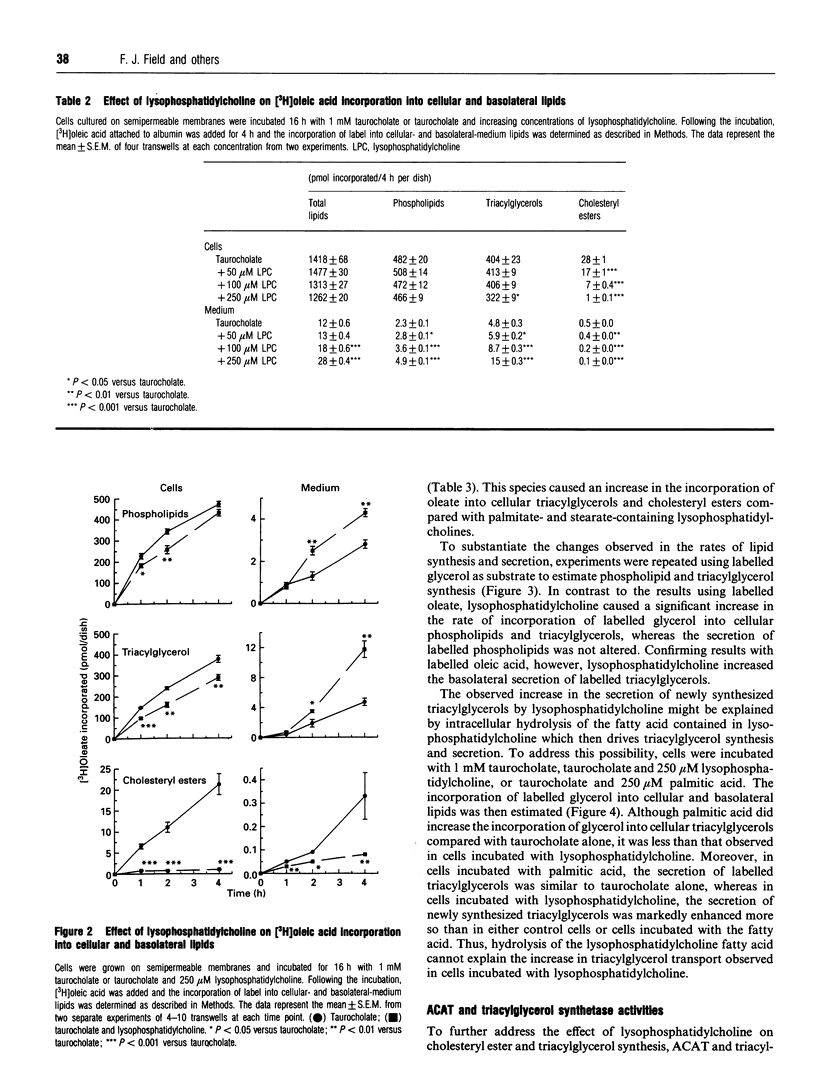
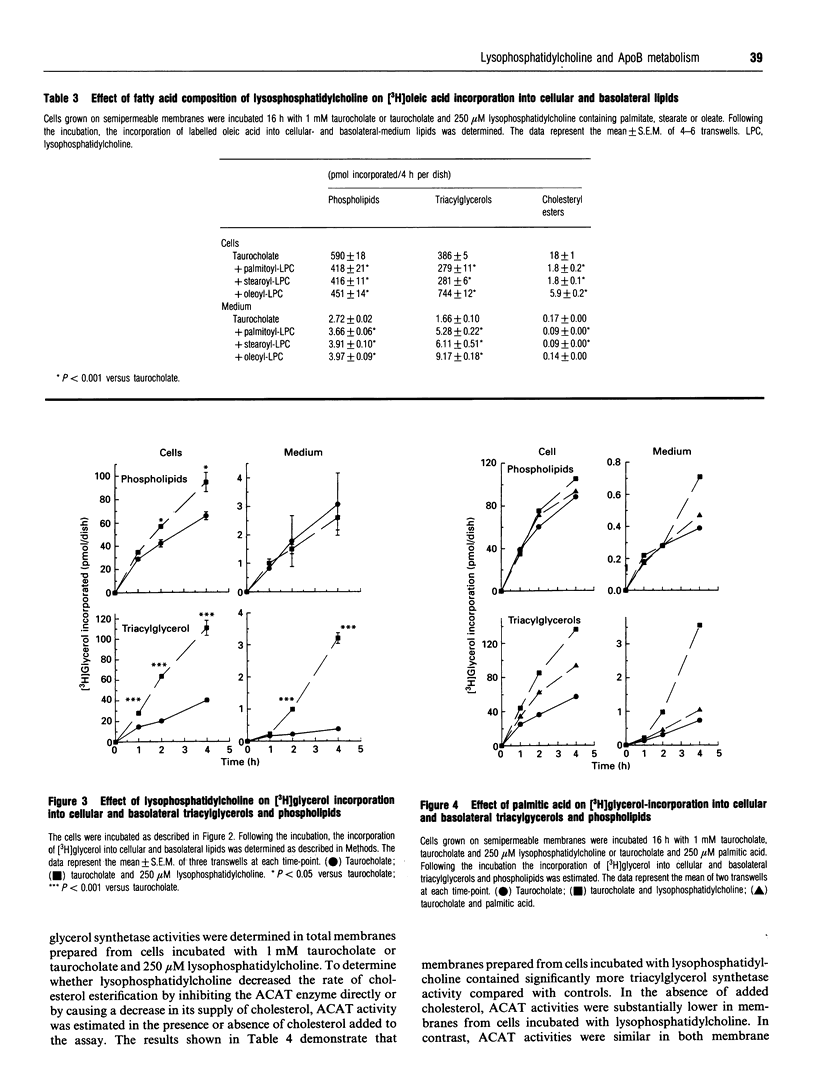
To address the effect of lysophosphatidylcholine on triacylglycerol transport in intestine, CaCo-2 cells, grown on semipermeable supports, were incubated with lysophosphatidylcholine solubilized in 1 mM taurocholate. [14C]Palmitoyllysophosphatidylcholine was readily taken up and incorporated predominantly into cellular phospholipids, particularly phosphatidylcholine. Twenty-five percent of the label was found in triacylglycerols. Compared with labelled cellular phospholipids, labelled triacylglycerols were preferentially secreted. Lysophosphatidylcholine caused a profound decrease in cholesteryl ester synthesis and secretion, whereas cellular triacylglycerol mass and triacylglycerol synthesis and secretion were increased. The effect was more pronounced with oleoyllysophosphatidylcholine than with either palmitoyl- or stearyl-lysophosphatidylcholine. Lysophosphatidylcholine increased the secretion of immunoreactive and newly-synthesized apoprotein B (apoB) without altering the rate of apoB synthesis. Thus, luminal lysophosphatidylcholine and/or its uptake decreases cholesterol esterification and secretion, but increases triacylglycerol synthesis and secretion, triacylglycerol mass accumulation and the secretion of apoB by CaCo-2 cells.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chalvardjian A., Rudnicki E. Determination of lipid phosphorus in the nanomolar range. Anal Biochem. 1970 Jul;36(1):225–226. doi: 10.1016/0003-2697(70)90352-0. [DOI] [PubMed] [Google Scholar]
- Clark S. B. Chylomicron composition during duodenal triglyceride and lecithin infusion. Am J Physiol. 1978 Aug;235(2):E183–E190. doi: 10.1152/ajpendo.1978.235.2.E183. [DOI] [PubMed] [Google Scholar]
