Abstract
Purpose
Regional nodal irradiation (RNI) for breast cancer yields improvements in disease outcomes, yet comprehensive target coverage often increases cardiac radiation therapy (RT) dose. Volumetric modulated arc therapy (VMAT) may mitigate high-dose cardiac exposure, although it often increases the volume of low-dose exposure. The cardiac implications of this dosimetric configuration (in contrast to historic 3D conformal techniques) remain uncertain.
Methods and Materials
Eligible patients receiving adjuvant RNI using VMAT for locoregional breast cancer were prospectively enrolled in an IRB-approved study. Echocardiograms were performed prior to RT, at the conclusion of RT, and 6 months following RT. Echocardiographic parameters were measured by a single reader and measures were compared pre- and post-RT via the signed-rank test. Changes in echocardiographic parameters over time were compared to mean and max heart doses via the Spearman correlation test.
Results
Among 19 evaluable patients (median age 38 years), 89% (n = 17) received doxorubicin and 37% (n = 7) received trastuzumab/pertuzumab combination therapy. All patients received VMAT-based whole-breast/chest wall and RNI. The average mean heart dose was 456 cGy (range, 187-697 cGy) and the average max heart dose was 3001 cGy (1560-4793 cGy). Among salient echocardiographic parameters, no significant decrement in cardiac function was observed when comparing pre-RT to 6 months post-RT: mean left ventricular ejection fraction (LVEF) was 61.8% (SD 4.4%) pre-RT and 62.7% (SD 3.8%) 6 months post-RT (P = .493); mean global longitudinal strain (GLS) was –19.3% (SD 2.2%) pre-RT and –19.6% (SD 1.8%) 6 months post-RT (P = .627). No individual patient exhibited reduced LVEF or sustained decrement in GLS. No correlations were observed for changes in LVEF or GLS when compared to mean or maximum heart doses (P > .1 for all).
Conclusions
VMAT for left-sided RNI yielded no significant early decrement in echocardiographic parameters of cardiac function, including LVEF and GLS, within this limited cohort. No patient exhibited significant LVEF changes, and none exhibited sustained decrements in GLS. VMAT may be a reasonable approach to cardiac avoidance in patients requiring RNI, including those receiving anthracyclines and HER2-directed therapy. Larger cohorts with longer follow-ups will be needed to validate these findings.
Introduction
Over 3 million cancer survivors in the United States have undergone treatment comprising some measure of cardiac radiation therapy (RT) exposure. Several series have demonstrated that chest/breast RT may result in increased cardiovascular (CV) morbidity, including cardiomyopathy, coronary disease, and valvular dysfunction.1, 2, 3, 4, 5, 6, 7, 8 Moreover, whereas the traditional understanding of RT-associated cardiotoxicity assumed a long latency period for the manifestation of cardiac events via accelerated fibrotic changes, recent studies suggest that early cardiotoxicity can be observed.1,3
Many epidemiologic analyses that associate RT with CV toxicity are based on outdated RT delivery techniques (eg, conventional, 2D, or 3D conformal RT) and systemic therapies. The applicability of these findings to current RT approaches and systemic agents remains unknown, and uncertainty persists regarding the mechanisms and predictors of cardiotoxicity.9 Moreover, with the implementation of advanced RT delivery technologies (eg surface imaging, respiratory gating, protons, etc.), improvements in cardiac dose reduction remain of hypothetical benefit with limited clinical indications of long-term superiority.10 Further elucidation of RT-associated CV toxicity is of particular importance given a series of landmark studies that prompted increased utilization of regional nodal irradiation (RNI).11,12 This comprehensive breast RT approach typically yields elevated cardiac exposure because of the proximity of the internal mammary nodal (IMN) target to the heart.
