Abstract
Background:
In this review, we provide an updated assessment of available evidence on the pharmacokinetics (PK) of CBD and explore the impact of different factors on PK outcomes.
Materials and Methods:
This systematic review and meta-regression analysis was preregistered (PROSPERO: CRD42021269857). We systematically searched Medline, Embase, PsycInfo, and Web of Science Core Collection up to November 19, 2022. Trials of CBD in healthy adults were included if they reported at least one of the PK parameters of interest, including Tmax, Cmax, AUC0–t, AUC0–inf, and T1/2, in serum or plasma. Studies of patient populations or CBD co-administration with other medications were excluded. The National Heart, Lung, and Blood Institute's Quality Assessment Tool for Before-After Studies with no Control Group was used. Random-effects multivariable meta-regression analysis was conducted.
Results:
A total of 112 trial arms from 39 studies were included; 26 trial arms had a “Good” quality, 70 “Fair,” and 16 “Poor.” Eight arms used inhalation CBD, 29 oromucosal, 73 oral, and 2 intravenous. CBD formulations could be categorized to nanotech (n=14), oil-based (n=21), alcohol-based (n=10), water-based (n=12), Sativex (n=17), and Epidiolex® (n=22). For single-dose studies, CBD doses ranged between 2 and 100 mg in inhalation, 5–50 mg in oromucosal, and 0.42–6000 mg in oral administration. Sixty-six trial arms had only male participants or a higher number of male than female participants. The duration of the PK session was between 4 and 164 h. A higher CBD dose was associated with higher Cmax, AUC0–t, and AUC0–inf. Compared with oral administration, oromucosal administration was associated with lower Cmax, AUC0–t, and AUC0–inf. Fed status was associated with higher Cmax and AUC0–t when compared with the fasting status. A higher ratio of female participants was associated with lower Tmax in oral administration and higher Cmax.
Conclusion:
As expected, CBD dose, route of administration, and diet were major determinants of CBD PK with oral routes providing higher bioavailability and nanotechnology formulations a faster onset. Although CBD appeared to have a faster onset and longer duration in women, more studies are required to delineate the role of biological sex. Factors that influence CBD PK have implications for medication development and appropriate dosing in clinical practice.
Keywords: pharmacokinetics, CBD, cannabis, clinical trials, bioavailability
Introduction
CBD, a cannabinoid constituent of the cannabis plant, has exponentially gained attention in both research and clinical applications as a potential treatment of several neuropsychiatric and general medical conditions.1 Epidiolex® was the first FDA-approved plant-derived CBD medication. Today, many CBD-based formulations are in development aiming for FDA approval and numerous nonapproved CBD preparations are available over the counter often implying beneficial “medicinal” properties.2 However, many questions raised by clinicians, researchers, and consumers of CBD products often relates to dosing and administration.
Pharmacokinetics (PK) is fundamental to medication development and guides appropriate dosing to achieve clinical effectiveness. Important factors normally considered for medication development include the route of administration and bioavailability. Keeping in line with the route of administration most preferred for medicinal purposes, the majority of CBD products currently available are for oral use. However, similar to other cannabinoids, CBD generally has poor bioavailability when consumed orally.3 That challenge has sparked a growth in the industry for the development of new nanotechnologies to improve bioavailability, thus increasing the diversity of formulations and delivery systems being used. There are now multiple CBD products being investigated in clinical studies with varying routes of administration, formulations, and administration conditions. Data generated from such studies should help to shed significant light on CBD PK parameters relevant to identifying those products potentially most suitable for subsequent trials regarding CBD's pharmacodynamic properties to alleviate specific clinical conditions.
However, one of the major challenges across studies and even in previous reviews has been the poor comparability of outcomes across different CBD PK studies owing to the different units and scales of reporting PK parameters, mainly geometric versus arithmetic scales.3 Although both scales have been commonly used in reporting PK data, they are not readily or precisely convertible, which contributes to confusion regarding PK outcomes. Of the scales, geometric method is preferred for reporting certain PK parameters owing to its greater robustness.4 No study has yet integrated the various CBD PK data on one scale.
In this review, we aimed to provide an updated systematic assessment of available evidence on the PK of CBD, covering recently published studies that were not included in previous reviews; provide comparable values of PK parameters from different studies on the same scale, and demonstrate patterns in outcomes based on the CBD dose and route of administration; and explore the simultaneous impact of different factors on PK outcomes using meta-regression models.
Materials and Methods
Protocol
This systematic review and meta-regression analysis was preregistered on PROSPERO (https://www.crd.york.ac.uk/prospero, ID: CRD42021269857) and conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines as much as it applied to pharmacokinetic studies.
Research objectives and outcomes
This review systematically assessed PK studies of CBD (pure CBD or in combination with THC) in healthy adults for both the quality of the studies and PK outcomes. The primary outcome was patterns of PK parameters of interest classified based on the route of entry and CBD dose to find potential meaningful patterns in outcomes that could help predict the PK of CBD relevant for clinical applications. The secondary outcome was the statistical significance level for each of the variables/factors that could potentially influence PK parameters, based on the available literature resulting from meta-regression models.
There were five PK parameters of interest:
-
(1)
Tmax: time from CBD administration to the maximum concentration of CBD in plasma/serum,
-
(2)
Cmax: maximum concentration of CBD in plasma/serum,
-
(3)
AUC0–t: the area under the curve of serum/plasma concentrations plotted against time from CBD administration to a specific time point, usually the end of the PK session,
-
(4)
AUC0–inf: the extrapolation of AUC0–t to the infinity time point,
-
(5)
Half-life, T1/2: the time needed for the concentration of the drug in the plasma to be reduced by 50%.
Potential variables of interest were the following: route of administration, CBD dose, CBD formulation, dietary (fasting/fed) status before CBD administration, abstinence from cannabis before CBD administration, sex, and duration of the PK session.3,5,6
Inclusion/exclusion criteria
Trials of any design were considered for inclusion if conducted on healthy adults (age between 18–65 years) and reported at least one of the PK parameters of interest after CBD administration in serum or plasma. CBD could be administered in any form, through any entry route, in any formulation or product. Studies were excluded if there was a history of major psychiatric disorders or general medical conditions in the sample, or if CBD was co-administered with another medication.
Search strategy
We systematically searched Medline, Embase, PsycInfo, Web of Science Core Collection, LILACS, and OpenGrey from inception to September 19, 2021, and in the case of Google Scholar, for the first 200 citations, using the following search terms: [Title/abstract➔ (CBD OR cannabidiol OR Sativex OR Epidiolex)] AND [Title/abstract➔ (pharmacokinetic OR concentration OR serum OR plasma OR blood)]. The above search was systematically updated on November 19, 2022 to cover publications since 2021 to date. Search terms were in English, but no language or publication period restriction was applied. Appropriate special characters/suffixes were used to search for any extension of the above terms. After the systematic search and screening, previous reviews of PK studies on CBD were searched manually for relevant original studies in addition to the reference lists of original articles published in the past 5 years. Experts in the field were consulted to seek any missed literature that could be included.
Study selection and data extraction
Two doctorate-level authors were co-trained and calibrated for the screening and data extraction on a sample of CBD PK studies. Any discrepancy would be discussed and resolved between the two screeners and the senior author at each stage. Conference abstracts and thesis reports were also included if the minimum required data were reported. Title-abstract and full-text screening were carried out in parallel using EndNote v. 9 (Cleverbridge, Inc.). Data extraction was carried out by one of the two authors, and then all the data were checked against the original source for accuracy by the other author.
Microsoft Excel sheets with predefined columns were used for extracting data, including but not limited to full citation details, study design, participants' age/sex/health status, sample size and dropouts, cannabis use pattern and abstinence status, fast/fed status before CBD administration, CBD source/supplier, details of CBD formulation, route of administration, CBD dosing, duration of the study session, values of any reported PK parameter in their original format, any relevant considerations of statistical methods, and finally any other relevant methodological information. Published articles, supplementary material, and pharmaceutical providers' websites were all used to collect relevant information, and in some cases, authors were contacted to obtain further details.
CBD formulation determination
CBD formulations were determined using three primary factors. First, when a patent was held and the specific ingredients were not provided in the methodology section for each study, a CBD sample would be designated as its own formulation (i.e., Epidiolex or Sativex). Second, when a methodology section indicated that the solution used a specific technology type (e.g., nanoparticle), these solutions were determined to be their own formulation. Third, when a study provided specific ingredients in the methodology section, the main base of the solution was determined to categorize different CBD formulation types. For example, most formulations consist mainly of oil-based, ethanol-based, gelatin-based, or water-based solutions. All CBD solutions were categorized using these three factors.
Quality assessment
The two authors who carried out the screening and data extraction also conducted a parallel quality assessment of all the studies. Any differences in ratings would be discussed and resolved between the two screeners and the senior author. Given the lack of a widely accepted quality assessment tool for PK studies, the US National Heart, Lung, and Blood Institute Quality Assessment Tool for Before-After Studies with no Control Group was deemed the best choice.7 The tool consists of 12 items assessing different aspects of before-after studies, from the clarity of the study question to the very end analysis. Each item is rated as Yes, No, Cannot Determine (CD), or Not Reported (NR). Specifically for the sample size question (Q5), a sample of >19 was considered as “Yes,” <10 was considered “No,” and in between was rated as “CD.”8
The assessor assigned an overall rating to each study as Poor, Fair, or Good. Toward a more precise and replicable application of this tool for PK studies, the authors weighted some items and developed a stratification strategy for overall rating, which can be found in Section A in the Supplementary Data.
Narrative synthesis
Study characteristics were organized, summarized, and presented separately for each trial arm in Table 1 in the order of date of publication. Similarities and differences among trial arms were described, including characteristics of the population such as sex, cannabis use, study design, diet status, CBD formulations, CBD dose, and reported outcomes. Numerical values of PK parameters were given in Supplementary Table S1 and described in the Quantitative Synthesis section.
Table 1.
