Background
X-linked agammaglobulinemia (XLA), also referred to as Bruton’s tyrosine kinase deficiency, is a rare genetic disorder that affects the immune system. We conducted genetic analysis on patients suffering from immunodeficiency by utilizing Next-Generation Sequencing techniques, as well as their closest relatives, to facilitate accurate diagnosis, offer genetic counseling services, and enhance our comprehension of XLA.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12879-024-09743-w.
Keywords: Primary immunodeficiency, X-linked agammaglobulinemia, Mycoplasma pneumonia
Case presentation
In this study, we present the case of a 12-year-old boy who experienced a 6-day-long episode of fever and cough, initially diagnosed with mycoplasma pneumonia and immunodeficiency. The patient had previously received anti-infection treatment including the use of antibiotics, corticosteroids, and antiviral agents before admission; however, the treatment’s efficacy was unsatisfactory. It is worth pointing out that, we present the first case of XLA in a Chinese individual carrying a BTK (c.753G > A) mutation.
Conclusions.
Based on previous reports in the literature and our case, we emphasize the importance of increased importance of early diagnosis of XLA so that patients can begin treatment quickly.
Background
X-linked agammaglobulinemia (XLA) is a primary immunodeficiency disorder characterized by mutations in the gene encoding Bruton’s tyrosine kinase (Btk), leading to a significant reduction in B-cell counts, a condition known as B-cell lymphopenia. Among the most prevalent congenital immune deficiency disorders, XLA primarily affects male children. Individuals with XLA, particularly during childhood, experience recurrent infections, commonly triggered by bacteria such as Streptococcus pneumoniae, Staphylococcus aureus, and Haemophilus influenzae. Common complications of XLA include otitis media, sinusitis, and pneumonia, which require immediate diagnostic assessment and therapeutic intervention to prevent further complications [1]. Patients with atypical XLA frequently suffer from misdiagnosis due to the difficulty in distinguishing their symptoms from other immunodeficiency diseases, complicating the treatment courses [2, 3]. Given the absence of targeted XLA treatments, patients typically receive immunoglobulin replacements and use antibiotics prophylactically to prevent recurrent infections. Community-acquired pneumonia caused by Mycoplasma pneumoniae is a significant concern for individuals with immunodeficiencies, potentially extending the clinical course [4]. In our investigation, we leveraged Next-Generation Sequencing (NGS) to perform comprehensive exome analysis, targeting the entire coding regions of 23,000 genes on individuals with immunodeficiencies and their closest family members to facilitate diagnostic precision and offer genetic consultations.
Case presentation
A 12-year-old boy sought medical attention at our emergency department due to a fever and cough for 6 days. The patient, known for poor physical fitness and susceptibility to respiratory infections, had a history of encephalitis two years ago, followed by purulent meningitis two weeks later, and pneumonia six months prior.
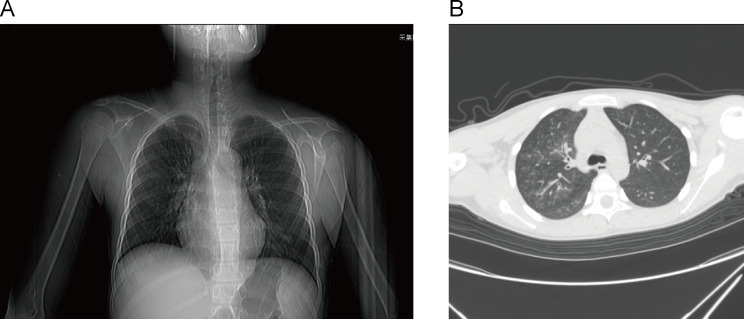
Before admission, a treatment protocol was employed that encompassed the use of antibiotics, corticosteroids, and antiviral agents, however, the treatment effect was not satisfactory. A sputum culture revealed positive cocci with short chains, while throat swabs detected Mycoplasma pneumoniae and influenza B virus. No causative organisms in blood and urine samples were identified. Blood tests showed the changes in lymphocyte percentage (Table 1). The chest X-ray revealed thickening and disorder of the lung bronchus. Chest CT imaging showed patchy and small patchy increased density in the right lobes of the lung and the lower lobe of the left lung (Fig. 1). Lung auscultation of the patient revealed the presence of fever, cough, and breathy sound in both lungs. The original diagnosis of pneumonia was confirmed. Following admission, infection and inflammation were treated with azithromycin, levofloxacin, cefoperazone sodium sulbactam, ceftazidime, and mabaloxavir, respectively. Three days following admission, the patient was transferred to the infectious disease department where he received a diagnosis of Mycoplasma pneumonia and severe influenza caused by the Influenza B virus, in addition to recurrent sinusitis. On the tenth day after admission, due to reduced immunoglobulin levels and persistent fever, immune deficiency was suspected (Table 1). Human immunoglobulin was administered as supplementing the insufficient immunity while continuing anti-infective treatment. The patient was discharged on the thirteenth day with normalized temperature and significant improvement. The discharge diagnosis included Mycoplasma pneumonia, severe influenza caused by influenza B virus, otitis media, sinusitis, and immune deficiency.
