Abstract
Early pregnancy loss (EPL) is the most common pregnancy complication, found in approximately 15% of all clinically recognized pregnancy complications. Up to date, various maternal as well as fetal factors are reported as a cause of EPLs. However, in approximately 50% of EPL cases, the exact cause is not clearly identified and these cases are referred as idiopathic.
The aim of our study was to examine the association of four distinct variants in the ANXA5 gene and two variants within the VEGFA gene in a cohort of women with EPLs from North Macedonia. This group was compared to a control group of women matched by ethnic background without pregnancy loss and at least one live birth. We also aimed to establish an effective and cost-efficient method for their detection based on multiplex single-base extension.
Among 190 women experiencing EPLs, and 190 samples from women without a history of pregnancy loss (control group), our results demonstrated a statistically significant prevalence of heterozygotes for the M2/ANXA5 haplotype in women with EPLs, compared to the control group (p=0.0006). In the analyses comparing genotypic frequencies for the variants in the VEGFA gene, higher frequencies were generally observed among women experiencing EPLs, however without statistical significance.
Our study aligns with multiple studies showing that M2 and M1 ANXA5 haplotypes are more prevalent in patients with pregnancy loss and presents an affordable genotyping technique for the specific ANXA5 and VEGFA variants.
Keywords: ANXA5, haplotypes, VEGFA, early pregnancy loss (EPL), multiplex single-base extension
INTRODUCTION
Early pregnancy loss (EPL), defined as a loss of the conceptus before the 12th week of gestation, is the most common pregnancy complication and is found in approximately 15% of all clinically recognized pregnancies [1]. About 5% of the couples trying for childbirth, experience recurrent pregnancy loss (RPL), a condition defined as two or more consecutive pregnancy losses, according to the European Society of Human Reproduction and Embryology (ESHRE) and American Society for Reproductive Medicine (ASRM) [2, 3], as well as three or more first trimester miscarriages, according to the Royal College of Obstetricians and Gynecologists, UK (RCOG) [4]. Up to now, various studies have reported that maternal as well as fetal factors may lead to RPL. Fetal chromosomal abnormalities are acknowledged as a significant contributor to pregnancy loss in approximately 50% of cases. This pattern is mirrored in our previous study concerning chromosomal abnormalities in early pregnancy losses (EPLs). In that study, chromosomal abnormalities were detected in 56.25% of uncontaminated products of conceptions (POCs), aligning with this observed trend [5]. Additionally, maternal endocrine dysregulations, autoimmune disorders, anatomical abnormalities, maternal thrombophilia, as well as genetic factors can contribute to RPL [6]. Concerning genetic factors, in a recent study that was designed and executed in our laboratory, we detected a high incidence of Joubert Syndrome among a group of EPLs (2.03%), indicating that fetal monogenic diseases can be a common cause of EPLs [7]. Still, in a large portion of RPL cases, the exact cause is not clearly identified, and these cases are referred as idiopathic RPL [8].
Angiogenesis is a physiological process through which new blood vessels are formed. Decreased angiogenesis has been associated with several adverse pregnancy outcomes, such as infertility, preeclampsia, miscarriage, intrauterine fetal distress/growth restriction, and in severe cases, fetal demise. The Annexin A5 protein, encoded by the ANXA5 gene, is found in abundance to hinder coagulation within the placenta and during the early angiogenesis. When calcium is present, Annexin A5 can attach to phosphatidylserine situated on the upper surface of syncytiotrophoblasts in the placenta to prevent clotting of maternal blood in the intervillous space. Additionally, the ANXA5 gene plays a crucial role in promoting epithelial repair, upholding placental integrity [9]. Studies have indicated that the activity of ANXA5 in placental tissue samples collected from patients with RPL was significantly diminished, accompanied by a suppressed ANXA5 expression. These findings imply that a decrease in Annexin A5 protein expression elevates the risk of RPL, especially in early pregnancy [10]. Bogdanova et al. first reported that the combination of certain single nucleotide changes: rs28717001 (c.-210A>C), rs28651243 (c.-184T>C), rs112782763 (c.-229G>A) and rs113588187 (c.-135G>A) in the promoter region of ANXA5 was associated with an increased risk of recurrent miscarriages. All four changes were defined as M2 haplotype, and the first two were designated as M1 haplotype [11]. As mentioned in a recent meta-analysis study [12], and a review article [13], dealing with the effect of the ANXA5 haplotypes on RPL, several case-control studies have shown a favorable correlation between the M2/ANXA5 haplotype and RPL across diverse ethnic populations, including cohorts from Germany [11, 14, 15], Japan [16], the United Kingdom [17] Italy [18] China [19] Malaysia [20] and Greece [21]. However, contrasting results were observed in studies involving the Chinese and Danish/Estonian ethnic groups. These groups did not reveal any association between the M2/ANXA5 haplotype and RPL [22, 23].