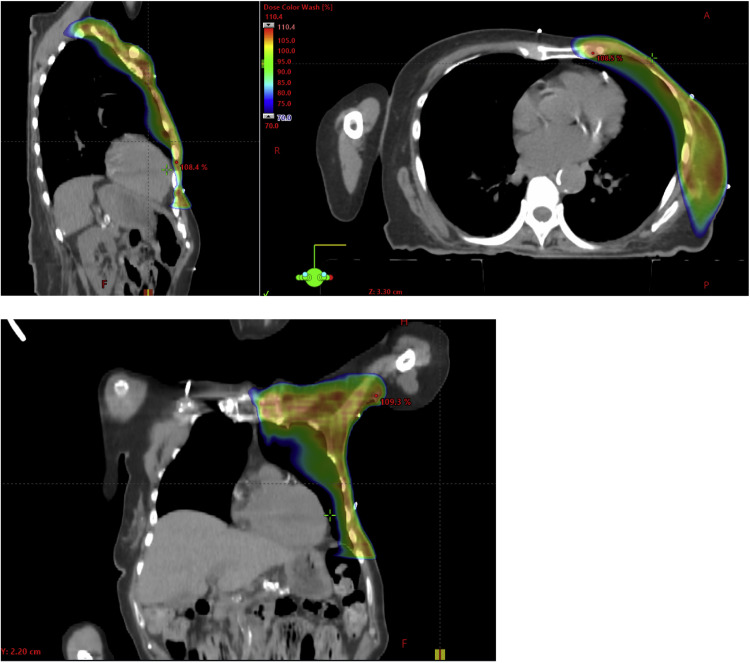
Volumetric modulated arc therapy (VMAT) is one of several approaches designed to reduce cardiac exposure from RNI (and thereby mitigate putative CV risk),13, 14, 15 yet the cardiac implications of this technique are unknown. Indeed, although VMAT improves the conformality of high-dose RT regions, it necessarily exposes more nontarget tissue to low doses of radiation (Fig. 1). This typically reduces the maximum heart dose as intended, while simultaneously increasing the mean heart dose (MHD) with, as yet, indeterminate consequences. MHDs approximating 7 Gy have been reported with VMAT, as has ipsilateral lung V5 approaching 100%.14
Figure 1.
Representative dose distribution of a volumetric modulated arc therapy (VMAT) plan targeting the left breast and regional lymph nodes.
Echocardiography is the primary modality for assessing structural and functional changes secondary to the cardiotoxic effects of RT. Myocardial strain imaging using speckle-tracking echocardiography is an established tool for quantitative measurement of cardiac contractile function.16 Myocardial strain parameters such as global longitudinal strain (GLS) are more sensitive compared with left ventricular ejection fraction (LVEF) for detecting early stages of cardiotoxicity related to RT and have established prognostic value in multiple treatment settings.17, 18, 19 Myocardial work (MW) indices are novel echocardiography-based measures that have been proposed to provide a complementary assessment of cardiac function over GLS; however, the value of MW indices to detect the cardiotoxic effects of RT is unknown.
Thus, we undertook a prospective single-arm cohort study to quantify the longitudinal change in myocardial strain and MW indices among breast cancer patients receiving RNI via VMAT. Imaging was conducted pre-RT, at the end of RT, and 6 months following RT, to assess for putative subclinical changes in cardiac function arising from treatment.
Methods and Materials
Study design
This was a single-arm prospective cohort study designed to evaluate changes in myocardial function based on 2D echocardiographic indices among breast cancer patients receiving left-sided RNI using VMAT. Patients with invasive locoregional breast cancer (ie, nonmetastatic) were prospectively screened and approached for participation in this institutional review board–approved study by the treating physician(s). Eligible and consenting patients were enrolled at the time of treatment consent for adjuvant RNI using VMAT (inclusive of RT to the axilla, supraclavicular fossa, IMN basin, and chest wall/breast to a total dose of 50 Gy in 25 fractions). Relevant dosimetric constraints include an ipsilateral lung V20 ≤ 27% and V10 ≤ 60%, contralateral lung V20 ≤ 5%, heart V25 ≤ 3%, heart mean ≤ 6 Gy, heart Dmax ≤ 50 Gy, left anterior descending artery Dmax ≤ 25 Gy. Planning was conducted using the Varian Eclipse Treatment Planning System (Varian Medical Systems). Thereafter, echocardiograms were performed at 3 timepoints: prior to RT (typically between the time of simulation and treatment start), at the conclusion of RT (typically within 1-2 weeks of the last RT fraction), and 6 months following RT. All study patients provided fully informed written consent prior to participation. This study protocol was approved by the Memorial Sloan Kettering Cancer Center institutional review board and was compliant with the Health Insurance Portability and Accountability Act.