Table 1.
Patient laboratory results
| Investigations | Results (Reference Range) |
|---|---|
| Lymphocyte percentage (%) | |
| T CD3+ | 88.88 (55–84) |
| T CD3 + CD8+ | 41.42 (11–41) |
| B CD19+ | 0.2 (6–25) |
| Immunoglobulins (g/L) | |
| IgG | 0.16 (7-15.5) |
| IgM | 0.11 (0.49–2.4) |
| IgA | 0.15 (0.58–2.91) |
Fig. 1.
Chest X-ray and computed tomography images of patient
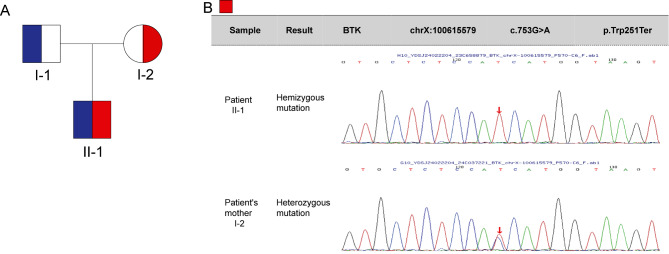
Following discharge, the patient was diagnosed with X-linked agammaglobulinemia through NGS, identifying a BTK gene mutation (c.753G > A) inherited from his mother. The parents were advised to bring the child for regular hospital visits for review and immunoglobulin replacement, while genetic testing was recommended for the child’s siblings. Through NGS testing, hemizygous for BTK (c.753G > A) was found in patient. The variant is non-sense, it leads to protein truncation at 251 (p. Trp251Ter). Pathogenicity analysis was conducted according to the American College of Medical Genetics and Genomics (ACMG) guidelines for variant interpretation (Fig. 2). Furthermore, we investigated the patient’s family history and discovered that neither parent nor any relatives had the condition.
Fig. 2.
The sequencing map of patient. Pedigree diagram and sanger sequencing of the patients
Discussion and conclusions
XLA is a common primary immunodeficiency disease, predominantly impacting male infants and young children [3]. It is characterized by a reduction or complete lack of B cells, leading to diminished immunoglobulin levels and compromised T cell function. As a result, patients are susceptible to recurrent bacterial infections [5]. Otitis media, sinusitis, and pneumonia are common complications of X-linked agammaglobulinemia (XLA), underscoring the significance of timely diagnosis and treatment [6]. Individuals with genetic alterations may exhibit an increased susceptibility to respiratory viral infections [7]. Mycoplasma pneumoniae, a prominent etiological cause of community-acquired pneumonia, particularly affects children with immunodeficiency, often resulting in more severe illness [8]. Taylor-Robinson et al. described a patient with hypogammaglobulinemia, who developed severe pneumonia due to Mycoplasma pneumoniae infection. Despite receiving multiple antibiotic treatments, the clinical course of pneumonia persisted for an extended duration [4]. In this study, the patient displayed markedly reduced levels of immunoglobulins and lymphocyte counts, accompanied by a recurrent respiratory tract infections and general debility. Previous diagnoses included encephalitis, purulent meningitis, pneumonia, and the current recurrence of sinusitis, all of which align with the clinical profile of XLA. Genetic sequencing in this case led to the diagnosis of XLA, revealing a hemizygous mutation in the BTK gene (c.753G > A). The same mutation was detected in heterozygous form in the patient’s mother [9]. Following immunoglobulin treatment, the patient’s condition gradually improved. However, given the chronic nature of XLA, lifelong administration of immunoglobulins and additional interventions to manage infections or other complications are likely necessary. Regular monitoring and provision of supportive care are essential for optimal management of XLA.