Summary of Pharmacokinetic Studies of Cannabidiol
| M:F | Abstinence status before medication | Diet status | Study design | Route of administration | Technology/formulation | CBD dose, mg | Duration of the PK session | Reported PK parameters | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Abbotts et al. (2022)12 | Arm1 | 14 M | 3 Days | Fast | Probably open-label, randomized, crossover (unclear washout) | Oral solution | Water-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
| Arm2 | 14 M | 3 Days | Fed | Probably single-blinded, randomized, crossover (unclear washout) | Oral solution | Water-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
|
| Arm3 | 14 M | 3 Days | Fast | Probably open-label, randomized, crossover (unclear washout) | Oral oil | Oil-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
|
| Arm4 | 14 M | 3 Days | Fast | Probably open-label, randomized, crossover (unclear washout) | Oral solution | Water-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
|
| Arm5 | 14 M | 3 Days | Fast | Probably open-label, randomized, crossover (unclear washout) | Oral solution | Water-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
|
| Arm6 | 14 M | 3 Days | Fast | Probably open-label, randomized, crossover (unclear washout) | Oral solution | Water-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ke Vd |
|
| Bergeria et al. (2022)13 | Arm1a | 3:3 | Yes (urine negative) | Fed | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Oral capsule | Water-based | 100, Single dose | 58 | Cmax Tmax |
| Arm1b | 3:3 | Yes (urine negative) | Fed | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Oral solution | Epidiolex® formulation | 100, Single dose | 58 | Cmax Tmax |
|
| Arm1c | 3:3 | Yes (urine negative) | Fed | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Oral solution | Water-based | 100, Single dose | 58 | Cmax Tmax |
|
| Arm2 | 9:9 | Yes (urine negative) | Fed | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Inhalation/vaporization | N/A | 100, Single dose | 58 | Cmax Tmax |
|
| Arm3 | 9:9 | Yes (urine negative) | Fed | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Inhalation/vaporization | N/A | 100, Single dose | 58 | Cmax Tmax |
|
| Arm4 | 3:3 | Yes (urine negative) | Fast | Double-blind, double-dummy, randomized, crossover (washout 1 week) | Oral solution | Water-based | 100, Single dose | 58 | None | |
| Berl et al. (2022)14 | Arm1 | 8:8 | 30 Days | Fed | Triple-blind, randomized, parallel group | Oral solution | Nanotech | 9.76, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ka tlag λZ λ |
| Arm2 | 7:8 | 30 Days | Fed | Triple-blind, randomized, parallel group | Oral solution | Oil-based | 9.92, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 Ka tlag λZ λ |
|
| Busardò et al. (2021)15 | 12:2 | 5 Days | Fast | Open-label, nonrandomized, single arm | Inhalation/vaporization | N/A | 8, Single dose | 24 | Tmax Cmax AUC0–t T1/2 Kel |
|
| Hosseini et al. (2021)16 | Arm1 | 11:1 | 6 Months | Fast | Open-label, randomized, crossover (24 h washout) | Oromucosal/sublingual | Nanotech | 25, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf CL/F |
| Arm2 | 11:1 | 6 Months | Fast | Open-label, randomized, crossover (24 h washout) | Oromucosal/sublingual | Nanotech | 50, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf CL/F |
|
| Arm3 | 11:1 | 6 Months | Fast | Open-label, randomized, crossover (24 h washout) | Oral oil | Oil-based | 50, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf CL/F |
|
| Arm4 | 11:1 | 6 Months | Fast | Open-label, randomized, crossover (24 h washout) | Oromucosal spray | Sativex formulation | 25, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf CL/F |
|
| Arm5 | 3 Total | 6 Months | Fast | Double-blind, randomized, placebo-controlled | Oromucosal/sublingual | Nanotech | 50, Multiple dose | 12 | Tmax Cmax AUC0–t T1/2 |
|
| Vitetta et al. (2021)17 | Arm1 | 11 Total (25% male in the original sample) | Yes (urine negative) | Fast | Single-blind, randomized placebo-controlled | Oromucosal spray buccal | Nanotech | 6, Single dose | 12 | Tmax Cmax AUC0–t T1/2 CL/F |
| Arm2 | 12 Total (25% male in the original sample) | Yes (urine negative) | Fast | Single-blind, randomized placebo-controlled | Oromucosal spray buccal | Nanotech | 18, Multiple dose | 12 | Tmax Cmax AUC0–t T1/2 CL/F |
|
| Williams et al. (2021)18 | Arm1 | 9:6 | 3 Days | Fast | Double-blind, randomized, crossover (72 h washout) | Oral solution | Oil-based | 30, Single dose | 4 | Tmax Cmax AUC0–t Ka |
| Arm2 | 9:6 | 3 Days | Fast | Double-blind, randomized, crossover (72 h washout) | Oral solution | Water-based | 30, Single dose | 4 | Tmax Cmax AUC0–t Ka |
|
| Arm3 | 9:6 | 3 Days | Fast | Double-blind, randomized, crossover (72 h washout) | Oral solution | Oil-based | 30, Single dose | 4 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel Vd Ka |
|
| Arm4 | 9:6 | 3 Days | Fast | Double-blind, randomized, crossover (72 h washout) | Oral solution | Oil-based | 30, Single dose | 4 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel Vd Ka |
|
| Arm5 | 9:6 | 3 Days | Fast | Double-blind, randomized, crossover (72 h washout) | Oral solution | Oil-based | 30, Single dose | 4 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel Vd Ka |
|
| Crockett et al. (2020)19 | Arm1 | 12:17 | 1 Month | Fast | Open-label, randomized, crossover (14 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 96 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
| Arm2 | 9:6 | 1 Month | Fed | Open-label, randomized, crossover (14 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 96 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm3 | 3:11 | 1 Month | Fed | Open-label, randomized, crossover (14 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 96 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm4 | 6:9 | 1 Month | Fed | Open-label, randomized, crossover (14 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 96 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm5 | 6:9 | 1 Month | Fed | Open-label, randomized, crossover (14 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 96 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Hobbs et al. (2020)20 | Arm1 | 2:3 | 3 Days | Fast | Double-blind, randomized, parallel arm | Oral solution | Oil-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel Vd Ka Ke |
| Arm2 | 2:3 | 3 Days | Fast | Double-blind, randomized, parallel arm | Oral solution | Oil-based | 30, Single dose | 6 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel Vd Ka Ke |
|
| Izgelov et al. (2020)21 | Arm1 | 12 M | 30 Days | Fast | Blinded, randomized, crossover (3 weeks washout) | Oral capsule | Nanotech | 90, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf Kel CL/F V/F |
| Arm2 | 12 M | 30 Days | Fast | Blinded, randomized, crossover (3 weeks washout) | Oral capsule | Oil-based | 90, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf Kel CL/F V/F |
|
| Arm3 | 12 M | 30 Days | Fast | Blinded, randomized, crossover (3 weeks washout) | Oral capsule | Water-based | 90, Single dose | 24 | Tmax Cmax AUC0–t |
|
| Pérez-Acevedo et al. (2021)22 | Arm1 | 11:2 | Yes (urine negative) | Fast | Open-label, nonrandomized, crossover (15 days washout) | Oral oil | Oil-based | 0.9, Single dose | 24 | Tmax Cmax AUC0–t T1/2 Kel |
| Arm2 | 11:2 | Yes (urine negative) | Fast | Open-label, nonrandomized, crossover (15 days washout) | Oral decoction | Water-based | 0.7, Single dose | 24 | Tmax Cmax AUC0–t T1/2 Kel |
|
| Perkins et al. (2020)23 | Arm1 | 4:2 | 3 Months | Fed | Double-blind, randomized, placebo-controlled | Oral solution | Oil-based | 5 mg/kg, Single dose | 168 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
| Arm2 | 5:1 | 3 Months | Fed | Double-blind, randomized, placebo-controlled | Oral solution | Oil-based | 10 mg/kg, Single dose | 168 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Arm3 | 5:1 | 3 Months | Fed | Double-blind, randomized, placebo-controlled | Oral solution | Oil-based | 20 mg/kg, Single dose | 168 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Pichini et al. (2020)24 | Arm1 | 1 M | NR | NR | Open-label, nonrandomized, crossover (2 weeks washout) | Oral decoction | Water-based | 0.42, Single dose | 24 | Tmax Cmax AUC0–t T1/2 |
| Arm2 | 1 M | NR | NR | Open-label, non-randomized, crossover (2 weeks washout) | Oral oil | Oil-based | 0.86, Single dose | 24 | Tmax Cmax AUC0–t T1/2 |
|
| Tayo et al. (2020)25 | 3:5 | 1 Month | Fed | Open-label, nonrandomized, parallel-group | Oral solution | Epidiolex formulation | 200, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Knaub et al. (2019)26 | Arm1 | 8:8 | 7 Days | Fast | Double-blind, randomized, crossover (14 days washout) | Oral capsule | Nanotech | 25, Single dose | 24 | Tmax Cmax AUC0–t |
| Arm2 | 8:8 | 7 Days | Fast | Double-blind, randomized, crossover (14 days washout) | Oral capsule | Oil-based | 25, Single dose | 24 | Tmax Cmax AUC0–t |
|
| Morrison et al. (2019)27 | Arm1 | 9 Total | 3 Months | Fed | Open-label, nonrandomized, parallel arm | Oral solution | Epidiolex formulation | 750, Multiple dose | 12 | Tmax Cmax AUC0–t |
| Arm2 | 8 Total | |||||||||
| Arm3 | 6 Total | 3 Months | Fed | Open-label, nonrandomized, parallel arm | Oral solution | Epidiolex formulation | 250, Multiple dose | 12 | Tmax Cmax AUC0–t |
|
| Arm4 | 4 Total | |||||||||
| Arm5 | 14 Total | |||||||||
| Arm6 | 9:6 | 3 Months | Fed | Open-label, nonrandomized, parallel arm | Oral solution | Epidiolex formulation | 750, Multiple dose | 12 | Tmax Cmax AUC0–t |
|
| Arm7 | 8:4 | |||||||||
| Arm8 | 9:5 | |||||||||
| Patrician et al. (2019)28 | Arm1 | 12 M | Yes (time NR) | Fed | Double-blind, placebo-controlled, crossover (6 days washout) | Oral capsule | Nanotech | 45, Single dose | 6 | Tmax Cmax AUC0–t |
| Arm2 | 12 M | Yes (time NR) | Fed | Double-blind, placebo-controlled, crossover (6 days washout) | Oral capsule | Nanotech | 90, Single dose | 6 | Tmax Cmax AUC0–t |
|
| Arm3 | 12 M | Yes (time NR) | Fed | Double-blind, placebo-controlled, crossover (6 days washout) | Oral capsule | Oil-based | 45, Single dose | 6 | Tmax Cmax AUC0–t |
|
| Arm4 | 12 M | Yes (time NR) | Fed | Double-blind, placebo-controlled, crossover (6 days washout) | Oral capsule | Oil-based | 90, Single dose | 6 | Tmax Cmax AUC0–t |
|
| Taylor et al. (2019)29 | 4:4 | 1 Month | Fed | Open-label, nonrandomized, parallel group | Oral solution | Epidiolex formulation | 200, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Atsmon et al. (2018a)30 | 15 M | 30 Days | Fed | Open-label, randomized, crossover (7 days washout) | Oral capsule | Nanotech | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel |
|
| Atsmon et al. (2018b)31 | Arm1 | 15 M | Yes (urine negative) | Fed | Open-label, randomized, crossover (7 days washout) | Oral capsule | Nanotech | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
| Arm2 | 15 M | Yes (urine negative) | Fed | Open-label, randomized, crossover (7 days washout) | Oral capsule | Nanotech | 100, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Arm3 | 15 M | Yes (urine negative) | Fed | Open-label, randomized, crossover (7 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel |
|
| Meyer et al. (2018)32 | 8, Total | Yes (urine negative) | Fasted, irrelevant | Open-label, nonrandomized, single arm | IV | N/A | 1.6, Single dose | 58 | Tmax Cmax AUC0–t |
|
| Schoedel et al. (2018)33 | Arm1 | 38 Total (72.1% male in original sample) | >12 Days | Fast | Double-blind, randomized, placebo-controlled crossover (8 days washout) | Oral solution | Epidiolex formulation | 750, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf |
| Arm2 | 39 Total (72.1% male in original sample) | >12 Days | Fast | Double-blind, randomized, placebo-controlled crossover (8 days washout) | Oral solution | Epidiolex formulation | 1500, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf |
|