The human placenta is rich in angiogenic factors such as VEGF, standing out as the most potent stimulator of angiogenesis. VEGF plays a crucial role in endometrial readiness, implantation, and the development of placental and fetal blood vessels in early pregnancy, and in the vascular adaptation during pregnancy in the mother [24]. Several single nucleotide changes (SNPs) have been identified in the VEGFA gene, affecting VEGF-A activity and expression. The most significant finding in most studies is that presence of the c.-1154G/A (rs1570360) variant is associated with recurrent early pregnancy losses in contrast to normal control groups. The earliest study demonstrating a significant difference goes back to 2005 in Greece [25]. The study involved 52 women with 3 or more recurrent pregnancy losses and 82 controls with live births and no history of pregnancy loss. The analysis of allele frequency for the polymorphic variant c.-1154G/A (rs1570360) yielded a p-value of 0.016, indicating an increased allelic frequency of the mutant allele A in patients with early pregnancy losses. The c.*237C>T variant (rs3025039) increases VEGFA expression as well and acts in a similar direction [26, 27]. Similarly, the effect of the variants in the ANXA5 gene have been shown in some studies to contribute negatively to the rs1570360 and rs3025039 variants in susceptibility to recurrent miscarriages in different geographic groups [27, 28, 29]. Yet others have not confirmed this association [30, 31], therefore the need for further research to see if these relations exist. Hence, these allelic variants are unquestionably noteworthy in investigating the predisposition to RPL development.
In establishing an effective and cost-efficient method for variant detection based on multiplex single-base extension, we aimed to examine the association of the c.-210A>C, c.-184T>C, c.-229G>A, c.-135G>A variants in the ANXA5 gene and c.-1154G>A, c.*237C>T variants in the VEGFA gene in a cohort of women with early pregnancy losses, compared to a control group of women without pregnancy loss and at least one live birth. Moreover, the determination of the status of these variants would provide a direction for future therapies in women with unexplained pregnancy losses, since the large amount of data suggest that the M2/ANXA5 haplotype may contribute to the occurrence of this condition.
MATERIALS AND METHODS
Materials
For this study, a total of 380 DNA samples were observed using a multiplex single-base extension reaction assay. The samples were taken from 190 women experiencing EPLs, 104 from Macedonian and 86 from Albanian ethnic backgrounds and the same number of samples were obtained from women without a history of pregnancy loss as well as at least one healthy live birth as control group.
Following a methodology outlined by Noveski et al., the examined women with EPLs were additionally selected because no fetal chromosomal abnormalities were detected in their POCs [32]. Of the total number of women with pregnancy losses, 81 had single pregnancy loss (sporadic), and 109 had two or more pregnancy losses (recurrent). Also, from the total number of women with early pregnancy losses, 151 did not have a previous live birth, and 39 had.
All participants gave informed consent for participation in the study. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the Macedonian Academy of Sciences and Arts (09-1047/6 from 04.05.2016). This work was financially supported by the project “Molecular basis of spontaneous abortion” (project number: 08-707) funded by the Macedonian Academy of Sciences and Arts.
Methods
Blood samples were collected from each participant and utilized for DNA isolation from leukocytes using the conventional method involving phenol/chloroform extraction and ethanol precipitation, as described by Efremov et al. (1999). All individuals were analyzed for the presence of 4 different variants in the ANXA5 gene: rs28717001 (c.-210A>C), rs28651243 (c.-184T>C), rs112782763 (c.-229G>A), rs113588187 (c.-135G>A) and 2 variants in the VEGFA gene: c.-1154G>A (rs1570360) and c.*237C>T (rs3025039). Taking inspiration from the methodology previously employed in our laboratory [33, 34, 35], we designed a multiplex single-base extension method for simultaneous detection of the 6 variants utilizing the Multiplex SNaPshot kit (Multiplex SNaPshot; Applied Biosystems, Warrington, WA) for the reaction, followed by capillary electrophoresis on an ABI Prism 3130 Genetic Analyzer (Applied Biosystems, Foster City, CA).
The PCR primers were designed to function in a multiplex mix and to produce PCR fragments of 427–550 bp. Briefly, the PCR amplification reaction contained 2 µL of 100 ng genomic DNA, 1.5 µL of a multiplex mix where each specific forward and reverse oligonucleotide primers are with a 10 pmol concentration (Table 1), 2 µL 2.5mM of each deoxyribonucleotide triphosphate (dNTP), 1.5 µL 25mM magnesium chloride, 5 µL GC rich enhancer and 0.2 µL of 1U Taq polymerase (Hot Fire polymerase; Solis Biodyne, Tartu, Estonia) in a total volume of 25 µL.
Table 1.
Nucleotide sequences of PCR primers
| Primer ID | Sequence (5′-3′) | Length (bp) |
|---|---|---|
| ANXA5-F | CAGCTACCGGGACAGCTC | 18 |
| ANXA5-R | CTCCAAAACCCCGAGCCC | 18 |
| VEGFA-F_IGU | TTCCTAGCAAAGAGGGAACG | 20 |
| VEGFA-R_IGU | GCTGACCGGTCCACCTAAC | 19 |
| VEGFA-F1_3’utr | ACACCATCACCATCGACAGA | 20 |
| VEGFA-R1_3’utr | GTCAGGATCTGAGTGGGAACA | 21 |
| rs112782763_ SShot | CCCCCCGCGGCCGGCCTGCGGTTG | 24 |
| rs28717001_ SShot | CCCCCCCTGCCCGGCTTGGCCCG | 23 |
| rs28651243_ SShot | CCCCCCCCCCCCCCCCGGAAACGCCAGCGGCCCC | 34 |
| rs113588187_ SShot | CCCCCCCGCCGAGATGCAGACGCTGAAGGATC | 32 |
| rs1570360_Sshot | CCCCCCCCCCCCCCCCGCCCGAGCCGCGTGTGGA | 34 |
| rs3025039_Sshot | CCCCCCCCCCCCCCTGGCGAATCCAATTCCAAGAGGGACC | 40 |
The PCR conditions were as follows: 10 min. initial denaturation at 95°C, followed by 33 cycles of 1 min at 95°C,1 min at 61°C for the primer annealing and 1 min at 72°C. The final elongation was set at 72°C for 10 min. Afterwards, purification was carried out using 1µL of ExoSAP-IT® (USB, Cleveland, OH) per 2.5 µL of PCR product. The process included an incubation step at 37°C for 20 minutes and subsequent inactivation of the enzyme at 86°C for 20 minutes.