Echocardiography
Two-dimensional and Doppler echocardiography was performed using a Vivid E9 ultrasound scanner (GE Medical Systems) at the following timepoints: pre-RT, post-RT, and 6 months post-RT. LVEF was calculated from the apical 4- and 2-chamber views using a modified Simpson biplane method according to the American Society of Echocardiography guidelines.20 During the standard 2D echocardiogram, apical 2-, 3-, and 4-chamber views and short-axis views at the midpapillary level were acquired at a frame rate of 40 to 80 frames per second. Speckle-tracking strain analysis was performed offline to calculate peak systolic GLS, global circumferential strain, and global radial strain (Echopac, GE Medical) as previously described.21 All strain analysis was performed by a board-certified cardiologist (A.Y.), independent of the clinical interpretation of the 2D echocardiogram. Strain analysis was conducted in a manner blinded to the patient RT plan or dosimetry.
MW indices were measured with commercially available software (GE Echopac), as previously described.22 The peak systolic LV pressure was estimated based on brachial systolic blood pressure measurement. Timing of mitral and aortic valve opening and closure, measured by pulse-wave Doppler and confirmed by 2D assessment in the apical long-axis view, were used to define isovolumic and systolic ejection periods. The software then used the peak systolic LV pressure, valvular timing events, and GLS measurements to automatically generate left ventricular pressure strain loops. The following indices were then calculated: (1) global work index (GWI), which represents the total area of the pressure strain loop from mitral valve closure to opening; (2) global constructive work (GCW), which represents MW during systolic shortening and negative work (ie, work performed by the blood on the ventricle) during lengthening in isovolumic relaxation; (3) global wasted work (GWW), which represents negative work during lengthening in systole and MW during shortening in isovolumic relaxation; and (4) global work efficiency, which is calculated as GCW divided by the sum of GCW and GWW.
Statistical analysis
Continuous measures were summarized as mean and standard deviation and categorical measures were summarized as frequency and percent. Differences in echocardiographic parameters among timepoints (pre-RT, post-RT, and 6 months post-RT) were explored by analysis of variance for repeated measures. Differences between the pre-RT and post-RT timepoints were further compared using the Wilcoxon signed-rank test. The Spearman's correlation test was used to assess the association between mean and max heart doses as a function of changes in echocardiography-based parameters from pre-RT to 6 months post-RT.
Interobserver and intraobserver variability for MW indices was assessed in 20 randomly selected studies by the intraclass correlation coefficient (ICC) and standard error of measurement (SEM). For interobserver variability, studies were remeasured by 2 observers blinded to one another's findings (A.Y. and J.L.). For intraobserver variability, studies were remeasured by the same observer at different points in time.
Results
Patient and treatment characteristics
From 2018 to 2020, 19 patients were enrolled in this prospective study (median age 38 years; range, 26-61) (Table 1). Of these, 16 (84%) underwent mastectomy, 3 (16%) underwent breast conservation, 4 (21%) had sentinel lymph node biopsy alone, and 15 (79%) had a complete axillary lymph node dissection. Axillary evaluations yielded a median of 2 positive nodes (range, 0-18), after recovering a median of 16 nodes in total (range, 2-27). Most patients received doxorubicin-based chemotherapy (n = 17; 89%), and 7 (39%) further received HER2-directed therapy comprising trastuzumab and pertuzumab. All patients received RNI using the VMAT technique, yielding a median MHD of 475 cGy (range, 187-698) and a median maximum heart dose of 2999 cGy (range, 1560-4793). No patients exhibited cardiac events over the follow-up period and none were admitted for cardiac diagnoses.
Table 1.
Patient and treatment characteristics
| N = 19 | Median (range) |
|---|---|
| Age (y) | 38 (26-61) |
| Surgery | |
| Breast conservation | 3 |
| Mastectomy | 16 |
| Sentinel node biopsy | 4 |
| Axillary lymph node dissection | 15 |
| Postmastectomy reconstruction | 12 |
| Axillary nodes removed | 16 (2-27) |
| Lymph nodes positive | 2 (0-18) |
| Systemic therapy | |
| Neoadjuvant chemotherapy | 15 |
| Adjuvant chemotherapy | 2 |
| Doxorubicin | 17 |
| HER2-directed therapy | 7 |
| Trastuzumab | 7 |
| Pertuzumab | 7 |
| Hormone therapy | 13 |
| Dosimetry | |
| Cardiac mean (cGy) | 475 (187-698) |
| Cardiac max (cGy) | 2999 (1560-4793) |
| Laterality | |
| Right | 1 |
| Left | 18 |
Longitudinal changes in cardiac structure and function
Echocardiographic parameters of cardiac structure and function over time are shown in Table 2. LVEF was normal at pre-RT (61.8% ± 4.4%) and did not significantly change post-RT (62.2% ± 4.3%) or at 6 months post-RT (62.7% ± 3.8%). Similarly, no changes in diastolic indices were observed during or after RT. We then measured indices of myocardial strain to assess for subclinical changes in cardiac function during RT. There were no significant differences from pre-RT to 6 months post-RT in GLS (–19.3% ± 2.2% vs –19.6% ± 1.8%, P = .627), global circumferential strain (–16.6% ± 3.6% vs –16.2% ± 3.5%, P = .986), or global radial strain (45.8 ± 23.9% vs 43.3 ± 15.7%, P = .958).
