Abstract
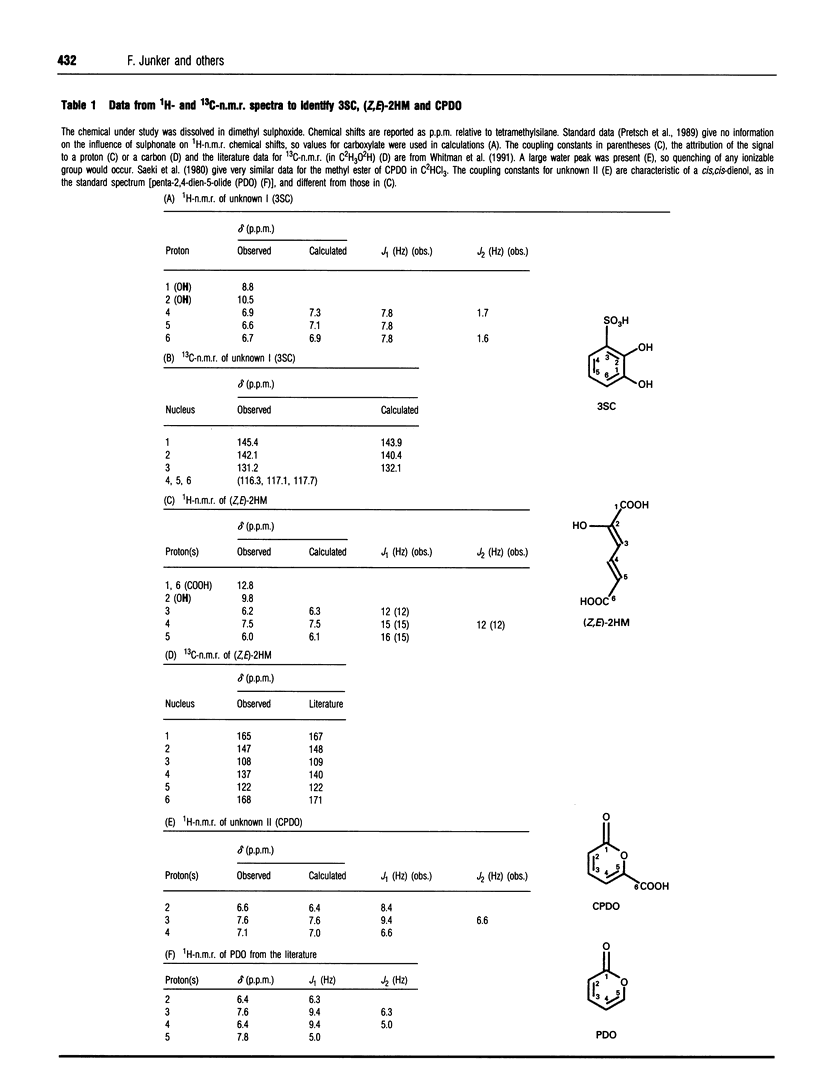
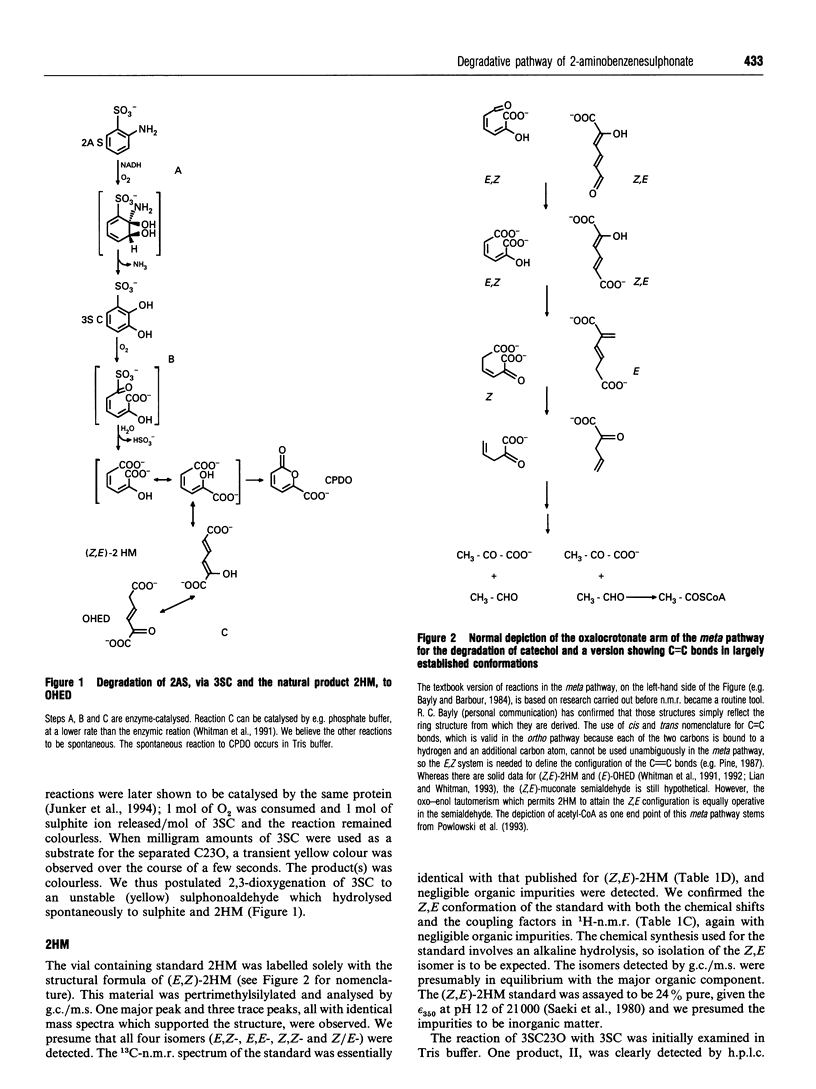
2-Aminobenzenesulphonic acid (2AS) is degraded by Alcaligenes sp. strain O-1 via a previously detected but unidentified intermediate. A mutant of strain O-1 was found to excrete this intermediate, which was isolated and identified by m.s., 1H- and 13C-n.m.r. as 3-sulphocatechol (3SC). Proteins from cell extracts of strain O-1 were separated by anion-exchange chromatography. A multicomponent oxygenase was observed to convert 1 mol each of NADH, O2 and 2AS into 1 mol each of 3SC, NH3 and NAD+. The enzyme presumably catalysed formation of the ring of a 2-amino-2,3-diol moiety, and elimination in the amino group led to a rearomatization. 3SC was further degraded via meta ring cleavage, which could be prevented by inactivation of the 3-sulphocatechol-2,3-dioxygenase (3SC23O) with 3-chlorocatechol. In Tris buffer, the separated 3SC23O catalysed the reaction of 1 mol each of 3SC and O2 involving a transient yellow intermediate, and release of 1 mol of sulphite and two organic products. The major product was identified by n.m.r. and by g.c./m.s. as 5-carboxypenta-2,4-dien-5-olide (CPDO), an indicator of formation of 2-hydroxymuconic acid (2HM). The second product was identified as the Z,E isomer of 2HM by comparison with authentic material. When the CPDO in the product mixture was chemically hydrolysed to (Z,E)-2HM, 1 mol of (Z,E)-2HM/mol of 3SC was observed. If oxygenation of 3SC by 3SC23O was carried out in phosphate buffer, only a single product was detected, a keto form of 2HM. This dioate was also formed from authentic (Z,E)-2HM in phosphate buffer. Formation of the natural product (Z,E)-2HM from the xenobiotic, 3SC, seems to involve oxygenation to the unstable 2-hydroxy-6-sulphonomuconic acid semialdehyde, which hydrolyses spontaneously to 2HM. There would appear to be at least one spontaneous reaction per enzyme reaction in this pathway.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Assinder S. J., Williams P. A. The TOL plasmids: determinants of the catabolism of toluene and the xylenes. Adv Microb Physiol. 1990;31:1–69. doi: 10.1016/s0065-2911(08)60119-8. [DOI] [PubMed] [Google Scholar]
- Bartels I., Knackmuss H. J., Reineke W. Suicide Inactivation of Catechol 2,3-Dioxygenase from Pseudomonas putida mt-2 by 3-Halocatechols. Appl Environ Microbiol. 1984 Mar;47(3):500–505. doi: 10.1128/aem.47.3.500-505.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernhardt F. H., Bill E., Trautwein A. X., Twilfer H. 4-Methoxybenzoate monooxygenase from Pseudomonas putida: isolation, biochemical properties, substrate specificity, and reaction mechanisms of the enzyme components. Methods Enzymol. 1988;161:281–294. doi: 10.1016/0076-6879(88)61031-7. [DOI] [PubMed] [Google Scholar]