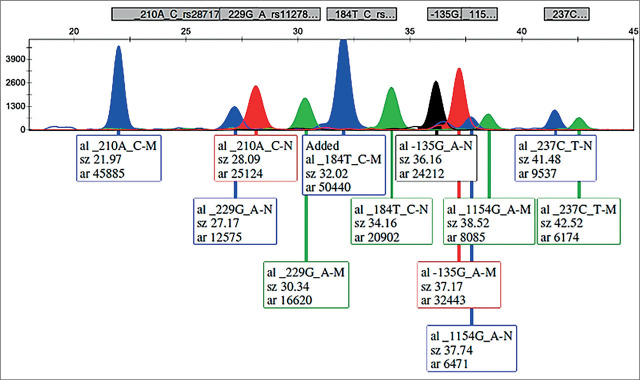
The refined PCR products served as the basis for identifying the six mutations. In the subsequent single-base extension reaction, the detection primers (SNaPshot primers) were aligned adjacent to the single-nucleotide polymorphism position. Various lengths of poly (dC) tails were appended to the single-base extension primers (refer to Table 2). The SNaPshot reaction included a 1.5µL primer mix comprising all 6 single-base extension primers at 3.5µL of purified PCR products, and 1.5µL of the SNaPshot Multiplex kit (Applied Biosystems), adding up to a final volume of 6.5 µL. The cycling profile comprised of 25 cycles of 95°C for 10 s, 55°C for 10 s, and 60°C for 30 s. After the reaction, the 5′-phosphophoryl groups of unincorporated dideoxynucleotide triphosphates were eliminated by adding 1.0U of shrimp alkaline phosphatase (SAP; USB), followed by an incubation step at 37°C for 40 min and at 86°C for 20 min to deactivate the enzyme. Capillary electrophoresis was conducted using an ABI PRISM 3130 Genetic Analyzer, and the results were analyzed using Gene Mapper, Version 4.0 (Applied Biosystems, Foster City, CA). Specifics of the multiplex PCR and SNaPshot reaction products can be found in Table 2. Due to the impact of the dye, size, and nucleotide composition on the mobility shift of DNA fragments, the reported sizes may deviate by a few bases from the actual sizes, especially with shorter fragments where the relative contribution of the dye is more pronounced. Figure 1 displays a representative electrophoretogram from a patient heterozygous for all six variants.
Table 2.
Single base extension primers data
| Variant | Nucleotide change | PCR fragment size (bp) | SNaPshot primer orientation | SNaPshot result (N/M) | SNaPshot fragment size-N (bp) | SNaPshot fragment size-M (bp) |
|---|---|---|---|---|---|---|
| rs112782763 (c.-229G>A) | G/A | 551 | Forward | G/A | 27.1 | 30.3 |
| rs28717001 (c.-210A>C) | A/C | Reverse | T/G | 21.9 | 28.1 | |
| rs28651243 (c.-184T>C) | T/C | Reverse | A/G | 34.1 | 32 | |
| rs113588187 (c.-135G>A) | G/A | Reverse | C/T | 36.1 | 37.1 | |
| rs1570360 (c.1154G>A) | G/A | 450 | Forward | G/A | 37.5 | 38.5 |
| rs3025039 (c.*237C>T) | C/T | 427 | Reverse | G/A | 41.5 | 42.5 |
Figure 1.
Electrophoretogram of an individual heterozygous for all ANXA5 and VEGFA examined variants.
The Chi-square test as well as Fisher’s exact test, were employed to assess the significance of the results in both the examined and control groups was determined based on the p-value. A p-value <0.05 was considered statistically significant.
RESULTS
ANXA5 variants
Haplotype-based classification
Given that the four alterations collectively constitute a haplotype, it is possible to categorize them based on the haplotype’s representation, the extent to which the M1 haplotype (comprising two changes) and the M2 haplotype (encompassing all four changes) are present. Moreover, various subcategories can be delineated to observe different combinations (haplotypes), including N/N (normal genotype), N/M1 (heterozygous for haplotype M1), M1/M1 (homozygous for haplotype M1), N/M2 (heterozygous for haplotype M2), M2/M2 (homozygous for haplotype M2), and M1/M2 (double heterozygous). All our examined samples belonged to one of the above-mentioned categories, i.e., a non-haplotype affiliation was not present in either of our cohorts.
We observed that the suggested pathological haplotypes (M1, M2 and their combinations) were more abundant among our group of women with EPLs, evident by the fact that the normal genotype was found to be significantly more frequent in the control group, with a p-value=0.0009. The M2 heterozygous haplotype was significantly more prevalent among the women with EPLs group, reaching a p-value of 0.0006. The heterozygous M1 and homozygous M2 haplotypes were both more frequent among the group of women with EPLs, however these differences did not reach statistical significance. Interestingly, no homozygous M1 and M1/M2 compound heterozygous haplotypes were detected among the patients group, while the control group included 1 and 5 representatives for each haplotype combination. These findings are presented in Table 3.
Table 3.