| Arm3 | 40 Total (72.1% male in original sample) | >12 Days | Fast | Double-blind, randomized, placebo-controlled crossover (8 days washout) | Oral solution | Epidiolex formulation | 4500, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf |
|
| Taylor et al. (2018)34 | Arm1 | 1:5 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 1500, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
| Arm2 | 3:3 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 3000, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm3 | 0:6 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 4500, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm4 | 2:4 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 6000, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm5 | 2:7 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 750, Single dose and multiple dose | 12 | Tmax Cmax AUC0–t T1/2 |
|
| Arm6 | 5:4 | 1 Month | Fast | Double-blind, randomized, placebo-controlled | Oral solution | Epidiolex formulation | 1500, Single dose and multiple dose | 12 | Tmax Cmax AUC0–t T1/2 |
|
| Arm7 | 4:8 | 1 Month | Fast | Open-label, randomized, crossover (7 days washout) | Oral solution | Epidiolex formulation | 1500, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Arm8 | 4:8 | 1 Month | Fed | Open-label, randomized, crossover (7 days washout) | Oral solution | Epidiolex formulation | 1500, Single dose | 48 | Tmax Cmax AUC0–t AUC0–inf T1/2 CL/F V/F |
|
| Cherniakov et al. (2017)35 | Arm1 | 9 M | 28 Days | Fast | Open-label, randomized, crossover (21 days washout) | Oral capsule | Nanotech | 10, Single dose | 24 | Tmax Cmax AUC0–t Kel |
| Arm2 | 9 M | 28 Days | Fast | Open-label, randomized, crossover (21 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t Kel |
|
| Haney et al. (2016)36 | 8 Total | No (urine positive) | Fed | Probably open-label, nonrandomized, single arm | Oral capsule | NR | 800, Single dose | 6 | Tmax Cmax |
|
| Desrosiers et al. (2014)37 | Arm1 | 10:4 | No (urine positive) | NR_ irrelevant | Open-label, nonrandomized, parallel arm | Inhalation/smoking | N/A | 2, Single dose | 30 | Tmax Cmax |
| Arm2 | 8:3 | No (urine positive) | NR_irrelevant | Open-label, nonrandomized, parallel arm | Inhalation/smoking | N/A | 2, Single dose | 30 | Tmax Cmax |
|
| Sellers et al. (2013)38 | Arm1 | 60 Total (64.2%:35.8%) | 90 Days | Fast | Double-blind, randomized, placebo-controlled, parallel arm | Oromucosal spray | Sativex formulation | 20, Multiple dose | 24 | Tmax Cmax AUC0–t AUC0–inf |
| Arm2 | 51 Total (64.2%:35.8%) | 90 Days | Fast | Double-blind, randomized, placebo-controlled, parallel arm | Oromucosal spray | Sativex formulation | 60–90, Multiple dose | 24 | Tmax Cmax AUC0–t AUC0–inf |
|
| Stott et al. (2013a)39 | Arm1 | 6 M | 30 Days | Fast | Open-label, randomized, parallel arm | Oromucosal spray | Sativex formulation | 5, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F |
| Arm3 | 6 M | 30 Days | Fast | Open-label, randomized, parallel arm | Oromucosal spray | Sativex formulation | 20, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F |
|
| Arm4 | 6 M | 30 Days | Fast | Open-label, randomized, parallel arm | Oromucosal spray | Sativex formulation | 5, Multiple dose | 24 | Tmax Cmax AUC0–t |
|
| Arm5 | 12 M | 30 Days | Fast | Open-label, randomized, parallel arm | Oromucosal spray | Sativex formulation | 10, Multiple dose | 24 | Tmax Cmax AUC0–t |
|
| Arm6 | 6 M | 30 Days | Fast | Open-label, randomized, parallel arm | Oromucosal spray | Sativex formulation | 20, Multiple dose | 24 | Tmax Cmax AUC0–t |
|
| Stott et al. (2013b)40 | Arm1 | 12 M | 30 Days | Fed | Open-label, probably nonrandomized, crossover (3 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F |
| Arm2 | 12 M | 30 Days | Fast | Open-label, probably nonrandomized, crossover (3 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F |
|
| Stott et al. (2013c)41 | Arm1 | 12 M | 30 Days | NR | Open-label, randomized, intra-arm crossover (7 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F V/F |
| Arm2 | 12 M | 30 Days | NR | Open-label, randomized, intra-arm crossover study (5 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F V/F |
|
| Arm3 | 12 M | 30 Days | NR | Open-label, randomized, intra-arm crossover study (2 days washout) | Oromucosal spray | Sativex formulation | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel CL/F V/F |
|
| Eichler et al. (2012)42 | Arm1 | 9 M | Yes (urine negative) | Fast | Double-blind, randomized, crossover (2 weeks washout) | Oral capsule | Alcohol-based | 27.8, Single dose | 24 | Tmax Cmax AUC0–t |
| Arm2 | 9 M | Yes (urine negative) | Fast | Double-blind, randomized, crossover (2 weeks washout) | Oral capsule | Alcohol-based | 14.8, Single dose | 24 | Tmax Cmax AUC0–t |
|
| Karschner et al. (2011)43 | Arm1 | 6:3 | Yes (urine negative) | Fed | Double-blind, randomized, placebo-controlled, double dummy | Oromucosal spray | Sativex formulation | 5, Single dose | 10 | Tmax Cmax AUC0–t |
| Arm2 | 6:3 | Yes (urine negative) | Fed | Double-blind, randomized, placebo-controlled, double dummy | Oromucosal spray | Sativex formulation | 15, Single dose | 10 | Tmax Cmax AUC0–t |
|
| Schwope et al. (2011)44 | 9:1 | No (urine positive) | NR_ irrelevant | Probably open-label, nonrandomized, single arm | Inhalation/smoking | N/A | 2, Single dose | 6 | Tmax Cmax |
|
| Nadulski et al. (2005a)45 | Arm1 | 12:12 | 30 Days | Fast | Double-blind, randomized, placebocontrolled crossover (1 week washout) | Oral capsule | Oil-based | 5.4, Single dose | 24 | Tmax Cmax AUC0–t |
| Arm2 | 12 Total | 30 Days | Fast | Open-label, nonrandomized, crossover (1 week washout) | Oral capsule | Oil-based | 5.4, Single dose | 24 | Tmax Cmax AUC0–t |
|
| Nadulski et al. (2005b)46 | 24 Total | NR | Fast | Double-blind, probably randomized, placebo-controlled | Oral capsule | NR | 5.4, Single dose | 24 | Tmax Cmax |
|
| Guy and Flint (2004)47 | Arm1 | 3:3 | 30 Days | Fast | Double-blind, placebo-controlled, crossover (6 days washout) | Oromucosal/sublingual | Alcohol-based | 20, Single dose | 12 | Tmax Cmax AUC0–t |
| Arm2 | 3:3 | 30 Days | Fast | Open-label, crossover (6 days washout) | Inhalation/nebulizer | Oil-based | 20, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel |
|
| Arm3 | 3:3 | 30 Days | Fast | Open-label, crossover (6 days washout) | Oromucosal/aerosol/sublingual | Alcohol-based | 20, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel |
|
| Arm4 | 3:3 | 30 Days | Fast | Open-label, crossover (6 days washout) | Oromucosal/sublingual | Alcohol-based | 20, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 Kel |
|
| Guy and Robson (2004a)48 | 24 M | Yes (urine negative) | Fed | Double-blind, randomized placebo-controlled | Oromucosal spray sublingual | Alcohol-based | 10, Single dose | 24 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Guy and Robson (2004b)49 | Arm1 | 6:6 | 30 Days | Fast | Open-label, randomized, crossover (6 days washout) | Oromucosal spray sublingual | Alcohol-based | 10, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
| Arm2 | 6:6 | 30 Days | Fast | Open-label, randomized, crossover (6 days washout) | Oromucosal spray buccal | Alcohol-based | 10, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Arm3 | 6:6 | 30 Days | Fast | Open-label, randomized, crossover (6 days washout) | Oromucosal spray oro-pharyngeal | Alcohol-based | 10, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Arm4 | 6:6 | 30 Days | Fast | Open-label, nonrandomized, crossover (6 days washout) | Oral capsule | Gelatin-based | 10, Single dose | 12 | Tmax Cmax AUC0–t AUC0–inf T1/2 |
|
| Ohlsson et al. (1986)50 | Arm1 | 5 M | 72 h | NR_irrelevant | Open-label, randomized, crossover (1 week washout) | IV | Alcohol-based | 20, Single dose | 72 | Tmax Cmax AUC0–t T1/2 CL/F Vd |
| Arm2 | 5 M | 72 h | NR_irrelevant | Open-label, randomized, crossover (1 week washout) | Inhalation/smoking | N/A | 19.2, Single dose | 72 | Tmax Cmax AUC0–t T1/2 |
|
N/A, not applicable; NR, not reported; IV, intravenous; PK, pharmacokinetics.
Statistical analyses
To make between-study comparisons and meta-regression analysis feasible, the reported values for PK parameters went through two steps of conversion/estimation, using online calculators and SPSS v. 28 (IBM Statistics).
In the first step, values were converted into a unified form consisting of hours for Tmax and T1/2, ng/mL for Cmax, h×ng/mL for AUC0–t and AUC0–inf. In the second step, we converted the values for Tmax and T1/2 into arithmetic mean (standard deviation [SD]) format and all the values for Cmax, AUC0–t, and AUC0–inf in geometric mean (coefficient of variation [CV%]) format as the preferred method for reporting of PK parameters.4 This was carried out because Cmax, AUC0–t, and AUC0–inf are usually highly variable and skewed, so a geometric scale is preferrable. In contrast, Tmax and T1/2 are less variable and an arithmetic scale is an acceptable method, which also would require less conversions in this review, thus allowing for higher accuracy. For the linear meta-regression analysis, confidence intervals were estimated and log-transformed for every parameter's values as necessary. Step 2 conversions are based on previously published methods and the latest guidelines,9–11 summarized and simplified in the Section B in the Supplementary Data to help with utilization in other PK reviews and replicability of this work.
Missing data were addressed as follows: (1) if the mean or median was not reported for a specific PK parameter, it was considered missing and was not imputed; and (2) if no measure of distribution (e.g., SD or CV%) was reported for a PK parameter, but the mean or median was reported, the SD (for Tmax and T1/2) or CV% (for Cmax, AUC0–t, AUC0–inf) was imputed using the largest SD or CV% value available for that parameter among all single-dose trial arms.
Comprehensive Meta-Analysis v. 3 (Biostat, Inc.) software was used for random-effects multivariable meta-regression analysis, with PK parameters as dependent outcomes and route of administration, CBD dose, diet status, CBD formulation, female ratio, and duration of PK session as independent predictors. Details of considerations for modeling are provided in the Section C in the Supplementary Data.
Overall, three groups of models were built for each PK parameter across single-dose trial arms: (1) models including all the three routes of CBD administration (inhalation, oromucosal, and oral); (2) models including only oromucosal and oral trial arms; and (3) models including only oral CBD trial arms. For sensitivity analysis, models were conducted once with all the single-dose trial arms irrespective of the quality rating and then a second time only including trial arms with a “Good” or “Fair” quality rating. For each model, the number of included arms was reported alongside the R-squared value to measure model fit, indicating the amount of variability in the data that the model could explain. For each variable in the model, the significance level (alpha=0.05) and the positive or negative sign of the regression coefficient were reported to indicate a positive or negative association, respectively. Because models were based on log-transformed data, the net regression coefficient values were not interpretable in terms of effect size, and thus only their positive/negative sign was reported.
Results
Study selection and overview
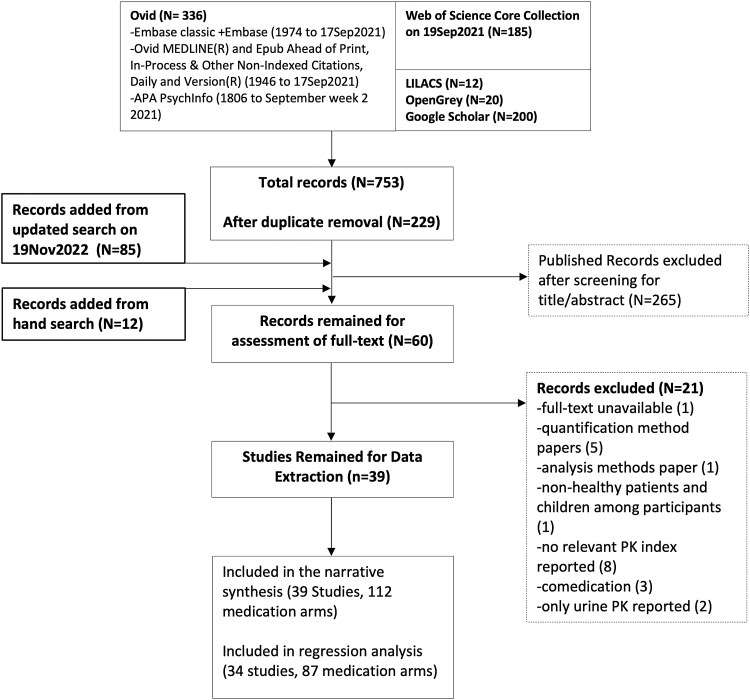
After processing all the retrieved records, 39 studies comprising 112 trial arms were included in the narrative synthesis,12–50 out of which 34 studies comprising 87 arms were used for quantitative comparisons and regression analysis (Fig. 1). Of the 25 trial arms that were excluded from the analysis, but included in the narrative synthesis, 15 trial arms administered multiple doses of CBD (Table 1), 2 arms had only 1 participant,24 2 arms were intravenous administration of CBD,32,50 PK values was not reported for 1 trial arm,13 and whole blood was used instead of plasma or serum for 5 trial arms.13
FIG. 1.