Table 2.
Longitudinal changes in conventional echocardiographic parameters during breast RT
| Pre-RT (n = 19) |
Post-RT (n = 19) |
6-month F/U (n = 18) | P value* | |
|---|---|---|---|---|
| LVEDD/BSA | 2.6 ± 0.3 | 2.6 ± 0.3 | 2.7 ± 0.2 | .818 |
| LV mass index | 74.7 ± 13.1 | 67.0 ± 15.4 | 68.3 ± 12.3 | .213 |
| LA volume index | 25.3 ± 5.8 | 21.4 ± 6.6 | 22.2 ± 7.3 | .200 |
| LVEF (%) | .493 | |||
| Median (IQR) | 61 (58.5, 65.5) | 62 (59.5, 65) | 63 (61, 65) | |
| Range | 54-69 | 55-72 | 53-68 | |
| Mean ± SD | 61.8 ± 4.4 | 62.2 ± 4.3 | 62.7 ± 3.8 | |
| Global longitudinal strain (%) | –19.3 ± 2.2 | –19.2 ± 1.8 | –19.6 ± 1.8 | .627 |
| A3C | –18.9 ± 2.4 | –19.6 ± 1.8 | –19.7 ± 2.6 | |
| A4C | –19.3 ± 2.5 | –19.2 ± 2.1 | –19.4 ± 2.0 | |
| A2C | –19.7 ± 2.3 | –19.2 ± 2.4 | –19.8 ± 2.0 | |
| Global circumferential strain (%) | –16.6 ± 3.6 | –17.0 ± 2.7 | –16.2 ± 3.5 | .986 |
| Global radial strain (%) | 45.8 ± 23.9 | 44.9 ± 10.4 | 43.3 ± 15.7 | .958 |
| Diastolic parameters | ||||
| Mitral E velocity (cm/s) | 77.3 ± 17.5 | 74.8 ± 15.7 | 77.2 ± 20.4 | .775 |
| Mitral A velocity (cm/s) | 60.5 ± 16.3 | 61.7 ± 11.8 | 62.9 ± 13.8 | .648 |
| Mitral E/A ratio | 1.3 ± 0.4 | 1.3 ± 0.4 | 1.3 ± 0.4 | .417 |
| Septal e’ (cm/s) | 10.3 ± 2.55 | 9.7 ± 3.0 | 10.0 ± 2.9 | .669 |
| Lateral e’ (cm/s) | 13.0 ± 3.2 | 11.7 ± 2.9 | 12.1 ± 3.6 | .323 |
| Septal E/e’ ratio | 7.6 ± 1.9 | 8.0 ± 2.3 | 8.0 ± 1.5 | .274 |
| Lateral E/e’ ratio | 6.2 ± 1.7 | 6.5 ± 1.5 | 6.6 ± 1.2 | .191 |
Abbreviations: A2C = apical 2-chamber; A3C = apical 3-chamber; A4C = apical 4-chamber; BSA = body surface area; IQR = interquartile range; LA = left atrial; LV = left ventricular; LVEDD = left ventricular end diastolic diameter; LVEF = Left ventricular ejection fraction; RT = radiation therapy.
Wilcoxon signed-rank test between pre-RT and 6-month F/U (N = 17).
Values are mean ± SD.
MW indices of the study population over time are shown in Table 3. All patients had normal MW indices pre-RT based on published normal reference values.23 There were no significant differences in GWI, GCW, GWW, or global work efficiency from pre-RT to 6 months post-RT.
Table 3.