The general results of the variants in the ANXA5 gene in the group of women with EPLs and Control group
| Haplotype | Women with EPLs (n=190) | % | p-value | Controls (n=190) | % |
|---|---|---|---|---|---|
| N/N | 135 | 71.05 | 0.0009 | 161 | 84.73 |
| N/M1 | 15 | 7.89 | 0.09 | 8 | 4.21 |
| M1/M1 | 0 | 0.00 | / | 1 | 0.52 |
| N/M2 | 36 | 18.95 | 0.0006 | 14 | 7.37 |
| M2/M2 | 4 | 2.11 | 0.18 | 1 | 0.52 |
| M1/M2 | 0 | 0.00 | / | 5 | 7.90 |
| Total | 190 | 100.00 | 190 | 100.00 |
Comparison based on history of pregnancy loss and live births
Similarly, in these categorizations, our findings indicated a significantly higher frequency of the normal genotype observed in the control group, highlighting that the pathological M1, M2 haplotypes and their combinations were more prevalent among the women who had experienced EPL in our study (p-value=0.0001). Remarkably, statistically significant outcomes were noted for the presence of heterozygotes for the M2 haplotype in individuals who had experienced one early pregnancy loss, when compared to the controls, a pattern also observed in those with recurrent EPLs, however with borderline p-value of 0.057. Further, the occurrence of heterozygous M1 and homozygous M2 haplotypes were equally distributed in both categorizations and did not achieve statistical significance in comparison to the corresponding haplotypes observed in the controls (data presented in Supplementary Table 1).
Notably, among the women with EPLs and without a live birth, there was a statistically significant higher prevalence of heterozygotes for haplotype M2 compared to the control group. Also, the homozygotes for the M2 haplotype belonged to this subgroup of women with EPLs and without a live birth. The women with EPLs and a live birth showed higher presence of heterozygotes for the M1 haplotype, with a borderline statistical significance (p-value=0.05), as presented in Supplementary Table 1.
Comparison based on maternal age and on gestational week of the current pregnancy among the women with EPLs
When the maternal age (at the time of admission) was taken in consideration in our cohort, we observed statistically significant higher prevalence of the heterozygous M2 haplotype among the women with EPLs, ≤ 30 years of age (p-value=0.003) and ≥ 36 years of age (p-value=0.01), in comparison to the control group of the same age range. In contrast, the group of examined women between 31 and 35 years of age, presented a statistically significant difference in the heterozygous M1 haplotype amid the two cohorts (p-value=0.03), with this haplotype being more frequent among the women with EPLs. These data are presented in Supplementary Table 2.
A comparison based on gestational week of the current pregnancy was also performed and these findings are presented in Supplementary Table 2 as well. Unfortunately, data on this parameter was missing for more than a third (35.3%) of the women with EPLs. We divided the women in two major groups, the first having pregnancy loss between weeks 6–9, and the second group consisted of women with EPLs in weeks 10 and 11, based on the presumption that women carrying the M2 haplotype are believed to face over twice the risk of fetal loss between 10 and 15 weeks of gestation compared to those who do not carry this haplotype. Again, the heterozygous M2 haplotype was significantly more present in both subgroups of women with EPLs, compared to the controls, reaching a p-value of 0.008 and 0.007 for both subgroups respectively.
VEGFA gene
The genotypic frequency of the two examined variants in the two specified groups are displayed in Table 4. The results indicate the absence of a statistically significant difference of the selected VEGFA variants between women with EPLs and the control group. The VEGFA c.-1154G/A (rs1570360) variant was identified in 76 patients (40%), in heterozygous state and in 22 (11.58%) patients in homozygous state. In the control group, 70 individuals were heterozygous (36.84%), while 21 (11.05%) were homozygous for this variant. The c.*237C/T (rs3025039) presented similar distributions between the women with EPLs and the controls, however, the homozygous mutant genotype was more rarely found in both groups, when compared to the c.-1154G/A (rs1570360) variant. Further, two women with EPL were found to be homozygous for both VEGFA variants (2/190, 1.05%), and 20 women with EPLs were heterozygous for the both VEGFA variants (20/190, 10.53%). Among the control group, those numbers were one (1/190, 0.53%), and 14 (14/190, 7.37%), respectively. Both differences were not statistically significant (p-value=0.8, and p-value=0.2, accordingly).
Table 4.
The genotypic frequency of the examined VEGFA variants in the group of women with EPLs and Control groups.
| c.-1154G>A | Women with EPLs (n=190) | % | p-value | Controls (n=190) | % |
|---|---|---|---|---|---|
| GG | 92 | 48.42 | 0.47 | 99 | 52.11 |
| GA | 76 | 40.00 | 0.53 | 70 | 36.84 |
| AA | 22 | 11.58 | 0.87 | 21 | 11.05 |
| Total | 190 | 100.00 | 190 | 100.00 | |
| c.*237C>T | |||||
| CC | 137 | 72.10 | 0.64 | 141 | 74.21 |
| CT | 50 | 26.32 | 0.64 | 46 | 24.21 |
| TT | 3 | 1.58 | 1 | 3 | 1.58 |
| Total | 190 | 100.00 | 190 | 100.00 |
Comparison based on history of pregnancy loss and previous live birth
The results of the comparison based on history of pregnancy loss and previous live birth between the two groups are presented in Supplementary Table 3. Examining c.-1154G/A (rs1570360), among women with 2 or more early pregnancy losses, the mutant homozygous form A/A is represented by 14.68%, compared to 11.05% in the control group. However, this discrepancy did not reach statistical significance.