PRISMA flow diagram. PRISMA, preferred reporting items for systematic reviews and meta-analyses.
Quality assessment
Twenty-six trial arms were rated as “Good,” 70 as “Fair,” and 16 as “Poor” based on 12 criteria (Q1–Q12) (Table 2). Most studies clearly stated the study question (Q1), described eligibility criteria (Q2) and had a representative sample in terms of inclusion/exclusion criteria (Q3). At the same time, they mostly failed to report whether all the eligible potential participants were included (Q4), and most had either moderate or low sample sizes (Q5), which was an essential consideration for rating. The intervention was well described in most studies (Q6), and PK outcomes were well-defined and consistently assessed (Q7). The majority of trial arms were non–double blind (Q8). Subject loss at follow-up (Q9) was reported in most studies. Statistical methods primarily measured before-after changes (Q10), for multiple times (Q11), except in some cases where it was not clearly stated. All studies were conducted at the individual participant level with no group interventions applicable to any of the studies (Q12).
Table 2.
Quality Assessment of Pharmacokinetic Studies of Cannabidiol
| Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Q9 | Q10 | Q11 | Q12 | Overall | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbotts et al. (2022)12 | Arm1 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair | |
| Arm5 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair | |
| Arm6 | Yes | Yes | Yes | Yes | CD | Yes | Yes | NR | Yes | Yes | Yes | N/A | Fair | |
| Bergeria et al. (2022)13 | Arm1a | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair |
| Arm1b | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Arm1c | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Berl et al. (2022)14 | Arm1 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Busardò et al. (2021)15 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Hosseini et al. (2021)16 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm5 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Vitetta et al. (2021)17 | Arm1 | Yes | Yes | Yes | Yes | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | Yes | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Good | |
| Williams et al. (2021)18 | Arm1 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm3 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm4 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm5 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Crockett et al. (2020)19 | Arm1 | Yes | Yes | Yes | NR | Yes | Yes | Yes | No | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm5 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Hobbs et al. (2020)20 | Arm1 | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Izgelov et al. (2020)21 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Pérez-Acevedo et al. (2021)22 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Perkins et al. (2020)23 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Pichini et al. (2020)24 | Arm1 | Yes | No | CD | No | No | Yes | Yes | NR | N/A | Yes | Yes | N/A | Poor |
| Arm2 | Yes | No | CD | No | No | Yes | Yes | NR | N/A | Yes | Yes | N/A | Poor | |
| Tayo et al. (2020)25 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Knaub et al. (2019)26 | Arm1 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | Yes | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Morrison et al. (2019)27 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | NR | N/A | Poor |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm5 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm6 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm7 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Arm8 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | CD | NR | N/A | Poor | |
| Patrician et al. (2019)28 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm4 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Taylor et al. (2019)29 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Atsmon et al. (2018a)30 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Atsmon et al. (2018b)31 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Meyer et al. (2018)32 | Yes | Yes | Yes | NR | No | Yes | Yes | No | No | Yes | Yes | N/A | Poor | |
| Schoedel et al. (2018)33 | Arm1 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm3 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Taylor et al. (2018)34 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm5 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm6 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair | |
| Arm7 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Arm8 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Cherniakov et al. (2017)35 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | Yes | N/A | Poor |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | CD | Yes | N/A | Poor | |
| Haney et al. (2016)36 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | No | NR | Yes | N/A | Poor | |
| Desrosiers et al. (2014)37 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | NR | NR | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | NR | NR | Yes | Yes | N/A | Fair | |
| Sellers et al. (2013)38 | Arm1 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | N/A | Good | |
| Stott et al. (2013a)39 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm5 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm6 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Stott et al. (2013b)40 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair | |
| Stott et al. (2013c)41 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair | |
| Eichler et al. (2012)42 | Arm1 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Karschner et al. (2011)43 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | NR | Yes | Yes | N/A | Fair | |
| Schwope et al. (2011)44 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair | |
| Nadulski et al. (2005a)45 | Arm1 | Yes | Yes | Yes | NR | Yes | Yes | Yes | Yes | NR | Yes | Yes | N/A | Good |
| Arm2 | Yes | Yes | Yes | NR | CD | Yes | Yes | No | NR | Yes | Yes | N/A | Fair | |
| Nadulski et al. (2005b)46 | Yes | Yes | Yes | NR | Yes | Yes | Yes | Yes | NR | Yes | Yes | N/A | Good | |
| Guy and Flint (2004)47 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Guy and Robson (2004a)48 | Yes | Yes | Yes | NR | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Good | |
| Guy and Robson (2004b)49 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm3 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Arm4 | Yes | Yes | Yes | NR | No | Yes | Yes | No | Yes | Yes | Yes | N/A | Fair | |
| Ohlsson et al. (1986)50 | Arm1 | Yes | Yes | Yes | NR | No | Yes | Yes | No | NR | Yes | Yes | N/A | Poor |
| Arm2 | Yes | Yes | Yes | NR | No | Yes | Yes | No | NR | Yes | Yes | N/A | Poor | |
Q1: Was the study question or objective clearly stated? Q2: Were eligibility/selection criteria for the study population prespecified and clearly described? Q3: Were the participants in the study representative of those who would be eligible for the test/service/intervention in the general or clinical population of interest? Q4: Were all eligible participants that met the prespecified entry criteria enrolled? Q5: Was the sample size sufficiently large to provide confidence in the findings? Q6: Was the test/service/intervention clearly described and delivered consistently across the study population? Q7: Were the outcome measures prespecified, clearly defined, valid, reliable, and assessed consistently across all study participants? Q8: Were the people assessing the outcomes blinded to the participants' exposures/interventions? Q9: Was the loss to follow-up after baseline 20% or less? Were those lost to follow-up accounted for in the analysis? Q10: Did the statistical methods examine changes in outcome measures from before to after the intervention? Were statistical tests done that provided p-values for the pre-to-post changes? Q11: Were outcome measures of interest taken multiple times before the intervention and multiple times after the intervention (i.e., did they use an interrupted time-series design)? Q12: If the intervention was conducted at a group level (a whole hospital, a community, etc.) did the statistical analysis take into account the use of individual-level data to determine effects at the group level?
CD, cannot determine.
Narrative synthesis
Thirty studies had more than one treatment arm, of which 22 studies, comprising 69 arms, had crossover designs with washout periods ranging between 24 h and 21 days (Table 1). Seventeen studies, comprising 42 arms, had at least one double-blind arm. Sixty-six trial arms had either only male participants or a higher number of males than females, while the sex ratio was either equal to one or favored females in 36 arms. Participants' sex was not clearly reported for 10 arms. Participants were abstinent from cannabis before study initiation in 105 arms and had fasted before CBD administration in 63 arms. Eight arms used inhalation as the route of administration, 29 oromucosal, 73 oral, and 2 intravenous. A variety of formulations were used consisting of nanotech (n=14), oil-based (n=21), alcohol-based (n=10), and water-based (n=12), alongside Sativex (n=17) and Epidiolex (n=22) formulations. For single-dose studies, CBD doses ranged between 2–100 mg in inhalation, 5–50 mg in oromucosal, and 0.42–6000 mg in oral administration (Table 1). The duration of the PK session was between 4–164 h. All trial arms reported Tmax and Cmax except one that did not report any values, 96 reported AUC0–t, 59 reported AUC0–inf, and 60 reported T1/2. Only 22 treatment arms from 4 studies reported Cmax and AUC in geometric scale (Supplementary Table S1 and Showcase in the Supplementary Data). At least one PK parameter from each and all treatment arms needed to go through second step conversions to geometric values to conform to the reporting format in Supplementary Table S1.
Quantitative synthesis
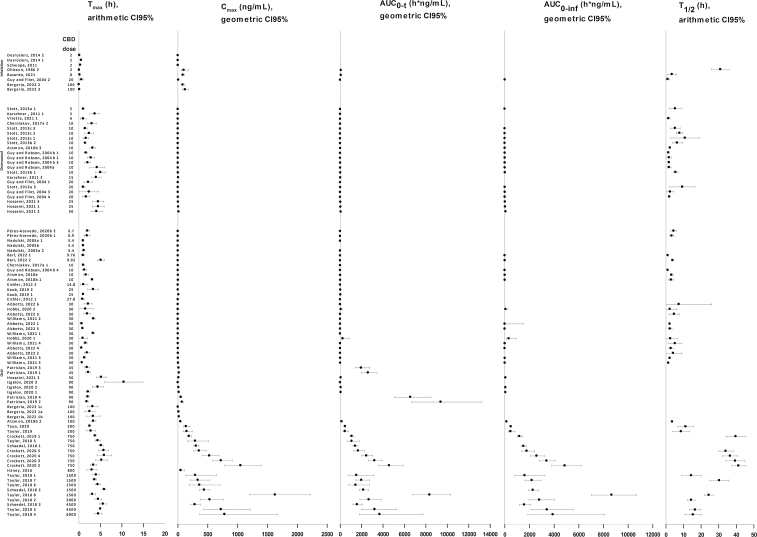
Reported PK values
Given the large variability of the doses studied, Table 3 provides a summary of the PK parameters for doses consisting of more than two trials. Supplementary Table S1 presents the PK parameters from all single-dose trial arms. Figure 2 demonstrates the PK parameters in order of increasing CBD dose for all the trial arms. Supplementary Figure S1 is a magnified version of Figure 2 for CBD doses ≤100 mg. Among single-dose trial arms, the arithmetic mean Tmax ranged between 0.00 and 0.60 h for inhalation, 1.00–5.01 h for oromucosal, and 0.59–10.45 h for oral administration (Supplementary Table S1 and Fig. 2). Geometric mean Cmax ranged between 0.42 and 120.77 ng/mL for inhalation, 0.38–12.90 ng/mL for oromucosal, and 0.22–1628 ng/mL for oral administration.
Table 3.
