Abstract
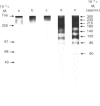
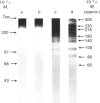
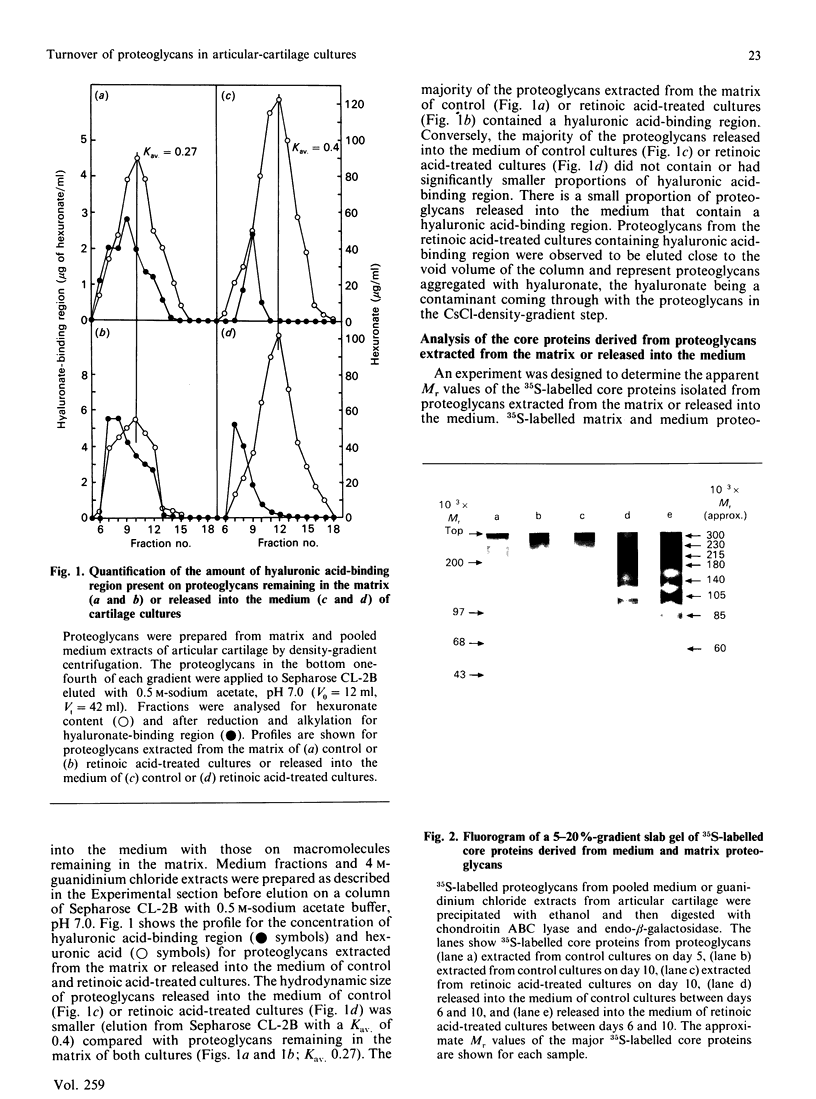
By using an e.l.i.s.a. method it was demonstrated that the majority of proteoglycans released into the medium of both control and retinoic acid-treated explant cultures of bovine articular cartilage did not contain a hyaluronate-binding region. This supports our previous findings [Campbell & Handley (1987) Arch. Biochem. Biophys. 258, 143-155] that proteoglycans released into the medium of both cultures were of smaller hydrodynamic size, more polydisperse and unable to form aggregates with hyaluronate. Analysis of 35S-labelled core proteins associated with proteoglycans released into the medium of both cultures by using SDS/polyacrylamide-gel electrophoresis and fluorography indicated the presence of a series of core-protein bands (Mr approx. 300,000, 230,000, 215,000, 200,000, 180,000, 140,000, 135,000, 105,000, 85,000 and 60,000) compared with three core proteins derived from the proteoglycans remaining in the matrix (Mr 300,000, 230,000 and 215,000). Further analysis of the core proteins released into the medium indicated that the larger core proteins associated with medium proteoglycans contain both chondroitin sulphate and keratan sulphate glycosaminoglycans whereas the smaller core proteins contain only chondroitin sulphate chains. These experiments provide definitive evidence that the loss of proteoglycans from the matrix involves proteolytic cleavage at various sites along the proteoglycan core protein.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Campbell M. A., Handley C. J., Hascall V. C., Campbell R. A., Lowther D. A. Turnover of proteoglycans in cultures of bovine articular cartilage. Arch Biochem Biophys. 1984 Oct;234(1):275–289. doi: 10.1016/0003-9861(84)90350-3. [DOI] [PubMed] [Google Scholar]
- Campbell M. A., Handley C. J. The effect of retinoic acid on proteoglycan biosynthesis in bovine articular cartilage cultures. Arch Biochem Biophys. 1987 Mar;253(2):462–474. doi: 10.1016/0003-9861(87)90200-1. [DOI] [PubMed] [Google Scholar]
- Campbell M. A., Handley C. J. The effect of retinoic acid on proteoglycan turnover in bovine articular cartilage cultures. Arch Biochem Biophys. 1987 Oct;258(1):143–155. doi: 10.1016/0003-9861(87)90331-6. [DOI] [PubMed] [Google Scholar]