Likewise, in the case of the polymorphic variant c.*237C/T (rs3025039), no notable difference was observed in the subgroups of women with EPLs, compared to the controls. The distribution of the mutant form T/T was higher in the group of women with 2 or more EPLs, although the difference compared to the control group was not statistically significant.
Regarding previous live births, the patients were categorized into two groups: one comprised patients with early pregnancy loss and no prior live births, and the other included those with early pregnancy loss and at least one live birth. The group of women with EPLs and a live birth consisted of 39 cases, among which, 53.85% expressed heterozygosity for the c.-1154G/A (rs1570360) variant, compared to the 36.84% among the controls, and this difference was found to be statistically significant (p-value=0.04). Amid the subgroup of women with EPLs and a live birth, the genotypes containing the mutant allele of the c.-1154G/A (rs1570360) variant, were found to be more common, compared to the control group where the normal genotype was most frequent, reaching a borderline p-value of 0.05. The data on c.*237C/T (rs3025039), for this parameter did not show statistical significance. These data are depicted in Supplementary Table 3.
Comparison based on maternal age and gestational week of the current pregnancy among the women with EPLs
When maternal age was taken into account, in order to observe the distribution of the genotypes for the c.-1154G/A (rs1570360) and c.*237C/T (rs3025039) VEGFA variants, between the different age groups of the women with EPLs and controls, no statistically significant differences were obtained in any subcategory. These data are presented in Supplementary Table 4.
An analysis was conducted considering the gestational week of the current pregnancy, and the genotypes of the VEGFA c.-1154G/A (rs1570360) and c.*237C/T (rs3025039) are displayed in Supplementary Table 4 as well. As previously noted, information regarding this parameter was unavailable for over a third (35.3%) of the women who experienced EPLs. The heterozygous c.-1154G/A (rs1570360) was much more common among the subgroup of women with EPLs in week 10–11, reaching p-value 0.03. In the same subgroup, the heterozygous c.*237C/T (rs3025039) was also more frequent, compared to the controls, with a borderline p-value of 0.05.
DISCUSSION
In this study, we have developed a SNaPshot genotyping technique, which identifies four distinct variants in the ANXA5 gene and two variants in the VEGFA gene. The method efficiently and concurrently identifies all six selected variants and also stands out for its simplicity, accuracy, ease of execution, and cost-effectiveness. By employing this method, we have assessed the occurrence rates of the two haplotypes (M1 and M2) within the ANXA5 gene and the two variants (c.-1154G/A (rs1570360) and c.*237C/T (rs3025039)) within the VEGFA gene among selected group of women of Macedonian and Albanian ethnic origin experiencing early pregnancy loss. Our analysis involved comparing these rates to controls matched by ethnicity and age.
Given the inconsistencies in the literature regarding the impact of the specified variants, as outlined in the introduction, the primary objective of our study was to investigate whether the six selected variants are linked to an elevated risk of early pregnancy loss, particularly within our two major ethnical groups (Macedonian and Albanian). It is important to emphasize that in our examined population fetal aneuploidy as a reason for EPLs was excluded.
Considering that the majority of studies have focused on individuals with subsequent pregnancy losses, and given the predominant nulliparous status among most patients, it became crucial to examine the distribution of haplotypes within various subgroups of our patients. Bogdanova et al. revealed four consecutive single nucleotide changes in the ANXA5 promoter, which are transmitted as a haplotype called M2 that reduces promoter activity, thereby leading to reduced production of Annexin A5 mRNA as well as M1 haplotype covering the first two nucleotide substitutions. Certain analyzes showed that haplotypes M1 and M2 reduce ANXA5 promoter activity by 40% and 60%, respectively, thereby significantly affecting ANXA5 expression. Women with the M2 haplotype are thought to have more than a 2-fold higher risk of fetal loss between 10 and 15 weeks of gestation than non-carriers [11]. As highlighted in a recent meta-analysis [12] and a review [13] examining the impact of the ANXA5 haplotypes on recurrent pregnancy loss (RPL), multiple case-control studies have demonstrated a positive association between the M2/ANXA5 haplotype and RPL across a range of ethnic backgrounds [11, 14,15,16,17,18,19,20,21].
In line with numerous studies, supporting the concept that M2 and M1 ANXA5 haplotypes are more common in patients experiencing pregnancy loss [10,11,12,13,14,15,16,17,18,19,20,21], our study presented higher frequency of the M2 haplotype among the women with EPLs compared to controls (p-value=0.0006).
Furthermore, when we did several sub categorizations, we observed evident statistical significance of the M2 haplotype among: women with recurrent as well as sporadic early pregnancy loss (p-value=0.057 and p-value<0.00001 respectively), women with EPLs and no live birth (p-value=0.0003), women ≤30 and ≥36 years of age (p-value=0.003 and p-value<0.001 respectively), and also in both subgroups of pregnancy loss between 6-9 GW and 10-11GW (p-value=0.008 and p-value<0.007 accordingly). The M1 haplotype was found to be more prevalent in the subgroup of women with EPLs and a live birth (p-value=0.05) and in the subgroup of women with ages between 31 and 35 years with a p-value of 0.01.
Furthermore, there are studies indicating that low-molecular-weight heparin might have a positive effect on miscarriage rate and recurrent implantation failure in treated M2/ANXA5 haplotype carriers [36, 37], so, a research in this direction could be a further step.