Pharmacokinetic Parameters of Cannabidiol in Single-Dose Studies in Accordance with Increasing Cannabidiol Dose
| CBD dose |
Tmax, arithmetic
|
Cmax, geometric
|
AUC0–t, geometric
|
AUC0–inf, geometric
|
T1/2, arithmetic
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Lower a | Upper a | Mean | Lower | Upper | Mean | Lower | Upper | Mean | Lower | Upper | Mean | Lower | Upper | ||
| Oromucosal formulations | ||||||||||||||||
| Cherniakov (2017) 2 | 10 | 3 | 1.96 | 4.04 | 0.43 | 0.28 | 0.66 | 2.89 | 2.17 | 3.85 | ||||||
| Stott (2013c) 3 | 10 | 1.46 | 0.97 | 1.95 | 0.52 | 0.35 | 0.77 | 1.53 | 1.05 | 2.24 | 2.71 | 2.03 | 3.61 | 5.22 | 2.35 | 8.09 |
| Stott (2013c) 2 | 10 | 2.38 | 1.36 | 3.40 | 0.58 | 0.41 | 0.8 | 1.58 | 1.13 | 2.21 | 3.45 | 3 | 3.98 | 7.81 | 5.90 | 9.72 |
| Stott (2013c) 1 | 10 | 1.63 | 0.95 | 2.31 | 0.81 | 0.52 | 1.26 | 2.7 | 1.84 | 3.95 | 4.37 | 3.08 | 6.22 | 10.86 | 2.78 | 18.94 |
| Stott (2013b) 2 | 10 | 1.45 | 1.16 | 1.74 | 0.97 | 0.67 | 1.41 | 3.57 | 2.31 | 5.54 | 4.57 | 3.02 | 6.9 | 6.39 | 3.54 | 9.24 |
| Atsmon (2018b) 3 | 10 | 3.18 | 2.55 | 3.81 | 1.81 | 1.37 | 2.39 | 6.8 | 5.51 | 8.38 | 7.35 | 6.06 | 8.92 | 2.31 | 1.91 | 2.71 |
| Guy and Robson (2004b) 1 | 10 | 1.63 | 1.20 | 2.06 | 2.02 | 1.33 | 3.06 | 5.75 | 3.97 | 8.32 | 6.09 | 4.27 | 8.69 | 1.44 | 0.94 | 1.94 |
| Guy and Robson (2004b) 2 | 10 | 2.79 | 1.96 | 3.62 | 2.09 | 1.21 | 3.61 | 5.19 | 3.44 | 7.83 | 5.69 | 3.89 | 8.32 | 1.81 | 0.51 | 3.11 |
| Guy and Robson (2004b) 3 | 10 | 2.04 | 1.32 | 2.76 | 2.11 | 1.39 | 3.19 | 6.53 | 4.46 | 9.55 | 6.97 | 4.8 | 10.12 | 1.76 | 1.25 | 2.27 |
| Guy and Robson (2004a) | 10 | 4.22 | 2.44 | 6.00 | 2.21 | 1.51 | 3.24 | 6.83 | 4.46 | 10.45 | 8.29 | 5.78 | 11.91 | 1.81 | 0.76 | 2.86 |
| Stott (2013b) 1 | 10 | 5.01 | 3.84 | 6.18 | 3.11 | 2.16 | 4.47 | 18.65 | 14.46 | 24.06 | 21.46 | 16.79 | 27.44 | 5.49 | 4.11 | 6.87 |
| Range | 1.46–5.01 | 0.95–3.84 | 1.74–6.18 | 0.43–3.11 | 0.28–2.16 | 0.66–4.47 | 1.53–18.65 | 1.05–14.46 | 2.21–24.06 | 2.71–21.46 | 2.03–16.79 | 3.61–27.44 | 1.44–10.86 | 0.51–5.90 | 1.94–19.94 | |
| Guy and Flint (2004) 1 | 20 | 2.17 | 1.14 | 3.20 | 1.87 | 1.19 | 2.93 | 1.57 | 0.54 | 4.51 | ||||||
| Stott (2013a) 3 | 20 | 1 | 0.58 | 1.42 | 1.89 | 1.09 | 3.29 | 7.36 | 3.26 | 16.61 | 9.54 | 4.06 | 22.4 | 9.36 | 2.21 | 16.51 |
| Guy and Flint (2004) 3 | 20 | 2.35 | 0.12 | 4.58 | 2.3 | 1.36 | 3.87 | 3.68 | 1.46 | 9.29 | 13.07 | 9.9 | 17.24 | 2.4 | 0.28 | 4.52 |
| Guy and Flint (2004) 4 | 20 | 1.67 | 0.81 | 2.53 | 2.5 | 1.9 | 3.28 | 2.78 | 1.37 | 5.64 | 8.91 | 5.85 | 13.56 | 1.97 | 1.32 | 2.62 |
| Range | 1–2.35 | 0.12–1.14 | 1.42–4.58 | 1.87–2.5 | 1.09–1.9 | 2.93–3.87 | 1.57–7.36 | 0.54–3.26 | 4.51–16.61 | 8.91–13.07 | 4.06–9.9 | 13.56–22.4 | 1.97–9.36 | 0.28–2.21 | 2.62–16.51 | |
| Oral formulations | ||||||||||||||||
| Cherniakov (2017) 1 | 10 | 1 | 0.74 | 1.26 | 1.82 | 1.21 | 2.74 | 6.01 | 4.01 | 9.00 | ||||||
| Guy and Robson (2004b) 4 | 10 | 1.27 | 0.74 | 1.80 | 1.83 | 1.12 | 2.99 | 4.37 | 2.73 | 7.01 | 4.65 | 2.95 | 7.35 | 1.09 | 0.80 | 1.38 |
| Atsmon (2018a) | 10 | 1.64 | 0.99 | 2.29 | 2.85 | 2.49 | 3.27 | 8.97 | 7.06 | 11.4 | 9.66 | 7.69 | 12.14 | 3.21 | 2.31 | 4.11 |
| Atsmon (2018b) 1 | 10 | 3.1 | 2.85 | 3.35 | 2.99 | 2.42 | 3.7 | 8.91 | 7.15 | 11.1 | 9.57 | 7.72 | 11.85 | 2.95 | 1.52 | 4.38 |
| Range | 1–3.1 | 0.74–2.85 | 1.26–3.35 | 1.82–2.99 | 1.12–2.49 | 2.74–3.7 | 4.37–8.97 | 2.73–7.15 | 7.01–11.4 | 4.65–9.66 | 2.95–7.72 | 7.35–12.14 | 1.09–3.21 | 0.80–2.31 | 1.38–4.38 | |
| Abbotts (2022) 6 | 30 | 2.16 | 1.16 | 3.16 | 0.2219 | 0.107 | 0.4601 | 0.4727 | 0.2435 | 0.9179 | 7.38 | 0 | 26.05 | |||
| Hobbs (2020) 2 | 30 | 1.5 | 0 | 3.36 | 0.43 | 0.14 | 1.33 | 54.48 | 15.59 | 190.35 | 68.24 | 23.55 | 197.69 | 2.3 | 0 | 6.21 |
| Abbotts (2022) 3 | 30 | 1.94 | 1.13 | 2.75 | 0.4642 | 0.3634 | 0.593 | 0.948 | 0.7113 | 1.2634 | 4.68 | 1.75 | 7.61 | |||
| Williams (2021) 2 | 30 | 3.39 | 3.03 | 3.75 | 0.72 | 0.39 | 1.31 | 1.47 | 0.87 | 2.48 | ||||||
| Abbotts (2022) 1 | 30 | 0.64 | 0.40 | 0.88 | 1.3828 | 0.9092 | 2.1031 | 2.548 | 1.8574 | 3.4953 | 4.0519 | 0.011 | 1491.9221 | 2.22 | 1.95 | 2.49 |
| Abbotts (2022) 5 | 30 | 0.86 | 0.63 | 1.09 | 1.6279 | 1.0399 | 2.5482 | 2.8186 | 1.9374 | 4.1006 | 5.2783 | 2.6764 | 10.4098 | 2.34 | 1.26 | 3.42 |
| Williams (2021) 1 | 30 | 3.29 | 2.95 | 3.63 | 1.67 | 1.11 | 2.52 | 3.5 | 2.33 | 5.25 | ||||||
| Hobbs (2020) 1 | 30 | 0.9 | 0 | 2.02 | 1.87 | 0.61 | 5.76 | 245.62 | 70.3 | 858.2 | 329.82 | 113.85 | 955.53 | 2.54 | 0 | 6.85 |
| Williams (2021) 4 | 30 | 1.53 | 0.97 | 2.09 | 2.18 | 1.45 | 3.3 | 5.12 | 3.58 | 7.34 | 7.72 | 5.2 | 11.46 | 5.18 | 1.26 | 9.10 |
| Abbotts (2022) 4 | 30 | 0.59 | 0.46 | 0.72 | 2.5665 | 1.7999 | 3.6598 | 3.8146 | 2.7132 | 5.3631 | 5.5323 | 3.1207 | 9.8074 | 2.85 | 1.61 | 4.09 |
| Abbotts (2022) 2 | 30 | 1.89 | 1.21 | 2.57 | 2.6463 | 2.067 | 3.388 | 6.1004 | 4.8302 | 7.7046 | 4.14 | 0 | 8.84 | |||
| Williams (2021) 3 | 30 | 1.28 | 0.94 | 1.62 | 3.21 | 2.51 | 4.1 | 6.98 | 5.37 | 9.08 | 11.87 | 8.76 | 16.1 | 2.2 | 1.57 | 2.83 |
| Williams (2021) 5 | 30 | 0.7 | 0.57 | 0.83 | 4.79 | 3.53 | 6.49 | 7.73 | 5.62 | 10.63 | 9.52 | 7.22 | 12.54 | 1.42 | 1.13 | 1.71 |
| Range | 0.59–3.39 | 0–3.03 | 0.72–3.75 | 0.22–4.79 | 0.107–3.53 | 0.46–6.49 | 0.47–245.62 | 0.24–70.3 | 0.92–858.2 | 4.05–329.82 | 0.01–113.85 | 10.41–1491.92 | 1.42–7.38 | 0–1.95 | 1.71–26.05 | |
| Izgelov (2020) 3 | 90 | 10.45 | 5.96 | 14.94 | 0.6 | 0.37 | 0.97 | 6.4 | 4.19 | 9.78 | ||||||
| Izgelov (2020) 2 | 90 | 4.38 | 3.12 | 5.64 | 12.52 | 9.28 | 16.91 | 60.76 | 46.92 | 78.68 | 65.73 | 51.21 | 84.36 | |||
| Izgelov (2020) 1 | 90 | 2.13 | 1.64 | 2.62 | 16.1 | 11.93 | 21.74 | 58.51 | 48.69 | 70.3 | 63.42 | 53.01 | 75.88 | |||
| Patrician (2019) 4 | 90 | 2.05 | 1.62 | 2.48 | 50.15 | 38.6 | 65.17 | 6563.29 | 5084.58 | 8472.05 | ||||||
| Patrician (2019) 2 | 90 | 1.83 | 1.40 | 2.26 | 68.76 | 50.3 | 93.98 | 9390.94 | 6663.48 | 13234.79 | ||||||
| Range | 1.83–10.45 | 1.40–5.96 | 2.26–14.94 | 0.6–68.76 | 0.37–50.3 | 0.97–93.98 | 6.4–9390.94 | 4.19–6663.48 | 9.78–13234.79 | 63.42–65.73 | 51.21–53.01 | 75.88–84.36 | ||||
| Bergeria (2022) 1c | 100 | 3.20 | 1.96 | 4.44 | 1.9941 | 0.8399 | 4.7344 | |||||||||
| Bergeria (2022) 1a | 100 | 2.50 | 1.26 | 3.74 | 12.1901 | 4.8913 | 30.3802 | |||||||||
| Bergeria(2022) 1b | 100 | 3.30 | 1.64 | 4.96 | 17.9012 | 10.3651 | 30.9167 | |||||||||
| Atsmon (2018b) 2 | 100 | 3.38 | 2.82 | 3.94 | 43.67 | 34.85 | 54.71 | 145.75 | 128.55 | 165.24 | 149.25 | 131.85 | 168.96 | 3.59 | 3.45 | 3.73 |
| Range | 2.50–3.38 | 1.26–2.82 | 3.74–4.96 | 1.99–43.67 | 0.84–34.85 | 4.73–54.71 | ||||||||||
| Tayo (2020) | 200 | 2.5 | 2.21 | 2.79 | 137.49 | 93.4 | 202.38 | 457.64 | 398.3 | 525.83 | 493.22 | 434.13 | 560.35 | 11.2 | 6.78 | 15.62 |
| Taylor (2019) | 200 | 2.78 | 1.75 | 3.81 | 148 | 90.09 | 243.13 | 449 | 259.17 | 777.87 | 474 | 273.11 | 822.67 | 8.58 | 3.67 | 13.49 |
| Range | 2.5–2.78 | 1.75–2.21 | 2.79–3.81 | 137.49–148 | 90.09–93.4 | 202.38–243.13 | 449–457.64 | 259.17–398.3 | 525.83–777.87 | 474–493.22 | 273.11–434.13 | 560.35–822.67 | 8.58–11.2 | 3.67–6.78 | 13.49–15.62 | |
| Crockett (2020) 1 | 750 | 3.75 | 3.47 | 4.03 | 187 | 155.16 | 225.38 | 1077 | 901.06 | 1287.29 | 1190 | 997.86 | 1419.14 | 39.7 | 34.27 | 45.13 |
| Taylor (2018) 5 | 750 | 4.38 | 3.73 | 5.03 | 290.8 | 163.88 | 516.01 | 1070 | 641.7 | 1784.17 | ||||||
| Schoedel (2018) 1 | 750 | 5.16 | 4.70 | 5.62 | 304.62 | 263.25 | 352.5 | 1407.75 | 1198.66 | 1653.31 | 1525.18 | 1318.03 | 1764.9 | |||
| Crockett (2020) 5 | 750 | 5.76 | 4.64 | 6.88 | 354 | 260.51 | 481.05 | 1676 | 1237.73 | 2269.46 | 1782 | 1323.65 | 2399.07 | 34 | 29.78 | 38.22 |
| Crockett (2020) 4 | 750 | 5.88 | 4.21 | 7.55 | 527 | 403.42 | 688.44 | 2450 | 1983.19 | 3026.69 | 2588 | 2084.54 | 3213.06 | 36.5 | 32.17 | 40.83 |
| Crockett (2020) 3 | 750 | 5.26 | 3.90 | 6.62 | 722 | 572.7 | 910.22 | 3202 | 2623.11 | 3908.64 | 3394 | 2789.24 | 4129.89 | 39.4 | 33.88 | 44.92 |
| Crockett (2020) 2 | 750 | 3.38 | 2.66 | 4.10 | 1050 | 786.26 | 1402.2 | 4584 | 3563.92 | 5896.06 | 4870 | 3806.16 | 6231.19 | 41.3 | 37.50 | 45.10 |
| Range | 3.38–5.88 | 2.66–4.70 | 4.03–7.55 | 187–1050 | 155.16–786.26 | 225.38–1402.2 | 1070–4584 | 641.7–3563.92 | 1287.29–5896.06 | 1190–4870 | 997.86–3806.16 | 1419.14–6231.19 | 34–41.3 | 29.78–37.50 | 38.22–45.13 | |
| Taylor (2018) 1 | 1500 | 4 | 3.17 | 4.83 | 292.4 | 132.16 | 646.95 | 1517 | 734.68 | 3132.35 | 1618 | 804.95 | 3252.28 | 14.43 | 8.96 | 19.90 |