- Bernhardt F. H., Erdin N., Staudinger H., Ullrich V. Interactions of substrates with a purified 4-methoxybenzoate monooxygenase system (O-demethylating) from Pseudomonas putida. Eur J Biochem. 1973 May;35(1):126–134. doi: 10.1111/j.1432-1033.1973.tb02818.x. [DOI] [PubMed] [Google Scholar]
- Bird J. A., Cain R. B. Microbial degradation of alkylbenzenesulphonates. Metabolism of homologues of short alkyl-chain length by an Alcaligenes sp. Biochem J. 1974 May;140(2):121–134. doi: 10.1042/bj1400121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brilon C., Beckmann W., Knackmuss H. J. Catabolism of Naphthalenesulfonic Acids by Pseudomonas sp. A3 and Pseudomonas sp. C22. Appl Environ Microbiol. 1981 Jul;42(1):44–55. doi: 10.1128/aem.42.1.44-55.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bünz P. V., Cook A. M. Dibenzofuran 4,4a-dioxygenase from Sphingomonas sp. strain RW1: angular dioxygenation by a three-component enzyme system. J Bacteriol. 1993 Oct;175(20):6467–6475. doi: 10.1128/jb.175.20.6467-6475.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L. H., Kenyon G. L., Curtin F., Harayama S., Bembenek M. E., Hajipour G., Whitman C. P. 4-Oxalocrotonate tautomerase, an enzyme composed of 62 amino acid residues per monomer. J Biol Chem. 1992 Sep 5;267(25):17716–17721. [PubMed] [Google Scholar]
- Eaton R. W., Ribbons D. W. Biotransformation of 3-methylphthalate by Micrococcus sp. strain 12B. J Gen Microbiol. 1987 Sep;133(9):2473–2476. doi: 10.1099/00221287-133-9-2473. [DOI] [PubMed] [Google Scholar]
- Engesser K. H., Strubel V., Christoglou K., Fischer P., Rast H. G. Dioxygenolytic cleavage of aryl ether bonds: 1,10-dihydro-1,10-dihydroxyfluoren-9-one, a novel arene dihydrodiol as evidence for angular dioxygenation of dibenzofuran. FEMS Microbiol Lett. 1989 Nov;53(1-2):205–209. doi: 10.1016/0378-1097(89)90392-3. [DOI] [PubMed] [Google Scholar]
- Feigel B. J., Knackmuss H. J. Syntrophic interactions during degradation of 4-aminobenzenesulfonic acid by a two species bacterial culture. Arch Microbiol. 1993;159(2):124–130. doi: 10.1007/BF00250271. [DOI] [PubMed] [Google Scholar]
- Fetzner S., Müller R., Lingens F. Purification and some properties of 2-halobenzoate 1,2-dioxygenase, a two-component enzyme system from Pseudomonas cepacia 2CBS. J Bacteriol. 1992 Jan;174(1):279–290. doi: 10.1128/jb.174.1.279-290.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukumori F., Hausinger R. P. Alcaligenes eutrophus JMP134 "2,4-dichlorophenoxyacetate monooxygenase" is an alpha-ketoglutarate-dependent dioxygenase. J Bacteriol. 1993 Apr;175(7):2083–2086. doi: 10.1128/jb.175.7.2083-2086.