Longitudinal changes in myocardial work indices during breast RT
| Pre-RT (n = 19) | Post-RT (n = 19) | 6-month F/U (n = 18) | P value* | |
|---|---|---|---|---|
| Global work index, mm Hg% | 1834 ± 238 | 1847 ± 238 | 1885 ± 271 | .356 |
| Global constructive work, mm Hg% | 1992 ± 260 | 2026 ± 243 | 2073 ± 226 | .093 |
| Global wasted work, mm Hg% | 76 ± 39 | 71 ± 50 | 71 ± 28 | .287 |
| Global work efficiency, % | 96 ± 2 | 96 ± 2 | 96 ± 1 | .280 |
Abbreviations: RT = radiation therapy.
Wilcoxon signed-rank test between pre-RT and 6-month F/U (N = 17).
Values are mean ± SD.
Echocardiographic parameters and RT dose
We evaluated for associations between cardiac dosimetry and longitudinal changes in echocardiographic parameters of cardiac function during the study period (from pre-RT to 6 months post-RT). There was no correlation between change in LVEF (pre-RT to 6 months post-RT) and mean (r = –0.02, P = .931) or max (r = –0.02, P = .948) heart dose. Similarly, we found no correlation between change in GLS (pre-RT to 6 months post-RT) and mean (r = 0.35, P = .15) or max (r = 0.18, P = .475) heart dose.
We then evaluated for associations between cardiac dosimetry and longitudinal changes in MW indices. There was a significant correlation between MHD and decline in GWI from pre-RT to 6 months post-RT (r= –0.51, P = .037), but not max heart dose and GWI (r = –0.13, P = .619). GWI declined from pre-RT to 6 months post-RT for participants in the highest quartile of MHD (1787 mm Hg% to 1732 mm Hg%) but was stable or increased for participants in the first (1750 mm Hg% to 1904 mm Hg%), second (1937 mm Hg% to 2114 mm Hg%), and third (1883 mm Hg% to 1880 mm Hg%) quartiles. There was no correlation between mean or max heart dose with other MW indices including GCW, GWW, or GWI.
Reproducibility
Interobserver and intraobserver variability for MW indices as measured by ICC and SEM are summarized in Table 4. GWI showed the best repeatability and reproducibility with an intraobserver and interobserver ICC of 0.94 (95% CI, 0.87-0.97) and 0.94 (95% CI, 0.85-0.97), respectively, and an intraobserver and interobserver SEM of 45.6 mm Hg% and 47.4 mm Hg%, respectively.
Table 4.
Interobserver and intraobserver variability for myocardial work indices
| Interobserver variability |
Intraobserver variability |
|||||
|---|---|---|---|---|---|---|
| ICC | 95% CI | SEM | ICC | 95% CI | SEM | |
| Global work index (mm Hg%) | 0.94 | 0.867-0.974 | 45.64 | 0.936 | 0.853-0.972 | 47.43 |
| Global constructive work (mm Hg%) | 0.899 | 0.79-0.953 | 55.35 | 0.909 | 0.795-0.961 | 56.08 |
| Global wasted work (mm Hg%) | 0.849 | 0.661-0.937 | 14.5 | 0.829 | 0.64-0.923 | 14.2 |
| Global work efficiency (%) | 0.887 | 0.741-0.953 | 0.53 | 0.863 | 0.695-0.942 | 0.55 |
Abbreviations: ICC = intraclass correlation coefficient; SEM = standard error of measurement.
Discussion
In this prospective study of women with breast cancer receiving multimodal treatment including RNI with VMAT, we observed no early reductions in echocardiographic parameters of LV systolic function including LVEF, GLS, or MW indices. The absence of significant changes in echocardiography-based measures of cardiac contractile function early after RNI with VMAT supports further investigation of this contemporary RT technique which is being increasingly adopted. In addition, we found no correlation between mean or max cardiac RT dose and longitudinal changes in LVEF or GLS. Finally, we observed an association between MHD and change in GWI from pre-RT to 6 months post-RT. The utility of assessing MW (in particular, GWI) to detect subclinical cardiotoxicity of breast RT may therefore warrant further investigation.