VEGFA has gathered significant attention due to its pivotal role in angiogenesis, particularly notable for its implications in embryo development. Compelling evidence underscores its critical involvement in fetal and placental angiogenesis, suggesting that vascular formation irregularities or dysfunction contribute to RPL. Additionally, first-trimester trophoblast VEGFA expression was found to be weaker in placental samples from RPL cases compared to gestational age-matched normal placenta [38, 39]. The most significant finding in most studies is that presence of the c.-1154G/A (rs1570360) variant is associated with recurrent early pregnancy losses in contrast to normal control groups [25,26,27,28,29], while others have not confirmed the association [30, 31], so the need for further research of these relations exists. According to a meta study by Xu et al., [27] the c.-1154G/A (rs1570360) and c.*237C/T (rs3025039) variant demonstrated statistical significance concerning RPL risk across different geographical populations, and discrepancies such as in our present study may be explained by the small sample size and substantial errors from estimation.
In our examined cohort, we observed a general higher prevalence of the heterozygous and mutant homozygous genotypes for the both VEGFA variants, compared to the controls, however without statistical significance. Nevertheless, we noticed borderline statistically significant difference (p-value=0.05) in heterozygotes for the c.-1154G/A (rs1570360) variant between the women with EPLs and a live birth, compared to the controls. Also, when we analyzed the results from the division based on gestational week of the last pregnancy, we noticed statistically higher frequency of the heterozygous genotypes for both VEGFA variants with a p-value<0.00001 for c.-1154G/A (rs1570360) and p-value=0.05 for c.*237C/T (rs3025039).
CONCLUSION
Our current study introduces a cost-effective genotyping method for selected ANXA5 and VEGFA variants. Additionally, our study is in line with numerous studies, supporting the concept that M2 and M1 ANXA5 haplotypes are more common in patients experiencing pregnancy loss. Moreover, since there are data on some experimental therapies for the individuals carrying primarily the M2/ANXA5 haplotype, a research in that direction and clinical studies for therapy justification could be a further step.
Supplementary Material
Supplementary Material Details
ACKNOWLEDGMENT
The study was supported by grant No. 08-707/2023 by the Macedonian Academy of Sciences and Arts to DPK.
Footnotes
CONFLICT OF INTEREST
Not declared.
REFERRENCES
- 1.Macklon NS, Geraedts JP, Fauser BC. Conception to ongoing pregnancy: the ‘black box’ of early pregnancy loss. Human reproduction update. 2002 NaN 1;8(4):333–43. doi: 10.1093/humupd/8.4.333. [DOI] [PubMed] [Google Scholar]
- 2.Eshre Guideline Group on RPL. Bender Atik R, Christiansen OB, Elson J, Kolte AM, Lewis S, Middeldorp S, Nelen W, Peramo B, Quenby S, Vermeulen N. ESHRE guideline: recurrent pregnancy loss. Human reproduction open. 2018;2018(2):hoy004. doi: 10.1093/hropen/hoy004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Practice Committee of the American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertility and sterility. 2020 NaN 1;113(3):533–5. doi: 10.1016/j.fertnstert.2019.11.025. [DOI] [PubMed] [Google Scholar]
- 4.Regan L, Rai R, Saravelos S, Li TC. Royal College of Obstetricians and Gynaecologists. Recurrent Miscarriage Green-top Guideline No. 17. BJOG: An International Journal of Obstetrics &Gynaecology. 2023 NaN;130(12):e9–39. doi: 10.1111/1471-0528.17515. [DOI] [PubMed] [Google Scholar]
- 5.Bozhinovski G, Terzikj M, Kubelka-Sabit K, Jasar D, Lazarevski S, Livrinova V, Plaseska-Karanfilska D. Chromosomal Abnormalities in Early Pregnancy Losses: A Study of 900 Samples. Balkan J Med Genet. 2024 NaN 12;26(2):11–20. doi: 10.2478/bjmg-2023-0014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Turesheva A, Aimagambetova G, Ukybassova T, Marat A, Kanabekova P, Kaldygulova L, Amanzholkyzy A, Ryzhkova S, Nogay A, Khamidullina Z, Ilmaliyeva A. Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box. Journal of Clinical Medicine. 2023 NaN 15;12(12):4074. doi: 10.3390/jcm12124074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bozhinovski G, Terzikj M, Kubelka-Sabit K, Plaseska-Karanfilska D. High Incidence of CPLANE1-Related Joubert Syndrome in the Products of Conceptions from Early Pregnancy Losses. Balkan Med J. 2024 NaN 1;41(2):97–104. doi: 10.4274/balkanmedj.galenos.2024.2023-10-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Deng T, Liao X, Zhu S. Recent advances in treatment of recurrent spontaneous abortion. Obstetrical & gynecological survey. 2022 NaN;77(6):355. doi: 10.1097/OGX.0000000000001033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gerke V, Moss SE. Annexins: from structure to function. Physiological reviews. 2002 NaN 1;82(2):331–71. doi: 10.1152/physrev.00030.2001. [DOI] [PubMed] [Google Scholar]