- Doege K., Sasaki M., Horigan E., Hassell J. R., Yamada Y. Complete primary structure of the rat cartilage proteoglycan core protein deduced from cDNA clones. J Biol Chem. 1987 Dec 25;262(36):17757–17767. [PubMed] [Google Scholar]
- Hascall V. C., Handley C. J., McQuillan D. J., Hascall G. K., Robinson H. C., Lowther D. A. The effect of serum on biosynthesis of proteoglycans by bovine articular cartilage in culture. Arch Biochem Biophys. 1983 Jul 1;224(1):206–223. doi: 10.1016/0003-9861(83)90205-9. [DOI] [PubMed] [Google Scholar]
- Hascall V. C., Kimura J. H. Proteoglycans: isolation and characterization. Methods Enzymol. 1982;82(Pt A):769–800. doi: 10.1016/0076-6879(82)82102-2. [DOI] [PubMed] [Google Scholar]
- Hascall V. C., Sajdera S. W. Proteinpolysaccharide complex from bovine nasal cartilage. The function of glycoprotein in the formation of aggregates. J Biol Chem. 1969 May 10;244(9):2384–2396. [PubMed] [Google Scholar]
- Heinegård D., Hascall V. C. Aggregation of cartilage proteoglycans. 3. Characteristics of the proteins isolated from trypsin digests of aggregates. J Biol Chem. 1974 Jul 10;249(13):4250–4256. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K., Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. doi: 10.1016/0022-2836(73)90198-8. [DOI] [PubMed] [Google Scholar]
- Oike Y., Kimata K., Shinomura T., Suzuki S. Proteinase activity in chondroitin lyase (chondroitinase) and endo-beta-D-galactosidase (keratanase) preparations and a method to abolish their proteolytic effect on proteoglycan. Biochem J. 1980 Oct 1;191(1):203–207. doi: 10.1042/bj1910203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldberg A., Antonsson P., Heinegård D. The partial amino acid sequence of bovine cartilage proteoglycan, deduced from a cDNA clone, contains numerous Ser-Gly sequences arranged in homologous repeats. Biochem J. 1987 Apr 1;243(1):255–259. doi: 10.1042/bj2430255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratcliffe A., Tyler J. A., Hardingham T. E. Articular cartilage cultured with interleukin 1. Increased release of link protein, hyaluronate-binding region and other proteoglycan fragments. Biochem J. 1986 Sep 1;238(2):571–580. doi: 10.1042/bj2380571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens J. W., Oike Y., Handley C., Hascall V. C., Hampton A., Caterson B. Characteristics of the core protein of the aggregating proteoglycan from the Swarm rat chondrosarcoma. J Cell Biochem. 1984;26(4):247–259. doi: 10.1002/jcb.240260405. [DOI] [PubMed] [Google Scholar]