The cardiac implications of adjuvant treatment have been studied intently since the early days of postmastectomy RT. Indeed, owing to the limited precision of RT in prior eras, several reports raised the specter of significant RT-associated CV toxicity.2,3,7 More recently, as the multidisciplinary management of breast cancer has improved in lockstep with radiotherapeutic technologies, several studies now support the increasing benefit of comprehensive RT for a variety of breast cancer presentations.11,12,24 Two landmark trials, MA.20 and EORTC 22922, both demonstrated that RNI confers a significant 10-year disease-free survival benefit for those presenting even with a limited axillary disease burden. In parallel, the DBCG-IMN study affirmed the importance of including the IMN basin as a component of RNI, demonstrating an overall survival benefit when this region was encompassed in the radiotherapeutic target despite the elevated cardiac dose with this approach. Thus, an increasing proportion of breast cancer patients now receive RNI, along with the concomitant elevation in cardiac RT exposure, although without impinging on cardiac outcomes. And although none of these studies demonstrated elevated cardiac mortality with IMN coverage, most literature to date suggests that increasing cardiac dose can only be detrimental to long-term outcomes. To that end, the VMAT approach studied here is being increasingly employed for RNI, although as discussed above, lowering the maximum heart dose as afforded by VMAT often comes at the expense of raising the MHD with unclear implications that require further study.
Overall, the present findings are consistent with those from our previous study which showed no evidence of early subclinical LV systolic dysfunction after contemporary breast RT in women with HER2-positive breast cancer.25 We further extend these results in the current longitudinal cohort study with the inclusion of patients treated with RNI using VMAT. Studies assessing the subclinical cardiotoxicity of RT have previously demonstrated radiation-induced global and segmental changes in myocardial strain and strain rate in patients with breast cancer; however, breast RT in these studies was delivered using tangential photon beams with typical 3D-conformal dosimetry.26, 27, 28 More advanced breast RT techniques, such as VMAT, intensity modulated RT, and proton therapy are now available and are increasingly used in standard care. Notably, VMAT is typically associated with an increase in cardiac mean dose, in addition to pulmonary V5, V10, and V20.14
MW indices, which are measures of left ventricular mechanics that incorporate myocardial deformation with afterload and are less susceptible to fluctuations in blood pressure, have diagnostic and prognostic significance in several CV conditions including chemotherapy-related cardiac dysfunction.29,30 This study further contributes to the current literature by assessing for changes in MW in the setting of breast RT. Overall, no significant change in MW indices was observed in our patients throughout the RT treatment period. However, among participants in the highest quartile of MHD, there was a decline in GWI. These findings possibly suggest that GWI can detect early subclinical changes in myocardial function related to RT. Further studies are needed to replicate these findings and determine whether GWI provides incremental diagnostic and prognostic value to established echocardiographic parameters.
These findings must be interpreted in the context of the study design. Although our prospective cohort study was of limited size, it nonetheless provides novel data on the potential role of MW for the assessment of radiation-induced cardiotoxicity. Whether changes in MW indices are associated with clinical CV outcomes will require larger studies with longer follow-ups. In addition, most patients in this study were treated with anthracyclines and/or HER2-targeted therapies which can cause short- and long-term effects on cardiac function. Therefore, we cannot exclude the potential confounding effects of prior/concurrent breast cancer treatments on echocardiographic changes during RT. However, the absence of significant changes in LVEF or GLS within 6 months of radiation in the context of other cardiotoxic cancer treatment exposures suggests that radiation with VMAT does not enhance the cardiotoxic effects of other cardiotoxic breast cancer treatments.
In conclusion, in this small prospective study, VMAT for patients requiring RNI yielded no observable early decrement in echocardiographic parameters of cardiac function including LVEF and GLS. The cardiac implications of VMAT for patients requiring RNI merit further study, including among those receiving anthracyclines and HER2-directed therapy. Assessment of MW indices such as GWI may be useful to identify subclinical cardiotoxicity in patients receiving among the highest allowable MHDs and warrants further consideration. Larger cohorts with longer follow-ups will be needed to validate these findings.
Disclosures
The authors have no relevant conflicts of interest to disclose.
Acknowledgments
The authors would like to acknowledge Anthony Abaya and Michael Sullivan for their support with data management.
Footnotes
This study was presented at the 2022 American Society for Radiation Oncology (ASTRO) Annual Meeting, San Antonio, TX, October 24-28, 2022.
Sources of support: This study was supported by a grant from the Imaging and Radiation Sciences Program at Memorial Sloan Kettering Cancer Center, the Lois Green Fund, and in part by NIH/NCI Cancer Center Support Grant No. P30CA008748.