| Taylor (2018) 7 | 1500 | 3.63 | 3.13 | 4.13 | 335.4 | 213.31 | 527.38 | 1987 | 1443.87 | 2734.44 | 2198 | 1644.02 | 2938.65 | 30.33 | 24.84 | 35.82 |
| Taylor (2018) 6 | 1500 | 4.38 | 3.73 | 5.03 | 361.8 | 185.85 | 704.35 | 1444 | 756.56 | 2756.07 | ||||||
| Schoedel (2018) 2 | 1500 | 5.89 | 5.51 | 6.27 | 439.96 | 363.05 | 533.17 | 2169.62 | 1767.64 | 2663.01 | 2285.08 | 1889.86 | 2762.95 | |||
| Taylor (2018) 8 | 1500 | 3.13 | 2.45 | 3.81 | 1628 | 1196.87 | 2214.42 | 8347 | 6760.99 | 10305.05 | 8669 | 7030.04 | 10690.06 | 24.4 | 21.92 | 26.88 |
| Range | 3.13–5.89 | 3.13–5.51 | 3.81–6.27 | 292.4–1628 | 132.16–1196.87 | 527.38–2214.42 | 1444–2169.62 | 734.68–6760.99 | 2663.01–10305.05 | 1618–8669 | 804.95–7030.04 | 2762.95–10690.06 | 14.43–30.33 | 8.96–24.84 | 19.90–35.82 | |
| Schoedel (2018) 3 | 4500 | 5.62 | 4.87 | 6.37 | 283.2 | 211.95 | 378.39 | 1576.25 | 1186.4 | 2094.2 | 1586.63 | 1206.29 | 2086.9 | |||
| Taylor (2018) 3 | 4500 | 5 | 5.00 | 5.00 | 722.1 | 430.97 | 1209.88 | 3215 | 1952.97 | 5292.58 | 3426 | 2118.62 | 5540.15 | 16.61 | 13.35 | 19.87 |
| Range | 5–5.62 | 4.87–5 | 5–6.37 | 283.2–722.1 | 211.95–430.97 | 378.39–1209.88 | 1576.25–3215 | 1186.4–1952.97 | 2094.2–5292.58 | 1586.63–3426 | 1206.29–2118.62 | 2086.9–5540.15 | ||||
Numbers next each reference is in accordance with the trial arm number in Table 1, for example, Abbotts (2022) 3 means Abbotts et al. (2022) study, trial arm number 3.
CI 95% reported as lower limit and upper limit.
CI, confidence interval.
FIG. 2.
Pharmacokinetic parameters of single dose CBD studies in increasing order of CBD dose.
Geometric mean AUC0–t ranged between 6.18 and 76.77 h×ng/mL for inhalation, 0.69–61.64 h×ng/mL for oromucosal, and 0.47–9390.94 h×ng/mL for oral administration. The geometric mean AUC0–inf was 9.03 for the only inhalation study that reported this parameter, ranging between 1.59 and 70.98 h×ng/mL for oromucosal and 3.32–8669 h×ng/mL for oral administration. The arithmetic mean T1/2 ranged between 1.10 and 31.00 h for inhalation, 1.44–10.86 h for oromucosal, and 1.09–70.3 h for oral administration.
Meta-regression analysis
Overall, of a total number of 87 trial arms that were included in analysis, 84 were used in regression models for Tmax, 81 for Cmax, 78 for AUC0–t, and 51 for AUC0–inf, and 53 for T1/2. The amount of variability in the data that the models could explain (R2) ranged between 0 and 83% for Tmax, indicating a very low to high model fit, 41–49% for Cmax, indicating a moderate fit, 44–52% for AUC0–t indicating a moderate fit, 35–70% for AUC0–inf indicating a low to high fit, and 84–87% for T1/2 indicating a high fit. Removing the “Poor” quality trial arms from the models did not result in a noticeable change in model fit or a change in the significance of variables.
Higher CBD dose was consistently associated with higher Cmax, AUC0–t, and AUC0–inf across all models (Table 4, Fig. 2, and Supplementary Fig. S1). Compared with oral administration as a reference, inhalation was associated with lower Tmax in a poorly fitted model (Table 4, Model Nos. 1 and 2), but did not show any significant difference for Cmax (Model Nos. 7 and 8), AUC0–t (Model Nos. 13 and 14), or AUC0–inf (Model No. 19). In addition, compared with oral administration as a reference, oromucosal administration was associated with lower Cmax (Model Nos. 7–10), AUC0–t (Model Nos. 13–16), and AUC0–inf (Model Nos. 19, 20), but there was no significant difference for Tmax (Model Nos. 1–4). Compared with the Epidiolex formulation as a reference, nanotech, and oil-based formulations were associated with a lower Tmax (Model Nos. 5 and 6).
Table 4.
Meta-Regression Models of Pharmacokinetic Parameters in Single-Dose Cannabidiol Studies
| Parameter | Model No. | Route of administration | Number of arms | Model fit R 2 | CBD dose |
Route
a
|
Formulation
a
|
Diet: fed a | Female b /total ratio | Duration | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Inhalation | Oromucosal | Nanotech | Oil-based | |||||||||
| Tmax | 1 | All routes | 84 | 0 | (−) <0.001 | (−) 0.960 | (+) 0.213 | |||||
| 2 | All routes—fair/good | 81 | 0 | (−) <0.001 | (−) 0.856 | (+) 0.294 | ||||||
| 3 | Oral and oromucosal | 75 | 0.35 | (+) 0.843 | (+) 0.088 | (+) 0.158 | ||||||
| 4 | Oral and oromucosal—fair/good | 73 | 0.35 | (+) 0.962 | (+) 0.097 | (+) 0.177 | ||||||
| 5 | Oral | 48 | 0.83 | (−) <0.001 | (−) <0.001 | (+) 0.51 | (−) 0.006 | |||||
| 6 | Oral—fair/good | 47 | 0.83 | (−) <0.001 | (−) <0.001 | (+) 0.717 | (−) 0.004 | |||||
| Cmax | 7 | All routes | 81 | 0.41 | (+) <0.001 | (−) 0.973 | (−) 0.009 | (+) 0.022 | ||||
| 8 | All routes—fair/good | 78 | 0.41 | (+) <0.001 | (−) 0.417 | (−) 0.013 | (+) 0.018 | |||||
| 9 | Oral and oromucosal | 72 | 0.49 | (+) <0.001 | (−) 0.037 | (+) <0.001 | (+) 0.006 | |||||
| 10 | Oral and oromucosal—fair/good | 70 | 0.48 | (+) <0.001 | (−) 0.049 | (+) <0.001 | (+) 0.008 | |||||
| 11 | Oral | 53 | 0.48 | (+) <0.001 | (+) <0.001 | (+) 0.010 | ||||||
| 12 | Oral—fair/good | 52 | 0.47 | (+) <0.001 | (+) <0.001 | (+) 0.011 | ||||||
| AUC0–t | 13 | All routes | 78 | 0.44 | (+) <0.001 | (−) 0.747 | (−) <0.003 | (+) 0.583 | (+) 0.001 | |||
| 14 | All routes—fair/good | 75 | 0.44 | (+) 0.001 | (−) 0.887 | (−) <0.004 | (+) 0.723 | (+) 0.001 | ||||
| 15 | Oral and oromucosal | 72 | 0.52 | (+) <0.001 | (−) <0.010 | (+) <0.001 | (+) 0.302 | (+) 0.094 | ||||
| 16 | Oral and oromucosal—fair/good | 70 | 0.51 | (+) <0.001 | (−) <0.012 | (+) <0.001 | (+) 0.348 | (+) 0.094 | ||||
| 17 | Oral | 53 | 0.49 | (+) <0.001 | (+) <0.001 | (+) 0.128 | (+) 0.510 | |||||
| 18 | Oral—fair/good | 52 | 0.49 | (+) <0.001 | (+) <0.001 | (+) 0.143 | (+) 0.505 | |||||
| AUC0–inf | 19 | All routes—fair/good | 51 | 0.36 | (+) 0.001 | (−) 0.463 | (−) 0.025 | (+) 0.201 | (+) <0.001 | |||
| 20 | Oral and oromucosal—fair/good | 47 | 0.35 | (+) 0.001 | (−) 0.054 | (−) 0.600 | (+) 0.350 | (+) <0.001 | ||||
| 21 | Oral—fair/good | 33 | 0.70 | (+) 0.002 | (−) 0.531 | (+) 0.341 | (+) 0.001 | |||||
| T1/2 | 22 | All routes | 53 | 0.84 | (+) 0.493 | (+) <0.001 | ||||||
| 23 | All routes—fair/good | 52 | 0.86 | (+) 0.277 | (+) <0.001 | |||||||
| 24 | Oral and oromucosal—fair/good | 50 | 0.86 | (+) 0.230 | (+) <0.001 | |||||||
| 25 | Oral—fair/good | 35 | 0.87 | (+) 0.005 | (+) <0.001 | |||||||
This table does not include regression coefficients because they were in log scale and not interpretable in terms of effect size. Hereby only the sig of regression coefficients, that is, negative or positive, and statistical significance level are reported.
Reference group for route of administration was “oral,” for CBD formulation was “Epidiolex,” and for diet was “fast” status.
The number of female participants was divided by the total participants, and the result ratio was a number between 0 and 1, which was included in the model.
Fed status was associated with higher Cmax (Model Nos. 9–12) and AUC0–t values for both oromucosal and oral administration models (Model Nos. 15–18) compared with the fasting status. No significant association of fed status was observed with either Tmax (Model Nos. 3–6) or AUC0–inf (Model Nos. 20 and 21). A higher ratio of female participants in the sample was associated with lower Tmax (Model Nos. 5 and 6) and higher T1/2 (Model No. 25) only among oral administration arms. A higher ratio of female participants was also associated with higher Cmax (Model Nos. 9–12) in all models. There was no significant association of female/total ratio with AUC0–t (Model Nos. 13–18) or AUC0–inf (Model Nos. 19–21). Finally, longer study duration was associated with higher AUC0–t only in the regression models that included all routes of administration (Model Nos. 13 and 14), and higher AUC0–inf and T1/2 in all the regression models (Model Nos. 19–25).
Discussion
In this review, we aimed to provide an updated systematic assessment of available evidence on the PK of CBD, provide comparable PK parameters from different studies on the same scale, and explore the impact of different relevant factors on PK outcomes. There was considerable heterogeneity in the available PK data for CBD both in terms of the study conducted and reported outcomes. Nevertheless, despite the heterogeneity and quality aspects, several meaningful patterns emerged for factors expected to influence the pharmacokinetic outcomes of CBD including the route of administration, dose, formulation, diet status, sex ratio, and study duration.