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harayama S., Rekik M., Ngai K. L., Ornston L. N. Physically associated enzymes produce and metabolize 2-hydroxy-2,4-dienoate, a chemically unstable intermediate formed in catechol metabolism via meta cleavage in Pseudomonas putida. J Bacteriol. 1989 Nov;171(11):6251–6258. doi: 10.1128/jb.171.11.6251-6258.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy S. I., Fewson C. A. Enzymes of the mandelate pathway in Bacterium N.C.I.B. 8250. Biochem J. 1968 Apr;107(4):497–506. doi: 10.1042/bj1070497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kersten P. J., Dagley S., Whittaker J. W., Arciero D. M., Lipscomb J. D. 2-pyrone-4,6-dicarboxylic acid, a catabolite of gallic acids in Pseudomonas species. J Bacteriol. 1982 Dec;152(3):1154–1162. doi: 10.1128/jb.152.3.1154-1162.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klecka G. M., Gibson D. T. Inhibition of catechol 2,3-dioxygenase from Pseudomonas putida by 3-chlorocatechol. Appl Environ Microbiol. 1981 May;41(5):1159–1165. doi: 10.1128/aem.41.5.1159-1165.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornberg A. Why purify enzymes? Methods Enzymol. 1990;182:1–5. doi: 10.1016/0076-6879(90)82003-k. [DOI] [PubMed] [Google Scholar]
- Locher H. H., Leisinger T., Cook A. M. 4-Sulphobenzoate 3,4-dioxygenase. Purification and properties of a desulphonative two-component enzyme system from Comamonas testosteroni T-2. Biochem J. 1991 Mar 15;274(Pt 3):833–842. doi: 10.1042/bj2740833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locher H. H., Leisinger T., Cook A. M. 4-Toluene sulfonate methyl-monooxygenase from Comamonas testosteroni T-2: purification and some properties of the oxygenase component. J Bacteriol. 1991 Jun;173(12):3741–3748. doi: 10.1128/jb.173.12.3741-3748.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Locher H. H., Leisinger T., Cook A. M. Degradation of p-toluenesulphonic acid via sidechain oxidation, desulphonation and meta ring cleavage in Pseudomonas (Comamonas) testosteroni T-2. J Gen Microbiol. 1989 Jul;135(7):1969–1978. doi: 10.1099/00221287-135-7-1969. [DOI] [PubMed] [Google Scholar]
- Markus A., Krekel D., Lingens F. Purification and some properties of component A of the 4-chlorophenylacetate 3,4-dioxygenase from Pseudomonas species strain CBS. J Biol Chem. 1986 Sep 25;261(27):12883–12888. [PubMed] [Google Scholar]
- Pfeifer F., Trüper H. G., Klein J., Schacht S. Degradation of diphenylether by Pseudomonas cepacia Et4: enzymatic release of phenol from 2,3-dihydroxydiphenylether. Arch Microbiol. 1993;159(4):323–329. doi: 10.1007/BF00290914. [DOI] [PubMed] [Google Scholar]
- Powlowski J. B., Dagley S., Massey V., Ballou D. P. Properties of anthranilate hydroxylase (deaminating), a flavoprotein from Trichosporon cutaneum. J Biol Chem. 1987 Jan 5;262(1):69–74. [PubMed] [Google Scholar]