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
- 1.Bradley JD, Paulus R, Komaki R, et al. Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): A randomised, two-by-two factorial phase 3 study. Lancet Oncol. 2015;16:187–199. doi: 10.1016/S1470-2045(14)71207-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Darby SC, Cutter DJ, Boerma M, et al. Radiation-related heart disease: Current knowledge and future prospects. Int J Radiat Oncol Biol Phys. 2010;76:656–665. doi: 10.1016/j.ijrobp.2009.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med. 2013;368:987–998. doi: 10.1056/NEJMoa1209825. [DOI] [PubMed] [Google Scholar]
- 4.Demirci S, Nam J, Hubbs JL, Nguyen T, Marks LB. Radiation-induced cardiac toxicity after therapy for breast cancer: interaction between treatment era and follow-up duration. Int J Radiat Oncol Biol Phys. 2009;73:980–987. doi: 10.1016/j.ijrobp.2008.11.016. [DOI] [PubMed] [Google Scholar]
- 5.Emens LA, Davidson NE. The follow-up of breast cancer. Semin Oncol. 2003;30:338–348. doi: 10.1016/s0093-7754(03)00094-0. [DOI] [PubMed] [Google Scholar]
- 6.Evans SB, Sioshansi S, Moran MS, Hiatt J, Price LL, Wazer DE. Prevalence of poor cardiac anatomy in carcinoma of the breast treated with whole-breast radiotherapy: Reconciling modern cardiac dosimetry with cardiac mortality data. Am J Clin Oncol. 2012;35:587–592. doi: 10.1097/COC.0b013e31822d9cf6. [DOI] [PubMed] [Google Scholar]
- 7.Giordano SH, Kuo Y-F, Freeman JL, Buchholz TA, Hortobagyi GN, Goodwin JS. Risk of cardiac death after adjuvant radiotherapy for breast cancer. J Natl Cancer Inst. 2005;97:419–424. doi: 10.1093/jnci/dji067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van den Bogaard VAB, Ta BDP, van der Schaaf A, et al. Validation and modification of a prediction model for acute cardiac events in patients with breast cancer treated with radiotherapy based on three-dimensional dose distributions to cardiac substructures. J Clin Oncol. 2017;35:1171–1178. doi: 10.1200/JCO.2016.69.8480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bekelman JE, Lu H, Pugh S, et al. Pragmatic randomised clinical trial of proton versus photon therapy for patients with non-metastatic breast cancer: The Radiotherapy Comparative Effectiveness (RadComp) Consortium trial protocol. BMJ Open. 2019;9 doi: 10.1136/bmjopen-2018-025556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Braunstein LZ, Warren LEG. Policy implications of proton radiation therapy: Toward an evidence-based approach for implementing novel oncologic technologies. Int J Radiat Oncol Biol Phys. 2016;95:560–561. doi: 10.1016/j.ijrobp.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 11.Poortmans PM, Weltens C, Fortpied C, et al. Internal mammary and medial supraclavicular lymph node chain irradiation in stage I-III breast cancer (EORTC 22922/10925): 15-year results of a randomised, phase 3 trial. Lancet Oncol. 2020;21:1602–1610. doi: 10.1016/S1470-2045(20)30472-1. [DOI] [PubMed] [Google Scholar]
- 12.Whelan TJ, Olivotto IA, Parulekar WR, et al. Regional nodal irradiation in early-stage breast cancer. N Engl J Med. 2015;373:307–316. doi: 10.1056/NEJMoa1415340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bernstein MB, Walker K, Gillespie E, et al. Bilateral regional nodal irradiation using volumetric modulated arc therapy: dosimetric analysis and feasibility. Pract Radiat Oncol. 2022;12:189–194. doi: 10.1016/j.prro.2021.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kuo L, Ballangrud ÅM, Ho AY, Mechalakos JG, Li G, Hong L. A VMAT planning technique for locally advanced breast cancer patients with expander or implant reconstructions requiring comprehensive postmastectomy radiation therapy. Med Dosim. 2019;44:150–154. doi: 10.1016/j.meddos.2018.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dumane VA, Saksornchai K, Zhou Y, Hong L, Powell S, Ho AY. Reduction in low-dose to normal tissue with the addition of deep inspiration breath hold (DIBH) to volumetric modulated arc therapy (VMAT) in breast cancer patients with implant reconstruction receiving regional nodal irradiation. Radiat Oncol. 2018;13:187. doi: 10.1186/s13014-018-1132-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu JE, Barac A, Thavendiranathan P, Scherrer-Crosbie M. Strain imaging in cardio-oncology. JACC CardioOncol. 2020;2:677–689. doi: 10.1016/j.jaccao.2020.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thavendiranathan P, Poulin F, Lim K-D, Plana JC, Woo A, Marwick TH. Use of myocardial strain imaging by echocardiography for the early detection of cardiotoxicity in patients during and after cancer chemotherapy: A systematic review. J Am Coll Cardiol. 2014;63:2751–2768. doi: 10.1016/j.jacc.2014.01.073. [DOI] [PubMed] [Google Scholar]