- Dixon J. L., Furukawa S., Ginsberg H. N. Oleate stimulates secretion of apolipoprotein B-containing lipoproteins from Hep G2 cells by inhibiting early intracellular degradation of apolipoprotein B. J Biol Chem. 1991 Mar 15;266(8):5080–5086. [PubMed] [Google Scholar]
- Dixon J. L., Ginsberg H. N. Regulation of hepatic secretion of apolipoprotein B-containing lipoproteins: information obtained from cultured liver cells. J Lipid Res. 1993 Feb;34(2):167–179. [PubMed] [Google Scholar]
- Field F. J., Albright E., Mathur S. N. Regulation of cholesterol esterification by micellar cholesterol in CaCo-2 cells. J Lipid Res. 1987 Sep;28(9):1057–1066. [PubMed] [Google Scholar]
- Field F. J., Salome R. G. Effect of dietary fat saturation, cholesterol and cholestyramine on acyl-CoA: cholesterol acyltransferase activity in rabbit intestinal microsomes. Biochim Biophys Acta. 1982 Sep 14;712(3):557–570. doi: 10.1016/0005-2760(82)90284-3. [DOI] [PubMed] [Google Scholar]
- Kam N. T., Albright E., Mathur S. N., Field F. J. Inhibition of acylcoenzyme A:cholesterol acyltransferase activity in CaCo-2 cells results in intracellular triglyceride accumulation. J Lipid Res. 1989 Mar;30(3):371–377. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lange Y., Strebel F., Steck T. L. Role of the plasma membrane in cholesterol esterification in rat hepatoma cells. J Biol Chem. 1993 Jul 5;268(19):13838–13843. [PubMed] [Google Scholar]
- Mansbach C. M., 2nd, Arnold A., Cox M. A. Factors influencing triacylglycerol delivery into mesenteric lymph. Am J Physiol. 1985 Nov;249(5 Pt 1):G642–G648. doi: 10.1152/ajpgi.1985.249.5.G642. [DOI] [PubMed] [Google Scholar]
- Murthy S., Albright E., Mathur S. N., Davidson N. O., Field F. J. Apolipoprotein B mRNA abundance is decreased by eicosapentaenoic acid in CaCo-2 cells. Effect on the synthesis and secretion of apolipoprotein B. Arterioscler Thromb. 1992 Jun;12(6):691–700. doi: 10.1161/01.atv.12.6.691. [DOI] [PubMed] [Google Scholar]