- Powlowski J., Sahlman L., Shingler V. Purification and properties of the physically associated meta-cleavage pathway enzymes 4-hydroxy-2-ketovalerate aldolase and aldehyde dehydrogenase (acylating) from Pseudomonas sp. strain CF600. J Bacteriol. 1993 Jan;175(2):377–385. doi: 10.1128/jb.175.2.377-385.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saeki Y., Nozaki M., Senoh S. Cleavage of pyrogallol by non-heme iron-containing dioxygenases. J Biol Chem. 1980 Sep 25;255(18):8465–8471. [PubMed] [Google Scholar]
- Sariaslani F. S. Microbial cytochromes P-450 and xenobiotic metabolism. Adv Appl Microbiol. 1991;36:133–178. doi: 10.1016/s0065-2164(08)70453-2. [DOI] [PubMed] [Google Scholar]
- Spanggord R. J., Spain J. C., Nishino S. F., Mortelmans K. E. Biodegradation of 2,4-dinitrotoluene by a Pseudomonas sp. Appl Environ Microbiol. 1991 Nov;57(11):3200–3205. doi: 10.1128/aem.57.11.3200-3205.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suen W. C., Spain J. C. Cloning and characterization of Pseudomonas sp. strain DNT genes for 2,4-dinitrotoluene degradation. J Bacteriol. 1993 Mar;175(6):1831–1837. doi: 10.1128/jb.175.6.1831-1837.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thurnheer T., Zürrer D., Höglinger O., Leisinger T., Cook A. M. Initial steps in the degradation of benzene sulfonic acid, 4-toluene sulfonic acids, and orthanilic acid in Alcaligenes sp. strain O-1. Biodegradation. 1990;1(1):55–64. doi: 10.1007/BF00117051. [DOI] [PubMed] [Google Scholar]
- Topp E., Xun L. Y., Orser C. S. Biodegradation of the herbicide bromoxynil (3,5-dibromo-4-hydroxybenzonitrile) by purified pentachlorophenol hydroxylase and whole cells of Flavobacterium sp. strain ATCC 39723 is accompanied by cyanogenesis. Appl Environ Microbiol. 1992 Feb;58(2):502–506. doi: 10.1128/aem.58.2.502-506.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xun L., Topp E., Orser C. S. Diverse substrate range of a Flavobacterium pentachlorophenol hydroxylase and reaction stoichiometries. J Bacteriol. 1992 May;174(9):2898–2902. doi: 10.1128/jb.174.9.2898-2902.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamanian M., Mason J. R. Benzene dioxygenase in Pseudomonas putida. Subunit composition and immuno-cross-reactivity with other aromatic dioxygenases. Biochem J. 1987 Jun 15;244(3):611–616. doi: 10.1042/bj2440611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zürrer D., Cook A. M., Leisinger T. Microbial desulfonation of substituted naphthalenesulfonic acids and benzenesulfonic acids. Appl Environ Microbiol. 1987 Jul;53(7):1459–1463. doi: 10.1128/aem.53.7.1459-1463.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]