- 18.Negishi K, Negishi T, Hare JL, Haluska BA, Plana JC, Marwick TH. Independent and incremental value of deformation indices for prediction of trastuzumab-induced cardiotoxicity. J Am Soc Echocardiogr. 2013;26:493–498. doi: 10.1016/j.echo.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 19.Oikonomou EK, Kokkinidis DG, Kampaktsis PN, et al. Assessment of prognostic value of left ventricular global longitudinal strain for early prediction of chemotherapy-induced cardiotoxicity: A systematic review and meta-analysis. JAMA Cardiol. 2019;4:1007–1018. doi: 10.1001/jamacardio.2019.2952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16:233–270. doi: 10.1093/ehjci/jev014. [DOI] [PubMed] [Google Scholar]
- 21.Yu AF, Raikhelkar J, Zabor EC, et al. Two-dimensional speckle tracking echocardiography detects subclinical left ventricular systolic dysfunction among adult survivors of childhood, adolescent, and young adult cancer. BioMed Res Int. 2016;2016 doi: 10.1155/2016/9363951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Russell K, Eriksen M, Aaberge L, et al. A novel clinical method for quantification of regional left ventricular pressure-strain loop area: A non-invasive index of myocardial work. Eur Heart J. 2012;33:724–733. doi: 10.1093/eurheartj/ehs016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Manganaro R, Marchetta S, Dulgheru R, et al. Echocardiographic reference ranges for normal non-invasive myocardial work indices: Results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging. 2019;20:582–590. doi: 10.1093/ehjci/jey188. [DOI] [PubMed] [Google Scholar]
- 24.Thorsen LBJ, Offersen BV, Danø H, et al. DBCG-IMN: A population-based cohort study on the effect of internal mammary node irradiation in early node-positive breast cancer. J Clin Oncol. 2016;34:314–320. doi: 10.1200/JCO.2015.63.6456. [DOI] [PubMed] [Google Scholar]
- 25.Yu AF, Ho AY, Braunstein LZ, et al. Assessment of early radiation-induced changes in left ventricular function by myocardial strain imaging after breast radiation therapy. J Am Soc Echocardiogr. 2019;32:521–528. doi: 10.1016/j.echo.2018.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lo Q, Hee L, Batumalai V, et al. Subclinical cardiac dysfunction detected by strain imaging during breast irradiation with persistent changes 6 weeks after treatment. Int J Radiat Oncol Biol Phys. 2015;92:268–276. doi: 10.1016/j.ijrobp.2014.11.016. [DOI] [PubMed] [Google Scholar]
- 27.Lo Q, Hee L, Batumalai V, et al. Strain imaging detects dose-dependent segmental cardiac dysfunction in the acute phase after breast irradiation. Int J Radiat Oncol Biol Phys. 2017;99:182–190. doi: 10.1016/j.ijrobp.2017.05.030. [DOI] [PubMed] [Google Scholar]
- 28.Trivedi SJ, Tang S, Byth K, et al. Segmental cardiac radiation dose determines magnitude of regional cardiac dysfunction. J Am Heart Assoc. 2021;10 doi: 10.1161/JAHA.120.019476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kosmala W, Negishi T, Thavendiranathan P, et al. Incremental value of myocardial work over global longitudinal strain in the surveillance for cancer-treatment-related cardiac dysfunction: A case-control study. J Clin Med. 2022:11. doi: 10.3390/jcm11040912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Calvillo-Argüelles O, Thampinathan B, Somerset E, et al. Diagnostic and prognostic value of myocardial work indices for identification of cancer therapy-related cardiotoxicity. JACC Cardiovasc Imaging. 2022;15:1361–1376. doi: 10.1016/j.jcmg.2022.02.027. [DOI] [PubMed] [Google Scholar]