- Murthy S., Albright E., Mathur S. N., Field F. J. Effect of eicosapentaenoic acid on triacylglycerol transport in CaCo-2 cells. Biochim Biophys Acta. 1990 Jul 16;1045(2):147–155. doi: 10.1016/0005-2760(90)90144-m. [DOI] [PubMed] [Google Scholar]
- Nilsson A., Borgström B. Absorption and metabolism of lecithin and lysolecithin by intestinal slices. Biochim Biophys Acta. 1967 Apr 4;137(2):240–254. doi: 10.1016/0005-2760(67)90100-2. [DOI] [PubMed] [Google Scholar]
- Nilsson A. Intestinal absorption of lecithin and lysolecithin by lymph fistula rats. Biochim Biophys Acta. 1968 Mar 4;152(2):379–390. doi: 10.1016/0005-2760(68)90047-7. [DOI] [PubMed] [Google Scholar]
- O'Doherty P. J., Kakis G., Kuksis A. Role of luminal lecithin in intestinal fat absorption. Lipids. 1973 May;8(5):249–255. doi: 10.1007/BF02531899. [DOI] [PubMed] [Google Scholar]
- O'Doherty P. J., Yousef I. M., Kuksis A. Effect of phosphatidylcholine on triacylglycerol synthesis in rat intestinal mucosa. Can J Biochem. 1974 Sep;52(9):726–733. doi: 10.1139/o74-103. [DOI] [PubMed] [Google Scholar]
- Scow R. O., Stein Y., Stein O. Incorporation of dietary lecithin and lysolecithin into lymph chylomicrons in the rat. J Biol Chem. 1967 Nov 10;242(21):4919–4924. [PubMed] [Google Scholar]
- Tso P., Lam J., Simmonds W. J. The importance of the lysophosphatidylcholine and choline moiety of bile phosphatidylcholine in lymphatic transport of fat. Biochim Biophys Acta. 1978 Mar 30;528(3):364–372. doi: 10.1016/0005-2760(78)90025-5. [DOI] [PubMed] [Google Scholar]
- Yao Z. M., Vance D. E. The active synthesis of phosphatidylcholine is required for very low density lipoprotein secretion from rat hepatocytes. J Biol Chem. 1988 Feb 25;263(6):2998–3004. [PubMed] [Google Scholar]