For the parameters related to bioavailability, that is, Cmax, AUC0–t, AUC0–inf, it appeared that inhalation and oral administration had comparable outcomes. However, oromucosal administration consistently resulted in a lower bioavailability than oral administration, which is in line with a previous systematic review3 and some of the within-study comparisons using a similar dose for both routes of administration.31,35 There are though other within-study comparisons indicating that bioavailability was comparable between oral and oromucosal administration,16 or that oral administration resulted in lower bioavailability.49 Different CBD formulations for oral and oromucosal administration, as well as the possibility of swallowing the oromucosally administered CBD leading to oral absorption, could potentially explain in part these within-study inconsistencies.
However, owing to the low CBD doses used particularly in oromucosal trial arms, comparability is limited and reliable interpretation warrants additional studies. Regarding rate of absorption, as would be expected, inhalation resulted in the lower Tmax/faster absorption compared with oral administration. There was a lack of significant difference between oromucosal and oral administration regarding absorption rate, which is in line with a previous systematic review3 as well as some of the direct within-study comparisons.16,31,35
Given the general low bioavailability of oral administration of cannabinoids, recent years have seen an increase in CBD nanoformulations in the hope of increasing absorption and bioavailability through the oral and oromucosal administration routes. In the regression models for Tmax, model fit was noticeably improved by accounting for formulation. Nanotech and oil-based formulations were associated with lower Tmax than the Epidiolex formulation indicating that they would have a faster onset of action. To our knowledge, there has been no published direct side-by-side comparison of Epidiolex with nanotech or oil-based formulations. In this review, owing to the methodological considerations that are discussed in the Statistical analyses section and Supplementary Methods in the Supplementary Data, only certain formulations could be included in the regression models for Tmax. Therefore, we could not explore other formulations or other PK parameters, thus limiting interpretation of different formulations on their comparable bioavailability for CBD.
As expected, higher CBD dose was consistently associated with higher bioavailability in all the models for Cmax, AUC0–t, and AUC0–inf. The information provided in Table 3 and Figure 2 serve as a useful reference to help predict CBD dose to reach a certain serum level and potential clinical effect for specific conditions. Although various CBD formulations have been used in lower doses, all the studies with a CBD dose of >100 mg were only conducted with Epidiolex. As such, there is still a lack of PK information about CBD doses expected to be more aligned with a “medicinal” range.
Food consumption can influence bioavailability of many medications, especially lipophilic compounds like CBD through increased transit time and lymphatic absorption in the intestines. We observed that fed condition was associated with a higher bioavailability of CBD, but had no significant impact in the time to reach maximum serum concentrations, compared with the fasted status. Specifically, we observed a higher Cmax and AUC0–t across all the models for fed condition compared with fasted status, which is in line with within-study direct comparisons.12,19,34,40 The lack of a significant effect of diet status on Tmax, is in line with two multiple-arm comprehensive studies,19,34 but inconsistent with two other studies where Tmax was considerably longer in the fed group.12,40
Although we would expect a significant effect of diet on AUC0–inf, both theoretically and based on within-study comparisons,19,34,40 we did not detect such an effect in our models. This could in part be attributed to the lower number of studies that reported AUC0–inf and thus lower power of these models.
The importance of sex for drug bioavailability and clearance could also have significant implications, including a probably lower required dose of CBD in women to reach a certain blood level and clinical effect. There are noted sex differences in CYP450 family of enzymes that contribute to the metabolism of CBD.51 In addition, the literature suggests slower clearance in women compared with men.52,53 We observed that for the PK studies in which sex ratio was reported, a higher female ratio was associated with a faster absorption and higher maximum concentrations through oral administration, evident with lower Tmax and higher Cmax, respectively. These findings are consistent with direct within-study CBD PK comparisons showing higher Cmax and lower Tmax in female participants compared with male participants.26,45
A recent PK study with very low doses of CBD reported no effect for sex on Tmax, but female sex was associated with significantly higher Cmax and AUC0–t.14 A higher Cmax and lower T1/2 with oral administration would mathematically be expected to be associated with a higher AUC0–t and AUC0–inf and also in line with direct within-study sex comparisons.14,26,45 However, this relationship was not evident in our analysis perhaps owing to the few studies conducted with women. Well-powered studies with clinically relevant higher doses in men and women are still needed.
As expected, duration of PK session was a determinant of overall bioavailability and clearance of CBD with implications for designing future studies. A longer duration of PK session was associated with a higher AUC0–inf in all models, and a higher AUC0–t in models that included data from all routes of administration of CBD. A higher duration of the PK session was also consistently associated with a longer T1/2 across all models. In line with this later finding, the literature suggests that cannabinoids in humans may need extended durations to adequately determine cannabinoid half-lives, given that the terminal half-life of cannabinoids is longer than the initial half-life.6 Indeed, half-lives of >60 h were reported for Epidiolex with a PK duration of 168 h.23
A number of limitations should be considered in interpreting the results. For example, certain study characteristics could limit cross-study comparisons, including different reporting scales, potential variations in the equipment and methods used for CBD quantification by different laboratories, and heterogeneity of the study conditions. There was also a lack of detail regarding some formulations, particularly for commercialized products. In addition, the presence of other cannabinoids in the preparations is an important consideration; whereas to the best of the authors' knowledge, there are no human PK study to date that has systematically compared CBD bioavailability with and without THC, hypothetically the presence of THC could influence CBD PK at the level of shared metabolic pathways. Such a pharmacokinetic interaction is supported by a study of CBD oil in cats that demonstrated increased serum CBD levels in the presence of THC54 and a recent study in humans where THC serum levels were increased in the presence of CBD.55 In addition, cannabidiolic acid in some of the preparations could readily convert to CBD in the human body and alter pharmacokinetic outcomes. Unfortunately, given the variability in formulations, presence of cannabinoids other than THC in many of the preparations, and lack of any direct evidence from well-controlled human studies on how THC may influence CBD PK, we did not include THC dose in our analyses. Another factor for consideration is the washout period among the crossover studies. Of the studies included in our analysis, Hosseini et al.16 reported the shortest washout of 24 h that would not be expected to provide sufficient time for the cannabinoid to be fully cleared. Although CBD T1/2 is >24 h, Hossieni et al.16 demonstrated a considerably low serum CBD concentration at 24 h compared with peak concentrations, and excluding this study from the meta-regression analysis did not change our findings. As such, this study was included in final analysis.
There were also certain limitations related to the methods of this review. Comprehensive quality assessment of PK studies requires a specifically designed assessment tool beyond the ones used for general before-after studies. Although there have been efforts to introduce such quality assessment tools,56 there is still no commonly accepted one available. In addition to the items covered by the NIHLB tool, preregistration of study protocol as a clinical trial, the potential effect of other constituents of the administered product on the PK outcomes, sufficient length of study, accounting for the potential effect of important covariates like sex and body mass, would be important considerations. In regard to the statistical methods, the log-transformed regression coefficients could not be interpreted in terms of effect size, limiting our prediction of each factor's impact on PK parameters. Finally, we did not include the PK outcomes of CBD metabolites reported by some of the included studies since the CBD metabolism is affected by multiple factors thus requiring a focused and extensive exploration that was outside the scope of this review. Understanding the PK of CBD metabolites can provide further insights into the CBD half/life and sex differences.
Conclusion
We provided an updated overview of the current status of evidence of PK of CBD. In exploring how different factors potentially influenced the PK outcomes of CBD, consuming food while taking CBD, female sex, and oral administration were associated with higher bioavailability. Recommendations for future research would mainly concern conducting original systematic studies to elucidate the impact of biological sex and different formulations in single studies with multiple arms. It would also be beneficial for future studies to examine clinically relevant doses of CBD, for example, >200 mg, given the increasing number of such preparations on the market and their potential application in clinical practice. Finally, reporting PK parameters in both arithmetic and geometric scales would help improve comparisons across studies and would also enhance knowledge aggregation and replicability.
Abbreviations Used
- CBD
cannabidiol
- CD
cannot determine
- CI
confidence interval
- CV
coefficient of variation
- NR
not reported
- PK
pharmacokinetics
- SD
standard deviation
- THC
tetrahydrocannabinol
Authors' Contributions
E.M.-Z., K.B., Y.L.H.: conceptualization; E.M.-Z. and A.C.: methodology, data extraction, and quality assessments; E.M.-Z.: narrative and quantitative synthesis, and original article draft preparation; and A.C., K.B., and Y.L.H. edited the article. All authors have read and agreed to the final version of the article.
Author Disclosure Statement
Authors have no conflicts or competing interests to disclose.
Funding Information
Authors were supported by National Institutes of Health NIDA UG3DA050323 and K23-DA045928.
Supplementary Material
Cite this article as: Moazen-Zadeh E, Chisholm A, Bachi K, Hurd YL (2023) Pharmacokinetics of cannabidiol: a systematic review and meta-regression analysis, Cannabis and Cannabinoid Research X:X, 1–28, DOI: 10.1089/can.2023.0025.
References
- 1. VanDolah HJ, Bauer BA, Mauck KF. Clinicians' Guide to Cannabidiol and Hemp Oils. Mayo Clin Proc 2019;94(9):1840–1851. [DOI] [PubMed] [Google Scholar]
- 2. WHO Expert Committee on Drug Dependence. Cannabidiol (CBD) Critical Review Report. Fortieth Meeting, Geneva, June 4–7, 2018. [Google Scholar]
- 3. Millar SA, Stone NL, Yates AS, et al. A systematic review on the pharmacokinetics of cannabidiol in humans. Front Pharmacol 2018;9:1365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Julious SA, Debarnot CA. Why are pharmacokinetic data summarized by arithmetic means? J Biopharm Stat 2000;10(1):55–71. [DOI] [PubMed] [Google Scholar]
- 5. Lucas CJ, Galettis P, Schneider J. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br J Clin Pharmacol 2018;84(11):2477–2482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Huestis MA. Human cannabinoid pharmacokinetics. Chem Biodivers 2007;4(8):1770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. National Heart, Lung, and Blood Institute. Study Quality Assessment Tools: For Observational Cohort and Cross-Sectional Studies, for Before-After (Pre-Post) Studies with No Control Group, for Case-Control Studies. Maryland, USA Im Internet (Stand: 1001 2019); 2019. Available from: www nhlbi nih gov/health-topics/study-quality-assessment-tools [Last accessed: September 21, 2022].
- 8. Aarons L, Ogungbenro K. Optimal design of pharmacokinetic studies. Basic Clin Pharmacol Toxicol 2010;106(3):250–255. [DOI] [PubMed] [Google Scholar]
- 9. Wan X, Wang W, Liu J, et al. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 2014;14(1):1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Higgins JP, White IR, Anzures-Cabrera J. Meta-analysis of skewed data: Combining results reported on log-transformed or raw scales. Stat Med 2008;27(29):6072–6092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Higgins JPT, Thomas J, Chandler J, et al. , (eds). Cochrane Handbook for Systematic Reviews of Interventions Version 6.3 (updated February 2022). Cochrane; 2022. Available from: wwwtrainingcochraneorg/handbook [Last accessed: September 21, 2022].