- 10.Cai Z, Zheng X, Chen Y, Chen F, Chen L, Deng X. Genetic analysis of ANXA5 haplotype and its effect on recurrent pregnancy loss. Molecular medicine reports. 2022 NaN 1;25(2):1–7. doi: 10.3892/mmr.2021.12559. [DOI] [PubMed] [Google Scholar]
- 11.Bogdanova N, Horst J, Chlystun M, Croucher PJ, Nebel A, Bohring A, Todorova A, Schreiber S, Gerke V, Krawczak M, Markoff A. A common haplotype of the annexin A5 (ANXA5) gene promoter is associated with recurrent pregnancy loss. Human molecular genetics. 2007 NaN 1;16(5):573–8.10. doi: 10.1093/hmg/ddm017. [DOI] [PubMed] [Google Scholar]
- 12.Ang KC, Bogdanova N, Markoff A, Ch’ng ES, Tang TH. Association between M2/ANXA5 haplotype and repeated pregnancy loss: a meta-analysis. Fertility and sterility. 2019 NaN 1;111(5):971–81. doi: 10.1016/j.fertnstert.2019.01.015. [DOI] [PubMed] [Google Scholar]
- 13.Peng L, Yang W, Deng X, Bao S. Research progress on ANXA5 in recurrent pregnancy loss. Journal of Reproductive Immunology. 2022 NaN 28;:103679. doi: 10.1016/j.jri.2022.103679. [DOI] [PubMed] [Google Scholar]
- 14.Tüttelmann F, Ivanov P, Dietzel C, Sofroniou A, Tsvyatkovska TM, Komsa-Penkova RS, Markoff A, Wieacker P, Bogdanova N. Further insights into the role of the annexin A5 M2 haplotype as recurrent pregnancy loss factor, assessing timing of miscarriage and partner risk. Fertility and sterility. 2013 NaN 1;100(5):1321–5. doi: 10.1016/j.fertnstert.2013.06.046. [DOI] [PubMed] [Google Scholar]
- 15.Rogenhofer N, Engels L, Bogdanova N, Tüttelmann F, Markoff A, Thaler C. Paternal and maternal carriage of the annexin A5 M2 haplotype are equal risk factors for recurrent pregnancy loss: a pilot study. Fertility and sterility. 2012 NaN 1;98(2):383–8. doi: 10.1016/j.fertnstert.2012.04.026. [DOI] [PubMed] [Google Scholar]
- 16.Miyamura H, Nishizawa H, Ota S, Suzuki M, Inagaki A, Egusa H, Nishiyama S, Kato T, Pryor-Koishi K, Nakanishi I, Fujita T. Polymorphisms in the annexin A5 gene promoter in Japanese women with recurrent pregnancy loss. Molecular human reproduction. 2011 NaN 1;17(7):447–52. doi: 10.1093/molehr/gar008. [DOI] [PubMed] [Google Scholar]
- 17.Demetriou C, Abu-Amero S, White S, Peskett E, Markoff A, Stanier P, Moore GE, Regan L. Investigation of the Annexin A5 M2 haplotype in 500 white European couples who have experienced recurrent spontaneous abortion. Reproductive biomedicine online. 2015 NaN 1;31(5):681–8. doi: 10.1016/j.rbmo.2015.07.004. [DOI] [PubMed] [Google Scholar]
- 18.Tiscia G, Colaizzo D, Chinni E, Pisanelli D, Sciannamè N, Favuzzi G, Margaglione M, Grandone E. Haplotype M2 in the annexin A5 (ANXA5) gene and the occurrence of obstetric complications. Thrombosis and haemostasis. 2009;102(08):309–13. doi: 10.1160/TH09-02-0123. [DOI] [PubMed] [Google Scholar]
- 19.Lan Y, Wang J, Zhang Q, Yang X, Li L, Yin J, Li H, Song X, Chen Z, Liu Y. Genetic variations and haplotypes in the annexin A5 gene are associated with the risk of recurrent pregnancy loss. Journal of cellular physiology. 2019 NaN;234(10):18308–16. doi: 10.1002/jcp.28463. [DOI] [PubMed] [Google Scholar]
- 20.Ang KC, Kathirgamanathan S, Ch’ng ES, Lee YY, Roslani AL, Naidu B, Kumar K, Abdullah R, Kadir SN, Yusoff NM, Abdullah WZ. Genetic analysis of the M2/ANXA5 haplotype as recurrent pregnancy loss predisposition in the Malay population. Journal of assisted reproduction and genetics. 2017 NaN;34:517–24. doi: 10.1007/s10815-017-0871-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dryllis G, Giannopoulos A, Zoi C, Pouliakis A, Logothetis E, Voulgarelis M, Zoi K, Kouskouni E, Dinou A, Stavropoulos-Giokas C, Kreatsas G. Correlation of single nucleotide polymorphisms in the promoter region of the ANXA5 (annexin A5) gene with recurrent miscarriages in women of Greek origin. The Journal of Maternal-Fetal & Neonatal Medicine. 2020 NaN 2;33(9):1538–43. doi: 10.1080/14767058.2018.1521799. [DOI] [PubMed] [Google Scholar]
- 22.Cao Y, Zhang Z, Xu J, Yuan W, Wang J, Huang X, Shen Y, Du J. The association of idiopathic recurrent pregnancy loss with polymorphisms in hemostasis-related genes. Gene. 2013 NaN 10;530(2):248–52. doi: 10.1016/j.gene.2013.07.080. [DOI] [PubMed] [Google Scholar]
- 23.Nagirnaja L, Nõmmemees D, Rull K, Christiansen OB, Nielsen HS, Laan M. Annexin A5 promoter haplotype M2 is not a risk factor for recurrent pregnancy loss in Northern Europe. PLoS One. 2015 NaN 2;10(7):e0131606. doi: 10.1371/journal.pone.0131606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sinem AY, Fatma S, Servet OH, Ahmet U, Emine C, Ozturk O. Fetal Vegf genotype is more important for abortion risk than mother genotype. CanakkaleOnSekiz Mart University; Turkey: 2014. [Google Scholar]