Atypical XLA patients are often initially misdiagnosed with other diseases, such as common variable immunodeficiency, chronic respiratory infections, autoimmune diseases, and so on. Clinical differentiation from these common diseases can be challenging, complicating the treatment process [10]. To minimize the risk of misdiagnosis, clinicians should maintain a high suspicion for XLA in patients who experience recurrent infections and have a suggestive family history. Following 8 to 15 years of patient follow-up, during which individuals with XLA received intramuscular immunoglobulin replacement and long-term antibiotics, it was observed that all 10 patients displayed normal chest X-ray findings and none developed bronchiectasis [11]. The study by Foy et al. [12] elucidated that patients with acute mycoplasma pneumoniae infection, who were immunocompromised, demonstrated a rapid clinical response to antibiotic therapy. Notably, individuals with XLA who experienced recurrent infections while receiving low-dose (< 200 mg/kg monthly) intravenous immunoglobulins exhibited clinical improvement when the dosage was increased to > 400 mg/kg every three weeks [13]. Additionally, research has demonstrated that individualizing the dosage of IgG is imperative to optimize clinical outcomes for patients with agammaglobulinemia [14]. Hence, monitoring the biological trough concentration of the drug is pivotal in evaluating both efficacy and safety of the treatment regimen. Documenting longitudinal changes in clinical symptoms and serum IgG levels on a personalized basis is anticipated to augment patient care [15, 16]. As patients with XLA have complete immunoglobulin defects and develop severe and recurrent infections, early diagnosis would be beneficial for the improvement of their quality of life. By identifying the specific gene mutations responsible for XLA, healthcare professionals can provide targeted treatments and interventions to manage the condition effectively, individualizing the dosage of IgG is imperative to optimize clinical outcomes for patients with agammaglobulinemia, which can significantly improve patients’ quality of life [14, 17]. In conclusion, the prognosis for patients with XLA can be favorably impacted through prompt diagnosis and the judicious use of immunoglobulin therapy. Immunological assays play a vital role in confirming immunodeficiency, necessitating collaboration with specialists for optimal management. Collaborating with immunologists and other specialists is essential for addressing the diagnostic challenges associated with atypical XLA.
Conclusion
This research aims to highlight that Mycoplasma pneumonia in individuals with X-linked agammaglobulinemia is a manageable condition. Health care providers are aware of the increased importance of early diagnosis of XLA so that patients can begin treatment quickly.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Not applicable.
Abbreviations
- XLA
X-linked agammaglobulinaemia
- Btk
Bruton’s tyrosine kinase
- NGS
Next-Generation Sequencing
Author contributions
Conceptualization, B.D. and Y.J.; methodology, data curation, writing—original draft preparation, B.D., J.H., Y.S., Z.L., H.C., Z.W., Y.Y., R.Z., C.W., J.Z., M.Q., Y.Z., X.Z., and F.W.; software, supervision, writing—review and editing, Y.J.; resources, project administration, Y.J.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82372229, No. 82002147); supported by China Postdoctoral Science Foundation (No. 2024T170246, No. 2024M750815); supported by Open Research Fund of National Health Commission Key Laboratory of Birth Defects Prevention & Henan Key Laboratory of Population Defects Prevention (No. ZD202301), supported by the Open Grant from the Pingyuan Laboratory (No. 2023PY-OP-0202), and supported by Open Project of Henan Province Engineering Research Center of Diagnosis and Treatment of Pediatric Infection and Critical Care (No. ERC202302).
Data availability
Data is provided within the manuscript or supplementary information files.
Declarations
Consent for publication
Written informed consent for publication of their clinical details and/or clinical images was obtained from the parent of the patient.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bowen Dai and Shujuan Han are co-first authors of the article.
Contributor Information
Fang Wang, Email: 13783637576@139.com.
Yuefei Jin, Email: jyf201907@zzu.edu.cn.
References
- 1.Kainulainen L, Nikoskelainen J, Vuorinen T, Tevola K, Liippo K, Ruuskanen O. Viruses and bacteria in bronchial samples from patients with primary hypogammaglobulinemia. Am J Respir Crit Care Med. 1999;159(4 Pt 1):1199–204. 10.1164/ajrccm.159.4.9807067 [DOI] [PubMed] [Google Scholar]