- 12. Abbotts KSS, Ewell TR, Butterklee HM, et al. Cannabidiol and cannabidiol metabolites: Pharmacokinetics, interaction with food, and influence on liver function. Nutrients 2022;14(10):2152; doi: 10.3390/nu14102152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Bergeria CL, Spindle TR, Cone EJ, et al. Pharmacokinetic profile of Δ9-tetrahydrocannabinol, cannabidiol and metabolites in blood following vaporization and oral ingestion of cannabidiol products. J Anal Toxicol 2022;46(6):583–591; doi: 10.1093/jat/bkab124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Berl V, Hurd YL, Lipshutz BH, et al. A randomized, triple-blind, comparator-controlled parallel study investigating the pharmacokinetics of cannabidiol and tetrahydrocannabinol in a novel delivery system, solutech, in association with cannabis use history. Cannabis Cannabinoid Res 2022;7(6):777–789; doi: 10.1089/can.2021.0176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Busardò FP, Pérez-Acevedo AP, Pacifici R, et al. Disposition of phytocannabinoids, their acidic precursors and their metabolites in biological matrices of healthy individuals treated with vaporized medical cannabis. Pharmaceuticals 2021;14(1):59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hosseini A, McLachlan AJ, Lickliter JD. A phase I trial of the safety, tolerability and pharmacokinetics of cannabidiol administered as single-dose oil solution and single and multiple doses of a sublingual wafer in healthy volunteers. Br J Clin Pharmacol 2021;87(4):2070–2077. [DOI] [PubMed] [Google Scholar]
- 17. Vitetta L, Butcher B, Henson JD, et al. A pilot safety, tolerability and pharmacokinetic study of an oro-buccal administered cannabidiol-dominant anti-inflammatory formulation in healthy individuals: A randomized placebo-controlled single-blinded study. Inflammopharmacology 2021;29(5):1361–1370. [DOI] [PubMed] [Google Scholar]
- 18. Williams NNB, Ewell TR, Abbotts KSS, et al. Comparison of five oral cannabidiol preparations in adult humans: Pharmacokinetics, body composition, and heart rate variability. Pharmaceuticals 2021;14(1):35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Crockett J, Critchley D, Tayo B, et al. A phase 1, randomized, pharmacokinetic trial of the effect of different meal compositions, whole milk, and alcohol on cannabidiol exposure and safety in healthy subjects. Epilepsia 2020;61(2):267–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hobbs JM, Vazquez AR, Remijan ND, et al. Evaluation of pharmacokinetics and acute anti-inflammatory potential of two oral cannabidiol preparations in healthy adults. Phytother Res 2020;34(7):1696–1703. [DOI] [PubMed] [Google Scholar]
- 21. Izgelov D, Davidson E, Barasch D, et al. Pharmacokinetic investigation of synthetic cannabidiol oral formulations in healthy volunteers. Eur J Pharm Biopharm 2020;154:108–115. [DOI] [PubMed] [Google Scholar]
- 22. Pérez-Acevedo AP, Pacifici R, Mannocchi G, et al. Disposition of cannabinoids and their metabolites in serum, oral fluid, sweat patch and urine from healthy individuals treated with pharmaceutical preparations of medical cannabis. Phytother Res 2021;35(3):1646–1657. [DOI] [PubMed] [Google Scholar]
- 23. Perkins D, Butler J, Ong K, et al. A phase 1, randomised, placebo-controlled, dose escalation study to investigate the safety, tolerability and pharmacokinetics of cannabidiol in fed healthy volunteers. Eur J Drug Metab Pharmacokinet 2020;45(5):575–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Pichini S, Mannocchi G, Gottardi M, et al. Fast and sensitive UHPLC-MS/MS analysis of cannabinoids and their acid precursors in pharmaceutical preparations of medical cannabis and their metabolites in conventional and non-conventional biological matrices of treated individual. Talanta 2020;209:120537. [DOI] [PubMed] [Google Scholar]
- 25. Tayo B, Taylor L, Sahebkar F, et al. A phase I, open-label, parallel-group, single-dose trial of the pharmacokinetics, safety, and tolerability of cannabidiol in subjects with mild to severe renal impairment. Clin Pharmacokinet 2020;59(6):747–755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Knaub K, Sartorius T, Dharsono T, et al. A novel self-emulsifying drug delivery system (SEDDS) based on VESIsorb® formulation technology improving the oral bioavailability of cannabidiol in healthy subjects. Molecules 2019;24(16):2967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Morrison G, Crockett J, Blakey G, et al. A phase 1, open-label, pharmacokinetic trial to investigate possible drug-drug interactions between clobazam, stiripentol, or valproate and cannabidiol in healthy subjects. Clin Pharmacol Drug Dev 2019;8(8):1009–1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Patrician A, Versic-Bratincevic M, Mijacika T, et al. Examination of a new delivery approach for oral cannabidiol in healthy subjects: A randomized, double-blinded, placebo-controlled pharmacokinetics study. Adv Ther 2019;36(11):3196–3210. [DOI] [PubMed] [Google Scholar]
- 29. Taylor L, Crockett J, Tayo B, et al. A phase 1, open-label, parallel-group, single-dose trial of the pharmacokinetics and safety of cannabidiol (CBD) in subjects with mild to severe hepatic impairment. J Clin Pharmacol 2019;59(8):1110–1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Atsmon J, Cherniakov I, Izgelov D, et al. PTL401, a new formulation based on pro-nano dispersion technology, improves oral cannabinoids bioavailability in healthy volunteers. J Pharm Sci 2018a;107(5):1423–1429. [DOI] [PubMed] [Google Scholar]
- 31. Atsmon J, Heffetz D, Deutsch L, et al. Single-dose pharmacokinetics of oral cannabidiol following administration of PTL101: A new formulation based on gelatin matrix pellets technology. Clin Pharmacol Drug Dev 2018b;7(7):751–758. [DOI] [PubMed] [Google Scholar]
- 32. Meyer P, Langos M, Brenneisen R. Human pharmacokinetics and adverse effects of pulmonary and intravenous THC-CBD formulations. Med Cannabis Cannabinoids 2018;1(1):36–43; doi: 10.1159/000489034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Schoedel KA, Szeto I, Setnik B, et al. Abuse potential assessment of cannabidiol (CBD) in recreational polydrug users: A randomized, double-blind, controlled trial. Epilepsy Behav 2018;88:162–171. [DOI] [PubMed] [Google Scholar]
- 34. Taylor L, Gidal B, Blakey G, et al. A phase I, randomized, double-blind, placebo-controlled, single ascending dose, multiple dose, and food effect trial of the safety, tolerability and pharmacokinetics of highly purified cannabidiol in healthy subjects. CNS Drugs 2018;32(11):1053–1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cherniakov I, Izgelov D, Barasch D, et al. Piperine-pro-nanolipospheres as a novel oral delivery system of cannabinoids: Pharmacokinetic evaluation in healthy volunteers in comparison to buccal spray administration. J Control Release 2017;266:1–7. [DOI] [PubMed] [Google Scholar]
- 36. Haney M, Malcolm RJ, Babalonis S, et al. Oral cannabidiol does not alter the subjective, reinforcing or cardiovascular effects of smoked cannabis. Neuropsychopharmacology 2016;41(8):1974–1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Desrosiers NA, Himes SK, Scheidweiler KB, et al. Phase I and II cannabinoid disposition in blood and plasma of occasional and frequent smokers following controlled smoked cannabis. Clin Chem 2014;60(4):631–643. [DOI] [PubMed] [Google Scholar]
- 38. Sellers EM, Schoedel K, Bartlett C, et al. A multiple-dose, randomized, double-blind, placebo-controlled, parallel-group QT/QTc study to evaluate the electrophysiologic effects of THC/CBD spray. Clin Pharmacol Drug Dev 2013;2(3):285–294. [DOI] [PubMed] [Google Scholar]
- 39. Stott C, White L, Wright S, et al. A phase I study to assess the single and multiple dose pharmacokinetics of THC/CBD oromucosal spray. Eur J Clin Pharmacol 2013a;69(5):1135–1147. [DOI] [PubMed] [Google Scholar]
- 40. Stott C, White L, Wright S, et al. A phase I study to assess the effect of food on the single dose bioavailability of the THC/CBD oromucosal spray. Eur J Clin Pharmacol 2013b;69(4):825–834. [DOI] [PubMed] [Google Scholar]
- 41. Stott C, White L, Wright S, et al. A phase I, open-label, randomized, crossover study in three parallel groups to evaluate the effect of Rifampicin, Ketoconazole, and Omeprazole on the pharmacokinetics of THC/CBD oromucosal spray in healthy volunteers. Springerplus 2013c;2(1):1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Eichler M, Spinedi L, Unfer-Grauwiler S, et al. Heat exposure of Cannabis sativa extracts affects the pharmacokinetic and metabolic profile in healthy male subjects. Planta Med 2012;78(7):686–691. [DOI] [PubMed] [Google Scholar]
- 43. Karschner EL, Darwin WD, Goodwin RS, et al. Plasma cannabinoid pharmacokinetics following controlled oral Δ9-tetrahydrocannabinol and oromucosal cannabis extract administration. Clin Chem 2011;57(1):66–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Schwope DM, Karschner EL, Gorelick DA, et al. Identification of recent cannabis use: Whole-blood and plasma free and glucuronidated cannabinoid pharmacokinetics following controlled smoked cannabis administration. Clin Chem 2011;57(10):1406–1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Nadulski T, Pragst F, Weinberg G, et al. Randomized, double-blind, placebo-controlled study about the effects of cannabidiol (CBD) on the pharmacokinetics of Δ9-tetrahydrocannabinol (THC) after oral application of THC verses standardized cannabis extract. Ther Drug Monit 2005a;27(6):799–810. [DOI] [PubMed] [Google Scholar]
- 46. Nadulski T, Sporkert F, Schnelle M, et al. Simultaneous and sensitive analysis of THC, 11-OH-THC, THC-COOH, CBD, and CBN by GC-MS in plasma after oral application of small doses of THC and cannabis extract. J Anal Toxicol 2005b;29(8):782–789. [DOI] [PubMed] [Google Scholar]
- 47. Guy G, Flint M. A single centre, placebo-controlled, four period, crossover, tolerability study assessing, pharmacodynamic effects, pharmacokinetic characteristics and cognitive profiles of a single dose of three formulations of cannabis based medicine extracts (CBMEs)(GWPD9901), plus a two period tolerability study comparing pharmacodynamic effects and pharmacokinetic characteristics of a single dose of a cannabis based medicine extract given via two administration routes (GWPD9901 EXT). J Cannabis Ther 2004;3(3):35–77. [Google Scholar]
- 48. Guy G, Robson P.. A phase I, double blind, three-way crossover study to assess the pharmacokinetic profile of cannabis based medicine extract (CBME) administered sublingually in variant cannabinoid ratios in normal healthy male volunteers (GWPK0215). J Cannabis Ther 2004a;3(4):121–152. [Google Scholar]
- 49. Guy G, Robson P.. A phase I, open label, four-way crossover study to compare the pharmacokinetic profiles of a single dose of 20 mg of a cannabis based medicine extract (CBME) administered on 3 different areas of the buccal mucosa and to investigate the pharmacokinetics of CBME per oral in healthy male and female volunteers (GWPK0112). J Cannabis Ther 2004b;3(4):79–120. [Google Scholar]
- 50. Ohlsson A, Lindgren JE, Andersson S, et al. Single-dose kinetics of deuterium-labelled cannabidiol in man after smoking and intravenous administration. Biomed Environ Mass Spectrom 1986;13(2):77–83. [DOI] [PubMed] [Google Scholar]
- 51. Soldin OP, Mattison DR. Sex differences in pharmacokinetics and pharmacodynamics. Clin Pharmacokinet 2009;48(3):143–157; doi: 10.2165/00003088-200948030-00001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Meibohm B, Beierle I, Derendorf H. How important are gender differences in pharmacokinetics? Clin Pharmacokinet 2002;41(5):329–342. [DOI] [PubMed] [Google Scholar]
- 53. Gandhi M, Aweeka F, Greenblatt RM, et al. Sex differences in pharmacokinetics and pharmacodynamics. Annu Rev Pharmacol Toxicol 2004;44:499–523. [DOI] [PubMed] [Google Scholar]
- 54. Kulpa JE, Paulionis LJ, Eglit GM, et al. Safety and tolerability of escalating cannabinoid doses in healthy cats. J Feline Med Surg 2021;23(12):1162–1175; doi: 10.1177/1098612x211004215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Zamarripa CA, Spindle TR, Surujunarain R, et al. Assessment of orally administered Δ9-tetrahydrocannabinol when coadministered with cannabidiol on Δ9-tetrahydrocannabinol pharmacokinetics and pharmacodynamics in healthy adults: A randomized clinical trial. JAMA Netw Open 2023;6(2):e2254752; doi: 10.1001/jamanetworkopen.2022.54752 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Soliman ABE, Pawluk SA, Wilby KJ, et al. Creation of an inventory of quality markers used to evaluate pharmacokinetic literature: A systematic review. J Clin Pharm Ther 2022;47(2):178–183. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.