- 25.Papazoglou D, Galazios G, Papatheodorou K, Liberis V, Papanas N, Maltezos E. et al. Vascular endothelial growth factor gene polymorphisms and idiopathic recurrent pregnancy loss. FertilSteril. 2005;83:959–6. doi: 10.1016/j.fertnstert.2004.12.017. [DOI] [PubMed] [Google Scholar]
- 26.Sun Y, Chen M, Mao B, Cheng X, Zhang X, Xu C. Association between vascular endothelial growth factor polymorphism and recurrent pregnancy loss: A systematic review and meta-analysis. European Journal of Obstetrics & Gynecology and Reproductive Biology. 2017 NaN 1;211:169–76. doi: 10.1016/j.ejogrb.2017.03.003. [DOI] [PubMed] [Google Scholar]
- 27.Xu X, Du C, Li H, Du J, Yan X, Peng L, Li G, Chen ZJ. Association of VEGF genetic polymorphisms with recurrent spontaneous abortion risk: a systematic review and meta-analysis. PLoS One. 2015 NaN 20;10(4):e0123696. doi: 10.1371/journal.pone.0123696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trifonova EA, Swarovskaya MG, Ganzha OA, Voronkova OV, Gabidulina TV, Stepanov VA. The interaction effect of angiogenesis and endothelial dysfunction-related gene variants increases the susceptibility of recurrent pregnancy loss. Journal of Assisted Reproduction and Genetics. 2019 NaN 15;36:717–26. doi: 10.1007/s10815-019-01403-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Su MT, Lin SH, Chen YC. Genetic association studies of angiogenesis-and vasoconstriction-related genes in women with recurrent pregnancy loss: a systematic review and meta-analysis. Human Reproduction Update. 2011 NaN 1;17(6):803–12. doi: 10.1093/humupd/dmr027. [DOI] [PubMed] [Google Scholar]
- 30.Eller AG, Branch DW, Nelson L, Porter TF, Silver RM. Vascular endothelial growth factor-A gene polymorphisms in women with recurrent pregnancy loss. Journal of reproductive immunology. 2011 NaN 1;88(1):48–52. doi: 10.1016/j.jri.2010.06.159. [DOI] [PubMed] [Google Scholar]
- 31.Fortis MF, Fraga LR, Boquett JA, Kowalski TW, Dutra CG, Gonçalves RO, Vianna FS, Schüler-Faccini L, Sanseverino MT. Angiogenesis and oxidative stress-related gene variants in recurrent pregnancy loss. Reproduction, Fertility and Development. 2018 NaN 13;30(3):498–506. doi: 10.1071/RD17117. [DOI] [PubMed] [Google Scholar]
- 32.Noveski P, Terzic M, Vujovic M, Kuzmanovska M, Sukarova Stefanovska E, Plaseska-Karanfilska D. Multilevel regression modeling for aneuploidy classification and physical separation of maternal cell contamination facilitates the QF-PCR based analysis of common fetal aneuploidies. Plos one. 2019 NaN 20;14(8):e0221227. doi: 10.1371/journal.pone.0221227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Madjunkova S, Volk M, Peterlin B, Plaseska-Karanfilska D. Detection of thrombophilic mutations related to spontaneous abortions by a multiplex SNaPshot method. Genetic testing and molecular biomarkers. 2012 NaN 1;16(4):259–64. doi: 10.1089/gtmb.2011.0173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Atanasovska B, Bozhinovski G, Plaseska-Karanfilska D, Chakalova L. Efficient detection of Mediterranean β-thalassemia mutations by multiplex single-nucleotide primer extension. PLoS One. 2012 NaN 26;7(10):e48167. doi: 10.1371/journal.pone.0048167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Plaseski T, Noveski P, Popeska Z, Efremov GD, Plaseska-Karanfilska D. Association study of single-nucleotide polymorphisms in FASLG, JMJDIA, LOC203413, TEX15, BRDT, OR2W3, INSR, and TAS2R38 genes with male infertility. Journal of andrology. 2012 NaN 8;33(4):675–83. doi: 10.2164/jandrol.111.013995. [DOI] [PubMed] [Google Scholar]
- 36.Rogenhofer N, Markoff A, Wagner A, Klein HG, Petroff D, Schleussner E. EThIG II Group. Thaler CJ. Lessons from the EThIGII trial: proper putative benefit assessment of low-molecular-weight heparin treatment in M2/ANXA5 haplotype carriers. Clinical and Applied Thrombosis/Hemostasis. 2017 NaN;23(1):27–33. doi: 10.1177/1076029616658117. [DOI] [PubMed] [Google Scholar]
- 37.Fishel S, Baker DJ, Greer IA. LMWH in IVF–Biomarkers and benefits. Thrombosis Research. 2017 NaN 1;151:S65–9. doi: 10.1016/S0049-3848(17)30071-3. [DOI] [PubMed] [Google Scholar]
- 38.Vuorela P, Carpen O, Tulppala M, Halmesmäki E. VEGF, its receptors and the tie receptors in recurrent miscarriage. Molecular human reproduction. 2000 NaN 1;6(3):276–82. doi: 10.1093/molehr/6.3.276. [DOI] [PubMed] [Google Scholar]
- 39.Plaisier M, Dennert I, Rost E, Koolwijk P, Van Hinsbergh VW, Helmerhorst FM. Decidual vascularization and the expression of angiogenic growth factors and proteases in first trimester spontaneous abortions. Human reproduction. 2009 NaN 1;24(1):185–97. doi: 10.1093/humrep/den296. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material Details