- 2.Kornfeld SJ, Kratz J, Haire RN, Litman GW, Good R. A., X-linked agammaglobulinemia presenting as transient hypogammaglobulinemia of infancy. J Allergy Clin Immunol. 1995;95(4):915–7. 10.1016/S0091-6749(95)70138-9 [DOI] [PubMed] [Google Scholar]
- 3.Lim LM, Chang JM, Wang IF, Chang WC, Hwang DY, Chen HC. Atypical X-linked agammaglobulinaemia caused by a novel BTK mutation in a selective immunoglobulin M deficiency patient. BMC Pediatr. 2013;13:150. 10.1186/1471-2431-13-150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Taylor-Robinson D, Webster AD, Furr PM, Asherson GL. Prolonged persistence of Mycoplasma pneumoniae in a patient with hypogammaglobulinaemia. J Infect. 1980;2(2):171–5. 10.1016/S0163-4453(80)91284-0 [DOI] [PubMed] [Google Scholar]
- 5.Carrillo-Tapia E, Garcia-Garcia E, Herrera-Gonzalez NE, Yamazaki-Nakashimada MA, Staines-Boone AT, Segura-Mendez NH, Scheffler-Mendoza SC, P OF-R, Gonzalez-Serrano ME, Rodriguez-Alba JC, Santos-Argumedo L, Berron-Ruiz L, Sanchez-Flores A, Lopez-Herrera G. Delayed diagnosis in X-linked agammaglobulinemia and its relationship to the occurrence of mutations in BTK non-kinase domains. Expert Rev Clin Immunol. 2018;14(1):83–93. 10.1080/1744666X.2018.1413349 [DOI] [PubMed] [Google Scholar]
- 6.Wang Y, Kanegane H, Wang X, Han X, Zhang Q, Zhao S, Yu Y, Wang J, Miyawaki T. Mutation of the BTK gene and clinical feature of X-linked agammaglobulinemia in mainland China. J Clin Immunol. 2009;29(3):352–6. 10.1007/s10875-008-9262-8 [DOI] [PubMed] [Google Scholar]
- 7.Kainulainen L, Vuorinen T, Rantakokko-Jalava K, Osterback R, Ruuskanen O. Recurrent and persistent respiratory tract viral infections in patients with primary hypogammaglobulinemia. J Allergy Clin Immunol. 2010;126(1):120–6. 10.1016/j.jaci.2010.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krafft C, Christy C. Mycoplasma Pneumonia in Children and adolescents. Pediatr Rev. 2020;41(1):12–9. 10.1542/pir.2018-0016 [DOI] [PubMed] [Google Scholar]
- 9.Kuehn HS, Niemela JE, Sreedhara K, Stoddard JL, Grossman J, Wysocki CA, de la Morena MT, Garofalo M, Inlora J, Snyder MP, Lewis DB, Stratakis CA, Fleisher TA, Rosenzweig SD. Novel nonsense gain-of-function NFKB2 mutations associated with a combined immunodeficiency phenotype. Blood. 2017;130(13):1553–64. 10.1182/blood-2017-05-782177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nifosi F, Violato E, Pavan C, Sifari L, Novello G, Guarda Nardini L, Manfredini D, Semenzin M, Pavan L, Marini M. Psychopathology and clinical features in an Italian sample of patients with myofascial and temporomandibular joint pain: preliminary data. Int J Psychiatry Med. 2007;37(3):283–300. 10.2190/PM.37.3.f [DOI] [PubMed] [Google Scholar]
- 11.Sweinberg SK, Wodell RA, Grodofsky MP, Greene JM, Conley ME. Retrospective analysis of the incidence of pulmonary disease in hypogammaglobulinemia. J Allergy Clin Immunol. 1991;88(1):96–104. 10.1016/0091-6749(91)90306-9 [DOI] [PubMed] [Google Scholar]
- 12.Foy HM, Ochs H, Davis SD, Kenny GE, Luce RR. Mycoplasma pneumoniae infections in patients with immunodeficiency syndromes: report of four cases. J Infect Dis. 1973;127(4):388–93. 10.1093/infdis/127.4.388 [DOI] [PubMed] [Google Scholar]
- 13.Trakultivakorn M, Ochs HD. X-linked agammaglobulinemia in northern Thailand. Asian Pac J Allergy Immunol. 2006;24(1):57–63. [PubMed] [Google Scholar]
- 14.Bonagura VR, Marchlewski R, Cox A, Rosenthal DW. Biologic IgG level in primary immunodeficiency disease: the IgG level that protects against recurrent infection. J Allergy Clin Immunol. 2008;122(1):210–2. 10.1016/j.jaci.2008.04.044 [DOI] [PubMed] [Google Scholar]
- 15.Angarola E, Peuchot VA, Warley F, Liberatore DI. Secondary hypogammaglobulinemia in adults-A large retrospective cohort study. Med Clin (Barc). 2024;162(4):170–8. 10.1016/j.medcli.2023.10.005 [DOI] [PubMed] [Google Scholar]
- 16.Li Z, Follman K, Freshwater E, Engler F, Yel L. Integrated population pharmacokinetics of immunoglobulin G following intravenous or subcutaneous administration of various immunoglobulin products in patients with primary immunodeficiencies. Int Immunopharmacol. 2022;113:109331. (Pt A). 10.1016/j.intimp.2022.109331 [DOI] [PubMed] [Google Scholar]
- 17.Alizadeh Z, Dashti P, Mazinani M, Nourizadeh M, Shakerian L, Tajik S, Movahedi M, Mamishi S, Pourpak Z, Fazlollahi MR. Clinical and genetic study of X-linked agammaglobulinemia patients (the benefit of early diagnosis). Iran J Allergy Asthma Immunol. 2020;19(3):305–9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is provided within the manuscript or supplementary information files.